In Vitro Evaluation of Combined Commercialized Ophthalmic Solutions Against Acanthamoeba Strains
Abstract
1. Introduction
2. Results
2.1. In Vitro Effect of Systane® Ultra and Naviblef® Daily Care Against the Trophozoite Stage of Acanthamoeba Strains
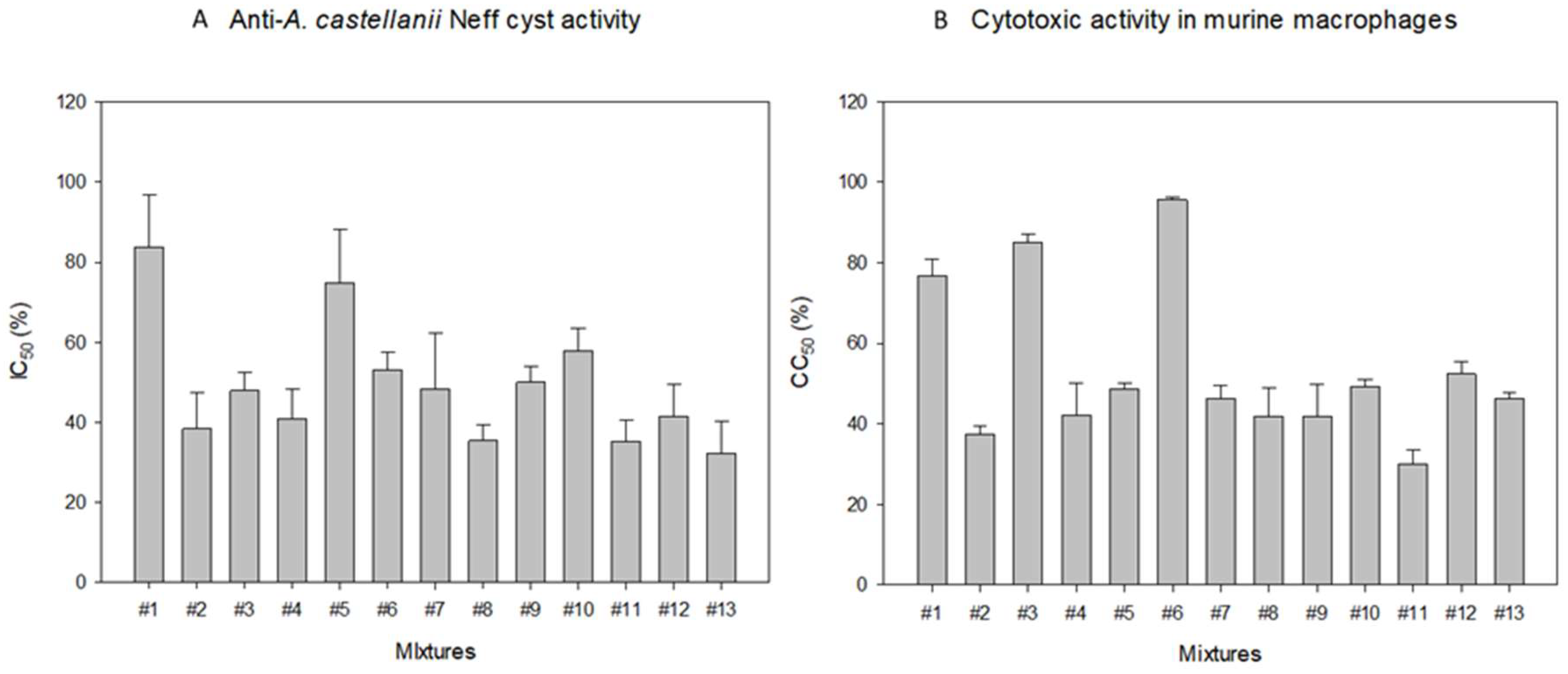
2.2. In Vitro Effect of Systane® Ultra and Naviblef® Daily Care Against the Cyst Stage of A. Castellanii Neff Strain
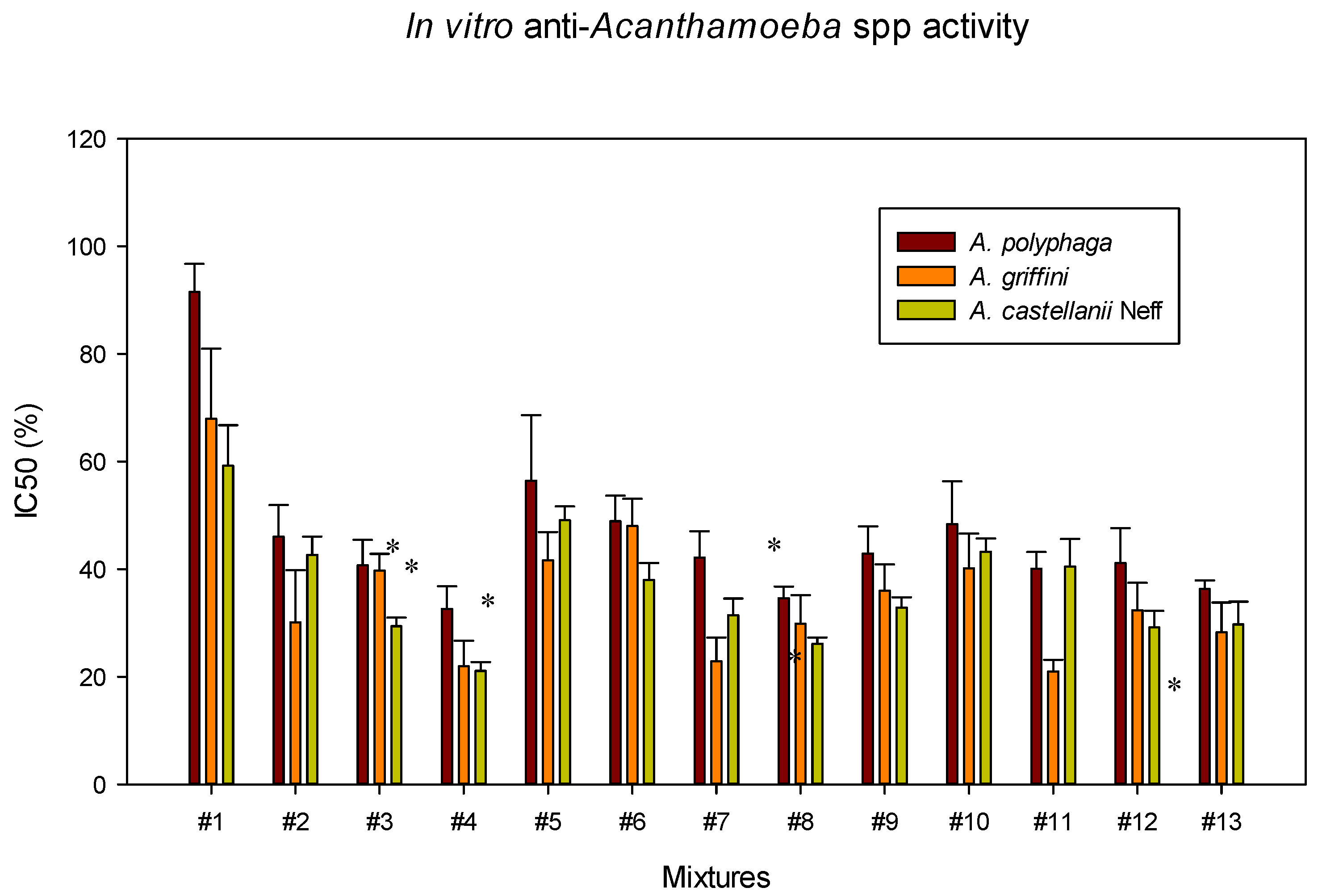
2.3. In Vitro Effect of Systane® Ultra and Naviblef® Daily Care Mixtures
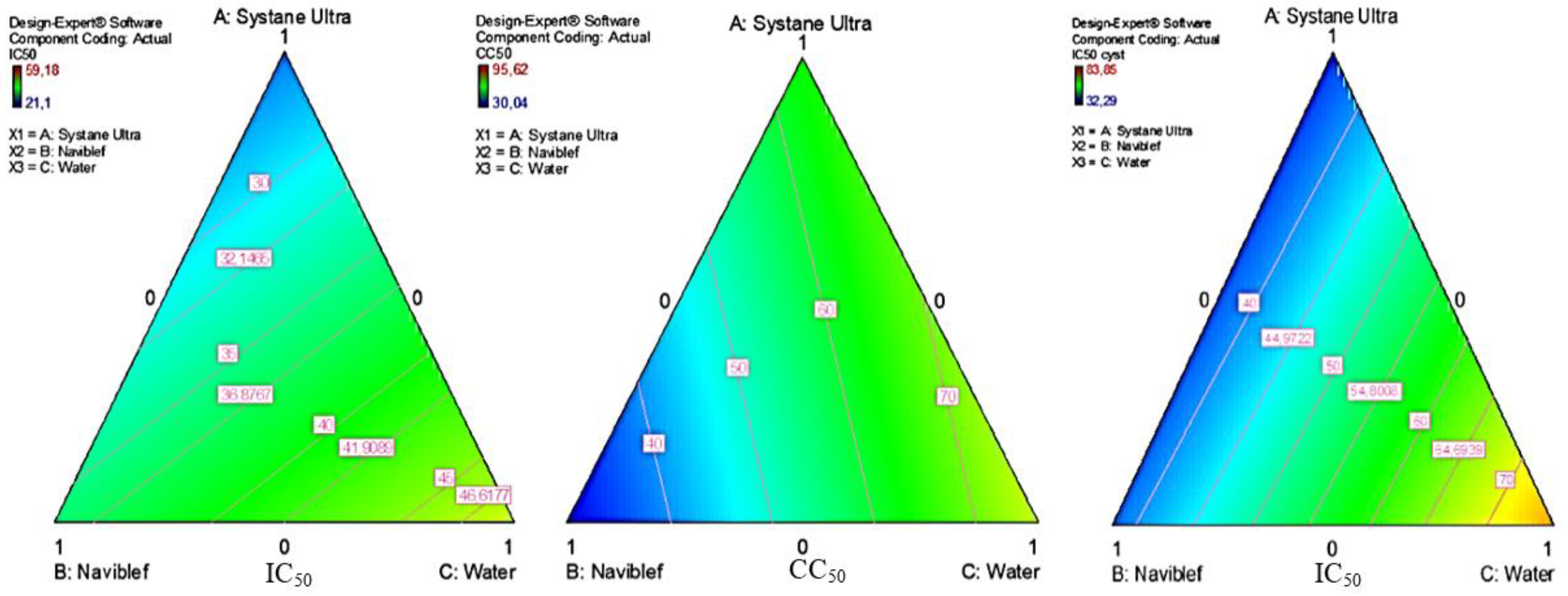
2.4. Response Surface Analysis
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Cell Cultures
4.2.1. Acanthamoeba Strains
4.2.2. Macrophage Cell Line
4.3. Anti-Acanthamoeba Assays
4.3.1. In Vitro Effect of Systane® Ultra and Naviblef® Daily Care Against the Trophozoite Stage of Acanthamoeba Strains
4.3.2. In Vitro Effect of Systane® Ultra and Naviblef® Daily Care Against the Cyst Stage of A. castellanii Neff Strain
4.3.3. In Vitro Effect Against Acanthamoeba spp. Trophozoites of Systane® Ultra and Naviblef® Daily Care Mixtures
4.4. In Vitro Effect of Systane® Ultra and Naviblef® Daily Care Mixtures Against Acanthamoeba castellanii Neff Cysts Evaluated by the Alamarblue™ Colorimetric Assay
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Siddiqui, R.; Khan, N.A. Biology and pathogenesis of Acanthamoeba. Parasites Vectors 2012, 5, 6. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo-Morales, J.; Khan, N.A.; Walochnik, J. An update on Acanthamoeba keratitis: Diagnosis, pathogenesis and treatment. Parasite 2015, 22, 10. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.A. Acanthamoeba biology and increasing importance in human health. FEMS Microbiol. Rev. 2006, 30, 564–595. [Google Scholar] [CrossRef] [PubMed]
