Detection of New Leptospira Genotypes Infecting Symptomatic Dogs: Is a New Vaccine Formulation Needed?
Abstract
1. Introduction
2. Results
2.1. Real-Time PCR
2.2. Isolation
2.3. Genotyping Analyzsis
2.3.1. Multilocus Sequence Typing (MLST)
2.3.2. Multiple Loci Variable-Number Tandem Repeat Analysis (MLVA)
2.4. Sequencing A Tract of the lic12008 Gene on Strains Belonging to ST17
2.5. Data Analysis
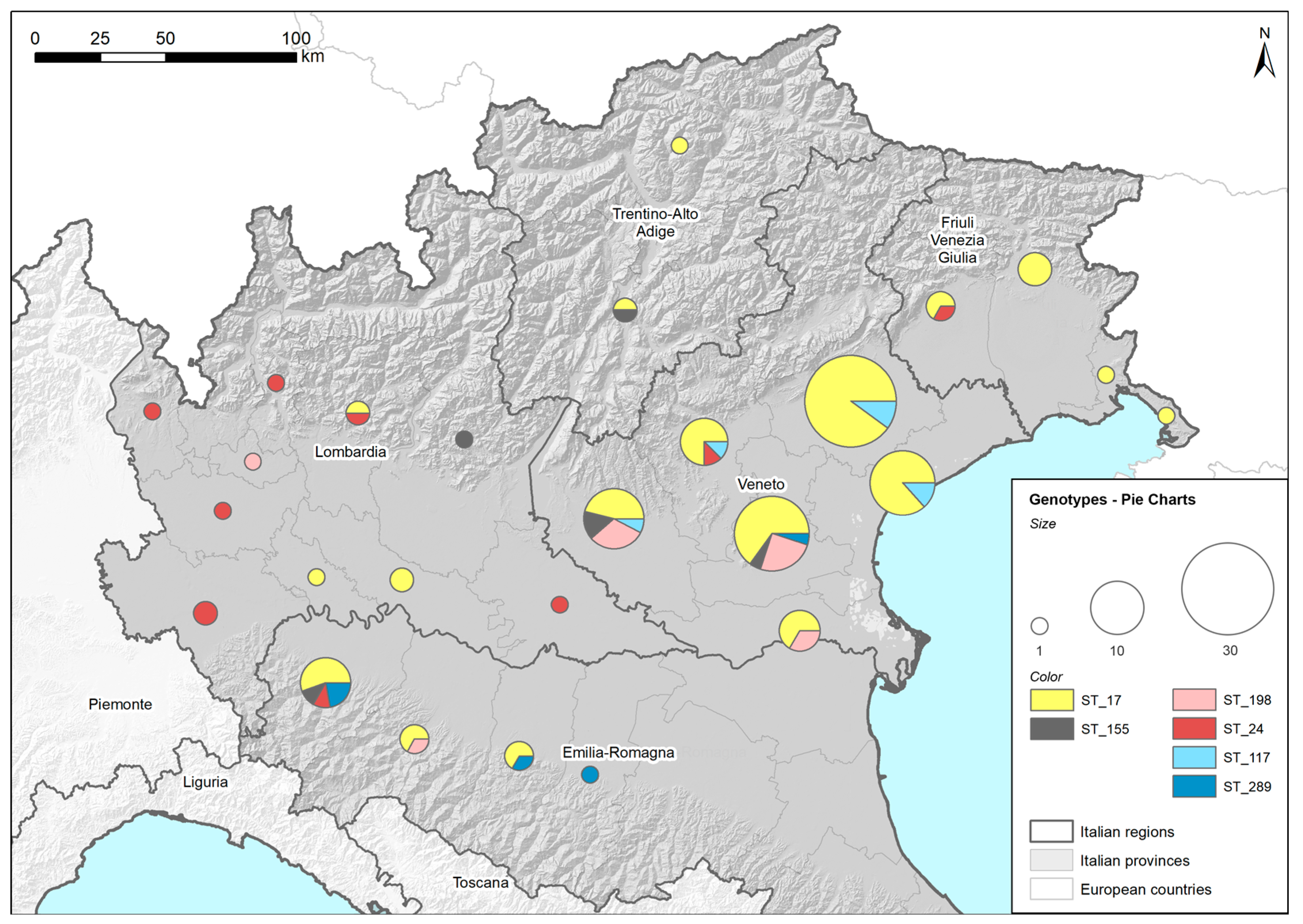
2.5.1. Geographical Maps Based on Found Genotypes
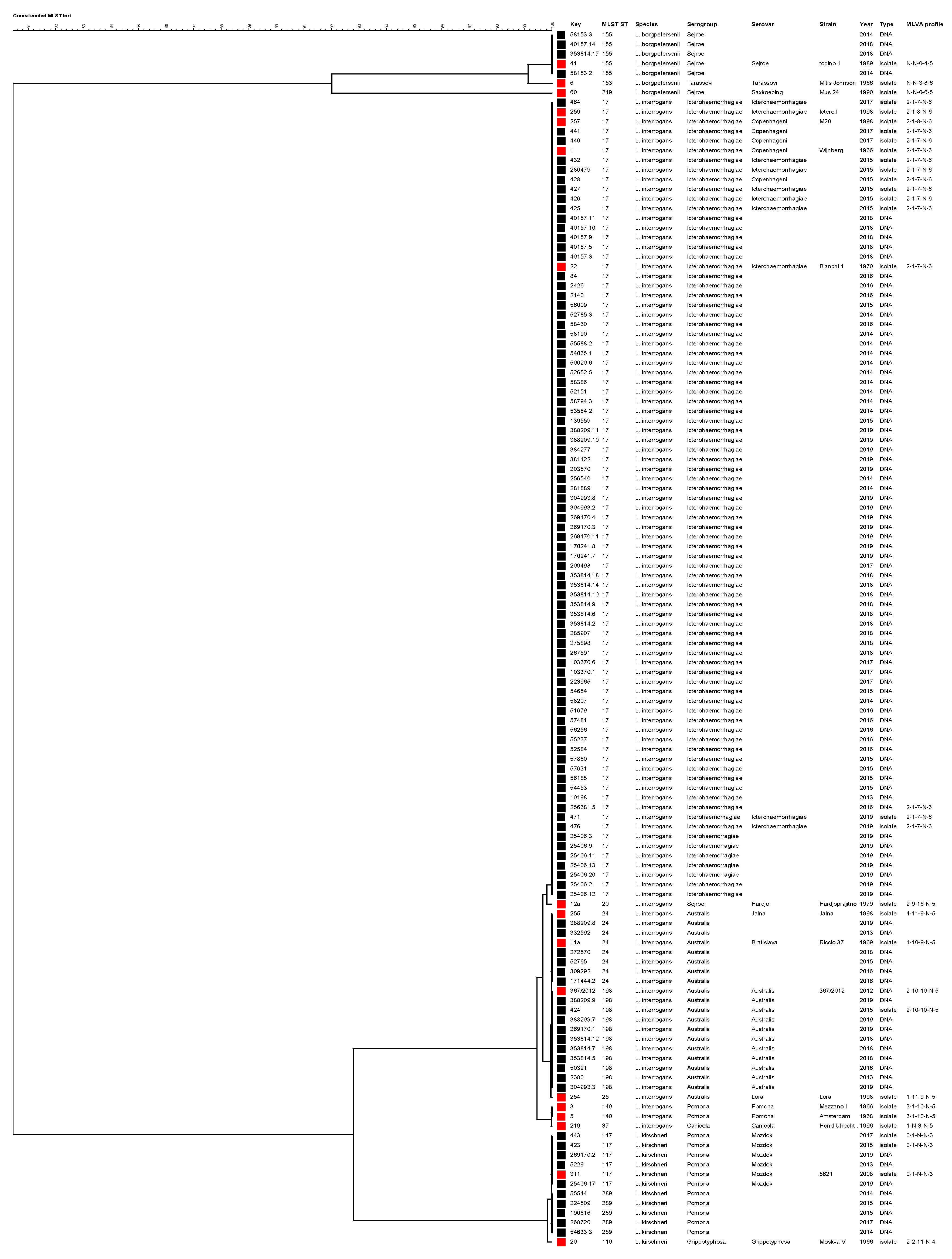
2.5.2. Phylogenetic Analysis Based on Concatenated MLST Loci
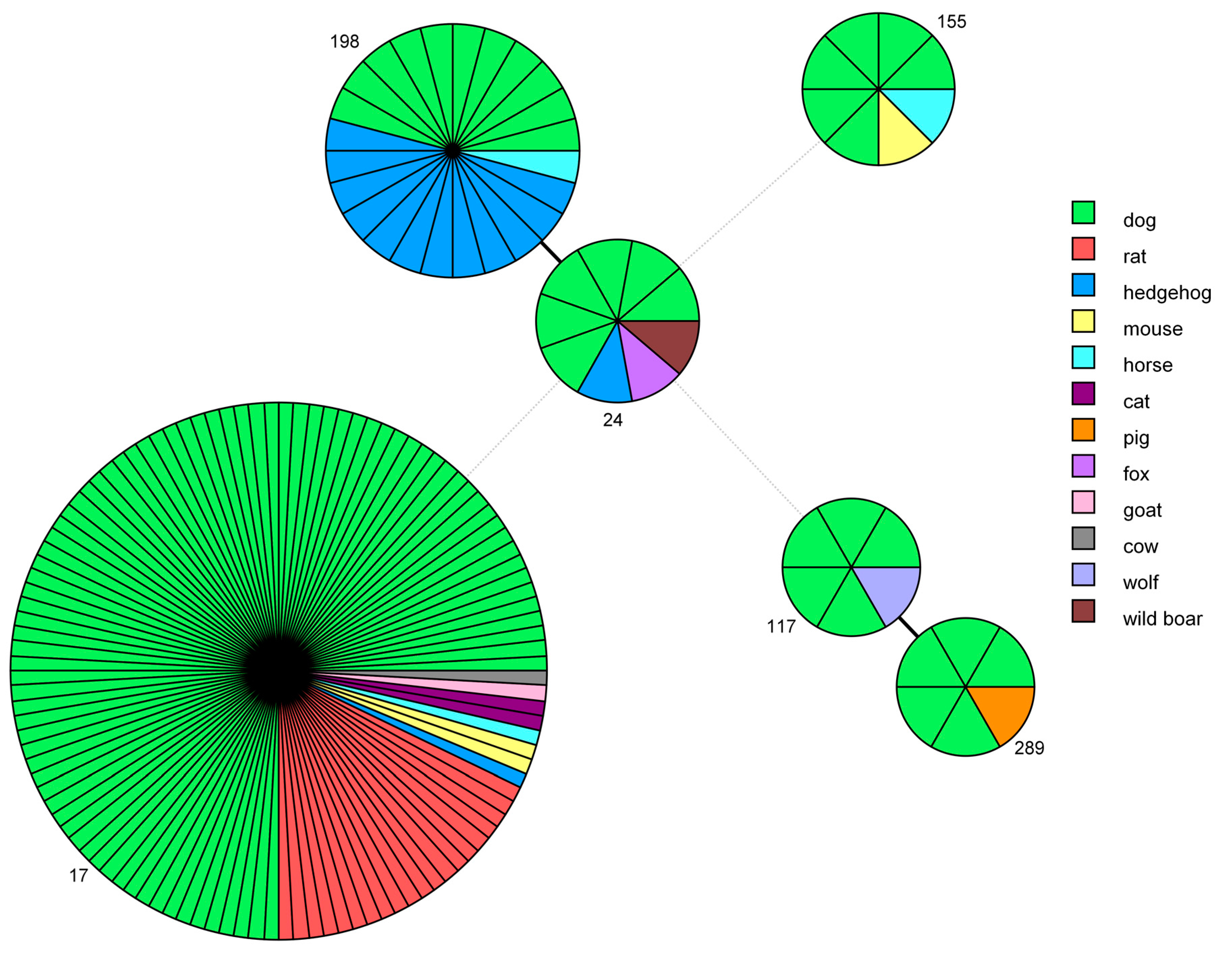
2.5.3. Minimum Spanning Tree Based on MLST Data
3. Discussion
4. Materials and Methods
4.1. Sampling, DNA Extraction and Real-Time PCR
4.2. Isolation and Serological Typing of Isolates
4.3. Genotyping
4.3.1. MLST
4.3.2. MLVA
4.4. Sequencing of a Tract of the lic12008 Gene
4.5. Analysis of Data
4.5.1. Geographical Map
4.5.2. Phylogenetic Analysis of Concatenated MLST Loci
4.5.3. Minimum Spanning Tree based on STs
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schuller, S.; Francey, T.; Hartmann, K.; Hugonnard, M.; Kohn, B.; Nally, J.E.; Sykes, J. European consensus statement on leptospirosis in dogs and cats. J. Small Anim. Pract. 2015, 56, 159–179. [Google Scholar] [CrossRef] [PubMed]
- Klarenbeek, A.; Schuffner, W.A.P. Appearance in Holland of Leptospira differing from Weil Strain. Ned. Tijdschr. Geneeskd. 1933, 77, 4271–4276. [Google Scholar]
- Azócar-Aedo, L.; Monti, G. Meta-Analyses of Factors Associated with Leptospirosis in Domestic Dogs. Zoonoses Public Health 2016, 63, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Zwijnenberg, R.J.G.; Smythe, L.D.; Symonds, M.I.; Dohnt, M.F.; Toribio, J.L.M.L. Cross-sectional study of canine leptospirosis in animal shelter populations in mainland Australia. Aust. Vet. J. 2008, 86, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Shi, D.; Liu, M.; Guo, S.; Liao, S.; Sun, M.; Liu, J.; Wang, L.; Wang, Z.; Wang, S.; Yang, D.; et al. Serological survey of canine leptospirosis in Southern China. Pak. Vet. J. 2012, 32, 280–282. [Google Scholar]
- De Abreu, J.A.P.; da Silva Krawczak, F.; Guedes, I.B.; de Souza-Filho, A.F.; de Souza, G.O.; de Campos Binder, L.; de Oliveira, C.S.; Sponchiado, J.; Melo, G.L.; Labruna, M.B.; et al. Frequency of anti-Leptospira spp. antibodies in dogs and wild small mammals from rural properties and conservation units in southern Brazil. One Health 2019, 8, 100104. [Google Scholar] [CrossRef]
- Khalili, M.; Sakhaee, E.; Amiri, F.B.; Safat, A.A.; Afshar, D.; Esmaeili, S. Serological evidence of leptospirosis in Iran; A systematic review and meta-analysis. Microb. Pathog. 2020, 138, 103833. [Google Scholar] [CrossRef]
- Davis, M.A.; Evermann, J.F.; Petersen, C.R.; VancerSchalie, J.; Besser, T.E.; Huckabee, J.; Daniels, J.B.; Hancock, D.D.; Leslie, M.; Baer, R. Serological survey for antibodies to Leptospira in dogs and raccoons in Washington State. Zoonoses Public Health 2008, 55, 436–442. [Google Scholar] [CrossRef]
