Opportunistic Pathogens and Microbial Communities and Their Associations with Sediment Physical Parameters in Drinking Water Storage Tank Sediments
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. qPCR Analysis
2.3. Next Generation Sequencing
2.4. Data Analysis
3. Results
3.1. Opportunistic Pathogens
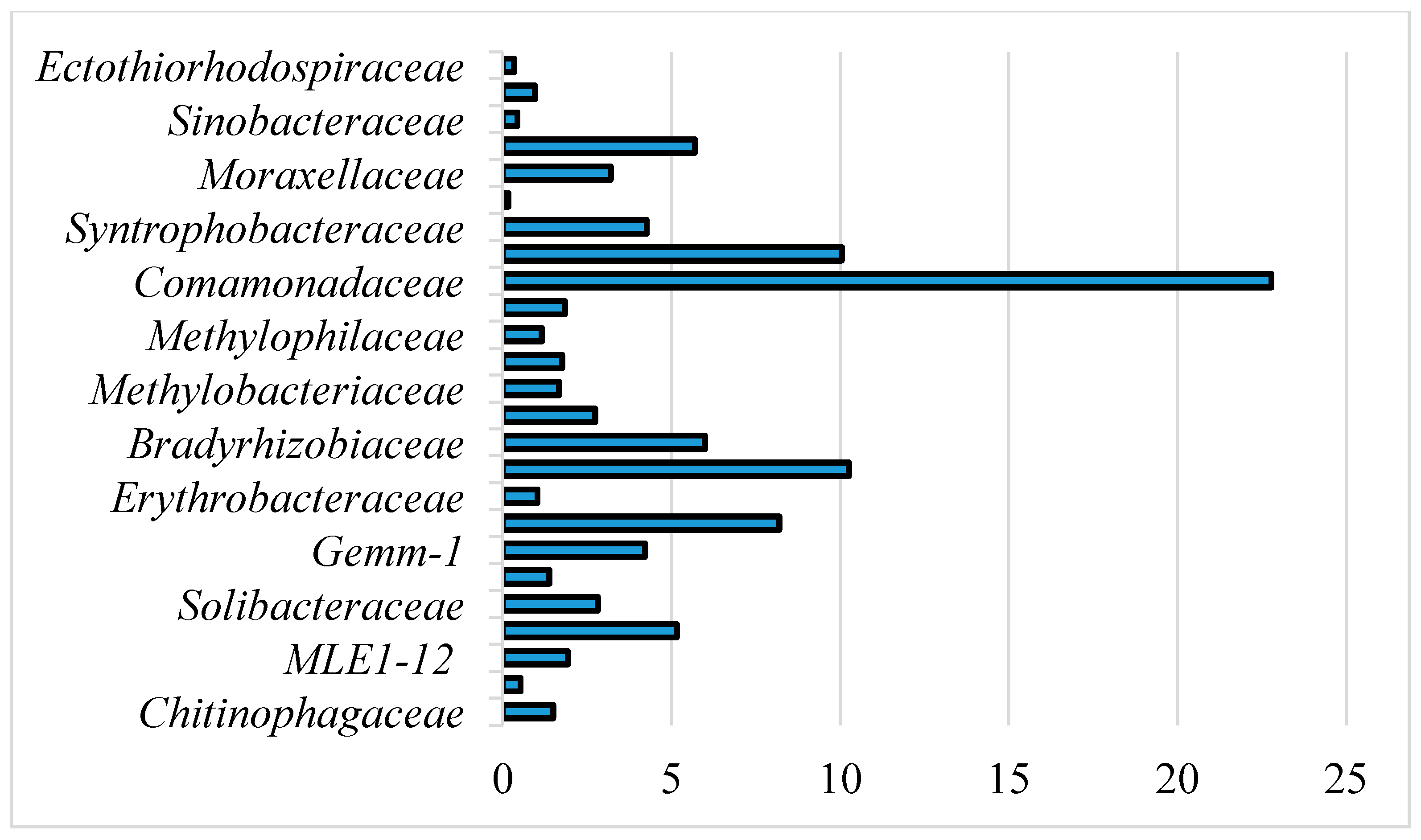
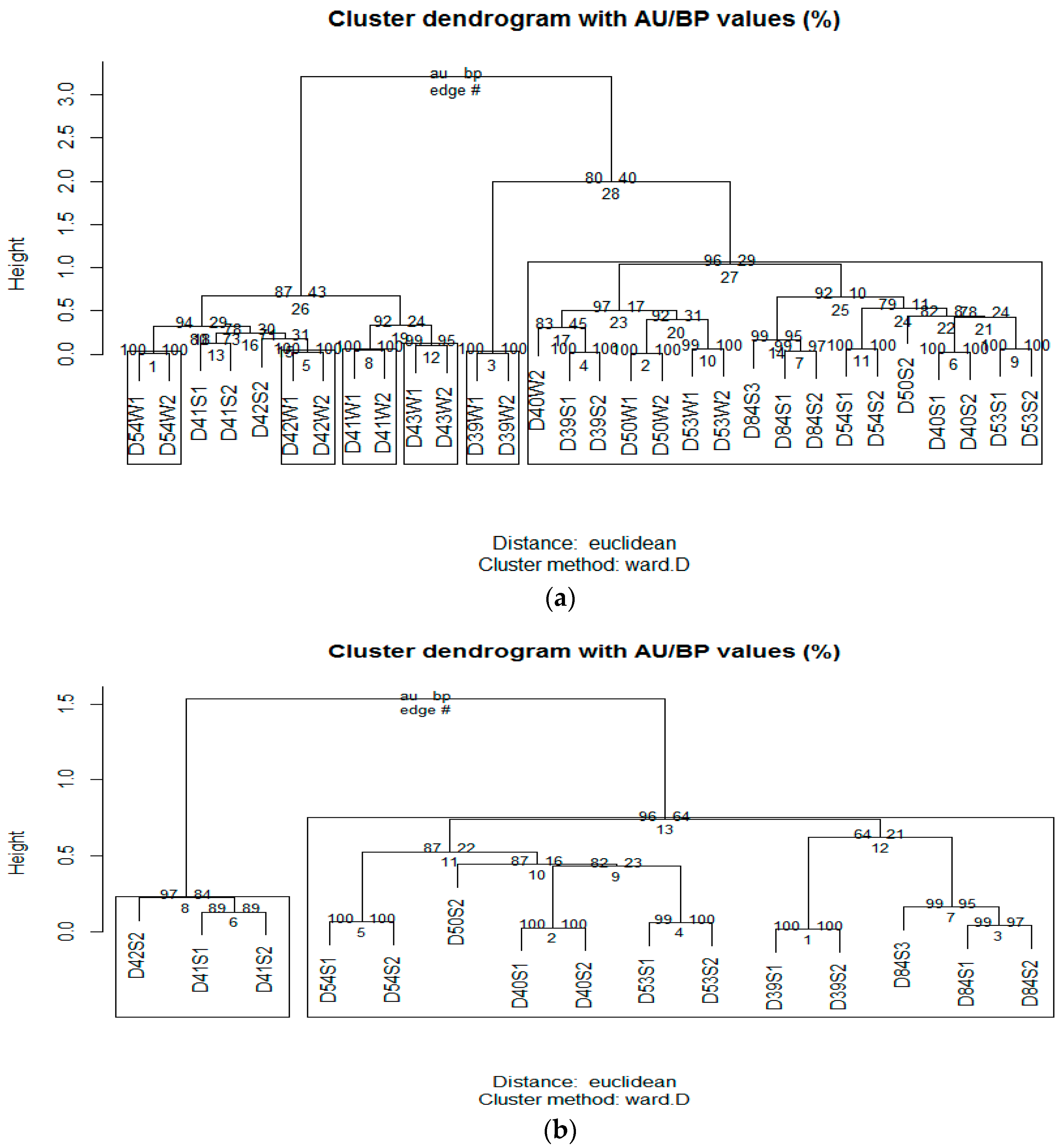
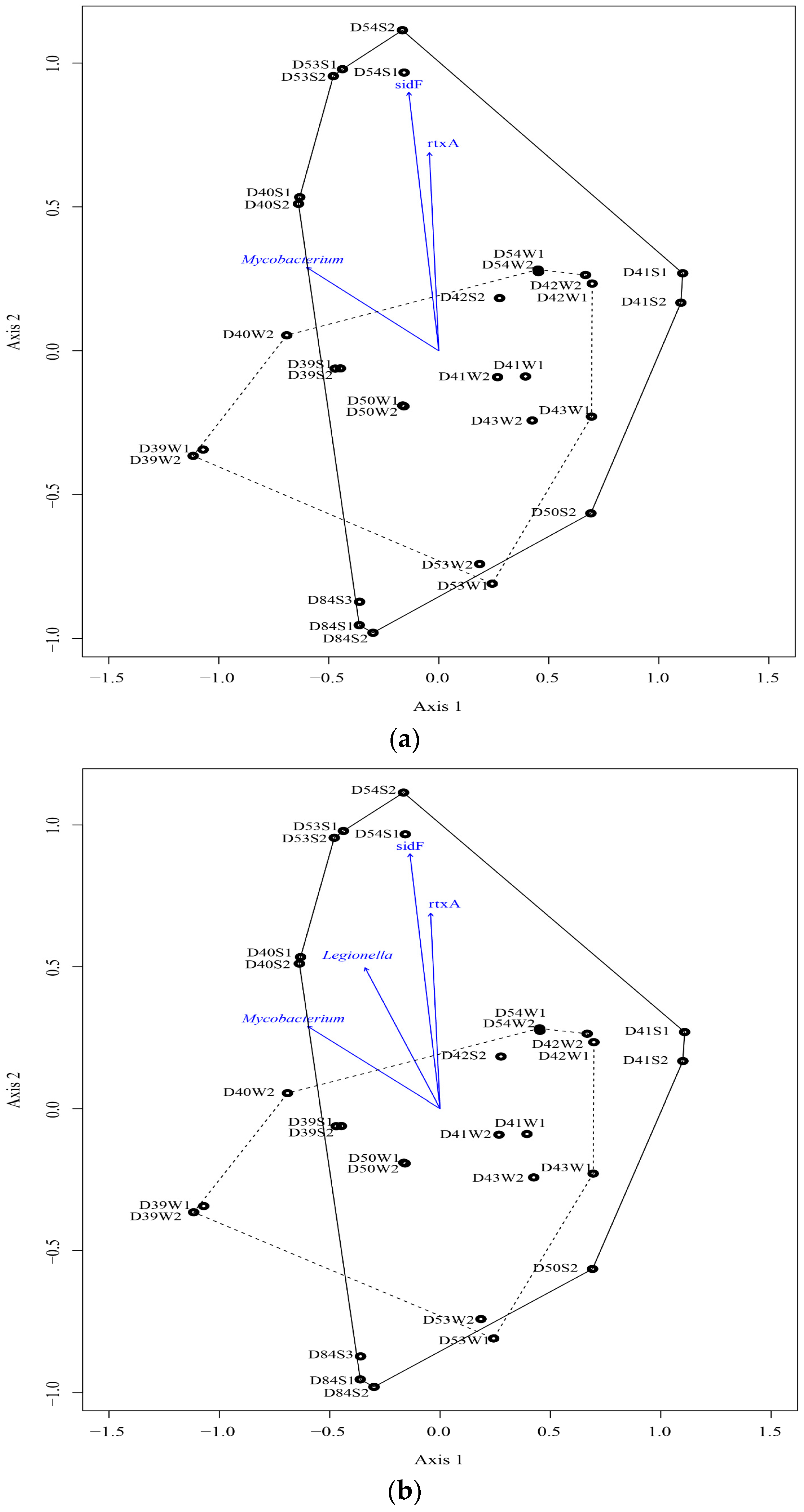
3.2. Microbial Community
4. Discussion
4.1. OPs in the Samples of Tank Water and Sediments
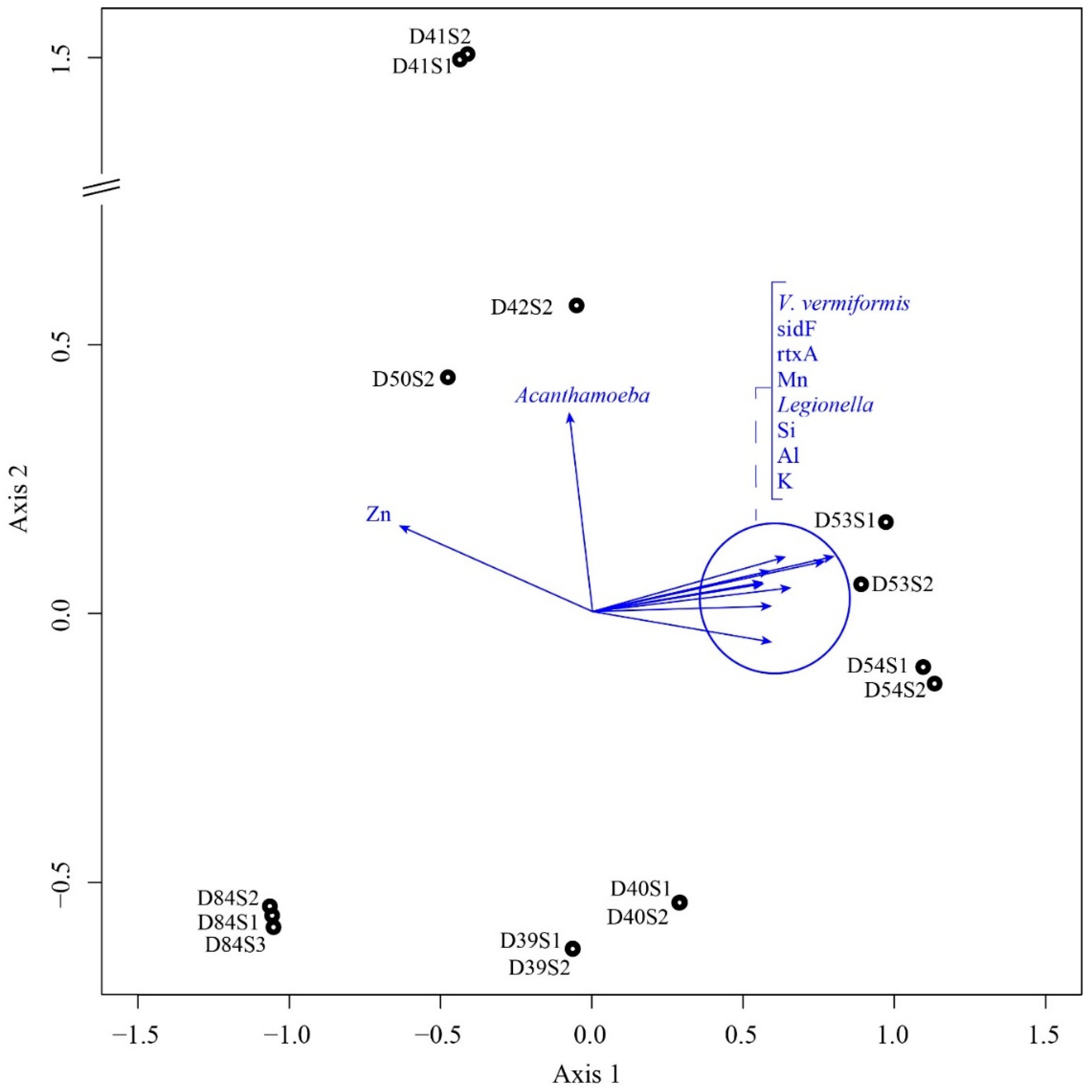
4.2. Microbial Communities and Their Relationship with Elements
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Revetta, R.P.; Pemberton, A.; Lamendella, R.; Iker, B.; Santo Domingo, J.W. Identification of bacterial populations in drinking water using 16s rrna-based sequence analyses. Water Res. 2010, 44, 1353–1360. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Struewing, I.; Vereen, E.; Kirby, A.E.; Levy, K.; Moe, C.; Ashbolt, N. Molecular detection of Legionella spp. And their associations with Mycobacterium spp., Pseudomonas aeruginosa and amoeba hosts in a drinking water distribution system. J. Appl. Microbiol. 2016, 120, 509–521. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Alvarez, V.; Revetta, R.P.; Santo Domingo, J.W. Metagenomic analyses of drinking water receiving different disinfection treatments. Appl. Environ. Microbiol. 2012, 78, 6095–6102. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Struewing, I.; Yelton, S.; Ashbolt, N. Molecular survey of occurrence and quantity of Legionella spp., Mycobacterium spp., Pseudomonas aeruginosa and amoeba hosts in municipal drinking water storage tank sediments. J. Appl. Microbiol. 2015, 119, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Van der Wielen, P.W.; Lut, M.C. Distribution of microbial activity and specific microorganisms across sediment size fractions and pipe wall biofilm in a drinking water distribution system. Water Sci. Technol. 2016, 16, 896–904. [Google Scholar] [CrossRef]
- Buse, H.Y.; Schoen, M.E.; Ashbolt, N.J. Legionellae in engineered systems and use of quantitative microbial risk assessment to predict exposure. Water Res. 2012, 46, 921–933. [Google Scholar] [CrossRef] [PubMed]
