Gut Microbiome in Psoriasis: An Updated Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Inclusion/Exclusion Criteria
- Subjects aged 18 years or older;
- Human case-control studies investigating the association between gut microbiota and psoriasis
- usage of culture-independent, high-throughput sequencing methods for gut microbiota quantification;
- Articles published in English.
2.3. Study Selection
2.4. Quality Assessment
- Selection (adequate case definition, representativeness of the cases, selection of controls, definition of controls);
- Comparability (factors that the study controlled for by design or analysis to improve the comparability of baseline characteristics of cases and controls);
- Exposure (ascertainment of exposure, same method of ascertainment for cases and controls, nonresponse rate).
2.5. Data Extraction
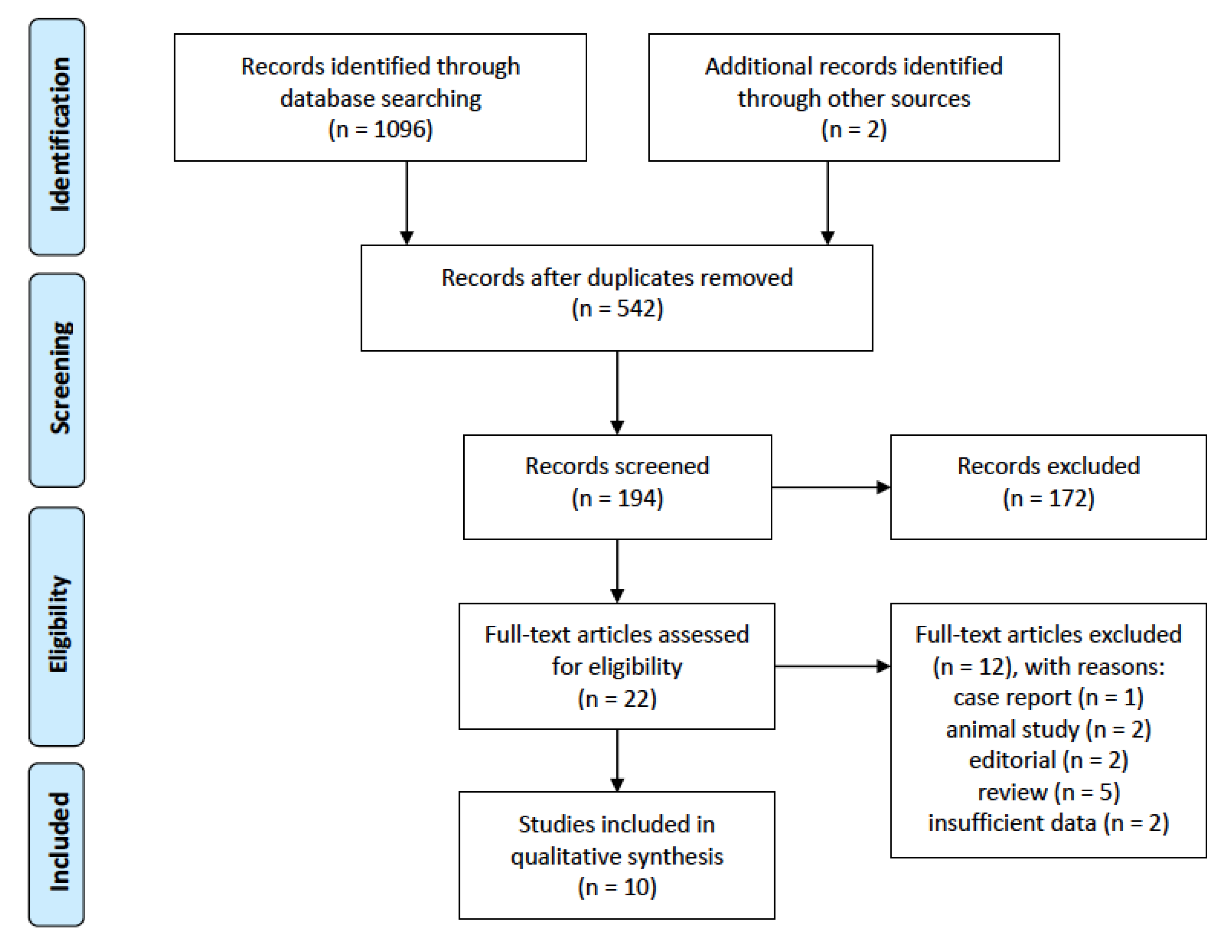
3. Results
3.1. Search Results and Study Characteristics
3.1.1. Gut Microbiome Diversity in Psoriasis
3.1.2. Gut Microbiome Alterations in Psoriasis
3.1.3. Changes in Gut Microbiota after Antipsoriatic Treatment
3.1.4. Quality of the Evidence
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Korman, N.J. Management of psoriasis as a systemic disease: What is the evidence? Br. J. Dermatol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Adarsh, M.B.; Dogra, S.; Vaiphei, K.; Vaishnavi, C.; Sinha, S.K.; Sharma, A. Evaluation of subclinical gut inflammation using faecal calprotectin levels and colonic mucosal biopsy in patients with psoriasis and psoriatic arthritis. Br. J. Dermatol. 2019, 181, 401–402. [Google Scholar] [CrossRef] [PubMed]
- Sikora, M.; Chrabaszcz, M.; Maciejewski, C.; Zaremba, M.; Waskiel, A.; Olszewska, M.; Rudnicka, L. Intestinal barrier integrity in patients with plaque psoriasis. J. Dermatol. 2018, 45, 1468–1470. [Google Scholar] [CrossRef]
- Lin, S.; Wang, Z.; Lam, K.L.; Zeng, S.; Tan, B.K.; Hu, J. Role of intestinal microecology in the regulation of energy metabolism by dietary polyphenols and their metabolites. Food Nutr. Res. 2019, 63. [Google Scholar] [CrossRef] [PubMed]
- Spencer, S.P.; Fragiadakis, G.K.; Sonnenburg, J.L. Pursuing Human-Relevant Gut Microbiota-Immune Interactions. Immunity 2019, 51, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Chelakkot, C.; Ghim, J.; Ryu, S.H. Mechanisms regulating intestinal barrier integrity and its pathological implications. Exp. Mol. Med. 2018, 50, 103. [Google Scholar] [CrossRef] [PubMed]
- Vallianou, N.; Stratigou, T.; Christodoulatos, G.S.; Dalamaga, M. Understanding the Role of the Gut Microbiome and Microbial Metabolites in Obesity and Obesity-Associated Metabolic Disorders: Current Evidence and Perspectives. Curr. Obes. Rep. 2019, 8, 317–332. [Google Scholar] [CrossRef] [PubMed]
- Scher, J.U.; Littman, D.R.; Abramson, S.B. Microbiome in Inflammatory Arthritis and Human Rheumatic Diseases. Arthritis Rheumatol. 2016, 68, 35–45. [Google Scholar] [CrossRef]
