Use of Proteomics in the Study of Mastitis in Ewes
Abstract
1. Introduction
2. Use of Proteomics in the Study of Milk Production in Healthy Animals
2.1. General Considerations
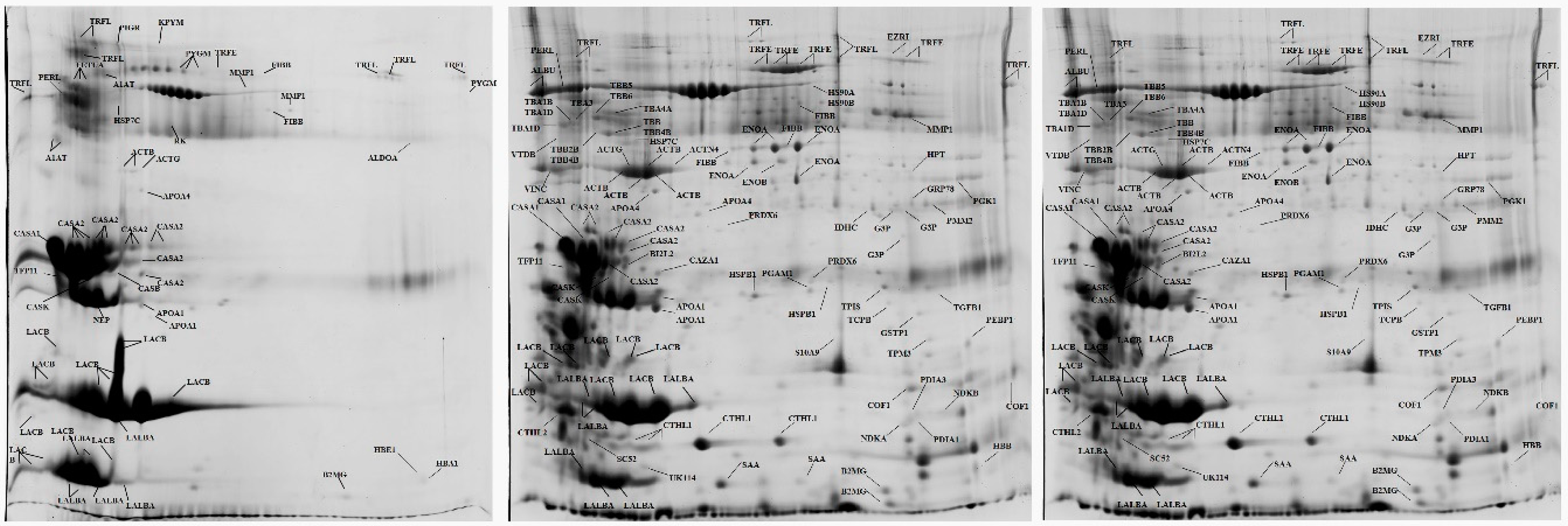
2.2. Appraisal of Proteomics Studies in Milk
3. Proteomics Studies in Mastitis in Sheep—Significance of Findings for the Study of the Infection
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Katsafadou, A.I. Proteomic Study of Ovine Mastitis Associated with Mannheimia haemolytica. Ph.D. Thesis, University of Thessaly, Karditsa, Greece, 2017. [Google Scholar]
- Lérias, J.R.; Hernández-Castellano, L.E.; Suárez-Trujillo, A.; Castro, N.; Pourlis, A.; Almeida, A.M. The mammary gland in small ruminants: Major morphological and functional events underlying milk production-a review. J. Dairy Res. 2014, 81, 304–318. [Google Scholar] [CrossRef] [PubMed]
- Athanasiadou, S.; Huntley, J.F. Emerging technologies and their applications in interactions between nutrition and immunity to gastrointestinal parasites in sheep. Parasite Immunol. 2008, 30, 101–111. [Google Scholar] [CrossRef] [PubMed]
- Gingras, A.C.; Gstaiger, M.; Raught, B.; Aebersold, R. Analysis of protein complexes using mass spectrometry. Nat. Rev. Mol. Cell Biol. 2007, 8, 645–654. [Google Scholar] [CrossRef] [PubMed]
- Witze, E.S.; Old, W.M.; Resing, K.A.; Ahn, N.G. Mapping protein post-translational modifications with mass spectrometry. Nat. Meth. 2007, 4, 798–806. [Google Scholar] [CrossRef] [PubMed]
- Anagnostopoulos, A.K.; Katsafadou, A.I.; Pierros, V.; Kontopodis, E.; Fthenakis, G.C.; Arsenos, G.; Karkabounas, S.C.; Tzora, A.; Skoufos, I.; Tsangaris, G.T. Milk of Greek sheep and goat breeds; characterization by means of proteomics. J. Proteom. 2016, 147, 76–84. [Google Scholar] [CrossRef] [PubMed]
- Katsafadou, A.I. From veterinary science to ‘Veterinomics’. SM J. Bioinform Proteom. 2016, 1, 1003. [Google Scholar]
- Boyce, J.D.; Cullen, P.A.; Nguyen, V.; Wilkie, I.; Adler, B. Analysis of the Pasteurella multocida outer membrane sub-proteome and its response to the in vivo environment of the natural host. Proteomics 2006, 6, 870–880. [Google Scholar] [CrossRef]
- Lippolis, J.D.; Peterson-Burch, B.D.; Reinhardt, T.A. Differential expression analysis of proteins from neutrophils in the periparturient period and neutrophils from dexamethasone treated dairy cows. Vet. Immunol. Immunopathol. 2006, 111, 149–164. [Google Scholar] [CrossRef]
- Vasileiou, N.G.C.; Cripps, P.J.; Ioannidi, K.S.; Chatzopoulos, D.C.; Gougoulis, D.A.; Sarrou, S.; Orfanou, D.C.; Politis, A.; Calvo Gonzalez-Valerio, T.; Argyros, S.; et al. Extensive countrywide field investigation of subclinical mastitis in sheep in Greece. J. Dairy Sci. 2018, 101, 7297–7310. [Google Scholar] [CrossRef]