- Biyela, P.T.; Ryu, H.; Brown, A.; Alum, A.; Abbaszadegan, M.; Rittmann, B.E. Distribution systems as reservoirs of Naegleria fowleri and other amoebae. J. Am. Water Work Assoc. 2012, 104, 49–50. [Google Scholar] [CrossRef]
- Craun, G.F.; Brunkard, J.M.; Yoder, J.S.; Roberts, V.A.; Carpenter, J.; Wade, T.; Calderon, R.L.; Roberts, J.M.; Beach, M.J.; Roy, S.L. Causes of outbreaks associated with drinking water in the united states from 1971 to 2006. Clin. Microbiol. Rev. 2010, 23, 507–528. [Google Scholar] [CrossRef] [PubMed]
- Whiley, H.; Keegan, A.; Fallowfield, H.; Bentham, R. Detection of Legionella, L. pneumophila and mycobacterium avium complex (mac) along potable water distribution pipelines. Int. J. Environ. Res. Public Health 2014, 11, 7393–7405. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Edwards, M.; Falkinham, J.O.; Pruden, A. Molecular survey of the occurrence of Legionella spp., Mycobacterium spp., Pseudomonas aeruginosa, and amoeba hosts in two chloraminated drinking water distribution systems. Appl. Environ. Microbiol. 2012, 78, 6285–6294. [Google Scholar] [CrossRef] [PubMed]
- Hallam, N.; West, J.; Forster, C.; Simms, J. The potential for biofilm growth in water distribution systems. Water Res. 2001, 35, 4063–4071. [Google Scholar] [CrossRef]
- Teng, F.; Guan, Y.; Zhu, W. Effect of biofilm on cast iron pipe corrosion in drinking water distribution system: Corrosion scales characterization and microbial community structure investigation. Corros. Sci. 2008, 50, 2816–2823. [Google Scholar] [CrossRef]
- Lytle, D.A.; Gerke, T.L.; Maynard, J.B. Effect of bacterial sulfate reduction on iron-corrosion scales. Am. Water Works Assoc. J. 2005, 97, 109. [Google Scholar]
- Friedman, M.; Hill, A.; Reiber, S.; Valentine, R.; Larsen, G.; Young, A.; Korshin, G.; Peng, C. Assessment of Inorganics Accumulation in Drinking Water System Scales and Sediments; Water Research Foundation: Denver, CO, USA, 2010. [Google Scholar]
- Lytle, D.A.; Sorg, T.J.; Frietch, C. Accumulation of arsenic in drinking water distribution systems. Environ. Sci. Technol. 2004, 38, 5365–5372. [Google Scholar] [CrossRef] [PubMed]