- Stevens, B.R.; Roesch, L.; Thiago, P.; Russell, J.T.; Pepine, C.J.; Holbert, R.C.; Raizada, M.K.; Triplett, E.W. Depression phenotype identified by using single nucleotide exact amplicon sequence variants of the human gut microbiome. Mol. Psychiatry 2020. [Google Scholar] [CrossRef]
- Kitai, T.; Tang, W.H.W. Gut microbiota in cardiovascular disease and heart failure. Clin. Sci. 2018, 132, 85–91. [Google Scholar] [CrossRef]
- Nishida, A.; Inoue, R.; Inatomi, O.; Bamba, S.; Naito, Y.; Andoh, A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin. J. Gastroenterol. 2018, 11, 1–10. [Google Scholar] [CrossRef]
- Zakostelska, Z.; Malkova, J.; Klimesova, K.; Rossmann, P.; Hornova, M.; Novosadova, I.; Stehlikova, Z.; Kostovcik, M.; Hudcovic, T.; Stepankova, R.; et al. Intestinal Microbiota Promotes Psoriasis-Like Skin Inflammation by Enhancing Th17 Response. PLoS ONE 2016, 11, e0159539. [Google Scholar] [CrossRef]
- Stehlikova, Z.; Kostovcikova, K.; Kverka, M.; Rossmann, P.; Dvorak, J.; Novosadova, I.; Kostovcik, M.; Coufal, S.; Srutkova, D.; Prochazkova, P.; et al. Crucial Role of Microbiota in Experimental Psoriasis Revealed by a Gnotobiotic Mouse Model. Front. Microbiol. 2019, 10, 236. [Google Scholar] [CrossRef]
- Walecka, I.; Olszewska, M.; Rakowska, A.; Slowinska, M.; Sicinska, J.; Piekarczyk, E.; Kowalska-Oledzka, E.; Goralska, B.; Rudnicka, L. Improvement of psoriasis after antibiotic therapy with cefuroxime axetil. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 957–958. [Google Scholar] [CrossRef] [PubMed]
- Szanto, M.; Dozsa, A.; Antal, D.; Szabo, K.; Kemeny, L.; Bai, P. Targeting the gut-skin axis-Probiotics as new tools for skin disorder management? Exp. Dermatol. 2019, 28, 1210–1218. [Google Scholar] [CrossRef]
- Yin, G.; Li, J.F.; Sun, Y.F.; Ding, X.; Zeng, J.Q.; Zhang, T.; Peng, L.H.; Yang, Y.S.; Zhao, H. Fecal microbiota transplantation as a novel therapy for severe psoriasis. Zhonghua Nei Ke Za Zhi 2019, 58, 782–785. [Google Scholar] [CrossRef]
- Sacchetti, L.; Nardelli, C. Gut microbiome investigation in celiac disease: From methods to its pathogenetic role. Clin. Chem. Lab. Med. 2020, 58, 340–349. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; Group, P. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. BMJ 2009, 339, b2535. [Google Scholar] [CrossRef]
- Yeh, N.L.; Hsu, C.Y.; Tsai, T.F.; Chiu, H.Y. Gut Microbiome in Psoriasis is Perturbed Differently During Secukinumab and Ustekinumab Therapy and Associated with Response to Treatment. Clin. Drug Investig. 2019, 39, 1195–1203. [Google Scholar] [CrossRef]
- Shapiro, J.; Cohen, N.A.; Shalev, V.; Uzan, A.; Koren, O.; Maharshak, N. Psoriatic patients have a distinct structural and functional fecal microbiota compared with controls. J. Dermatol. 2019, 46, 595–603. [Google Scholar] [CrossRef]
