The Uptake and Metabolism of Amino Acids, and Their Unique Role in the Biology of Pathogenic Trypanosomatids
Abstract
1. Introduction
2. Amino Acid Uptake
2.1. Amino Acid Transporters and the Identification of Their Putative Genes in Trypanosomatids
2.2. Biochemical Characterization of Amino Acid Transporters
2.2.1. Trypanosoma brucei
2.2.2. Trypanosoma cruzi
2.2.3. Leishmania spp.
3. Amino Acid Metabolism in TriTryps
3.1. Proline
3.2. Glutamine, Histidine, and Glutamate
3.3. Arginine and Ornithine
3.4. Branched-Chain Amino Acids
3.5. Asparagine and Aspartate
3.6. Cysteine and Methionine
3.7. Other Amino Acids: Phenylalanine, Tryptophan, Tyrosine, Serine, Glycine, Threonine, Alanine, and Lysine
3.7.1. Aromatic Amino Acids (Phe, Trp and Tyr)
3.7.2. Glycine and Serine
3.7.3. Threonine
3.7.4. Alanine
3.7.5. Lysine
4. Non-Obvious Roles of Amino Acids
4.1. Host-Parasite Interaction
4.2. Role of Amino Acids in the Regulation of Autophagy and Apoptosis
4.3. Osmotic Control by Amino Acids
5. Concluding Remarks
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Rodrigues, J.C.F.; Godinho, J.L.P.; de Souza, W. Biology of human pathogenic trypanosomatids: Epidemiology, lifecycle and ultrastructure. In Proteins and Proteomics of Leishmania and Trypanosoma; Springer: Dordrecht, The Netherlands, 2014; Volume 74, pp. 1–42. [Google Scholar]
- Bringaud, F.; Barrett, M.P.; Zilberstein, D. Multiple roles of proline transport and metabolism in trypanosomatids. Front. Biosci. (Landmark Ed.) 2012, 17, 349–374. [Google Scholar] [CrossRef] [PubMed]
- Garcia, E.S.; Genta, F.A.; De Azambuja, P.; Schaub, G.A. Interactions between intestinal compounds of triatomines and Trypanosoma cruzi. Trends Parasitol. 2010, 26, 499–505. [Google Scholar] [CrossRef] [PubMed]
- De Souza, W.; de Carvalho, T.M.U.; Barrias, E.S. Review on Trypanosoma cruzi: Host cell interaction. Int. J. Cell Biol. 2010, 2010, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Almeida-de-Faria, M.; Freymüller, E.; Colli, W.; Alves, M.J.M.; Freymu, E. Trypanosoma cruzi: Characterization of an intracellular epimastigote-like form. Exp. Parasitol. 1999, 92, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Alves, M.J.M.; Colli, W. Trypanosoma cruzi: Adhesion to the host cell and intracellular survival. IUBMB Life 2007, 59, 274–279. [Google Scholar] [CrossRef] [PubMed]
- Ponte-Sucre, A. An overview of Trypanosoma brucei infections: An intense host–parasite interaction. Front. Microbiol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Mony, B.M.; Matthews, K.R. Assembling the components of the quorum sensing pathway in African trypanosomes. Mol. Microbiol. 2015, 96, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Silvester, E.; McWilliam, K.; Matthews, K. The cytological events and molecular control of life cycle development of Trypanosoma brucei in the mammalian bloodstream. Pathogens 2017, 6, 29. [Google Scholar] [CrossRef] [PubMed]
- Rotureau, B.; Van Den Abbeele, J. Through the dark continent: African trypanosome development in the tsetse fly. Front. Cell. Infect. Microbiol. 2013, 3, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mogk, S.; Boßelmann, C.M.; Mudogo, C.N.; Stein, J.; Wolburg, H.; Duszenko, M. African trypanosomes and brain infection—The unsolved question. Biol. Rev. 2017, 92, 1675–1687. [Google Scholar] [CrossRef] [PubMed]
- Bates, P.A. Transmission of Leishmania metacyclic promastigotes by phlebotomine sand flies. Int. J. Parasitol. 2007, 37, 1097–1106. [Google Scholar] [CrossRef] [PubMed]
- Dostálová, A.; Volf, P. Leishmania development in sand flies: Parasite-vector interactions overview. Parasit. Vectors 2012, 5, 276. [Google Scholar] [CrossRef] [PubMed]
- Lestinova, T.; Rohousova, I.; Sima, M.; de Oliveira, C.I.; Volf, P. Insights into the sand fly saliva: Blood-feeding and immune interactions between sand flies, hosts, and Leishmania. PLoS Negl. Trop. Dis. 2017, 11, e0005600. [Google Scholar] [CrossRef] [PubMed]
- Podinovskaia, M.; Descoteaux, A. Leishmania and the macrophage: A multifaceted interaction. Future Microbiol. 2015, 10, 111–129. [Google Scholar] [CrossRef] [PubMed]
- Geiger, A.; Bossard, G.; Sereno, D.; Pissarra, J.; Lemesre, J.-L.; Vincendeau, P.; Holzmuller, P. Escaping deleterious immune response in their hosts: Lessons from trypanosomatids. Front. Immunol. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Tanowitz, H.B.; Scherer, P.E.; Mota, M.M.; Figueiredo, L.M. Adipose tissue: A safe haven for parasites? Trends Parasitol. 2017, 33, 276–284. [Google Scholar] [CrossRef] [PubMed]
- Higgins, C.F. ABC transporters: From microorganisms to man. Annu. Rev. Cell Biol. 1992, 8, 67–113. [Google Scholar] [CrossRef] [PubMed]
- Saier, M.H. A functional-phylogenetic classification system for transmembrane solute transporters. Microbiol. Mol. Biol. Rev. 2000, 64, 354–411. [Google Scholar] [CrossRef] [PubMed]
- El-Sayed, N.M.; Myler, P.J.; Blandin, G.; Berriman, M.; Crabtree, J.; Aggarwal, G.; Caler, E.; Renauld, H.; Worthey, E.A.; Hertz-Fowler, C.; et al. Comparative genomics of trypanosomatid parasitic protozoa. Science 2005, 309, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Young, G.B.; Jack, D.L.; Smith, D.W.; Saier, M.H. The amino acid/auxin: Proton symport permease family. Biochim. Biophys. Acta 1999, 1415, 306–322. [Google Scholar] [CrossRef]
- Bouvier, L.A.; Silber, A.M.; Galvão Lopes, C.; Canepa, G.E.; Miranda, M.R.; Tonelli, R.R.; Colli, W.; Alves, M.J.M.; Pereira, C.A. Post genomic analysis of permeases from the amino acid/auxin family in protozoan parasites. Biochem. Biophys. Res. Commun. 2004, 321, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Jackson, A.P. Origins of amino acid transporter loci in trypanosomatid parasites. BMC Evol. Biol. 2007, 7, 1–17. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baker, N.; Alsford, S.; Horn, D. Genome-wide RNAi screens in African trypanosomes identify the nifurtimox activator NTR and the eflornithine transporter AAT6. Mol. Biochem. Parasitol. 2011, 176, 55–57. [Google Scholar] [CrossRef] [PubMed]
- Schumann Burkard, G.; Jutzi, P.; Roditi, I. Genome-wide RNAi screens in bloodstream form trypanosomes identify drug transporters. Mol. Biochem. Parasitol. 2011, 175, 91–94. [Google Scholar] [CrossRef] [PubMed]
- Vincent, I.M.; Creek, D.; Watson, D.G.; Kamleh, M.A.; Woods, D.J.; Wong, P.E.; Burchmore, R.J.S.; Barrett, M.P. A molecular mechanism for eflornithine resistance in African trypanosomes. PLoS Pathog. 2010, 6, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ngô, H.; Tschudi, C.; Gull, K.; Ullu, E. Double-stranded RNA induces mRNA degradation in Trypanosoma brucei. Proc. Natl. Acad. Sci. USA 1998, 95, 14687–14692. [Google Scholar] [CrossRef] [PubMed]
- Beneke, T.; Madden, R.; Makin, L.; Valli, J.; Sunter, J.; Gluenz, E. A CRISPR Cas9 high-throughput genome editing toolkit for kinetoplastids. R. Soc. Open Sci. 2017, 4, 170095. [Google Scholar] [CrossRef] [PubMed]
- Tielens, A.G.M.; Van Hellemond, J.J. Differences in energy metabolism between Trypanosomatidae. Parasitol. Today 1998, 14, 265–271. [Google Scholar] [CrossRef]
- Bringaud, F.; Rivière, L.; Coustou, V. Energy metabolism of trypanosomatids: Adaptation to available carbon sources. Mol. Biochem. Parasitol. 2006, 149, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Tielens, A.G.M.; van Hellemond, J.J. Surprising variety in energy metabolism within Trypanosomatidae. Trends Parasitol. 2009, 25, 482–490. [Google Scholar] [CrossRef] [PubMed]
- Voorheis, H.P. Amino-acid transport in Trypanosoma brucei. Trans. R. Soc. Trop. Med. Hyg. 1971, 65, 241–242. [Google Scholar] [CrossRef]
- Clark Southworth, G.; Read, C.P. Absorption of some amino acids by the haemoflagellate, Trypanosoma gambiense. Comp. Biochem. Physiol. Part A Physiol. 1972, 41, 905–911. [Google Scholar] [CrossRef]
- Hansen, B.D. Trypanosoma gambiense: Membrane transport of amino acids. Exp. Parasitol. 1979, 48, 296–304. [Google Scholar] [CrossRef]
- Fricker, S.P.; Jones, S.E.M.; Clive Ellory, J.; Angus, J.M.; Klein, R.A. Threonine uptake in Trypanosoma brucei. Mol. Biochem. Parasitol. 1984, 11, 215–223. [Google Scholar] [CrossRef]
- Duszenko, M.; Ferguson, M.A.; Lamont, G.S.; Rifkin, M.R.; Cross, G.A. Cysteine eliminates the feeder cell requirement for cultivation of Trypanosoma brucei bloodstream forms in vitro. J. Exp. Med. 1985, 162, 1256–1263. [Google Scholar] [CrossRef] [PubMed]
- Geigy, R.; Huber, M.; Weinman, D.; Wyatt, G.R. Demonstration of trehalose in the vector of African trypanosomiasis: The tsetse fly. Acta Trop. 1959, 16, 255–262. [Google Scholar] [PubMed]
- Lamour, N.; Riviere, L.; Coustou, V.; Coombs, G.H.; Barrett, M.P.; Bringaud, F.F.F. Proline metabolism in procyclic Trypanosoma brucei is down-regulated in the presence of glucose. J. Biol. Chem. 2005, 280, 11902–11910. [Google Scholar] [CrossRef] [PubMed]
- Mantilla, B.S.; Marchese, L.; Casas-Sánchez, A.; Dyer, N.A.; Ejeh, N.; Biran, M.; Bringaud, F.; Lehane, M.J.; Acosta-Serrano, A.; Silber, A.M. Proline metabolism is essential for Trypanosoma brucei brucei survival in the tsetse vector. PLoS Pathog. 2017, 13, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Bursell, E. The role of proline in energy metabolism. In Energy Metabolism in Insects; Springer: Boston, MA, USA, 1981; pp. 135–154. [Google Scholar]
- L’Hostis, C.; Geindre, M.; Deshusses, J. Active transport of L-proline in the protozoan parasite Trypanosoma brucei brucei. Biochem. J. 1993, 291, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Mathieu, C.; Salgado, A.G.; Wirdnam, C.; Meier, S.; Grotemeyer, M.S.; Inbar, E.; Mäser, P.; Zilberstein, D.; Sigel, E.; Bütikofer, P.; et al. Trypanosoma brucei eflornithine transporter AAT6 is a low-affinity low-selective transporter for neutral amino acids. Biochem. J. 2014, 463, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, B.; Rattendi, D.; Lloyd, D.; Yarlett, N.; Bacchi, C.J. Kinetics of methionine transport and metabolism by Trypanosoma brucei brucei and Trypanosoma brucei rhodesiense. Arch. Biochem. Biophys. 2000, 377, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Hasne, M.P.; Barrett, M.P. Transport of methionine in Trypanosoma brucei brucei. Mol. Biochem. Parasitol. 2000, 111, 299–307. [Google Scholar] [CrossRef]
- Mathieu, C.; Macêdo, J.P.; Hürlimann, D.; Wirdnam, C.; Haindrich, A.C.; Grotemeyer, M.S.; González-Salgado, A.; Schmidt, R.S.; Inbar, E.; Mäser, P.; et al. Arginine and lysine transporters are essential for Trypanosoma brucei. PLoS ONE 2017, 12, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Macedo, J.P.; Currier, R.B.; Wirdnam, C.; Horn, D.; Alsford, S.; Rentsch, D. Ornithine uptake and the modulation of drug sensitivity in Trypanosoma brucei. FASEB J. 2017, 31, 4649–4660. [Google Scholar] [CrossRef] [PubMed]
- Hampton, J.R. Lysine uptake in cultured Trypanosoma cruzi: Interactions of competitive inhibitors. J. Protozool. 1970, 17, 597–600. [Google Scholar] [CrossRef] [PubMed]
