Investigation of Macrolide Resistance Genotypes in Mycoplasma bovis Isolates from Canadian Feedlot Cattle
Abstract
1. Introduction
2. Results
2.1. Culture and Reference Antimicrobial Susceptibilities
2.2. Genome Sequencing and Assembly
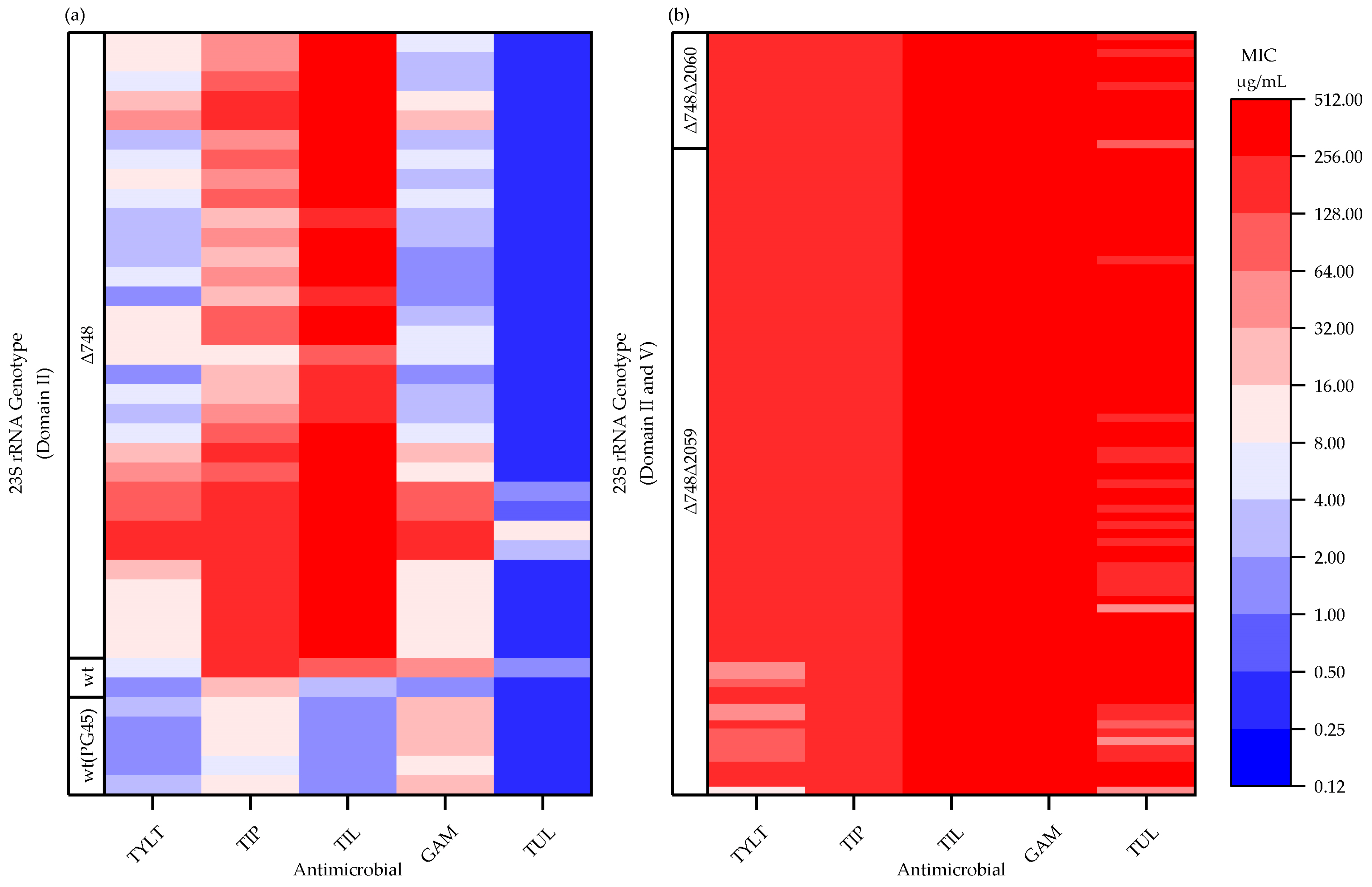
2.3. 23 S rRNA Gene
2.4. L4 and L22 Ribosomal Proteins
3. Discussion
4. Materials and Methods
4.1. Animals and Sample Collection
4.2. Mycoplasma Culture and Isolation
4.3. DNA Extraction and Identification
4.4. Whole Genome Sequencing and Assembly
4.5. Antimicrobial Susceptibility Testing
4.6. Clinical Breakpoints
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nicholas, R.A.J.; Ayling, R.D. Mycoplasma bovis: Disease, diagnosis, and control. Res. Vet. Sci. 2003, 74, 105–112. [Google Scholar] [CrossRef]
- Maunsell, F.P.; Woolums, A.R.; Francoz, D.; Rosenbusch, R.F.; Step, D.L.; Wilson, D.J.; Janzen, E.D. Mycoplasma bovis infections in cattle. J. Vet. Intern. Med. 2011, 25, 772–783. [Google Scholar] [CrossRef]
- Gagea, M.I.; Bateman, K.G.; Shanahan, R.A.; van Dreumel, T.; McEwen, B.J.; Carman, S.; Archambault, M.; Caswell, J.L. Naturally Occurring Mycoplasma Bovis—Associated Pneumonia and Polyarthritis in Feedlot Beef Calves. J. Vet. Diagn. Investig. 2006, 18, 29–40. [Google Scholar] [CrossRef]
- Caswell, J.L.; Bateman, K.G.; Cai, H.Y.; Castillo-Alcala, F. Mycoplasma bovis in Respiratory Disease of Feedlot Cattle. Vet. Clin. N. Am. Food Anim. 2010, 26, 365–379. [Google Scholar] [CrossRef]
- Krysak, D.E. Chronic pneumonia and polyarthritis syndrome in a feedlot calf. Can. Vet. J. 2006, 47, 1019–1022. [Google Scholar]
- Caswell, J.L.; Archambault, M. Mycoplasma bovis pneumonia in cattle. Anim. Health Res. Rev. 2007, 8, 161–186. [Google Scholar] [CrossRef]
- Lysnyansky, I.; Ayling, R.D. Mycoplasma bovis: Mechanisms of Resistance and Trends in Antimicrobial Susceptibility. Front. Microbiol. 2016, 7, 595. [Google Scholar] [CrossRef] [PubMed]
- Hendrick, S.H.; Bateman, K.G.; Rosengren, L.B. The effect of antimicrobial treatment and preventive strategies on bovine respiratory disease and genetic relatedness and antimicrobial resistance of Mycoplasma bovis isolates in a western Canadian feedlot. Can. Vet. J. 2013, 54, 1146–1156. [Google Scholar] [PubMed]
- Anholt, R.M.; Klima, C.; Allan, N.; Matheson-Bird, H.; Schatz, C.; Ajitkumar, P.; Otto, S.J.; Peters, D.; Schmid, K.; Olson, M.; et al. Antimicrobial Susceptibility of Bacteria That Cause Bovine Respiratory Disease Complex in Alberta, Canada. Front. Vet. Sci. 2017, 4, 207. [Google Scholar] [CrossRef] [PubMed]
- Cai, H.Y.; McDowall, R.; Parker, L.; Kaufman, E.I.; Caswell, J.L. Changes in antimicrobial susceptibility profiles of Mycoplasma bovis over time. Can. J. Vet. Res. 2019, 83, 34–41. [Google Scholar] [PubMed]
