Comparative Pathology of West Nile Virus in Humans and Non-Human Animals
Abstract
1. Introduction
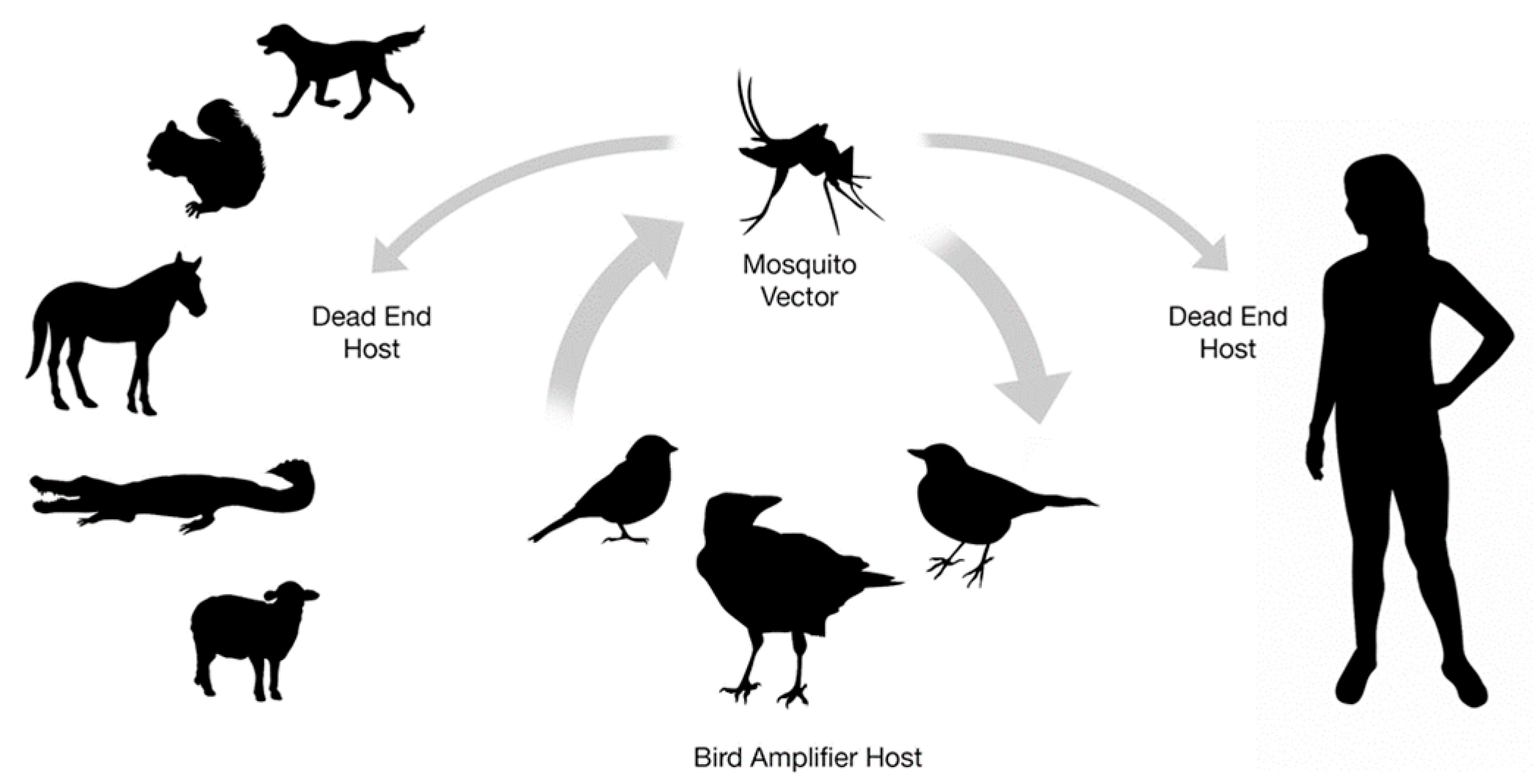
2. The Virus and Its Ecology
3. Natural WNV Disease
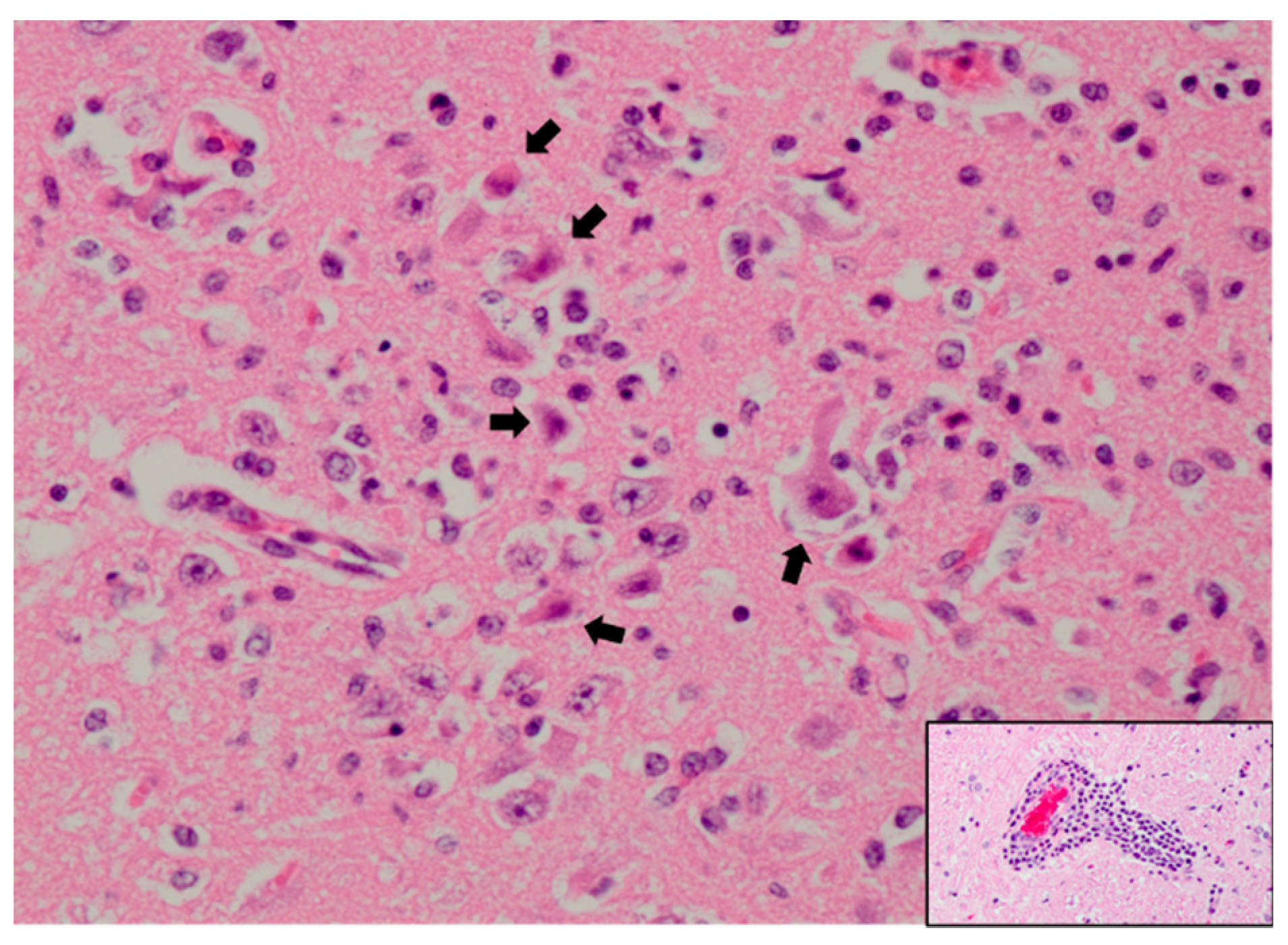
3.1. Humans
3.2. Birds
3.3. Horses
3.4. Additional Affected Vertebrate Species
4. Biomedical Models of WNV Infection
4.1. Mice
4.2. Hamsters
4.3. Non-Human Primates
4.4. Additional Animal Species Used in WNV Biomedical Research
5. Summary and Future Directions
Funding
Acknowledgments
Conflicts of Interest
References
- Curren, E.J.; Lehman, J.; Kolsin, J.; Walker, W.L.; Martin, S.W.; Staples, J.E.; Hills, S.L.; Gould, C.V.; Rabe, I.B.; Fischer, M.; et al. West nile virus and other nationally notifiable arboviral diseases—United States, 2017. Morb. Mortal. Wkly. Rep. 2018, 67, 1137–1142. [Google Scholar] [CrossRef]
- Smithburn, K.C.; Hughes, T.P.; Burke, A.W.; Paul, J.H. A Neurotropic Virus Isolated from the Blood of a Native of Uganda. Am. J. Trop. Med. Hyg. 1940, 1, 471–492. [Google Scholar] [CrossRef]
- Melnick, J.L.; Paul, J.R.; Riordan, J.T.; Barnett, V.H.; Goldblum, N.; Zabin, E. Isolation from Human Sera in Egypt of a Virus Apparently Identical to West Nile Virus. Exp. Biol. Med. 1951, 77, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Hurlbut, H.S.; Rizk, F.; Taylor, R.M.; Work, T.H. A study of the ecology of West Nile virus in Egypt. Am. J. Trop. Med. Hyg. 1956, 5, 579–620. [Google Scholar] [PubMed]
- Perelman, A.; Stern, J. Acute pancreatitis in West Nile fever. Am. J. Trop. Med. Hyg. 1974, 23, 1150–1152. [Google Scholar] [CrossRef] [PubMed]
- Lvov, D.K.; Butenko, A.M.; Gromashevsky, V.L.; Larichev, V.P.; Gaidamovich, S.Y.; Vyshemirsky, O.I.; Zhukov, A.N.; Lazorenko, V.V.; Salko, V.N.; Kovtunov, A.I.; et al. Isolation of two strains of West Nile virus during an outbreak in southern Russia, 1999. Emerg. Infect. Dis. 2000, 6, 373–376. [Google Scholar] [CrossRef]
- Siegel-Itzkovich, J. Twelve die of West Nile virus in Israel. Br. Med. J. 2000, 321, 724. [Google Scholar]
- Rossi, S.L.; Ross, T.M.; Evans, J.D. West Nile virus. Clin. Lab. Med. 2010, 30, 47–65. [Google Scholar] [CrossRef]
- Weaver, S.C.; Barrett, A.D.T. Transmission cycles, host range, evolution and emergence of arboviral disease. Nat. Rev. Microbiol. 2004, 2, 789–801. [Google Scholar] [CrossRef]
- O’Leary, D.R.; Marfin, A.A.; Montgomery, S.P.; Kipp, A.M.; Lehman, J.A.; Biggerstaff, B.J.; Elko, V.L.; Collins, P.D.; Jones, J.E.; Campbell, G.L. The epidemic of West Nile virus in the United States, 2002. Vector-Borne Zoonotic Dis. 2004, 4, 61–70. [Google Scholar] [CrossRef]
- Outbreak of West Nile–like viral encephalitis—New York, 1999. Morb. Mortal. Wkly. Rep. 1999, 48, 845–849.
