An Update on Sexual Transmission of Zika Virus
Abstract
1. Introduction
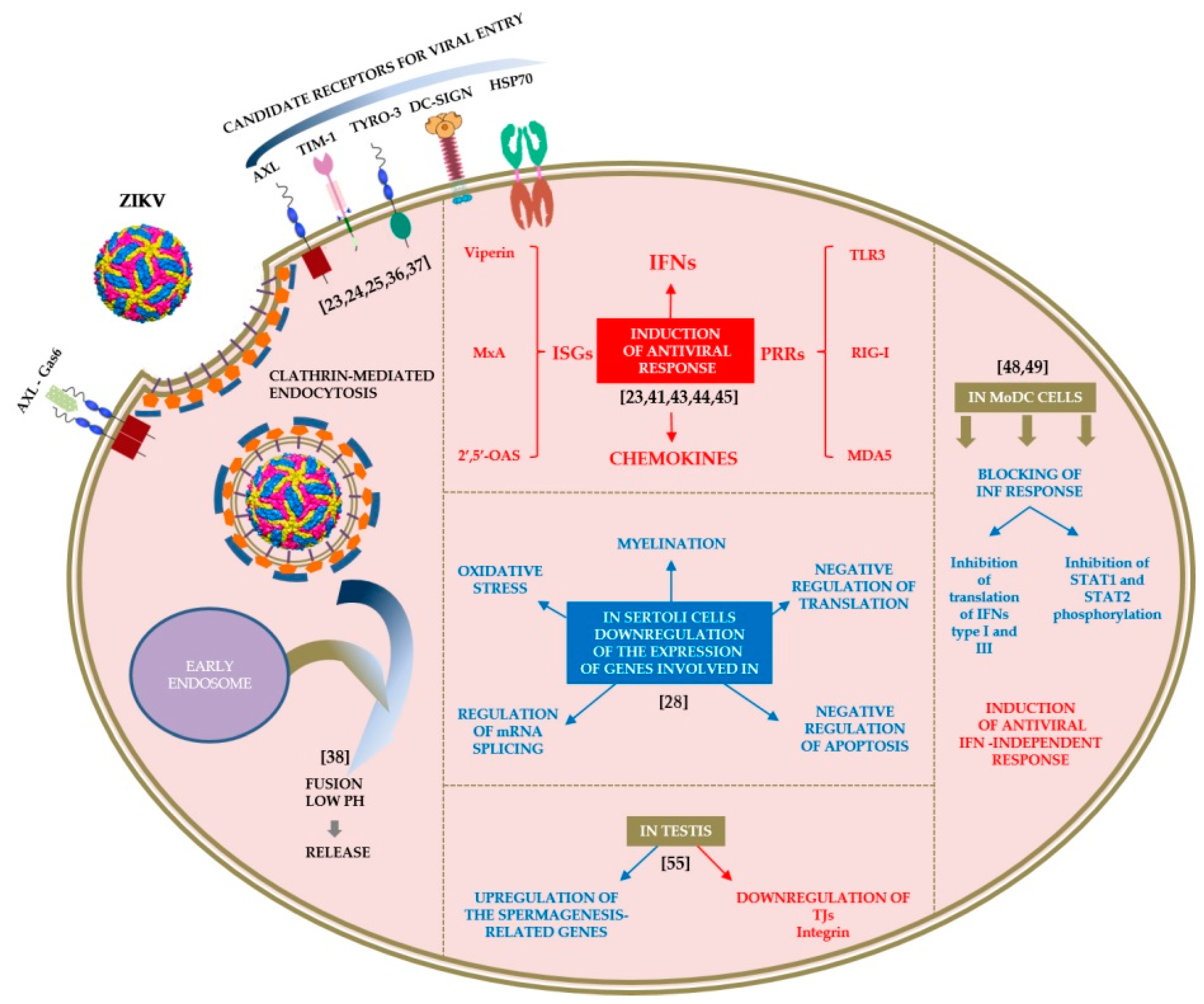
2. Host–Virus Interactions
2.1. Tropism
2.2. Cell Interactions
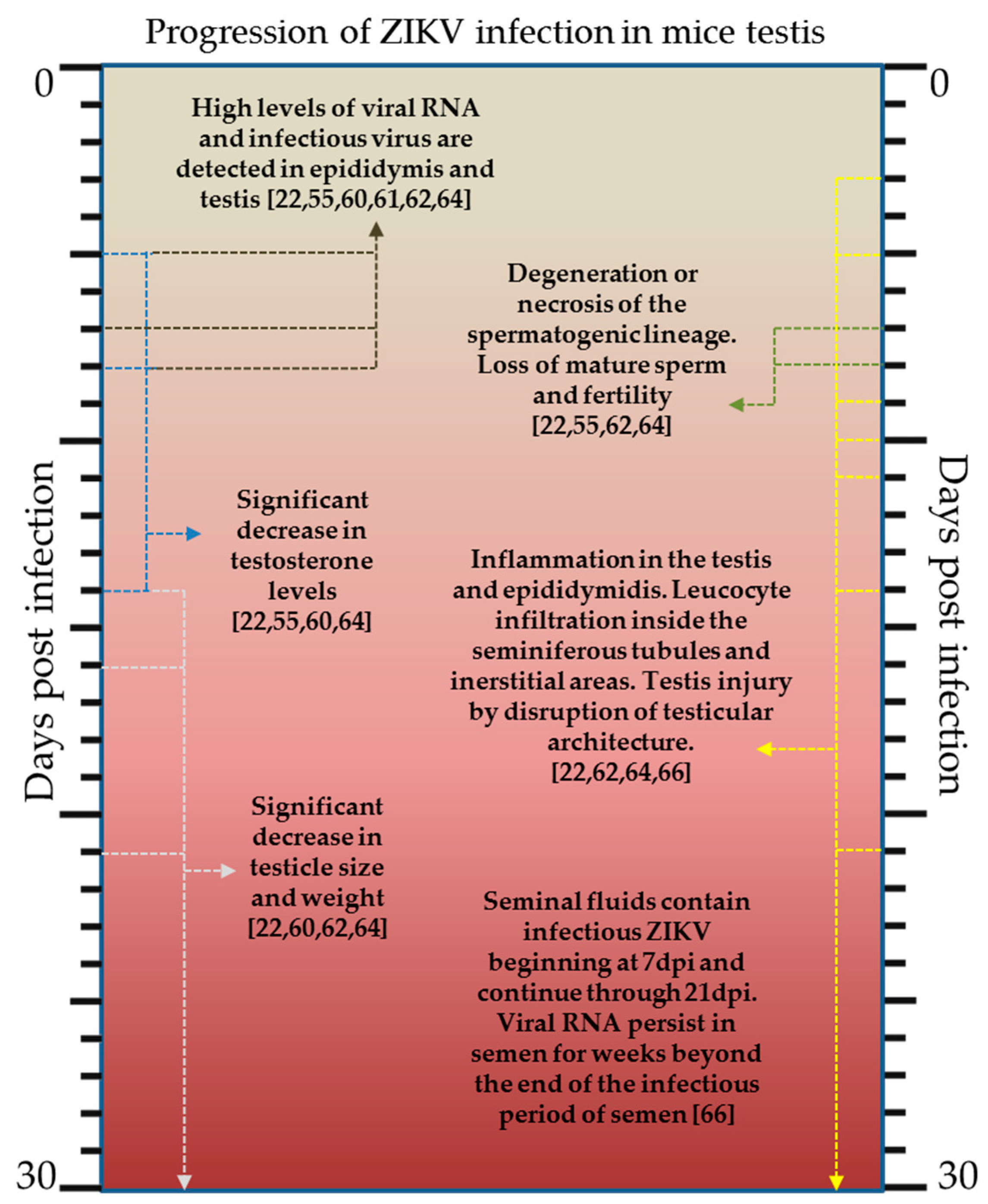
2.3. Laboratory Animals
3. Epidemiology
4. Clinical and Laboratory Findings
5. Management and Prevention
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dick, G.W.; Kitchen, S.F.; Haddow, A.J. Zika virus. I. Isolations and serological specificity. Trans. R. Soc. Trop. Med. Hyg. 1952, 46, 509–520. [Google Scholar] [CrossRef]
- Duffy, M.; Chen, T.H.; Hancock, T.; Powers, A.; Kool, J.; Lanciotti, R.; Pretrick, M.; Marfel, M.; Holzbauer, S.; Dubray, C.; et al. Zika virus outbreak on Yap Island, Federated States of Micronesia. N. Eng. J. Med. 2009, 360, 2536–2543. [Google Scholar] [CrossRef] [PubMed]
- Cao-Lormeau, V.M.; Roche, C.; Teissier, A.; Robin, E.; Berry, A.L.; Mallet, H.P.; Sall, A.; Musso, D. Zika virus, French Polynesia, South Pacific, 2013. Emerg. Infect. Dis. 2014, 20, 1085. [Google Scholar] [CrossRef] [PubMed]
- Lowe, R.; Barcellos, C.; Brasil, P.; Cruz, O.; Honorio, N.A.; Kuper, H.; Carvalho, M.S. The Zika virus epidemic in Brazil: From discovery to future implications. Int. J. Environ. Res. Public Health 2018, 15, 96. [Google Scholar] [CrossRef] [PubMed]
- WHO 2017. Zika Virus Country Classification Scheme: Interim Guidance, March 2017, Geneva. Available online: http://apps.who.int/iris/bitstream/handle/10665/254619/WHO-ZIKV-SUR-17.1-eng.pdf (accessed on 20 July 2018).