- Jelinski, M.; Kinnear, A.; Gesy, K.; Andrés-Lasheras, S.; Zaheer, R.; Weese, S.; McAllister, T.A. Antimicrobial Sensitivity Testing of Mycoplasma bovis Isolates Derived from Western Canadian Feedlot Cattle. Microorganisms 2020, 8, 124. [Google Scholar] [CrossRef] [PubMed]
- Rosenbusch, R.F.; Kinyon, J.M.; Apley, M.; Funk, N.D.; Smith, S.; Hoffman, L.J. In Vitro Antimicrobial Inhibition Profiles of Mycoplasma Bovis Isolates Recovered from Various Regions of the United States from 2002 to 2003. J. Vet. Diagn. Investig. 2005, 17, 436–441. [Google Scholar] [CrossRef]
- Hata, E.; Harada, T.; Itoh, M. Relationship between Antimicrobial Susceptibility and Multilocus Sequence Type of Mycoplasma bovis Isolates and Development of a Method for Rapid Detection of Point Mutations Involved in Decreased Susceptibility to Macrolides, Lincosamides, Tetracyclines, and Spectinomycin. Appl. Environ. Microbiol. 2019, 85, e00575-19. [Google Scholar] [CrossRef] [PubMed]
- Ayling, R.D.; Baker, S.E.; Nicholas, R.A.J.; Peek, M.L.; Simon, A.J. Comparison of in vitro activity of danofloxacin, florfenicol, oxytetracycline, spectinomycin and tilmicosin against recent field isolates of Mycoplasma bovis. Vet. Rec. 2000, 146, 745–747. [Google Scholar] [CrossRef] [PubMed]
- Gerchman, I.; Levisohn, S.; Mikula, I.; Lysnyansky, I. In vitro antimicrobial susceptibility of Mycoplasma bovis isolated in Israel from local and imported cattle. Vet. Microbiol. 2009, 137, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Gautier-Bouchardon, A.V.; Ferré, S.; Le Grand, D.; Paoli, A.; Gay, E.; Poumarat, F. Overall decrease in the susceptibility of Mycoplasma bovis to antimicrobials over the past 30 years in France. PLoS ONE 2014, 9, e87672. [Google Scholar] [CrossRef]
- Sulyok, K.M.; Kreizinger, Z.; Fekete, L.; Hrivnák, V.; Magyar, T.; Jánosi, S.; Schweitzer, N.; Turcsányi, I.; Makrai, L.; Erdélyi, K.; et al. Antibiotic susceptibility profiles of Mycoplasma bovis strains isolated from cattle in Hungary, Central Europe. BMC Vet. Res. 2014, 10, 256. [Google Scholar] [CrossRef]
- Heuvelink, A.; Reugebrink, C.; Mars, J. Antimicrobial susceptibility of Mycoplasma bovis isolates from veal calves and dairy cattle in the Netherlands. Vet. Microbiol. 2016, 189, 1–7. [Google Scholar] [CrossRef]
- Klein, U.; de Jong, A.; Moyaert, H.; El Garch, F.; Leon, R.; Richard-Mazet, A.; Rose, M.; Maes, D.; Pridmore, A.; Thomson, J.R.; et al. Antimicrobial susceptibility monitoring of Mycoplasma hyopneumoniae and Mycoplasma bovis isolated in Europe. Vet. Microbiol. 2017, 204, 188–193. [Google Scholar] [CrossRef]