- Visvesvara, G.S.; Moura, H.; Schuster, F.L. Pathogenic and opportunistic free-living amoebae: Acanthamoeba spp., Balamuthia mandrillaris, Naegleria fowleri, and Sappinia diploidea. FEMS Immunol. Med. Microbiol. 2007, 50, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo-Morales, J.; Martín-Navarro, C.M.; López-Arencibia, A.; Arnalich-Montiel, F.; Piñero, J.E.; Valladares, B. Acanthamoeba keratitis: An emerging disease gathering importance worldwide? Trends Parasitol. 2013, 29, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Trabelsi, H.; Dendana, F.; Sellami, A.; Sellami, H.; Cheikhrouhou, F.; Neji, S.; Makni, F.; Ayadi, A. Pathogenic free-living amoebae: Epidemiology and clinical review. Pathol. Biol. 2012, 60, 399–405. [Google Scholar] [CrossRef]
- Martín-Navarro, C.M.; Lorenzo-Morales, J.; Cabrera-Serra, M.G.; Rancel, F.; Coronado-Álvarez, N.M.; Piñero, J.E.; Valladares, B. The potential pathogenicity of chlorhexidine-sensitive Acanthamoeba strains isolated from contact lens cases from asymptomatic individuals in Tenerife, Canary Islands, Spain. J. Med. Microbiol. 2008, 57, 1399–1404. [Google Scholar] [CrossRef]
- Martín-Navarro, C.M.; Lorenzo-Morales, J.; Machin, R.P.; López-Arencibia, A.; García-Castellano, J.M.; de Fuentes, I.; Loftus, B.; Maciver, S.K.; Valladares, B.; Piñero, J.E. Inhibition of HMG-CoA reductase and the application of statins as a novel effective therapeutic approach against Acanthamoeba infections. Antimicrob. Agents Chemother. 2013, 57, 375–381. [Google Scholar] [CrossRef]
- Cabello-Vílchez, A.M.; Martín-Navarro, C.M.; López-Arencibia, A.; Reyes-Batlle, M.; Sifaoui, I.; Valladares, B.; Piñero, J.E.; Lorenzo-Morales, J. Voriconazole as a first-line treatment against potentially pathogenic Acanthamoeba strains from Peru. Parasitol. Res. 2014, 113, 755–759. [Google Scholar] [CrossRef]