- Hubálek, Z.; Halouzka, J. West Nile fever—A reemerging mosquito–borne viral disease in Europe. Emerg. Infect. Dis. 1999, 5, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Lanciotti, R.S.; Roehrig, J.T.; Deubel, V.; Smith, J.; Parker, M.; Steele, K.; Crise, B.; Volpe, K.E.; Crabtree, M.B.; Scherret, J.H.; et al. Origin of the West Nile virus responsible for an outbreak of encephalitis in the Northeastern United States. Science 1999, 286, 2333–2337. [Google Scholar] [CrossRef] [PubMed]
- Giladi, M.; Metzkor-Cotter, E.; Martin, D.A.; Siegman-Igra, Y.; Korczyn, A.D.; Rosso, R.; Berger, S.A.; Campbell, G.L.; Lanciotti, R.S. West Nile Encephalitis in Israel, 1999: The New York Connection. Emerg. Infect. Dis. 2001, 7, 659–661. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.F.; Andreadis, T.G.; Vossbrinck, C.R.; Tirrell, S.; Wakem, E.M.; French, R.A.; Garmendia, A.E.; Van Kruiningen, H.J. Isolation of West Nile virus from mosquitoes, crows, and a Cooper’s hawk in Connecticut. Science 1999, 286, 2331–2333. [Google Scholar] [CrossRef] [PubMed]
- Bakonyi, T.; Hubálek, Z.; Rudolf, I.; Nowotny, N. Novel flavivirus or new lineage of West Nile virus, central Europe. Emerg. Infect. Dis. 2005, 11, 225. [Google Scholar] [CrossRef]
- May, F.J.; Davis, C.T.; Tesh, R.B.; Barrett, A.D.T. Phylogeography of West Nile Virus: From the Cradle of Evolution in Africa to Eurasia, Australia, and the Americas. J. Virol. 2011, 85, 2964–2974. [Google Scholar] [CrossRef]
- Bondre, V.P.; Jadi, R.S.; Mishra, A.C.; Yergolkar, P.N.; Correspondence, V.A.A. West Nile virus isolates from India: Evidence for a distinct genetic lineage. J. Gen. Virol. 2007, 88, 875–884. [Google Scholar] [CrossRef]
- Vázquez, A.; Sánchez-Seco, M.; Ruiz, S.; Molero, F.; Hernandez, L.; Moreno, J.; Magallanes, A.; Tejedor, C.G.; Tenorio, A. Putative new lineage of West Nile virus, Spain. Emerg. Infect. Dis. 2010, 16, 549–552. [Google Scholar] [CrossRef]
- King, A.; Lefkowitz, E.; Adams, M.; Carstens, E. Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses; Elsevier: New York, NY, USA, 2011. [Google Scholar]
- Charrel, R.N.; Brault, A.C.; Gallian, P.; Lemasson, J.J.; Murgue, B.; Murri, S.; Pastorino, B.; Zeller, H.; De Chesse, R.; De Micco, P.; et al. Evolutionary relationship between Old World West Nile virus strains: Evidence for viral gene flow between Africa, the Middle East, and Europe. Virology 2003, 315, 381–388. [Google Scholar] [CrossRef]
- Lanciotti, R.S.; Ebel, G.D.; Deubel, V.; Kerst, A.J.; Murri, S.; Meyer, R.; Bowen, M.; McKinney, N.; Morrill, W.E.; Crabtree, M.B.; et al. Complete genome sequences and phylogenetic analysis of West Nile virus strains isolated from the United States, Europe, and the Middle East. Virology 2002, 298, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Bakonyi, T.; Ivanics, E.; Erdélyi, K.; Ursu, K.; Ferenczi, E.; Weissenböck, H.; Nowotny, N. Lineage 1 and 2 strains of encephalitic West Nile virus, central Europe. Emerg. Infect. Dis. 2006, 12, 618–623. [Google Scholar] [CrossRef] [PubMed]
- Papa, A.; Xanthopoulou, K.; Gewehr, S.; Mourelatos, S. Detection of West Nile virus lineage 2 in mosquitoes during a human outbreak in Greece. Clin. Microbiol. Infect. 2011, 17, 1176–1180. [Google Scholar] [CrossRef] [PubMed]
- Valiakos, G.; Touloudi, A.; Iacovakis, C.; Athanasiou, L.; Birtsas, P.; Spyrou, V.; Billinis, C. Molecular detection and phylogenetic analysis of West Nile virus lineage 2 in sedentary wild birds (Eurasian magpie), Greece, 2010. Euro Surveill. 2011, 16, 19862. [Google Scholar]
- Shahhosseini, N.; Chinikar, S.; Moosa-Kazemi, S.H.; Sedaghat, M.M.; Kayedi, M.H.; Lühken, R.; Schmidt-Chanasit, J. West Nile Virus lineage-2 in Culex specimens from Iran. Trop. Med. Int. Heal. 2017, 22, 1343–1349. [Google Scholar] [CrossRef]
- Cotar, A.I.; Fǎlcuţǎ, E.; Dinu, S.; Necula, A.; Bîrluţiu, V.; Ceianu, C.S.; Prioteasa, F.L. West Nile virus lineage 2 in Romania, 2015–2016: Co-circulation and strain replacement. Parasites Vectors 2018, 11, 562. [Google Scholar] [CrossRef]
- Kolodziejek, J.; Seidel, B.; Jungbauer, C.; Dimmel, K.; Kolodziejek, M.; Rudolf, I.; Hubálek, Z.; Allerberger, F.; Nowotny, N. West Nile Virus Positive Blood Donation and Subsequent Entomological Investigation, Austria, 2014. PLoS ONE 2015, 10, e0126381. [Google Scholar] [CrossRef]
- Wodak, E.; Richter, S.; Bagó, Z.; Revilla-Fernández, S.; Weissenböck, H.; Nowotny, N.; Winter, P. Detection and molecular analysis of West Nile virus infections in birds of prey in the eastern part of Austria in 2008 and 2009. Vet. Microbiol. 2011, 149, 358–366. [Google Scholar] [CrossRef]
- Busquets, N.; Laranjo-González, M.; Soler, M.; Nicolás, O.; Rivas, R.; Talavera, S.; Villalba, R.; San Miguel, E.; Torner, N.; Aranda, C.; et al. Detection of West Nile virus lineage 2 in North-Eastern Spain (Catalonia). Transbound. Emerg. Dis. 2019, 66, 617–621. [Google Scholar] [CrossRef]
- Vilibic-Cavlek, T.; Savic, V.; Petrovic, T.; Toplak, I.; Barbic, L.; Petric, D.; Tabain, I.; Hrnjakovic-Cvjetkovic, I.; Bogdanic, M.; Klobucar, A.; et al. Emerging Trends in the Epidemiology of West Nile and Usutu Virus Infections in Southern Europe. Front. Vet. Sci. 2019, 6, 437. [Google Scholar] [CrossRef]
- Kramer, L.D.; Styer, L.M.; Ebel, G.D. A Global Perspective on the Epidemiology of West Nile Virus. Annu. Rev. Entomol. 2008, 53, 61–81. [Google Scholar] [CrossRef] [PubMed]
- Godsey, M.S.; King, R.J.; Burkhalter, K.; Delorey, M.; Colton, L.; Charnetzky, D.; Sutherland, G.; Ezenwa, V.O.; Wilson, L.A.; Coffey, M.; et al. Ecology of potential West Nile virus vectors in Southeastern Louisiana: Enzootic transmission in the relative absence of Culex quinquefasciatus. Am. J. Trop. Med. Hyg. 2013, 88, 986–996. [Google Scholar] [CrossRef] [PubMed]
- Bolling, B.G.; Moore, C.G.; Anderson, S.L.; Blair, C.D.; Beaty, B.J. Entomological Studies along the Colorado Front Range during a Period of Intense West Nile virus activity. J. Am. Mosq. Control Assoc. 2007, 23, 37–46. [Google Scholar] [CrossRef]
- Hamer, G.L.; Chaves, L.F.; Anderson, T.K.; Kitron, U.D.; Brawn, J.D.; Ruiz, M.O.; Loss, S.R.; Walker, E.D.; Goldberg, T.L. Fine-scale variation in vector host use and force of infection drive localized patterns of West Nile virus transmission. PLoS ONE 2011, 6, e23767. [Google Scholar] [CrossRef]
- Kent, R.; Juliusson, L.; Weissmann, M.; Evans, S.; Komar, N. Seasonal Blood–Feeding Behavior of Culex tarsalis (Diptera: Culicidae) in Weld County, Colorado, 2007. J. Med. Entomol. 2009, 46, 380–390. [Google Scholar] [CrossRef] [PubMed]
- Kilpatrick, A.M.; Daszak, P.; Jones, M.J.; Marra, P.P.; Kramer, L.D.; Marm Kilpatrick, A.; Daszak, P.; Jones, M.J.; Marra, P.P.; Kramer, L.D. Host heterogeneity dominates West Nile virus transmission. Proc. R. Soc. B Biol. Sci. 2006, 273, 2327–2333. [Google Scholar] [CrossRef]
- Kilpatrick, A.M.; Kramer, L.D.; Jones, M.J.; Marra, P.P.; Daszak, P. West Nile virus epidemics in North America are driven by shifts in mosquito feeding behavior. PLoS Biol. 2006, 4, 606–610. [Google Scholar] [CrossRef]
- Dawson, J.R.; Stone, W.B.; Ebel, G.D.; Young, D.S.; Galinski, D.S.; Pensabene, J.P.; Franke, M.A.; Eidson, M.; Kramer, L.D. Crow deaths caused by West Nile virus during winter. Emerg. Infect. Dis. 2007, 13, 1912–1914. [Google Scholar] [CrossRef]
- Hinton, M.G.; Reisen, W.K.; Wheeler, S.S.; Townsend, A.K. West Nile Virus Activity in a Winter Roost of American Crows (Corvus brachyrhynchos): Is Bird-To-Bird Transmission Important in Persistence and Amplification? J. Med. Entomol. 2015, 52, 683–692. [Google Scholar] [CrossRef]
- Ip, H.S.; Van Wettere, A.J.; McFarlane, L.; Shearn-Bochsler, V.; Dickson, S.L.; Baker, J.; Hatch, G.; Cavender, K.; Long, R.; Bodenstein, B. West Nile Virus Transmission in Winter: The 2013 Great Salt Lake Bald Eagle and Eared Grebes Mortality Event. PLoS Curr. 2014, 6. [Google Scholar] [CrossRef]
- Banet-Noach, C.; Simanov, L.; Malkinson, M. Direct (non-vector) transmission of West Nile virus in geese. Avian Pathol. 2003, 32, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, N.; Young, G.; Ndaluka, C.; Bielefeldt-Ohmann, H.; Komar, N.; Bowen, R. Persistent West Nile virus infection in the house sparrow (Passer domesticus). Arch. Virol. 2009, 154, 783–789. [Google Scholar] [CrossRef]
- Nasci, R.S.; Savage, H.M.; White, D.J.; Miller, J.R.; Cropp, B.C.; Godsey, M.S.; Kerst, A.J.; Bennett, P.; Gottfried, K.; Lanciotti, R.S. West Nile virus in overwintering culex mosquitoes, New York City, 2000. Emerg. Infect. Dis. 2001, 7, 742–744. [Google Scholar] [CrossRef] [PubMed]
- Nelms, B.M.; Fechter-Leggett, E.; Carroll, B.D.; Macedo, P.; Kluh, S.; Reisen, W.K. Experimental and natural vertical transmission of West Nile virus by California Culex (Diptera: Culicidae) mosquitoes. J. Med. Entomol. 2013, 50, 371–378. [Google Scholar] [CrossRef] [PubMed]
- Laboratory-acquired West Nile virus infections—United States, 2002. Morb. Mortal. Wkly. Rep. 2002, 51, 1133–1135.