- Hampton, J.R. Arginine transport in the culture form of Trypanosoma cruzi. J. Protozool. 1971, 18, 701–703. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, S.S.; Pereira, A.A.S.; Chiari, E.; Mares-Guia, M.; Gazzinelli, G. Comparative kinetics of arginine and lysine transport by epimastigotes and trypomastigotes from two strains of Trypanosoma cruzi. J. Protozool. 1976, 23, 179–186. [Google Scholar] [CrossRef]
- Canepa, G.E.; Silber, A.M.; Bouvier, L.A.; Pereira, C.A. Biochemical characterization of a low-affinity arginine permease from the parasite Trypanosoma cruzi. FEMS Microbiol. Lett. 2004, 236, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.A.; Alonso, G.D.; Paveto, M.C.; Flawiá, M.M.; Torres, H.N. L-arginine uptake and L-phosphoarginine synthesis in Trypanosoma cruzi. J. Eukaryot. Microbiol. 1999, 46, 566–570. [Google Scholar] [CrossRef] [PubMed]
- Inbar, E.; Canepa, G.E.; Carrillo, C.; Glaser, F.; Grotemeyer, M.S.; Rentsch, D.; Zilberstein, D.; Pereira, C.A. Lysine transporters in human trypanosomatid pathogens. Amino Acids 2012, 42, 347–360. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, C.; Canepa, G.E.; Giacometti, A.; Bouvier, L.A.; Miranda, M.R.; de los Milagros Camara, M.; Pereira, C.A. Trypanosoma cruzi amino acid transporter TcAAAP411 mediates arginine uptake in yeasts. FEMS Microbiol. Lett. 2010, 306, 97–102. [Google Scholar] [CrossRef] [PubMed]
- Miranda, M.R.; Sayé, M.; Bouvier, L.A.; de los Milagros Cámara, M.; Montserrat, J.; Pereira, C.A. Cationic amino acid uptake constitutes a metabolic regulation mechanism and occurs in the flagellar pocket of Trypanosoma cruzi. PLoS ONE 2012, 7, 3–9. [Google Scholar] [CrossRef] [PubMed]
- Henriques, C.; Miller, M.P.; Catanho, M.; De Carvalho, T.M.U.; Krieger, M.A.; Probst, C.M.; De Souza, W.; Degrave, W.; Amara, S.G. Identification and functional characterization of a novel arginine/ornithine transporter, a member of a cationic amino acid transporter subfamily in the Trypanosoma cruzi genome. Parasites Vectors 2015, 8, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Silber, A.M.; Tonelli, R.R.; Martinelli, M.; Colli, W.; Alves, M.J.M. Active transport of L-proline in Trypanosoma cruzi. J. Eukaryot. Microbiol. 2002, 49, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Saye, M.; Miranda, M.R.; Di Girolamo, F.; De Los Milagros Cámara, M.; Pereira, C.A. Proline modulates the Trypanosoma cruzi resistance to reactive oxygen species and drugs through a novel D, L-proline transporter. PLoS ONE 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Caballero, Z.C.; Costa-Martins, A.G.; Ferreira, R.C.; P Alves, J.M.; Serrano, M.G.; Camargo, E.P.; Buck, G.A.; Minoprio, P.; Teixeira, G.M.M. Phylogenetic and syntenic data support a single horizontal transference to a Trypanosoma ancestor of a prokaryotic proline racemase implicated in parasite evasion from host defences. Parasites Vectors 2015, 8, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Canepa, G.E.; Bouvier, L.A.; Urias, U.; Miranda, M.R.; Colli, W.; Alves, M.J.M.; Pereira, C.A. Aspartate transport and metabolism in the protozoan parasite Trypanosoma cruzi. FEMS Microbiol. Lett. 2005, 247, 65–71. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Silber, A.M.; Rojas, R.L.G.; Urias, U.; Colli, W.; Alves, M.J.M. Biochemical characterization of the glutamate transport in Trypanosoma cruzi. Int. J. Parasitol. 2006, 36, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Barisón, M.J.; Damasceno, F.S.; Mantilla, B.S.; Silber, A.M. The active transport of histidine and its role in ATP production in Trypanosoma cruzi. J. Bioenerg. Biomembr. 2016, 48, 437–449. [Google Scholar] [CrossRef] [PubMed]
- Galvez Rojas, R.L.; Ahn, I.Y.; Suárez Mantilla, B.; Sant’Anna, C.; Pral, E.M.F.; Silber, A.M. The uptake of GABA in Trypanosoma cruzi. J. Eukaryot. Microbiol. 2015, 62, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Canepa, G.E.; Bouvier, L.A.; Miranda, M.R.; Uttaro, A.D.; Pereira, C.A. Characterization of Trypanosoma cruzi L-cysteine transport mechanisms and their adaptive regulation. FEMS Microbiol. Lett. 2009, 292, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Manchola, N.C.; Rapado, L.N.; Barison, M.J.; Silber, A.M. Biochemical characterization of branched chain amino acids uptake in Trypanosoma cruzi. J. Eukaryot. Microbiol. 2015, 63, 299–308. [Google Scholar] [CrossRef] [PubMed]
- Mukkada, A.J.; Simon, M.W. Leishmania tropica: Uptake of methionine by promastigotes. Exp. Parasitol. 1977, 42, 87–96. [Google Scholar] [CrossRef]
- Simon, M.W.; Mukkada, A.J. Leishmania tropica: Regulation and specificity of the methionine transport system in promastigotes. Exp. Parasitol. 1977, 42, 97–105. [Google Scholar] [CrossRef]
- Law, S.S.; Mukkada, A.J. Transport of L-proline and its regulation in Leishmania tropica promastigotes. J. Protozool. 1979, 26, 295–301. [Google Scholar] [CrossRef] [PubMed]
- Bonay, P.; Cohen, E. Neutral amino acid transport in Leishmania promastigotes. Biochim. Biophys. Acta 1983, 731, 222–228. [Google Scholar] [CrossRef]
- Zilberstein, D.; Dwyer, D.M. Protonmotive force-driven active transport of D-glucose and L-proline in the protozoan parasite Leishmania donovani. Proc. Natl. Acad. Sci. USA 1985, 82, 1716–1720. [Google Scholar] [CrossRef] [PubMed]
- Zilberstein, D.; Philosoph, H.; Gepstein, A. Maintenance of cytoplasmic pH and proton motive force in promastigotes of Leishmania donovani. Mol. Biochem. Parasitol. 1989, 36, 109–117. [Google Scholar] [CrossRef]
- Glaser, T.A.; Mukkada, A.J. Proline transport in Leishmania donovani amastigotes: Dependence on pH gradients and membrane potential. Mol. Biochem. Parasitol. 1992, 51, 1–8. [Google Scholar] [CrossRef]
- Ter Kuile, B.H.; Opperdoes, F.R. A chemostat study on proline uptake and metabolism of Leishmania donovani. J. Protozool. 1992, 39, 555–558. [Google Scholar] [CrossRef] [PubMed]
- Zilberstein, D.; Gepstein, A. Regulation of L-proline transport in Leishmania donovani by extracellular pH. Mol. Biochem. Parasitol. 1993, 61, 197–205. [Google Scholar] [CrossRef]
- Mazareb, S.; Fu, Z.Y.; Zilberstein, D. Developmental regulation of proline transport in Leishmania donovani. Exp. Parasitol. 1999, 91, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Kandpal, M.; Fouce, R.B.; Pal, A.; Guru, P.Y.; Tekwani, B.L. Kinetics and molecular characteristics of arginine transport by Leishmania donovani promastigotes. Mol. Biochem. Parasitol. 1995, 71, 193–201. [Google Scholar] [CrossRef]
- Paes, L.S.; Galvez Rojas, R.L.; Daliry, A.; Floeter-Winter, L.M.; Ramirez, M.I.; Silber, A.M. Active transport of glutamate in Leishmania (Leishmania) amazonensis. J. Eukaryot. Microbiol. 2008, 55, 382–387. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, M.G.; Paes, L.S.; Zampieri, R.A.; da Silva, M.F.L.; Silber, A.M.; Floeter-Winter, L.M. Biochemical characterization of serine transport in Leishmania (Leishmania) amazonensis. Mol. Biochem. Parasitol. 2009, 163, 107–113. [Google Scholar] [CrossRef] [PubMed]
- Inbar, E.; Schlisselberg, D.; Suter Grotemeyer, M.; Rentsch, D.; Zilberstein, D. A versatile proline/alanine transporter in the unicellular pathogen Leishmania donovani regulates amino acid homoeostasis and osmotic stress responses. Biochem. J. 2013, 449, 555–566. [Google Scholar] [CrossRef] [PubMed]
- Shaked-Mishan, P.; Suter-Grotemeyer, M.; Yoel-Almagor, T.; Holland, N.; Zilberstein, D.; Rentsch, D. A novel high-affinity arginine transporter from the human parasitic protozoan Leishmania donovani. Mol. Microbiol. 2006, 60, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Darlyuk, I.; Goldman, A.; Roberts, S.C.; Ullmarr, B.; Rentsch, D.; Zilberstein, D. Arginine homeostasis and transport in the human pathogen Leishmania donovani. J. Biol. Chem. 2009, 284, 19800–19807. [Google Scholar] [CrossRef] [PubMed]
- Castilho-Martins, E.A.; da Silva, M.F.L.; dos Santos, M.G.; Muxel, S.M.; Floeter-Winter, L.M. Axenic Leishmania amazonensis promastigotes sense both the external and internal arginine pool distinctly regulating the two transporter-coding genes. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Aoki, J.I.; Muxel, S.M.; Zampieri, R.A.; Acuña, S.M.; Fernandes, J.C.R.; Vanderlinde, R.H.; Sales, M.C.O.P.; Floeter-Winter, L.M. L-arginine availability and arginase activity: Characterization of amino acid permease 3 in Leishmania amazonensis. PLoS Negl. Trop. Dis. 2017, 11, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.; Miller, R.; Block, K. Branched-chain amino acid metabolism. Annu. Rev. Nutr. 1984, 4, 409–454. [Google Scholar] [CrossRef] [PubMed]
- Curien, G.; Biou, V.; Mas-Droux, C.; Robert-Genthon, M.; Ferrer, J.L.; Dumas, R. Amino acid biosynthesis: New architectures in allosteric enzymes. Plant Physiol. Biochem. 2008, 46, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Nayak, A.; Akpunarlieva, S.; Barrett, M.; Burchmore, R. A defined medium for Leishmania culture allows definition of essential amino acids. Exp. Parasitol. 2018, 185, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, K.; Roos, D.S. Protozoan genomics for drug discovery. Nat. Biotechnol. 2005, 23, 1089–1091. [Google Scholar] [CrossRef] [PubMed]
- Canepa, G.E.; Carrillo, C.; Miranda, M.R.; Sayé, M.; Pereira, C.A. Arginine kinase in Phytomonas, a trypanosomatid parasite of plants. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 2011, 160, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Vincent, I.M.; Creek, D.J.; Burgess, K.; Woods, D.J.; Burchmore, R.J.S.; Barrett, M.P. Untargeted metabolomics reveals a lack of synergy between nifurtimox and eflornithine against Trypanosoma brucei. PLoS Negl. Trop. Dis. 2012, 6. [Google Scholar] [CrossRef] [PubMed]
- Berriman, M.; Ghedin, E.; Hertz-Fowler, C.; Blandin, G.; Renauld, H.; Bartholomeu, D.C.; Lennard, N.J.; Caler, E.; Hamlin, N.E.; Haas, B.; et al. The genome of the African trypanosome Trypanosoma brucei. Science 2005, 309, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Guttman, H.N. Patterns of methionine and lysine biosynthesis in the Trypanosomatidae during growth. J. Protozool. 1967, 14, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Opperdoes, F.R.; Butenko, A.; Flegontov, P.; Yurchenko, V.; Lukeš, J. Comparative metabolism of free-living Bodo saltans and parasitic trypanosomatids. J. Eukaryot. Microbiol. 2016, 63, 657–678. [Google Scholar] [CrossRef] [PubMed]
- Krassner, S.M.; Flory, B. Proline metabolism in Leishmania donovani promastigotes. J. Protozool. 1972, 19, 682–685. [Google Scholar] [CrossRef] [PubMed]
- Sylvester, D.; Krassner, S.M.; In, P.M. Proline metabolism in Trypanosoma cruzi epimastigotes. Comp. Biochem. Physiol. Part B Biochem. 1976, 55, 443–447. [Google Scholar] [CrossRef]
- Coustou, V.; Biran, M.; Breton, M.; Guegan, F.; Rivière, L.; Plazolles, N.; Nolan, D.; Barrett, M.P.; Franconi, J.M.; Bringaud, F.; et al. Glucose-induced remodeling of intermediary and energy metabolism in procyclic Trypanosoma brucei. J. Biol. Chem. 2008, 283, 16343–16354. [Google Scholar] [CrossRef] [PubMed]