- Rocha-Cabrera, P.; Reyes-Batlle, M.; Martín-Navarro, C.M.; Dorta-Gorrín, A.; López-Arencibia, A.; Sifaoui, I.; Martínez-Carretero, E.; Piñero, J.E.; Martín-Barrera, F.; Valladares, B.; et al. Detection of Acanthamoeba on the ocular surface in a Spanish population using the Schirmer strip test: Pathogenic potential, molecular classification and evaluation of the sensitivity to chlorhexidine and voriconazole of the isolated Acanthamoeba strains. J. Med. Microbiol. 2015, 64, 849–853. [Google Scholar] [CrossRef]
- Montiel, F.; Martín-Navarro, C.M.; Alió, J.L.; López-Vélez, R.; Martínez-Carretero, E.; Valladares, B.; Piñero, J.E.; Lorenzo-Morales, J. Successful monitoring and treatment of intraocular dissemination of Acanthamoeba. Arch. Ophthalmol. 2012, 130, 1474–1475. [Google Scholar] [CrossRef] [PubMed]
- Hammersmith, K.M. Diagnosis and management of Acanthamoeba keratitis. Curr. Opin. Ophthalmol. 2006, 7, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Niyyati, M.; Dodangeh, S.; Lorenzo-Morales, J. A Review of the Current Research Trends in the Application of Medicinal Plants as a Source for Novel Therapeutic Agents Against Acanthamoeba Infections. Iran. J. Pharm. Res. 2016, 5, 893–900. [Google Scholar]
- Lin, T.Y.; Yeh, L.K.; Ma, D.H.; Chen, P.Y.; Lin, H.C.; Sun, C.C.; Tan, H.Y.; Chen, H.C.; Chen, S.Y.; Hsiao, C.H. Risk factors and microbiological features of patients hospitalized for microbial keratitis: A 10-year study in a referral center in Taiwan. Medicine 2015, 94, e1905. [Google Scholar] [CrossRef] [PubMed]
- Torkildsen, G.; Brujic, M.; Cooper, M.S.; Karpecki, P.; Majmudar, P.; Trattler, W.; Reis, M.; Ciolino, J.B. Evaluation of a new artificial tear formulation for the management of tear film stability and visual function in patients with dry eye. Clin. Ophthalmol. 2017, 11, 1883–1889. [Google Scholar] [CrossRef] [PubMed]