- Huang, L.; Gao, R.; Yu, N.; Zhu, Y.; Ding, Y.; Qin, H. Dysbiosis of gut microbiota was closely associated with psoriasis. Sci. China Life Sci. 2019, 62, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Hidalgo-Cantabrana, C.; Gomez, J.; Delgado, S.; Requena-Lopez, S.; Queiro-Silva, R.; Margolles, A.; Coto, E.; Sanchez, B.; Coto-Segura, P. Gut microbiota dysbiosis in a cohort of patients with psoriasis. Br. J. Dermatol. 2019, 181, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.J.; Ho, H.J.; Tseng, C.H.; Lai, Z.L.; Shieh, J.J.; Wu, C.Y. Intestinal microbiota profiling and predicted metabolic dysregulation in psoriasis patients. Exp. Dermatol. 2018, 27, 1336–1343. [Google Scholar] [CrossRef] [PubMed]
- Codoner, F.M.; Ramirez-Bosca, A.; Climent, E.; Carrion-Gutierrez, M.; Guerrero, M.; Perez-Orquin, J.M.; Horga de la Parte, J.; Genoves, S.; Ramon, D.; Navarro-Lopez, V.; et al. Gut microbial composition in patients with psoriasis. Sci. Rep. 2018, 8, 3812. [Google Scholar] [CrossRef]
- Tan, L.; Zhao, S.; Zhu, W.; Wu, L.; Li, J.; Shen, M.; Lei, L.; Chen, X.; Peng, C. The Akkermansia muciniphila is a gut microbiota signature in psoriasis. Exp. Dermatol. 2018, 27, 144–149. [Google Scholar] [CrossRef]
- Eppinga, H.; Sperna Weiland, C.J.; Thio, H.B.; van der Woude, C.J.; Nijsten, T.E.; Peppelenbosch, M.P.; Konstantinov, S.R. Similar Depletion of Protective Faecalibacterium prausnitzii in Psoriasis and Inflammatory Bowel Disease, but not in Hidradenitis Suppurativa. J. Crohns Colitis 2016, 10, 1067–1075. [Google Scholar] [CrossRef]
- Masallat, D.; Moemen, D.; State, A.F. Gut bacterial microbiota in psoriasis: A case control study. Afr. J. Microbiol. Res. 2016, 10, 1337–1343. [Google Scholar]
- Scher, J.U.; Ubeda, C.; Artacho, A.; Attur, M.; Isaac, S.; Reddy, S.M.; Marmon, S.; Neimann, A.; Brusca, S.; Patel, T.; et al. Decreased bacterial diversity characterizes the altered gut microbiota in patients with psoriatic arthritis, resembling dysbiosis in inflammatory bowel disease. Arthritis Rheumatol. 2015, 67, 128–139. [Google Scholar] [CrossRef]
- Mirza, A.; Forbes, J.D.; Zhu, F.; Bernstein, C.N.; Van Domselaar, G.; Graham, M.; Waubant, E.; Tremlett, H. The multiple sclerosis gut microbiota: A systematic review. Mult. Scler. Relat. Disord. 2020, 37, 101427. [Google Scholar] [CrossRef]
- Chen, Z.; Qi, J.; Wei, Q.; Zheng, X.; Wu, X.; Li, X.; Liao, Z.; Lin, Z.; Gu, J. Variations in gut microbial profiles in ankylosing spondylitis: Disease phenotype-related dysbiosis. Ann. Transl. Med. 2019, 7, 571. [Google Scholar] [CrossRef]
- Pittayanon, R.; Lau, J.T.; Leontiadis, G.I.; Tse, F.; Yuan, Y.; Surette, M.; Moayyedi, P. Differences in Gut Microbiota in Patients With vs Without Inflammatory Bowel Diseases: A Systematic Review. Gastroenterology 2020, 158, 930–946.e931. [Google Scholar] [CrossRef]