- López, M.C.; Vila, A.; Rodón, J.; Roura, X. Leptospira seroprevalence in owned dogs from Spain. Heliyon 2019, 5, e02373. [Google Scholar] [CrossRef]
- Ambily, R.; Mini, M.; Joseph, S.; Krishna, S.V.; Abhinai, G. Canine leptospirosis—A seroprevalence study from Kerala, India. Vet. World 2013, 6, 42–44. [Google Scholar] [CrossRef]
- Scanziani, E.; Origgi, F.; Giusti, A.M.; Iacchia, G.; Vasino, A.; Pirovano, G.; Scarpa, P.; Tagliabue, S. Serological survey of leptospiral infection in kennelled dogs in Italy. J. Small Anim. Pract. 2002, 43, 154–157. [Google Scholar] [CrossRef] [PubMed]
- Tagliabue, S.; Figarolli, B.M.; D’Incau, M.; Foschi, G.; Gennero, M.S.; Giordani, R.; Natale, A.; Papa, P.; Ponti, N.; Scaltrito, D.; et al. Serological surveillance of Leptospirosis in Italy: Two–year national data (2010-2011). Vet. Ital. 2016, 52, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Bertelloni, F.; Cilia, G.; Turchi, B.; Pinzauti, P.; Cerri, D.; Fratini, F. Epidemiology of leptospirosis in North-Central Italy: Fifteen years of serological data (2002–2016). Comp. Immunol. Microbiol. Infect. Dis. 2019, 65, 14–22. [Google Scholar] [CrossRef] [PubMed]
- Jull, D.J.; Heath, K.R. The Evaluation of a Combined L.canicola and L.icterohaemorrhagiae Vaccine on Hamsters and Dogs. J. Small Anim. Pract. 1960, 1, 245–258. [Google Scholar] [CrossRef]
- André-Fontaine, G. Canine leptospirosis-Do we have a problem? Vet. Microbiol. 2006, 117, 19–24. [Google Scholar] [CrossRef]
- Sykes, J.E.; Hartmann, K.; Lunn, K.F.; Moore, G.E.; Stoddard, R.A.; Goldstein, R.E. 2010 ACVIM Small Animal Consensus Statement on Leptospirosis: Diagnosis, Epidemiology, Treatment, and Prevention. J. Vet. Intern. Med. 2011, 25, 1–13. [Google Scholar] [CrossRef]
- Ellis, W.A. Control of canine leptospirosis in Europe: Time for a change? Vet. Rec. 2010, 167, 602–605. [Google Scholar] [CrossRef]
- Mayer-Scholl, A.; Luge, E.; Draeger, A.; Nöckler, K.; Kohn, B. Distribution of Leptospira Serogroups in Dogs from Berlin, Germany. Vector-Borne Zoonotic Dis. 2013, 13, 200–202. [Google Scholar] [CrossRef]
- Renaud, C.; Andrews, S.; Djelouadji, Z.; Lecheval, S.; Corrao-Revol, N.; Buff, S.; Demont, P.; Kodjo, A. Prevalence of the Leptospira serovars bratislava, grippotyphosa, mozdok and pomona in French dogs. Vet. J. 2013, 196, 126–127. [Google Scholar] [CrossRef]
- Klaasen, H.L.B.M.; Van Der Veen, M.; Molkenboer, M.J.C.H.; Sutton, D. A novel tetravalent Leptospira bacterin protects against infection and shedding following challenge in dogs. Vet. Rec. 2013, 172, 181. [Google Scholar] [CrossRef]
- Klaasen, H.L.B.M.; van der Veen, M.; Sutton, D.; Molkenboer, M.J.C.H. A new tetravalent canine leptospirosis vaccine provides at least 12 months immunity against infection. Vet. Immunol. Immunopathol. 2014, 158, 26–29. [Google Scholar] [CrossRef] [PubMed]
- Levett, P.N. Leptospirosis. Clin. Microbiol. Rev. 2001, 14, 296–326. [Google Scholar] [CrossRef]