- Paes, L.S.; Suárez Mantilla, B.; Zimbres, F.M.; Pral, E.M.F.; Diogo de Melo, P.; Tahara, E.B.; Kowaltowski, A.J.; Elias, M.C.; Silber, A.M. Proline dehydrogenase regulates redox state and respiratory metabolism in Trypanosoma cruzi. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Mantilla, B.S.; Paes, L.S.; Pral, E.M.F.; Martil, D.E.; Thiemann, O.H.; Fernández-Silva, P.; Bastos, E.L.; Silber, A.M. Role of Δ1-pyrroline-5-carboxylate dehydrogenase supports mitochondrial metabolism and host-cell invasion of Trypanosoma cruzi. J. Biol. Chem. 2015, 290, 7767–7790. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A. B. Strecker The interconversion of glutamic acid and proline. J. Biol. Chem. 1962, 237, 1876–1882. [Google Scholar] [PubMed]
- Phang, J.M.; Yeh, G.C.; Hagedorn, C.H. The intercellular proline cycle. Life Sci. 1981, 28, 53–58. [Google Scholar] [CrossRef]
- Vogel, H.J.; Davis, J. Glutamic g-semialdehyde and Δ1-pyrroline-5-carboxylic acid, intermediates in the biosynthesis of proline. J. Am. Chem. Soc. 1952, 74, 109–112. [Google Scholar] [CrossRef]
- Strecker, H.J. The interconversion of glutamic acid and proline. J. Biol. Chem. 1960, 235, 3218–3223. [Google Scholar] [CrossRef]
- Tonelli, R.R.; Silber, A.M.; Almeida-de-Faria, M.; Hirata, I.Y.; Colli, W.; Alves, J.M. L-proline is essential for the intracellular differentiation of Trypanosoma cruzi. Cell. Microbiol. 2004, 6, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Opperdoes, F.R.; Coombs, G.H. Metabolism of Leishmania: Proven and predicted. Trends Parasitol. 2007, 23, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Akpunarlieva, S.; Weidt, S.; Lamasudin, D.; Naula, C.; Henderson, D.; Barrett, M.; Burgess, K.; Burchmore, R. Integration of proteomics and metabolomics to elucidate metabolic adaptation in Leishmania. J. Proteom. 2017, 155, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Hayzer, D.J.; Leisinger, T. Proline biosynthesis in Escherichia coli. Eur. J. Biochem. 1982, 121, 561–565. [Google Scholar] [CrossRef] [PubMed]
- Csonka, L.N.; Leisinger, T. Biosynthesis of proline. EcoSal Plus 2007, 2. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.A.; Delauney, A.J.; Verma, D.P. A bifunctional enzyme (delta 1-pyrroline-5-carboxylate synthetase) catalyzes the first two steps in proline biosynthesis in plants. Proc. Natl. Acad. Sci. USA 1992, 89, 9354–9358. [Google Scholar] [CrossRef] [PubMed]
- Turchetto-Zolet, A.C.; Margis-Pinheiro, M.; Margis, R. The evolution of pyrroline-5-carboxylate synthase in plants: A key enzyme in proline synthesis. Mol. Genet. Genom. 2009, 281, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Yura, T.; Vogel, H.J. Pyrroline-5-carboxylate reductase of Neurospora crassa; partial purification and some properties. J. Biol. Chem. 1959, 234, 335–338. [Google Scholar] [PubMed]
- Rossi, J.J.; Vender, J.; Berg, C.M.; Coleman, W.H. Partial purification and some properties of Δ1-pyrroline-5-carboxylate reductase from Escherichia coli. J. Bacteriol. 1977, 129, 108–114. [Google Scholar] [PubMed]
- Szoke, A.; Miao, G.H.; Hong, Z.; Verma, D.P. Subcellular location of delta-pyrroline-5-carboxylate reductase in root/nodule and leaf of soybean. Plant Physiol. 1992, 99, 1642–1649. [Google Scholar] [CrossRef] [PubMed]
- Murahama, M.; Yoshida, T.; Hayashi, F.; Ichino, T.; Sanada, Y.; Wada, K. Purification and characterization of delta(1)-pyrroline-5-carboxylate reductase isoenzymes, indicating differential distribution in spinach (Spinacia oleracea L.) leaves. Plant Cell Physiol. 2001, 42, 742–750. [Google Scholar] [CrossRef] [PubMed]
- Delauney, A.J.; Hu, C.A.A.; Kavi Kishor, P.B.; Verma, D.P.S. Cloning of ornithine δ-anrinotransferase cDNA from Vigna aconitifolia by trans-complementation in Escherichia coli and regulation of proline biosynthesis. J. Biol. Chem. 1993, 268, 18673–18678. [Google Scholar] [PubMed]
- Yoshida, N.; Plessmann Camargo, E. Ureotelism and ammonotelism in trypanosomatids. J. Bacteriol. 1978, 136, 1184–1186. [Google Scholar] [PubMed]
- König, S.; Marco, H.; Gäde, G. D-Proline: Comment to “An overview on D-amino acids”. Amino Acids 2018, 50, 359–361. [Google Scholar] [CrossRef] [PubMed]
- Reina-San-Martín, B.; Degrave, W.; Rougeot, C.; Cosson, A.; Chamond, N.; Cordeiro-da-Silva, A.; Arala-Chaves, M.; Coutinho, A.; Minoprio, P. A B-cell mitogen from a pathogenic trypanosome is a eukaryotic proline racemase. Nat. Med. 2000, 6, 890–897. [Google Scholar] [PubMed]
- Chamond, N.; Grégoire, C.; Coatnoan, N.; Rougeot, C.; Freitas-Junior, L.H.; Da Silveira, J.F.; Degrave, W.M.; Minoprio, P. Biochemical characterization of proline racemases from the human protozoan parasite Trypanosoma cruzi and definition of putative protein signatures. J. Biol. Chem. 2003, 278, 15484–15494. [Google Scholar] [CrossRef] [PubMed]
- Chamond, N.; Goytia, M.; Coatnoan, N.; Barale, J.C.; Cosson, A.; Degrave, W.M.; Minoprio, P. Trypanosoma cruzi proline racemases are involved in parasite differentiation and infectivity. Mol. Microbiol. 2005, 58, 46–60. [Google Scholar] [CrossRef] [PubMed]
- Cazzulo, J.J.; Juan, S.M.; Segura, E.L. Glutamate dehydrogenase and aspartate aminotransferase in Trypanosoma cruzi. Comp. Biochem. Physiol. B 1977, 56, 301–303. [Google Scholar] [CrossRef]
- Barderi, P.; Campetella, O.; Carlos, A.; Frasch, A.C.C.; Santome, J.A.; Hellman, U.; Petterssonr, U.; Cazzulo, J.J. The NADP+-linked glutamate dehydrogenase from Trypanosoma cruzi: Sequence, genomic organization and expression. Biochem. J. 1998, 330, 951–958. [Google Scholar] [CrossRef] [PubMed]
- Cannata, J.J.B.; Frasch, A.C.C.; Cataldi de Flombaum, M.A.; Segura, E.L.; Cazzulo, J.J. Two forms of malic enzyme with different regulatory properties in Trypanosoma cruzi. Biochem. J. 1979, 184, 409–419. [Google Scholar] [CrossRef] [PubMed]
- Zeledon, R. Comparative physiological studies on four species of hemoflagellates in culture. II. Effect of carbohydrates and related substances and some amino compounds on the respiration. J. Parasitol. 1960, 46, 541–551. [Google Scholar] [CrossRef] [PubMed]
- Zelada, C.; Jo, J.; Montemartini, M.; Cazzulo, J.J.; Nowicki, C. Purification and partial structural and kinetic characterization of an alanine aminotransferase from epimastigotes of Trypanosoma cruzi. Mol. Biochem. Parasitol. 1996, 79, 225–228. [Google Scholar] [CrossRef]
- Li, Y.; Shah-Simpson, S.; Okrah, K.; Belew, A.T.; Choi, J.; Caradonna, K.L.; Padmanabhan, P.; Ndegwa, D.M.; Temanni, M.R.; Corrada Bravo, H.; et al. Transcriptome remodeling in Trypanosoma cruzi and human cells during intracellular infection. PLoS Pathog. 2016, 12, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Barisón, M.J.; Rapado, L.N.; Merino, E.F.; Pral, E.M.F.; Mantilla, B.S.; Marchese, L.; Nowicki, C.; Silber, A.M.; Cassera, M.B. Metabolomic profiling reveals a finely tuned, starvation induced metabolic switch in Trypanosoma cruzi epimastigotes. J. Biol. Chem. 2017, 292, 8964–8977. [Google Scholar] [CrossRef] [PubMed]
- Crispim, M.; Damasceno, S.; Hernandez, A.; Barison, M.J.; Sauter, I.P.; Pavani, R.S.; Moura, A.S.; Pral, E.M.F.; Cortez, M.; Elias, M.C.; et al. The glutamine synthetase of Trypanosoma cruzi is required for its resistance to ammonium accumulation and evasion of the parasitophorous vacuole during host-cell infection. PLoS Negl. Trop. Dis. 2018, 12, e0006170. [Google Scholar] [CrossRef] [PubMed]
- Gao, G.; Nara, T.; Nakajima-Shimada, J.; Aoki, T. Novel organization and sequences of five genes encoding all six enzymes for de novo pyrimidine biosynthesis in Trypanosoma cruzi. J. Mol. Biol. 1999, 285, 149–161. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, M.; Morales, J.; Fukai, Y.; Suzuki, S.; Takamiya, S.; Tsubouchi, A.; Inoue, S.; Inoue, M.; Kita, K.; Harada, S.; et al. Critical importance of the de novo pyrimidine biosynthesis pathway for Trypanosoma cruzi growth in the mammalian host cell cytoplasm. Biochem. Biophys. Res. Commun. 2012, 417, 1002–1006. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Leija, C.; Rijo-Ferreira, F.; Chen, J.; Cestari, I.; Stuart, K.; Tu, B.P.; Phillips, M.A. GMP synthase is essential for viability and infectivity of Trypanosoma brucei despite a redundant purine salvage pathway. Mol. Microbiol. 2015, 97, 1006–1020. [Google Scholar] [CrossRef] [PubMed]
- Marciano, D.; Maugeri, D.A.; Cazzulo, J.J.; Nowicki, C. Functional characterization of stage-specific aminotransferases from trypanosomatids. Mol. Biochem. Parasitol. 2009, 166, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Yadav, S.; Soumya, N.; Kumar, R.; Kishore, N.; Singh, S. Biochemical and inhibition studies of glutamine synthetase from Leishmania donovani. Microb. Pathog. 2017, 107, 164–174. [Google Scholar] [CrossRef] [PubMed]
- Naderer, T.; Wee, E.; McConville, M.J. Role of hexosamine biosynthesis in Leishmania growth and virulence. Mol. Microbiol. 2008, 69, 858–869. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.M. Recent advances in arginine metabolism: Roles and regulation of the arginases. Br. J. Pharmacol. 2009, 157, 922–930. [Google Scholar] [CrossRef] [PubMed]
- Pegg, A.E. The function of spermine. IUBMB Life 2014, 66, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Ariyanayagam, M.R.; Fairlamb, A.H. Diamine auxotrophy may be a universal feature of Trypanosoma cruzi epimastigotes. Mol. Biochem. Parasitol. 1997, 84, 111–121. [Google Scholar] [CrossRef]
- Ariyanayagam, M.R.; Fairlamb, A.H. Ovothiol and trypanothione as antioxidants in trypanosomatids. Mol. Biochem. Parasitol. 2001, 115, 189–198. [Google Scholar] [CrossRef]
- Oza, S.L.; Tetaud, E.; Ariyanayagam, M.R.; Warnon, S.S.; Fairlamb, A.H. A single enzyme catalyses formation of trypanothione from glutathione and spermidine in Trypanosoma cruzi. J. Biol. Chem. 2002, 277, 35853–35861. [Google Scholar] [CrossRef] [PubMed]
- Oza, S.L.; Ariyanayagam, M.R.; Aitcheson, N.; Fairlamb, A.H. Properties of trypanothione synthetase from Trypanosoma brucei. Mol. Biochem. Parasitol. 2003, 131, 25–33. [Google Scholar] [CrossRef]
- Oza, S.L.; Shaw, M.P.; Wyllie, S.; Fairlamb, A.H. Trypanothione biosynthesis in Leishmania major. Mol. Biochem. Parasitol. 2005, 139, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Jockers-Scherubl, M.C.; Schirmer, R.H.; Krauth-Siegel, R.L. Trypanothione reductase from Trypanosoma cruzi. Catalytic properties of the enzyme and inhibition studies with trypanocidal compounds. Eur. J. Biochem. 1989, 180, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Jones, D.C.; Ariza, A.; Chow, W.H.A.; Oza, S.L.; Fairlamb, A.H. Comparative structural, kinetic and inhibitor studies of Trypanosoma brucei trypanothione reductase with T. cruzi. Mol. Biochem. Parasitol. 2010, 169, 12–19. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.C.; Tancer, M.J.; Polinsky, M.R.; Michael Gibson, K.; Heby, O.; Ullman, B. Arginase plays a pivotal role in polyamine precursor metabolism in Leishmania: Characterization of gene deletion mutants. J. Biol. Chem. 2004, 279, 23668–23678. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, E.R.; da Silva, M.F.L.; Fischer, H.; Mortara, R.A.; Mayer, M.G.; Framesqui, K.; Silber, A.M.; Floeter-Winter, L.M. Biochemical and biophysical properties of a highly active recombinant arginase from Leishmania (Leishmania) amazonensis and subcellular localization of native enzyme. Mol. Biochem. Parasitol. 2008, 159, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Jenkinson, C.P.; Grody, W.W.; Cederbaum, S.D. Comparative properties of arginases. Comp. Biochem. Physiol. B Biochem. Mol. Biol. 1996, 114, 107–132. [Google Scholar] [CrossRef]
