Rapid Discrimination of Methicillin-Resistant Staphylococcus aureus by MALDI-TOF MS
Abstract
1. Introduction
2. Results
2.1. Specific Peaks for MRSA and MSSA
2.2. Specific Peaks According to MRSA SCCmec Type
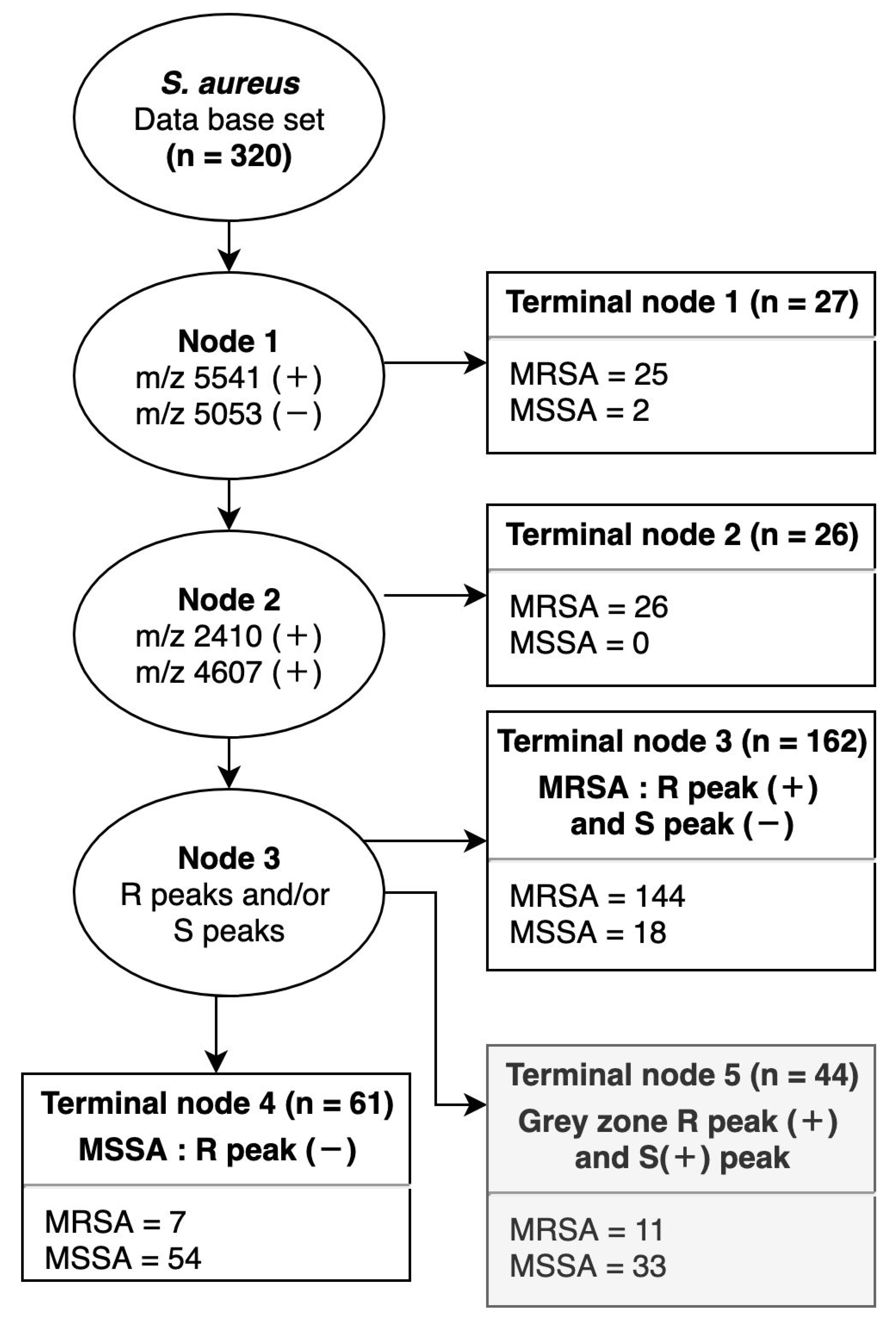
2.3. Decision Tree for MRSA and MSSA
2.4. Simple Prediction Model Based on a Combination of Specific Peaks
2.5. Evaluation of the Test Set
3. Discussion
4. Materials and Methods
4.1. Bacterial Strains
4.2. MALDI-TOF Measurements of Bacterial Cells
4.3. Screening for the mecA Gene and SCCmec Type
4.4. Analysis of Mass Spectra
4.5. Ethical Approval
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Tong, S.Y.; Davis, J.S.; Eichenberger, E.; Holland, T.L.; Fowler, V.G., Jr. Staphylococcus aureus infections: Epidemiology, pathophysiology, clinical manifestations, and management. Clin. Microbiol. Rev. 2015, 28, 603–661. [Google Scholar] [CrossRef] [PubMed]
- Lakhundi, S.; Zhang, K. Methicillin-Resistant Staphylococcus aureus: Molecular Characterization, Evolution, and Epidemiology. Clin. Microbiol. Rev. 2018, 31. [Google Scholar] [CrossRef]
- Asadollahi, P.; Farahani, N.N.; Mirzaii, M.; Khoramrooz, S.S.; van Belkum, A.; Asadollahi, K.; Dadashi, M.; Darban-Sarokhalil, D. Distribution of the Most Prevalent Spa Types among Clinical Isolates of Methicillin-Resistant and -Susceptible Staphylococcus aureus around the World: A Review. Front. Microbiol. 2018, 9, 163. [Google Scholar] [CrossRef] [PubMed]
- Pastagia, M.; Kleinman, L.C.; Lacerda de la Cruz, E.G.; Jenkins, S.G. Predicting risk for death from MRSA bacteremia. Emerg. Infect. Dis. 2012, 18, 1072–1080. [Google Scholar] [CrossRef] [PubMed]
- Kang, C.I.; Song, J.H. Antimicrobial resistance in Asia: Current epidemiology and clinical implications. Infect. Chemother. 2013, 45, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Edwards-Jones, V.; Claydon, M.A.; Evason, D.J.; Walker, J.; Fox, A.J.; Gordon, D.B. Rapid discrimination between methicillin-sensitive and methicillin-resistant Staphylococcus aureus by intact cell mass spectrometry. J. Med. Microbiol. 2000, 49, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Hiramatsu, K.; Katayama, Y.; Yuzawa, H.; Ito, T. Molecular genetics of methicillin-resistant Staphylococcus aureus. Int. J. Med. Microbiol. 