- Scepanovic, P.; Hodel, F.; Mondot, S.; Partula, V.; Byrd, A.; Hammer, C.; Alanio, C.; Bergstedt, J.; Patin, E.; Touvier, M.; et al. A comprehensive assessment of demographic, environmental, and host genetic associations with gut microbiome diversity in healthy individuals. Microbiome 2019, 7, 130. [Google Scholar] [CrossRef] [PubMed]
- Wagner, B.D.; Grunwald, G.K.; Zerbe, G.O.; Mikulich-Gilbertson, S.K.; Robertson, C.E.; Zemanick, E.T.; Harris, J.K. On the Use of Diversity Measures in Longitudinal Sequencing Studies of Microbial Communities. Front. Microbiol. 2018, 9, 1037. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N.; Yamashita, T.; Hirata, K.I. Gut Microbiome and Cardiovascular Diseases. Diseases 2018, 6, 56. [Google Scholar] [CrossRef] [PubMed]
- Crovesy, L.; Masterson, D.; Rosado, E.L. Profile of the gut microbiota of adults with obesity: A systematic review. Eur. J. Clin. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Indias, I.; Sanchez-Alcoholado, L.; Garcia-Fuentes, E.; Cardona, F.; Queipo-Ortuno, M.I.; Tinahones, F.J. Insulin resistance is associated with specific gut microbiota in appendix samples from morbidly obese patients. Am. J. Transl. Res. 2016, 8, 5672–5684. [Google Scholar] [PubMed]
- Sobhonslidsuk, A.; Chanprasertyothin, S.; Pongrujikorn, T.; Kaewduang, P.; Promson, K.; Petraksa, S.; Ongphiphadhanakul, B. The Association of Gut Microbiota with Nonalcoholic Steatohepatitis in Thais. Biomed. Res. Int. 2018, 2018, 9340316. [Google Scholar] [CrossRef]
- Valguarnera, E.; Wardenburg, J.B. Good Gone Bad: One Toxin Away From Disease for Bacteroides fragilis. J. Mol. Biol. 2020, 432, 765–785. [Google Scholar] [CrossRef]
- Zhou, C.; Zhao, H.; Xiao, X.Y.; Chen, B.D.; Guo, R.J.; Wang, Q.; Chen, H.; Zhao, L.D.; Zhang, C.C.; Jiao, Y.H.; et al. Metagenomic profiling of the pro-inflammatory gut microbiota in ankylosing spondylitis. J. Autoimmun. 2019, 102360. [Google Scholar] [CrossRef]
- Guerreiro, C.S.; Calado, A.; Sousa, J.; Fonseca, J.E. Diet, Microbiota, and Gut Permeability-The Unknown Triad in Rheumatoid Arthritis. Front. Med. 2018, 5, 349. [Google Scholar] [CrossRef]
- De Filippis, F.; Pasolli, E.; Tett, A.; Tarallo, S.; Naccarati, A.; De Angelis, M.; Neviani, E.; Cocolin, L.; Gobbetti, M.; Segata, N.; et al. Distinct Genetic and Functional Traits of Human Intestinal Prevotella copri Strains Are Associated with Different Habitual Diets. Cell Host Microbe 2019, 25, 444–453.e443. [Google Scholar] [CrossRef] [PubMed]
- Tett, A.; Huang, K.D.; Asnicar, F.; Fehlner-Peach, H.; Pasolli, E.; Karcher, N.; Armanini, F.; Manghi, P.; Bonham, K.; Zolfo, M.; et al. The Prevotella copri Complex Comprises Four Distinct Clades Underrepresented in Westernized Populations. Cell Host Microbe 2019, 26, 666–679.e667. [Google Scholar] [CrossRef] [PubMed]