- Badirzadeh, A.; Taheri, T.; Taslimi, Y.; Abdossamadi, Z.; Heidari-Kharaji, M.; Gholami, E.; Sedaghat, B.; Niyyati, M.; Rafati, S. Arginase activity in pathogenic and non-pathogenic species of Leishmania parasites. PLoS Negl. Trop. Dis. 2017, 11, e0005774. [Google Scholar] [CrossRef] [PubMed]
- Boitz, J.M.; Gilroy, C.A.; Olenyik, T.D.; Paradis, D.; Perdeh, J.; Dearman, K.; Davis, M.J.; Yates, P.A.; Li, Y.; Riscoe, M.K.; et al. Arginase is essential for survival of Leishmania donovani promastigotes but not intracellular amastigotes. Infect. Immun. 2017, 85. [Google Scholar] [CrossRef] [PubMed]
- Hai, Y.; Kerkhoven, E.J.; Barrett, M.P.; Christianson, D.W. Crystal structure of an arginase-like protein from Trypanosoma brucei that evolved without a binuclear manganese cluster. Biochemistry 2015, 54, 458–471. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.A.; Alonso, G.D.; Torres, H.N.; Flawiá, M.M. Arginine kinase: A common feature for management of energy reserves in African and American flagellated trypanosomatids. J. Eukaryot. Microbiol. 2002, 49, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.A.; Alonso, G.D.; Paveto, M.C.; Iribarren, A.; Cabanas, M.L.; Torres, H.N.; Flawiá, M.M. Trypanosoma cruzi arginine kinase characterization and cloning. A novel energetic pathway in protozoan parasites. J. Biol. Chem. 2000, 275, 1495–1501. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.A.; Alonso, G.D.; Ivaldi, S.; Silber, A.; Alves, M.J.M.; Bouvier, L.A.; Flawiá, M.M.; Torres, H.N. Arginine metabolism in Trypanosoma cruzi is coupled to parasite stage and replication. FEBS Lett. 2002, 526, 111–114. [Google Scholar] [CrossRef]
- Voncken, F.; Gao, F.; Wadforth, C.; Harley, M.; Colasante, C. The phosphoarginine energy-buffering system of Trypanosoma brucei involves multiple arginine kinase isoforms with different subcellular locations. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Ooi, C.P.; Rotureau, B.; Gribaldo, S.; Georgikou, C.; Julkowska, D.; Blisnick, T.; Perrot, S.; Subota, I.; Bastin, P. The flagellar arginine kinase in Trypanosoma brucei is important for infection in tsetse flies. PLoS ONE 2015, 10, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Krassner, S.M.; Flory, B. Essential amino acids in the culture of Leishmania tarentolae. J. Parasitol. 1971, 57, 917–920. [Google Scholar] [CrossRef] [PubMed]
- Cadavid-Restrepo, G.; Gastardelo, T.S.; Faudry, E.; de Almeida, H.; Bastos, I.M.; Negreiros, R.S.; Lima, M.M.; Assumpção, T.C.; Almeida, K.C.; Ragno, M.; et al. The major leucyl aminopeptidase of Trypanosoma cruzi (LAPTc) assembles into a homohexamer and belongs to the M17 family of metallopeptidases. BMC Biochem. 2011, 12. [Google Scholar] [CrossRef] [PubMed]
- Timm, J.; Valente, M.; García-Caballero, D.; Wilson, K.S.; González-Pacanowska, D. Structural characterization of acidic M17 leucine aminopeptidases from the TriTryps and evaluation of their role in nutrient starvation in Trypanosoma brucei. mSphere 2017, 2, e00226-16. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, T.; Nishida, N.; Bakthavatsalam, S.; Kataoka, K.; Takada, H.; Yoshimura, T.; Soda, K.; Esaki, N. The purification, characterization, cloning and sequencing of the gene for a halostable and thermostable leucine dehydrogenase from Thermoactinomyces intermedius. Eur. J. Biochem. 1994, 222, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, T.; Yamanaka, K.; Kazuoka, T.; Kanzawa, N.; Soda, K. Psychrophilic valine dehydrogenase of the antarctic psychrophile, Cytophaga sp. KUC-1. Eur. J. Biochem. 2001, 4383, 4375–4383. [Google Scholar] [CrossRef]
- Bixel, M.G.; Hutson, S.M.; Hamprecht, B. Cellular distribution of branched-chain amino acid aminotransferase isoenzymes among rat brain glial cells in culture. J. Histochem. Cytochem. 1997, 45, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Brosnan, J.T.; Brosnan, M.E. Branched-chain amino acids: Metabolism, physiological function, and application. J. Nutr. 2006, 3, 207s–211s. [Google Scholar] [CrossRef]
- Ikeda, Y.; Tanaka, K. Purification and characterization of isovaleryl coenzyme A dehydrogenase from rat liver mitochondria. J. Biol. Chem. 1983, 258, 1077–1085. [Google Scholar] [PubMed]
- Pohl, B.; Raichle, T.; Ghisla, S. Studies on the reaction mechanism of general acyl-CoA dehydrogenase: Determination of selective isotope effects in the dehydrogenation of butyryl-CoA. Eur. J. Biochem. 1986, 160, 109–115. [Google Scholar] [CrossRef] [PubMed]
- Ghisla, S.; Thorpe, C. Acyl-CoA dehydrogenases: A mechanistic overview. Eur. J. Biochem. 2004, 271, 494–508. [Google Scholar] [CrossRef] [PubMed]
- Mancilla, R.; Naquira, C.; Lanas, C. Protein biosynthesis in trypanosomidae. Exp. Parasitol. 1967, 21, 154–159. [Google Scholar] [CrossRef]
- Blum, J.J. Oxidation of leucine by Leishmania donovani. J. Protozool. 1991, 38, 527–531. [Google Scholar] [CrossRef] [PubMed]
- Ginger, M.L.; Chance, M.L.; Goad, L.J. Elucidation of carbon sources used for the biosynthesis of fatty acids and sterols in the trypanosomatid Leishmania mexicana. Biochem. J. 1999, 342, 397–405. [Google Scholar] [CrossRef] [PubMed]
- Ginger, M.L.; Prescott, M.C.; Reynolds, D.G.; Chance, M.L.; Goad, J.L. Utilization of leucine and acetate as carbon sources for sterol and fatty acid biosynthesis by Old and New World Leishmania species, Endotrypanum monterogeii and Trypanosoma cruzi. Eur. J. Biochem. 2000, 267, 2555–2566. [Google Scholar] [CrossRef] [PubMed]
- Ginger, M.L.; Chance, M.L.; Sadler, I.H.; Goad, L.J. The biosynthetic incorporation of the intact leucine skeleton into sterol by the trypanosomatid Leishmania mexicana. J. Biol. Chem. 2001, 276, 11674–11682. [Google Scholar] [CrossRef] [PubMed]
- Nes, C.R.; Singha, U.K.; Liu, J.; Ganapathy, K.; Villalta, F.; Waterman, M.R.; Lepesheva, G.I.; Chaudhuri, M.; Nes, W.D. Novel sterol metabolic network of Trypanosoma brucei procyclic and bloodstream forms. Biochem. J. 2012, 443, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Riviere, L.; Moreau, P.; Allmann, S.; Hahn, M.; Biran, M.; Plazolles, N.; Franconi, J.-M.; Boshart, M.; Bringaud, F. Acetate produced in the mitochondrion is the essential precursor for lipid biosynthesis in procyclic trypanosomes. Proc. Natl. Acad. Sci. USA 2009, 106, 12694–12699. [Google Scholar] [CrossRef] [PubMed]
- Millerioux, Y.; Ebikeme, C.; Biran, M.; Morand, P.; Bouyssou, G.; Vincent, I.M.; Mazet, M.; Riviere, L.; Franconi, J.M.; Burchmore, R.J.S.; et al. The threonine degradation pathway of the Trypanosoma brucei procyclic form: The main carbon source for lipid biosynthesis is under metabolic control. Mol. Microbiol. 2013, 90, 114–129. [Google Scholar] [PubMed]
- Montamat, E.E.; Arauzo, S.S.; Blanco, A. Subcellular localization of leucine aminotransferase and a-hydroxyacid dehydrogenase in Trypanosoma cruzi. Mol. Biochem. Parasitol. 1987, 22, 185–193. [Google Scholar] [CrossRef]
- Manchola, N.C.; Silber, A.M.; Nowicki, C. The non-canonical substrates of Trypanosoma cruzi tyrosine and aspartate aminotransferases: Branched-chain amino acids. J. Eukaryot. Microbiol. 2018, 65, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Contreras, V.T.; Salles, J.M.; Thomas, N.; Morel, C.M.; Goldenberg, S. In vitro differentiation of Trypanosoma cruzi under chemically defined conditions. Mol. Biochem. Parasitol. 1985, 16, 315–327. [Google Scholar] [CrossRef]
- Berger, L.C.; Wilson, J.; Wood, P.; Berger, J.; Berger, B.J. Methionine regeneration and aspartate aminotransferase in parasitic protozoa. J. Bacteriol. 2001, 183, 4421–4434. [Google Scholar] [CrossRef] [PubMed]
- Marciano, D.; Llorente, C.; Maugeri, D.A.; de la Fuente, C.; Opperdoes, F.; Cazzulo, J.J.; Nowicki, C. Biochemical characterization of stage-specific isoforms of aspartate aminotransferases from Trypanosoma cruzi and Trypanosoma brucei. Mol. Biochem. Parasitol. 2008, 161, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Creek, D.J.; Mazet, M.; Achcar, F.; Anderson, J.; Kim, D.H.; Kamour, R.; Morand, P.; Millerioux, Y.; Biran, M.; Kerkhoven, E.J.; et al. Probing the metabolic network in bloodstream-form Trypanosoma brucei using untargeted metabolomics with stable isotope labelled glucose. PLoS Pathog. 2015, 11, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Loureiro, I.; Faria, J.; Clayton, C.; Ribeiro, S.M.; Roy, N.; Santarem, N.; Tavares, J.; Cordeiro-da-Silva, A.; Santarem, N.; Tavares, J.; et al. Knockdown of asparagine synthetase A renders Trypanosoma brucei auxotrophic to asparagine. PLoS Negl. Trop. Dis. 2013, 7, e2578. [Google Scholar] [CrossRef]
- Faria, J.; Loureiro, I.; Santarém, N.; Macedo-ribeiro, S.; Tavares, J.; Cordeiro-da-Silva, A. Leishmania infantum asparagine synthetase A is dispensable for parasites survival and infectivity. PLoS Negl. Trop. Dis. 2016, 10, e0004365. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Srivastava, A.; Jha, P.; Sinha, K.K.; Kundu, B. Asparaginase as a new molecular target against leishmaniasis: Insights into the mechanism of action and structure-based inhibitor design. Mol. Biosyst. 2015, 11, 1887–1896. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Ali, V.; Pratap Singh, K.; Gupta, P.; Suman, S.S.; Ghosh, A.K.; Bimal, S.; Pandey, K.; Das, P. Deciphering the interplay between cysteine synthase and thiol cascade proteins in modulating Amphotericin B resistance and survival of Leishmania donovani under oxidative stress. Redox Biol. 2017, 12, 350–366. [Google Scholar] [CrossRef] [PubMed]
- Finkelstein, J.D. Methionine metabolism in mammals. J. Nutr. Biochem. 1990, 1, 228–237. [Google Scholar] [CrossRef]
- Reguera, R.M.; Redondo, C.M.; Pérez-Pertejo, Y.; Balaña-Fouce, R. S-adenosylmethionine in protozoan parasites: Functions, synthesis and regulation. Mol. Biochem. Parasitol. 2007, 152, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Ciman, M.; Siliprandi, N. On the oxidation of a-oxibutyrate by isolated mammalian mitochondria. Biochim. Biophys. Acta 1968, 162, 164–169. [Google Scholar] [CrossRef]
- Bremer, J. Pyruvate dehydrogenase, substrate specificity and product inhibition. Eur. J. Biochem. 1969, 8, 535–540. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.C.; Banerjee, R.V.; Huang, S.; Sumner, J.S.; Matthews, R.G. Comparison of cobalamin-independent and cobalamin-dependent methionine synthases from Escherichia coli: Two solutions to the same chemical problem. Biochemistry 1992, 31, 6045–6056. [Google Scholar] [CrossRef] [PubMed]
- Cazzulo, J.J. Amino acid and protein metabolism. In Molecular Medical Parasitology; Academic Press: Cambridge, MA, USA, 2003; pp. 171–195. [Google Scholar]