- Lee, I.; Bos, S.; Wang, S.; Gadea, G.; Desprès, P.; Zhao, R.Y. Probing Molecular Insights into Zika Virus-Host Interactions. Viruses 2018, 10, 233. [Google Scholar] [CrossRef] [PubMed]
- Ioos, S.; Mallet, H.P.; Leparc-Goffart, I.; Gauthier, V.; Cardoso, T.; Herida, M. Current Zika virus epidemiology and recent epidemics. Med. Mal. Infect. 2014, 44, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Oehler, E.; Watrin, L.; Larre, P.; Goffart-Leparc, I.; Lasterem, S.; Valour, F.; Baudouin, L.; Mallet, H.P.; Musso, D. Zika virus infection complicated by Guillen-Barre syndrome–case report, French Polynesia, December 2013. Eurosurveillance 2014, 19, 20720. [Google Scholar] [CrossRef] [PubMed]
- Ventura, C.; Maia, M.; Bravo-Filho, V.; Gois, A.; Belford, R. Zika virus in Brazil and macular atrophy in a child with microcephaly. Lancet 2015, 387, 228. [Google Scholar] [CrossRef]
- Sakkas, H.; Economou, V.; Papadopoulou, C. Zika virus infection: Past and present of another emerging vector-borne disease. J. Vector Borne Dis. 2016, 53, 305–311. [Google Scholar] [PubMed]
- Aliota, M.; Bassit, L.; Bradrick, S.; Cox, B.; Garcia-Blanco, M.; Gavegnano, C.; Friedrich, T.; Golos, T.; Griffin, D.; Haddow, A. Zika in the Americas, year 2: What have we learned? What gaps remain? A report from the Global Virus Network. Antivir. Res. 2017, 144, 223–246. [Google Scholar] [CrossRef] [PubMed]
- Foy, B.D.; Kobylinski, K.C.; Foy, J.L.C.; Blitvich, B.J.; Travassos da Rosa, A.; Haddow, A.D.; Lanciotti, R.; Tesh, R. Probable non-vector-borne transmission of Zika virus, Colorado, USA. Emerg. Infect. Dis. 2011, 17, 880–882. [Google Scholar] [CrossRef] [PubMed]
- Beshard, M.; Lastere, S.; Teissier, A.; Cao-Lormeau, V.; Musso, D. Evidence of perinatal transmission of Zika virus, French Polynesia, December 2013 and February 2014. Eurosurveillance 2014, 19, 13. [Google Scholar]
- Holtzman, M.; Golden, W.; Sheffield, J. Zika virus infection in the pregnant woman. Clin. Obs. Gynecol. 2018, 61, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.; Oduyebo, T.; Brault, A.; Brooks, J.; Chang, K.W.; Hills, S.; Kuehnert, M.; Mead, P.; Meaney-Delman, D.; Rabe, I.; et al. Modes of transmission of Zika virus. J. Infect. Dis. 2017, 216, S875–S883. [Google Scholar] [CrossRef] [PubMed]
- Barjas-Castro, M.; Angerami, R.; Cunha, M.; Suzuki, A.; Nogueira, J.; Rocco, I.; Maeda, A.; Vasami, F.; Katz, G.; Boin, I.; et al. Probable transfusion-transmitted Zika virus in Brazil. Transfusion 2016, 56, 1684–1688. [Google Scholar] [CrossRef] [PubMed]
- Motta, I.; Spencer, B.; Cordeiro da Silva, S.; Arruda, M.; Dobbin, J.; Gonzaga, Y.; Arcuri, I.; Tavares, R.; Atta, E.; Fernandes, R.; et al. Evidence for transmission of Zika virus by platelet transfusion. N. Engl. J. Med. 2016, 375, 1101–1103. [Google Scholar] [CrossRef] [PubMed]
- Hamel, R.; Liegeois, F.; Wichit, S.; Pompon, J.; Diop, F.; Talignani, L.; Thomas, F.; Despres, P.; Yssel, H.; Misse, D. Zika virus: Epidemiology, clinical features and host-virus interactions. Microbes Infect. 2016, 18, 441–449. [Google Scholar] [CrossRef] [PubMed]
- Grischott, F.; Puhan, M.; Hatz, C.; Schlagenhauf, P. Non-vector borne transmission of Zika-Virus: A systematic review. Travel Med. Infect. Dis. 2016, 14, 313–330. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Wilson, M.E. Update on non-vector transmission of dengue: Relevant studies with Zika and other flaviviruses. Trop. Dis. Travel Med. Vaccines 2016, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Coelho, F.C.; Durovni, B.; Saraceni, V.; Lemos, C.; Codeco, C.T.; Camargo, S.; de Carvalho, L.M.; Bastos, L.; Arduini, D.; Villela, D.A.M.; et al. Higher incidence of ZIka in adult women than adult men in Rio de Janeiro suggests a significant contribution of sexual transmission from men to women. Int. J. Infect. Dis. 2016, 51, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Govero, J.; Esakky, P.; Scheaffer, S.; Fernandez, E.; Drury, A.; Platt, D.; Gorman, M.; Richner, J.; Caine, E.; Salazar, V.; et al. Zika virus infection damages the testes in mice. Nature 2016, 540, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Hamel, R.; Dejarnac, O.; Wichit, S.; Ekchariyawat, P.; Neyret, A.; Luplertlop, N.; Perera-Lecoin, M.; Surasombatpattana, P.; Talignani, L.; Thomas, F.; et al. Biology of Zika Virus Infection in Human Skin Cells. J. Virol. 2015, 8880–8896. [Google Scholar] [CrossRef] [PubMed]