- Centers for Disease Control and Prevention (CDC). West Nile virus infection among turkey breeder farm workers—Wisconsin, 2002. MMWR. Morb. Mortal. Wkly. Rep. 2003, 52, 1017–1019. [Google Scholar]
- Centers for Disease Control and Prevention (CDC) Intrauterine West Nile virus infection—New York, 2002. Morb. Mortal. Wkly. Rep. 2002, 51, 1135–1136.
- Hinckley, A.F.; O’Leary, D.R.; Hayes, E.B. Transmission of West Nile virus through human breast milk seems to be rare. Pediatrics 2007, 119, e666–e671. [Google Scholar] [CrossRef]
- Dodd, R.Y.; Foster, G.A.; Stramer, S.L. Keeping Blood Transfusion Safe From West Nile Virus: American Red Cross Experience, 2003 to 2012. Transfus. Med. Rev. 2015, 29, 153–161. [Google Scholar] [CrossRef]
- Anesi, J.A.; Silveira, F.P. AST Infectious Diseases Community of Practice Arenaviruses and West Nile Virus in solid organ transplant recipients: Guidelines from the American Society of Transplantation Infectious Diseases Community of Practice. Clin. Transplant. 2019, 33, e13576. [Google Scholar] [CrossRef]
- Morales, M.A.; Barrandeguy, M.; Fabbri, C.; Garcia, J.B.; Vissani, A.; Trono, K.; Gutierrez, G.; Pigretti, S.; Menchaca, H.; Garrido, N.; et al. West Nile virus isolation from equines in Argentina, 2006. Emerg. Infect. Dis. 2006, 12, 1559–1561. [Google Scholar] [CrossRef]
- Blitvich, B.J.; Fernandez-Salas, I.; Contreras-Cordero, J.F.; Marlenee, N.L.; Gonzalez-Rojas, J.I.; Komar, N.; Gubler, D.J.; Calisher, C.H.; Beaty, B.J. Serologic evidence of West Nile virus infection in horses, Coahuila State, Mexico. Emerg. Infect. Dis. 2003, 9, 853–856. [Google Scholar] [CrossRef]
- Komar, N.; Clark, G.G. West Nile virus activity in Latin America and the Caribbean. Rev. Panam. Salud Publica/Pan Am. J. Public Health 2006, 19, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.S.G.; Matos, A.C.D.; da Cunha, M.A.C.R.; Rehfeld, I.S.; Galinari, G.C.F.; Marcelino, S.A.C.; Saraiva, L.H.G.; Martins, N.R.; Maranhão, R.D.; Lobato, Z.I.P.; et al. West Nile virus associated with equid encephalitis in Brazil, 2018. Transbound. Emerg. Dis. 2019, 66, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Yamshchikov, G.; Borisevich, V.; Kwok, C.W.; Nistler, R.; Kohlmeier, J.; Seregin, A.; Chaporgina, E.; Benedict, S.; Yamshchikov, V. The suitability of yellow fever and Japanese encephalitis vaccines for immunization against West Nile virus. Vaccine 2005, 23, 4785–4792. [Google Scholar] [CrossRef] [PubMed]
- Swaddle, J.P.; Calos, S.E. Increased avian diversity is associated with lower incidence of human West Nile infection: Observation of the dilution effect. PLoS ONE 2008, 3, e2488. [Google Scholar] [CrossRef]
- Grubaugh, N.D.; Smith, D.R.; Brackney, D.E.; Bosco-Lauth, A.M.; Fauver, J.R.; Campbell, C.L.; Felix, T.A.; Romo, H.; Duggal, N.K.; Dietrich, E.A.; et al. Experimental Evolution of an RNA Virus in Wild Birds: Evidence for Host-Dependent Impacts on Population Structure and Competitive Fitness. PLoS Pathog. 2015, 11, e1004874. [Google Scholar] [CrossRef]
- Brault, A.C.; Huang, C.Y.-H.; Langevin, S.A.; Kinney, R.M.; Bowen, R.A.; Ramey, W.N.; Panella, N.A.; Holmes, E.C.; Powers, A.M.; Miller, B.R. A single positively selected West Nile viral mutation confers increased virogenesis in American crows. Nat. Genet. 2007, 39, 1162. [Google Scholar] [CrossRef]
- VanDalen, K.K.; Hall, J.S.; Clark, L.; McLean, R.G.; Smeraski, C. West Nile virus infection in American Robins: New insights on dose response. PLoS ONE 2013, 8, e68537. [Google Scholar] [CrossRef]
- Klenk, K.; Snow, J.; Morgan, K.; Bowen, R.; Stephens, M.; Foster, F.; Gordy, P.; Beckett, S.; Komar, N.; Gubler, D.; et al. Alligators as West Nile Virus Amplifiers. Emerg. Infect. Dis. 2004, 10, 2150–2155. [Google Scholar] [CrossRef]
- Platt, K.B.; Tucker, B.J.; Halbur, P.G.; Blitvich, B.J.; Fabiosa, F.G.; Mullin, K.; Parikh, G.R.; Kitikoon, P.; Bartholomay, L.C.; Rowley, W.A. Fox squirrels (Sciurus niger) develop West Nile virus viremias sufficient for infecting select mosquito species. Vector Borne Zoonotic Dis. 2008, 8, 225–233. [Google Scholar] [CrossRef] [PubMed]
- Tiawsirisup, S.; Platt, K.B.; Tucker, B.J.; Rowley, W.A. Eastern cottontail rabbits (Sylvilagus floridanus) develop West Nile virus viremias sufficient for infecting select mosquito species. Vector-Borne Zoonotic Dis. 2005, 5, 342–350. [Google Scholar] [CrossRef]
- Saxena, V.; Bolling, B.G.; Wang, T. Emerging Pathogens. In Clinics in Laboratory Medicine; Ismail, N., Snyder, J., William Pasculle, A., Eds.; Saunders: Philadelphia, PA, USA, 2017. [Google Scholar]
- Sejvar, J.J. Clinical manifestations and outcomes of West Nile virus infection. Viruses 2014, 6, 606–623. [Google Scholar] [CrossRef] [PubMed]
- Sejvar, J.J.; Haddad, M.B.; Tierney, B.C.; Campbell, G.L.; Marfin, A.A.; Van Gerpen, J.A.; Fleischauer, A.; Leis, A.A.; Stokic, D.S.; Petersen, L.R. Neurologic Manifestations and Outcome of West Nile Virus Infection. J. Am. Med. Assoc. 2003, 290, 511–515. [Google Scholar] [CrossRef]
- Agamanolis, D.P.; Leslie, M.J.; Caveny, E.A.; Guarner, J.; Shieh, W.J.; Zaki, S.R. Neuropathological findings in West Nile virus encephalitis: A case report. Ann. Neurol. 2003, 54, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Doron, S.I.; Dashe, J.F.; Adelman, L.S.; Brown, W.F.; Werner, B.G.; Hadley, S. Histopathologically proven poliomyelitis with quadriplegia and loss of brainstem function due to West Nile virus infection. Clin. Infect. Dis. 2003, 37, e74–e77. [Google Scholar] [CrossRef]
- Fan, E.; Needham, D.M.; Brunton, J.; Kern, R.Z.; Stewart, T.E. West Nile virus infection in the intensive care unit: A case series and literature review. Can. Respir. J. 2004, 11, 354–358. [Google Scholar] [CrossRef]
- Kelley, T.W.; Prayson, R.A.; Ruiz, A.I.; Isada, C.M.; Gordon, S.M.; Kelley, T.W.; Ruiz, A.I.; Prayson, R.A.; Gordon, S.M.; Isada, C.M. The Neuropathology of West Nile Virus Meningoencephalitis: A Report of Two Cases and Review of the Literature. Am. J. Clin. Pathol. 2003, 119, 749–753. [Google Scholar] [CrossRef]
- Bouffard, J.P.; Riudavets, M.A.; Holman, R.; Rushing, E.J. Neuropathology of the brain and spinal cord in human West Nile virus infection. Clin. Neuropathol. 2004, 23, 59–61. [Google Scholar]
- Shieh, W.J.; Guarner, J.; Layton, M.; Fine, A.; Miller, J.; Nash, D.; Campbell, G.L.; Roehrig, J.T.; Gubler, D.J.; Zaki, S.R. The role of pathology in an investigation of an outbreak of West Nile encephalitis in New York, 1999. Emerg. Infect. Dis. 2000, 6, 370–372. [Google Scholar] [CrossRef]
- Schafernak, K.T.; Bigio, E.H. West Nile virus encephalomyelitis with polio-like paralysis & nigral degeneration. Can. J. Neurol. Sci. 2006, 33, 407–410. [Google Scholar]
- Fratkin, J.D.; Leis, A.A.; Stokic, D.S.; Slavinski, S.A.; Geiss, R.W. Spinal Cord Neuropathology in Human West Nile Virus Infection. Arch. Pathol. Lab. Med. 2004, 128, 533–537. [Google Scholar]
- Guarner, J.; Shieh, W.-J.; Hunter, S.; Paddock, C.D.; Morken, T.; Campbell, G.L.; Marfin, A.A.; Zaki, S.R. Clinicopathologic study and laboratory diagnosis of 23 cases with West Nile virus encephalomyelitis. Hum. Pathol. 2004, 35, 983–990. [Google Scholar] [CrossRef]
- Omalu, B.I.; Shakir, A.A.; Wang, G.; Lipkin, W.I.; Wiley, C.A. Fatal fulminant pan-meningo-polioencephalitis due to West Nile virus. Brain Pathol. 2003, 13, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Leis, A.A.; Fratkin, J.; Stokic, D.S.; Harrington, T.; Webb, R.M.; Slavinski, S.A. West nile poliomyelitis. Lancet Infect. Dis. 2003, 3, 9–10. [Google Scholar] [CrossRef]
- Sampson, B.A.; Nields, H.; Armbrustmacher, V.; Asnis, D.S. Muscle weakness in West Nile encephalitis is due to destruction of motor neurons. Hum. Pathol. 2003, 34, 628–629. [Google Scholar] [CrossRef]
- Singh, S.; Kumar, A. Ocular manifestations of emerging flaviviruses and the blood-retinal barrier. Viruses 2018, 10, 530. [Google Scholar] [CrossRef] [PubMed]
- Khairallah, M.; Ben Yahia, S.; Ladjimi, A.; Zeghidi, H.; Ben Romdhane, F.; Besbes, L.; Zaouali, S.; Messaoud, R. Chorioretinal involvement in patients with West Nile virus infection. Ophthalmology 2004, 111, 2065–2070. [Google Scholar] [CrossRef]