- Las Heras, A.; Dominguez, L.; Fernandez-Garayzabal, J.F. Prevalence and aetiology of subclinical mastitis in dairy ewes of the Madrid region. Small Rumin. Res. 1999, 32, 21–29. [Google Scholar] [CrossRef]
- Ergun, Y.; Aslantas, O.; Dogruer, G.; Kirecci, E.; Saribay, M.K.; Ates, C.T.; Ulku, A.; Demir, C. Prevalence and etiology of subclinical mastitis in Awassi dairy ewes in southern Turkey. Turk. J. Vet. Anim. Sci. 2009, 33, 477–483. [Google Scholar]
- Gelasakis, A.I.; Mavrogianni, V.S.; Petridis, I.G.; Vasileiou, N.G.; Fthenakis, G.C. Mastitis in sheep—The last 10 years and the future of research. Vet. Microbiol. 2015, 181, 136–146. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific opinion on the welfare risks related to the farming of sheep for wool, meat and milk production. EFSA J. 2014, 12, 3933–4060. [Google Scholar]
- Gougoulis, D.A.; Kyriazakis, I.; Fthenakis, G.C. Diagnostic significance of behavioural changes in sheep health management. Small Rumin. Res. 2010, 92, 52–56. [Google Scholar] [CrossRef]
- Giadinis, N.D.; Arsenos, G.; Tsakos, P.; Psychas, V.; Dovas, C.I.; Papadopoulos, E.; Karatzias, H.; Fthenakis, G.C. “Milk-drop syndrome of ewes”: Investigation of the causes in dairy sheep in Greece. Small Rumin. Res. 2012, 106, 33–35. [Google Scholar] [CrossRef]
- Katsafadou, A.I.; Tsangaris, G.T.; Billinis, C.; Fthenakis, G.C. Use of proteomics in the study of microbial diseases of small ruminants. Vet. Microbiol. 2015, 181, 27–33. [Google Scholar] [CrossRef]
- Chen, Q.; Ke, X.; Zhang, J.S.; Lai, S.Y.; Fang, F.; Mo, W.M.; Ren, Y.P. Proteomics method to quantify the percentage of cow, goat, and sheep milks in raw materials for dairy products. J. Dairy Sci. 2016, 99, 9483–9492. [Google Scholar] [CrossRef]
- Roncada, P.; Piras, C.; Soggiu, A.; Turk, R.; Urbani, A.; Bonizzi, L. Farm animal milk proteomics. J. Proteom. 2012, 75, 4259–4274. [Google Scholar] [CrossRef]
- Casado, B.; Affolter, M.; Kussmann, M. Omics-rooted studies of milk proteins, oligosaccharides and lipids. J. Proteom. 2009, 73, 196–208. [Google Scholar] [CrossRef]
- Roncada, P.; Gaviraghi, A.; Liberatori, S.; Canas, B.; Bini, L.; Greppi, G.F. Identification of caseins in goat milk. Proteomics 2002, 2, 723–726. [Google Scholar] [CrossRef]
- Guarino, C.; Fuselli, F.; La Mantia, A.; Longo, L.; Faberi, A.; Marianella, R.M. Peptidomic approach, based on liquid chromatography/electrospray ionization tandem mass spectrometry, for detecting sheep’s milk in goat’s and cow’s cheeses. Rapid Commun. Mass Spectrom. 2010, 24, 705–713. [Google Scholar] [CrossRef]
- Anagnostopoulos, A.K.; Tsangaris, G.T. Feta cheese proteins: Manifesting the identity of Greece’s national treasure. Data Brief 2018, 19, 2037–2040. [Google Scholar] [CrossRef] [PubMed]
