Multifocal Equine Influenza Outbreak with Vaccination Breakdown in Thoroughbred Racehorses
Abstract
:1. Introduction
2. Results
2.1. Overview
2.2. Diagnosis
2.3. Factors Contributing to Disease Spread
2.4. Serological Data
Virus Characterisation
3. Discussion
4. Materials and Methods
4.1. Sample Collection and Clinical Histories
4.2. RNA Extraction and Real-Time RT-PCR
4.3. Equine Influenza Antibody Detection and Quantification
4.4. Virus Isolation
4.5. Haemagglutinin and Neuraminidase Gene Sequencing
4.6. Genetic Sequence Analysis
4.7. Antigenic Characterisation
4.8. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bryant, N.A.; Rash, A.S.; Russell, C.A.; Ross, J.; Cooke, A.; Bowman, S.; MacRae, S.; Lewis, N.S.; Paillot, R.; Zanoni, R.; et al. Antigenic and genetic variations in European and North American equine influenza virus strains (H3N8) isolated from 2006 to 2007. Vet. Microbiol. 2009, 138, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Bryant, N.A.; Rash, A.S.; Woodward, A.L.; Medcalf, E.; Helwegen, M.; Wohlfender, F.; Cruz, F.; Herrmann, C.; Borchers, K.; Tiwari, A.; et al. Isolation and characterisation of equine influenza viruses (H3N8) from Europe and North America from 2008 to 2009. Vet. Microbiol. 2011, 147, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Quinlivan, M.; Arkins, S.; Cullinane, A. The molecular epidemiology of equine influenza in Ireland from 2007–2010 and its international significance. Equine Vet. J. 2012, 44, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Fitzpatrick, D.A.; Cullinane, A. Epidemiological and virological investigations of equine influenza outbreaks in Ireland (2010–2012). Influ. Other Respir. Viruses 2013, 7 (Suppl. 4), 61–72. [Google Scholar] [CrossRef] [PubMed]
- Woodward, A.L.; Rash, A.S.; Blinman, D.; Bowman, S.; Chambers, T.M.; Daly, J.M.; Damiani, A.; Joseph, S.; Lewis, N.; McCauley, J.W.; et al. Development of a surveillance scheme for equine influenza in the UK and characterisation of viruses isolated in Europe, Dubai and the USA from 2010–2012. Vet. Microbiol. 2014, 169, 113–127. [Google Scholar] [CrossRef] [PubMed]
- Legrand, L.J.; Pitel, P.H.; Cullinane, A.A.; Fortier, G.D.; Pronost, S.L. Genetic evolution of equine influenza strains isolated in France from 2005 to 2010. Equine Vet. J. 2015, 47, 207–211. [Google Scholar] [CrossRef] [PubMed]
- Back, H.; Berndtsson, L.T.; Grondahl, G.; Stahl, K.; Pringle, J.; Zohari, S. The first reported Florida clade 1 virus in the Nordic countries, isolated from a Swedish outbreak of equine influenza in 2011. Vet. Microbiol. 2016, 184, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Rash, A.; Morton, R.; Woodward, A.; Maes, O.; McCauley, J.; Bryant, N.; Elton, D. Evolution and divergence of H3N8 equine influenza viruses circulating in the United Kingdom from 2013 to 2015. Pathogens 2017, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Newton, J.R.; Daly, J.M.; Spencer, L.; Mumford, J.A. Description of the outbreak of equine influenza (H3N8) in the United Kingdom in 2003, during which recently vaccinated horses in Newmarket developed respiratory disease. Vet. Record 2006, 158, 185–192. [Google Scholar] [CrossRef]
- Martella, V.; Elia, G.; Decaro, N.; Di Trani, L.; Lorusso, E.; Campolo, M.; Desario, C.; Parisi, A.; Cavaliere, N.; Buonavoglia, C. An outbreak of equine influenza virus in vaccinated horses in Italy is due to an H3N8 strain closely related to recent North American representatives of the Florida sub-lineage. Vet. Microbiol. 2007, 121, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, T.; Niwa, H.; Tsujimura, K.; Kondo, T.; Matsumura, T. Epidemic of equine influenza among vaccinated racehorses in Japan in 2007. J. Vet. Med. Sci. 2008, 70, 623–625. [Google Scholar] [CrossRef] [PubMed]
- Barbic, L.; Madic, J.; Turk, N.; Daly, J. Vaccine failure caused an outbreak of equine influenza in Croatia. Vet. Microbiol. 2009, 133, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Perglione, C.O.; Gildea, S.; Rimondi, A.; Mino, S.; Vissani, A.; Carossino, M.; Cullinane, A.; Barrandeguy, M. Epidemiological and virological findings during multiple outbreaks of equine influenza in South America in 2012. Influ. Other Respir. Viruses 2016, 10, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Heine, H.G.; Trinidad, L.; Selleck, P.; Lowther, S. Rapid detection of highly pathogenic avian influenza H5N1 virus by TaqMan reverse transcriptase-polymerase chain reaction. Avian Dis. 2007, 51, 370–372. [Google Scholar] [CrossRef] [PubMed]
- Quinlivan, M.; Dempsey, E.; Ryan, F.; Arkins, S.; Cullinane, A. Real-time reverse transcription PCR for detection and quantitative analysis of equine influenza virus. J. Clin. Microbiol. 2005, 43, 5055–5057. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Arkins, S.; Cullinane, A. Management and environmental factors involved in equine influenza outbreaks in Ireland 2007–2010. Equine Vet. J. 2011, 43, 608–617. [Google Scholar] [CrossRef] [PubMed]
- OIE Report 2015. Available online: http://www.oie.int/wahis_2/public/wahid.php/Reviewreport/Review?reportid=18572 (accessed on 9 April 2018).