- Garg, S.; Jampol, L.M. Systemic and intraocular manifestations of West Nile virus infection. Surv. Ophthalmol. 2005, 50, 3–13. [Google Scholar] [CrossRef]
- Bains, H.S.; Jampol, L.M.; Caughron, M.C.; Parnell, J.R. Vitritis and chorioretinitis in a patient with West Nile virus infection. Arch. Ophthalmol. 2003, 121, 205–207. [Google Scholar]
- Hershberger, V.S.; Augsburger, J.J.; Hutchins, R.K.; Miller, S.A.; Horwitz, J.A.; Bergmann, M. Chorioretinal lesions in nonfatal cases of West Nile virus infection. Ophthalmology 2003, 110, 1732–1736. [Google Scholar] [CrossRef]
- Murray, K.O.; Baraniuk, S.; Resnick, M.; Arafat, R.; Kilborn, C.; Shallenberger, R.; York, T.L.; Martinez, D.; Malkoff, M.; Elgawley, N.; et al. Clinical investigation of hospitalized human cases of West Nile virus infection in Houston, Texas, 2002–2004. Vector Borne Zoonotic Dis. 2008, 8, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Lindsey, N.P.; Sejvar, J.J.; Bode, A.V.; Pape, W.J.; Campbell, G.L. Delayed mortality in a cohort of persons hospitalized with West Nile virus disease in Colorado in 2003. Vector Borne Zoonotic Dis. 2012, 12, 230–235. [Google Scholar] [CrossRef] [PubMed]
- Albagali, C.; Chaimoff, R. A case of West Nile myocarditis. Harefuah 1959, 57, 274–276. [Google Scholar]
- Kushawaha, A.; Jadonath, S.; Mobarakai, N. West nile virus myocarditis causing a fatal arrhythmia: A case report. Cases J. 2009, 2, 7147. [Google Scholar] [CrossRef]
- Pergam, S.A.; DeLong, C.E.; Echevarria, L.; Scully, G.; Goade, D.E. Myocarditis in West Nile Virus infection. Am. J. Trop. Med. Hyg. 2006, 75, 1232–1233. [Google Scholar] [CrossRef]
- Mathiot, C.C.; Gonzalez, J.P.; Georges, A.J. Current problems of arboviruses in central Africa. Bull. Soc. Pathol. Exot. Fil. 1988, 81, 396–401. [Google Scholar]
- Smith, R.D.; Konoplev, S.; DeCourten-Myers, G.; Brown, T. West Nile virus encephalitis with myositis and orchitis. Hum. Pathol. 2004, 35, 254–258. [Google Scholar] [CrossRef] [PubMed]
- Sampson, B.A.; Ambrosi, C.; Charlot, A.; Reiber, K.; Veress, J.F.; Armbrustmacher, V. The pathology of human West Nile virus infection. Hum. Pathol. 2000, 31, 527–531. [Google Scholar] [CrossRef]
- Sejvar, J.J.; Curns, A.T.; Welburg, L.; Jones, J.F.; Lundgren, L.M.; Capuron, L.; Pape, J.; Reeves, W.C.; Campbel, G.L. Neurocognitive and functional outcomes in persons recovering from West Nile virus illness. J. Neuropsychol. 2008, 2, 477–499. [Google Scholar] [CrossRef]
- Murray, K.O.; Nolan, M.S.; Ronca, S.E.; Datta, S.; Govindarajan, K.; Narayana, P.A.; Salazar, L.; Woods, S.P.; Hasbun, R. The neurocognitive and Mri Outcomes of West nile Virus infection: Preliminary analysis Using an external control group Kristy. Front. Neurol. 2018, 9, 111. [Google Scholar] [CrossRef] [PubMed]
- Carson, P.J.; Konewko, P.; Wold, K.S.; Mariani, P.; Goli, S.; Bergloff, P.; Crosby, R.D. Long-Term Clinical and Neuropsychological Outcomes of West Nile Virus Infection. Clin. Infect. Dis. 2006, 43, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Cook, R.L.; Xu, X.; Yablonsky, E.J.; Sakata, N.; Tripp, J.H.; Hess, R.; Piazza, P.; Rinaldo, C.R. Demographic and clinical factors associated with persistent symptoms after West Nile virus infection. Am. J. Trop. Med. Hyg. 2010, 83, 1133–1136. [Google Scholar] [CrossRef] [PubMed]
- Busch, M.P.; Kleinman, S.H.; Tobler, L.H.; Kamel, H.T.; Norris, P.J.; Walsh, I.; Matud, J.L.; Prince, H.E.; Lanciotti, R.S.; Wright, D.J.; et al. Virus and Antibody Dynamics in Acute West Nile Virus Infection. J. Infect. Dis. 2008, 198, 984–993. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.; Walker, C.; Herrington, E.; Lewis, J.A.; McCormick, J.; Beasley, D.W.C.; Tesh, R.B.; Fisher-Hoch, S. Persistent Infection with West Nile Virus Years after Initial Infection. J. Infect. Dis. 2010, 201, 2–4. [Google Scholar] [CrossRef] [PubMed]
- Penn, R.G.; Guarner, J.; Sejvar, J.J.; Hartman, H.; McComb, R.D.; Nevins, D.L.; Bhatnagar, J.; Zaki, S.R. Persistent neuroinvasive West Nile virus infection in an immunocompromised patient. Clin. Infect. Dis. 2006, 42, 680–683. [Google Scholar] [CrossRef]
- Nolan, M.S.; Podoll, A.S.; Hause, A.M.; Akers, K.M.; Finkel, K.W.; Murray, K.O. Prevalence of chronic kidney disease and progression of disease over time among patients enrolled in the Houston West Nile virus cohort. PLoS ONE 2012, 7, e40374. [Google Scholar] [CrossRef]
- Steele, K.E.; Linn, M.J.; Schoepp, R.J.; Komar, N.; Geisbert, T.W.; Manduca, R.M.; Calle, P.P.; Raphael, B.L.; Clippinger, T.L.; Larsen, T.; et al. Pathology of fatal West Nile virus infections in native and exotic birds during the 1999 outbreak in New York City, New York. Vet. Pathol. 2000, 37, 208–224. [Google Scholar] [CrossRef]
- Gamino, V.; Höfle, U. Pathology and tissue tropism of natural West Nile virus infection in birds: A review. Vet. Res. 2013, 44, 39. [Google Scholar] [CrossRef]
- Komar, N.; Langevin, S.; Hinten, S.; Nemeth, N.; Edwards, E.; Hettler, D.; Davis, B.; Bowen, R.; Bunning, M. Experimental infection of North American birds with the New York 1999 strain of West Nile virus. Emerg. Infect. Dis. 2003, 9, 311–322. [Google Scholar] [CrossRef]
- Nemeth, N.; Gould, D.; Bowen, R.; Komar, N. Natural and experimental West Nile virus infection in five raptor species. J. Wildl. Dis. 2006, 42, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, C.; Franca, M.; Uzal, F.; Anderson, M.; Barr, B.; Woods, L.; Moore, J.; Woolcock, P.; Shivaprasad, H.L. Pathology and Immunohistochemical Findings of West Nile Virus Infection in Psittaciformes. Vet. Pathol. 2011, 48, 975–984. [Google Scholar] [CrossRef] [PubMed]
- Eckstrand, C.D.; Woods, L.W.; Diab, S.S.; Crossley, B.M.; Giannitti, F. Diagnostic exercise: High mortality in a flock of chukar partridge chicks (Alectoris chukar) in California. Vet. Pathol. 2015, 52, 189–192. [Google Scholar] [CrossRef] [PubMed]
- Nemeth, N.M.; Bosco-Lauth, A.M.; Williams, L.M.; Bowen, R.A.; Brown, J.D. West Nile Virus Infection in Ruffed Grouse (Bonasa umbellus): Experimental Infection and Protective Effects of Vaccination. Vet. Pathol. 2017, 54, 901–911. [Google Scholar] [CrossRef]
- Bakonyi, T.; Gajdon, G.K.; Schwing, R.; Vogl, W.; Häbich, A.C.; Thaller, D.; Weissenböck, H.; Rudolf, I.; Hubálek, Z.; Nowotny, N. Chronic West Nile virus infection in kea (Nestor notabilis). Vet. Microbiol. 2016, 183, 135–139. [Google Scholar] [CrossRef]
- Gamino, V.; Escribano-Romero, E.; Gutiérrez-Guzmán, A.V.; Blázquez, A.B.; Saiz, J.C.; Höfle, U. Oculopathologic Findings in Flavivirus-Infected Gallinaceous Birds. Vet. Pathol. 2014, 51, 1113–1116. [Google Scholar] [CrossRef]
- Ziegler, U.; Angenvoort, J.; Fischer, D.; Fast, C.; Eiden, M.; Rodriguez, A.V.; Revilla-Fernández, S.; Nowotny, N.; de la Fuente, J.G.; Lierz, M.; et al. Pathogenesis of West Nile virus lineage 1 and 2 in experimentally infected large falcons. Vet. Microbiol. 2013, 161, 263–273. [Google Scholar] [CrossRef]
- Wünschmann, A.; Timurkaan, N.; Armien, A.G.; Bueno Padilla, I.; Glaser, A.; Redig, P.T. Clinical, pathological, and immunohistochemical findings in bald eagles (Haliaeetus leucocephalus) and golden eagles (Aquila chrysaetos) naturally infected with West Nile virus. J. Vet. Diagn. Investig. 2014, 26, 599–609. [Google Scholar] [CrossRef]
- Hofmeister, E.; Porter, R.E.; Christian Franson, J. Experimental susceptibility of Wood Ducks (Aix sponsa) for West Nile virus. J. Wildl. Dis. 2015, 51, 411–418. [Google Scholar] [CrossRef]
- Cox, S.L.; Campbell, G.D.; Nemeth, N.M. Outbreaks of West Nile virus in captive waterfowl in Ontario, Canada. Avian Pathol. 2015, 44, 135–141. [Google Scholar] [CrossRef]
- Gamino, V.; Escribano-Romero, E.; Blázquez, A.B.; Gutiérrez-Guzmán, A.V.; Martín-Acebes, M.; Saiz, J.C.; Höfle, U. Experimental North American West Nile Virus Infection in the Red-legged Partridge (Alectoris rufa). Vet. Pathol. 2016, 53, 585–593. [Google Scholar] [CrossRef] [PubMed]
- Hofmeister, E.K.; Lund, M.; Shearn Bochsler, V. West Nile Virus Infection in American Singer Canaries: An Experimental Model in a Highly Susceptible Avian Species. Vet. Pathol. 2018, 55, 531–538. [Google Scholar] [CrossRef] [PubMed]