2002, 292, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Grmek-Kosnik, I.; Dermota, U.; Ribic, H.; Storman, A.; Petrovic, Z.; Zohar-Cretnik, T. Evaluation of single vs pooled swab cultures for detecting MRSA colonization. J. Hosp. Infect. 2018, 98, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; Fox, A.J.; Edwards-Jones, V.; Gordon, D.B. Intact cell mass spectrometry (ICMS) used to type methicillin-resistant Staphylococcus aureus: Media effects and inter-laboratory reproducibility. J. Microbiol. Methods 2002, 48, 117–126. [Google Scholar] [CrossRef]
- Luo, J.; Li, J.; Yang, H.; Yu, J.; Wei, H. Accurate Detection of Methicillin-Resistant Staphylococcus aureus in Mixtures by Use of Single-Bacterium Duplex Droplet Digital PCR. J. Clin. Microbiol. 2017, 55, 2946–2955. [Google Scholar] [CrossRef] [PubMed]
- Van Belkum, A.; Rochas, O. Laboratory-Based and Point-of-Care Testing for MSSA/MRSA Detection in the Age of Whole Genome Sequencing. Front. Microbiol. 2018, 9, 1437. [Google Scholar] [CrossRef] [PubMed]
- Patel, R. MALDI-TOF MS for the diagnosis of infectious diseases. Clin. Chem. 2015, 61, 100–111. [Google Scholar] [CrossRef] [PubMed]
- Bohme, K.; Morandi, S.; Cremonesi, P.; Fernandez No, I.C.; Barros-Velazquez, J.; Castiglioni, B.; Brasca, M.; Canas, B.; Calo-Mata, P. Characterization of Staphylococcus aureus strains isolated from Italian dairy products by MALDI-TOF mass fingerprinting. Electrophoresis 2012, 33, 2355–2364. [Google Scholar] [CrossRef] [PubMed]
- Ostergaard, C.; Hansen, S.G.; Moller, J.K. Rapid first-line discrimination of methicillin resistant Staphylococcus aureus strains using MALDI-TOF MS. Int. J. Med. Microbiol. 2015, 305, 838–847. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Ding, J.; Rao, X.; Yu, J.; Chu, M.; Ren, W.; Wang, L.; Xue, W. Analysis of methicillin-resistant Staphylococcus aureus major clonal lineages by Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry (MALDI-TOF MS). J. Microbiol. Methods 2015, 117, 122–127. [Google Scholar] [CrossRef] [PubMed]