- Sifaoui, I.; Reyes-Batlle, M.; López-Arencibia, A.; Chiboub, O.; Rodríguez-Martín, J.; Rocha-Cabrera, P.; Valladares, B.; Piñero, J.E.; Lorenzo-Morales, J. Toxic effects of selected proprietary dry eye drops on Acanthamoeba. Sci. Rep. 2018, 8, 8520. [Google Scholar] [CrossRef]
- Paimela, T.; Ryhanen, T.; Kauppinen AMarttila, L.; Salminen, A.; Kaarniranta, K. The preservative polyquaternium-1 increases cytoxicity and NF-kappaB linked inflammation in human corneal epithelial cells. Mol. Vis. 2012, 18, 1189–1196. [Google Scholar]
- Chirife, J.; Herszage, L.; Joseph, A.; Bozzini, J.P.; Leardini, N.; Kohn, E.S. In vitro antibacterial activity of concentrated polyethylene glycol 400 solutions. Antimicrob. Agents Chemother. 1983, 24, 409–412. [Google Scholar] [CrossRef]
- Bozzini, J.P.; Kohn, E.S.; Joseph, A.; Herszage, L.; Chirife, J. Submicroscopical changes in Klebsiella pneumoniae cells treated with concentrated sucrose and polyethylene glycol 400 solutions. J. Appl. Microbiol. 1986, 60, 375–379. [Google Scholar]
- Kinnunen, T.; Koskela, M. Antibacterial and antifungal properties of propylene glycol, hexylene glycol, and 1, 3-butylene glycol in vitro. Acta Derm.-Venereol. 1991, 71, 148–150. [Google Scholar]
- Nalawade, T.M.; Bhat, K.; Sogi, S.H. Bactericidal activity of propylene glycol, glycerine, polyethylene glycol 400, and polyethylene glycol 1000 against selected microorganisms. J. Int. Soc. Prevent. Communit. Dent. 2015, 5, 114. [Google Scholar] [CrossRef] [PubMed]