- Lytle, D.A.; Sorg, T.; Wang, L.; Chen, A. The accumulation of radioactive contaminants in drinking water distribution systems. Water Res. 2014, 50, 396–407. [Google Scholar] [CrossRef] [PubMed]
- Stein, L.Y.; La Duc, M.T.; Grundl, T.J.; Nealson, K.H. Bacterial and archaeal populations associated with freshwater ferromanganous micronodules and sediments. Environ. Microbiol. 2001, 3, 10–18. [Google Scholar] [CrossRef] [PubMed]
- White, C.; Tancos, M.; Lytle, D.A. Microbial community profile of a lead service line removed from a drinking water distribution system. Appl. Environ. Microbiol. 2011, 77, 5557–5561. [Google Scholar] [CrossRef] [PubMed]
- Cornelis, P. Iron uptake and metabolism in pseudomonads. Appl. Microbiol. Biotechnol. 2010, 86, 1637–1645. [Google Scholar] [CrossRef] [PubMed]
- Robinson, D.A.; Griffith, R.W.; Shechtman, D.; Evans, R.B.; Conzemius, M.G. In vitro antibacterial properties of magnesium metal against Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus. Acta Biomater. 2010, 6, 1869–1877. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Suarez, A.; Dellunde, J.; Salvado, H.; Cervero-Arago, S.; Mendez, J.; Canals, O.; Blanco, S.; Arcas, A.; Araujo, R. Microbial and physicochemical parameters associated with Legionella contamination in hot water recirculation systems. Environ. Sci. Pollut. Res. 2013, 20, 5534–5544. [Google Scholar] [CrossRef] [PubMed]
- Iivanainen, E.; Martikainen, P.; Katila, M.L. Environmental factors affecting the occurrence of mycobacteria in brook sediments. J. Appl. Microbiol. 1999, 86, 673–681. [Google Scholar] [CrossRef] [PubMed]
- Pusch, M.; Fiebig, D.; Brettar, I.; Eisenmann, H.; Ellis, B.K.; Kaplan, L.A.; Lock, M.A.; Naegeli, M.W.; Traunspurger, W. The role of micro-organisms in the ecological connectivity of running waters. Freshw. Biol. 1998, 40, 453–495. [Google Scholar] [CrossRef]
- Li, Y.C.; Yu, S.; Strong, J.; Wang, H.L. Are the biogeochemical cycles of carbon, nitrogen, sulfur, and phosphorus driven by the “fe-iii-fe-ii redox wheel” in dynamic redox environments? J. Soils Sediments 2012, 12, 683–693. [Google Scholar] [CrossRef]