- Hubálek, Z.; Kosina, M.; Rudolf, I.; Mendel, J.; Straková, P.; Tomešek, M. Mortality of Goshawks (Accipiter gentilis) due to west nile virus lineage 2. Vector-Borne Zoonotic Dis. 2018, 18, 624–627. [Google Scholar] [CrossRef] [PubMed]
- Wünschmann, A.; Armién, A.G.; Khatri, M.; Martinez, L.C.; Willette, M.; Glaser, A.; Alvarez, J.; Redig, P. Ocular Lesions in Red-Tailed Hawks (Buteo jamaicensis ) With Naturally Acquired West Nile Disease. Vet. Pathol. 2017, 54, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Lopes, H.; Redig, P.; Glaser, A.; Armien, A.; Wünschmann, A. Clinical findings, lesions, and viral antigen distribution in great gray owls (Strix nebulosa) and barred owls (Strix varia) with spontaneous West Nile virus infection. Avian Dis. 2007, 51, 140–145. [Google Scholar] [CrossRef]
- Wünschmann, A.; Shivers, J.; Bender, J.; Carroll, L.; Fuller, S.; Saggese, M.; van Wettere, A.; Redig, P. Pathologic and Immunohistochemical Findings in Goshawks (Accipiter gentilis) and Great Horned Owls (Bubo virginianus) Naturally Infected with West Nile Virus. Avian Dis. 2005, 49, 252–259. [Google Scholar] [CrossRef]
- Pauli, A.M.; Cruz-Martinez, L.A.; Ponder, J.B.; Redig, P.T.; Glaser, A.L.; Klauss, G.; Schoster, J.V.; Wünschmann, A. Ophthalmologic and oculopathologic findings in red-tailed hawks and Cooper’s hawks with naturally acquired West Nile virus infection. J. Am. Vet. Med. Assoc. 2007, 231, 1240–1248. [Google Scholar] [CrossRef]
- Nemeth, N.M.; Thomsen, B.V.; Spraker, T.R.; Benson, J.M.; Bosco-Lauth, A.M.; Oesterle, P.T.; Bright, J.M.; Muth, J.P.; Campbell, T.W.; Gidlewski, T.L.; et al. Clinical and Pathologic Responses of American Crows (Corvus brachyrhynchos) and Fish Crows (C ossifragus) to Experimental West Nile Virus Infection. Vet. Pathol. 2011, 48, 1061–1074. [Google Scholar] [CrossRef]
- Ernest, H.B.; Woods, L.W.; Hoar, B.R. Pathology associated with West Nile virus infections in the yellow-billed magpie (Pica nuttalli): A California endemic bird. J. Wildl. Dis. 2010, 46, 401–408. [Google Scholar] [CrossRef]
- Swayne, D.E.; Beck, J.R.; Zaki, S. Pathogenicity of West Nile Virus for Turkeys. Avian Dis. 2000, 44, 932. [Google Scholar] [CrossRef]
- Senne, D.A.; Pedersen, J.C.; Hutto, D.L.; Taylor, W.D.; Schmitt, B.J.; Panigrahy, B. Pathogenicity of West Nile Virus in Chickens. Avian Dis. 2000, 44, 642. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, K.V.; Dokter, A.M.; Blancher, P.J.; Sauer, J.R.; Smith, A.C.; Smith, P.A.; Stanton, J.C.; Panjabi, A.; Helft, L.; Parr, M.; et al. Decline of the North American avifauna. Science 2019, 366, 120–124. [Google Scholar] [CrossRef] [PubMed]
- George, T.L.; Harrigan, R.J.; Lamanna, J.A.; Desante, D.F.; Saracco, J.F.; Smith, T.B. Persistent impacts of West Nile virus on North American bird populations. Proc. Natl. Acad. Sci. USA 2015, 112, 14290–14294. [Google Scholar] [CrossRef] [PubMed]
- LaDeau, S.L.; Kilpatrick, A.M.; Marra, P.P. West Nile virus emergence and large-scale declines of North American bird populations. Nature 2007, 447, 710–713. [Google Scholar] [CrossRef]
- Wheeler, S.S.; Barker, C.M.; Fang, Y.; Armijos, M.V.; Carroll, B.D.; Husted, S.; Johnson, W.O.; Reisen, W.K. Differential Impact of West Nile Virus on California Birds. Condor 2009, 111, 1–20. [Google Scholar] [CrossRef]
- Kilpatrick, A.M.; Wheeler, S.S. Impact of West Nile Virus on Bird Populations: Limited Lasting Effects, Evidence for Recovery, and Gaps in Our Understanding of Impacts on Ecosystems. J. Med. Entomol. 2019, 56, 1491–1497. [Google Scholar] [CrossRef]
- Gibbs, S.E.J.; Wimberly, M.C.; Madden, M.; Masour, J.; Yabsley, M.J.; Stallknecht, D.E. Factors affecting the geographic distribution of West Nile virus in Georgia, USA: 2002–2004. Vector Borne Zoonotic Dis. 2006, 6, 73–82. [Google Scholar] [CrossRef]
- Crowder, D.W.; Dykstra, E.A.; Brauner, J.M.; Duffy, A.; Reed, C.; Martin, E.; Peterson, W.; Carrière, Y.; Dutilleul, P.; Owen, J.P. West nile virus prevalence across landscapes is mediated by local effects of agriculture on vector and host communities. PLoS ONE 2013, 8, e55006. [Google Scholar] [CrossRef]
- Bradley, C.A.; Gibbs, S.E.J.; Altizer, S. Urban land use predicts West Nile virus exposure in songbirds. Ecol. Appl. 2008, 18, 1083–1092. [Google Scholar] [CrossRef]
- Reisen, W.K.; Hahn, D.C. Comparison of immune responses of brown-headed cowbird and related blackbirds to West Nile and other mosquito-borne encephalitis viruses. J. Wildl. Dis. 2007, 43, 439–449. [Google Scholar] [CrossRef]
- Ward, M.P.; Schuermann, J.A.; Highfield, L.D.; Murray, K.O. Characteristics of an outbreak of West Nile virus encephalomyelitis in a previously uninfected population of horses. Vet. Microbiol. 2006, 118, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Paré, J.; Moore, A. West Nile virus in horses-What do you need to know to diagnose the disease? Can. Vet. J. 2018, 59, 1119–1120. [Google Scholar] [PubMed]
- Venter, M.; Pretorius, M.; Fuller, J.A.; Botha, E.; Rakgotho, M.; Stivaktas, V.; Weyer, C.; Romito, M.; Williams, J. West Nile virus lineage 2 in horses and other animals with neurologic disease, South Africa, 2008–2015. Emerg. Infect. Dis. 2017, 23, 2060–2064. [Google Scholar] [CrossRef] [PubMed]
- Porter, M.B.; Long, M.T.; Getman, L.M.; Giguère, S.; MacKay, R.J.; Lester, G.D.; Alleman, A.R.; Wamsley, H.L.; Franklin, R.P.; Jacks, S.; et al. West Nile virus encephalomyelitis in horses: 46 cases (2001). J. Am. Vet. Med. Assoc. 2003, 222, 1241–1247. [Google Scholar] [CrossRef]
- Delcambre, G.H.; Liu, J.; Streit, W.J.; Shaw, G.P.J.; Vallario, K.; Herrington, J.; Wenzlow, N.; Barr, K.L.; Long, M.T. Phenotypic characterisation of cell populations in the brains of horses experimentally infected with West Nile virus. Equine Vet. J. 2017, 49, 815–820. [Google Scholar] [CrossRef]
- Cantile, C.; Del Piero, F.; Di Guardo, G.; Arispici, M.; Del Piero, F.; Di Guardo, G.; Arispici, M. Pathologic and Immunohistochemical Findings in Naturally Occurring West Nile Virus Infection in Horses. Vet Pathol. 2001, 38, 414–421. [Google Scholar] [CrossRef]
- Yaeger, M.; Yoon, K.-J.; Schwartz, K.; Berkland, L. West Nile virus meningoencephalitis in a Suri alpaca and Suffolk ewe. J. Vet. Diagn. Investig. 2004, 16, 64–66. [Google Scholar] [CrossRef]
- Del Piero, F.; Stremme, D.W.; Habecker, P.L.; Cantile, C. West Nile Flavivirus Polioencephalomyelitis in a harbor seal (Phoca vitulina). Vet. Pathol. 2006, 43, 58–61. [Google Scholar] [CrossRef]
- Palmer, M.V.; Stoffregen, W.C.; Rogers, D.G.; Hamir, A.N.; Richt, J.A.; Pedersen, D.D.; Waters, W.R. West Nile virus infection in reindeer (Rangifer tarandus). J. Vet. Diagn. Investig. 2004, 16, 219–222. [Google Scholar] [CrossRef]
- Dutton, C.J.; Quinnell, M.; Lindsay, R.; DeLay, J.; Barker, I.K. Paraparesis in a polar bear (Ursus maritimus) associated with West Nile virus infection. J. Zoo Wildl. Med. 2009, 40, 568–571. [Google Scholar] [CrossRef]
- Miller, D.L.; Radi, Z.A.; Baldwin, C.; Ingram, D. Fatal West Nile virus infection in a white-tailed deer (Odocoileus virginianus). J. Wildl. Dis. 2005, 41, 246–249. [Google Scholar] [CrossRef] [PubMed]
- Ølberg, R.A.; Barker, I.K.; Crawshaw, G.J.; Bertelsen, M.F.; Drebot, M.A.; Andonova, M. West Nile virus encephalitis in a Barbary macaque (Macaca sylvanus). Emerg. Infect. Dis. 2004, 10, 712–714. [Google Scholar] [CrossRef] [PubMed]
- Tyler, J.W.; Turnquist, S.E.; David, A.T.; Kleiboeker, S.B.; Middleton, J.R. West Nile virus encephalomyelitis in a sheep. J. Vet. Intern. Med. 2003, 17, 242–244. [Google Scholar] [CrossRef] [PubMed]
- Rimoldi, G.; Mete, A.; Adaska, J.M.; Anderson, M.L.; Symmes, K.P.; Diab, S. West Nile Virus Infection in Sheep. Vet. Pathol. 2017, 54, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Root, J.J.; Oesterle, P.T.; Nemeth, N.M.; Klenk, K.; Gould, D.H.; McLean, R.G.; Clark, L.; Hall, J.S. Experimental infection of fox squirrels (Sciurus niger) with West Nile virus. Am. J. Trop. Med. Hyg. 2006, 75, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Nevarez, J.G.; Mitchell, M.A.; Morgan, T.; Roy, A.; Johnson, A. Association of West Nile Virus with Lymphohistiocytic Proliferative Cutaneous Lesions in American Alligators (Alligator mississippiensis) Detected by RT-PCR. J. Zoo Wildl. Med. 2008, 39, 562–567. [Google Scholar] [CrossRef] [PubMed]