- Mzyk, D.A.; Bublitz, C.M.; Martinez, M.N.; Davis, J.L.; Baynes, R.E.; Smith, G.W. Impact of bovine respiratory disease on the pharmacokinetics of danofloxacin and tulathromycin in different ages of calves. PLoS ONE 2019, 14, e0218864. [Google Scholar] [CrossRef]
- Griffin, D. Economic Impact Associated with Respiratory Disease in Beef Cattle. Vet. Clin. N. Am. Large Anim. Pract. 1997, 13, 367–377. [Google Scholar] [CrossRef]
- Nickell, J.S.; White, B.J. Metaphylactic Antimicrobial Therapy for Bovine Respiratory Disease in Stocker and Feedlot Cattle. Vet. Clin. N. Am. Food Anim. 2010, 26, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Brault, S.A.; Hannon, S.J.; Gow, S.P.; Warr, B.N.; Withell, J.; Song, J.; Williams, C.M.; Otto, S.J.G.; Booker, C.W.; Morley, P.S. Antimicrobial Use on 36 Beef Feedlots in Western Canada: 2008–2012. Front. Vet. Sci. 2019, 6, 329. [Google Scholar] [CrossRef] [PubMed]
- Owen, J.R.; Noyes, N.; Young, A.E.; Prince, D.J.; Blanchard, P.C.; Lehenbauer, T.W.; Aly, S.S.; Davis, J.H.; O’Rourke, S.M.; Abdo, Z.; et al. Whole-Genome Sequencing and Concordance Between Antimicrobial Susceptibility Genotypes and Phenotypes of Bacterial Isolates Associated with Bovine Respiratory Disease. G3 Genes Genomes Genet. 2017, 7, 3059–3071. [Google Scholar] [CrossRef] [PubMed]
- Calcutt, M.J.; Lysnyansky, I.; Sachse, K.; Fox, L.K.; Nicholas, R.A.J.; Ayling, R.D. Gap analysis of Mycoplasma bovis disease, diagnosis and control: An aid to identify future development requirements. Transbound. Emerg. Dis. 2018, 65, 91–109. [Google Scholar] [CrossRef]
- Zaheer, R.; Cook, S.R.; Klima, C.L.; Stanford, K.; Alexander, T.; Topp, E.; Read, R.R.; McAllister, T.A. Effect of subtherapeutic vs. therapeutic administration of macrolides on antimicrobial resistance in Mannheimia haemolytica and enterococci isolated from beef cattle. Front. Microbiol. 2013, 4, 133. [Google Scholar] [CrossRef]
- Hansen, L.H.; Mauvais, P.; Douthwaite, S. The macrolide–ketolide antibiotic binding site is formed by structures in domains II and V of 23S ribosomal RNA. Mol. Microbiol. 1999, 31, 623–631. [Google Scholar] [CrossRef]
- Moore, P.B.; Steitz, T.A. The Structural Basis of Large Ribosomal Subunit Function. Annu. Rev. Biochem. 2003, 72, 813–850. [Google Scholar] [CrossRef]
- Dinos, G.P. The macrolide antibiotic renaissance. Br. J. Pharmacol. 2017, 174, 2967–2983. [Google Scholar] [CrossRef]
- Demczuk, W.; Martin, I.; Peterson, S.; Bharat, A.; Van Domselaar, G.; Graham, M.; Lefebvre, B.; Allen, V.; Hoang, L.; Tyrrell, G.; et al. Genomic Epidemiology and Molecular Resistance Mechanisms of Azithromycin-Resistant Neisseria gonorrhoeae in Canada from 1997 to 2014. J. Clin. Microbiol. 2016, 54, 1304–1313. [Google Scholar] [CrossRef]