- Walker, J.; Barrett, J. Parasite sulphur amino acid metabolism. Int. J. Parasitol. 1997, 27, 883–897. [Google Scholar] [CrossRef]
- Pegg, A.E. S-Adenosylmethionine decarboxylase: A brief review. Cell Biochem. Funct. 1984, 2, 11–15. [Google Scholar] [CrossRef] [PubMed]
- Pegg, A.E.; Xiong, H.; Feith, D.J.; Shantz, L.M. S-adenosylmethionine decarboxylase: Structure, function and regulation by polyamines. Biochem. Soc. Trans. 1998, 26, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Berger, B.J.; Dai, W.W.; Wang, H.; Stark, R.E.; Cerami, A. Aromatic amino acid transamination and methionine recycling in trypanosomatids. Proc. Natl. Acad. Sci. USA 1996, 93, 4126–4130. [Google Scholar] [CrossRef] [PubMed]
- Moskovitz, J. Methionine sulfoxide reductases: Ubiquitous enzymes involved in antioxidant defense, protein regulation, and prevention of aging-associated diseases. Biochim. Biophys. Acta Proteins Proteom. 2005, 1703, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Reguera, R.M.; Perez-Pertejo, Y.; Ordóñez, C.; Cubría, J.C.; Tekwani, B.L.; Balaña-Fouce, R.; Ordóñez, D. S-adenosylmethionine synthesis in Leishmania infantum. Cell Biol. Int. 1999, 23, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Reguera, R.M.; Balaña-Fouce, R.; Pérez-Pertejo, Y.; Fernández, F.J.; García-Estrada, C.; Cubría, J.C.; Ordóñez, C.; Ordóñez, D. Cloning expression and characterization of methionine adenosyltransferase in Leishmania infantum promastigotes. J. Biol. Chem. 2002, 277, 3158–3167. [Google Scholar] [CrossRef] [PubMed]
- Willert, E.K.; Fitzpatrick, R.; Phillips, M.A. Allosteric regulation of an essential trypanosome polyamine biosynthetic enzyme by a catalytically dead homolog. Proc. Natl. Acad. Sci. USA 2007, 104, 8275–8280. [Google Scholar] [CrossRef] [PubMed]
- Willert, E.K.; Phillips, M.A. Regulated expression of an essential allosteric activator of polyamine biosynthesis in African trypanosomes. PLoS Pathog. 2008, 4, e1000183. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Singh, S.P.; Agnihotri, P.; Pratap, J.V. Characterization of a novel putative S-adenosylmethionine decarboxylase-like protein from Leishmania donovani. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Roberts, S.C.; Scott, J.; Gasteier, J.E.; Jiang, Y.; Brooks, B.; Jardim, A.; Carter, N.S.; Heby, O.; Ullman, B. S-adenosylmethionine decarboxylase from Leishmania donovani: Molecular, genetic, and biochemical characterization of null mutants and overproducers. J. Biol. Chem. 2002, 277, 5902–5909. [Google Scholar] [CrossRef] [PubMed]
- Sansom, F.M.; Tang, L.; Ralton, J.E.; Saunders, E.C.; Naderer, T.; McConville, M.J. Leishmania major methionine sulfoxide reductase A Is required for resistance to oxidative stress and efficient replication in macrophages. PLoS ONE 2013, 8, 15–17. [Google Scholar] [CrossRef] [PubMed]
- Bitonti, A.J.; Dumont, J.A.; McCann, P.P. Characterization of Trypanosoma brucei brucei S-adenosyl-L-methionine decarboxylase and its inhibition by berenil, pentamidine and methylglyoxal bis(guanylhydrazone). Biochem. J. 1986, 237, 685–689. [Google Scholar] [CrossRef] [PubMed]
- Yarlett, N.; Bacchi, C.J. Effect of dl-α-difluoromethylornithine on methionine cycle intermediates in Trypanosoma brucei brucei. Mol. Biochem. Parasitol. 1988, 27, 1–10. [Google Scholar] [CrossRef]
- Yarlett, N.; Garofalo, J.; Goldberg, B.; Ciminelli, M.A.; Ruggiero, V.; Sufrin, J.R.; Bacchi, C.J. S-Adenosylmethionine synthetase in bloodstream Trypanosoma brucei. BBA—Mol. Basis Dis. 1993, 1181, 68–76. [Google Scholar] [CrossRef]
- Bacchi, C.J.; Goldberg, B.; Garofalo-Hannan, J.; Rattendi, D.; Lyte, P.; Yarlett, N. Fate of soluble methionine in African trypanosomes: Effects of metabolic inhibitors. Biochem. J. 1995, 309, 737–743. [Google Scholar] [CrossRef] [PubMed]
- Tekwani, B.L.; Bacchi, C.J.; Pegg, A.E. Putrescine activated S-adenosylmethionine decarboxylase from Trypanosoma brucei brucei. Mol. Cell. Biochem. 1992, 117, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Willert, E.K.; Phillips, M.A. Cross-species activation of trypanosome S-adenosylmethionine decarboxylase by the regulatory subunit prozyme. Mol. Biochem. Parasitol. 2009, 168, 1–6. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Arias, D.G.; Cabeza, M.S.; Erben, E.D.; Carranza, P.G.; Lujan, H.D.; Iñón, M.T.T.; Iglesias, A.A.; Guerrero, S.A. Functional characterization of methionine sulfoxide reductase A from Trypanosoma spp. Free Radic. Biol. Med. 2011, 50, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Guerrero, S.A.; Arias, D.G.; Cabeza, M.S.; Law, M.C.Y.; D’Amico, M.; Kumar, A.; Wilkinson, S.R. Functional characterisation of the methionine sulfoxide reductase repertoire in Trypanosoma brucei. Free Radic. Biol. Med. 2017, 112, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Kinch, L.N.; Scott, J.R.; Ullman, B.; Phillips, M.A. Cloning and kinetic characterization of the Trypanosoma cruzi. Mol. Biochem. Parasitol. 1999, 101, 1–11. [Google Scholar] [CrossRef]
- Parker, N.B.; Yang, X.; Hanke, J.; Mason, K.A.; Schowen, R.L.; Borchardt, R.T.; Yin, D.H. Trypanosoma cruzi: Molecular cloning and characterization of the S-adenosylhomocysteine hydrolase. Exp. Parasitol. 2003, 105, 149–158. [Google Scholar] [CrossRef] [PubMed]
- Duszenko, M.; Mühlstädt, K.; Broder, A. Cysteine is an essential growth factor for Trypanosoma brucei bloodstream forms. Mol. Biochem. Parasitol. 1992, 50, 269–273. [Google Scholar] [CrossRef]
- Baltz, T.; Baltz, D.; Giroud, C.; Crockett, J. Cultivation in a semi-defined medium of animal infective forms of Trypanosoma brucei, T. equiperdum, T. evansi, T. rhodesiense and T. gambiense. EMBO J. 1985, 4, 1273–1277. [Google Scholar] [PubMed]
- Williams, R.A.M.; Westrop, G.D.; Coombs, G.H. Two pathways for cysteine biosynthesis in Leishmania major. Biochem. J. 2009, 420, 451–462. [Google Scholar] [CrossRef] [PubMed]
- Abendroth, J.; Choi, R.; Wall, A.; Clifton, M.C.; Lukacs, C.M.; Staker, B.L.; Van Voorhis, W.; Myler, P.; Lorimer, D.D.; Edwards, T.E. Structures of aspartate aminotransferases from Trypanosoma brucei, Leishmania major and Giardia lamblia. Acta Crystallogr. Sect. F Struct. Biol. Commun. 2015, 71, 566–571. [Google Scholar] [CrossRef] [PubMed]
- Nozaki, T.; Shigeta, Y.; Saito-Nakano, Y.; Imada, M.; Kruger, W.D. Characterization of transsulfuration and cysteine biosynthetic pathways in the protozoan hemoflagellate, Trypanosoma cruzi: Isolation and molecular characterization of cystathionine gamma-synthase and serine acetyltransferase from trypanosoma. J. Biol. Chem. 2001, 276, 6516–6523. [Google Scholar] [CrossRef] [PubMed]
- Marciano, D.; Santana, M.; Nowicki, C. Functional characterization of enzymes involved in cysteine biosynthesis and H2S production in Trypanosoma cruzi. Mol. Biochem. Parasitol. 2012, 185, 114–120. [Google Scholar] [CrossRef] [PubMed]
- Romero, I.; Téllez, J.; Romanha, A.J.; Steindel, M.; Grisard, E.C. Upregulation of cysteine synthase and cystathionine β-synthase contributes to Leishmania braziliensis survival under oxidative stress. Antimicrob. Agents Chemother. 2015, 59, 4770–4781. [Google Scholar] [CrossRef] [PubMed]
- Giordana, L.; Mantilla, B.S.; Santana, M.; Silber, A.M.; Nowicki, C. Cystathionine γ-lyase, an enzyme related to the reverse transsulfuration pathway, is functional in Leishmania spp. J. Eukaryot. Microbiol. 2014, 61, 204–213. [Google Scholar] [CrossRef] [PubMed]
- Marciano, D.; Santana, M.; Mantilla, B.S.; Silber, A.M.; Marino-Buslje, C.; Nowicki, C. Biochemical characterization of serine acetyltransferase and cysteine desulfhydrase from Leishmania major. Mol. Biochem. Parasitol. 2010, 173, 170–174. [Google Scholar] [CrossRef] [PubMed]
- Wirtz, M.; Berkowitz, O.; Droux, M.; Hell, R. The cysteine synthase complex from plants: Mitochondrial serine acetyltransferase from Arabidopsis thaliana carries a bifunctional domain for catalysis and protein-protein interaction. Eur. J. Biochem. 2001, 268, 686–693. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, C.; Montemartini, M.; Duschak, V.; Santomé, J.A.; Cazzulo, J.J. Presence and subcellular localization of tyrosine aminotransferase and p-hydroxyphenyllactate dehydrogenase in epimastigotes of Trypanosoma cruzi. FEMS Microbiol. Lett. 1992, 92, 119–124. [Google Scholar] [CrossRef][Green Version]
- Montemartini, M.; Santomé, J.; Cazzulo, J.J.; Nowicki, C. Production of aromatic α-hydroxyacids by epimastigotes of Trypanosoma cruzi, and its possible role in NADH reoxidation. FEMS Microbiol. Lett. 1994, 118, 89–92. [Google Scholar] [CrossRef]
- Bontempi, E.J.; Aslund, L.; Porcel, B.M.; Segura, E.; Henriksson, J.; Örn, A.; Petterson, U.; Ruiz, A.M. Isolation and characterization of a gene from Trypanosoma cruzi encoding a 46-kilodalton prtein with homology to human and rat tyrosine aminotransferase. Mol. Biochem. Parasitol. 1993, 59, 253–262. [Google Scholar] [CrossRef]
- Blankenfeldt, W.; Nowicki, C.; Montemartini-Kalisz, M.; Kalisz, H.M.; Hecht, H.J. Crystal structure of Trypanosoma cruzi tyrosine aminotransferase: Substrate specificity is influenced by cofactor binding mode. Protein Sci. 1999, 8, 2406–2417. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, C.; Hunter, G.R.; Montemartini-Kalisz, M.; Blankenfeldt, W.; Hecht, H.J.; Kalisz, H.M. Recombinant tyrosine aminotransferase from Trypanosoma cruzi: Structural characterization and site directed mutagenesis of a broad substrate specificity enzyme. Biochim. Biophys. Acta Protein Struct. Mol. Enzymol. 2001, 1546, 268–281. [Google Scholar] [CrossRef]
- Cazzulo Franke, M.C.; Vernal, J.; Cazzulo, J.J.; Nowicki, C. The NAD-linked aromatic α-hydroxy acid dehydrogenase from Trypanosoma cruzi. A new member of the cytosolic malate dehydrogenases group without malate dehydrogenase activity. Eur. J. Biochem. 1999, 266, 903–910. [Google Scholar] [CrossRef] [PubMed]
- Nowicki, C.; Cazzulo, J.J. Aromatic amino acid catabolism in trypanosomatids. Comp. Biochem. Physiol. Part A Mol. Integr. Physiol. 2008, 151, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, A.N.; Ghosh, J.J. Transaminases of Leishmania donovani, the causative organism of Kala-azar. Nature 1957, 180, 1425. [Google Scholar] [CrossRef] [PubMed]
- Le Blancq, S.M.; Lanham, S.M. Aspartate aminotransferase in Leishmania is a broad-spectrum transaminase. Trans. R. Soc. Trop. Med. Hyg. 1984, 78, 373–375. [Google Scholar] [CrossRef]
- Vernal, J.; Jose, J.; Nowicki, C. Isolation and partial characterization of a broad specificity aminotransferase from Leishmania mexicana promastigotes. Mol. Biochem. Parasitol. 1998, 96, 83–92. [Google Scholar] [CrossRef]
- Vernal, J.; Cazzulo, J.J.; Nowicki, C. Cloning and heterologous expression of a broad specificity aminotransferase of Leishmania mexicana promastigotes. FEMS Microbiol. Lett. 2003, 229, 217–222. [Google Scholar] [CrossRef]
- Lye, L.F.; Kang, S.O.; Nosanchuk, J.D.; Casadevall, A.; Beverley, S.M. Phenylalanine hydroxylase (PAH) from the lower eukaryote Leishmania major. Mol. Biochem. Parasitol. 2011, 175, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Stibbs, H.H.; Seed, J.R. Further studies on the metabolism of tryptophan in Trypanosoma brucei gambiense: Cofactors, inhibitors, and end-products. Specialia 1974, 31, 274–278. [Google Scholar]