- Borch, T.; Kretzschmar, R.; Kappler, A.; Van Cappellen, P.; Ginder-Vogel, M.; Voegelin, A.; Campbell, K. Biogeochemical redox processes and their impact on contaminant dynamics. Environ. Sci. Technol. 2010, 44, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Lovley, D.R.; Coates, J.D. Bioremediation of metal contamination. Curr. Opin. Biotechnol. 1997, 8, 285–289. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Lauber, C.L.; Walters, W.A.; Berg-Lyons, D.; Huntley, J.; Fierer, N.; Owens, S.M.; Betley, J.; Fraser, L.; Bauer, M. Ultra-high-throughput microbial community analysis on the illumina hiseq and miseq platforms. ISME J. 2012, 6, 1621. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J. Introducing mothur: Open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microbiol. 2009, 75, 7537–7541. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Gevers, D.; Westcott, S.L. Reducing the effects of pcr amplification and sequencing artifacts on 16s rrna-based studies. PLoS ONE 2011, 6, e27310. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.D.; Sun, Y.J.; Zhao, X.; Wang, L.; Ding, A.Z.; Zhao, X.H. Sequencing insights into microbial communities in the water and sediments of fenghe river, china. Arch. Environ. Contam. Toxicol. 2016, 71, 122–132. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.B.; Wang, J.L. Denitrification performance and biofilm characteristics using biodegradable polymers pcl as carriers and carbon source. Chemosphere 2013, 91, 1310–1316. [Google Scholar] [CrossRef] [PubMed]
- Aguirre-von-Wobeser, E.; Eguiarte, L.E.; Souza, V.; Soberon-Chavez, G. Theoretical analysis of the cost of antagonistic activity for aquatic bacteria in oligotrophic environments. Front. Microbiol. 2015, 6, 8. [Google Scholar] [CrossRef] [PubMed]
- Kuever, J. The family syntrophobacteraceae. In The Prokaryotes: Deltaproteobacteria and Epsilonproteobacteria; Rosenberg, E., DeLong, E.F., Lory, S., Stackebrandt, E., Thompson, F., Eds.; Springer: Berlin/Heidelberg, Germany, 2014; pp. 289–299. [Google Scholar]
- Bahr, M.; Crump, B.C.; Klepac-Ceraj, V.; Teske, A.; Sogin, M.L.; Hobbie, J.E. Molecular characterization of sulfate-reducing bacteria in a New England salt marsh. Environ. Microbiol. 2005, 7, 1175–1185. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Zhen, Y.; Mi, T.Z.; Xu, B.C.; Wang, G.S.; Zhang, Y.; Yu, Z.G. Community composition and distribution of sulfate- and sulfite-reducing prokaryotes in sediments from the changjiang estuary and adjacent east china sea. Estuar. Coast. Shelf Sci. 2015, 165, 75–85. [Google Scholar] [CrossRef]