- Lerner, U.; Amram, E.; Ayling, R.D.; Mikula, I.; Gerchman, I.; Harrus, S.; Teff, D.; Yogev, D.; Lysnyansky, I. Acquired resistance to the 16-membered macrolides tylosin and tilmicosin by Mycoplasma bovis. Vet. Microbiol. 2014, 168, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Sulyok, K.M.; Kreizinger, Z.; Wehmann, E.; Lysnyansky, I.; Bányai, K.; Marton, S.; Jerzsele, Á.; Rónai, Z.; Turcsányi, I.; Makrai, L.; et al. Mutations Associated with Decreased Susceptibility to Seven Antimicrobial Families in Field and Laboratory-Derived Mycoplasma bovis Strains. Antimicrob. Agents Chemother. 2017, 61, e01983-16. [Google Scholar] [CrossRef] [PubMed]
- Pereyre, S.; Gonzalez, P.; de Barbeyrac, B.; Darnige, A.; Renaudin, H.; Charron, A.; Raherison, S.; Bébéar, C.; Bébéar, C.M. Mutations in 23S rRNA Account for Intrinsic Resistance to Macrolides in Mycoplasma hominis and Mycoplasma fermentans and for Acquired Resistance to Macrolides in M. hominis. Antimicrob. Agents Chemother. 2002, 46, 3142–3150. [Google Scholar] [CrossRef] [PubMed]
- Chrisment, D.; Charron, A.; Cazanave, C.; Pereyre, S.; Bebear, C. Detection of macrolide resistance in Mycoplasma genitalium in France. J. Antimicrob. Chemother. 2012, 67, 2598–2601. [Google Scholar] [CrossRef] [PubMed]
- Canu, A.; Malbruny, B.; Coquemont, M.; Davies, T.A.; Appelbaum, P.C.; Leclercq, R. Diversity of Ribosomal Mutations Conferring Resistance to Macrolides, Clindamycin, Streptogramin, and Telithromycin in Streptococcus pneumoniae. Antimicrob. Agents Chemother. 2002, 46, 125–131. [Google Scholar] [CrossRef]
- Jalava, J.; Vaara, M.; Huovinen, P. Mutation at the position 2058 of the 23S rRNA as a cause of macrolide resistance in Streptococcus pyogenes. Ann. Clin. Microbiol. Antimicrob. 2004, 3, 5. [Google Scholar] [CrossRef][Green Version]
- Karlsson, E.; Golovliov, I.; Lärkeryd, A.; Granberg, M.; Larsson, E.; Öhrman, C.; Niemcewicz, M.; Birdsell, D.; Wagner, D.M.; Forsman, M.; et al. Clonality of erythromycin resistance in Francisella tularensis. J. Antimicrob. Chemother. 2016, 71, 2815–2823. [Google Scholar] [CrossRef]
- Zaman, S.; Fitzpatrick, M.; Lindahl, L.; Zengel, J. Novel mutations in ribosomal proteins L4 and L22 that confer erythromycin resistance in Escherichia coli. Mol. Microbiol. 2007, 66, 1039–1050. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, H.-P.; Jiang, Y.; Hou, S.-P.; Liu, Y.-J.; Liu, Q.-Z. Mutations in 23S rRNA and ribosomal protein L4 account for resistance in Chlamydia trachomatis strains selected in vitro by macrolide passage. Andrologia 2010, 42, 274–280. [Google Scholar] [CrossRef]
- Peric, M.; Bozdogan, B.; Jacobs, M.R.; Appelbaum, P.C. Effects of an Efflux Mechanism and Ribosomal Mutations on Macrolide Susceptibility of Haemophilus influenzae Clinical Isolates. Antimicrob. Agents Chemother. 2003, 47, 1017–1022. [Google Scholar] [CrossRef]