- Leptospirosis. In OIE Terrestrial Manual; World Organization for Animal Health: Paris, France, 2018; pp. 503–516.
- Strutzberg-Minder, K.; Tschentscher, A.; Beyerbach, M.; Homuth, M.; Kreienbrock, L. Passive surveillance of Leptospira infection in swine in Germany. Porc. Health Manag. 2018, 4, 10. [Google Scholar] [CrossRef] [PubMed]
- Levett, P.N. Usefulness of Serologic Analysis as a Predictor of the Infecting Serovar in Patients with Severe Leptospirosis. Clin. Infect. Dis. 2003, 36, 447–452. [Google Scholar] [CrossRef] [PubMed]
- Martin, L.E.R.; Wiggans, K.T.; Wennogle, S.A.; Curtis, K.; Chandrashekar, R.; Lappin, M.R. Vaccine-Associated Leptospira Antibodies in Client-Owned Dogs. J. Vet. Intern. Med. 2014, 28, 789–792. [Google Scholar] [CrossRef]
- Stoddard, R.A.; Gee, J.E.; Wilkins, P.P.; McCaustland, K.; Hoffmaster, A.R. Detection of pathogenic Leptospira spp. through TaqMan polymerase chain reaction targeting the LipL32 gene. Diagn. Microbiol. Infect. Dis. 2009, 64, 247–255. [Google Scholar] [CrossRef]
- Levett, P.N.; Morey, R.E.; Galloway, R.L.; Turner, D.E.; Steigerwalt, A.G.; Mayer, L.W. Detection of pathogenic leptospires by real-time quantitative PCR. J. Med. Microbiol. 2005, 54, 45–49. [Google Scholar] [CrossRef]
- Smythe, L.D.; Smith, I.L.; Smith, G.A.; Dohnt, M.F.; Symonds, M.L.; Barnett, L.J.; McKay, D.B. A quantitative PCR (TaqMan) assay for pathogenic Leptospira spp. BMC Infect. Dis. 2002, 2, 13. [Google Scholar] [CrossRef]
- Merien, F.; Portnoi, D.; Bourhy, P.; Charavay, F.; Berlioz-Arthaud, A.; Baranton, G. A rapid and quantitative method for the detection of Leptospira species in human leptospirosis. FEMS Microbiol. Lett. 2005, 249, 139–147. [Google Scholar] [CrossRef]
- Ahmed, A.; Engelberts, M.F.M.; Boer, K.R.; Ahmed, N.; Hartskeerl, R.A. Development and validation of a real-time PCR for detection of pathogenic Leptospira species in clinical materials. PLoS ONE 2009, 4, e7093. [Google Scholar] [CrossRef]
- Slack, A.T.; Symonds, M.L.; Dohnt, M.F.; Smythe, L.D. Identification of pathogenic Leptospira species by conventional or real-time PCR and sequencing of the DNA gyrase subunit B encoding gene. BMC Microbiol. 2006, 6, 95. [Google Scholar] [CrossRef] [PubMed]
- Bourhy, P.; Bremont, S.; Zinini, F.; Giry, C.; Picardeau, M. Comparison of real-time PCR assays for detection of pathogenic Leptospira spp. in blood and identification of variations in target sequences. J. Clin. Microbiol. 2011, 49, 2154–2160. [Google Scholar] [CrossRef] [PubMed]
- Harkin, K.R.; Roshto, Y.M.; Sullivan, J.T.; Purvis, T.J.; Chengappa, M.M. Comparison of polymerase chain reaction assay, bacteriologic culture, and serologic testing in assessment of prevalence of urinary shedding of leptospires in dogs. J. Am. Vet. Med. Assoc. 2003, 222, 1230–1233. [Google Scholar] [CrossRef]
- Midence, J.N.; Leutenegger, C.M.; Chandler, A.M.; Goldstein, R.E. Effects of recent Leptospira vaccination on whole blood real-time PCR testing in healthy client-owned dogs. J. Vet. Intern. Med. 2012, 26, 149–152. [Google Scholar] [CrossRef] [PubMed]