- Zhang, Y.; He, Q. Characterization of bacterial diversity in drinking water by pyrosequencing. Water Sci. Technol. 2013, 13, 358–367. [Google Scholar] [CrossRef]
- Berry, D.; Horn, M.; Xi, C.; Raskin, L. Mycobacterium avium infections of Acanthamoeba strains: Host strain variability, grazing-acquired infections, and altered dynamics of inactivation with monochloramine. Appl. Environ. Microbiol. 2010, 76, 6685–6688. [Google Scholar] [CrossRef] [PubMed]
- Buse, H.; Ashbolt, N. Differential growth of Legionella pneumophila strains within a range of amoebae at various temperatures associated with in-premise plumbing. Lett. Appl. Microbiol. 2011, 53, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Cirillo, S.L.G.; Bermudez, L.E.; El-Etr, S.H.; Duhamel, G.E.; Cirillo, J.D. Legionella pneumophila entry gene rtxa is involved in virulence. Infect. Immun. 2001, 69, 508–517. [Google Scholar] [CrossRef] [PubMed]
- Hsu, F.S.; Zhu, W.H.; Brennan, L.; Tao, L.L.; Luo, Z.Q.; Mao, Y.X. Structural basis for substrate recognition by a unique Legionella phosphoinositide phosphatase. Proc. Natl. Acad. Sci. USA 2012, 109, 13567–13572. [Google Scholar] [CrossRef] [PubMed]
- Tison, D.; Pope, D.; Cherry, W.; Fliermans, C. Growth of Legionella pneumophila in association with blue-green algae (cyanobacteria). Appl. Environ. Microbiol. 1980, 39, 456–459. [Google Scholar] [PubMed]
- Wadowsky, R.; Butler, L.; Cook, M.; Verma, S.; Paul, M.; Fields, B.; Keleti, G.; Sykora, J.; Yee, R. Growth-supporting activity for Legionella pneumophila in tap water cultures and implication of hartmannellid amoebae as growth factors. Appl. Environ. Microbiol. 1988, 54, 2677–2682. [Google Scholar] [PubMed]
- Greub, G.; Raoult, D. Morphology of Legionella pneumophila according to their location within hartmanella vermiformis. Res. Microbiol. 2003, 154, 619–621. [Google Scholar] [CrossRef] [PubMed]