- CLSI. Methods for Antimicrobial Susceptibility Testing for Human Mycoplasmas; Approved Guideline; CLSI Document M43-A; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2011. [Google Scholar]
- CLSI. Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals, 4th ed.; CLSI Supplement VET08; Clinical and Laboratory Standards Institute: Wayne, PA, USA, 2018. [Google Scholar]
- Gautier-Bouchardon, A.V. Antimicrobial resistance in mycoplasma spp. Microbiol. Spectrum. 2018, 6, 425–446. [Google Scholar] [CrossRef]
- Rottem, S. Interaction of Mycoplasmas with Host Cells. Physiol. Rev. 2003, 83, 417–432. [Google Scholar] [CrossRef] [PubMed]
- Sulyok, K.M.; Bekő, K.; Kreizinger, Z.; Wehmann, E.; Jerzsele, Á.; Rónai, Z.; Turcsányi, I.; Makrai, L.; Szeredi, L.; Jánosi, S.; et al. Development of molecular methods for the rapid detection of antibiotic susceptibility of Mycoplasma bovis. Vet. Microbiol. 2018, 213, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Wise, K.S.; Calcutt, M.J.; Foecking, M.F.; Röske, K.; Madupu, R.; Methé, B.A. Complete Genome Sequence of Mycoplasma bovis Type Strain PG45 (ATCC 25523). Infect. Immun. 2011, 79, 982–983. [Google Scholar] [CrossRef]
- Khalil, D.; Becker, C.A.M.; Tardy, F. Monitoring the Decrease in Susceptibility to Ribosomal RNAs Targeting Antimicrobials and Its Molecular Basis in Clinical Mycoplasma bovis Isolates over Time. Microb. Drug Resist. 2017, 23, 799–811. [Google Scholar] [CrossRef]
- Andersen, N.M.; Poehlsgaard, J.; Warrass, R.; Douthwaite, S. Inhibition of Protein Synthesis on the Ribosome by Tildipirosin Compared with Other Veterinary Macrolides. Antimicrob. Agents Chemother. 2012, 56, 6033–6036. [Google Scholar] [CrossRef]
- Kong, L.C.; Gao, D.; Jia, B.Y.; Wang, Z.; Gao, Y.H.; Pei, Z.H.; Liu, S.M.; Xin, J.Q.; Ma, H.X. Antimicrobial susceptibility and molecular characterization of macrolide resistance of Mycoplasma bovis isolates from multiple provinces in China. J. Vet. Med. Sci. 2016, 78, 293–296. [Google Scholar] [CrossRef]
- Liu, M.; Douthwaite, S. Resistance to the macrolide antibiotic tylosin is conferred by single methylations at 23S rRNA nucleotides G748 and A2058 acting in synergy. Proc. Natl. Acad. Sci. USA 2002, 99, 14658–14663. [Google Scholar] [CrossRef]
- Zhao, F.; Liu, J.; Shi, W.; Huang, F.; Liu, L.; Zhao, S.; Zhang, J. Antimicrobial susceptibility and genotyping of Mycoplasma pneumoniae isolates in Beijing, China, from 2014 to 2016. Antimicrob. Resist. Infect. Control 2019, 8, 18. [Google Scholar] [CrossRef]
- Auerbach-Nevo, T.; Baram, D.; Bashan, A.; Belousoff, M.; Breiner, E.; Davidovich, C.; Cimicata, G.; Eyal, Z.; Halfon, Y.; Krupkin, M.; et al. Ribosomal Antibiotics: Contemporary Challenges. Antibiotics 2016, 5, 24. [Google Scholar] [CrossRef]