- Lai, A.C.; Chambers, T.M.; Holland, R.E., Jr.; Morley, P.S.; Haines, D.M.; Townsend, H.G.; Barrandeguy, M. Diverged evolution of recent equine-2 influenza (H3N8) viruses in the western hemisphere. Arch. Virol. 2001, 146, 1063–1074. [Google Scholar] [CrossRef] [PubMed]
- Karamendin, K.; Kydyrmanov, A.; Kasymbekov, Y.; Khan, E.; Daulbayeva, K.; Asanova, S.; Zhumatov, K.; Seidalina, A.; Sayatov, M.; Fereidouni, S.R. Continuing evolution of equine influenza virus in central Asia, 2007–2012. Arch. Virol. 2014, 159, 2321–2327. [Google Scholar] [CrossRef] [PubMed]
- World Organisation for Animal Health (OIE). Equine influenza vaccine composition—Conclusions and recommendations. Bull. Off. Int. Epizoot. 2015, 2, 53–55. [Google Scholar]
- Van Maanen, C.; Cullinane, A. Equine influenza virus infections: An update. Vet. Q. 2002, 24, 79–94. [Google Scholar] [CrossRef] [PubMed]
- Burrows, R.; Denyer, M.; Goodridge, D.; Hamilton, F. Field and laboratory studies of equine influenza viruses isolated in 1979. Vet. Rec. 1981, 109, 353–356. [Google Scholar] [CrossRef] [PubMed]
- Cowled, B.; Ward, M.P.; Hamilton, S.; Garner, G. The equine influenza epidemic in Australia: Spatial and temporal descriptive analyses of a large propagating epidemic. Prev. Vet. Med. 2009, 92, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.S.; Williams, C.C.; Meinkoth, J.H.; Malayer, J.R.; Royer, C.M.; Williamson, K.K.; McKenzie, E.C. Influx of neutrophils and persistence of cytokine expression in airways of horses after performing exercise while breathing cold air. Am. J. Vet. Res. 2007, 68, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Folsom, R.W.; Littlefield-Chabaud, M.A.; French, D.D.; Pourciau, S.S.; Mistric, L.; Horohov, D.W. Exercise alters the immune response to equine influenza virus and increases susceptibility to infection. Equine Vet. J. 2001, 33, 664–669. [Google Scholar] [CrossRef] [PubMed]
- Newton, J.R.; Wood, J.L.; Jessett, D.; Yates, P.J.; Mumford, J.A. ‘Cross-protection’ and ‘cross-reaction’ with equine influenza vaccines. Vet. Rec. 1999, 145, 647. [Google Scholar] [PubMed]