- Boonsilp, S.; Thaipadungpanit, J.; Amornchai, P.; Wuthiekanun, V.; Bailey, M.S.; Holden, M.T.G.; Zhang, C.; Jiang, X.; Koizumi, N.; Taylor, K.; et al. A single multilocus sequence typing (MLST) scheme for seven pathogenic Leptospira species. PLoS Negl. Trop. Dis. 2013, 7, e1954. [Google Scholar] [CrossRef] [PubMed]
- Salaün, L.; Mérien, F.; Gurianova, S.; Baranton, G.; Picardeau, M. Application of Multilocus Variable-Number Tandem-Repeat Analysis for Molecular Typing of the Agent of Leptospirosis. J. Clin. Microbiol. 2006, 44, 3954–3962. [Google Scholar] [CrossRef]
- Weiss, S.; Menezes, A.; Woods, K.; Chanthongthip, A.; Dittrich, S.; Opoku-Boateng, A.; Simuli, M.; Chalke, V. An Extended Multilocus Sequence Typing (MLST) Scheme for Rapid Direct Typing of Leptospira from Clinical Samples. PLoS Negl. Trop. Dis. 2016, 10, e0004996. [Google Scholar] [CrossRef]
- Klaasen, H.L.B.M.; Molkenboer, M.J.C.H.; Vrijenhoek, M.P.; Kaashoek, M.J. Duration of immunity in dogs vaccinated against leptospirosis with a bivalent inactivated vaccine. Vet. Microbiol. 2003, 95, 121–132. [Google Scholar] [CrossRef]
- Wilson, S.; Stirling, C.; Thomas, A.; King, V.; Plevová, E.; Chromá, L.; Siedek, E.; Illambas, J.; Salt, J.; Sture, G. Duration of immunity of a multivalent (DHPPi/L4R) canine vaccine against four Leptospira serovars. Vaccine 2013, 31, 3126–3130. [Google Scholar] [CrossRef]
- Boniotti, M.B.; Gelmini, L.; Carra, E.; Figarolli, B.M.; D’incau, M.; Tagliabue, S. Leptospira Interrogans Serogroup Australis In Hedgehog In Northern Italy. Proocedings of the International Leptospirosis Society of the Conference, Fukuoka, Japan, 7–11 October 2013. [Google Scholar]
- Bertasio, C.; Papetti, A.; Scaltriti, E.; Tagliabue, S.; D’Incau, M.; Boniotti, M.B. Serological Survey and Molecular Typing Reveal New Leptospira Serogroup Pomona Strains among Pigs of Northern Italy. Pathogens 2020, 9, 332. [Google Scholar] [CrossRef]
- Modric, Z.; Culjak, K.; Hahn, V. Leptospirosis in a dog caused by Leptospira interrogans serotype pomona. Vet. Glas. 1987, 41, 43–47. [Google Scholar]
- Vojinović, D.; Bogićević, N.; Vasić, A.; Manić, M.; Elezović Radovanović, M.; Rogožarski, D.; Marić, J.; Valčić, M. Seroepidemiological survey of leptospiral infection in stray dogs in Serbia. Turkish J. Vet. Anim. Sci. 2015, 39, 719–723. [Google Scholar] [CrossRef]
- Burriel, A.R.; Dalley, C.; Woodward, M.J. Prevalence of Leptospira species among farmed and domestic animals in Greece. Vet. Rec. 2003, 153, 146–148. [Google Scholar] [CrossRef]
- Cerri, D.; Ebani, V.V.; Fratini, F.; Pinzauti, P.; Andreani, E. Epidemiology of leptospirosis: Observations on serological data obtained by a “diagnostic laboratory for leptospirosis” from 1995 to 2001. New Microbiol. 2003, 26, 383–389. [Google Scholar]
- Felzemburgh, R.D.M.; Ribeiro, G.S.; Costa, F.; Reis, R.B.; Hagan, J.E.; Melendez, A.X.T.O.; Fraga, D.; Santana, F.S.; Mohr, S.; dos Santos, B.L.; et al. Prospective Study of Leptospirosis Transmission in an Urban Slum Community: Role of Poor Environment in Repeated Exposures to the Leptospira Agent. PLoS Negl. Trop. Dis. 2014, 8, e2927. [Google Scholar] [CrossRef]
