Schistosoma mansoni and Haematobium: Radiological Diagnostic Clues and Pathophysiology
Abstract
1. Introduction
2. Methods
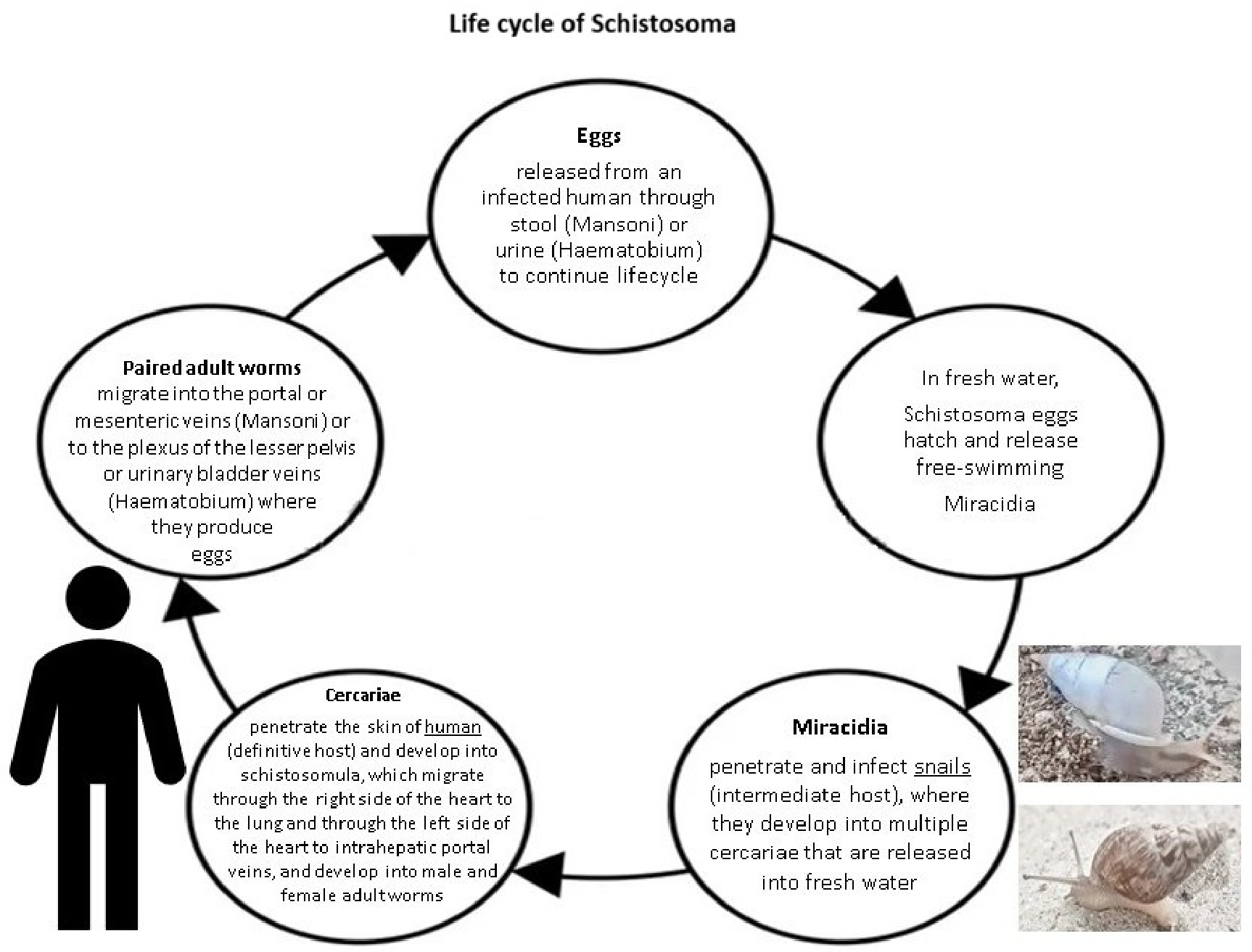
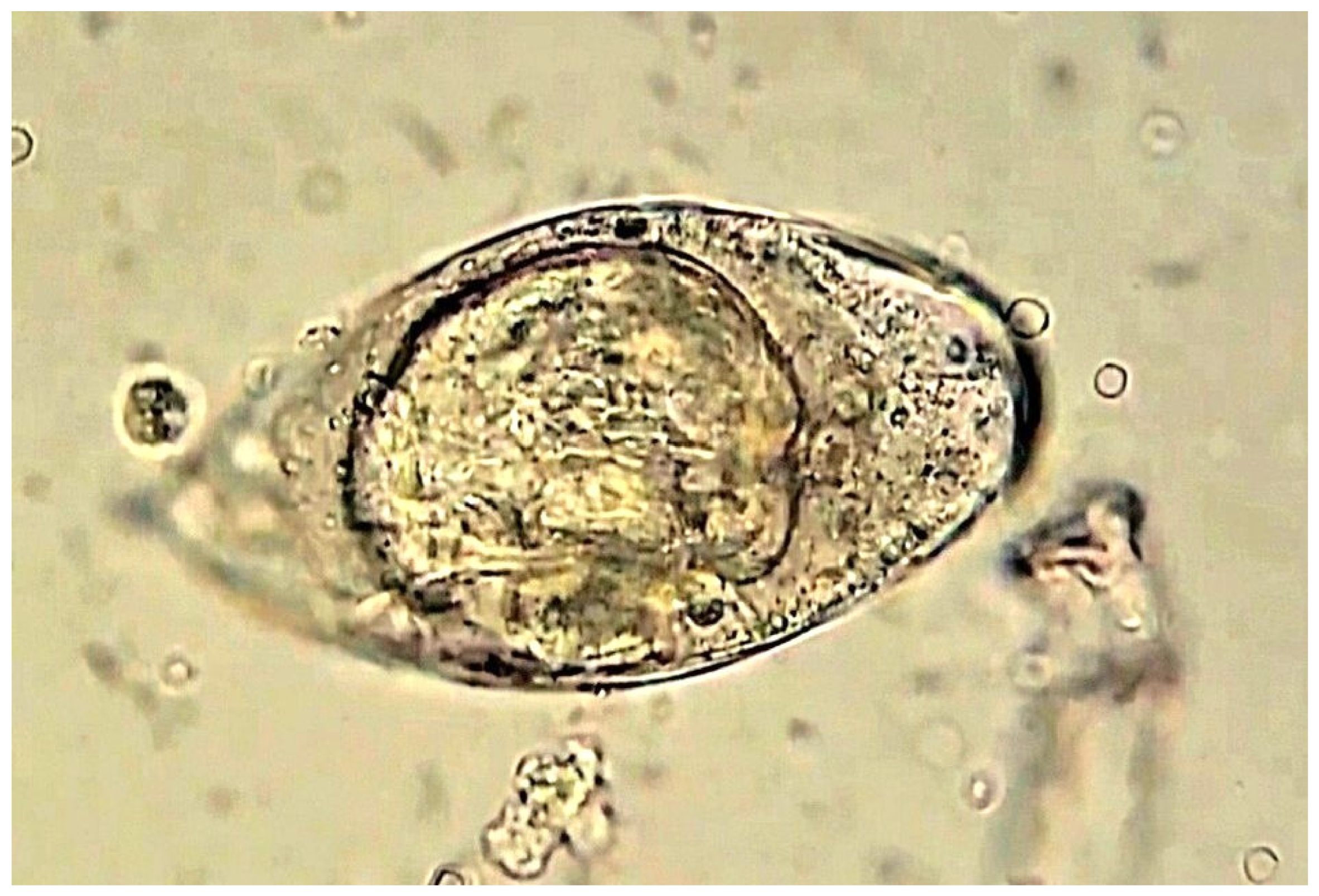
3. Life Cycle of Schistosoma
4. Pathophysiology of Schistosomiasis
5. Diagnosis of Schistosoma
6. Medical Imaging of Schistosoma
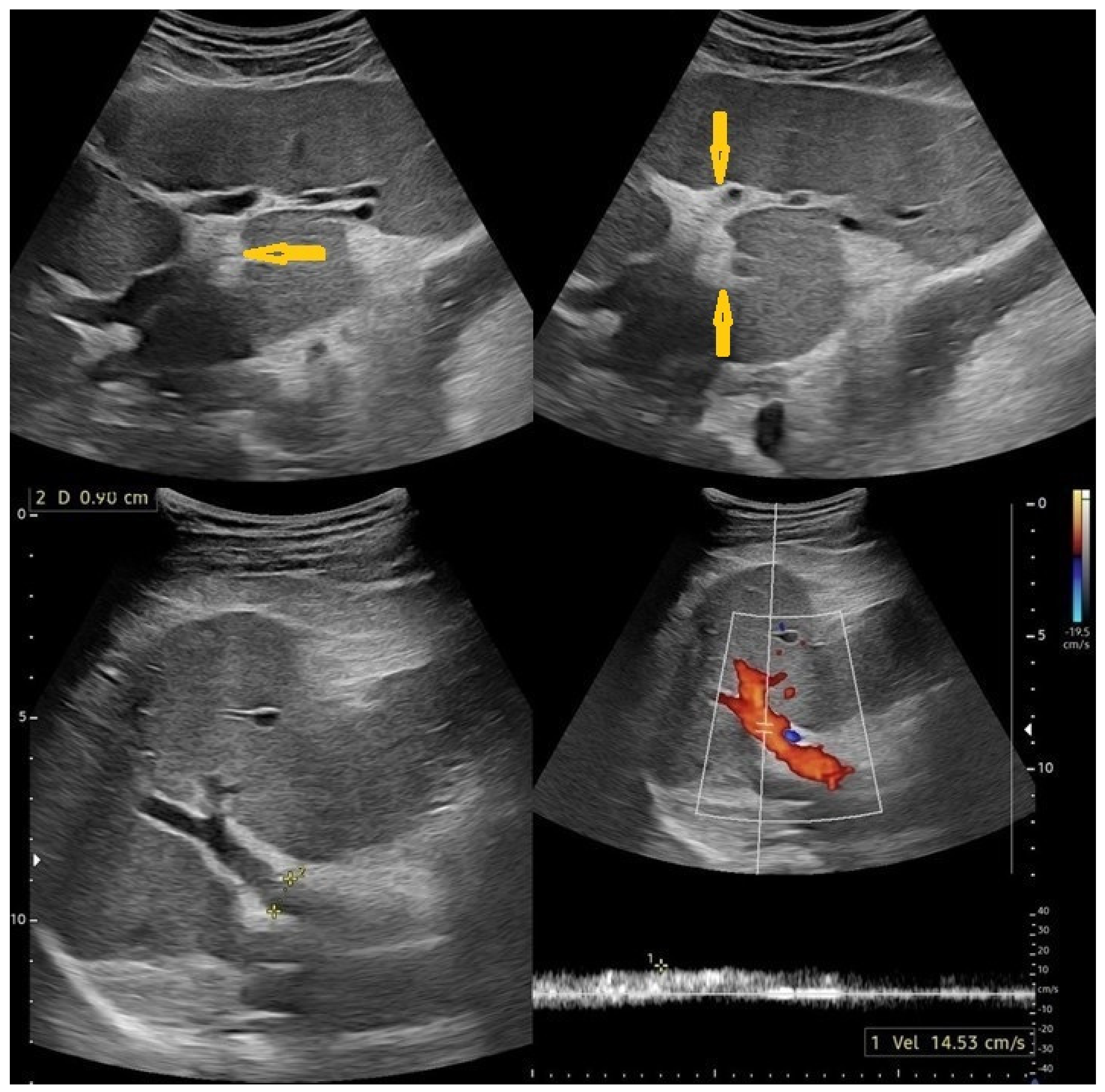
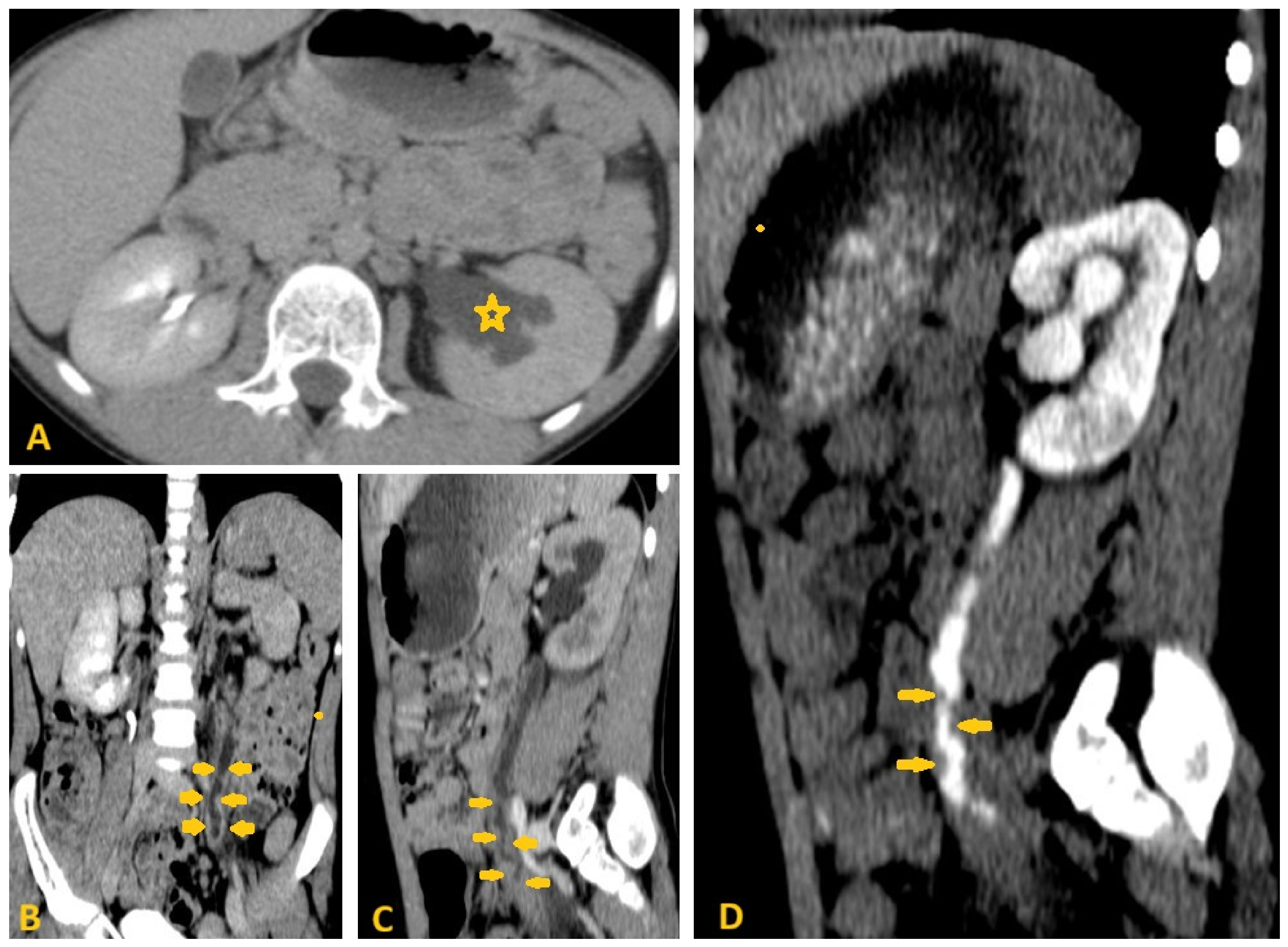
6.1. Medical Imaging Clues in Hepatointestinal Schistosomiasis
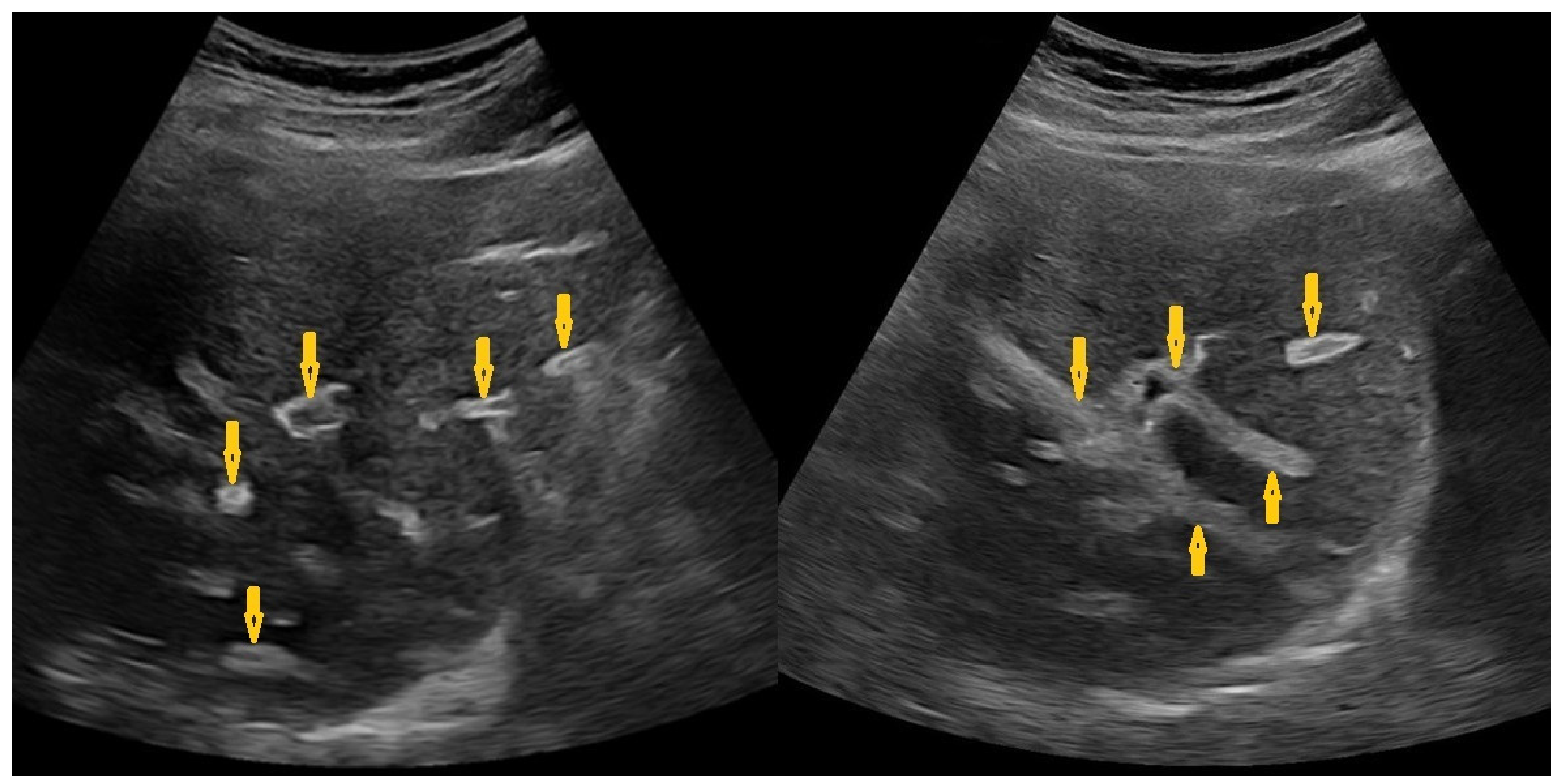
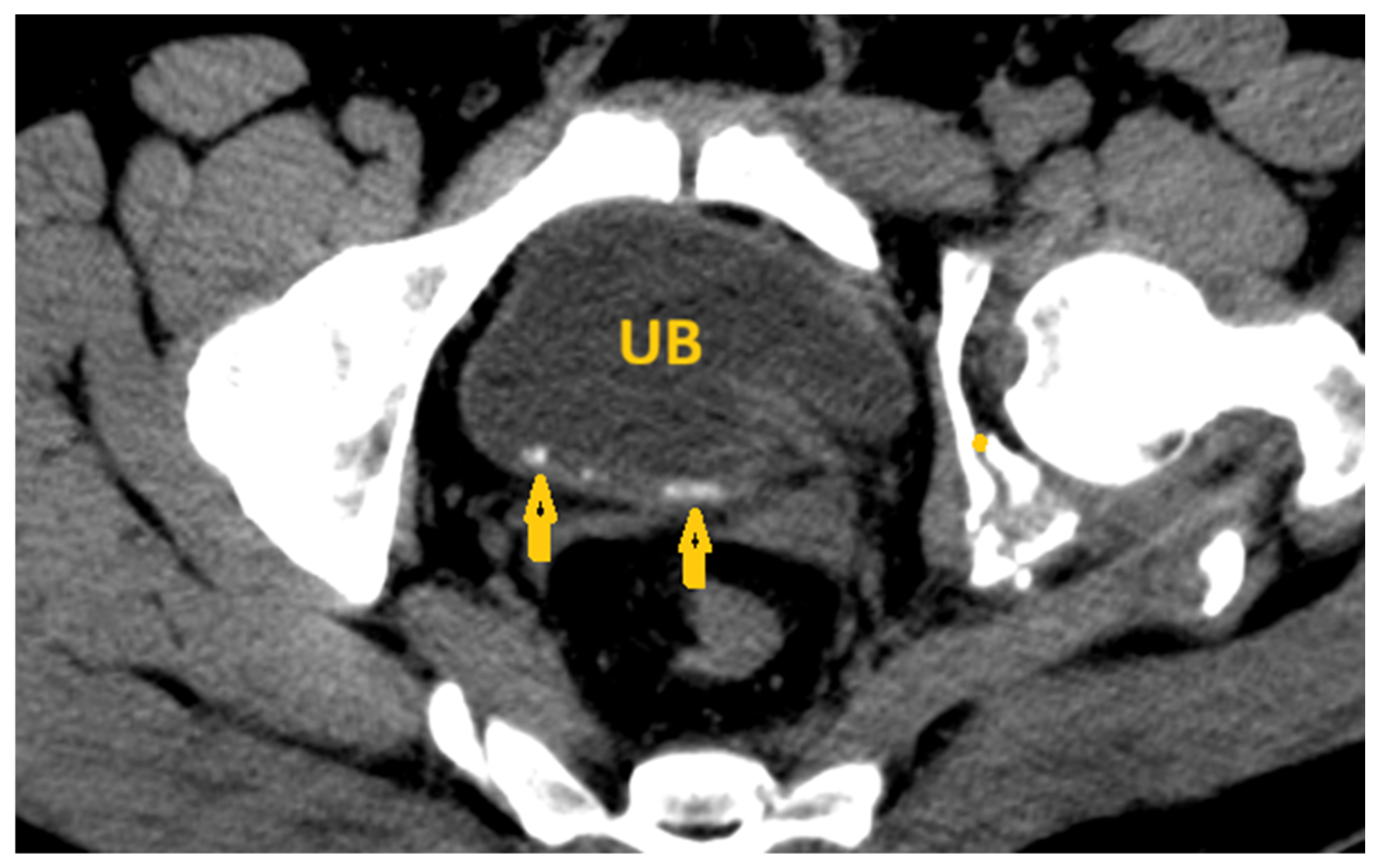
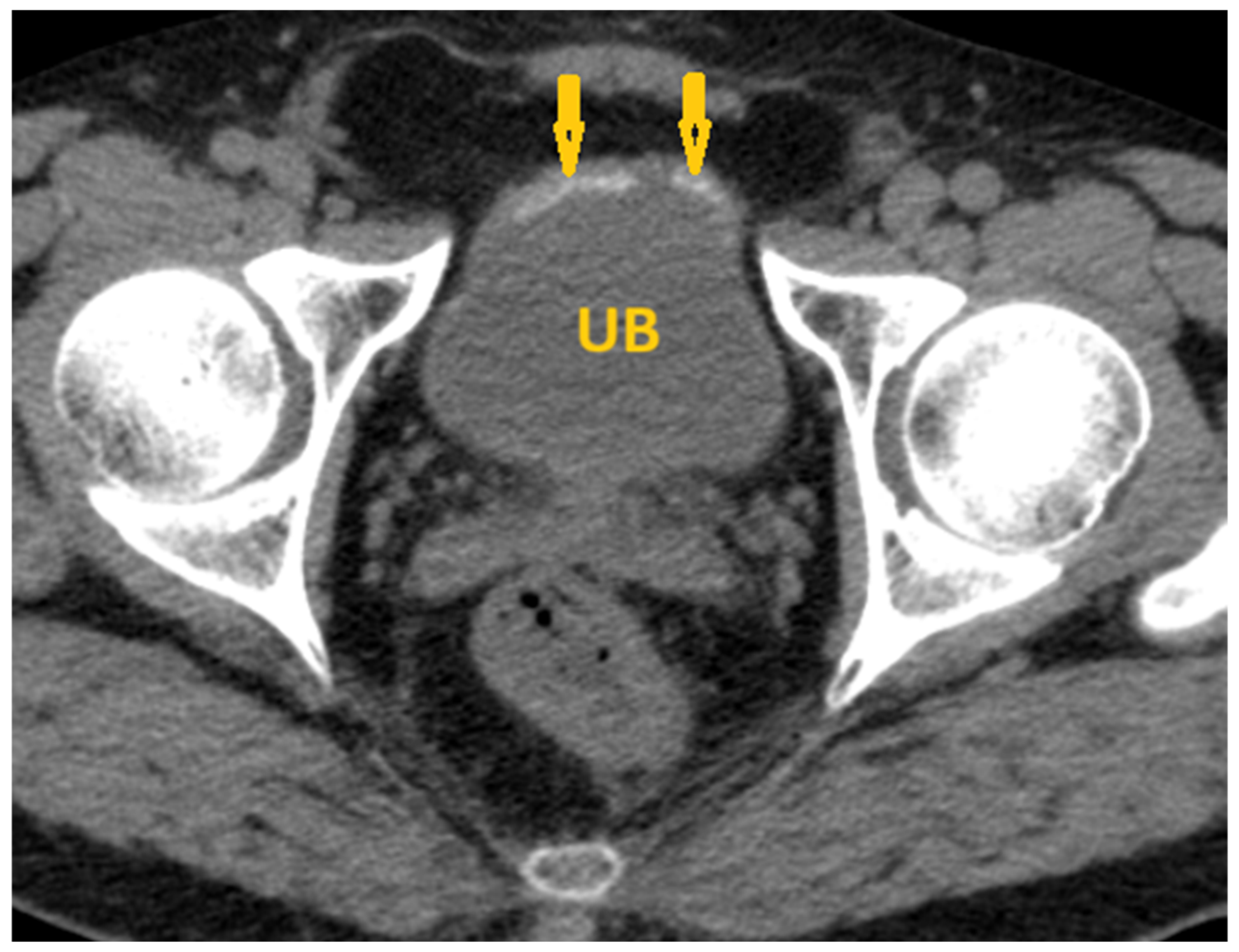
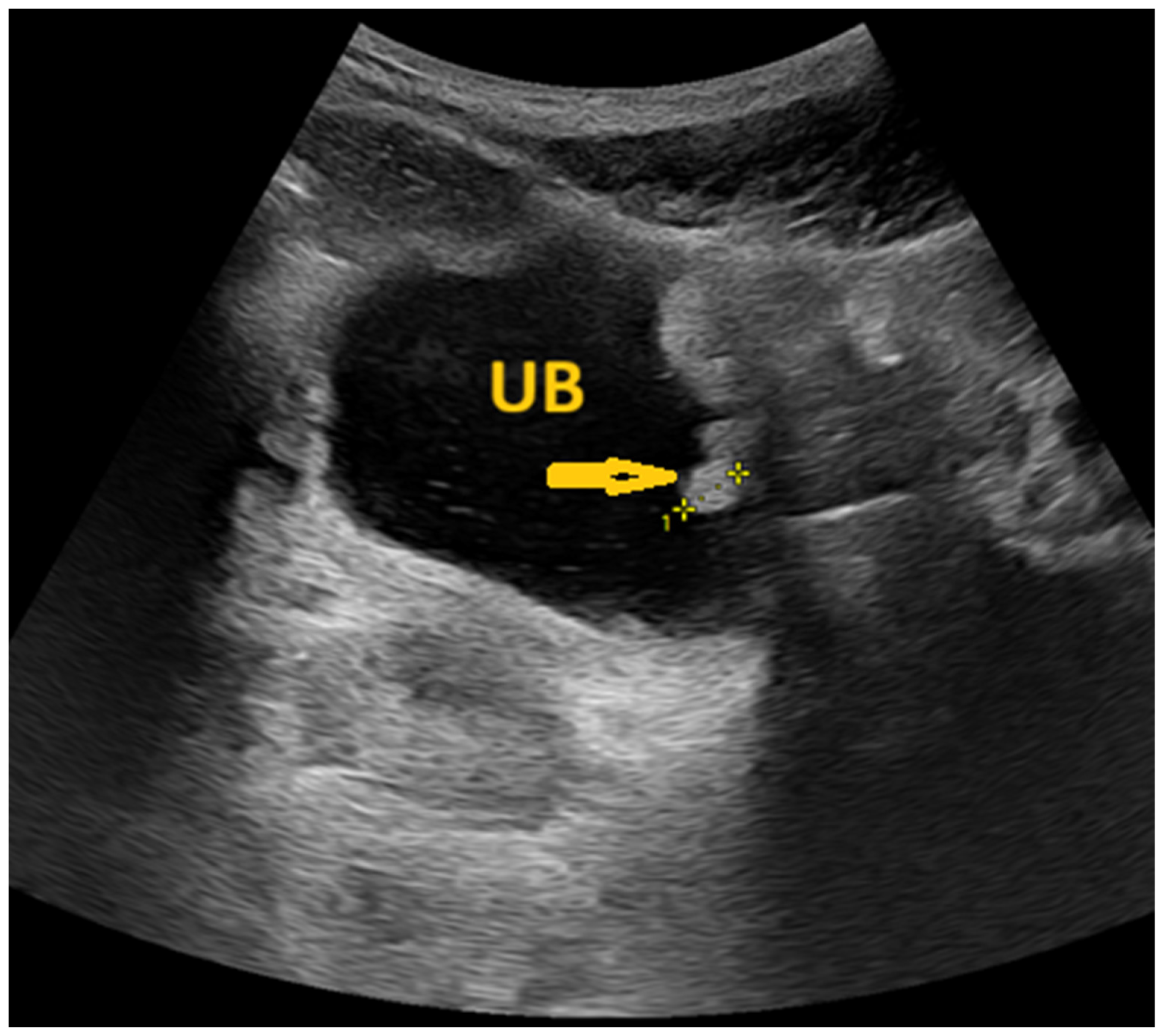
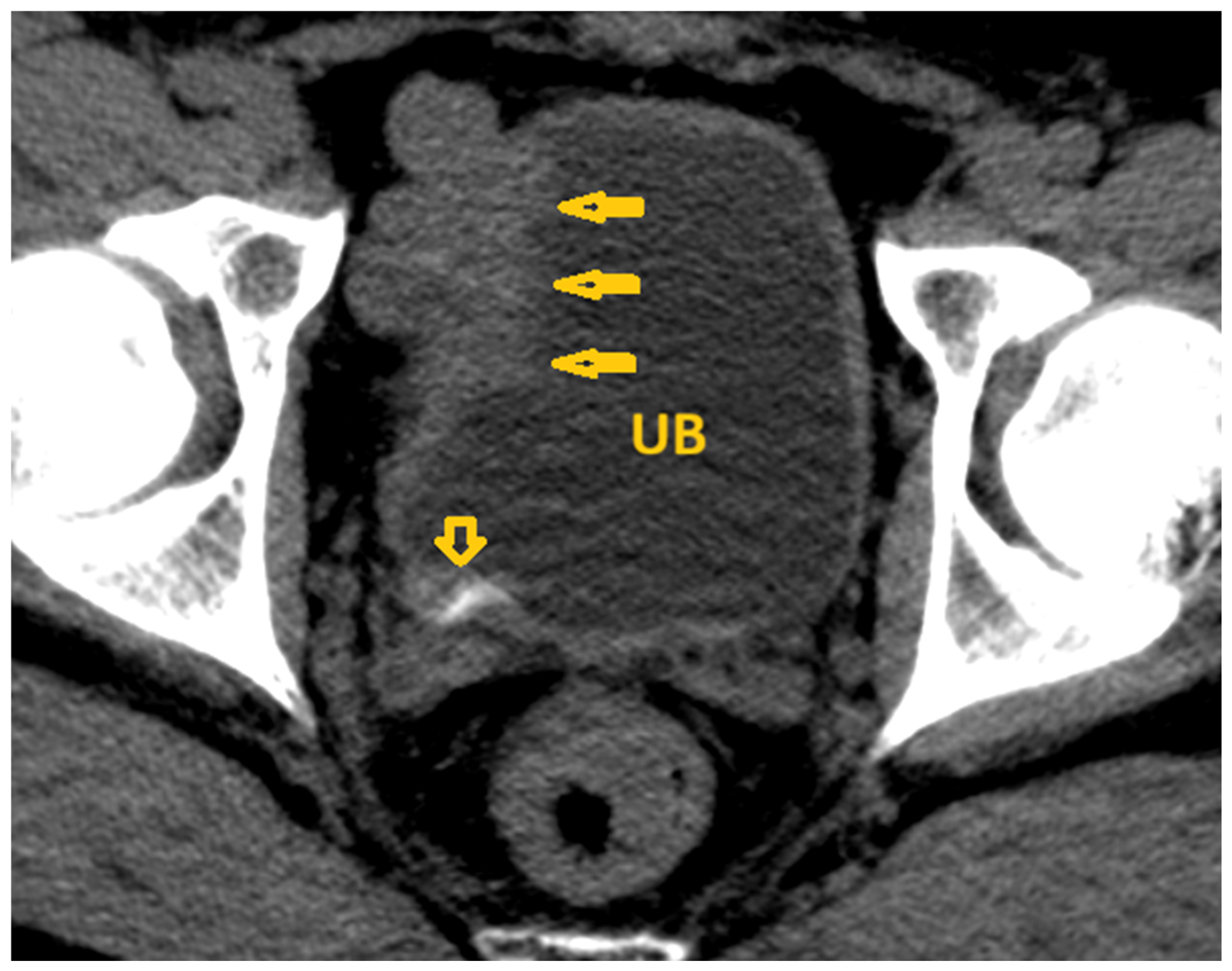
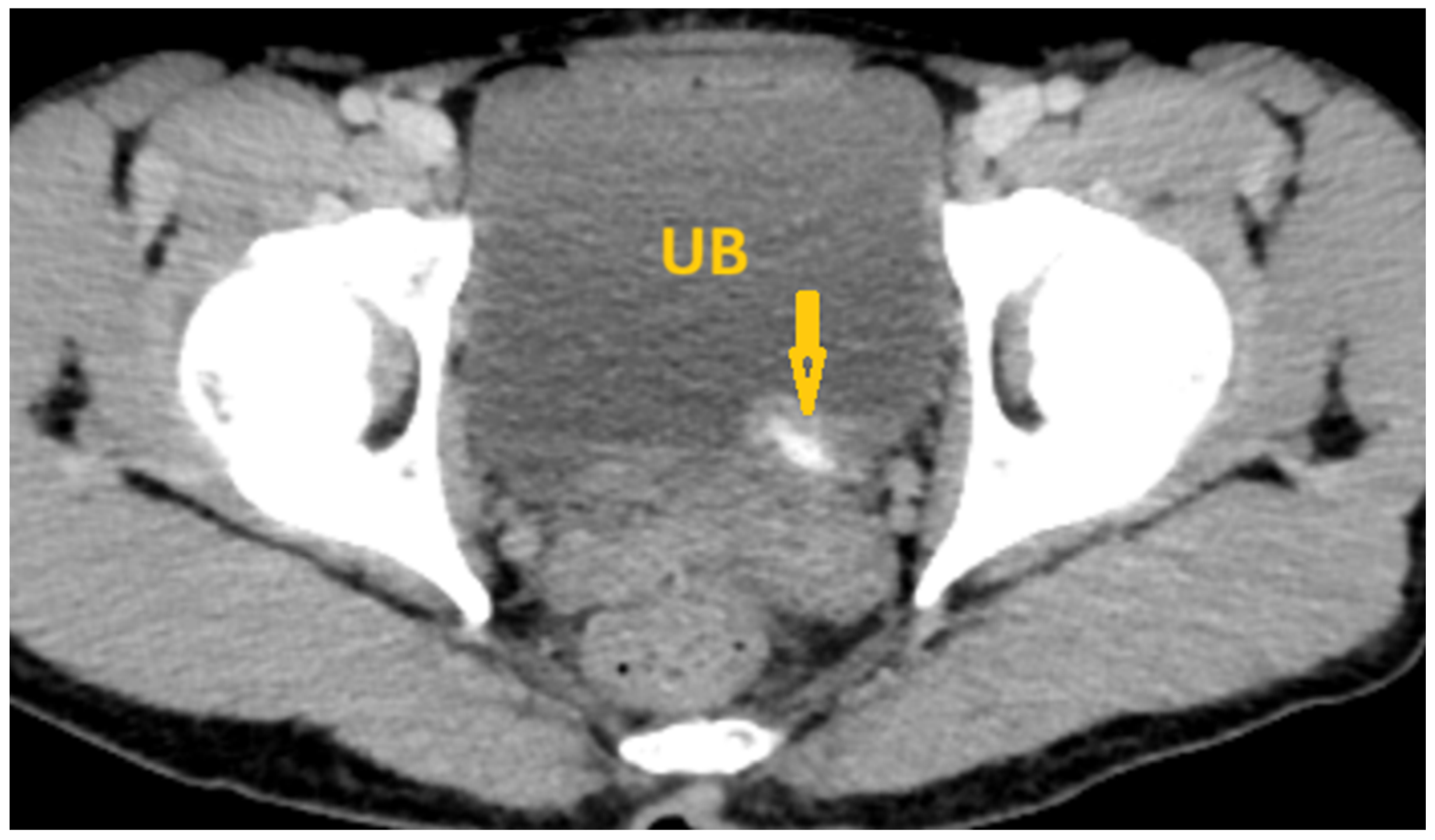
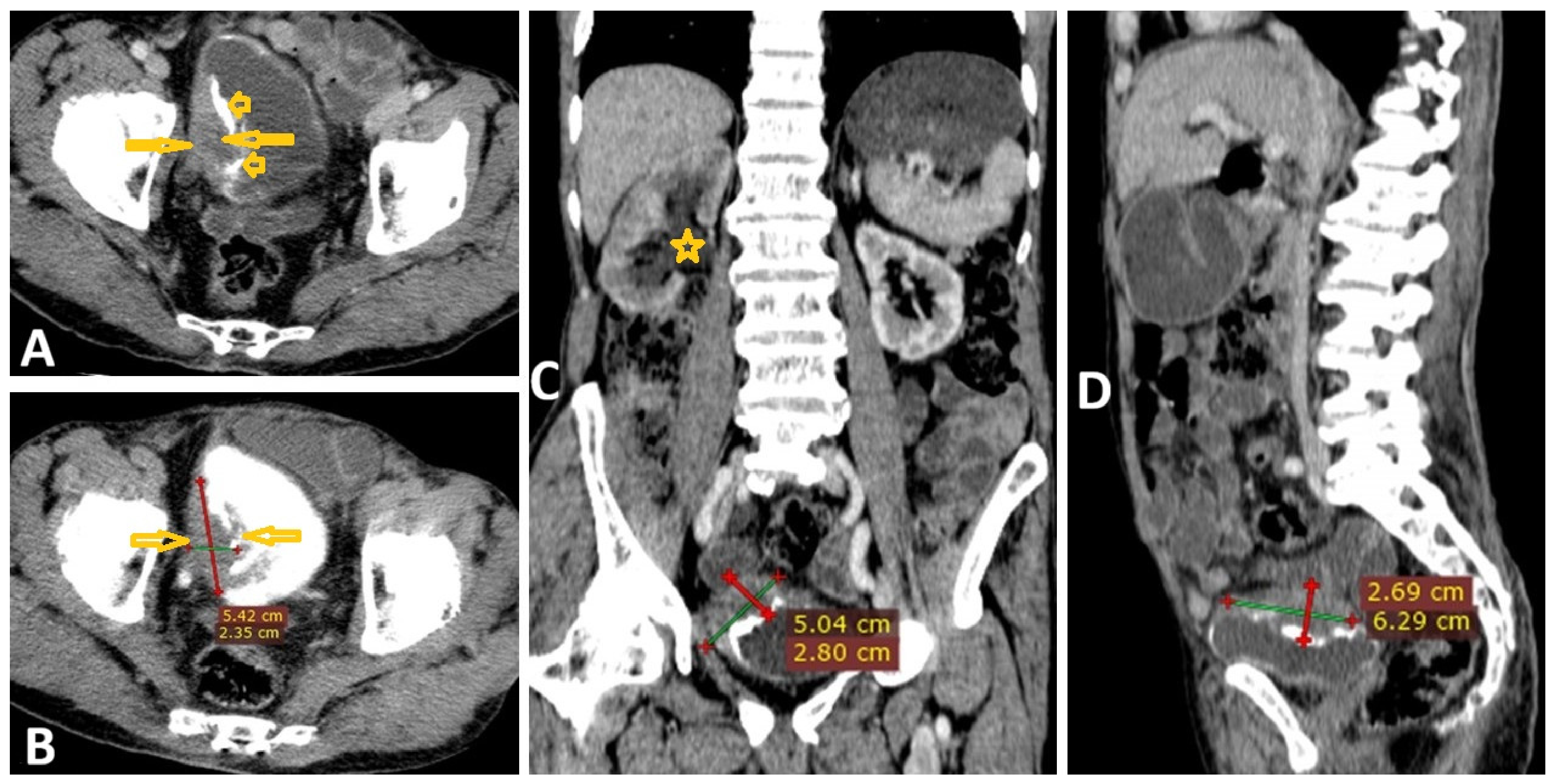
6.2. Medical Imaging Clues in the Genitourinary Schistosomiasis
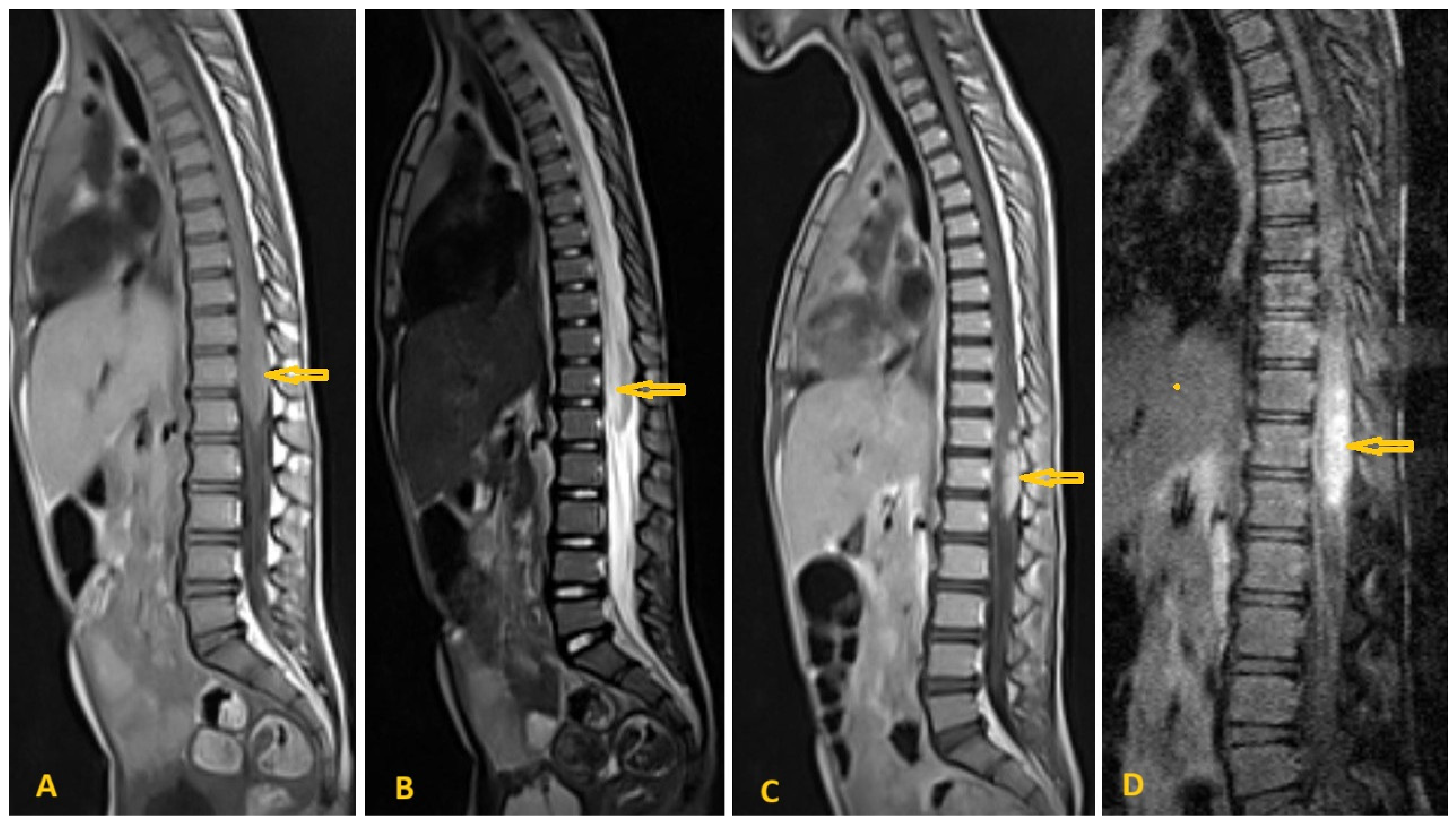
6.3. Medical Imaging Clues in Central Nervous System Schistosomiasis
7. Limitations
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| CT | Computed Tomography |
| MRI | Magnetic Resonance Imaging |
| WHO | World Health Organization |
| CNS | Central Nervous System |
| S | Schistosoma |
| POC-CCA | The Point-Of-Care Circulating Cathodic Antigen |
| NAATs | Nucleic Acid Amplification Tests |
| PCR | Polymerase Chain Reaction |
| cm | Centimeter |
References