- Daly, J.M.; Yates, R.J.; Browse, G.; Swann, Z.; Newton, J.R.; Jessett, D.; Davis-Poynter, N.; Mumford, J.A. Comparison of hamster and pony challenge models for evaluation of effect of antigenic drift on cross protection afforded by equine influenza vaccines. Equine Vet. J. 2003, 35, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Cullinane, A.; Elton, D.; Mumford, J. Equine influenza—Surveillance and control. Influ. Other Respir. Viruses 2010, 4, 339–344. [Google Scholar] [CrossRef] [PubMed]
- World Organisation for Animal Health (OIE). Expert Surveillance panel on equine influenza vaccine composition—Conclusions and recommendations. Bull. Off. Int. Epizoot. 2010, 2, 44–45. [Google Scholar]
- Paillot, R.; Rash, N.L.; Garrett, D.; Prowse-Davis, L.; Montesso, F.; Cullinane, A.; Lemaitre, L.; Thibault, J.C.; Wittreck, S.; Dancer, A. How to meet the last OIE expert surveillance panel recommendations on equine influenza (EI) vaccine composition: A review of the process required for the recombinant canarypox-based EI vaccine. Pathogens 2016, 5, 64. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, T.; Cullinane, A.; Gildea, S.; Bannai, H.; Nemoto, M.; Tsujimura, K.; Kondo, T.; Matsumura, T. The potential impact of a single amino-acid substitution on the efficacy of equine influenza vaccines. Equine Vet. J. 2015, 47, 456–462. [Google Scholar] [CrossRef] [PubMed]
- Van Oirschot, J.T.; Bruin, G.; de Boer-Luytze, E.; Smolders, G. Maternal antibodies against equine influenza virus in foals and their interference with vaccination. Zentralblatt Vet. Reihe B J. Vet. Med. Ser. B 1991, 38, 391–396. [Google Scholar] [CrossRef]
- Van Maanen, C.; Bruin, G.; de Boer-Luijtze, E.; Smolders, G.; de Boer, G.F. Interference of maternal antibodies with the immune response of foals after vaccination against equine influenza. Vet. Q. 1992, 14, 13–17. [Google Scholar] [CrossRef] [PubMed]
- Cullinane, A.; Weld, J.; Osborne, M.; Nelly, M.; McBride, C.; Walsh, C. Field studies on equine influenza vaccination regimes in Thoroughbred foals and yearlings. Vet. J. 2001, 161, 174–185. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Arkins, S.; Cullinane, A. A comparative antibody study of the potential susceptibility of Thoroughbred and non-Thoroughbred horse populations in Ireland to equine influenza virus. Influenza Other Respir. Viruses 2010, 4, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Arkins, S.; Walsh, C.; Cullinane, A. A comparison of antibody responses to commercial equine influenza vaccines following primary vaccination of thoroughbred weanlings—A randomised blind study. Vaccine 2011, 29, 9214–9223. [Google Scholar] [CrossRef] [PubMed]
- Gildea, S.; Arkins, S.; Walsh, C.; Cullinane, A. A comparison of antibody responses to commercial equine influenza vaccines following annual booster vaccination of national hunt horses—A randomised blind study. Vaccine 2011, 29, 3917–3922. [Google Scholar] [CrossRef] [PubMed]
- Cullinane, A.; Gildea, S.; Weldon, E. Comparison of primary vaccination regimes for equine influenza: Working towards an evidence-based regime. Equine Vet. J. 2014, 46, 669–673. [Google Scholar] [CrossRef] [PubMed]
- Park, A.W.; Wood, J.L.; Newton, J.R.; Daly, J.; Mumford, J.A.; Grenfell, B.T. Optimising vaccination strategies in equine influenza. Vaccine 2003, 21, 2862–2870. [Google Scholar] [CrossRef]
- Foord, A.J.; Selleck, P.; Colling, A.; Klippel, J.; Middleton, D.; Heine, H.G. Real-time RT-PCR for detection of equine influenza and evaluation using samples from horses infected with A/Equine/Sydney/2007 (H3N8). Vet. Microbiol. 2009, 137, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Read, A.J.; Finlaison, D.S.; Gu, X.; Davis, R.J.; Arzey, K.E.; Kirkland, P.D. Application of real-time PCR and ELISA assays for equine influenza virus to determine the duration of viral RNA shedding and onset of antibody response in naturally infected horses. Aust. Vet. J. 2011, 89 (Suppl. 1), 42–43. [Google Scholar] [CrossRef] [PubMed]
- Mumford, J.A.; Wood, J. Establishing an acceptability threshold for equine influenza vaccines. Dev. Biol. Stand. 1992, 79, 137–146. [Google Scholar] [PubMed]