- Stefia, L.V.; Lee, J.; Patel, J.; de Sousa, S.R.; Legrand, J.; Rodero, M.; Burman, S.; Linedale, R.; Morrison, M.; Khosrotehrani, K. Secretome components from Faecalibacterium prausnitzii strains A2-165 and AHMP21 Modulate Cutaneous Wound Inflammation. J. Investig. Dermatol. 2020. [Google Scholar] [CrossRef]
- Toya, T.; Corban, M.T.; Marrietta, E.; Horwath, I.E.; Lerman, L.O.; Murray, J.A.; Lerman, A. Coronary artery disease is associated with an altered gut microbiome composition. PLoS ONE 2020, 15, e0227147. [Google Scholar] [CrossRef]
- Ufnal, M.; Pham, K. The gut-blood barrier permeability - A new marker in cardiovascular and metabolic diseases? Med. Hypotheses 2017, 98, 35–37. [Google Scholar] [CrossRef]
- Sikora, M.; Chrabaszcz, M.; Waskiel-Burnat, A.; Rakowska, A.; Olszewska, M.; Rudnicka, L. Claudin-3—A new intestinal integrity marker in patients with psoriasis: Association with disease severity. J. Eur. Acad. Dermatol. Venereol. 2019, 33, 1907–1912. [Google Scholar] [CrossRef]
- Sikora, M.; Stec, A.; Chrabaszcz, M.; Waskiel-Burnat, A.; Zaremba, M.; Olszewska, M.; Rudnicka, L. Intestinal Fatty Acid Binding Protein, a Biomarker of Intestinal Barrier, is Associated with Severity of Psoriasis. J. Clin. Med. 2019, 8, 1021. [Google Scholar] [CrossRef]
- Knox, N.C.; Forbes, J.D.; Peterson, C.L.; Van Domselaar, G.; Bernstein, C.N. The Gut Microbiome in Inflammatory Bowel Disease: Lessons Learned From Other Immune-Mediated Inflammatory Diseases. Am. J. Gastroenterol. 2019, 114, 1051–1070. [Google Scholar] [CrossRef]
- Panek, M.; Cipcic Paljetak, H.; Baresic, A.; Peric, M.; Matijasic, M.; Lojkic, I.; Vranesic Bender, D.; Krznaric, Z.; Verbanac, D. Methodology challenges in studying human gut microbiota—Effects of collection, storage, DNA extraction and next generation sequencing technologies. Sci. Rep. 2018, 8, 5143. [Google Scholar] [CrossRef]
- Mottawea, W.; Butcher, J.; Li, J.; Abujamel, T.; Manoogian, J.; Mack, D.; Stintzi, A. The mucosal-luminal interface: An ideal sample to study the mucosa-associated microbiota and the intestinal microbial biogeography. Pediatr. Res. 2019, 85, 895–903. [Google Scholar] [CrossRef]
- Castelino, M.; Eyre, S.; Moat, J.; Fox, G.; Martin, P.; Ho, P.; Upton, M.; Barton, A. Optimisation of methods for bacterial skin microbiome investigation: Primer selection and comparison of the 454 versus MiSeq platform. BMC Microbiol. 2017, 17, 23. [Google Scholar] [CrossRef]
| Study | Country | Case Number | Case Age (Mean ± SD) | Case Gender Men, (%) | Antipsoriatic Therapy | Control Number | Control Age (Mean ± SD) | Control Gender Men, (%) | NOS |
|---|---|---|---|---|---|---|---|---|---|
| Yeh et al., 2019 [19] | Taiwan | 34 | SEC: 51 ± 12 UST: 48.4 ± 12.7 | 25 (73.5%) | No treatment except for topical agents * | 12 | 48.4 ± 13.3 | 10 (83.3%) | 8 |
| Shapiro et al., 2019 [20] | Israel | 24 | 52.7 ± 11.6 | 16 (66.7%) | Topical treatment (22) Biologics (2) | 22 | 43.9 ± 12.7 | 16 (72.7%) | 8 |