- Singer, B.J.; Gomes, M.; Coulibaly, J.T.; Daigavane, M.; Tan, S.T.; Bogoch, I.I.; Lo, N.C. Population-level impact of mass drug administration against schistosomiasis with anthelmintic drugs targeting juvenile schistosomes: A modelling study. Lancet Microbe 2025, 6, 101065. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Caldwell, N.; Afshar, R.; Baragaña, B.; Bustinduy, A.L.; Caffrey, C.R.; Collins, J.J.; Fusco, D.; Garba, A.; Gardner, M.; Gomes, M.; et al. Perspective on Schistosomiasis Drug Discovery: Highlights from a Schistosomiasis Drug Discovery Workshop at Wellcome Collection, London, September 2022. ACS Infect. Dis. 2023, 9, 1046–1055. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ally, O.; Kanoi, B.N.; Ochola, L.; Nyanjom, S.G.; Shiluli, C.; Misinzo, G.; Gitaka, J. Schistosomiasis diagnosis: Challenges and opportunities for elimination. PLoS Negl. Trop. Dis. 2024, 18, e0012282. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ponzo, E.; Midiri, A.; Manno, A.; Pastorello, M.; Biondo, C.; Mancuso, G. Insights into the epidemiology, pathogenesis, and differential diagnosis of schistosomiasis. Eur. J. Microbiol. Immunol. 2024, 14, 86–96. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- McManus, D.P.; Dunne, D.W.; Sacko, M.; Utzinger, J.; Vennervald, B.J.; Zhou, X. Schistosomiasis. Nat. Rev. Dis. Primers 2018, 4, 13. [Google Scholar] [CrossRef] [PubMed]
- Olveda, D.U.; Olveda, R.M.; Lam, A.K.; Chau, T.N.; Li, Y.; Gisparil, A.D., II; Ross, A.G. Utility of Diagnostic Imaging in the Diagnosis and Management of Schistosomiasis. Clin. Microbiol. 2014, 3, 142. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cozzi, D.; Bertelli, E.; Savi, E.; Verna, S.; Zammarchi, L.; Tilli, M.; Rinaldi, F.; Pradella, S.; Agostini, S.; Miele, V. Ultrasound findings in urogenital schistosomiasis: A pictorial essay. J. Ultrasound 2020, 23, 195–205. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Alsaedi, H.I.; Krsoom, A.M.; Alshoabi, S.A.; Alsharif, W.M. Investigation Study of Ultrasound Practitioners’ Awareness about Artefacts of Hepatobiliary Imaging in Almadinah Almunawwarah. Pak. J. Med. Sci. 2022, 38, 1526–1533. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cimini, A.; Ricci, M.; Gigliotti, P.E.; Pugliese, L.; Chiaravalloti, A.; Danieli, R.; Schillaci, O. Medical Imaging in the Diagnosis of Schistosomiasis: A Review. Pathogens 2021, 10, 1058. [Google Scholar] [CrossRef] [PubMed]
- Nation, C.S.; Da’dara, A.A.; Marchant, J.K.; Skelly, P.J. Schistosome migration in the definitive host. PLoS Negl. Trop. Dis. 2020, 14, e0007951. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Richter, J.; da Costa, C.P. The Parasite’s Life Cycle and the Patient. In Praziquantel; Spangenberg, T., Ed.; Springer: Cham, Switzerland, 2026. [Google Scholar] [CrossRef]
- Formenti, F.; Cortés, A.; Deiana, M.; Salter, S.; Parkhill, J.; Berriman, M.; Rinaldi, G.; Cantacessi, C. The Human Blood Fluke, Schistosoma mansoni, Harbors Bacteria Throughout the Parasite’s Life Cycle. J. Infect. Dis. 2023, 228, 1299–1303. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Llanwarne, F.; Helmby, H. Granuloma formation and tissue pathology in Schistosoma japonicum versus Schistosoma mansoni infections. Parasite Immunol. 2021, 43, e12778. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Santos, L.L.; Santos, J.; Gouveia, M.J.; Bernardo, C.; Lopes, C.; Rinaldi, G.; Brindley, P.J.; Costa, J.M.C.D. Urogenital Schistosomiasis-History, Pathogenesis, and Bladder Cancer. J. Clin. Med. 2021, 10, 205. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Miller, K.; Choudry, J.; Mahmoud, E.S.; Lodh, N. Accurate Diagnosis of Schistosoma mansoni and S. haematobium from Filtered Urine Samples Collected in Tanzania, Africa. Pathogens 2024, 13, 59. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vaillant, M.T.; Philippy, F.; Neven, A.; Barré, J.; Bulaev, D.; Olliaro, P.L.; Utzinger, J.; Keiser, J.; Garba, A.T. Diagnostic tests for human Schistosoma mansoni and Schistosoma haematobium infection: A systematic review and meta-analysis. Lancet Microbe 2024, 5, e366–e378. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hoekstra, P.T.; van Dam, G.J.; van Lieshout, L. Context-Specific Procedures for the Diagnosis of Human Schistosomiasis—A Mini Review. Front. Trop. Dis. 2021, 2, 722438. [Google Scholar] [CrossRef]
- Dessein, H.; Duflot, N.; Romano, A.; Opio, C.; Pereira, V.; Mola, C.; Kabaterene, N.; Coutinho, A.; Dessein, A. Genetic algorithms identify individuals with high risk of severe liver disease caused by schistosomes. Hum. Genet. 2020, 139, 821–831, Correction in Hum. Genet. 2020, 139, 833. https://doi.org/10.1007/s00439-020-02181-z. [Google Scholar] [CrossRef] [PubMed]