- Newton, J.R.; Townsend, H.G.; Wood, J.L.; Sinclair, R.; Hannant, D.; Mumford, J.A. Immunity to equine influenza: Relationship of vaccine-induced antibody in young Thoroughbred racehorses to protection against field infection with influenza A/equine-2 viruses (H3N8). Equine Vet. J. 2000, 32, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Daly, J.; Daas, A.; Behr-Gross, M.E. Collaborative study for the establishment of a candidate equine influenza subtype 2 American-like strain A/EQ/South Africa/4/03—Horse antiserum biological reference preparation. Pharmeuropa Bio 2007, 2007, 7–14. [Google Scholar] [PubMed]
- Mumford, J.A. Biology, epidemiology and vaccinology of equine influenza. In Proceedings of the International Symposium, Budapest, Hungary, 10–11 December 2001; pp. 7–12. [Google Scholar]
- Morley, P.S.; Hanson, L.K.; Bogdan, J.R.; Townsend, H.G.; Appleton, J.A.; Haines, D.M. The relationship between single radial hemolysis, hemagglutination inhibition, and virus neutralization assays used to detect antibodies specific for equine influenza viruses. Vet. Microbiol. 1995, 45, 81–92. [Google Scholar] [CrossRef]
- Yamagishi, H.; Nagamine, T.; Shimoda, K.; Ide, S.; Igarashi, Y.; Yoshioka, I.; Matumoto, M. Comparative measurement of equine influenza virus antibodies in horse sera by single radial hemolysis, neutralization, and hemagglutination inhibition tests. J. Clin. Microbiol. 1982, 15, 660–662. [Google Scholar] [PubMed]
- Spackman, E.; Senne, D.A.; Myers, T.J.; Bulaga, L.L.; Garber, L.P.; Perdue, M.L.; Lohman, K.; Daum, L.T.; Suarez, D.L. Development of a real-time reverse transcriptase PCR assay for type A influenza virus and the avian H5 and H7 hemagglutinin subtypes. J. Clin. Microbiol. 2002, 40, 3256–3260. [Google Scholar] [CrossRef] [PubMed]
- World Organisation for Animal Health (OIE). Chapter 2.5.7 Equine Influenza. In Manual of Diagnostic Tests and Vaccines for Terrestrial Animals; OIE: Paris, France, 2016; pp. 1–16. [Google Scholar]
- Rash, A.; Woodward, A.; Bryant, N.; McCauley, J.; Elton, D. An efficient genome sequencing method for equine influenza [H3N8] virus reveals a new polymorphism in the PA-X protein. Virol. J. 2014, 11, 159. [Google Scholar] [CrossRef] [PubMed]
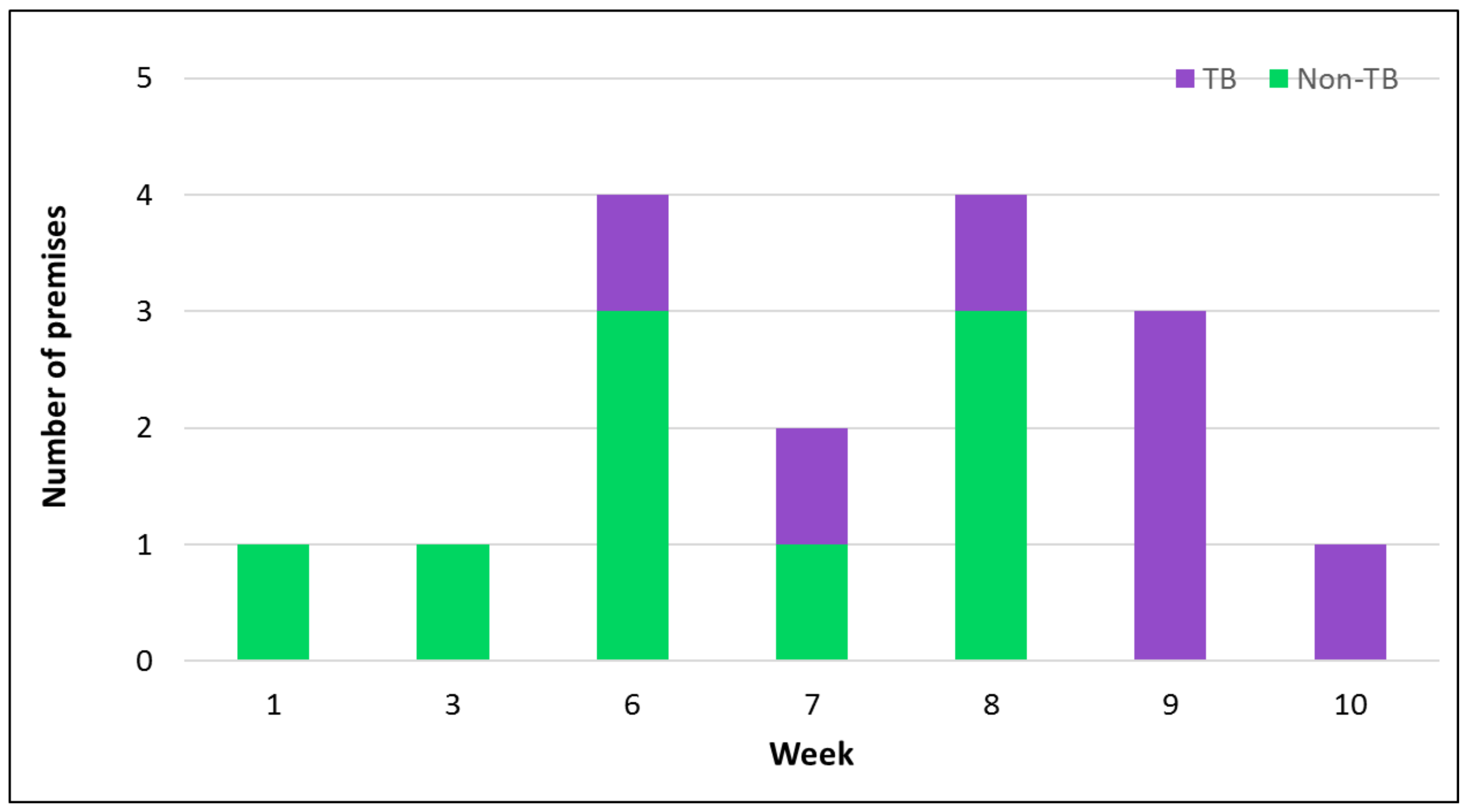
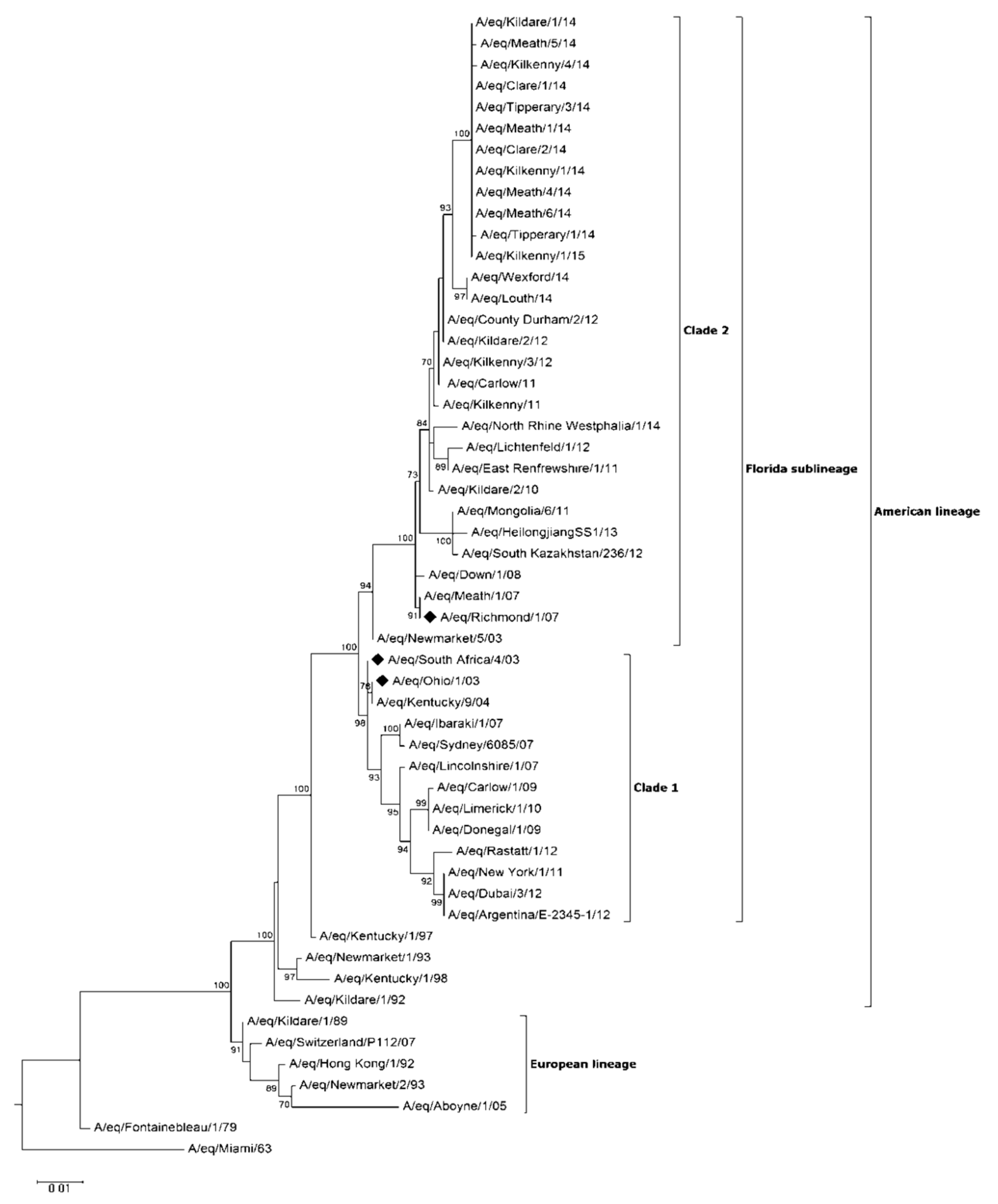
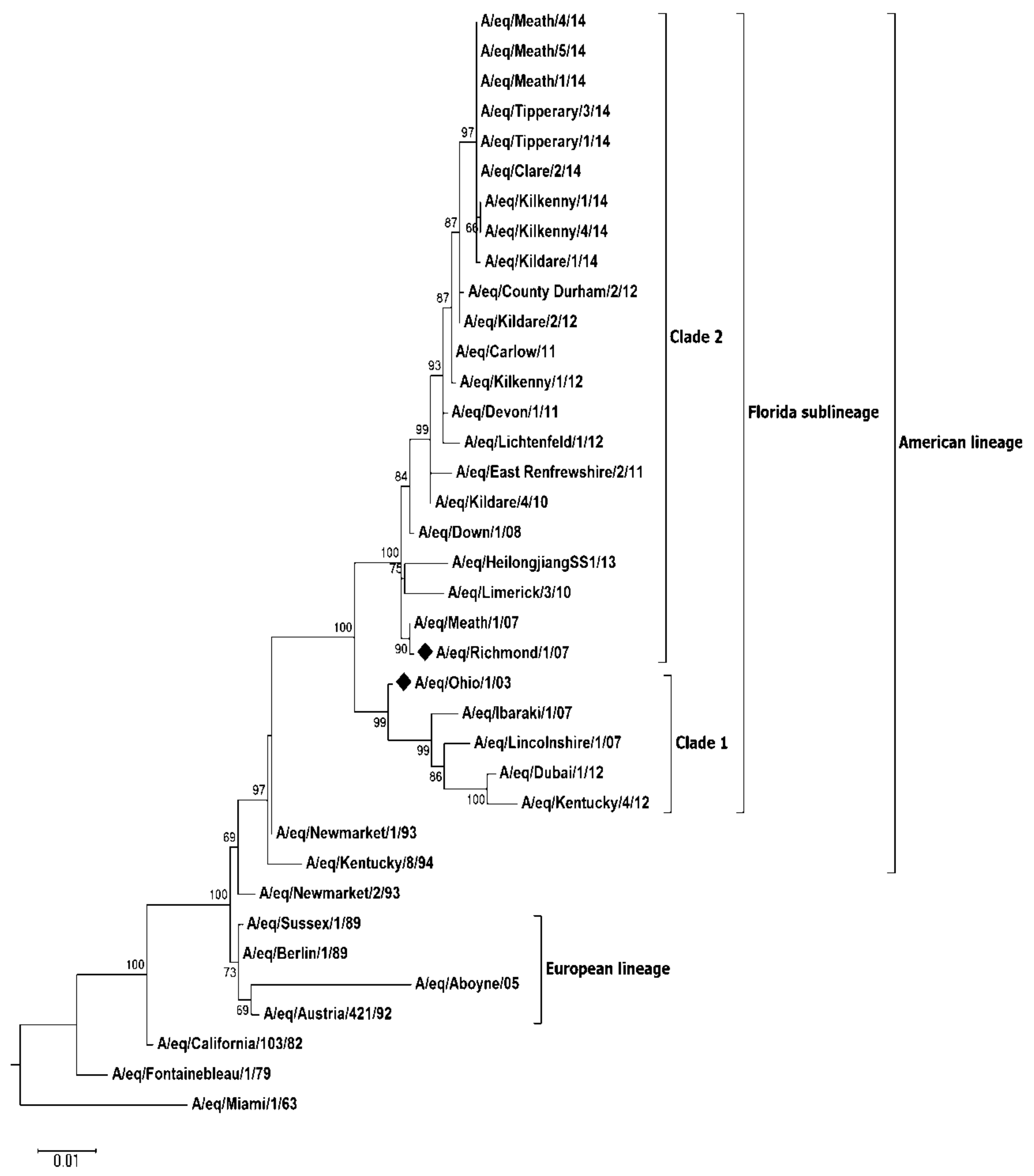