- Gagnaire, V.; Jardin, J.; Jan, G. Proteomics of milk and bacteria used in fermented dairy products: From qualitative to quantitative advances. J. Dairy Sci. 2009, 92, 811–825. [Google Scholar] [CrossRef] [PubMed]
- Mavromati, J. Brucellosis and proteomics: An approach in Albania. In Proceedings of the 2012 Meeting of Cost Action FA 1002 Farm Animal Proteomics, Algarve, Portugal, 12–13 April 2012; pp. 80–83. [Google Scholar]
- Cunsolo, V.; Muccilli, V.; Saletti, R.; Foti, S. Applications of mass spectrometry techniques in the investigation of milk proteome. Eur. J. Mass Spectrom. 2011, 17, 305–320. [Google Scholar] [CrossRef] [PubMed]
- Signorelli, F.; Cifuni, G.F.; Miarelli, M. Differentially expressed mammary proteins during lactation in dairy sheep. Livest. Sci. 2012, 149, 224–231. [Google Scholar] [CrossRef]
- Ha, M.; Sabherwal, M.; Duncan, E.; Stevens, S.; Stockwell, P.; McConnell, M.; Bekhit, A.-D.; Carne, A. In-depth characterization of sheep (Ovis aries) milk whey proteome and comparison with cow (Bos taurus). PLoS ONE 2015, 10, e0139774. [Google Scholar] [CrossRef] [PubMed]
- Anagnostopoulos, A.K.; Katsafadou, A.I.; Pierros, V.; Kontopodis, E.; Fthenakis, G.C.; Arsenos, G.; Karkabounas, S.C.; Tzora, A.; Skoufos, I.; Tsangaris, G.T. Dataset of milk whey proteins of three indigenous Greek sheep breeds. Data Brief 2016, 8, 877–880. [Google Scholar] [CrossRef]
- Beddek, A.J.; Rawson, P.; Peng, L.; Snell, R.; Lehnert, K.; Ward, H.E.; Jordan, T.W. Profiling the metabolic proteome of bovine mammary tissue. Proteomics 2008, 8, 1502–1515. [Google Scholar] [CrossRef]
- Peng, L.F.; Rawson, P.; McLauchlan, D.; Lehnert, K.; Snell, R.; Lordan, T.W. Proteomic analysis of microsomes from lactating bovine mammary gland. J. Proteome Res. 2008, 7, 1427–1432. [Google Scholar] [CrossRef]
- Rawson, P.; Stockum, C.; Peng, L.; Manivannan, B.; Lehnert, K.; Ward, H.E.; Berry, S.D.; Davis, S.R.; Snell, R.G.; McLauchlan, D.; et al. Metabolic proteomics of the liver and mammary gland during lactation. J. Proteom. 2012, 75, 4429–4435. [Google Scholar] [CrossRef]
- Addis, M.F.; Pisanu, S.; Ghisaura, S.; Pagnozzi, D.; Marogna, G.; Tanca, A.; Biosa, G.; Cacciotto, C.; Alberti, A.; Pittau, M.; et al. Proteomics and pathway analyses of the milk fat globule in sheep naturally infected by Mycoplasma agalactiae provide indications of the in vivo response of the mammary epithelium to bacterial infection. Infect. Immun. 2011, 79, 3833–3845. [Google Scholar] [CrossRef]
- Pisanu, S.; Ghisaura, S.; Pagnozzi, D.; Biosa, G.; Tanca, A.; Roggio, T.; Uzzau, S.; Addis, M.F. The sheep milk fat globule membrane proteome. J. Proteom. 2011, 74, 350–358. [Google Scholar] [CrossRef] [PubMed]
- Pisanu, S.; Ghisaura, S.; Pagnozzi, D.; Falchi, G.; Biosa, G.; Tanca, A.; Roggio, T.; Uzzau, S.; Addis, M.F. Characterization of sheep milk fat globule proteins by two-dimensional polyacrylamide gel electrophoresis/mass spectrometry and generation of a reference map. Int. Dairy J. 2012, 24, 78–86. [Google Scholar] [CrossRef]