- Elbaz, T.; Esmat, G. Hepatic and intestinal schistosomiasis: Review. J. Adv. Res. 2013, 4, 445–452. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Andrianah, G.E.P.; Rakotomena, D.; Rakotondrainibe, A.; Rajaonarison Ny Ony, L.H.N.; Ranoharison, H.D.; Ratsimba, H.R.; Rajaonera, T.; Ahmad, A. Contribution of Ultrasonography in the Diagnosis of Periportal Fibrosis Caused by Schistosomiasis. J. Med. Ultrasound 2020, 28, 41–43. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lim, M.C.; Quek, E.J.W.; Chan, T.Y.H.; Teng, M.; Ng, C.H.; Gnanavelou, M.; Chen, V.L.; Fallowfield, J.A.; Zhang, H.; Noureddin, M.; et al. Systematic Review: Imaging-Based Morphological Criteria for Liver Cirrhosis-A Call to Standardise. Aliment. Pharmacol. Ther. 2026, 63, 1201–1214. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lu, P.; Dong, C.; Yang, G.; Zhou, Y.; Jia, N. The Imaging Manifestations of Liver Cirrhosis. In Radiology of Hepatobiliary Diseases; Li, H., Ed.; Springer: Singapore, 2025. [Google Scholar] [CrossRef]
- Chou, E.; Gadani, S.; Liu, X. Noninvasive and Invasive Methods for the Diagnosis of Portal Hypertension. Tech. Vasc. Interv. Radiol. 2025, 28, 101054. [Google Scholar] [CrossRef] [PubMed]
- Strickland, G.T. Gastrointestinal manifestations of schistosomiasis. Gut 1994, 35, 1334–1337. [Google Scholar] [CrossRef] [PubMed] [PubMed Central][Green Version]
- Emara, M.H.; Mahros, A.M.; Rasheda, A.M.A.; Radwan, M.I.; Mohamed, B.; Abdelrazik, O.; Elazab, M.; Elbatae, H. Schistosomal (bilharzial) polyps: Travel through the colon and beyond. World J. Gastroenterol. 2023, 29, 4156–4165. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Akhtar, M.M.; ALJuhani, N.; Younus, D.; Alsahafi, A.H.; Abouhamda, A. Schistosomiasis Mansoni Manifesting as Multiple Colonic Polyps. Cureus 2020, 12, e11755. [Google Scholar] [CrossRef]