- Stibbs, H.H.; Seed, J.R. Short-term metabolism of (14-C) tryptophan in rats infected with Trypanosoma brucei gambiense. J. Infect. Dis. 1975, 131, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Stibbs, H.H.; Seed, J.R. Metabolism of tyrosine and phenylalanine in Trypanosoma brucei gambiense. Int. J. Biochem. 1975, 6, 197–203. [Google Scholar] [CrossRef]
- Seed, J.R.; Hall, J.E.; Sechelski, J. Phenylalanine metabolism in Microtus montanus chronically infected with Trypanosoma brucei gambiense. Comp. Biochem. Physiol. B 1982, 71, 209–215. [Google Scholar] [CrossRef]
- Hall, J.E.; Seed, J.R. Increased urinary excretion of aromatic amino acid catabolites by Microtus montanus chronically infected with Trypanosoma brucei gambiense. Compative Biochem. 1984, 77, 755–760. [Google Scholar] [CrossRef]
- El Sawalhy, A.; Seed, J.R.; Hall, J.E.; El Attar, H. Increased excretion of aromatic amino acid catabolites in animals infected with Trypanosoma brucei evansi. J. Parasitol. 1998, 84, 469–473. [Google Scholar] [CrossRef] [PubMed]
- McGettrick, A.F.; Corcoran, S.E.; Barry, P.J.G.; McFarland, J.; Crès, C.; Curtis, A.M.; Franklin, E.; Corr, S.C.; Mok, K.H.; Cummins, E.P.; et al. Trypanosoma brucei metabolite indolepyruvate decreases HIF-1α and glycolysis in macrophages as a mechanism of innate immune evasion. Proc. Natl. Acad. Sci. USA 2016, 113, E7778–E7787. [Google Scholar] [CrossRef] [PubMed]
- Schirch, V.; Szebenyi, D.M.E. Serine hydroxymethyltransferase revisited. Curr. Opin. Chem. Biol. 2005, 9, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Salcedo, E.; Sims, P.F.G.; Hyde, J.E. A glycine-cleavage complex as part of the folate one-carbon metabolism of Plasmodium falciparum. Trends Parasitol. 2005, 21, 406–411. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hampton, J.R. Serine metabolism in Trypanosoma cruzi: Synthesis of other amino acids. Comp. Biochem. Physiol. B 1971, 39, 999–1003. [Google Scholar] [CrossRef]
- Nosei, C.; Avila, J. Serine hydroxymethyltransferase activity in Trypanosoma cruzi, Trypanosoma rangeli and American Leishmania spp. Comp. Biochem. Physiol. Part B Biochem. 1985, 81, 701–704. [Google Scholar] [CrossRef]
- Gagnon, D.; Foucher, A.; Girard, I.; Ouellette, M. Stage specific gene expression and cellular localization of two isoforms of the serine hydroxymethyltransferase in the protozoan parasite Leishmania. Mol. Biochem. Parasitol. 2006, 150, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Roy, G.; Ouellette, M. Inactivation of the cytosolic and mitochondrial serine hydroxymethyl transferase genes in Leishmania major. Mol. Biochem. Parasitol. 2015, 204, 106–110. [Google Scholar] [CrossRef] [PubMed]
- Capelluto, D.G.; Hellman, U.; Cazzulo, J.J.; Cannata, J.J. Purification and some properties of serine hydroxymethyltransferase from Trypanosoma cruzi. Eur. J. Biochem. 2000, 267, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Scott, D.A.; Hickerson, S.M.; Vickers, T.J.; Beverley, S.M. The role of the mitochondrial glycine cleavage complex in the metabolism and virulence of the protozoan parasite Leishmania major. J. Biol. Chem. 2008, 283, 155–165. [Google Scholar] [CrossRef] [PubMed]
- Danson, M.J.; Conroy, K.; McQuattie, A.; Stevenson, K.J. Dihydrolipoamide dehydrogenase from Trypanosoma brucei. Characterization and cellular location. Biochem. J. 1987, 243, 661–665. [Google Scholar] [CrossRef] [PubMed]
- Cook, I.D.; Jackman, S.A.; Danson, M.J.; Eisenthal, R.; Hough, D.W.; Whish, W.J. Identification of dihydrolipoamide dehydrogenase in the procyclic form of Trypanosoma brucei. Biochem. Soc. Trans. 1990, 18, 862–863. [Google Scholar] [CrossRef] [PubMed]
- Jackman, S.A.; Hough, D.W.; Danson, M.J.; Stevenson, K.J.; Opperdoes, F.R. Subcellular localisation of dihydrolipoamide dehydrogenase and detection of lipoic acid in bloodstream forms of Trypanosoma brucei. Eur. J. Biochem. 1990, 193, 91–95. [Google Scholar] [CrossRef] [PubMed]
- Else, A.J.; Hough, D.W.; Danson, M.J. Cloning, sequencing, and expression of Trypanosoma brucei dihydrolipoamide dehydrogenase. Eur. J. Biochem. 1993, 212, 423–429. [Google Scholar] [CrossRef] [PubMed]
- Roldán, A.; Comini, M.A.; Crispo, M.; Krauth-Siegel, R.L. Lipoamide dehydrogenase is essential for both bloodstream and procyclic Trypanosoma brucei. Mol. Microbiol. 2011, 81, 623–639. [Google Scholar] [CrossRef] [PubMed]
- Lohrer, H.; Krauth-Siegel, R.L. Purification and characterization of lipoamide dehydrogenase from Trypanosoma cruzi. Eur. J. Biochem. 1990, 194, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Else, A.J.; Clarke, J.F.; Willis, A.; Jackman, S.A.; Hough, D.W.; Danson, M.J. Dihydrolipoamide dehydrogenase in the Trypanosoma subgenus, Trypanozoon. Mol. Biochem. Parasitol. 1994, 64, 233–239. [Google Scholar] [CrossRef]
- Gutiérrez-Correa, J. Trypanosoma cruzi dihydrolipoamide dehydrogenase as target of reactive metabolites generated by cytochrome c/hydrogen peroxide (or linoleic acid hydroperoxide)/phenol systems. Free Radic. Res. 2010, 44, 1345–1358. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, P.F.; Moreira, D.S.; Baba, E.H.; Volpe, C.M.O.; Ruiz, J.C.; Romanha, A.J.; Murta, S.M.F. Molecular characterization of lipoamide dehydrogenase gene in Trypanosoma cruzi populations susceptible and resistant to benznidazole. Exp. Parasitol. 2016, 170, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cross, G.A.M.; Klein, R.A.; Linstea, P.J.D. Utilization of amino acids by Trypanosoma brucei in culture: L-threonine as a precursor for acetate. Parasitology 1975, 71, 311–313. [Google Scholar] [CrossRef] [PubMed]
- Millerioux, Y.; Morand, P.; Biran, M.; Mazet, M.; Moreau, P.; Wargnies, M.; Ebikeme, C.; Deramchia, K.; Gales, L.; Portais, J.C.; et al. ATP synthesis-coupled and -uncoupled acetate production from acetyl-CoA by mitochondrial acetate: Succinate CoA-transferase and acetyl-CoA thioesterase in Trypanosoma. J. Biol. Chem. 2012, 287, 17186–17197. [Google Scholar] [CrossRef] [PubMed]
- Klein, R.A.; Linstead, D.J. Threonine as a preferred source of 2 carbon units for lipid synthesis in Trypanosoma brucei. Biochem. Soc. Trans. 1976, 4, 48–50. [Google Scholar] [CrossRef] [PubMed]
- Simon, M.W.; Jayasimhulu, K.; Mukkada, A.J. The free amino acid pool in Leishmania tropica promastigotes. Mol. Biochem. Parasitol. 1983, 9, 47–57. [Google Scholar] [CrossRef]
- Saunders, E.C.; Ng, W.W.; Chambers, J.M.; Ng, M.; Naderer, T.; Krömer, J.O.; Likić, V.A.; McConville, M.J. Isotopomer profiling of Leishmania mexicana promastigotes reveals important roles for succinate fermentation and aspartate uptake in Tricarboxylic Acid Cycle (TCA) anaplerosis, glutamate synthesis, and growth. J. Biol. Chem. 2011, 286, 27706–27717. [Google Scholar] [CrossRef] [PubMed]
- Ong, H.B.; Lee, W.S.; Patterson, S.; Wyllie, S.; Fairlamb, A.H. Homoserine and quorum-sensing acyl homoserine lactones as alternative sources of threonine: A potential role for homoserine kinase in insect-stage Trypanosoma brucei. Mol. Microbiol. 2015, 95, 143–156. [Google Scholar] [CrossRef] [PubMed]
- Williamson, J.; Desowitz, R.S. Chemical composition of Trypanosomes.1. protein, amino acid and sugar analysis. Exp. Parasitol. 1961, 11, 161–175. [Google Scholar] [CrossRef]
- Chappell, B.Y.L.H.; Southworth, G.C.; Read, C.P. Short-interval absorption and metabolism of some amino acids in Trypanosotna gambiense. Parasitology 1972, 64, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Panizzutti, R.; De Souza Leite, M.; Pinheiro, C.M.; Meyer-Fernandes, J.R. The occurrence of free D-alanine and an alanine racemase activity in Leishmania amazonensis. FEMS Microbiol. Lett. 2006, 256, 16–21. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Frydman, B.; de los Santos, C.; Cannata, J.J.B.; Cazzulo, J.J. Carbon-13 nuclear magnetic resonance analysis of [1-13C]glucose metabolism in Trypanosoma cruzi. Eur. J. Biochem. 1990, 192, 363–368. [Google Scholar] [CrossRef] [PubMed]
- Westrop, G.D.; Williams, R.A.M.; Wang, L.; Zhang, T.; Watson, D.G.; Silva, A.M.; Coombs, G.H. Metabolomic analyses of Leishmania reveal multiple species differences and large differences in amino acid metabolism. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Newport, G.R.; Page, C.R. Free amino acids in brain, liver, and skeletal muscle tissue of voles infected with Trypanosoma brucei gambiense. J. Parasitol. 1977, 63, 1060–1064. [Google Scholar] [CrossRef] [PubMed]
- Newport, G.R.; Page, C.R.; Ashman, P.U.; Stibbs, H.H.; Seed, J.R. Alteration of free serum amino acids in voles infected with Trypanosoma brucei gambiense. J. Parasitol. 1977, 63, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Isoun, T.T.; Isoun, M.J.; Anosa, V.O. Free plasma amino acid profiles of normal and Trypanosoma vivax infected sheep. Tropenmed. Parasitol. 1978, 29, 330–334. [Google Scholar] [PubMed]
- Bursell, E. Aspects of the metabolism of amino acids in the tsetse fly, Glossina (Diptera). J. Insect Physiol. 1963, 9, 439–452. [Google Scholar] [CrossRef]
- Spitznagel, D.; Ebikeme, C.; Biran, M.; Nicabháird, N.; Bringaud, F.; Henehan, G.T.M.; Nolan, D.P. Alanine aminotransferase of Trypanosoma brucei- a key role in proline metabolism in procyclic life forms. FEBS J. 2009, 276, 7187–7199. [Google Scholar] [CrossRef] [PubMed]
- Bentel, M.; Harder, S.; Wiesgigl, M.; Heukeshoven, J.; Gelhaus, C.; Krause, E.; Clos, J.; Bruchhaus, I. Developmentally induced changes of the proteome in the protozoan parasite Leishmania donovani. Proteomics 2003, 3, 1811–1829. [Google Scholar] [CrossRef] [PubMed]
- Antunes, L.C.M.; Han, J.; Pan, J.; Moreira, C.J.C.; Azambuja, P.; Borchers, C.H.; Carels, N. Metabolic signatures of triatomine vectors of Trypanosoma cruzi unveiled by metabolomics. PLoS ONE 2013, 8, e77283. [Google Scholar] [CrossRef] [PubMed]
- Harington, J.S. Histamine and Histidine in excreta of the blood-sucking bug Rhodnius prolixus. Nature 1956, 178, 268. [Google Scholar] [CrossRef] [PubMed]
- Michael Barrett, F. Changes in the concentration of free amino acids in the haemolymph of Rhodnius prolixus during the fifth instar. Comp. Biochem. Physiol. Part B Biochem. 1974, 48, 241–250. [Google Scholar] [CrossRef]
- Silber, A.M.; Colli, W.; Ulrich, H.; Alves, M.J.M.; Pereira, C.A.; Manso Alves, M.; Pereira, C.A. Amino acid metabolic routes in Trypanosoma cruzi: Possible therapeutic targets against Chagas’ disease. Curr. Drug Target Infect. Disord. 2005, 5, 53–64. [Google Scholar] [CrossRef][Green Version]
- Silva Paes, L.; Mantilla, B.A.; Barison, M.J.; Wrenger, C.; Silber, A.M. The uniqueness of the Trypanosoma cruzi mitochondrion: Opportunities to target new drugs against Chagas’ disease. Curr. Pharm. Des. 2011, 17, 2074–2099. [Google Scholar] [CrossRef]
- Magdaleno, A.; Ahn, I.Y.; Paes, L.S.; Silber, A.M. Actions of a proline analogue, L-thiazolidine-4-carboxylic acid (T4C), on Trypanosoma cruzi. PLoS ONE 2009, 4. [Google Scholar] [CrossRef] [PubMed]