- Camoez, M.; Sierra, J.M.; Dominguez, M.A.; Ferrer-Navarro, M.; Vila, J.; Roca, I. Automated categorization of methicillin-resistant Staphylococcus aureus clinical isolates into different clonal complexes by MALDI-TOF mass spectrometry. Clin. Microbiol. Infect. 2016, 22. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, K.; Pakulat, N.; Macht, M.; Krut, O.; Seifert, H.; Fleer, S.; Hunger, F.; Kronke, M. Identification and discrimination of Staphylococcus aureus strains using matrix-assisted laser desorption/ionization-time of flight mass spectrometry. Proteomics 2002, 2, 747–753. [Google Scholar] [CrossRef]
- Du, Z.; Yang, R.; Guo, Z.; Song, Y.; Wang, J. Identification of Staphylococcus aureus and determination of its methicillin resistance by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. Anal. Chem. 2002, 74, 5487–5491. [Google Scholar] [CrossRef] [PubMed]
- Josten, M.; Dischinger, J.; Szekat, C.; Reif, M.; Al-Sabti, N.; Sahl, H.G.; Parcina, M.; Bekeredjian-Ding, I.; Bierbaum, G. Identification of agr-positive methicillin-resistant Staphylococcus aureus harbouring the class A mec complex by MALDI-TOF mass spectrometry. Int. J. Med. Microbiol. 2014, 304, 1018–1023. [Google Scholar] [CrossRef] [PubMed]
- Van Belkum, A.; Welker, M.; Pincus, D.; Charrier, J.P.; Girard, V. Matrix-Assisted Laser Desorption Ionization Time-of-Flight Mass Spectrometry in Clinical Microbiology: What Are the Current Issues? Ann. Lab. Med. 2017, 37, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Gagnaire, J.; Dauwalder, O.; Boisset, S.; Khau, D.; Freydiere, A.M.; Ader, F.; Bes, M.; Lina, G.; Tristan, A.; Reverdy, M.E.; et al. Detection of Staphylococcus aureus delta-toxin production by whole-cell MALDI-TOF mass spectrometry. PLoS ONE 2012, 7, e40660. [Google Scholar] [CrossRef] [PubMed]
- Szabados, F.; Kaase, M.; Anders, A.; Gatermann, S.G. Identical MALDI TOF MS-derived peak profiles in a pair of isogenic SCCmec-harboring and SCCmec-lacking strains of Staphylococcus aureus. J. Infect. 2012, 65, 400–405. [Google Scholar] [CrossRef] [PubMed]
- Rhoads, D.D.; Wang, H.; Karichu, J.; Richter, S.S. The presence of a single MALDI-TOF mass spectral peak predicts methicillin resistance in staphylococci. Diagn. Microbiol. Infect. Dis. 2016, 86, 257–261. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.Y.; Lien, F.; Liu, T.P.; Chen, C.H.; Chen, C.J.; Lu, J.J. Application of a MALDI-TOF analysis platform (ClinProTools) for rapid and preliminary report of MRSA sequence types in Taiwan. PeerJ 2018, 6, e5784. [Google Scholar] [CrossRef] [PubMed]