- Ribas, F.; Perramon, J.; Terradillos, A.; Frias, J.; Lucena, F. The pseudomonas group as an indicator of potential regrowth in water distribution systems. J. Appl. Microbiol. 2000, 88, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Ling, F.Q.; Hwang, C.A.; LeChevallier, M.W.; Andersen, G.L.; Liu, W.T. Core-satellite populations and seasonality of water meter biofilms in a metropolitan drinking water distribution system. ISME J. 2016, 10, 582–595. [Google Scholar] [CrossRef] [PubMed]
- Luhrig, K.; Canback, B.; Paul, C.J.; Johansson, T.; Persson, K.M.; Radstrom, P. Bacterial community analysis of drinking water biofilms in southern sweden. Microbes Environ. 2015, 30, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Proctor, C.R.; Edwards, M.A.; Pruden, A. Microbial composition of purified waters and implications for regrowth control in municipal water systems. Environ. Sci. 2015, 1, 882–892. [Google Scholar] [CrossRef]
- Hwang, C.; Ling, F.Q.; Andersen, G.L.; LeChevallier, M.W.; Liu, W.T. Microbial community dynamics of an urban drinking water distribution system subjected to phases of chloramination and chlorination treatments. Appl. Environ. Microbiol. 2012, 78, 7856–7865. [Google Scholar] [CrossRef] [PubMed]
- Tokajian, S.; Hashwa, F. Microbiological quality and genotypic speciation of heterotrophic bacteria isolated from potable water stored in household tanks. Water Qual. Res. J. Can. 2004, 39, 64–73. [Google Scholar]
- Keinanen-Toivola, M.M.; Revetta, R.P.; Santo Domingo, J.W. Identification of active bacterial communities in a model drinking water biofilm system using 16s rrna-based clone libraries. FEMS Microbiol. Lett. 2006, 257, 182–188. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.G.; Ren, N.Q.; Wang, A.J. Contributions of fermentative acidogenic bacteria and sulfate-reducing bacteria to lactate degradation and sulfate reduction. Chemosphere 2008, 72, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Hard, B.C.; Walther, C.; Babel, W. Sorption of aluminum by sulfate-reducing bacteria isolated from uranium mine tailings. Geomicrobiol. J. 1999, 16, 267–275. [Google Scholar]
- Martins, M.; Taborda, R.; Silva, G.; Assuncao, A.; Matos, A.P.; Costa, M.C. Aluminum and sulphate removal by a highly al-resistant dissimilatory sulphate-reducing bacteria community. Biodegradation 2012, 23, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Cardenas, E.; Wu, W.M.; Leigh, M.B.; Carley, J.; Carroll, S.; Gentry, T.; Luo, J.; Watson, D.; Gu, B.; Ginder-Vogel, M.; et al. Microbial communities in contaminated sediments, associated with bioremediation of uranium to submicromolar levels. Appl. Environ. Microbiol. 2008, 74, 3718–3729. [Google Scholar] [CrossRef] [PubMed]