- Oliveira, M.A.A.; Leal, É.A.; Correia, M.A.; Serufo Filho, J.C.; Dias, R.S.; Serufo, J.C. Human leptospirosis: Occurrence of serovars of Leptospira spp. in the state of Minas Gerais, Brazil, from 2008 to 2012. Braz. J. Microbiol. 2017, 48, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Suepaul, S.M.; Carrington, C.V.F.; Campbell, M.; Borde, G.; Adesiyun, A.A. Serovars of Leptospira isolated from dogs and rodents. Epidemiol. Infect. 2010, 138, 1059–1070. [Google Scholar] [CrossRef] [PubMed]
- Boey, K.; Shiokawa, K.; Rajeev, S. Leptospira infection in rats: A literature review of global prevalence and distribution. PLoS Negl. Trop. Dis. 2019, 13, e0007499. [Google Scholar] [CrossRef]
- Ghneim, G.S.; Viers, J.H.; Chomel, B.B.; Kass, P.H.; Descollonges, D.A.; Johnson, M.L. Use of a case-control study and geographic information systems to determine environmental and demographic risk factors for canine leptospirosis. Vet. Res. 2007, 38, 37–50. [Google Scholar] [CrossRef]
- Major, A.; Schweighauser, A.; Francey, T. Increasing incidence of canine leptospirosis in Switzerland. Int. J. Environ. Res. Public Health 2014, 11, 7242–7260. [Google Scholar] [CrossRef]
- Schuller, S.; Arent, Z.J.; Gilmore, C.; Nally, J. Prevalence of antileptospiral serum antibodies in dogs in Ireland. Vet. Rec. 2015, 177, 126. [Google Scholar] [CrossRef] [PubMed]
- Santos, L.A.; Adhikarla, H.; Yan, X.; Wang, Z.; Fouts, D.E.; Vinetz, J.M.; Alcantara, L.C.J.; Hartskeerl, R.A.; Goris, M.G.A.; Picardeau, M.; et al. Genomic comparison among global isolates of L. interrogans serovars Copenhageni and Icterohaemorrhagiae identified natural genetic variation caused by an indel. Front. Cell. Infect. Microbiol. 2018, 8, 193. [Google Scholar] [CrossRef] [PubMed]
- Rissi, D.R.; Brown, C.A. Diagnostic features in 10 naturally occurring cases of acute fatal canine leptospirosis. J. Vet. Diagn. Investig. 2014, 26, 799–804. [Google Scholar] [CrossRef] [PubMed]
- CE, N.; GJ, K.; JJ, K. Serum biochemical changes in dogs with experimental Leptospira interrogans serovar icterohaemorrhagiae infection. Am. J. Vet. Res. 1981, 42, 1125–1129. [Google Scholar]
- Balboni, A.; Zamagni, S.; Bertasio, C.; Boniotti, M.B.; Troìa, R.; Battilani, M.; Dondi, F. Identification of Serogroups Australis and Icterohaemorrhagiae in Two Dogs with a Severe Form of Acute Leptospirosis in Italy. Pathogens 2020, 9, 351. [Google Scholar] [CrossRef]
- Jolley, K.A.; Maiden, M.C.J. BIGSdb: Scalable analysis of bacterial genome variation at the population level. BMC Bioinform. 2010, 11, 595. [Google Scholar] [CrossRef]
- Van den Broek, A.H.M.; Thrusfield, M.V.; Dobbiet, G.R.; Ellisi, W.A. A serological and bacteriological survey of leptospiral infection in dogs in Edinburgh and Glasgow. J. Small Anim. Pract. 1991, 32, 118–124. [Google Scholar] [CrossRef]