- Diner, E.J.; Hayes, C.S. Recombineering Reveals a Diverse Collection of Ribosomal Proteins L4 and L22 that Confer Resistance to Macrolide Antibiotics. J. Mol. Biol. 2009, 386, 300–315. [Google Scholar] [CrossRef] [PubMed]
- Pitt, R.; Fifer, H.; Woodford, N.; Alexander, S. Detection of markers predictive of macrolide and fluoroquinolone resistance in Mycoplasma genitalium from patients attending sexual health services in England. Sex. Transm. Infect. 2018, 94, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Day, M.; Cole, M.; Patel, H.; Fifer, H.; Woodford, N.; Pitt, R. P616 Predictive macrolide and fluoroquinolone resistance markers in mycoplasma genitalium from the UK and ireland. Sex. Transm. Infect. 2019, 95, A272. [Google Scholar] [CrossRef]
- Register, K.B.; Thole, L.; Rosenbush, R.F.; Minion, F.C. Multilocus sequence typing of Mycoplasma bovis reveals host-specific genotypes in cattle versus bison. Vet. Microbiol. 2015, 175, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Register, K.B.; Boatwright, W.D.; Gesy, K.M.; Thacker, T.C.; Jelinski, M.D. Mistaken identity of an open reading frame proposed for PCR-based identification of Mycoplasma bovis and the effect of polymorphisms and insertions on assay performance. J. Vet. Diagn. Investig. 2018, 30, 637–641. [Google Scholar] [CrossRef] [PubMed]
- Miles, K.; McAuliffe, L.; Ayling, R.D.; Nicholas, R.A.J. Rapid detection of Mycoplasma dispar and M. bovirhinis using allele specific polymerase chain reaction protocols. FEMS Microbiol. Lett. 2004, 241, 103–107. [Google Scholar] [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R. The Sequence Alignment/Map format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [Google Scholar] [CrossRef] [PubMed]
- Broad Institute; GitHub Repository. Picard Toolkit. Available online: http://broadinstitute.github.io/picard/ (accessed on 15 June 2020).
- Van de Auwera, G.A.; Carneiro, M.O.; Hartl, C.; Poplin, R.; del Angel, G.; Levy-Moonshine, A.; Jordan, T.; Shakir, K.; Roazen, D.; Thibault, J.; et al. From FastQ Data to High-Confidence Variant Calls: The Genome Analysis Toolkit Best Practices Pipeline. Curr. Protoc. Bioinf. 2013, 43, 11. [Google Scholar] [CrossRef]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinf. 2009, 10, 421. [Google Scholar] [CrossRef] [PubMed]
- Vester, B.; Douthwaite, S. Macrolide Resistance Conferred by Base Substitutions in 23S rRNA. Antimicrob. Agents Chemother. 2001, 45, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Sergeant; ESG. Epitools Epidemiological Calculators. Ausvet. Available online: http://epitools.ausvet.com.au. (accessed on 15 June 2020).