- Lasch, P.; Fleige, C.; Stammler, M.; Layer, F.; Nubel, U.; Witte, W.; Werner, G. Insufficient discriminatory power of MALDI-TOF mass spectrometry for typing of Enterococcus faecium and Staphylococcus aureus isolates. J. Microbiol. Methods 2014, 100, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Sauget, M.; van der Mee-Marquet, N.; Bertrand, X.; Hocquet, D. Matrix-assisted laser desorption ionization-time of flight Mass spectrometry can detect Staphylococcus aureus clonal complex 398. J. Microbiol. Methods 2016, 127, 20–23. [Google Scholar] [CrossRef] [PubMed]
- Kwon, S.S.; Hong, S.K.; Kim, M.S.; Yong, D.; Lee, K. Performance of Matrix-Assisted Laser Desorption Ionization Time-of-Fight Mass Spectrometry for Rapid Discrimination of Methicillin-Resistant Staphylococcus aureus (MRSA): First Report of a Relation Between Protein Peaks and MRSA spa Type. Ann. Lab. Med. 2017, 37, 553–555. [Google Scholar] [CrossRef] [PubMed]
- Josten, M.; Reif, M.; Szekat, C.; Al-Sabti, N.; Roemer, T.; Sparbier, K.; Kostrzewa, M.; Rohde, H.; Sahl, H.G.; Bierbaum, G. Analysis of the matrix-assisted laser desorption ionization-time of flight mass spectrum of Staphylococcus aureus identifies mutations that allow differentiation of the main clonal lineages. J. Clin. Microbiol. 2013, 51, 1809–1817. [Google Scholar] [CrossRef] [PubMed]
- Zetola, N.; Francis, J.S.; Nuermberger, E.L.; Bishai, W.R. Community-acquired meticillin-resistant Staphylococcus aureus: An emerging threat. Lancet Infect. Dis. 2005, 5, 275–286. [Google Scholar] [CrossRef]
- Wolters, M.; Rohde, H.; Maier, T.; Belmar-Campos, C.; Franke, G.; Scherpe, S.; Aepfelbacher, M.; Christner, M. MALDI-TOF MS fingerprinting allows for discrimination of major methicillin-resistant Staphylococcus aureus lineages. Int. J. Med. Microbiol. 2011, 301, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Bayer, A.; Cosgrove, S.E.; Daum, R.S.; Fridkin, S.K.; Gorwitz, R.J.; Kaplan, S.L.; Karchmer, A.W.; Levine, D.P.; Murray, B.E.; et al. Clinical practice guidelines by the infectious diseases society of america for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children: Executive summary. Clin. Infect. Dis. 2011, 52, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Udo, E.E. Community-acquired methicillin-resistant Staphylococcus aureus: The new face of an old foe? Med. Princ. Pract. 