- Gavegnano, C.; Bassit, L.; Cox, B.; Hsiao, H.M.; Johnson, E.; Suthar, M.; Chakraborty, R.; Schinazi, R. Jak Inhibitors Modulate Production of Replication–Competent Zika virus in Human Hofbauer, Trophoblasts and Neuroblastoma cells. Pathog. Immun. 2017, 2, 199–218. [Google Scholar] [CrossRef] [PubMed]
- Goo, L.; DeMaso, C.; Pelc, R.; Ledgerwood, J.; Graham, B.; Kuhnc, R.; Pierson, T. The Zika virus envelope protein glycan loop regulates virion antigenicity. Virology 2018, 515, 191–202. [Google Scholar] [CrossRef] [PubMed]
- Meertens, L.; Labeau, A.; Dejarnac, O.; Cipriani, S.; Sinigaglia, L.; Bonnet-Madin, L.; Le Charpentier, T.; Hafirassou, M.L.; Zamborlini, A.; Cao-Lormeau, V.M.; et al. Axl Mediates ZIKA Virus Entry in Human Glial Cells and Modulates Innate Immune Responses. Cell Rep. 2017, 18, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; DeLalio, L.; Isakson, B.; Wang, T. AXL-Mediated Productive Infection of Human Endothelial Cells by Zika Virus. Circ. Res. 2016, 119, 1183–1189. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Jovel, J.; Lopez-Orozco, J.; Limonta, D.; Airo, A.; Hou, S.; Stryapunina, I.; Fibke, C.; Moore, R.B.; Hobman, T.C. Human Sertoli cells support high levels of Zika virus replication and persistence. Sci. Rep. 2018, 8, 5477. [Google Scholar] [CrossRef] [PubMed]
- Richard, A.S.; Shim, B.S.; Kwon, Y.C.; Zhang, R.; Otsuka, Y.; Schmitt, K.; Berri, F.; Diamond, M.S.; Choe, H. AXL-dependent infection of human fetal endothelial cells distinguishes Zika virus from other pathogenic flaviviruses. Proc. Natl. Acad. Sci. USA 2017, 114, 2024–2029. [Google Scholar] [CrossRef] [PubMed]
- Hastings, A.K.; Yockey, L.J.; Jagger, B.W.; Hwang, J.; Uraki, R.; Gaitsch, H.F.; Parnell, L.A.; Cao, B.; Mysorekar, I.; Rothlin, C.V.; et al. TAM receptors are not required for Zika virus infection in mice. Cell Rep. 2017, 19, 558–568. [Google Scholar] [CrossRef] [PubMed]
- Retallack, H.; Di Lullo, E.; Arias, C.; Knopp, K.A.; Laurie, M.T.; Sandoval-Espinosa, C.; Mancia Leon, W.R.; Krencik, R.; Ullian, E.M.; Spatazza, J.; et al. Zika virus cell tropism in the developing human brain and inhibition by azithromycin. Proc. Natl. Acad. Sci. USA 2016, 113, 14408–14413. [Google Scholar] [CrossRef] [PubMed]
- Miner, J.J.; Sene, A.; Richner, J.M.; Smith, A.M.; Santeford, A.; Ban, N.; Weger-Lucarelli, J.; Manzella, F.; Ruckert, C.; Govero, J.; et al. Zika virus infection in mice causes panuveitis with shedding of virus in tears. Cell Rep. 2016, 16, 3208–3218. [Google Scholar] [CrossRef] [PubMed]
- Wells, M.F.; Salick, M.R.; Wiskow, O.; Ho, D.J.; Worringer, K.A.; Ihry, R.J.; Kommineni, S.; Bilican, B.; Klim, J.R.; Hill, E.J.; et al. Genetic Ablation of AXL Does Not Protect Human Neural Progenitor Cells and Cerebral Organoids from Zika Virus Infection. Cell Stem Cell 2016, 19, 703–708. [Google Scholar] [CrossRef] [PubMed]
- Tabata, T.; Petitt, M.; Puerta-Guardo, H.; Michlmayr, D.; Wang, C.; Fang-Hoover, J.; Harris, E.; Pereira, L. Zika Virus Targets Different Primary Human Placental Cells, Suggesting Two Routes for Vertical Transmission. Cell Host Microb. 2016, 10, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Yang, M.; Azar, S.R.; Soong, L.; Weaver, S.C.; Sun, J.; Chen, Y.; Rossi, S.L.; Cai, J. Viral Retinopathy in Experimental Models of Zika Infection. Investig. Ophthalmol. Vis. Sci. 2017, 58, 4355–4365. [Google Scholar] [CrossRef] [PubMed]