- Elhoseeny, M.M.; Sallam, A.; Eladl, A.E.; Othman, A.A.A. Bleeding rectal polyp as an atypical presentation of intestinal schistosomiasis: A case report from Egypt. PLoS Negl. Trop. Dis. 2026, 20, e0013972. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Shebel, H.M.; Elsayes, K.M.; Abou El Atta, H.M.; Elguindy, Y.M.; El-Diasty, T.A. Genitourinary schistosomiasis: Life cycle and radiologic-pathologic findings. RadioGraphics 2012, 32, 1031–1046. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Sung, W.S. Calcification of the urinary bladder and ureter in schistosomiasis. Kidney Res. Clin. Pract. 2018, 37, 304–305. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lackey, E.K.; Horrall, S. Schistosomiasis. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2026. Available online: https://www.ncbi.nlm.nih.gov/books/NBK554434/ (accessed on 1 April 2026).
- El Boté, H.; Boughaleb, A. Tuberculosis of seminal vesicles: A rare case report. IDCases 2025, 41, e02288. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ladumor, H.; Al-Mohannadi, S.; Ameerudeen, F.S.; Ladumor, S.; Fadl, S. TB or not TB: A comprehensive review of imaging manifestations of abdominal tuberculosis and its mimics. Clin. Imaging 2021, 76, 130–143. [Google Scholar] [CrossRef] [PubMed]