- Jacobson, E.R.; Ginn, P.E.; Troutman, J.M.; Farina, L.; Stark, L.; Klenk, K.; Burkhalter, K.L.; Komar, N. West Nile virus infection in farmed American alligators (Alligator mississippiensis) in Florida. J. Wildl. Dis. 2005, 41, 96–106. [Google Scholar] [CrossRef]
- Miller, D.L.; Mauel, M.J.; Baldwin, C.; Burtle, G.; Ingram, D.; Hines, M.E.; Frazier, K.S. West Nile virus in farmed alligators. Emerg. Infect. Dis. 2003, 9, 794–799. [Google Scholar] [CrossRef]
- Cannon, A.B.; Luff, J.A.; Brault, A.C.; MacLachlan, N.J.; Case, J.B.; Green, E.N.G.; Sykes, J.E. Acute encephalitis, polyarthritis, and myocarditis associated with West Nile virus infection in a dog. J. Vet. Intern. Med. 2006, 20, 1219–1223. [Google Scholar] [CrossRef]
- Lanthier, I.; Hébert, M.; Tremblay, D.; Harel, J.; Dallaire, A.D.; Girard, C. Natural West Nile virus infection in a captive juvenile Arctic wolf (Canis lupus). J. Vet. Diagn. Investig. 2004, 16, 326–329. [Google Scholar] [CrossRef]
- Paddock, C.D.; Nicholson, W.L.; Bhatnagar, J.; Goldsmith, C.S.; Greer, P.W.; Hayes, E.B.; Risko, J.A.; Henderson, C.; Blackmore, C.G.; Lanciotti, R.S.; et al. Fatal Hemorrhagic Fever Caused by West Nile Virus in the United States. Clin. Infect. Dis. 2006, 42, 1527–1535. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Tapia, D.; Hassett, D.E.; Mitchell, W.J.; Johnson, G.C.; Kleiboeker, S.B. West Nile virus encephalitis: Sequential histopathological and immunological events in a murine model of infection. J. Neurovirol. 2007, 13, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Kimura, T.; Sasaki, M.; Okumura, M.; Kim, E.; Sawa, H. Flavivirus encephalitis: Pathological aspects of mouse and other animal models. Vet. Pathol. 2010, 47, 806–818. [Google Scholar] [CrossRef] [PubMed]
- Nalca, A.; Fellows, P.F.; Whitehouse, C.A. Vaccines and animal models for arboviral encephalitides. Antivir. Res. 2003, 60, 153–174. [Google Scholar] [CrossRef]
- Mashimo, T.; Lucas, M.; Simon-Chazottes, D.; Frenkiel, M.P.; Montagutelli, X.; Ceccaldi, P.E.; Deubel, V.; Guénet, J.L.; Desprès, P. A nonsense mutation in the gene encoding 2′-5′-oligoadenylate synthetase/L1 isoform is associated with West Nile virus susceptibility in laboratory mice. Proc. Natl. Acad. Sci. USA 2002, 99, 11311–11316. [Google Scholar] [CrossRef]
- Perelygin, A.A.; Scherbik, S.V.; Zhulin, I.B.; Stockman, B.M.; Li, Y.; Brinton, M.A. Positional cloning of the murine flavivirus resistance gene. Proc. Natl. Acad. Sci. USA 2002, 99, 9322–9327. [Google Scholar] [CrossRef]
- Scherbik, S.V.; Kluetzman, K.; Perelygin, A.A.; Brinton, M.A. Knock-in of the Oas1br allele into a flavivirus-induced disease susceptible mouse generates the resistant phenotype. Virology 2007, 368, 232–237. [Google Scholar] [CrossRef][Green Version]
- Chambers, T.J.; Diamond, M.S. Pathogenesis of flavivirus encephalitis. Adv. Virus Res. 2003, 60, 273–342. [Google Scholar]
- Shrestha, B.; Gottlieb, D.; Diamond, M.S. Infection and Injury of Neurons by West Nile Encephalitis Virus. J. Virol. 2003, 77, 13203–13213. [Google Scholar] [CrossRef]
- Himsworth, C.G.; Gurney, K.E.B.; Neimanis, A.S.; Wobeser, G.A.; Leighton, F.A. An outbreak of West Nile virus infection in captive lesser scaup (Aythya affinis) ducklings. Avian Dis. 2009, 53, 129–134. [Google Scholar] [CrossRef]
- Williams, J.H.; Mentoor, J.D.L.; Van Wilpe, E.; Venter, M. Comparative Pathology of Neurovirulent Lineage 1 (NY99/385) and Lineage 2 (SPU93/01) West Nile Virus Infections in BALBc Mice. Vet. Pathol. 2015, 52, 140–151. [Google Scholar] [CrossRef] [PubMed]
- White, J.P.; Xiong, S.; Malvin, N.P.; Khoury-Hanold, W.; Heuckeroth, R.O.; Stappenbeck, T.S.; Diamond, M.S. Intestinal Dysmotility Syndromes following Systemic Infection by Flaviviruses. Cell 2018, 175, 1198–1212.e12. [Google Scholar] [CrossRef] [PubMed]
- Johnston, L.J.; Halliday, G.M.; King, N.J. Phenotypic changes in Langerhans’ cells after infection with arboviruses: A role in the immune response to epidermally acquired viral infection? J. Virol. 1996, 70, 4761–4766. [Google Scholar] [CrossRef] [PubMed]
- Byrne, S.N.; Halliday, G.M.; Johnston, L.J.; King, N.J.C. Interleukin-1β but not tumor necrosis factor is involved in West Nile virus-induced Langerhans cell migration from the skin in C57BL/6 mice. J. Investig. Dermatol. 2001, 117, 702–709. [Google Scholar] [CrossRef]
- Johnston, L.J.; Halliday, G.M.; King, N.J.C. Langerhans cells migrate to local lymph nodes following cutaneous infection with an arbovirus. J. Investig. Dermatol. 2000, 114, 560–568. [Google Scholar] [CrossRef]
- Diamond, M.S.; Shrestha, B.; Marri, A.; Mahan, D.; Engle, M. B Cells and Antibody Play Critical Roles in the Immediate Defense of Disseminated Infection by West Nile Encephalitis Virus. J. Virol. 2003, 77, 2578–2586. [Google Scholar] [CrossRef]
- Shrestha, B.; Wang, T.; Samuel, M.A.; Whitby, K.; Craft, J.; Fikrig, E.; Diamond, M.S. Gamma Interferon Plays a Crucial Early Antiviral Role in Protection against West Nile Virus Infection. J. Virol. 2006, 80, 5338–5348. [Google Scholar] [CrossRef]
- Shrestha, B.; Diamond, M.S. Role of CD8+ T Cells in Control of West Nile Virus Infection. J. Virol. 2004, 78, 8312–8321. [Google Scholar] [CrossRef]
- Shrestha, B.; Samuel, M.A.; Diamond, M.S. CD8+ T Cells Require Perforin To Clear West Nile Virus from Infected Neurons. J. Virol. 2006, 80, 119–129. [Google Scholar] [CrossRef]
- Wang, Y.; Lobigs, M.; Lee, E.; Mullbacher, A. CD8+ T Cells Mediate Recovery and Immunopathology in West Nile Virus Encephalitis. J. Virol. 2003, 77, 13323–13334. [Google Scholar] [CrossRef]
- Klein, R.S.; Lin, E.; Zhang, B.; Luster, A.D.; Tollett, J.; Samuel, M.A.; Engle, M.; Diamond, M.S. Neuronal CXCL10 Directs CD8+ T-Cell Recruitment and Control of West Nile Virus Encephalitis. J. Virol. 2005, 79, 11457–11466. [Google Scholar] [CrossRef] [PubMed]
- Glass, W.G.; Lim, J.K.; Cholera, R.; Pletnev, A.G.; Gao, J.L.; Murphy, P.M. Chemokine receptor CCR5 promotes leukocyte trafficking to the brain and survival in West Nile virus infection. J. Exp. Med. 2005, 202, 1087–1098. [Google Scholar] [CrossRef] [PubMed]
- Shirato, K.; Kimura, T.; Mizutani, T.; Kariwa, H.; Takashima, I. Different chemokine expression in lethal and non-lethal murine West Nile virus infection. J. Med. Virol. 2004, 74, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.Y.; Guzman, H.; Zhang, H.; Travassos da Rosa, A.P.; Tesh, R.B. West Nile virus infection in the golden hamster (Mesocricetus auratus): A model for West Nile encephalitis. Emerg. Infect. Dis. 2001, 7, 714–721. [Google Scholar] [CrossRef] [PubMed]
- Wang, T. Role of γδ T cells in West Nile virus-induced encephalitis: Friend or foe? J. Neuroimmunol. 2011, 240, 22–27. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Zukor, K.; Wang, H.; Hurst, B.L.; Siddharthan, V.; Van Wettere, A.; Pilowsky, P.M.; Morrey, J.D. Phrenic nerve deficits and neurological immunopathology associated with acute West Nile virus infection in mice and hamsters. J. Neurovirol. 2017, 23, 186–204. [Google Scholar] [CrossRef]
- Morrey, J.; Siddharthan, V.; Wang, H.; Hall, J.; Skirpstunas, R.; Olsen, A.; Nordstrom, J.; Koenig, S.; Johnson, S.; Diamond, M. West Nile virus-induced acute flaccid paralysis is prevented by monoclonal antibody treatment when administered after infection of spinal cord neurons. J. Neurovirol. 2008, 14, 152–163. [Google Scholar] [CrossRef]
- Samuel, M.A.; Wang, H.; Siddharthan, V.; Morrey, J.D.; Diamond, M.S. Axonal transport mediates West Nile virus entry into the central nervous system and induces acute flaccid paralysis. Proc. Natl. Acad. Sci. USA 2007, 104, 17140–17145. [Google Scholar] [CrossRef]
- Tesh, R.B.; Siirin, M.; Guzman, H.; Travassos da Rosa, A.P.A.; Wu, X.; Duan, T.; Lei, H.; Nunes, M.R.; Xiao, S. Persistent West Nile Virus Infection in the Golden Hamster: Studies on Its Mechanism and Possible Implications for Other Flavivirus Infections. J. Infect. Dis. 2005, 192, 287–295. [Google Scholar] [CrossRef]
- Manuelidis, E.E. Neuropathology of experimental West Nile virus infection in monkeys. J. Neuropathol. Exp. Neurol. 1956, 15, 448–460. [Google Scholar] [CrossRef]