- Tagliabue, S.; Farina, R. Inchiesta sieroepidemiologica sulla diffusione della leptospirosi ta gli animali domestici ed alcune specie selvatiche. Sel. Vet. 1995, 36, 11–12. [Google Scholar]
- Chiari, M.; Figarolli, B.M.; Tagliabue, S.; Alborali, G.L.; Bertoletti, M.; Papetti, A.; D’Incau, M.; Zanoni, M.; Boniotti, M.B. Seroprevalence and risk factors of leptospirosis in wild boars (Sus scrofa) in northern Italy. Hystrix 2016, 27, 2. [Google Scholar] [CrossRef]
- Das Neves Paiva-Cardoso, M.; Arent, Z.; Gilmore, C.; Hartskeerl, R.; Ellis, W.A. Altodouro, a new Leptospira serovar of the Pomona serogroup isolated from rodents in northern Portugal. Infect. Genet. Evol. 2013, 13, 211–217. [Google Scholar] [CrossRef]
- Rocha, T.; Ellis, W.A.; Montgomery, J.; Gilmore, C.; Regalla, J.; Brem, S. Microbiological and serological study of leptospirosis in horses at slaughter: First isolations. Res. Vet. Sci. 2004, 76, 199–202. [Google Scholar] [CrossRef]
- Rocha, T. Isolation of Leptospira interrogans serovar mozdok from aborted swine fetuses in Portugal. Vet. Rec. 1990, 126, 602. [Google Scholar]
- Majetic, Z.S.; Galloway, R.; Sabljic, E.R.; Milas, Z.; Perko, V.M.; Habus, J.; Margaletic, J.; Pernar, R.; Turk, N. Epizootiological survey of small mammals as Leptospira spp. reservoirs in Eastern Croatia. Acta Trop. 2014, 131, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Pritchard, D.G.; Todd, N.; Barlow, A.; Little, S.A. Outbreak of Leptospira interrogans serovar mozdok in sows in Dorset, England. Isr. J. Vet. Med. 1987, 43, 343. [Google Scholar]
- Barlow, A.M. Reproductive failure in sows associated with Leptospira Mozdok from a wildlife source. Pig J. 2004, 54, 123–131. [Google Scholar]
- Hathaway, S.C.; Todd, J.N.; Headlam, S.A.; Jeffrey, M. Possible role of leptospires of the Pomona serogroup in sporadic bovine abortion in the south west of England. Vet. Rec. 1984, 115, 623–626. [Google Scholar] [CrossRef]
- Da Cunha, C.E.P.; Felix, S.R.; Neto, A.C.P.S.; Campello-Felix, A.; Kremer, F.S.; Monte, L.G.; Amaral, M.G.; Nobre, M.D.O.; Da Silva, É.F.; Hartleben, C.P.; et al. Infection with Leptospira kirschneri Serovar Mozdok: First Report from the Southern Hemisphere. Am. J. Trop. Med. Hyg. 2016, 94, 519–521. [Google Scholar] [CrossRef] [PubMed]
- Greenlee, J.J.; Alt, D.P.; Bolin, C.A.; Zuerner, R.L.; Andreasen, C.B. Experimental canine leptospirosis caused by Leptospira interrogans serovars pomona and bratislava. Am. J. Vet. Res. 2005, 66, 1816–1822. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, R.E.; Lin, R.C.; Langston, C.E.; Scrivani, P.V.; Erb, H.N.; Barr, S.C. Influence of infecting serogroup on clinical features of leptospirosis in dogs. J. Vet. Intern. Med. 2006, 20, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Rühl-Fehlert, C.I.; Brem, S.; Feller, W.; Kopp, H.; Meyer, P.; Rinke, M. Clinical, microbiological and pathological observations in laboratory beagle dogs infected with leptospires of the serogroup sejroe. Exp. Toxicol. Pathol. 2000, 52, 201–207. [Google Scholar] [CrossRef]