- Ghiorse, W. Biology of iron-and manganese-depositing bacteria. Annu. Rev. Microbiol. 1984, 38, 515–550. [Google Scholar] [CrossRef] [PubMed]
- Nealson, K.H.; Saffarini, D. Iron and manganese in anaerobic respiration: Environmental significance, physiology, and regulation. Annu. Rev. Microbiol. 1994, 48, 311–343. [Google Scholar] [CrossRef] [PubMed]
- Conley, L.; Ceraso, M.; Stephenson, T.; Wolford, R.; Wadowsky, R.; McNamara, A.; Yee, R. Effects of metals on Legionella pneumophila growth in drinking water plumbing systems. Appl. Environ. Microbiol. 1985, 50, 1149–1154. [Google Scholar]
- Reeves, M.; Pine, L.; Hutner, S.; George, J.; Harrell, W. Metal requirements of Legionella pneumophila. J. Clin. Microbiol. 1981, 13, 688–695. [Google Scholar] [PubMed]
- Rogers, J.; Dowsett, A.; Dennis, P.; Lee, J.; Keevil, C. Influence of plumbing materials on biofilm formation and growth of Legionella pneumophila in potable water systems. Appl. Environ. Microbiol. 1994, 60, 1842–1851. [Google Scholar] [PubMed]
| Matrix | Location ID | Acanthamoeba spp. | Vermamoeba vermiformis | Mycobacterium spp. | Pseudomonas aeruginosa | Legionella spp. | L. pneumophila (rtxA) | L. pneumophila (sidF) |
|---|---|---|---|---|---|---|---|---|
| Sediment (GN G−1: genome copy number per gram sediment) | D39S | 5 ± 1 | 0 | (2.73 ± 0.68) × 104 | 0 | 335 ± 31 | 0 | 0 |
| D40S | 1 ± 1 | 4 | (8.10 ± 1.16) × 103 | 0 | 923 ± 34 | 0 | 0 | |
| D41S | 28 ± 7 | 0 | (8.00 ± 1.20) × 101 | 0 | 0 | 0 | 0 | |
| D42S | 0 | 0 | (8.47 ± 10.04) × 103 | 0 | 0 | 0 | 0 | |
| D43S | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| D50S | 0 | 14 ± 22 | (2.00 ± 4.00) × 101 | 0 | 1 ± 3 | 0 | 0 | |
| D53S | 7 | 99 ± 60 | (2.39 ± 2.24) × 103 | 0 | 282 ± 113 | 25 ± 51 | 85 ± 69 | |
| D54S | 3 | 120 ± 43 | (2.57 ± 0.88) × 105 | 0 | (7.20 ± 3.31) × 104 | 300 ± 38 | 173 ± 87 | |
| Average | 6 | 29 | 3.79 × 104 | 0 | 9.19 × 103 | 41 | 32 | |
| Occurrence | 63% | 50% | 88% | 0 | 50% | 25% | 25% | |