- Alshoabi, S.A.; Almas, K.M.; Aldofri, S.A.; Hamid, A.M.; Alhazmi, F.H.; Alsharif, W.M.; Abdulaal, O.M.; Qurashi, A.A.; Aloufi, K.M.; Alsultan, K.D.; et al. The Diagnostic Deceiver: Radiological Pictorial Review of Tuberculosis. Diagnostics 2022, 12, 306. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cheng, P.Y.; Huang, Y.Y.; Jaw, F.S.; Chung, S.D.; Tsai, C.Y. Diffused bladder wall calcification in a survivor with severe coronavirus disease 2019: A case report. Medicine 2022, 101, e30314. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Orish, V.N.; Morhe, E.K.S.; Azanu, W.; Alhassan, R.K.; Gyapong, M. The parasitology of female genital schistosomiasis. Curr. Res. Parasitol. Vector Borne Dis. 2022, 2, 100093. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- de Wilton, A.; Aggarwal, D.; Jäger, H.R.; Manji, H.; Chiodini, P.L. Delayed diagnosis of spinal cord schistosomiasis in a non-endemic country: A tertiary referral centre experience. PLoS Negl. Trop. Dis. 2021, 15, e0009161. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ferrari, T.C.; Moreira, P.R. Neuroschistosomiasis: Clinical symptoms and pathogenesis. Lancet Neurol. 2011, 10, 853–864. [Google Scholar] [CrossRef] [PubMed]
- Alanazi, R.F.; Al Karawi, M.; Almalki, A.; Sufiani, F.; Al Karawi, S. Schistosomiasis Involving the Central Nervous System: Case Report of a Rare Complication. Case Rep. Surg. 2023, 2023, 9968155. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Arega, G.; Adane, L.; Mekonnen, E.; Negussie, M.A. Spinal schistosomiasis masquerading as spinal cord tumor in a 12-year-old male adolescent: A case report. Radiol. Case Rep. 2025, 20, 1717–1720. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]








Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Alshoabi, S.A.; Magram, A.O.; Alkalady, A.H.; Al-Magtari, R.R.; Almas, K.M.; Al-Sayaghi, K.M.; Hamid, A.M.; Alhazmi, F.H.; Qurashi, A.A.; Alsharif, W.; et al. Schistosoma mansoni and Haematobium: Radiological Diagnostic Clues and Pathophysiology. Pathogens 2026, 15, 536. https://doi.org/10.3390/pathogens15050536
Alshoabi SA, Magram AO, Alkalady AH, Al-Magtari RR, Almas KM, Al-Sayaghi KM, Hamid AM, Alhazmi FH, Qurashi AA, Alsharif W, et al. Schistosoma mansoni and Haematobium: Radiological Diagnostic Clues and Pathophysiology. Pathogens. 2026; 15(5):536. https://doi.org/10.3390/pathogens15050536
Chicago/Turabian StyleAlshoabi, Sultan Abdulwadoud, Abdullatif O. Magram, Abdulaziz H. Alkalady, Rafat Rashed Al-Magtari, Khaled M. Almas, Khaled Mohammed Al-Sayaghi, Abdullgabbar M. Hamid, Fahad H. Alhazmi, Abdulaziz A. Qurashi, Walaa Alsharif, and et al. 2026. "Schistosoma mansoni and Haematobium: Radiological Diagnostic Clues and Pathophysiology" Pathogens 15, no. 5: 536. https://doi.org/10.3390/pathogens15050536
APA StyleAlshoabi, S. A., Magram, A. O., Alkalady, A. H., Al-Magtari, R. R., Almas, K. M., Al-Sayaghi, K. M., Hamid, A. M., Alhazmi, F. H., Qurashi, A. A., Alsharif, W., Alsaedi, A., AbuAzzah, E., Algahtani, A., Alqfail, K. A., & Alshamrani, K. M. (2026). Schistosoma mansoni and Haematobium: Radiological Diagnostic Clues and Pathophysiology. Pathogens, 15(5), 536. https://doi.org/10.3390/pathogens15050536