- Hajaji, S.; Sifaoui, I.; López-Arencibia, A.; Reyes-Batlle, M.; Valladares, B.; Pinero, J.E.; Lorenzo-Morales, J.; Akkari, H. Amoebicidal activity of α-bisabolol, the main sesquiterpene in chamomile (Matricaria recutita L.) essential oil against the trophozoite stage of Acanthamoeba castellanii Neff. Acta Parasitol. 2017, 62, 290–295. [Google Scholar] [CrossRef] [PubMed]
- Hajaji, S.; Sifaoui, I.; López-Arencibia, A.; Reyes-Batlle, M.; Jiménez, I.A.; Bazzocchi, I.L.; Valladares, B.; Pinero, J.E.; Lorenzo-Morales, J.; Akkari, H. Correlation of radical-scavenging capacity and amoebicidal activity of Matricaria recutita L. (Asteraceae). Exp. Parasitol. 2017, 183, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Su, C.-W.; Tighe, S.; Sheha, H.; Cheng, A.M.S.; Tsenget, S.C.G. Safety and efficacy of 4-terpineol against microorganisms associated with blepharitis and common ocular diseases. BMJ Open Ophthalmol. 2018, 3, e000094. [Google Scholar] [CrossRef] [PubMed]
- González-Robles, A.; Salazar-Villatoro, L.; Omaña-Molina, M.; Reyes-Batlle, M.; Martín-Navarro, C.M.; Lorenzo-Morales, J. Morphological features and in vitro cytopathic effect of Acanthamoeba griffini trophozoites isolated from a clinical case. J. Parasitol. Res. 2014. [Google Scholar] [CrossRef] [PubMed]
- Sifaoui, I.; Reyes-Batlle, M.; López-Arencibia, A.; Wagner, C.; Chiboub, O.; De Agustino Rodríguez, J.; Rocha-Cabrera, P.; Valladares, B.; Piñero, J.E.; Lorenzo-Morales, J. Evaluation of the anti-Acanthamoeba activity of two commercial eye drops commonly used to lower eye pressure. Exp. Parasitol. 2017, 183, 117–123. [Google Scholar] [CrossRef]
- Lorenzo-Morales, J.; Kliescikova, J.; Martinez-Carretero, E.; De Pablos, L.M.; Profotova, B.; Nohynkova, E.; Osuna, A.; Valladares, B. Glycogen phosphorylase in Acanthamoeba spp.: Determining the role of the enzyme during the encystment process. Eukaryot. Cell 2008, 7, 509–517. [Google Scholar] [CrossRef]