2013, 22 (Suppl. 1), 20–29. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.J.; Tsai, F.J.; Ho, C.M.; Liu, Y.C.; Chen, C.J. Peptide biomarker discovery for identification of methicillin-resistant and vancomycin-intermediate Staphylococcus aureus strains by MALDI-TOF. Anal. Chem. 2012, 84, 5685–5692. [Google Scholar] [CrossRef] [PubMed]
- Kostrzewa, M.; Sparbier, K.; Maier, T.; Schubert, S. MALDI-TOF MS: An upcoming tool for rapid detection of antibiotic resistance in microorganisms. Proteom. Clin. Appl. 2013, 7, 767–778. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Kuwahara-Arai, K.; Katayama, Y.; Uehara, Y.; Han, X.; Kondo, Y.; Hiramatsu, K. Staphylococcal Cassette Chromosome mec (SCCmec) analysis of MRSA. Methods Mol. Biol. 2014, 1085, 131–148. [Google Scholar] [CrossRef] [PubMed]
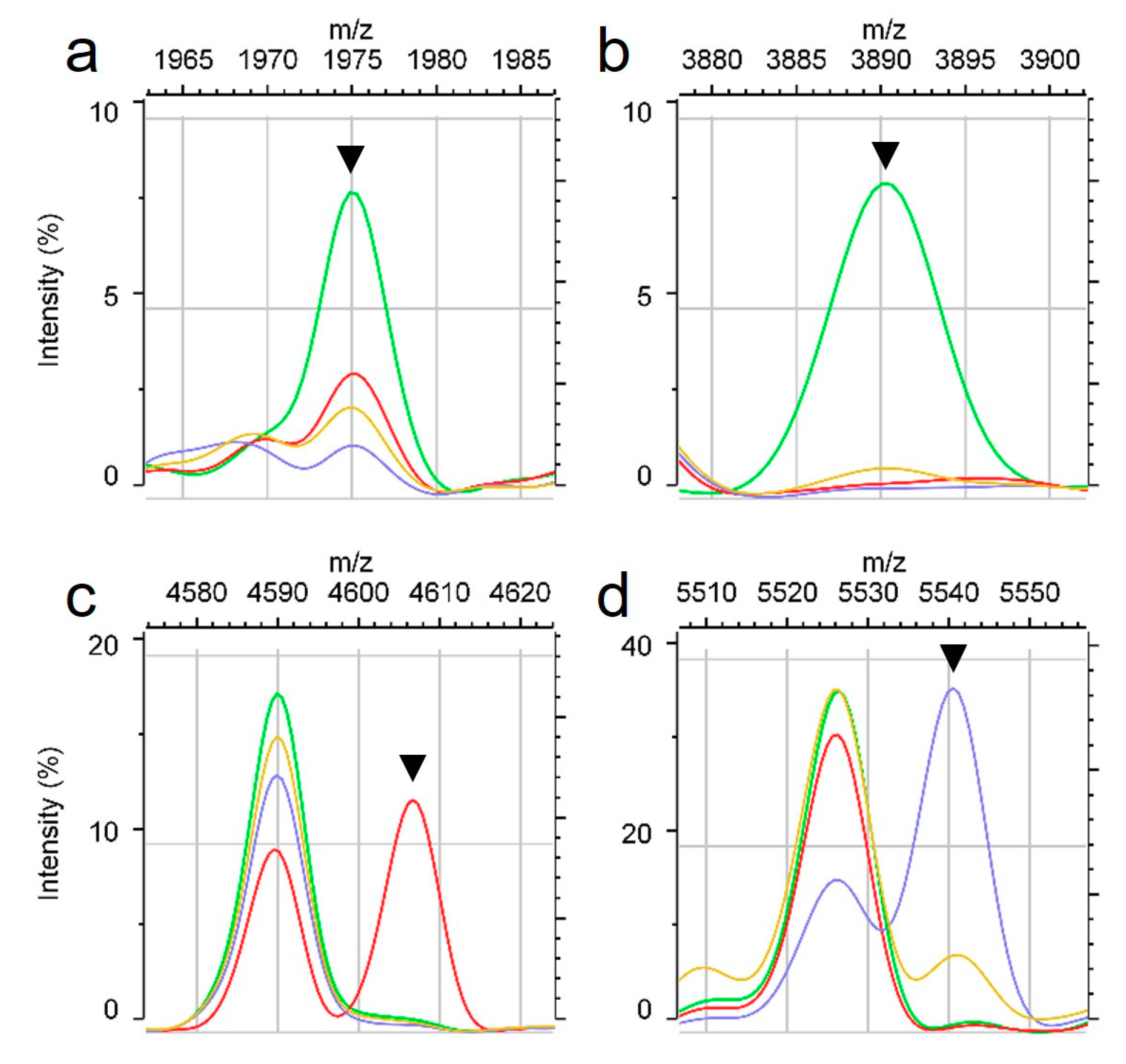
| Type | Peak (m/z) | SCCmec | MSSA | SE | SP | PPV | NPV | P | Decision Tree | Reference | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| II | III | IV | ||||||||||
| MRSA SCCmec type II | 1975 | 81 | 12 | 6 | 26 | 80.6 | 80.0 | 79.1 | 81.5 | <0.001 | node 3 | |
| 2134 | 65 | 52 | 27 | 30 | 65.2 | 67.3 | 65.2 | 67.3 | <0.001 | |||
| 2592 | 24 | 12 | 3 | 4 | 23.9 | 95.2 | 82.2 | 57.1 | <0.001 | node 3 | ||
| 3890 | 55 | 0 | 0 | 3 | 54.8 | 98.2 | 96.6 | 69.8 | <0.001 | node 3 | [9] *, [10] †, [12] †, [20] † | |
| MRSA SCCmec type III | 2204 | 3 | 68 | 0 | 0 | 68.0 | 98.3 | 77.3 | 97.3 | 0.001 | ||
| 2410 | 43 | 100 | 18 | 15 | 100.0 | 69.8 | 21.9 | 100.0 | <0.001 | node 2, 3 | [15] †, [17,18,19,20] † | |
| 2874 | 6 | 64 | 9 | 5 | 64.0 | 94.2 | 48.5 | 96.9 | <0.001 | |||
| 4607 | 1 | 100 | 0 | 0 | 100.0 | 99.7 | 96.2 | 100.0 | <0.001 | node 2, 3 | ||
| 6594 | 3 | 92 | 9 | 12 | 92.0 | 93.2 | 53.5 | 99.3 | <0.001 | [12] †, [20] †, [25] ※, [26] † | ||
| 9216 | 1 | 100 | 0 | 0 | 100.0 | 99.7 | 96.2 | 100.0 | <0.001 | |||
| MRSA SCCmec type IV | 5053 | 98 | 80 | 21 | 91 | N/A | <0.001 | node 1 | ||||
| 5541 | 1 | 0 | 76 | 24 | 75.8 | 90.2 | 47.2 | 97.0 | <0.001 | node 1 | [10] † | |