- Pogodina, V.V.; Frolova, M.P.; Malenko, G.V.; Fokina, G.I.; Koreshkova, G.V.; Kiseleva, L.L.; Bochkova, N.G.; Ralph, N.M. Study on West Nile virus persistence in monkeys. Arch. Virol. 1983, 75, 71–86. [Google Scholar] [CrossRef] [PubMed]
- Ratterree, M.S.; Gutierrez, R.A.; Travassos da Rosa, A.P.A.; Dille, B.J.; Beasley, D.W.C.; Bohm, R.P.; Desai, S.M.; Didier, P.J.; Bikenmeyer, L.G.; Dawson, G.J.; et al. Experimental infection of rhesus macaques with West Nile virus: Level and duration of viremia and kinetics of the antibody response after infection. J. Infect. Dis. 2004, 189, 669–676. [Google Scholar] [CrossRef] [PubMed]
- Verstrepen, B.E.; Fagrouch, Z.; van Heteren, M.; Buitendijk, H.; Haaksma, T.; Beenhakker, N.; Palù, G.; Richner, J.M.; Diamond, M.S.; Bogers, W.M.; et al. Experimental infection of rhesus macaques and common marmosets with a European strain of West Nile virus. PLoS Negl. Trop. Dis. 2014, 8, e2797. [Google Scholar] [CrossRef] [PubMed]
- Eldadah, A.H.; Nathanson, N.; Sarsitis, R. Pathogenesis of West Nile virus encephalitis in mice and rats. I. Influence of age and species on mortality and infection. Am. J. Epidemiol. 1967, 86, 765–775. [Google Scholar] [CrossRef]
- Eldadah, A.H.; Nathanson, N. Pathogenesis of West Nile virus encephalitis in mice and rats. II. Virus multiplication, evolution of immunofluorescence, and development of histological lesions in the brain. Am. J. Epidemiol. 1967, 86, 776–790. [Google Scholar] [CrossRef]
- Posadas-Herrera, G.; Inoue, S.; Fuke, I.; Muraki, Y.; Mapua, C.A.; Khan, A.H.; del Carmen Parquet, M.; Manabe, S.; Tanishita, O.; Ishikawa, T.; et al. Development and evaluation of a formalin-inactivated West Nile Virus vaccine (WN-VAX) for a human vaccine candidate. Vaccine 2010, 28, 7939–7946. [Google Scholar] [CrossRef]
- Bonafé, N.; Rininger, J.A.; Chubet, R.G.; Foellmer, H.G.; Fader, S.; Anderson, J.F.; Bushmich, S.L.; Anthony, K.; Ledizet, M.; Fikrig, E.; et al. A recombinant West Nile virus envelope protein vaccine candidate produced in Spodoptera frugiperda expresSF+ cells. Vaccine 2009, 27, 213–222. [Google Scholar] [CrossRef]
- Poore, E.A.; Slifka, D.K.; Raué, H.P.; Thomas, A.; Hammarlund, E.; Quintel, B.K.; Torrey, L.L.; Slifka, A.M.; Richner, J.M.; Dubois, M.E.; et al. Pre-clinical development of a hydrogen peroxide-inactivated West Nile virus vaccine. Vaccine 2017, 35, 283–292. [Google Scholar] [CrossRef]
- Seino, K.K.; Long, M.T.; Gibbs, E.P.J.; Bowen, R.A.; Beachboard, S.E.; Humphrey, P.P.; Dixon, M.A.; Bourgeois, M.A. Comparative efficacies of three commercially available vaccines against West Nile Virus (WNV) in a short-duration challenge trial involving an equine WNV encephalitis model. Clin. Vaccine Immunol. 2007, 14, 1465–1471. [Google Scholar] [CrossRef]
- Siger, L.; Bowen, R.; Karaca, K.; Murray, M.; Jagannatha, S.; Echols, B.; Nordgren, R.; Minke, J.M. Evaluation of the efficacy provided by a Recombinant Canarypox-Vectored Equine West Nile Virus vaccine against an experimental West Nile Virus intrathecal challenge in horses. Vet. Ther. 2006, 7, 249–256. [Google Scholar]
- Chaintoutis, S.C.; Diakakis, N.; Papanastassopoulou, M.; Banos, G.; Dovas, C.I. Evaluation of cross-protection of a lineage 1 West Nile virus inactivated vaccine against natural infections from a virulent lineage 2 strain in horses, under field conditions. Clin. Vaccine Immunol. 2015, 22, 1040–1049. [Google Scholar] [CrossRef][Green Version]
- Bunning, M.L.; Bowen, R.A.; Bruce Cropp, C.; Sullivan, K.G.; Davis, B.S.; Komar, N.; Godsey, M.S.; Baker, D.; Hettler, D.L.; Holmes, D.A.; et al. Experimental infection of horses with West Nile virus. Emerg. Infect. Dis. 2002, 8, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Olivares, J.; Mansfield, K.L.; Phipps, L.P.; Johnson, N.; Tearle, J.; Fooks, A.R. Antibody response in horses following experimental infection with West Nile Virus lineages 1 and 2. Transbound. Emerg. Dis. 2011, 58, 206–212. [Google Scholar] [CrossRef]
- Long, M.T.; Gibbs, E.P.J.; Mellencamp, M.W.; Zhang, S.; Barnett, D.C.; Seino, K.K.; Beachboard, S.E.; Humphrey, P.P. Safety of an attenuated West Nile virus vaccine, live Flavivirus chimera in horses. Equine Vet. J. 2007, 39, 486–490. [Google Scholar] [CrossRef]
- West Nile Virus|AAEP. Available online: https://aaep.org/guidelines/vaccination-guidelines/core-vaccination-guidelines/west-nile-virus (accessed on 6 December 2019).
- Suen, W.W.; Uddin, M.J.; Wang, W.; Brown, V.; Adney, D.R.; Broad, N.; Prow, N.A.; Bowen, R.A.; Hall, R.A.; Bielefeldt-Ohmann, H. Experimental west nile virus infection in rabbits: An alternative model for studying induction of disease and virus control. Pathogens 2015, 4, 529–558. [Google Scholar] [CrossRef] [PubMed]
- Teehee, M.L.; Bunning, M.L.; Stevens, S.; Bowen, R.A. Experimental infection of pigs with West Nile virus. Brief Report. Arch. Virol. 2005, 150, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Austgen, L.E.; Bowen, R.A.; Bunning, M.L.; Davis, B.S.; Mitchell, C.J.; Chang, G.J.J. Experimental Infection of Cats and Dogs with West Nile Virus. Emerg. Infect. Dis. 2004, 10, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Steinman, A.; Banet-Noach, C.; Simanov, L.; Grinfeld, N.; Aizenberg, Z.; Levi, O.; Lahav, D.; Malkinson, M.; Perk, S.; Shpigel, N.Y. Experimental infection of common garter snakes (Thamnophis sirtalis) with West Nile virus. Vector Borne Zoonotic Dis. 2006, 6, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Fagre, A.C.; Kading, R.C. Can bats serve as reservoirs for Arboviruses? Viruses 2019, 11, 215. [Google Scholar] [CrossRef] [PubMed]
- Dridi, M.; Van Den Berg, T.; Lecollinet, S.; Lambrecht, B. Evaluation of the pathogenicity of West Nile virus (WNV) lineage 2 strains in a SPF chicken model of infection: NS3-249Pro mutation is neither sufficient nor necessary for conferring virulence. Vet. Res. 2015, 46, 130. [Google Scholar] [CrossRef][Green Version]
- Deardorff, E.R.; Fitzpatrick, K.A.; Jerzak, G.V.S.; Shi, P.Y.; Kramer, L.D.; Ebel, G.D. West nile virus experimental evolution in vivo and the trade-off hypothesis. PLoS Pathog. 2011, 7, e1002335. [Google Scholar] [CrossRef] [PubMed]
- Jerzak, G.V.S.; Brown, I.; Shi, P.-Y.; Kramer, L.D.; Ebel, G.D. Genetic diversity and purifying selection in West Nile virus populations are maintained during host switching. Virology 2008, 374, 256–260. [Google Scholar] [CrossRef]
- Kading, R.C.; Biggerstaff, B.J.; Young, G.; Komar, N. Mosquitoes used to draw blood for arbovirus viremia determinations in small vertebrates. PLoS ONE 2014, 9, e99342. [Google Scholar] [CrossRef] [PubMed]
- Grubaugh, N.D.; Fauver, J.R.; Rückert, C.; Weger-Lucarelli, J.; Garcia-Luna, S.; Murrieta, R.A.; Gendernalik, A.; Smith, D.R.; Brackney, D.E.; Ebel, G.D. Mosquitoes Transmit Unique West Nile Virus Populations during Each Feeding Episode. Cell Rep. 2017, 19, 709–718. [Google Scholar] [CrossRef] [PubMed]
- Worwa, G.; Hutton, A.A.; Frey, M.; Duggal, N.K.; Brault, A.C.; Reisen, W.K. Increases in the competitive fitness of West Nile virus isolates after introduction into California. Virology 2018, 514, 170–181. [Google Scholar] [CrossRef]
- Duggal, N.K.; Bosco-Lauth, A.; Bowen, R.A.; Wheeler, S.S.; Reisen, W.K.; Felix, T.A.; Mann, B.R.; Romo, H.; Swetnam, D.M.; Barrett, A.D.T.; et al. Evidence for Co-evolution of West Nile Virus and House Sparrows in North America. PLoS Negl. Trop. Dis. 2014, 8, e3262. [Google Scholar] [CrossRef]
- Langevin, S.A.; Brault, A.C.; Panella, N.A.; Bowen, R.A.; Komar, N. Variation in virulence of West Nile virus strains for house sparrows (Passer domesticus). Am. J. Trop. Med. Hyg. 2005, 72, 99–102. [Google Scholar] [CrossRef]
- Langevin, S.A.; Bowen, R.A.; Reisen, W.K.; Andrade, C.C.; Ramey, W.N.; Maharaj, P.D.; Anishchenko, M.; Kenney, J.L.; Duggal, N.K.; Romo, H.; et al. Host Competence and Helicase Activity Differences Exhibited by West Nile Viral Variants Expressing NS3-249 Amino Acid Polymorphisms. PLoS ONE 2014, 9, e100802. [Google Scholar] [CrossRef]
- Dietrich, E.A.; Langevin, S.A.; Huang, C.Y.-H.; Maharaj, P.D.; Delorey, M.J.; Bowen, R.A.; Kinney, R.M.; Brault, A.C. West Nile Virus Temperature Sensitivity and Avian Virulence Are Modulated by NS1-2B Polymorphisms. PLoS Negl. Trop. Dis. 2016, 10, e0004938. [Google Scholar] [CrossRef]