- Magdaleno, A.; Suárez Mantilla, B.; Rocha, S.C.; Pral, E.M.F.; Silber, A.M. The involvement of glutamate metabolism in the resistance to thermal, nutritional, and oxidative stress in Trypanosoma cruzi. Enzym. Res. 2011, 2011, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Krassner, S.M.; Granger, B.; Phermsangngnam, P.; Le, T.; Linden, V. Further studies on substrates inducing metacyclogenesis in Trypanosoma cruzi. J. Protozool. 1990, 37, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Martins, R.M.; Covarrubias, C.; Rojas, R.G.; Silber, A.M.; Yoshida, N. Use of L-proline and ATP production by Trypanosoma cruzi metacyclic forms as requirements for host cell invasion. Infect. Immun. 2009, 77, 3023–3032. [Google Scholar] [CrossRef] [PubMed]
- Schenkman, S.; Robbins, E.S.; Nussenzweig, V. Attachment of Trypanosoma cruzi to mammalian cells requires parasite energy, and invasion can be independent of the target cell cytoskeleton. Infect. Immun. 1991, 59, 645–654. [Google Scholar] [PubMed]
- Silber, A.M.; Tonelli, R.R.; Lopes, C.G.; Cunha-e-Silva, N.; Torrecilhas, A.C.T.; Schumacher, R.I.; Colli, W.; Alves, M.J.M. Glucose uptake in the mammalian stages of Trypanosoma cruzi. Mol. Biochem. Parasitol. 2009, 168, 102–108. [Google Scholar] [CrossRef] [PubMed]
- Shah-Simpson, S.; Lentini, G.; Dumoulin, P.C.; Burleigh, B.A. Modulation of host central carbon metabolism and in situ glucose uptake by intracellular Trypanosoma cruzi amastigotes. PLoS Pathog. 2017, 13, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Toledo, D.A.M.; Roque, N.R.; Teixeira, L.; Milán-Garcés, E.A.; Carneiro, A.B.; Almeida, M.R.; Andrade, G.F.S.; Martins, J.S.; Pinho, R.R.; Freire-de-Lima, C.G.; et al. Lipid body organelles within the parasite Trypanosoma cruzi: A role for intracellular arachidonic acid metabolism. PLoS ONE 2016, 11, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Gazos-Lopes, F.; Martin, J.L.; Dumoulin, P.C.; Burleigh, B.A. Host triacylglycerols shape the lipidome of intracellular trypanosomes and modulate their growth. PLoS Pathog. 2017, 13, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Ley, V.; Robbins, E.S.; Nussenzweig, V.; Andrews, N.W.; Andrews, N.W. The exit of Trypanosoma cruzi from the phagosome is inhibited by raising the pH of acidic compartments. J. Exp. Med. 1990, 171, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Bustos, T.; Potapenko, E.; Storey, M.; Docampo, R. An intracellular ammonium transporter is necessary for replication, differentiation, and resistance to starvation and osmotic stress in Trypanosoma cruzi. mSphere 2018, 3, e00377-17. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, L.; Ferreira, M.A.; Cosson, A.; Batista, M.M.; Batista Dda, G.; Minoprio, P.; Degrave, W.M.; Berneman, A.; Soeiro Mde, N. Inhibition of Trypanosoma cruzi proline racemase affects host-parasite interactions and the outcome of in vitro infection. Mem. Inst. Oswaldo Cruz 2009, 104, 1055–1062. [Google Scholar] [CrossRef] [PubMed]
- Coatnoan, N.; Berneman, A.; Chamond, N.; Minoprio, P. Proline racemases: Insights into Trypanosoma cruzi peptides containing D-proline. Mem. Inst. Oswaldo Cruz 2009, 104, 295–300. [Google Scholar] [CrossRef] [PubMed]
- Buschiazzo, A.; Goytia, M.; Schaeffer, F.; Degrave, W.; Shepard, W.; Grégoire, C.; Chamond, N.; Cosson, A.; Berneman, A.; Coatnoan, N.; et al. Crystal structure, catalytic mechanism, and mitogenic properties of Trypanosoma cruzi proline racemase. Proc. Natl. Acad. Sci. USA 2006, 103, 1705–1710. [Google Scholar] [CrossRef] [PubMed]
- Ryley, J.F. Studies on the metabolism of the protozoa. 9. Comparative metabolism of bloodstream and culture forms of Trypanosoma rhodiense. Biochemistry 1962, 85, 211–223. [Google Scholar] [CrossRef]
- Hannaert, V.; Michels, P.A.M. Structure, function, and biogenesis of glycosomes in Kinetoplastida. J. Bioenerg. Biomembr. 1994, 26, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Vassella, E.; Reuner, B.; Yutzy, B.; Boshart, M. Differentiation of African trypanosomes is controlled by a density sensing mechanism which signals cell cycle arrest via the cAMP pathway. J. Cell Sci. 1997, 110, 2661–2671. [Google Scholar] [PubMed]
- Van Grinsven, K.W.A.; Van Den Abbeele, J.; Van Den Bossche, P.; Van Hellemond, J.J.; Tielens, A.G.M. Adaptations in the glucose metabolism of procyclic Trypanosoma brucei isolates from tsetse flies and during differentiation of bloodstream forms. Eukaryot. Cell 2009, 8, 1307–1311. [Google Scholar] [CrossRef] [PubMed]
- Maudlin, I. Infection of tsetse with Trypanosoma brucei rhodesiense and T. b. gambiense. In The Molecular Biology of Insect Disease Vectors; Springer: Dordrecht, The Netherlands, 1997; pp. 136–145. [Google Scholar]
- Overath, P.; Czichos, J.; Haas, C. The effect of citrate/cis-aconitate on oxidative metabolism during transformation of Trypanosoma brucei. Eur. J. Biochem. 1986, 160, 175–182. [Google Scholar] [CrossRef] [PubMed]
- Ziegelbauer, K.; Quinten, M.; Schwarz, H.; Pearson, T.W.; Overath, P. Synchronous differentiation of Trypanosoma brucei from bloodstream to procyclic forms in vitro. Eur. J. Biochem. 1990, 192, 373–378. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, B.; Biswas, S.; Mondal, S.; Bera, T. Stage specific developmental changes in the mitochondrial and surface membrane associated redox systems of Leishmania donovani promastigote and amastigote. Biochemistry 2010, 75, 494–518. [Google Scholar] [CrossRef] [PubMed]
- Warburg, A.; Schlein, Y. The effect of post-bloodmeal nutrition of Phlebotomus papatasi on the transmission of Leishmania major. Am. J. Trop. Med. Hyg. 1986, 35, 926–930. [Google Scholar] [CrossRef] [PubMed]
- Saunders, E.C.; Ng, W.W.; Kloehn, J.; Chambers, J.M.; Ng, M.; Mcconville, M.J. Induction of a stringent metabolic response in intracellular stages of Leishmania mexicana leads to increased dependence on mitochondrial metabolism. PLoS Pathog. 2014, 10. [Google Scholar] [CrossRef] [PubMed]
- McConville, M.J.; Saunders, E.C.; Kloehn, J.; Dagley, M.J. Leishmania carbon metabolism in the macrophage phagolysosome-feast or famine? F1000Research 2015, 4, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Saunders, E.C.; Naderer, T.; Chambers, J.; Landfear, S.M.; McConville, M.J. Leishmania mexicana can utilize amino acids as major carbon sources in macrophages but not in animal models. Mol. Microbiol. 2018, 1–29. [Google Scholar] [CrossRef] [PubMed]
- Debrabant, A.; Joshi, M.B.; Pimenta, P.F.P.; Dwyer, D.M. Generation of Leishmania donovani axenic amastigotes: Their growth and biological characteristics. Int. J. Parasitol. 2004, 34, 205–217. [Google Scholar] [CrossRef] [PubMed]
- Diaz, E.; Zacarias, A.K.; Pérez, S.; Vanegas, O.; Köhidai, L.; Padrón-Nieves, M.; Ponte-Sucre, A. Effect of aliphatic, monocarboxylic, dicarboxylic, heterocyclic and sulphur-containing amino acids on Leishmania spp. chemotaxis. Parasitology 2015, 142, 1621–1630. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Green, S.J.; Crawford, R.M.; Hockmeyer, J.T.; Meltzer, M.S.; Nacy, C.A. Leishmania major amastigotes initiate the L-arginine-dependent killing mechanism in INF-gamma-stimulated macrophages by induction of tumor necrosis factor-alpha. J. Immunol. 1990, 90, 4290–4297. [Google Scholar]
- Acuña, S.M.; Aoki, J.I.; Laranjeira-Silva, M.F.; Zampieri, R.A.; Fernandes, J.C.R.; Muxel, S.M.; Floeter-Winter, L.M. Arginase expression modulates nitric oxide production in Leishmania (Leishmania) amazonensis. PLoS ONE 2017, 12, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Rath, M.; Müller, I.; Kropf, P.; Closs, E.I.; Munder, M. Metabolism via arginase or nitric oxide synthase: Two competing arginine pathways in macrophages. Front. Immunol. 2014, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.F.L.; Floeter-Winter, L.M. Arginase in Leishmania. In Proteins and Proteomics of Leishmania and Trypanosoma; Springer: Dordrecht, The Netherlands, 2014; Volume 74, pp. 103–117. [Google Scholar]
- Goldman-Pinkovich, A.; Balno, C.; Strasser, R.; Zeituni-Molad, M.; Bendelak, K.; Rentsch, D.; Ephros, M.; Wiese, M.; Jardim, A.; Myler, P.J.; et al. An arginine deprivation response pathway is induced in Leishmania during macrophage invasion. PLoS Pathog. 2016, 12, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Muxel, S.M.; Laranjeira-Silva, M.F.; Zampieri, R.A.; Floeter-Winter, L.M. Leishmania (Leishmania) amazonensis induces macrophage miR-294 and miR-721 expression and modulates infection by targeting NOS2 and L-arginine metabolism. Sci. Rep. 2017, 7, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Kropf, P.; Fuentes, J.M.; Fahnrich, E.; Arpa, L.; Herath, S.; Weber, V.; Soler, G.; Celada, A.; Modolell, M.; Muller, I. Arginase and polyamine synthesis are key factors in the regulation of experimental leishmaniasis in vivo. FASEB J. 2005, 19, 1000–1002. [Google Scholar] [CrossRef] [PubMed]
- Stuehr, D.J. Mammalian nitric oxide synthases. Biochim. Biophys. Acta Bioenerg. 1999, 1411, 217–230. [Google Scholar] [CrossRef]
- Alderton, W.K.; Cooper, C.E.; Knowles, R.G. Nitric oxide synthases: Structure, function and inhibition. Biochem. J. 2001, 357, 593–615. [Google Scholar] [CrossRef] [PubMed]
- Duszenko, M.; Ginger, M.L.; Brennand, A.; Gualdrón-López, M.; Colombo, M.I.; Coombs, G.H.; Coppens, I.; Jayabalasingham, B.; Langsley, G.; De Castro, S.L.; et al. Autophagy in protists. Autophagy 2011, 7, 127–158. [Google Scholar] [CrossRef] [PubMed]
- Brennand, A.; Gualdrón-López, M.; Coppens, I.; Rigden, D.J.; Ginger, M.L.; Michels, P.A.M. Autophagy in parasitic protists: Unique features and drug targets. Mol. Biochem. Parasitol. 2011, 177, 83–99. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Lee, M. Autophagy—A key player in cellular and body metabolism. Nat. Rev. Endocrinol. 2014, 10, 322–337. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.S.; Buẗikofer, P. Autophagy in Trypanosoma brucei: Amino acid requirement and regulation during different growth phases. PLoS ONE 2014, 9, 1–10. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, F.-J.; Xu, Z.-S.; Soo, A.D.S.; Lun, Z.-R.; He, C.Y. ATP-driven and AMPK-independent autophagy in an early branching eukaryotic parasite. Autophagy 2017, 13, 715–729. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; He, C.Y. Autophagy in protozoan parasites: Trypanosoma brucei as a model. Future Microbiol. 2017, 12, 1337–1340. [Google Scholar] [CrossRef] [PubMed]
- Albina, J.E.; Cui, S.; Mateo, R.B.; Reichner, J.S. Nitric oxide-mediated apoptosis in murine peritoneal macrophages. J. Immunol. 1993, 150, 5080–5085. [Google Scholar] [PubMed]