| Huang et al., 2019 [21] | China | 35 | 52.1 ± 3.0 | 22 (62.9%) | NR | 27 | 52.9 ± 1.5 | 16 (59.3%) | 6 |
| Hidalgo-Cantabrana et al., 2019 [22] | Spain | 19 | 49 ± 11 | 12 (63.2%) | No treatment except for topical corticosteroids | 20 | 43 ± 11 | 5 (25%) | 6 |
| Chen et al., 2018 [23] | Taiwan | 32 | 42.8 ± 12.6 | 25 (78.1%) | Phototherapy (8) DMARDs/ Biologics (20) | 64 | 44.2±10.8 | 50 (78.1%) | 7 |
| Codoner et al., 2018 [24] | Spain | 52 | 41.2 ± 14.4 | 25 (48.1%) | NR | NR | NR | NR | 4 |
| Tan et al., 2018 [25] | China | 14 | 47.5 ± 4.7 | 10 (71.4%) | NR | 14 | 40.4 ± 2.5 | 8 (57.1%) | 7 |
| Eppinga et al., 2016 [26] | Netherlands | 29 | 46 ± 14.0 | 12 (41.4%) | No treatment (27) DMARDs (2) | 33 | 41 ± 14.9 | 10 (30.3%) | 6 |
| Massallat et al., 2016 [27] | Egypt | 45 | 42.3 ± 10 | 18 (40%) | NR | 45 | 44.2 ± 7.1 | 20 (44.4%) | 8 |
| Scher et al., 2015 [28] | US | 15 | 39.4 | 7 (46.7%) | No systemic treatment | 17 | 42.2 | 6 (35.3%) | 6 |
| Study | Sample | Sample Transport | DNA Extraction | Microbiota Analysis Technique | Sequencing Target | Sequencing Platform | Data Analysis Platform | Reference Sequences Database |
|---|---|---|---|---|---|---|---|---|
| Yeh et al., 2019 [19] | stool | DNA stabilizer, Immediate freezing, Transport on ice | QIAamp DNA Stool Mini Kit | 16S rRNA gene sequencing | V3–V4 | Ilumnia MiSeq platform | QIIME | Kyoto Encyclopedia of Genes and Genomies database |
| Shapiro et al., 2019 [20] | stool | Immediate freezing | PowerSoil HTP 96 Kit | 16S rRNA gene sequencing | V4 | Ilumnia MiSeq platform | QIIME | Greengenes database |
| Huang et al., 2019 [21] | stool | Transport on ice | PowerSoil HTP 96 Kit | 16S rRNA gene sequencing | V4–V5 | Ilumnia MiSeq platform | UPARSE | National Center for Biotechnology Information Sequence Read Archive database |
| Hidalgo-Cantabrana et al., 2019 [22] | stool | Immediate freezing | QIAamp DNA Stool Mini Kit | 16S rRNA gene sequencing | V2–V3 | Ion Gene Studio S5 sequencer | QIIME | SILVA database |
| Chen et al., 2018 [23] | stool | Transport on ice | QIAamp DNA Stool Mini Kit | 16S rRNA gene sequencing | V3–V4 | Ilumnia MiSeq platform | UPARSE | Greengenes database |
| Codoner et al., 2018 [24] | stool | Immediate freezing | QIAamp DNA Stool Mini Kit | 16S rRNA gene sequencing | V3–V4 | Ilumnia MiSeq platform | QIIME | National Center for Biotechnology Information |
| Tan et al., 2018 [25] | stool | Immediate freezing | E.Z.N.A. stool DNA Kit | 16S rRNA gene sequencing | V4 | Ilumnia MiSeq platform | USEARCH | Ribosomal Database Project (RDP) |
| Eppinga et al., 2016 [26] | stool | Ambient temperature | NR | real-time quantitative PCR | - | quantitative PCR | - | - |
| Massallat et al., 2016 [27] | stool | NR | QIAamp DNA Stool Mini Kit | real-time quantitative PCR | - | quantitative PCR | - | - |