- Marciano-Cabral, F.; Cabral, G. Acanthamoeba spp. as agents of disease in humans. Clin. Microbiol. Rev. 2003, 16, 273–307. [Google Scholar] [CrossRef]
| IC50 (%) | Trophozoites | Cysts | ||
|---|---|---|---|---|
| A. Castellanii Neff | A. Polyphaga | A. Griffini | A. Castellanii Neff | |
| Systane® Ultra | 4.97 ± 0.71 | 5.98 ± 0.60 | 7.99 ± 1.25 | 34.6 ± 1.38 |
| Naviblef® Daily Care | 0.68 ± 0.16 | 0.98 ± 0.28 | 1.13 ± 0.16 | 2.1 ± 0.04 |
| Mixture | IC50 (%) A. Castellanii Neff Trophozoites | Systane® Ultra Proportion (%) | Naviblef® Proportion (%) |
|---|---|---|---|
| #1 | 59.18 ± 7.56 | 0.59 | 0.06 |
| #2 | 42.67 ± 3.36 | 0.43 | 0.43 |
| #3 | 29.39 ± 1.57 | 3.53 | 0.03 |
| #4 | 21.10 ± 1.61 | 2.53 | 0.21 |
| #5 | 49.07 ± 2.56 | 0.49 | 0.27 |
| #6 | 37.96 ± 3.16 | 2.47 | 0.04 |
| #7 | 31.46 ± 3.07 | 2.05 | 0.31 |
| #8 | 26.12 ± 1.17 | 3.13 | 0.14 |
| #9 | 32.85 ± 1.91 | 2.13 | 0.18 |
| #10 | 43.23 ± 2.45 | 1.62 | 0.14 |
| #11 | 40.49 ± 5.11 | 1.52 | 0.31 |
| #12 | 29.16 ± 3.05 | 2.70 | 0.10 |
| #13 | 29.72 ± 4.23 | 2.75 | 0.23 |
| Systane® Ultra | Naviblef® |
|---|---|
| Polyetilene Glycol 400 Propylene Glycol Hidroxipropilguar Sorbitol Aminomethyl Propanol Boric acid Potassium chloride Sodium chloride POLYQUAD®(polidronium chloride) 0.001% as preservative. | Melaleuca alternifolia (tea tree oil, TTO) Anthemis nobilis (chamomile oil) D-Panthenol Alantoine Taurine Purified water |
| Mixture | Systane® Ultra (%) | Naviblef® (%) | Sterile Water (%) |
|---|---|---|---|
| #1 | 0.0100 | 0.0010 | 0.9890 |
| #2 | 0.0100 | 0.0100 | 0.9800 |
| #3 | 0.1200 | 0.0010 | 0.8790 |
| #4 | 0.1200 | 0.0100 | 0.8700 |
| #5 | 0.0100 | 0.0055 | 0.9845 |
| #6 | 0.0650 | 0.0010 | 0.9340 |
| #7 | 0.0650 | 0.0100 | 0.9250 |
| #8 | 0.1200 | 0.0055 | 0.8745 |