| PID | Date | Location (County) | Method of Case Confirmation | Premises Type | Veterinary Intervention | Clinical Cases (%) | Confirmed Cases (%) | Up to Date Vaccination Records Available (%) |
|---|---|---|---|---|---|---|---|---|
| 1 | January 2014 | Wexford | P 6, PS 2 | Non-TB | 14 days | 14/14 (100%) | 8/14 (57%) | 0/14 (0%) |
| 2 | February 2014 | Wexford | P 3, PS 1 | Mixed non-TB | 14 days | 4/5 (80%) | 4/5 (80%) | 1/5 (20%) |
| 3 | February 2014 | Louth | P 9, S 3, PS 2 | Equestrian centre/livery yard | 3 days | 5/29 (17%) | 14/29 (48%) | 1/29 (3%) |
| 4 | October 2014 | Meath | P 3 | Non-TB | 7 days | 3/3 (100%) | 3/3 (100%) | 0/3 (0%) |
| 5 | November 2014 | Tipperary | S 1, PS 4 | Mixed non-TB | 5 days | 5/7 (71%) | 5/7 (71%) | 0/7 (0%) |
| 6 | November 2014 | Tipperary | P 2, S 2, PS 5, PSV 1 | Mixed non-TB | 2 days | 5/16 (31%) | 10/16 (63%) | 6/16 (38%) |
| 7 | November 2014 | Clare | S 1, PS 1 | Show jumping | 4 days | 3/13 (23%) | 2/13 (15%) | 4/13 (31%) |
| 8 | November 2014 | Kildare | P 1, PSV 1 | TB (mixed ages and disciplines) | 1 day | 2/5 (40%) | 2/5 (40%) | 5/5 (100%) |
| 9 | November 2014 | Clare | PS 2 | TB (mixed ages and disciplines) | 17 days | 12/30 (40%) | 2/30 (7%) | 0/30 (0%) (18/30 reported vaccinated) * |
| 10 | December 2014 | Kildare | PS 1 | TB public stud | 3 days | 3/3 (100%) | 1/3 (33%) | 0/3 (0%) |
| 11 | December 2014 | Clare | PS 2, PSV 1 | Non-TB | 8 days | 3/4 (75%) | 3/4 (75%) | 0/4 (0%) |
| 12 | December 2014 | Meath | P 1 | Non-TB | 5 days | 1/1 (100%) | 1/1 (100%) | 0/1 (0%) |
| 13 | December 2014 | Dublin | P 6, S 2, PS 3 | Equestrian centre/livery yard | 16 days | 8/32 (25%) | 11/32 (34%) | 1/32 (3%) |
| 14 | December 2014 | Kilkenny | P 4, S 1, PS 2, PV 2, PSV 2 | Racing yard/TB breeding farm | 2 days | 13/25 (52%) | 11/25 (44%) | 10/25 (40%) |
| 15 | December 2014 | Meath | P 7 | Riding school/livery yard | 10 days | 6/19 (32%) | 7/19 (37%) | 0/19 (0%) |
| 16 | December 2014 | Meath | P 16, PS 1, PV 1 | Racing yard | 2 days | 17/47 (36%) | 18/47 (38%) | 41/47 (87%) |
| 17 | December 2014 | Meath | P 5, PV 1 | Racing yard | 6 days | 3/13 (23%) | 6/13 (46%) | 10/13 (77%) |
| 18 | December 2014 | Meath | P 4 | Mixed non-TB | 17 days | 2/6 (33%) | 4/6 (67%) | 0/6 (0%) |