| Scher et al., 2015 [28] | stool | NR | NR | 16S rRNA gene pyro-sequencing | V1–V2 | 454 GS FLX Titanium platform | Mothur | SILVA database |
| Study | Indices of α-Diversity | Gut Microbiota α-Diversity in Psoriasis | Indices of β-Diversity | Gut Microbiota β-Diversity in Psoriasis |
|---|---|---|---|---|
| Yeh et al. [19] | Shannon index, Simpson index | No differences were observed | UniFrac analysis (weighted and unweighted analyses), Bray Curtis index | Significant difference |
| Shapiro et al. [20] | Shannon index, rarefaction curves | No differences were observed | UniFrac analysis (weighted and unweighted analyses) | Significant difference |
| Huang et al. [21] | Shannon index, Simpson index, ACE index, Chao1 index | Shannon and Simpson indexes—no differences ACE and Chao indexes—decreased in psoriasis | PCA based on the Bray-Curtis dissimilarity distance | Significant difference |
| Hidalgo-Cantabrana et al. [22] | Shannon index, Chao1 index, Faith’s phylogenetic diversity index | Lower diversity in psoriasis | unweighted Unifrac analysis | Significant difference |
| Chen et al. [23] | Shannon index, Simpson index, Chao1 index, number of observed OTUs | No differences were observed | UniFrac analysis (weighted and unweighted analyses), Bray Curtis index | Significant difference (psoriasis patients with BMI < 25) |
| Codoner et al. [24] | Shannon index | Greater diversity in psoriasis | PCA | Significant difference |
| Tan et al. [25] | Shannon index, Simpson index, ACE index, Chao1 index | No differences were observed | PCA, UPGMA | Significant difference |
| Scher et al. [28] | Shannon index, Faith’s phylogenetic diversity index | Lower diversity in psoriasis | unweighted Unifrac analysis | Significant difference |
| Phylum | Class | Order | Family | Genus | Species |
|---|---|---|---|---|---|
| Bacteroidetes ↑ [21] ↓ [20,22,23] | Bacteroidia | Bacteroidales | Bacteroidaceae ↑ [25] ↓ [22,23] | Bacteroides ↑ [25] ↓ [22,24] | |
| Prevotellaceae ↓ [22,23] | Prevotella | Prevotella copri ↓ [20] | |||
| Paraprevotella ↓ [20,22] | |||||
| Barnesiellaceae ↓ [22] | Barnesiella ↓ [22] | ||||
| Tannerellaceae ↓ [22] | |||||
| Rikenellaceae ↓ [22] | Alistipes ↓ [22] | ||||
| Tannerellaceae | Parabacteroides ↑ [21] ↓ [22,28] | ||||
| Porphyromonadaceae ↓ [28] | |||||
| S24-7 ↓ [25] | |||||
| Marinilabiliales | Marinifilaceae ↓ [22] | ||||
| Firmicutes ↑ [20,22,23] ↓ [21] | Clostridia | Clostridiales | Ruminococcaceae ↑ [22,23] | Faecalibacterium ↑ [20,24] ↓ [22] | Faecalibacterium prausnitzii ↓ [26] |
| Ruminococcus ↑ [20,22] | Ruminococcus gnavus ↑ [20] | ||||
| Subdoligranulum ↑ [22] | |||||
| Lachnospiraceae ↑ [22,23] | Lachnospira ↑ [21] ↓ [20] | ||||
| Blautia ↑ [20,22] | |||||
| Coprococcus ↑ [20] | |||||
| Dorea ↑ [20] | Dorea formicigenerans ↑ [20] | ||||
| Clostridiales Family XIII ↑ [22] | |||||
| Peptostreptococcaceae ↑ [22] | |||||
| Christensenellaceae | Christensenella ↑ [20] | ||||