- McHugh, M.L. Interrater reliability: The kappa statistic. Biochem. Med. 2012, 22, 276–282. [Google Scholar] [CrossRef]
| Production Year | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| 2006 | 2007 | 2008 | 2014 | 2015 | 2016 | 2017 | 2018 | Total | |
| Anatomical Location | |||||||||
| Joint | 1 | 1 | 1 | 1 | 14 | 11 | 6 | 35 | |
| Lung | 3 | 3 | 17 | 15 | 6 | 44 | |||
| Nasopharynx | 5 | 28 | 9 | 5 | 47 | ||||
| Total | 5 | 29 | 1 | 4 | 4 | 31 | 35 | 17 | 126 |
| Health Status | |||||||||
| Healthy | 2 | 14 | 9 | 5 | 30 | ||||
| Diseased | 3 | 12 | 15 | ||||||
| Dead | 3 | 1 | 4 | 4 | 31 | 26 | 12 | 81 | |
| Total | 5 | 29 | 1 | 4 | 4 | 31 | 35 | 17 | 126 |
| Health Status | Phenotype (% Resistant) | ||||||
|---|---|---|---|---|---|---|---|
| GAM | TIL | TIP | TUL | TYLT | Total | ||
| Healthy | R/S (%R) | 19/11 (63.3) | 30/0 (100) | 30/0 (100) | 11/19 (36.7) | 22/8 (73.3) | 30 |
| Diseased | R/S (%R) | 9/6 (60.0) | 15/0 (100) | 15/0 (100) | 9/6 (60.0) | 11/4 (73.3) | 15 |
| Dead | R/S (%R) | 78/3 (96.3) | 80/1 (98.8) | 81/0 (100) | 72/9 (88.9) | 78/3 (96.3) | 81 |
| Total | R/S (%R) | 106/20 (84.1) | 125/1 (99.2) | 126/0 (100) | 92/34 (73.0) | 111/15 (88.1) | 126 |
| Genotype | 23S rRNA Gene Alleles+ | Percent (n) of Isolates | Phenotype# (% Resistant) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Domain II | Domain V | GAM | TIL | TIP | TUL | TYLT | |||
| 748 | 2059 | 2060 | |||||||
| Wildtype* | G748 | A2059 | A2060 | 1.6 (2) | 1 (50) | 1 (50) | 2 (100) | 0 (0) | 0 (0) |
| Total | 1.6 (2) | 1 (50) | 1 (50) | 2 (100) | 0 (0) | 0 (0) | |||
| Δ748 only | G748, G748A‡ | 4.0 (5) | 5 (100) | 5 (100) | 5 (100) | 0 (0) | 5 (100) | ||
| G748A | 21.4 (27) | 8 (29.6) | 27 (100) | 27 (100) | 0 (0) | 14 (51.9) | |||
| Total | 25.4 (32) | 13 (40.6) | 32 (100) | 32 (100) | 0 (0) | 19 (59.4) | |||
| Δ748Δ2059 | G748, G748A‡ | A2059, A2059G‡ | 0.8 (1) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | |
| G748A | A2059, A2059G‡ | 2.4 (3) | 3 (100) | 3 (100) | 3 (100) | 3 (100) | 3 (100) | ||
| G748A | A2059, A2059C‡ | 2.4 (3) | 3 (100) | 3 (100) | 3 (100) | 3 (100) | 3 (100) | ||
| G748A | A2059, A2059T‡ | 7.1 (9) | 9 (100) | 9 (100) | 9 (100) | 9 (100) | 9 (100) | ||
| G748A | A2059G | 49.2 (62) | 62 (100) | 62 (100) | 62 (100) | 62 (100) | 62 (100) | ||
| Total | 61.9 (78) | 78 (100) | 78 (100) | 78 (100) | 78 (100) | 78 (100) | |||
| Δ748Δ2060 | G748A | A2060, A2060C‡ | 0.8 (1) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | 1 (100) | |
| G748A | A2060C | 7.9 (10) | 10 (100) | 10 (100) | 10 (100) | 10 (100) | 10 (100) | ||
| G748A | A2060G | 2.4 (3) | 3 (100) | 3 (100) | 3 (100) | 3 (100) | 3 (100) | ||
| Total | 11.1 (14) | 14 (100) | 14 (100) | 14 (100) | 14 (100) | 14 (100) | |||
| 23S rRNA Genotype+ | |||||
|---|---|---|---|---|---|
| Wildtype | Δ748 only | Δ748Δ2059 | Δ748Δ2060 | ||
| TUL | R/S | 0/2 | 0/32 | 78/0 | 14/0 |