- Shelby, T.; Banerjee, T.; Zegar, I.; Santra, S. Highly Sensitive, Engineered Magnetic Nanosensors to Investigate the Ambiguous Activity of Zika Virus and Binding Receptors. Sci. Rep. 2017, 7, 7377. [Google Scholar] [CrossRef] [PubMed]
- Bagasra, O.; Addanki, K.C.; Goodwin, G.R.; Hughes, B.W.; Pandey, P.; McLean, E. Cellular Targets and Receptor of Sexual Transmission of Zika Virus. Appl. Immunohistochem. Mol. Morphol. 2017, 25, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Persaud, M.; Martinez-Lopez, A.; Buffone, C.; Porcelli, S.A.; Diaz-Griffero, F. Infection by Zika viruses requires the transmembrane protein AXL, endocytosis and low pH. Virology 2018, 518, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Joguet, G.; Mansuy, J.M.; Hamdi, S.; Walschaerts, M.; Pavili, L.; Guyomard, S.; Prisant, N.; Lamarre, P.; Dejucq-Rainsford, N.; Pasquier, C.; et al. Effect of acute Zika virus infection on sperm and virus clearance in body fluids: A prospective observational study. Lancet Infect. Dis. 2017, 17, 1200–1208. [Google Scholar] [CrossRef]
- Wang, A.; Thurmond, S.; Islas, L.; Hui, K.; Hai, R. Zika virus genome biology and molecular pathogenesis. Emerg. Microbes Infect. 2017, 6, e13. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Xu, D.; Ye, Q.; Hong, S.; Jiang, Y.; Liu, X.; Zhang, N.; Shi, L.; Qin, C.F.; Xu, Z. Zika Virus Disrupts Neural Progenitor Development and Leads to Microcephaly in Mice. Cell Stem Cell 2016, 19, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Tang, H.; Hammack, C.; Ogden, S.C.; Wen, Z.; Qian, X.; Li, Y.; Yao, B.; Shin, J.; Zhang, F.; Lee, E.M.; et al. Zika Virus Infects Human Cortical Neural Progenitors and Attenuates Their Growth. Cell Stem Cell 2016, 18, 587–590. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Hammack, C.; Ogden, S.C.; Cheng, Y.; Lee, E.M.; Wen, Z.; Qian, X.; Nguyen, H.N.; Li, Y.; Yao, B.; et al. Molecular signatures associated with ZIKV exposure in human cortical neural progenitors. Nucleic Acids Res. 2016, 44, 8610–8620. [Google Scholar] [CrossRef] [PubMed]
- Siemann, D.N.; Strange, D.P.; Maharaj, P.N.; Shi, P.Y.; Verma, S. Zika Virus Infects Human Sertoli Cells and Modulates the Integrity of the In Vitro Blood-Testis Barrier Model. J. Virol. 2017, 27, 91. [Google Scholar] [CrossRef] [PubMed]
- Urcuqui-Inchima, S.; Cabrera, J.; Haenni, A.L. Interplay between dengue virus and Toll-like receptors, RIG-I/MDA5 and microRNAs: Implications for pathogenesis. Antivir. Res. 2017, 147, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Vielle, N.J.; Zumkehr, B.; García-Nicolás, O.; Blank, F.; Stojanov, M.; Musso, D.; Baud, D.; Summerfield, A.; Alves, M.P. Silent infection of human dendritic cells by African and Asian strains of Zika virus. Sci. Rep. 2018, 8, 5440. [Google Scholar] [CrossRef] [PubMed]
- Iversen, M.B.; Ank, N.; Melchjorsen, J.; Paludan, S.R. Expression of type III interferon (IFN) in the vaginal mucosa is mediated primarily by dendriticcells and displays stronger dependence on NF-κB than type I IFNs. J. Virol. 2010, 84, 4579–4586. [Google Scholar] [CrossRef] [PubMed]
- Bowen, J.; Quicke, K.; Maddur, M.; O’Neal, J.; McDonald, C.; Fedorova, N.; Puri, V.; Shabman, R.; Pulendran, B.; Suthar, M. Zika Virus Antagonizes Type I Interferon Responses during Infection of Human Dendritic Cells. PLoS Pathog. 2017, 13, e1006164. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Hua, S.; Chen, H.R.; Quyang, Z.; Einkauf, K.; Tse, S.; Ard, K.; Ciaranello, A.; Yawetz, S.; Sax, P.; et al. Transcriptional changes during naturally-acquired Zika Virus infection render dendritic cells highly conducive to viral replication. Cell Rep. 2017, 21, 3471–3482. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, S.; Wang, X.; Liao, S.; Wu, Y.; Han, C. Endogenously produced FGF2 is essential for the survival and proliferation of cultured mouse spermatogonial stem cells. Cell Res. 2012, 22, 773–776. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Pi, Z.; Liu, L.; Zhang, B.; Huang, X.; Hu, P.; Chevet, E.; Yi, P.; Liu, J. FGF-2 prevents cancer cells from ER stress-mediated apoptosis via enhancing proteasome-mediated Nck degradation. Biochem. J. 2013, 452, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Liang, Q.; Luo, Z.; Zeng, J.; Chen, W.; Foo, S.S.; Lee, S.A.; Ge, J.; Wang, S.; Goldman, S.A.; Zlokovic, B.V.; et al. Zika Virus NS4A and NS4B Proteins Deregulate AktmTOR Signaling in Human Fetal Neural Stem Cells to Inhibit Neurogenesis and Induce Autophagy. Cell Stem Cell 2016, 19, 663–671. [Google Scholar] [CrossRef] [PubMed]