| Water (GN L−1: genome copy number per liter water) | D39w | 0 | 0 | (2.33 ± 3.28) × 105 | 70 ± 91 | (1.24 ± 1.70) × 104 | 0 | 0 |
| D40w | 0 | 0 | (5.63 ± 7.74) × 104 | 0 | (6.93 ± 9.76) × 104 | 0 | 0 | |
| D41w | 50 | 0 | 6.91 × 103 | 0 | 0 | 0 | 0 | |
| D42w | 0 | 0 | 3.37 × 104 | 0 | 0 | 0 | 0 | |
| D43w | 0 | 0 | 2.52 × 103 | 0 | 1.50 × 103 | 0 | 0 | |
| D50w | 0 | 480 ± 670 | (5.07 ± 0.03)×103 | 2770 ± 14 | (1.84 ± 1.46) × 104 | 82 ± 117 | 36 ± 52 | |
| D53w | 0 | 0 | 0 | 83 ± 117 | 0 | 0 | 0 | |
| D54w | 0 | 98 ± 136 | (2.68 ± 2.84) × 103 | 371 ± 150 | 0 | 0 | 0 | |
| Average | 6 | 613 | 4.25 × 104 | 412 | 1.27 × 104 | 10 | 4 | |
| Occurrence | 13% | 25% | 88% | 50% | 50% | 13% | 13% |
| Elements | Pathogens | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acanthamoeba | Vermamoeba vermiformis | Mycobacterium | Legionella spp. | L. pneumophila (rtxA) | L. pneumophila (sidF) | |||||||
| R2 | P | R2 | P | R2 | P | R2 | P | R2 | P | R2 | P | |
| Al | 0.93 | <0.001 | 0.93 | <0.001 | 0.92 | <0.001 | 0.80 | 0.017 | ||||
| K | 0.75 | <0.031 | 0.77 | <0.025 | 0.75 | <0.031 | ||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qin, K.; Struewing, I.; Domingo, J.S.; Lytle, D.; Lu, J. Opportunistic Pathogens and Microbial Communities and Their Associations with Sediment Physical Parameters in Drinking Water Storage Tank Sediments. Pathogens 2017, 6, 54. https://doi.org/10.3390/pathogens6040054
Qin K, Struewing I, Domingo JS, Lytle D, Lu J. Opportunistic Pathogens and Microbial Communities and Their Associations with Sediment Physical Parameters in Drinking Water Storage Tank Sediments. Pathogens. 2017; 6(4):54. https://doi.org/10.3390/pathogens6040054
Chicago/Turabian StyleQin, Ke, Ian Struewing, Jorge Santo Domingo, Darren Lytle, and Jingrang Lu. 2017. "Opportunistic Pathogens and Microbial Communities and Their Associations with Sediment Physical Parameters in Drinking Water Storage Tank Sediments" Pathogens 6, no. 4: 54. https://doi.org/10.3390/pathogens6040054
APA StyleQin, K., Struewing, I., Domingo, J. S., Lytle, D., & Lu, J. (2017). Opportunistic Pathogens and Microbial Communities and Their Associations with Sediment Physical Parameters in Drinking Water Storage Tank Sediments. Pathogens, 6(4), 54. https://doi.org/10.3390/pathogens6040054