- Scanziani, E.; Crippa, L.; Giusti, A.M.; Luini, M.; Pacciarini, M.L.; Tagliabue, S.; Cavalletti, E. Leptospira interrogans serovar sejroe infection in a group of laboratory dogs. Lab. Anim. 1995, 29, 300–306. [Google Scholar] [CrossRef] [PubMed]
- Miotto, B.A.; Tozzi, B.F.; de Souza Penteado, M.; Guilloux, A.G.A.; Moreno, L.Z.; Heinemann, M.B.; Moreno, A.M.; Lilenbaum, W.; Hagiwara, M.K. Diagnosis of acute canine leptospirosis using multiple laboratory tests and characterization of the isolated strains. BMC Vet. Res. 2018, 14, 222. [Google Scholar] [CrossRef]
- Miotto, B.A.; Moreno, L.Z.; Guilloux, A.G.A.; de Sousa, G.O.; Loureiro, A.P.; Moreno, A.M.; Lilenbaum, W.; Vasconcellos, S.A.; Heinemann, M.B.; Hagiwara, M.K. Molecular and serological characterization of the first Leptospira santarosai strain isolated from a dog. Acta Trop. 2016, 162, 1–4. [Google Scholar] [CrossRef]
- Claus, A.; Van de Maele, I.; Pasmans, F.; Gommeren, K.; Daminet, S. Leptospirosis in dogs: A retrospective study of seven clinical cases in Belgium. Vlaams Diergeneeskd. Tijdschr. 2008, 77, 259–263. [Google Scholar]
- Fennestad, K.L.; Borg-Petersen, C. Leptospirosis in Danish wild mammals. J. Wildl. Dis. 1972, 8, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Borčić, B.; Kovačić, H.; Šebek, Z.; Aleraj, B.; Tvrtković, N. Small terrestrial mammals as reservoirs of leptospires in Sava valley (Croatia). Folia Parasitol. 2005, 29, 177–182. [Google Scholar]
- Steinen, A.C.; Schuurman, J.L.; Gravekamp, C.; Korver, H.; Terpstra, W.J. Muskrats as carriers of pathogenic leptospires in The Netherlands. Antonie Van Leeuwenhoek 1992, 61, 43–50. [Google Scholar] [CrossRef]
- Korver, H.; Kolk, A.; Vingerhoed, J.; Van Leeuwen, J.; Terpstra, W. Classification of serovars of the icterohaemorrhagiae serogroup by monoclonal antibodies. Isr. J. Vet. 1988, 44, 15–18. [Google Scholar]
- Terpstra, W.J.; Korver, H.; Schoone, G.J.; von Leeuwen, J.; Schönemann, C.E.; de Jonge-Aglibut, S.; Kolk, A.H. Comparative classification of Leptospira serovars of the Pomona group by monoclonal antibodies and restriction-endonuclease analysis. Zentralbl. Bakteriol. Mikrobiol. Hyg. A. 1987, 266, 412–421. [Google Scholar] [CrossRef]
- Cinco, M.; Banfi, E.; Stornello, C.; Campo, F.; Korver, H. First human isolate of Leptospira interrogans as serovar bratislava in Italy. FEMS Microbiol. Lett. 1989, 47, 499–503. [Google Scholar] [CrossRef][Green Version]
| Urine | Blood | Kidney | Liver | Lung | Bladder | Spleen | Organ(s) | Not Defined | Total | |
|---|---|---|---|---|---|---|---|---|---|---|
| No. of Positives/Total | 162/1095 | 66/665 | 75/392 | 23/115 | 7/33 | 2/9 | 0/6 | 12/148 | 0/22 | 347/2485 |
| Percentage of Positives | 14.8% | 9.9% | 18.6% | 20.0% | 21.2% | 22.2% | 0.0% | 8.1% | 0.0% | 14.0% |
| No. of Isolates | Species | Serogroup | Serovar |
|---|---|---|---|
| 8 | L. interrogans | Icterohaemorrhagiae | Icterohaemorrhagiae |
| 3 | L. interrogans | Icterohaemorrhagiae | Copenhageni |
| 2 | L. kirschneri | Pomona | Mozdok |