- Heaton, N.S.; Randall, G. Dengue virus-induced autophagy regulates lipid metabolism. Cell Host Microbe 2010, 8, 422–432. [Google Scholar] [CrossRef] [PubMed]
- Sir, D.; Kuo, C.F.; Tian, Y.; Liu, H.M.; Huang, E.J.; Jung, J.U.; Machida, K.; Ou, J.H.J. Replication of hepatitis C virus RNA on autophagosomal membranes. J. Biol. Chem. 2012, 287, 18036–18043. [Google Scholar] [CrossRef] [PubMed]
- Sheng, Z.Y.; Gao, N.; Wang, Z.Y.; Cui, X.Y.; Zhou, D.S.; Fan, D.Y.; Chen, H.; Wang, P.G.; An, J. Sertoli Cells Are Susceptible to ZIKV Infection in Mouse Testis. Front. Cell. Infect. Microbiol. 2017, 21, 272. [Google Scholar] [CrossRef] [PubMed]
- Musso, D.; Roche, C.; Robin, E.; Nhan, T.; Teissier, A.; Cao-Lormeau, V.M. Potential sexual transmission of Zika virus. Emerg. Infect. Dis. 2015, 21, 359–361. [Google Scholar] [CrossRef] [PubMed]
- Deckard, D.T.; Chung, W.M.; Brooks, J.T.; Smith, J.C.; Woldai, S.; Hennessey, M.; Kwit, N.; Mead, P. Male-to-Male Sexual Transmission of Zika Virus-Texas. MMWR 2016, 65, 372–374. [Google Scholar] [PubMed]
- Grant, A.; Ponia, S.S.; Tripathi, S.; Balasubramaniam, V.; Miorin, L.; Sourisseau, M.; Schwarz, M.C.; Sánchez-Seco, M.P.; Evans, M.J.; Best, S.M.; et al. Zika Virus Targets Human STAT2 to Inhibit Type I Interferon Signaling. Cell Host Microbe 2016, 19, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Zellweger, R.; Shresta, S. Mouse Models to Study Dengue Virus Immunology and Pathogenesis. Front. Immunol. 2014, 5, 151. [Google Scholar] [CrossRef] [PubMed]
- Uraki, R.; Hwang, J.; Jurado, K.A.; Householder, S.; Vockey, L.; Hastings, A.; Homer, R.; Iwasaki, A.; Fikrig, E. Zika virus causes testicular atrophy. Sci. Adv. 2017, 3, e1602899. [Google Scholar] [CrossRef] [PubMed]
- Clancy, C.S.; Van Wettere, A.J.; Siddharthan, V.; Morrey, J.D.; Julander, J.G. Comparative histopathologic lesions of the male reproductive tract during acute Infection of Zika virus in AG129 and IFNAR−/− mice. Am. J. Pathol. 2018, 188, 904–915. [Google Scholar] [CrossRef] [PubMed]
- Griffin, B.D.; Muthumani, K.; Warner, B.; Majer, A.; Hagan, M.; Audet, J.; Stein, D.; Ranadheera, C.; Racine, T.; De La Vega, M.A.; et al. DNA vaccination protects mice against Zika virus-induced damage to the testes. Nat. Commun. 2017, 8, 15743. [Google Scholar] [CrossRef] [PubMed]
- Siddharthan, V.; Van Wettere, A.J.; Li, R.; Miao, J.; Morrey, J.D.; Julander, J.G. Zika virus infection of adult and fetal STAT2 knock-out hamsters. Virology 2017, 507, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Li, S.; Ma, S.; Jia, L.; Zhang, F.; Zhang, J.; Wong, G.; Zhang, S.; Lu, X.; Liu, M.; et al. Zika virus causes testis damage and leads to male infertility in mice. Cell 2016, 167, 1511.e10–1524.e10. [Google Scholar] [CrossRef] [PubMed]
- Duggal, N.K.; Ritter, J.M.; Pestorious, S.E.; Zaki, S.R.; Davis, B.S.; Chang, G.J.; Bowen, R.A.; Brault, A.C. Frequent Zika Virus Sexual Transmission and Prolonged Viral Shedding in an Immunodeficient Mouse Model. Cell Rep. 2017, 18, 1751–1760. [Google Scholar] [CrossRef] [PubMed]
- Morrison, T.E.; Diamond, M.S. Animal Models of Zika Virus Infection, Pathogenesis, and Immunity. J. Virol. 2017, 91, e00009-17. [Google Scholar] [CrossRef] [PubMed]
- WHO 2017. Situation Report, ZIKA Virus, Microcephaly, Guillain-Barre Syndrome, 10 March 2017. Available online: http://apps.who.int/iris/bitstream/handle/10665/254714/zikasitrep10Mar17eng.pdf (accessed on 7 May 2018).