| 5579 | 0 | 0 | 70 | 11 | 69.7 | 95.8 | 65.7 | 96.5 | <0.001 | |||
| MSSA | 2194 | 2 | 24 | 76 | 55 | 55.1 | 84.0 | 63.4 | 78.9 | <0.001 | node 3 | |
| 2232 | 1 | 8 | 55 | 37 | 37.4 | 90.1 | 65.6 | 74.1 | <0.001 | node 3 | ||
| 2301 | 6 | 56 | 94 | 74 | 73.8 | 74.6 | 59.4 | 85.0 | <0.001 | |||
| 2339 | 1 | 0 | 52 | 34 | 33.6 | 91.5 | 66.7 | 73.3 | <0.001 | |||
| 2631 | 5 | 44 | 85 | 66 | 66.4 | 77.9 | 60.2 | 82.2 | <0.001 | node 3 | ||
| 2668 | 17 | 44 | 67 | 42 | 42.1 | 72.3 | 43.3 | 71.3 | <0.001 | |||
| 3034 | 1 | 24 | 6 | 16 | 15.9 | 95.8 | 65.4 | 69.4 | <0.001 | [10] † | ||
| 5509 | 5 | 0 | 0 | 20 | 19.6 | 96.2 | 72.4 | 70.4 | <0.001 | [26] † | ||
| Combined Peaks | Database Set | Test Set | |||
|---|---|---|---|---|---|
| MRSA (%) | MSSA (%) | MRSA (%) | MSSA (%) | ||
| 4-peak determination: one or more peaks at m/z 1975, 2410, 3890, and 5541 (+) | 96.2 | 45.8 | 75.8 | 53.5 | |
| 4-peak determination with | 2194 (-) | 80.8 | 74.8 | 65.3 | 61.6 |
| 2230 (-) | 86.9 | 67.3 | 63.2 | 69.8 | |
| 2339 (-) | 88.3 | 67.3 | 67.4 | 58.1 | |
| 2630 (-) | 76.1 | 79.4 | 62.1 | 66.3 | |
| Number of Isolates | SE (%) | SP (%) | PPV (%) | NPV (%) | |
|---|---|---|---|---|---|
| Database set | 320 | 96.5 | 73.0 | 90.7 | 88.5 |
| Test set | 181 | 87.6 | 71.4 | 78.0 | 83.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, J.-M.; Kim, I.; Chung, S.H.; Chung, Y.; Han, M.; Kim, J.-S. Rapid Discrimination of Methicillin-Resistant Staphylococcus aureus by MALDI-TOF MS. Pathogens 2019, 8, 214. https://doi.org/10.3390/pathogens8040214
Kim J-M, Kim I, Chung SH, Chung Y, Han M, Kim J-S. Rapid Discrimination of Methicillin-Resistant Staphylococcus aureus by MALDI-TOF MS. Pathogens. 2019; 8(4):214. https://doi.org/10.3390/pathogens8040214
Chicago/Turabian StyleKim, Jung-Min, Inhee Kim, Sung Hee Chung, Yousun Chung, Minje Han, and Jae-Seok Kim. 2019. "Rapid Discrimination of Methicillin-Resistant Staphylococcus aureus by MALDI-TOF MS" Pathogens 8, no. 4: 214. https://doi.org/10.3390/pathogens8040214
APA StyleKim, J.-M., Kim, I., Chung, S. H., Chung, Y., Han, M., & Kim, J.-S. (2019). Rapid Discrimination of Methicillin-Resistant Staphylococcus aureus by MALDI-TOF MS. Pathogens, 8(4), 214. https://doi.org/10.3390/pathogens8040214