| 19 | January 2015 | Kilkenny | P 3, S 2, PS 1 | Racing yard | 21 days | 12/33 (36%) | 6/33 (18%) | 19/33 (58%) |
| Last Vaccine Dose (Manufacturer) | No. of Horses (%) | Vaccine Breakdown (%) | No. of Days Since V2 | No. of Days Since V3 or Subsequent Booster | Representative H3N8 American Lineage Vaccine Strain |
|---|---|---|---|---|---|
| Duvaxyn IET Plus (Elanco Animal Health) | 43 (44%) | 13/43 (30%) | 124 (n = 1) | 276 ± 27.7 SE (n = 12) | A/eq/Newmarket/1/93 |
| Equip FT (Zoetis) | 10 (10%) | 5/10 (50%) | 189 (n = 1) | 267 ± 51.2 SE (n = 4) | A/eq/Kentucky/98 |
| Equilis Prequenza Te (MSD Animal Health) | 32 (33%) | 9/32 (28%) | 135 ± 33.5 SE (n = 2) | 217 ± 35.7 SE (n = 7) | A/eq/South Africa/4/03 1 |
| ProteqFlu Te (Merial) | 13 (13%) | 3/13 (23%) | 42 (n = 1) | 257 ± 95.5 SE (n = 2) | A/eq/Ohio/03 2 |
| Total/average | 98 | 30/98 (31%) | 125 ± 25.9 SE (n = 5) | 261 ± 19.6 SE (n = 25) |
| HA1 | HA2 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Animo Acid Number | 6 | 103 | 112 | 144 | 208 | 291 | 300 | 113 | 187 | 199 | 204 |
| A/eq/1/Meath/07 | S | P | V | A | R | E | V | A | L | C | G |
| A/eq/Wexford/14 | . | L | I | V | . | D | I | T | M | . | . |
| A/eq/Louth/14 | . | L | I | V | . | D | I | T | M | . | . |
| A/eq/Meath/1/14 | . | L | I | V | . | D | I | . | M | . | S |
| A/eq/ Tipperary/1/14 | . | L | I | V | . | D | I | . | M | Y | . |
| A/eq/Tipperary/3/14 | . | L | I | V | . | D | I | . | M | . | . |
| A/eq/ Clare/1/14 | . | L | I | V | . | D | I | . | M | . | . |
| A/eq/Kildare/1/14 | . | L | I | V | . | D | I | . | M | . | . |
| A/eq/Clare/2/14 | . | L | I | V | . | D | I | . | M | . | . |
| A/eq/ Kilkenny/1/14 * | . | L | I | V | . | D | I | . | M | . | . |
| A/eq/Kilkenny/4/14 * | N | L | I | V | . | D | I | . | M | . | . |
| A/eq/Meath/4/14 | . | L | I | V | . | D | I | . | M | . | . |
| A/eq/Meath/5/14 | . | L | I | V | G | D | I | . | M | . | . |
| A/eq/Meath/6/14 | . | L | I | V | . | D | I | . | M | . | . |
| A/eq/Kilkenny/1/15 | . | L | I | V | . | D | I | . | M | . | . |
| NA | ||||||||
|---|---|---|---|---|---|---|---|---|
| Animo Acid Number | 12 | 22 | 25 | 42 | 109 | 410 | 415 | 434 |