- Depoux, A.; Philibert, A.; Rabier, S.; Philippe, H.J.; Fontanet, A.; Flahault, A. A multi-faceted pandemic: A review of the state of knowledge on the Zika virus. Public Health Rev. 2018, 39, 10. [Google Scholar] [CrossRef] [PubMed]
- Carabali, M.; Austin, N.; King, N.; Kaufman, J. The Zika epidemic and abortion in Latin America: A scoping review. Glob. Health Res. Policy 2018, 3, 15. [Google Scholar] [CrossRef] [PubMed]
- Moreira, J.; Peixoto, T.M.; Siqueira, A.M.; Lamas, S.S. Sexually acquired Zika virus: A systematic review. Clin. Microbiol. Infect. 2017, 23, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Freour, T.; Mirallie, S.; Hubert, B.; Splingart, C.; Barriere, P.; Maquart, M.; Leparc-Goffart, I. Sexual transmission of Zika virus in an entirely asymptomatic couple returning from a Zika epidemic area, France, April 2016. Eurosurveillance 2016, 21, 30254. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.W.; Young, M.P.; Mamidi, A.; Regla-Nava, J.A.; Kim, K.; Shresta, S. A mouse model of Zika virus sexual transmission and vaginal viral replication. Cell Rep. 2016, 17, 3091–3098. [Google Scholar] [CrossRef] [PubMed]
- WHO 2017. Sexual Transmission of Zika Virus: Current Status, Challenges and Research Priorities. Available online: http://apps.who.int/iris/bitstream/handle/10665/259583/WHO-RHR-17.23-eng.pdf?sequence=1. (accessed on 9 May 2018).
- Maxian, O.; Neufeld, A.; Talis, E.; Childs, L.; Blackwood, J. Zika virus dynamics: When does sexual transmission matter? Epidemics 2017, 21, 48–55. [Google Scholar] [CrossRef] [PubMed]
- CDC 2018. Zika Virus: 2017 Case Counts in the US. Available online: https://www.cdc.gov/zika/reporting/2017-case-counts.html (accessed on 9 May 2018).
- Brooks, R.; Carlos, M.P.; Myers, R.; White, M.G.; Bobo-Lenosi, T.; Aplan, D.; Blythe, D.; Feldman, K. Likely sexual transmission of Zika virus from a man with no symtoms of infection-Maryland, 2016. MMWR 2016, 65, 915–916. [Google Scholar] [PubMed]
- Prisant, N.; Bujan, L.; Benichou, H.; Hayot, P.-H.; Pavili, L.; Lurel, S.; Hermann, C.; Janky, E.; Joquet, G. Zika virus in the female genital tract. Lancet Infect. Dis. 2016, 16, 1000–1001. [Google Scholar] [CrossRef]
- Russell, K.; Hills, S.; Oster, A.; Porce, C.C.; Danyluk, G.; Cone, M.; Brooks, R.; Scotland, S.; Schiffman, E.; Fredette, C.; et al. Male-to-Female Sexual Transmission of Zika Virus—United States, January–April 2016. Clin. Infect. Dis. 2017, 64, 211–213. [Google Scholar] [CrossRef] [PubMed]
- Nicastri, E.; Castilletti, C.; Liuzzi, G.; Iannetta, M.; Capobianchi, M.R.; Ippolito, G. Persistent detection of Zika virus RNA in semen for six months after symptom onset in a traveler returning from Haiti to Italy, February 2016. Eurosurveillance 2016, 21, 30314. [Google Scholar] [CrossRef] [PubMed]
- De Laval, F.; Matheus, S.; Labrousse, T.; Enfissi, A.; Rousset, D.; Briolant, S. Kinetics of Zika viral load in semen. N. Eng. J. Med. 2017, 377, 697–699. [Google Scholar] [CrossRef] [PubMed]
- Mansuy, J.M.; Duterte, M.; Mengelle, C.; Fourcade, C.; Marchou, B.; Marchou, B.; Delobel, P.; Izopet, J.; Martin-Blondel, G. Zika virus: High infectious viral load in semen, a new sexually transmitted pathogen? Lancet Infect. Dis. 2016, 16, 405. [Google Scholar] [CrossRef]
- Arsuaga, M.; Bujalance, S.G.; Diaz-Menendez, M.; Vazquez, A.; Arribas, J.R. Probable sexual transmission from a vasectomised man. Lancet Infect. Dis. 2016, 16, 1107. [Google Scholar] [CrossRef]
- Turmel, J.M.; Abgueguen, P.; Hubert, B.; Vandamme, Y.M.; Maquart, M.; Le Guillou-Guillemette, H.; Leparc-Goffart, I. Late sexual transmission of Zika virus related to persistence in the semen. Lancet 2016, 387, 2501. [Google Scholar] [CrossRef]
- CDC 2018. Zika Virus: Sexual Transmission and Prevention. Available online: https://www.cdc.gov/zika/prevention/sexual-transmission-prevention.html (accessed on 7 May 2018).