- Chiaradia, E.; Valiani, A.; Tartaglia, M.; Scoppetta, F.; Renzone, G.; Arena, S.; Avellini, L.; Benda, S.; Gaiti, A.; Scaloni, A. Ovine subclinical mastitis: Proteomic analysis of whey and milk fat globules unveils putative diagnostic biomarkers in milk. J. Proteom. 2013, 83, 144–159. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, R.; Holland, J.W.; Deeth, H.C.; Alewood, P. Milk proteomics. Int. Dairy J. 2004, 14, 1013–1023. [Google Scholar] [CrossRef]
- Bianchi, L.; Puglia, M.; Landi, C.; Matteoni, S.; Perini, D.; Armini, A.; Verani, M.; Trombetta, C.; Soldani, P.; Roncada, P.; et al. Solubilization methods and reference 2-DE map of cow milk fat globules. J. Proteom. 2009, 72, 853–864. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Castellano, L.; Almeida, A.M.; Castro, N.; Arguello, A. The colostrum proteome, ruminant nutrition and immunity: A review. Curr. Protein Pept. Sci. 2014, 15, 64–74. [Google Scholar] [CrossRef]
- Hernandez-Castellano, L.; Almeida, A.; Ventosa, M.; Coelho, A.; Castro, N.; Arguello, A. The effect of colostrum intake on blood plasma proteome profiles in newborn lambs: Low abundance proteins. BMC Vet. Res. 2014, 10, 85. [Google Scholar] [CrossRef]
- Scumaci, D.; Trimboli, F.; Dell’Aquila, L.; Concolino, A.; Pappaianni, G.; Tammè, L.; Vignola, G.; Luciani, A.; Morelli, D.; Cuda, G.; et al. Proteomics-driven analysis of ovine whey colostrum. PLoS ONE 2015, 10, e0117433. [Google Scholar] [CrossRef]
- Hernández-Castellano, L.E.; Argüello, A.; Almeida, A.M.; Castro, N.; Bendixen, E. Colostrum protein uptake in neonatal lambs examined by descriptive and quantitative liquid chromatography-tandem mass spectrometry. J. Dairy Sci. 2015, 98, 135–147. [Google Scholar] [CrossRef]
- Calvano, C.D.; De Ceglie, C.; Monopoli, A.; Zambonin, C.G. Detection of sheep and goat milk adulterations by direct MALDI-TOF MS analysis of milk tryptic digests. J. Mass Spectrom. 2012, 47, 1141–1149. [Google Scholar] [CrossRef] [PubMed]
- Le Maréchal, C.; Jan, G.; Even, S.; McCulloch, J.A.; Azevedo, V.; Thiéry, R.; Vautor, E.; Le Loir, Y. Development of serological proteome analysis of mastitis by Staphylococcus aureus in ewes. J. Microbiol. Methods 2009, 79, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Fthenakis, G.C.; Jones, J.E.T. The effect of inoculation of coagulase-negative staphylococci into the ovine mammary gland. J. Comp. Pathol. 1990, 102, 211–219. [Google Scholar] [CrossRef]
- Le Maréchal, C.; Jardin, J.; Jan, G.; Even, S.; Pullido, C.; Guibert, J.M.; Hernandez, D.; Francois, P.; Scherenzel, J.; Demon, D.; et al. Staphylococcus aureus seroproteomes discriminate ruminant isolates causing mild or severe mastitis. BMC Vet. Res. 2011, 42, 35. [Google Scholar] [CrossRef] [PubMed]