| Clostridiaceae | Clostridium | Clostridium citroniae ↑ [25] | |||
| Erysipelotrichia | Erysipelotrichales ↓ [28] | Erysipelotrichaceae ↑ [22] ↓ [28] | Allobaculum ↓ [22] | ||
| Coprobacillus ↓ [28] | |||||
| Bacilli | Lactobacillales | Lactobacillaceae ↓ [22] | |||
| Streptococcaceae ↓ [22] | Streptococcus ↑ [21] | ||||
| Lactococcus ↑ [21] | |||||
| Carnobacteriaceae | Carnobacterium ↓ [21] | ||||
| Granulicatella ↓ [21] | |||||
| Enterococcaceae ↑ [25] | Enterococcus ↑ [25] | ||||
| Bacillales | Bacillaceae | Bacillus ↑ [21] | |||
| Negativicutes | Veillonellales | Veillonellaceae ↑ [25] ↓ [22] | |||
| Verrucomicrobia ↓ [25] | Verrucomicrobiae ↓ [25] | Verrucomicrobiales ↓ [25] | Akkermansiaceae | Akkermansia ↑ [24] ↓ [25] | Akkermiansia muciniphila ↓ [25] |
| Verrucomicrobiaceae↓ [25] | |||||
| Proteobacteria ↓ [20,22] | Gammaproteobacteria | Enterobacterales | Enterobacteriaceae | Escherichia | Escherichia coli ↑ [26] |
| Pasteurellales | Pasteurellaceae ↓ [22] | ||||
| Betaproteobacteria | Burkholderiales | Burkholderiaceae ↓ [22] | |||
| Sutterellaceae | Sutterella ↑ [21] ↓ [20] | ||||
| Deltaproteobacteria | Desulfovibrionales | Desulfovibrionaceae ↓ [22] | |||
| Actinobacteria ↑ [20,22] ↓ [27,28] | Actinobacteria ↓ [28] | Bifidobacteriales ↓ [28] | Bifidobacteriaceae ↑ [22] ↓ [28] | Bifidobacterium ↑ [20,22] ↓ [28] | |
| Micrococcales | Micrococcaceae | Rothia ↓ [21] | |||
| Coriobacteriia | Coriobacteriales | Coriobacteriaceae ↑ [22] | Collinsella ↑ [20,22] | Collinsella aerofaciens ↑ [20] | |
| Eggerthellales | Eggerthellaceae ↑ [22] | Slackia ↑ [22] | |||
| Gordonibacter ↓ [21] | |||||
| Lentisphaerae | Lentisphaeria | Victivallales | Victivallaceae ↓ [22] | ||
| Deinococcus–Thermus | Deinococci | Thermales | Thermaceae | Thermus ↓ [21] | |
| Tenericutes ↓ [25] | Mollicutes ↓ [25] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sikora, M.; Stec, A.; Chrabaszcz, M.; Knot, A.; Waskiel-Burnat, A.; Rakowska, A.; Olszewska, M.; Rudnicka, L. Gut Microbiome in Psoriasis: An Updated Review. Pathogens 2020, 9, 463. https://doi.org/10.3390/pathogens9060463
Sikora M, Stec A, Chrabaszcz M, Knot A, Waskiel-Burnat A, Rakowska A, Olszewska M, Rudnicka L. Gut Microbiome in Psoriasis: An Updated Review. Pathogens. 2020; 9(6):463. https://doi.org/10.3390/pathogens9060463
Chicago/Turabian StyleSikora, Mariusz, Albert Stec, Magdalena Chrabaszcz, Aleksandra Knot, Anna Waskiel-Burnat, Adriana Rakowska, Malgorzata Olszewska, and Lidia Rudnicka. 2020. "Gut Microbiome in Psoriasis: An Updated Review" Pathogens 9, no. 6: 463. https://doi.org/10.3390/pathogens9060463
APA StyleSikora, M., Stec, A., Chrabaszcz, M., Knot, A., Waskiel-Burnat, A., Rakowska, A., Olszewska, M., & Rudnicka, L. (2020). Gut Microbiome in Psoriasis: An Updated Review. Pathogens, 9(6), 463. https://doi.org/10.3390/pathogens9060463