- Zanluca, C.; dos Santos, C.N.D. Zika virus-an overview. Microbes Infect. 2016, 18, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Dhama, K.; Khandia, R.; Munjal, A.; Karthik, K.; Tiwari, R.; Chakraborty, S.; Malik, Y.S.; Bueno-Marí, R. Prevention and Control Strategies to Counter Zika Virus, a Special Focus on Intervention Approaches against Vector Mosquitoes—Current Updates. Front. Microbiol. 2018, 9, 87. [Google Scholar] [CrossRef] [PubMed]
- Rather, I.A.; Kumar, S.; Bajpai, V.K.; Lim, J.; Park, Y.H. Prevention and control strategies to counter Zika epidemic. Front. Microbiol. 2017, 8, 305. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Dhama, K.; Malik, Y.S.; Ramakrishnan, M.A.; Karthik, K.; Tiwari, R.; Saurabh, S.; Sachan, S.; Joshi, S.K. Zika virus–emergence, evolution, pathology, diagnosis, and control: Current global scenario and future perspectives—A comprehensive review. Vet. Q. 2016, 36, 150–175. [Google Scholar] [CrossRef] [PubMed]
- Mead, P.S.; Hills, S.L.; Brooks, J.T. Zika virus as a sexually transmitted pathogen. Curr. Opin. Infect. Dis. 2017, 31, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Bloch, E.M.; Ness, P.M.; Tobian, A.A.R.; Sugarman, J. Revisiting Blood Safety Practices Given Emerging Data about Zika Virus. N. Engl. J. Med. 2018, 378, 1837–1841. [Google Scholar] [CrossRef] [PubMed]
- Guerra-Reyes, L.; Iguiñiz-Romero, R.A. Performing purity: Reproductive decision-making and implications for a community under threat of Zika in Iquitos. Peru. Cult. Health Sex. 2018. [Google Scholar] [CrossRef] [PubMed]
- Richner, J.M.; Diamond, M.S. Zika virus vaccines: Immune response, current status, and future challenges. Curr. Opin. Immunol. 2018, 53, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Wilder-Smith, A.; Vannice, K.; Durbin, A.; Hombach, J.; Thomas, S.J.; Thevarjan, I.; Simmons, C.P. Zika vaccines and therapeutics: Landscape analysis and challenges ahead. BMC Med. 2018, 16, 84. [Google Scholar] [CrossRef] [PubMed]
- WHO 2018. International Clinical Trials Registry Platform (ICTRP). Available online: http://www.who.int/ictrp/en/ (accessed on 27 June 2018).
- WHO 2018. WHO Vaccine Pipeline Tracker. Available online: http://www.who.int/immunization/research /vaccine_pipeline_tracker_spreadsheet/en/ (accessed on 27 June 2018).
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sakkas, H.; Bozidis, P.; Giannakopoulos, X.; Sofikitis, N.; Papadopoulou, C. An Update on Sexual Transmission of Zika Virus. Pathogens 2018, 7, 66. https://doi.org/10.3390/pathogens7030066
Sakkas H, Bozidis P, Giannakopoulos X, Sofikitis N, Papadopoulou C. An Update on Sexual Transmission of Zika Virus. Pathogens. 2018; 7(3):66. https://doi.org/10.3390/pathogens7030066
Chicago/Turabian StyleSakkas, Hercules, Petros Bozidis, Xenofon Giannakopoulos, Nikolaos Sofikitis, and Chrissanthy Papadopoulou. 2018. "An Update on Sexual Transmission of Zika Virus" Pathogens 7, no. 3: 66. https://doi.org/10.3390/pathogens7030066
APA StyleSakkas, H., Bozidis, P., Giannakopoulos, X., Sofikitis, N., & Papadopoulou, C. (2018). An Update on Sexual Transmission of Zika Virus. Pathogens, 7(3), 66. https://doi.org/10.3390/pathogens7030066