- Seyffert, N.; Le Maréchal, C.; Jardin, J.; McCulloch, J.A.; Rosado, F.R.; Miyoshi, A.; Even, S.; Jan, G.; Berkova, N.; Vautor, E.; et al. Staphylococcus aureus proteins differentially recognized by the ovine immune response in mastitis or nasal carriage. Vet. Microbiol. 2012, 157, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Eckersall, P.D.; Young, F.J.; McComb, C.; Hogarth, C.J.; Safi, S.; Weber, A.; McDonald, T.; Nolan, A.M.; Fitzpatrick, J.L. Acute phase proteins in serum and milk from dairy cows with clinical mastitis. Vet. Rec. 2001, 148, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Eckersall, P.D.; Young, F.J.; Nolan, A.M.; Knight, C.H.; McComb, C.; Waterston, M.M.; Hogarth, C.J.; Scott, E.M.; Fitzpatrick, J.L. Acute phase proteins in bovine milk in an experimental model of Staphylococcus aureus subclinical mastitis. J. Dairy Sci. 2006, 89, 1488–1501. [Google Scholar] [CrossRef]
- Smolenski, G.; Haines, S.; Kwan, F.Y.S.; Bond, J.; Farr, V.; Davis, S.R.; Stelwagen, Κ.; Wheeler, Τ.Τ. Characterisation of host defense proteins in milk using a proteomic approach. J. Proteome Res. 2007, 6, 207–215. [Google Scholar] [CrossRef]
- Katsafadou, A.I.; Tsangaris, G.T.; Anagnostopoulos, A.K.; Billinis, C.; Barbagianni, M.S.; Vasileiou, N.G.C.; Spanos, S.A.; Mavrogianni, V.S.; Fthenakis, G.C. Differential quantitative proteomics study of experimental Mannheimia haemolytica mastitis in sheep. J. Proteom. 2019, 205, 103393. [Google Scholar] [CrossRef]
- Addis, M.F.; Pisanu, S.; Marogna, G.; Cubeddu, T.; Pagnozzi, D.; Cacciotto, C.; Campesi, F.; Schianchi, G.; Rocca, S.; Uzzau, S. Production and release of antimicrobial and immune defense proteins by mammary epithelial cells following Streptococcus uberis infection of sheep. Infect. Immun. 2013, 81, 3182–3197. [Google Scholar] [CrossRef]
- Pisanu, S.; Cubeddu, T.; Pagnozzi, D.; Rocca, S.; Cacciotto, C.; Alberti, A.; Marogna, G.; Uzzau, S.; Addis, M.F. Neutrophil extracellular traps in sheep mastitis. Vet. Res. 2015, 46, 59. [Google Scholar] [CrossRef] [PubMed]
- Mehrzad, J.; Desrosiers, C.; Lauzon, K.; Robitaille, G.; Zhao, X.; Lacasse, P. Proteases involved in mammary tissue damage during endotoxin-induced mastitis in dairy cows. J. Dairy Sci. 2005, 88, 211–222. [Google Scholar] [CrossRef]
- Lutzow, Y.C.S.; Donaldson, L.; Gray, C.P.; Vuocolo, T.; Pearson, R.D.; Reverter, A.; Byrne, K.A.; Sheehy, P.A.; Windon, R.; Tellam, R.L. Identification of immune genes and proteins involved in the response of bovine mammary tissue to Staphylococcus aureus infection. BMC Vet. Res. 2008, 4, 18. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, R.L.; Sainz, R.D. Energy partitioning and modeling in animal nutrition. Annu. Rev. Nutr. 1995, 15, 191–211. [Google Scholar] [CrossRef] [PubMed]