| A/eq/Meath/1/07 | S | V | H | G | R | I | K | T |
| A/eq/Meath/1/14 | . | . | N | C | K | V | R | S |
| A/eq/Tipperary/1/14 | . | . | N | C | K | V | R | S |
| A/eq/Tipperary/3/14 | . | . | N | C | K | V | R | S |
| A/eq/Kildare/1/14 | . | I | N | C | K | V | R | S |
| A/eq/Clare/2/14 | . | . | N | C | K | V | R | S |
| A/eq/Kilkenny/1/14 * | F | . | N | C | K | V | R | S |
| A/eq/Kilkenny/4/14 * | F | . | N | C | K | V | R | S |
| A/eq/Meath/4/14 | . | . | N | C | K | V | R | S |
| A/eq/Meath/5/14 | . | . | N | C | K | V | R | S |
| Reference Ferret Antisera | ||||||
|---|---|---|---|---|---|---|
| Reference Virus Strain | A/eq/NM/1/93 Am | A/eq/NM/2/93 Eu | A/eq/SA/4/03 FCL1 | A/eq/DL/1/09 FCL1 | A/eq/MH/1/07 FCL2 | A/eq/KE/2/12 FCL2 |
| A/eq/NM/1/93 | 181 | 32 | 181 | 256 | 724 | 1024 |
| A/eq/NM/2/93 | 64 | 256 | 64 | 64 | 256 | 256 |
| A/eq/SA/4/03 | 64 | 32 | 1448 | 1024 | 512 | 512 |
| A/eq/DL/1/09 | 256 | 128 | 1024 | 2896 | 256 | 256 |
| A/eq/MH/1/07 | 362 | 128 | 181 | 256 | 724 | 512 |
| A/eq/KE/2/12 | 181 | 91 | 128 | 256 | 1024 | 1024 |
| A/eq/Clare/2/14 | 181 | 91 | 128 | 256 | 1024 | 1024 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gildea, S.; Garvey, M.; Lyons, P.; Lyons, R.; Gahan, J.; Walsh, C.; Cullinane, A. Multifocal Equine Influenza Outbreak with Vaccination Breakdown in Thoroughbred Racehorses. Pathogens 2018, 7, 43. https://doi.org/10.3390/pathogens7020043
Gildea S, Garvey M, Lyons P, Lyons R, Gahan J, Walsh C, Cullinane A. Multifocal Equine Influenza Outbreak with Vaccination Breakdown in Thoroughbred Racehorses. Pathogens. 2018; 7(2):43. https://doi.org/10.3390/pathogens7020043
Chicago/Turabian StyleGildea, Sarah, Marie Garvey, Pamela Lyons, Rachel Lyons, Jacinta Gahan, Cathal Walsh, and Ann Cullinane. 2018. "Multifocal Equine Influenza Outbreak with Vaccination Breakdown in Thoroughbred Racehorses" Pathogens 7, no. 2: 43. https://doi.org/10.3390/pathogens7020043
APA StyleGildea, S., Garvey, M., Lyons, P., Lyons, R., Gahan, J., Walsh, C., & Cullinane, A. (2018). Multifocal Equine Influenza Outbreak with Vaccination Breakdown in Thoroughbred Racehorses. Pathogens, 7(2), 43. https://doi.org/10.3390/pathogens7020043