- Piacenza, L.; Peluffo, G.; Radi, R. L-arginine-dependent suppression of apoptosis in Trypanosoma cruzi: Contribution of the nitric oxide and polyamine pathways. Proc. Natl. Acad. Sci. USA 2001, 98, 7301–7306. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.; Das, S.; Roy, S.; Ghosh, A.K.; Sardar, A.H.; Verma, S.; Saini, S.; Singh, R.; Abhishek, K.; Kumar, A.; et al. Deprivation of L-arginine induces oxidative stress mediated apoptosis in Leishmania donovani promastigotes: Contribution of the polyamine pathway. PLoS Negl. Trop. Dis. 2016, 10, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Darling, T.N.; Blum, J.J. Changes in the shape of Leishmania major promastigotes in response to hexoses, proline, and hypo-osmotic stress. J. Protozool. 1990, 37, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Kollien, A.H.; Schaub, G.A. The development of Trypanosoma cruzi in triatominae. Parasitol. Today 2000, 16, 381–387. [Google Scholar] [CrossRef]
- Kollien, A.H.; Grospietsch, T.; Kleffmann, T.; Zerbst-Boroffka, I.; Schaub, G.A. Ionic composition of the rectal contents and excreta of the reduviid bug Triatoma infestans. J. Insect Physiol. 2001, 47, 739–747. [Google Scholar] [CrossRef]
- Louradour, I.; Monteiro, C.C.; Inbar, E.; Ghosh, K.; Merkhofer, R.; Lawyer, P.; Paun, A.; Smelkinson, M.; Secundino, N.; Lewis, M.; et al. The midgut microbiota plays an essential role in sand fly vector competence for Leishmania major. Cell. Microbiol. 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Lang, F. Mechanisms and significance of cell volume regulation. J. Am. Coll. Nutr. 2007, 26, 613S–623S. [Google Scholar] [CrossRef] [PubMed]
- Brocker, C.; Thompson, D.C.; Vasiliou, V. The role of hyperosmotic stress in inflammation and disease. Biomol. Concepts 2012, 3, 345–364. [Google Scholar] [CrossRef] [PubMed]
- Rohloff, P.; Docampo, R. A contractile vacuole complex is involved in osmoregulation in Trypanosoma cruzi. Exp. Parasitol. 2008, 118, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Docampo, R.; Ulrich, P.; Moreno, S.N.J. Evolution of acidocalcisomes and their role in polyphosphate storage and osmoregulation in eukaryotic microbes. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Docampo, R.; de Souza, W.; Miranda, K.; Rohloff, P.; Moreno, S.N.J. Acidocalcisomes—Conserved from bacteria to man. Nat. Rev. Microbiol. 2005, 3, 251–261. [Google Scholar] [CrossRef] [PubMed]
- Allen, R.D.; Naitoh, Y. Osmoregulation and contractile vacuoles of protozoa. Int. Rev. Cytol. 2002, 215, 351–394. [Google Scholar] [PubMed]
- Docampo, R.; Jimenez, V.; Lander, N.; Li, Z.H.; Niyogi, S. New insights into roles of acidocalcisomes and contractile vacuole complex in osmoregulation in protists. Int. Rev. Cell Mol. Biol. 2013, 305, 69–113. [Google Scholar] [PubMed]
- Ruiz, F.A.; Rodrigues, C.O.; Docampo, R. Rapid changes in polyphosphate content within acidocalcisomes in response to cell growth, differentiation, and environmental stress in Trypanosoma cruzi. J. Biol. Chem. 2001, 276, 26114–26121. [Google Scholar] [CrossRef] [PubMed]
- LeFurgey, A.; Ingram, P.; Blum, J.J. Compartmental responses to acute osmotic stress in Leishmania major result in rapid loss of Na+ and Cl−. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2001, 128, 385–394. [Google Scholar] [CrossRef]
- Lemercier, G.; Espiau, B.; Ruiz, F.A.; Vieira, M.; Luo, S.; Baltz, T.; Docampo, R.; Bakalara, N. A pyrophosphatase regulating polyphosphate metabolism in acidocalcisomes is essential for Trypanosoma brucei virulence in mice. J. Biol. Chem. 2004, 279, 3420–3425. [Google Scholar] [CrossRef] [PubMed]
- Vieira, L.L.; Lafuente, E.; Gamarro, F.; Cabantchik, Z. An amino acid channel activated by hypotonically induced swelling of Leishmania major promastigotes. Biochem. J. 1996, 319, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Rohloff, P.; Rodrigues, C.O.; Docampo, R. Regulatory volume decrease in Trypanosoma cruzi involves amino acid efflux and changes in intracellular calcium. Mol. Biochem. Parasitol. 2003, 126, 219–230. [Google Scholar] [CrossRef]
- LeFurgey, A.; Gannon, M.; Blum, J.; Ingram, P. Leishmania donovani amastigotes mobilize organic and inorganic osmolytes during regulatory volume decrease. J. Eukaryot. Microbiol. 2005, 52, 277–289. [Google Scholar] [CrossRef] [PubMed]
- Rohloff, P.; Docampo, R. Ammonium production during hypo-osmotic stress leads to alkalinization of acidocalcisomes and cytosolic acidification in Trypanosoma cruzi. Mol. Biochem. Parasitol. 2006, 150, 249–255. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.-H.; Alvarez, V.E.; De Gaudenzi, J.G.; Sant’Anna, C.; Frasch, A.C.C.; Cazzulo, J.J.; Docampo, R. Hyperosmotic stress induces aquaporin-dependent cell shrinkage, polyphosphate synthesis, amino acid accumulation, and global gene expression changes in Trypanosoma cruzi. J. Biol. Chem. 2011, 286, 43959–43971. [Google Scholar] [CrossRef] [PubMed]
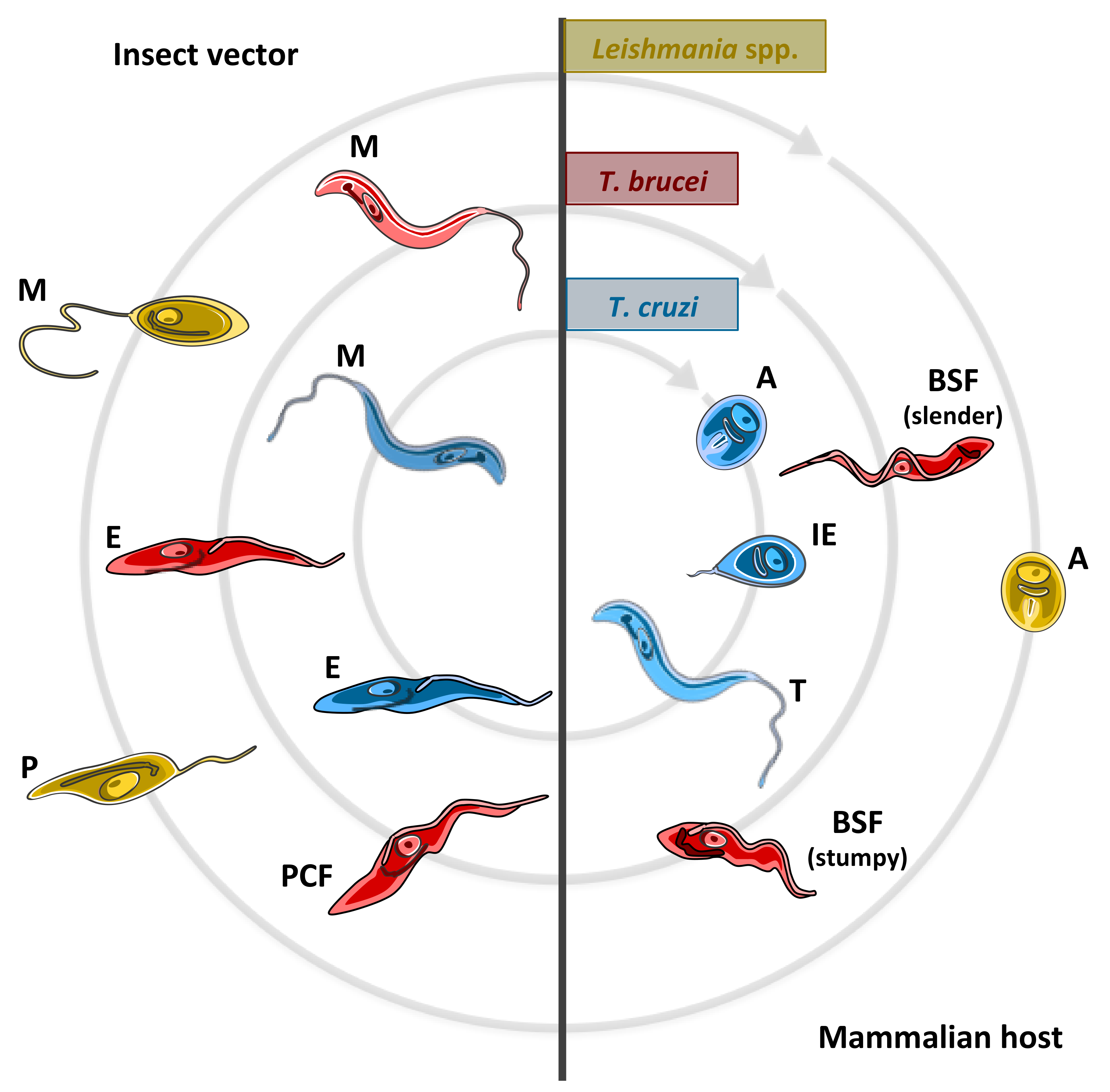
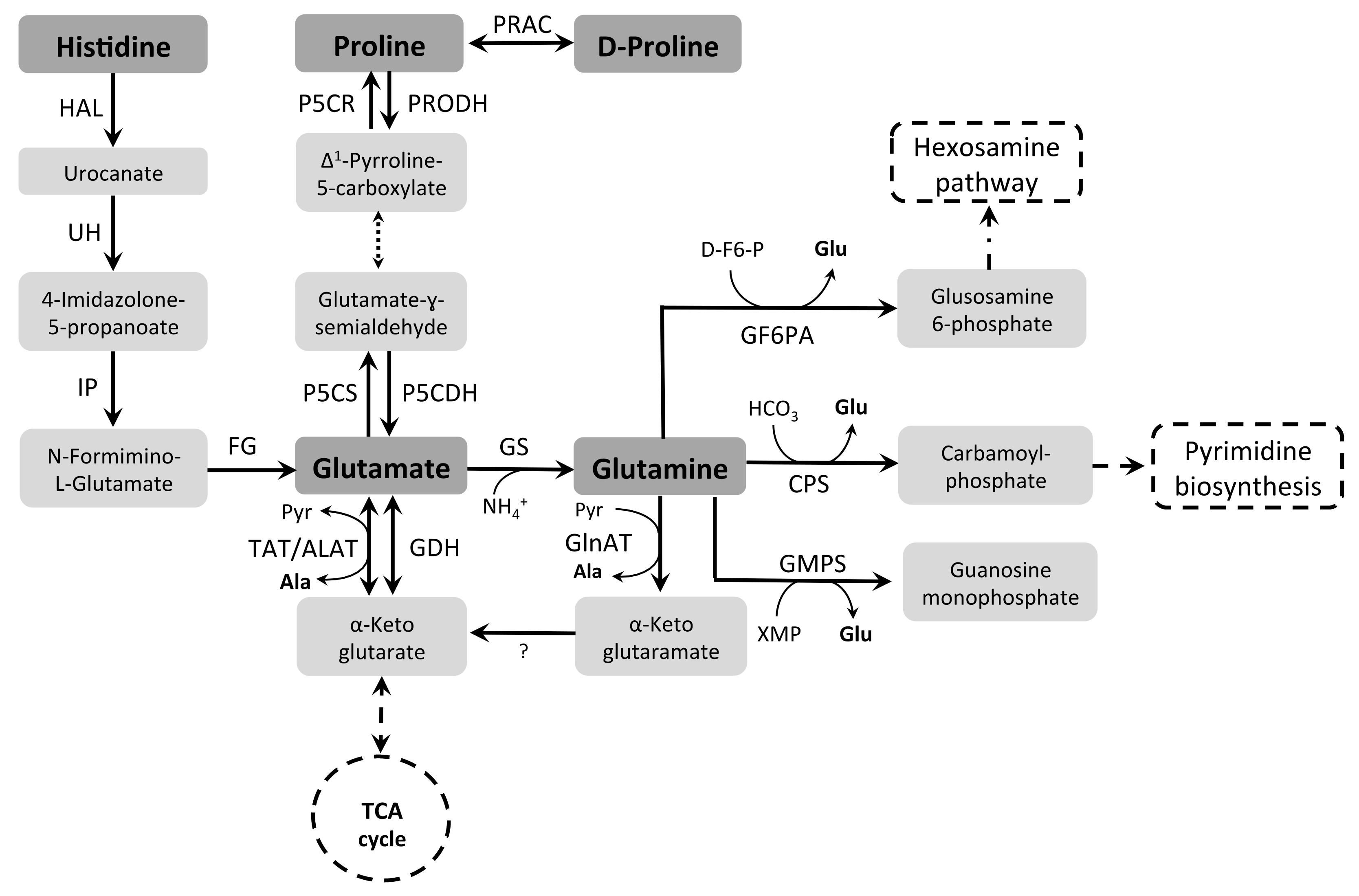
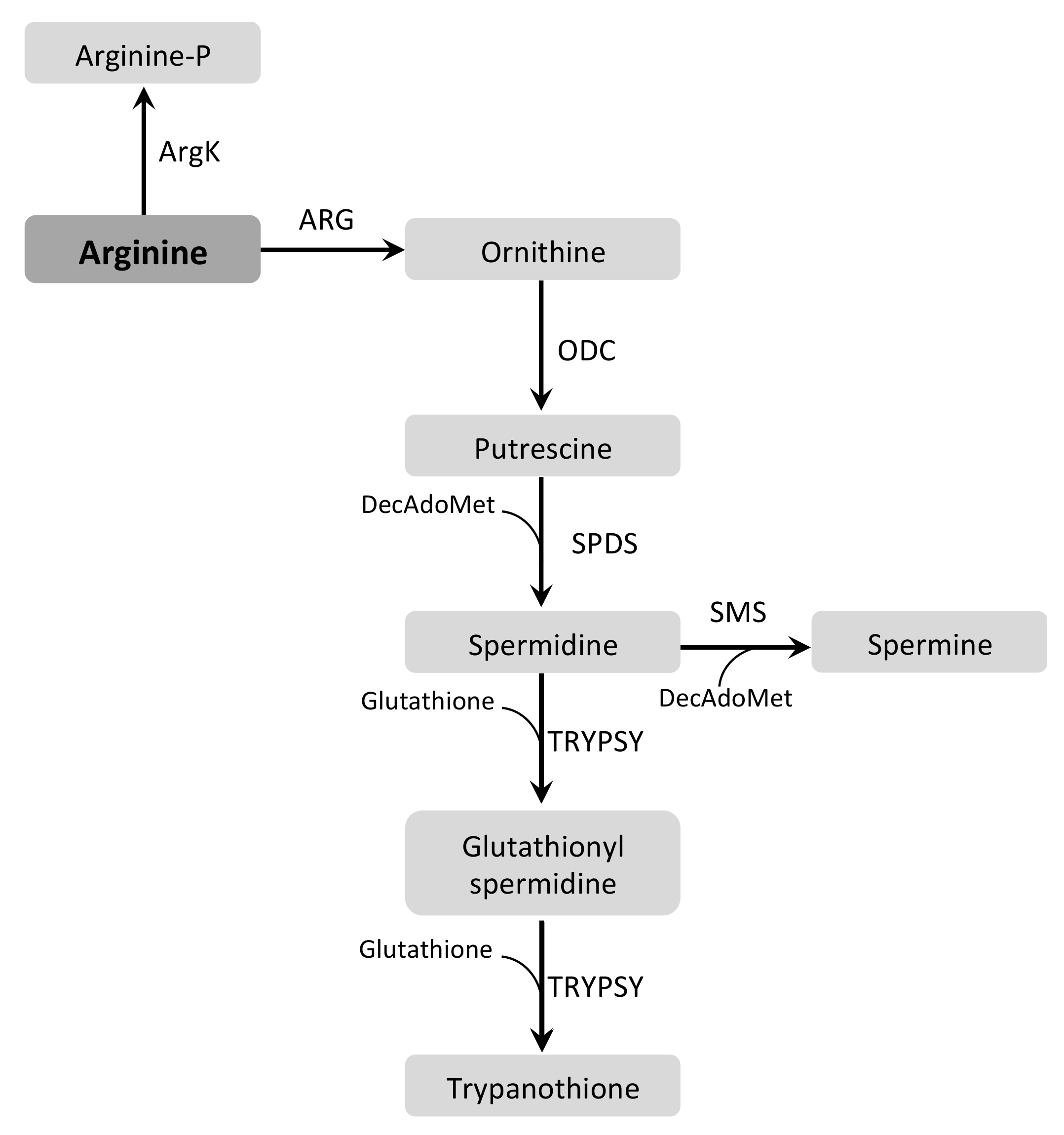
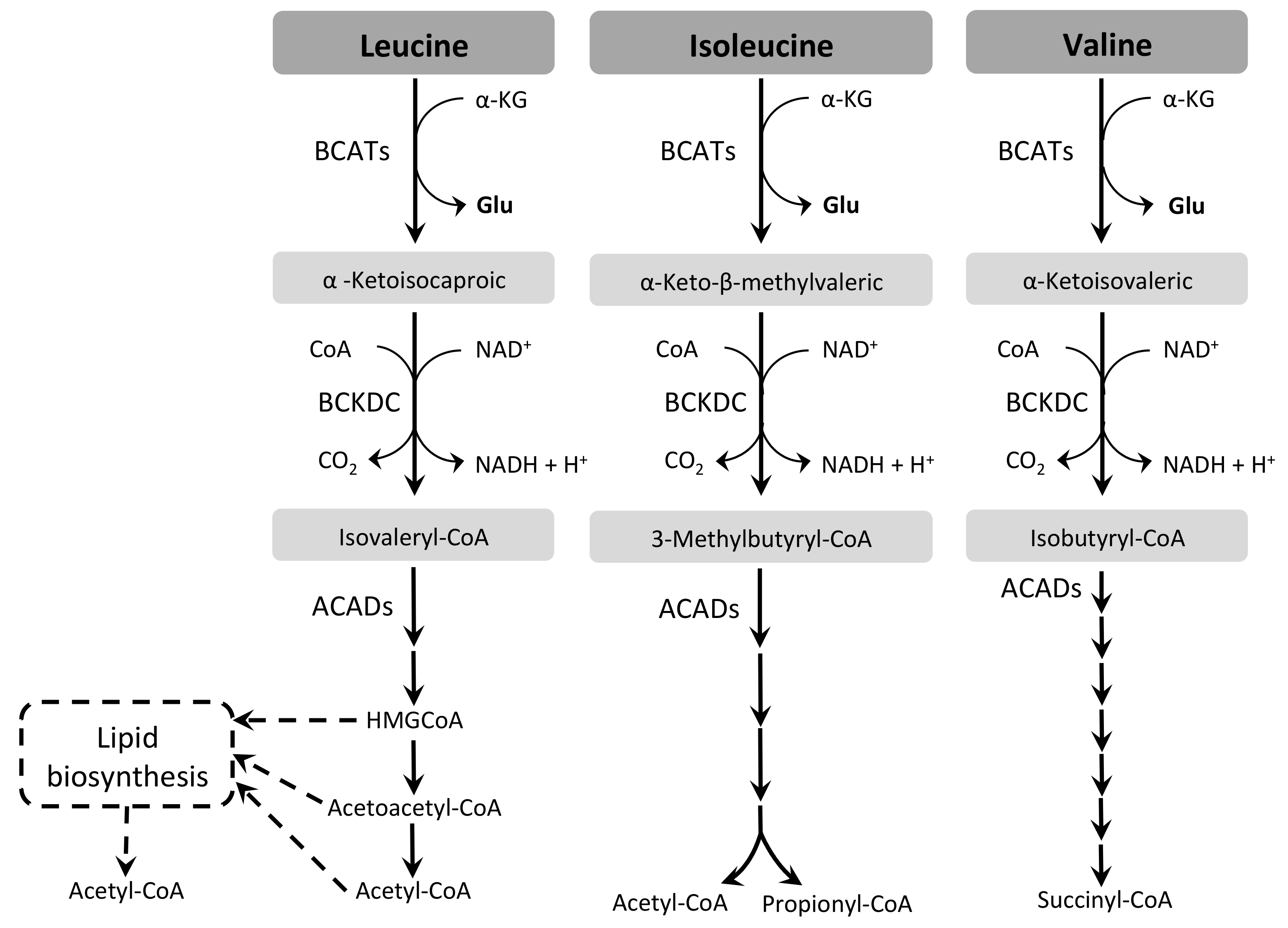
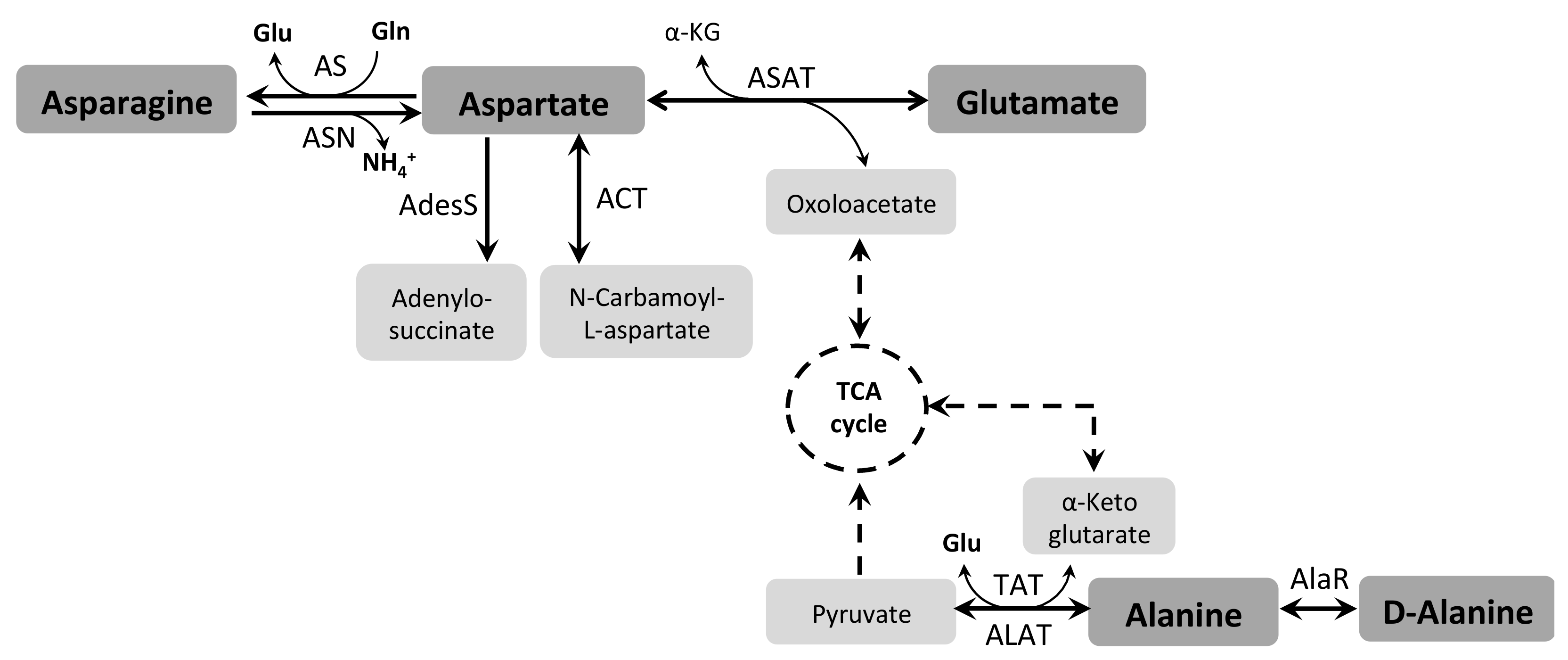
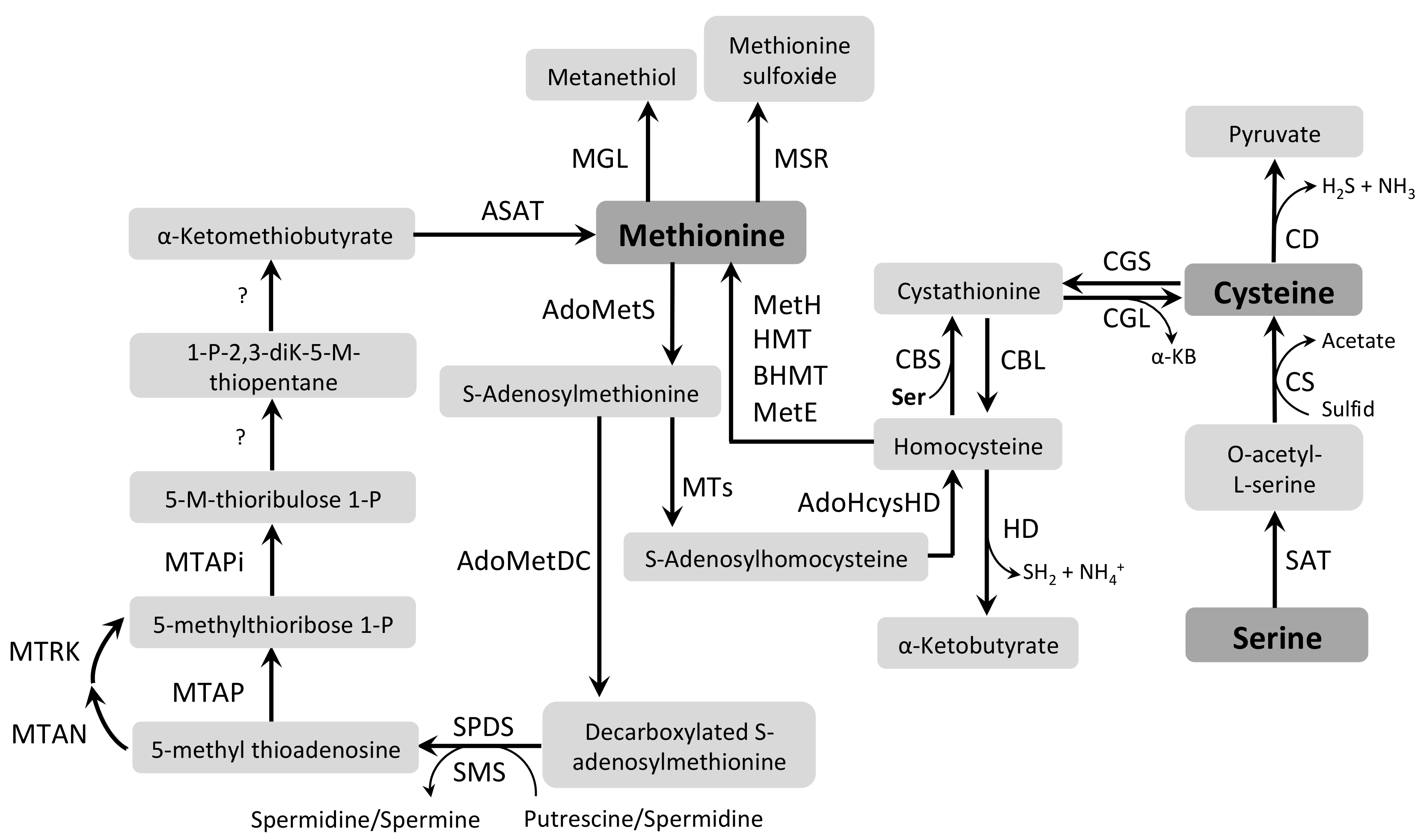
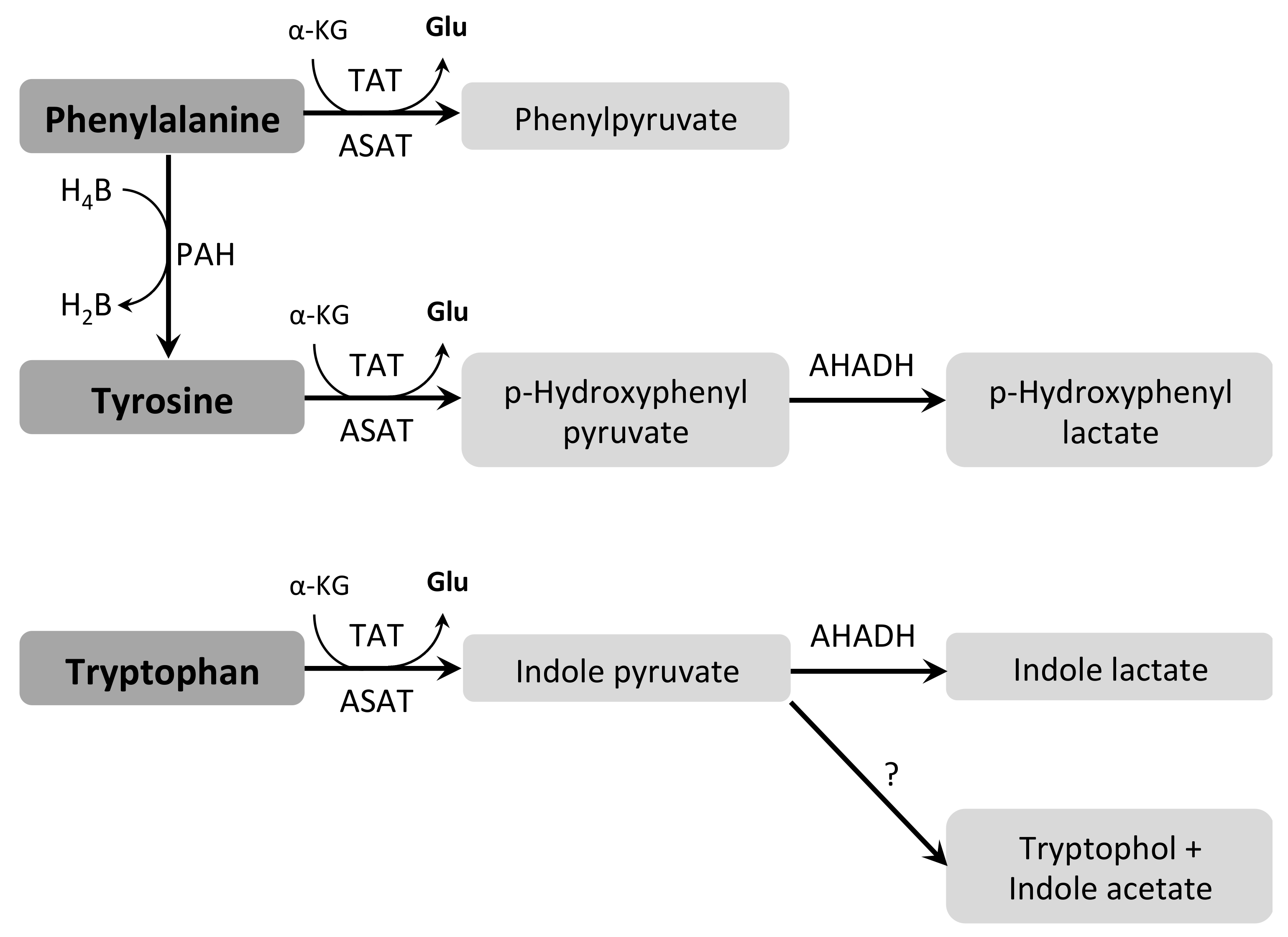
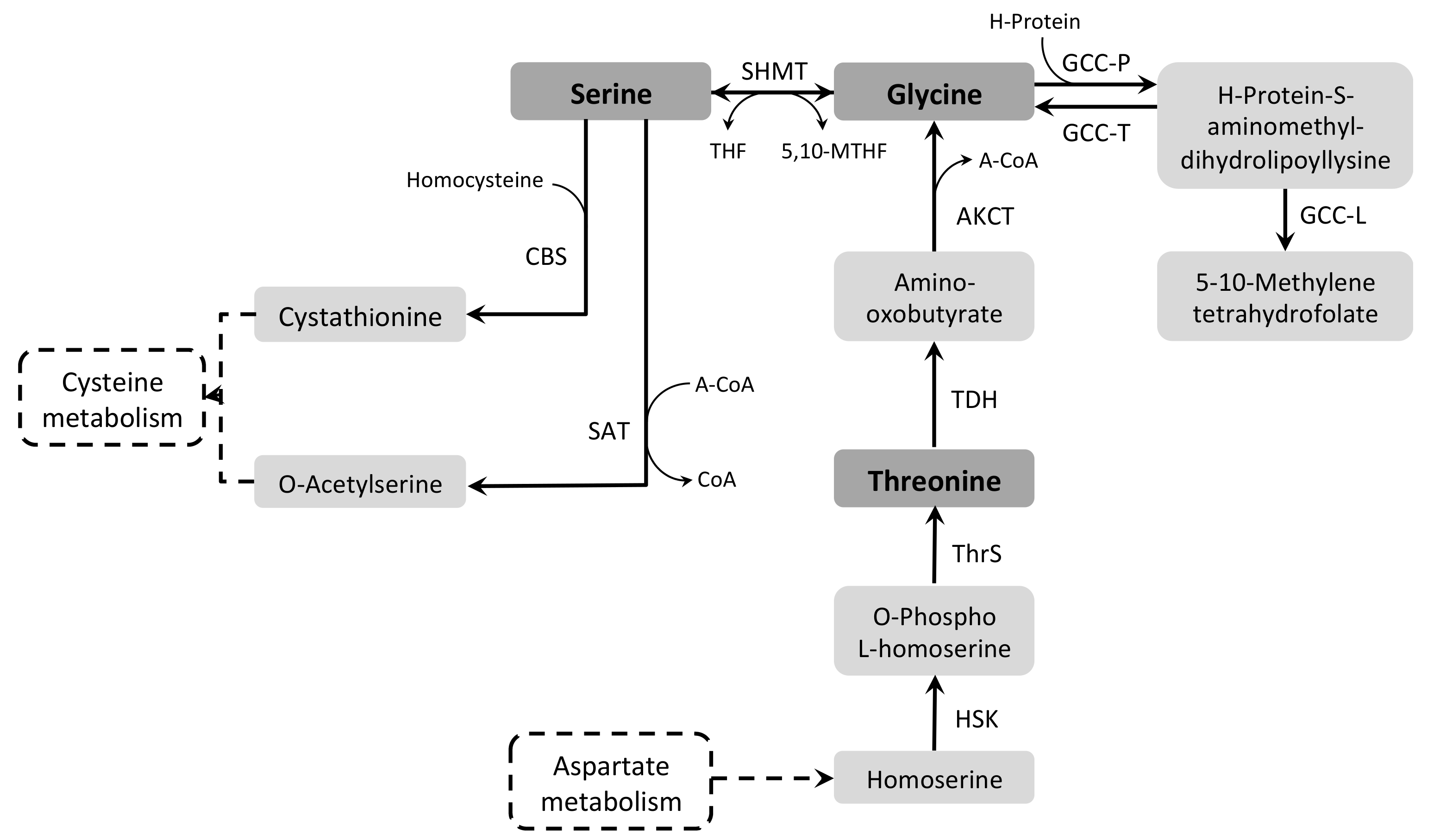
| Amino Acid | T. brucei | T. cruzi | Leishmania spp. | Gene Name |
|---|---|---|---|---|
| Proline a | Two systems [38,41,42] | Two systems [54] | Three systems [72] | TbAAT6 [40] |
| TcAAP24 [55] | ||||
| LmAAP24 [76] | ||||
| Glutamate | One system [60] | One system [76] | ||
| Glutamine | One system (Damasceno et al., submitted) | |||
| Isoleucine, Leucine and Valine b | One system [64] | |||
| Aspartate | One system [59] | |||
| Arginine c | Two systems [48,49] | One system [73] | TbAAT5-3 [45] | |
| TcAAAP411/TcAAP3 and TcCAT1.1 [51,53] | ||||
| LdAAP3 and LaAAP3 [75,79,80,81,82] | ||||
| Ornithine d | TbAAT10-1 and TbAAT2-4 [46] | |||
| TcCAT1.1 [55] | ||||
| Histidine e | One system [61] | TbAAT2-4 [46] | ||
| Methionine | Two systems [43,44] | One system [65,66] | ||
| Lysine f | One system [52] | One system [52] | TbAAT16-1 [45] | |
| TcAAP7 [52] | ||||
| LmAAP7 [52] | ||||
| Threonine | One system [35] | |||
| Cysteine | One system [36] | One system [63] | ||
| Serine | One system [77] |
| Amino Acid | Enzymes | Genes * | Characterization | ||
|---|---|---|---|---|---|
| T. brucei | T. cruzi | L. major | |||
| Ala | ALAT | Tb927.1.3950 | TcCLB.506529.420● | LmjF12.0630 | [129,269] |
| AlaR | Tb927.5.1280● | TcCLB.509767.90● | LmjF.23.1480 | [262] | |
| TAT | (see aromatic AA) | ||||
| Arg | ARG | - | - | LmjF.35.1480 | [141] |
| ArgK | Tb927.9.6210 | TcCLB.482369.29 | - | [51,148,150] | |
| Aromatic AA | ASAT/BSAT | Tb927.10.3660 | TcCLB.503841.70 | LmjF.35.0820 | [171,174,228] |
| TAT | - | TcCLB.508535.50 | LmjF.36.2360 | [129] | |
| Asn | ASN | - | TcCLB.504147.110● | LmjF.36.4430● | |
| Asp | AS | Tb927.7.1110 | TcCLB.503899.90● | LmjF.26.0830● | [176] |
| ASAT | (see aromatic AA) | ||||
| mASAT | Tb927.11.5090 | TcCLB.510945.70 | LmjF.24.0370● | [174] | |
| BCAA | BCAT | Tb927.2.4590● | LmjF27.2030● | ||
| TAT/ASAT | (see aromatic AA) | ||||
| Cys | CGS | Tb11.v5.0869● | TcCLB.510741.10● | LmjF14.0460 | [215] |
| CD | - | - | LmjF.32.2640 | [216] | |
| Gln | GLNAT | Tb927.10.11970 | TcCLB.503747.10● | LmjF.33.1330● | [129] |
| GF6PA | Tb927.7.5560● | TcCLB.510303.200 | LmjF.06.0950 | [131]; Crispim et al., unpublished data | |
| CPS | Tb927.5.3800● | TcCLB.508373.10 | LmjF.16.0590 | [126,127] | |
| GMPS | Tb927.7.2100 | TcCLB.508085.10● | LmjF.22.0110● | [128] | |
| Glu | GDH | Tb927.9.5900● | TcCLB.505843.10 | LmjF.15.1010● | [119] |
| GS | Tb927.7.4970● | TcCLB.503405.10 | LmjF.06.0370 | [125,130] | |
| P5CS | - | TcCLB.509067.70● | LmjF.32.3140● | ||
| TAT/ASAT | (see aromatic AA) | ||||
| Gly | GCC-P | Tb927.7.1910● | TcCLB.510911.50● | LmjF26.0030 | [244] |
| His | HAL | - | TcCLB.506247.220 | - | [61] |
| Lys | - | - | - | - | |
| Met | AdoMetS | Tb927.6.4840 | TcCLB.506945.160● | LmjF.30.3500 | [189,191] |
| MSR | Tb427.08.550● | TcCLB.510855.10● | LmjF.07.1140● | ||
| Pro | PRODH | Tb927.7.210 | TcCLB.506411.30 | LmjF.26.1610 | [38,95]; Ferraz et al., unpublished data |
| PRAC | - | TcCLB.430737.10● | - | ||
| Phe | PAH | - | - | LmjF.28.1280 | [229] |
| Ser | SHMT-L | - | - | LmjF.28.2370 | [241] |
| SHMT-S | - | TcCLB.510407.90 | LmjF.14.1320 | [241,243] | |
| SAT | TcCLB.510879.80 | LmjF.34.2850 | [210,216] | ||
| CBS | Tb11.02.5400● | TcCLB.508241.140 | LmjF17.0250 | [210,212] | |
| Thr | TDH | Tb927.6.2790 | TcCLB.507923.10● | - | [169] |
| ● Putative genes | |||||
| Amino Acid | Function | T. brucei | T. cruzi | Leishmania spp. | References |
|---|---|---|---|---|---|
| Ala | Osmotic control | [336,337,338] | |||
| Arg | Polyamine biosynthesis | [134,136] | |||
| Establishment of infection | [306] | ||||
| Apoptosis protection | [320] | ||||
| Energy management | [51,148,149] | ||||
| Asn | Metacyclogenesis | [278] | |||
| Asp | Metacyclogenesis | [172] | |||
| Pyrimidine biosynthesis | [175] | ||||
| Mitochondrial ATP synthesis | [93] | ||||
| Cys | Trypanothione synthesis | [214] | |||
| Gln | Pyrimidine biosynthesis | [126,127] | |||
| Hexosamine biosynthesis | Crispim et al., unpublished; [131] | ||||
| Metacyclogenesis | Damasceno et al., submitted | ||||
| Glu | Osmotic control | [336,337,338] | |||
| Metacyclogenesis | [172] | ||||
| Mitochondrial ATP synthesis | [93,121] | ||||
| Resistance to oxidative stress | [277] | ||||
| Resistance to nutritional stress | [277] | ||||
| Gly | Osmotic control | [336,337,338] | |||
| His | Resistance to nutritional stress | [61] | |||
| Autophagy protection | [316] | ||||
| Leu | Sterol biosynthesis | [165,167] | |||
| Fatty acid biosynthesis | [165] | ||||
| Met | Polyamine biosynthesis | [173] | |||
| Resistance to oxidative stress | [197,204,205] | ||||
| Pro | Mitochondrial ATP synthesis | [2,38,39,92,95,96] | |||
| Carbon source | [2,30,38,103] | ||||
| Cell differentiation | [101,172] | ||||
| Cell invasion | [96,279,337] | ||||
| Osmotic control | [337] | ||||
| Resistance to oxidative stress | [276] | ||||
| Resistance to thermal stress | [276] | ||||
| Resistance to nutritional stress | [276] | ||||
| Ser | Osmotic control | [336,338] | |||
| Thr | Carbon source | [169,256] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marchese, L.; Nascimento, J.D.F.; Damasceno, F.S.; Bringaud, F.; Michels, P.A.M.; Silber, A.M. The Uptake and Metabolism of Amino Acids, and Their Unique Role in the Biology of Pathogenic Trypanosomatids. Pathogens 2018, 7, 36. https://doi.org/10.3390/pathogens7020036
Marchese L, Nascimento JDF, Damasceno FS, Bringaud F, Michels PAM, Silber AM. The Uptake and Metabolism of Amino Acids, and Their Unique Role in the Biology of Pathogenic Trypanosomatids. Pathogens. 2018; 7(2):36. https://doi.org/10.3390/pathogens7020036
Chicago/Turabian StyleMarchese, Letícia, Janaina De Freitas Nascimento, Flávia Silva Damasceno, Frédéric Bringaud, Paul A. M. Michels, and Ariel Mariano Silber. 2018. "The Uptake and Metabolism of Amino Acids, and Their Unique Role in the Biology of Pathogenic Trypanosomatids" Pathogens 7, no. 2: 36. https://doi.org/10.3390/pathogens7020036
APA StyleMarchese, L., Nascimento, J. D. F., Damasceno, F. S., Bringaud, F., Michels, P. A. M., & Silber, A. M. (2018). The Uptake and Metabolism of Amino Acids, and Their Unique Role in the Biology of Pathogenic Trypanosomatids. Pathogens, 7(2), 36. https://doi.org/10.3390/pathogens7020036