| 2 | L. interrogans | Australis | Australis |
| Genotyped Samples | ST 17 | ST 198 | ST 24 | ST 117 | ST 155 | ST 289 | ST 17 Like | Total |
|---|---|---|---|---|---|---|---|---|
| No. of DNA | 65 (+17) | 8 (+3) | 6 (+4) | 3 (+2) | 4 (+2) | 5 | (1) | 91 (+29) |
| No. of Isolates | 11 | 2 | 2 | 15 | ||||
| Total | 76 (+17) | 10 (+3) | 6 (+4) | 5 (+2) | 4 (+2) | 5 | (1) | 135 |
| % | 68.9% | 9.6% | 7.4% | 5.2% | 4.4% | 3.7% | 0.7% | 100.0% |
| ST of Infecting Leptospira | Vaccination Status | Vital Status | Total | ||||
|---|---|---|---|---|---|---|---|
| Regular | Not Regular | Unknown | Alive | Died | Unknown | ||
| 17 | 3 | 23 | 65 | 5 | 32 | 56 | 91 |
| 198 | 6 | 1 | 6 | 4 | 1 | 8 | 13 |
| 24 | 1 | 9 | 1 | 9 | 10 | ||
| 117 | 1 | 1 | 5 | 1 | 3 | 3 | 7 |
| 155 | 1 | 2 | 3 | 1 | 5 | 6 | |
| 289 | 1 | 4 | 1 | 4 | 5 | ||
| 17 Like | 1 | 1 | 1 | ||||
| Total | 12 | 28 | 95 | 10 | 39 | 86 | 135 |
| No. | Type | Species | Sequence Type | Serovar Definition | |
|---|---|---|---|---|---|
| By lic12008 Sequencing | By MAT | ||||
| 8 | isolate | L. interrogans | 17 | Icterohaemorrhagiae | Icterohaemorrhagiae |
| 2 | Isolate | L. interrogans | 17 | Icterohaemorrhagiae | Copenhageni |
| 1 | Isolate | L. interrogans | 17 | Copenhageni | Copenhageni |
| 52 | DNA | L. interrogans | 17 | Icterohaemorrhagiae | N.A. |
| 3 | DNA | L. interrogans | 17 | Copenhageni | N.A. |
| Matrix | No. of DNA Extracted from Biological Samples | No. of Isolated Samples | Total |
|---|---|---|---|
| Urine | 101 | 15 | 120 |
| Blood | 45 | 45 | |
| Kidney | 34 | 34 | |
| Lung | 5 | 5 | |
| Organ(s) | 6 | 6 | |
| Liver | 4 | 4 | |
| Bladder | 3 | 3 | |
| Not defined | 10 | 10 | |
| Total | 208 | 15 | 223 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bertasio, C.; Boniotti, M.B.; Lucchese, L.; Ceglie, L.; Bellinati, L.; Mazzucato, M.; Furlanello, T.; D’Incau, M.; Natale, A. Detection of New Leptospira Genotypes Infecting Symptomatic Dogs: Is a New Vaccine Formulation Needed? Pathogens 2020, 9, 484. https://doi.org/10.3390/pathogens9060484
Bertasio C, Boniotti MB, Lucchese L, Ceglie L, Bellinati L, Mazzucato M, Furlanello T, D’Incau M, Natale A. Detection of New Leptospira Genotypes Infecting Symptomatic Dogs: Is a New Vaccine Formulation Needed? Pathogens. 2020; 9(6):484. https://doi.org/10.3390/pathogens9060484
Chicago/Turabian StyleBertasio, Cristina, Maria Beatrice Boniotti, Laura Lucchese, Letizia Ceglie, Laura Bellinati, Matteo Mazzucato, Tommaso Furlanello, Mario D’Incau, and Alda Natale. 2020. "Detection of New Leptospira Genotypes Infecting Symptomatic Dogs: Is a New Vaccine Formulation Needed?" Pathogens 9, no. 6: 484. https://doi.org/10.3390/pathogens9060484
APA StyleBertasio, C., Boniotti, M. B., Lucchese, L., Ceglie, L., Bellinati, L., Mazzucato, M., Furlanello, T., D’Incau, M., & Natale, A. (2020). Detection of New Leptospira Genotypes Infecting Symptomatic Dogs: Is a New Vaccine Formulation Needed? Pathogens, 9(6), 484. https://doi.org/10.3390/pathogens9060484