| Proportion (95% CI) | 0 (0–0.66) | 0 (0–0.11) | 1 (0.95–1) | 1 (0.78–1) | |
| GAM | R/S | 1/1 | 13/19 | 78/0 | 14/0 |
| Proportion (95% CI) | 0.50 (0.09–0.91) | 0.41 (0.26–0.58) | 1 (0.95–1) | 1 (0.78–1) | |
| TYLT | R/S | 0/2 | 19/13 | 78/0 | 14/0 |
| Proportion (95% CI) | 0 (0–0.66) | 0.59 (0.42–0.74) | 1 (0.95–1) | 1 (0.78–1) | |
| TIL | R/S | 1/1 | 32/0 | 78/0 | 14/0 |
| Proportion (95% CI) | 0.50 (0.09–0.91) | 1 (0.89–1) | 1 (0.95–1) | 1 (0.78–1) | |
| TIP | R/S | 2/0 | 32/0 | 78/0 | 14/0 |
| Proportion (95% CI) | 1 (0.34–1) | 1 (0.89–1) | 1 (0.95–1) | 1 (0.78–1) | |
| Total | 2 | 32 | 78 | 14 | |
| 23S rRNA Genotype+ | MIC (µg/mL) | Ribosomal Proteins‡ | |||||
|---|---|---|---|---|---|---|---|
| GAM | TIL | TIP | TUL | TYLT | L4 | L22 | |
| wildtype (PG45) | 8–16 | 1 | 4–8 | 0.25 | 1–2 | Gly185, Thr186 | Gln93 |
| wildtype | 32 | 64 | >128 | 1 | 4 | Gly185Arg | Gln93His |
| Δ748 | 128 | >256 | >128 | 2 | 128 | Gly185Ala, Thr186Pro | Gln93His |
| 128 | >256 | >128 | 8 | 128 | Gly185Ala, Thr186Pro | Gln93His | |
| 64 | >256 | >128 | 0.5 | 64 | Gly185Ala, Thr186Pro | Gln93His | |
| 64 | >256 | >128 | 1 | 64 | Gly185Ala, Thr186Pro | Gln93His | |
| 16 | 256 | 128 | 0.25 | 32 | Gly185Arg | Gln93His | |
| Δ748Δ2059 | >256 | >256 | >128 | >256 | >128 | Gly185Arg | Gln93His |
| >256 | >256 | >128 | 256 | >128 | Gly185Arg | Gln93His | |
| >256 | >256 | >128 | >256 | >128 | Gly185Arg | Gln93His | |
| >256 | >256 | >128 | 128 | 64 | Gly185Arg | Gln93His | |
| >256 | >256 | >128 | 128 | 64 | Gly185Arg | Gln93His | |
| >256 | >256 | >128 | 32 | 64 | Gly185Arg | Gln93His | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kinnear, A.; McAllister, T.A.; Zaheer, R.; Waldner, M.; Ruzzini, A.C.; Andrés-Lasheras, S.; Parker, S.; Hill, J.E.; Jelinski, M.D. Investigation of Macrolide Resistance Genotypes in Mycoplasma bovis Isolates from Canadian Feedlot Cattle. Pathogens 2020, 9, 622. https://doi.org/10.3390/pathogens9080622
Kinnear A, McAllister TA, Zaheer R, Waldner M, Ruzzini AC, Andrés-Lasheras S, Parker S, Hill JE, Jelinski MD. Investigation of Macrolide Resistance Genotypes in Mycoplasma bovis Isolates from Canadian Feedlot Cattle. Pathogens. 2020; 9(8):622. https://doi.org/10.3390/pathogens9080622
Chicago/Turabian StyleKinnear, Andrea, Tim A. McAllister, Rahat Zaheer, Matthew Waldner, Antonio C. Ruzzini, Sara Andrés-Lasheras, Sarah Parker, Janet E. Hill, and Murray D. Jelinski. 2020. "Investigation of Macrolide Resistance Genotypes in Mycoplasma bovis Isolates from Canadian Feedlot Cattle" Pathogens 9, no. 8: 622. https://doi.org/10.3390/pathogens9080622
APA StyleKinnear, A., McAllister, T. A., Zaheer, R., Waldner, M., Ruzzini, A. C., Andrés-Lasheras, S., Parker, S., Hill, J. E., & Jelinski, M. D. (2020). Investigation of Macrolide Resistance Genotypes in Mycoplasma bovis Isolates from Canadian Feedlot Cattle. Pathogens, 9(8), 622. https://doi.org/10.3390/pathogens9080622