- Melian, E.B.; Hall-Mendelin, S.; Du, F.; Owens, N.; Bosco-Lauth, A.M.; Nagasaki, T.; Rudd, S.; Brault, A.C.; Bowen, R.A.; Hall, R.A.; et al. Programmed ribosomal frameshift alters expression of west nile virus genes and facilitates virus replication in birds and mosquitoes. PLoS Pathog. 2014, 10, e1004447. [Google Scholar] [CrossRef]
- Maharaj, P.D.; Bosco-Lauth, A.M.; Langevin, S.A.; Anishchenko, M.; Bowen, R.A.; Reisen, W.K.; Brault, A.C. West Nile and St. Louis encephalitis viral genetic determinants of avian host competence. PLoS Negl. Trop. Dis. 2018, 12, e0006302. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.M.; Brault, A.C.; van Amerongen, G.; Bosco-Lauth, A.M.; Romo, H.; Sewbalaksing, V.D.; Bowen, R.A.; Osterhaus, A.D.M.E.; Koraka, P.; Martina, B.E.E. Susceptibility of Carrion Crows to Experimental Infection with Lineage 1 and 2 West Nile Viruses. Emerg. Infect. Dis. 2015, 21, 1357–1365. [Google Scholar] [CrossRef] [PubMed]
- Murray, K.O.; Kolodziej, S.; Ronca, S.E.; Gorchakov, R.; Navarro, P.; Nolan, M.S.; Podoll, A.; Finkel, K.; Mandayam, S. Visualization of West Nile Virus in Urine Sediment using Electron Microscopy and Immunogold up to Nine Years Postinfection. Am. J. Trop. Med. Hyg. 2017, 97, 1913–1919. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ogawa, M.; Okubo, H.; Tsuji, Y.; Yasui, N.; Someda, K. Chronic progressive encephalitis occurring 13 years after Russian spring–summer encephalitis. J. Neurol. Sci. 1973, 19, 363–373. [Google Scholar] [CrossRef]
| Order | ACCI | ANSE | CHAR | CICO | FALC | GALL | PASS | PELE | PHOE | PSIT | STRI | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Family | ACCI | ANAT | LARI | ARDE | FALC | PHAS | CORV | LANI | PASS | FRIN | PHAL | PHOE | PSIT | STRO | STRI |
| Brain | |||||||||||||||
| Perivascular cuffs | + | + | + | ND | + | + | + | + | + | − | − | + | + | + | + |
| Gliosis/glial nodules | + | + | + | ND | + | + | + | + | − | − | − | + | + | + | + |
| Meningeal inflammation | + | + | − | ND | + | − | + | − | − | − | + | − | − | − | + |
| Neuronal degeneration and necrosis | + | + | + | ND | + | + | + | − | − | − | + | − | − | + | + |
| Vasculitis | + | − | − | ND | − | − | + | − | + | − | − | − | − | − | − |
| Hemorrhage | + | − | − | ND | − | + | − | − | − | − | + | − | − | − | + |
| Spinal cord | |||||||||||||||
| Inflammation | + | + | ND | ND | ND | ND | ND | NT | ND | NT | ND | ND | ND | NT | + |
| Neuronal degeneration and necrosis | − | + | ND | ND | ND | ND | ND | NT | ND | NT | ND | ND | ND | NT | + |
| Peripheral nervous system | |||||||||||||||
| Inflammation | + | − | ND | ND | + | + | − | NT | NT | NT | ND | ND | ND | NT | + |
| Eye | |||||||||||||||
| Retinitis | + | NT | NT | NT | NT | − | ND | NT | NT | NT | NT | NT | + | NT | + |
| Retinal necrosis | + | NT | NT | NT | NT | − | ND | NT | NT | NT | NT | NT | − | NT | − |
| Pectenitis | + | NT | NT | NT | NT | + | ND | NT | NT | NT | NT | NT | − | NT | − |
| Uveitis (inc. iris, choroid, ciliary body) | + | NT | NT | NT | + | + | ND | NT | NT | NT | NT | NT | − | NT | − |
| Heart | |||||||||||||||
| Inflammation | + | + | + | + | + | + | + | + | − | − | + | + | + | NT | + |
| Myofiber necrosis | + | + | − | − | + | + | + | − | − | − | − | − | + | NT | + |
| Myofibril lysis and mineralization | + | + | + | + | + | + | + | − | − | − | + | + | + | NT | − |
| Vasculitis | − | − | − | − | + | − | + | − | − | − | − | − | − | NT | − |
| Hemorrhage | + | − | + | + | − | + | + | − | − | − | + | + | + | NT | − |
| Gastrointestinal tract | |||||||||||||||
| Inflammation | + | − | ND | ND | − | + | + | − | ND | − | − | − | + | NT | + |
| Enterocyte necrosis | − | + | ND | ND | − | + | + | − | ND | − | − | − | − | NT | + |
| Crypt necrosis | − | − | ND | ND | − | − | + | + | ND | − | + | + | + | NT | + |
| Hemorrhage | − | + | ND | ND | − | − | + | − | ND | − | − | + | − | NT | − |
| Liver | |||||||||||||||
| Inflammation | + | + | ND | ND | + | + | + | + | − | + | − | − | + | + | + |
| Hepatocyte necrosis | + | + | ND | ND | − | + | + | + | + | + | + | + | + | + | + |
| Vasculitis | − | − | ND | ND | + | − | − | − | − | − | − | − | − | − | − |
| Bile duct hyperplasia | + | − | ND | ND | − | − | − | − | − | − | − | + | − | − | − |
| Hemosiderosis | + | − | ND | ND | − | + | + | − | + | − | − | − | − | − | + |
| Hemorrhage | + | − | ND | ND | − | − | + | − | − | − | − | − | − | − | − |
| Kidney | |||||||||||||||
| Inflammation (interstitial) | + | + | ND | + | + | + | + | ND | + | + | + | + | + | NT | + |
| Tubular necrosis | + | + | ND | − | − | + | + | ND | + | + | − | − | + | NT | + |
| Glomerular necrosis | − | − | ND | − | − | − | − | ND | − | − | − | − | + | NT | + |
| Vasculitis | − | − | ND | − | + | − | − | ND | − | − | − | − | − | NT | − |
| Hemorrhage | − | + | ND | − | − | − | − | ND | − | − | − | − | − | NT | − |
| Lung | NT | ||||||||||||||
| Inflammation | + | − | ND | ND | − | + | + | − | + | − | ND | ND | − | NT | + |
| Necrosis | + | − | ND | ND | − | − | + | − | − | − | ND | ND | − | NT | + |
| Vasculitis | − | − | ND | ND | − | − | + | − | − | − | ND | ND | − | NT | − |
| Edema | + | − | ND | ND | + | − | − | + | + | − | ND | ND | − | NT | − |
| Spleen | |||||||||||||||
| Lymphoid necrosis/apoptosis | + | + | ND | + | − | + | + | + | − | + | + | ND | + | NT | + |
| Lymphoid depletion | + | + | ND | − | + | + | − | − | − | − | − | ND | − | NT | + |
| Fibrin deposition | − | − | ND | + | − | + | − | + | − | − | + | ND | − | NT | + |
| Hemorrhage | − | + | ND | + | − | − | − | − | − | − | + | ND | − | NT | − |
| Hemosiderosis | + | + | ND | − | − | + | + | − | + | − | − | ND | − | NT | + |
| Vasculitis | − | − | ND | − | + | − | − | − | − | − | − | ND | − | NT | − |
| Other lymphoid organs | |||||||||||||||
| Thymic lymphoid necrosis | NT | + | NT | NT | NT | + | NT | NT | NT | NT | NT | NT | − | NT | + |
| Bursal epithelial atrophy−apoptosis | + | − | NT | NT | NT | ND | NT | NT | − | NT | NT | NT | ND | NT | + |
| Bursal lymphoid atrophy−apoptosis | + | + | NT | NT | NT | + | NT | NT | + | NT | NT | NT | ND | NT | + |
| Bone marrow necrosis | ND | − | NT | NT | NT | − | + | NT | NT | NT | NT | NT | ND | NT | ND |
| Endocrine system | |||||||||||||||
| Pancreatic necrosis | − | + | ND | ND | − | + | − | + | ND | − | − | + | + | NT | + |
| Pancreatic inflammation | + | + | ND | ND | + | + | − | − | ND | − | + | + | + | NT | + |
| Adrenal gland necrosis | − | ND | ND | ND | NT | + | − | NT | NT | NT | ND | ND | + | NT | − |
| Adrenal gland inflammation | + | ND | ND | ND | NT | + | + | NT | NT | NT | ND | ND | + | NT | + |
| Thyroid gland necrosis | − | + | NT | NT | NT | NT | NT | NT | NT | NT | NT | NT | ND | NT | ND |
| Thyroid gland inflammation | + | − | NT | NT | NT | NT | NT | NT | NT | NT | NT | NT | ND | NT | ND |
| Skin | |||||||||||||||
| Inflammation | ND | + | NT | NT | NT | − | NT | ND | NT | − | NT | NT | + | NT | − |
| Skeletal muscle | |||||||||||||||
| Myofibril degeneration and necrosis | + | − | NT | NT | + | − | ND | NT | NT | − | NT | NT | + | NT | + |
| Inflammation | + | − | NT | NT | + | + | ND | NT | NT | − | NT | NT | + | NT | + |
| Fibrosis | + | − | NT | NT | − | − | ND | NT | NT | − | NT | NT | − | NT | + |
| Gonads | |||||||||||||||
| Inflammation | − | − | ND | ND | − | + | NT | ND | NT | NT | ND | ND | − | NT | + |
| Necrosis | − | − | ND | ND | − | − | NT | ND | NT | NT | ND | ND | − | NT | + |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byas, A.D.; Ebel, G.D. Comparative Pathology of West Nile Virus in Humans and Non-Human Animals. Pathogens 2020, 9, 48. https://doi.org/10.3390/pathogens9010048
Byas AD, Ebel GD. Comparative Pathology of West Nile Virus in Humans and Non-Human Animals. Pathogens. 2020; 9(1):48. https://doi.org/10.3390/pathogens9010048
Chicago/Turabian StyleByas, Alex D., and Gregory D. Ebel. 2020. "Comparative Pathology of West Nile Virus in Humans and Non-Human Animals" Pathogens 9, no. 1: 48. https://doi.org/10.3390/pathogens9010048
APA StyleByas, A. D., & Ebel, G. D. (2020). Comparative Pathology of West Nile Virus in Humans and Non-Human Animals. Pathogens, 9(1), 48. https://doi.org/10.3390/pathogens9010048