- Sandberg, F.B.; Emmans, G.C.; Kyriazakis, I. The effects of pathogen challenges on the performance of naïve and immune animals: The problem of prediction. Animal 2007, 1, 67–86. [Google Scholar] [CrossRef] [PubMed]
- Fragkou, I.A.; Skoufos, J.; Cripps, P.J.; Kyriazakis, I.; Papaioannou, N.; Boscos, C.M.; Tzora, A.; Fthenakis, G.C. Differences in susceptibility to Mannheimia haemolytica-associated mastitis between two breeds of dairy sheep. J. Dairy Res. 2007, 74, 349–355. [Google Scholar] [CrossRef] [PubMed]
- Vasileiou, N.G.C.; Gougoulis, D.A.; Riggio, V.; Ioannidi, K.S.; Chatzopoulos, D.C.; Mavrogianni, V.S.; Petinaki, E.; Fthenakis, G.C. Association of subclinical mastitis prevalence with sheep breeds in Greece. J. Dairy Res. 2018, 85, 317–320. [Google Scholar] [CrossRef] [PubMed]
- Addis, M.F.; Tedde, V.; Dore, S.; Pisanu, S.; Puggioni, G.M.G.; Roggio, A.M.; Pagnozzi, D.; Lollai, S.; Cannas, E.A.; Uzzau, S. Evaluation of milk cathelicidin for detection of dairy sheep mastitis. J. Dairy Sci. 2016, 99, 6446–6456. [Google Scholar] [CrossRef] [PubMed]
- Cubeddu, T.; Cacciotto, C.; Pisanu, S.; Tedde, V.; Alberti, A.; Pittau, M.; Dore, S.; Cannas, A.; Uzzau, S.; Rocca, S.; et al. Cathelicidin production and release by mammary epithelial cells during infectious mastitis. Vet. Immunol. Immunopathol. 2017, 189, 66–70. [Google Scholar] [CrossRef]
- Miglio, A.; Moscati, L.; Fruganti, G.; Pela, M.; Scoccia, E.; Valiani, A.; Maresca, C. Use of milk amyloid A in the diagnosis of subclinical mastitis in dairy ewes. J. Dairy Res. 2013, 80, 496–502. [Google Scholar] [CrossRef]
- Steffen, P.; Kwiatkowski, M.; Robertson, W.D.; Zarrine-Afsar, A.; Deterra, D.; Richter, V.; Schlüter, H. Protein species as diagnostic markers. J. Proteom. 2016, 34, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Smith, L.M.; Kelleher, N.L. Proteoforms as the next proteomics currency. Science 2018, 359, 1106–1107. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Katsafadou, A.I.; Vasileiou, N.G.C.; Fthenakis, G.C. Use of Proteomics in the Study of Mastitis in Ewes. Pathogens 2019, 8, 134. https://doi.org/10.3390/pathogens8030134
Katsafadou AI, Vasileiou NGC, Fthenakis GC. Use of Proteomics in the Study of Mastitis in Ewes. Pathogens. 2019; 8(3):134. https://doi.org/10.3390/pathogens8030134
Chicago/Turabian StyleKatsafadou, Angeliki I., Natalia G. C. Vasileiou, and George C. Fthenakis. 2019. "Use of Proteomics in the Study of Mastitis in Ewes" Pathogens 8, no. 3: 134. https://doi.org/10.3390/pathogens8030134
APA StyleKatsafadou, A. I., Vasileiou, N. G. C., & Fthenakis, G. C. (2019). Use of Proteomics in the Study of Mastitis in Ewes. Pathogens, 8(3), 134. https://doi.org/10.3390/pathogens8030134