| #9 | 0.0650 | 0.0055 | 0.9295 |
| #10 | 0.0375 | 0.0033 | 0.9593 |
| #11 | 0.0375 | 0.0078 | 0.9547 |
| #12 | 0.0925 | 0.0033 | 0.9043 |
| #13 | 0.0925 | 0.0078 | 0.8998 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reyes-Batlle, M.; Mura-Escorche, G.; Sifaoui, I.; Otero-Ruiz, A.; Alfaro-Sifuentes, R.; López-Arencibia, A.; Rocha-Cabrera, P.; Chiboub, O.; Rizo-Liendo, A.; Zamora-Herrera, J.; et al. In Vitro Evaluation of Combined Commercialized Ophthalmic Solutions Against Acanthamoeba Strains. Pathogens 2019, 8, 109. https://doi.org/10.3390/pathogens8030109
Reyes-Batlle M, Mura-Escorche G, Sifaoui I, Otero-Ruiz A, Alfaro-Sifuentes R, López-Arencibia A, Rocha-Cabrera P, Chiboub O, Rizo-Liendo A, Zamora-Herrera J, et al. In Vitro Evaluation of Combined Commercialized Ophthalmic Solutions Against Acanthamoeba Strains. Pathogens. 2019; 8(3):109. https://doi.org/10.3390/pathogens8030109
Chicago/Turabian StyleReyes-Batlle, María, Glorian Mura-Escorche, Ines Sifaoui, Alejandro Otero-Ruiz, Rosalía Alfaro-Sifuentes, Atteneri López-Arencibia, Pedro Rocha-Cabrera, Olfa Chiboub, Aitor Rizo-Liendo, Jonadab Zamora-Herrera, and et al. 2019. "In Vitro Evaluation of Combined Commercialized Ophthalmic Solutions Against Acanthamoeba Strains" Pathogens 8, no. 3: 109. https://doi.org/10.3390/pathogens8030109
APA StyleReyes-Batlle, M., Mura-Escorche, G., Sifaoui, I., Otero-Ruiz, A., Alfaro-Sifuentes, R., López-Arencibia, A., Rocha-Cabrera, P., Chiboub, O., Rizo-Liendo, A., Zamora-Herrera, J., Bethencourt-Estrella, C. J., Rodríguez-Expósito, R. L., San Nicolás-Hernández, D., Piñero, J. E., & Lorenzo-Morales, J. (2019). In Vitro Evaluation of Combined Commercialized Ophthalmic Solutions Against Acanthamoeba Strains. Pathogens, 8(3), 109. https://doi.org/10.3390/pathogens8030109








