Increasing Case Numbers and Earlier Age at Diagnosis of Alveolar Echinococcosis: Insights from a 13-Year Retrospective Swiss Study
Abstract
1. Introduction
2. Methodology
2.1. Study Design and Setting
2.2. Patient Identification
2.3. Data Collection
2.4. Statistical Analysis
3. Results
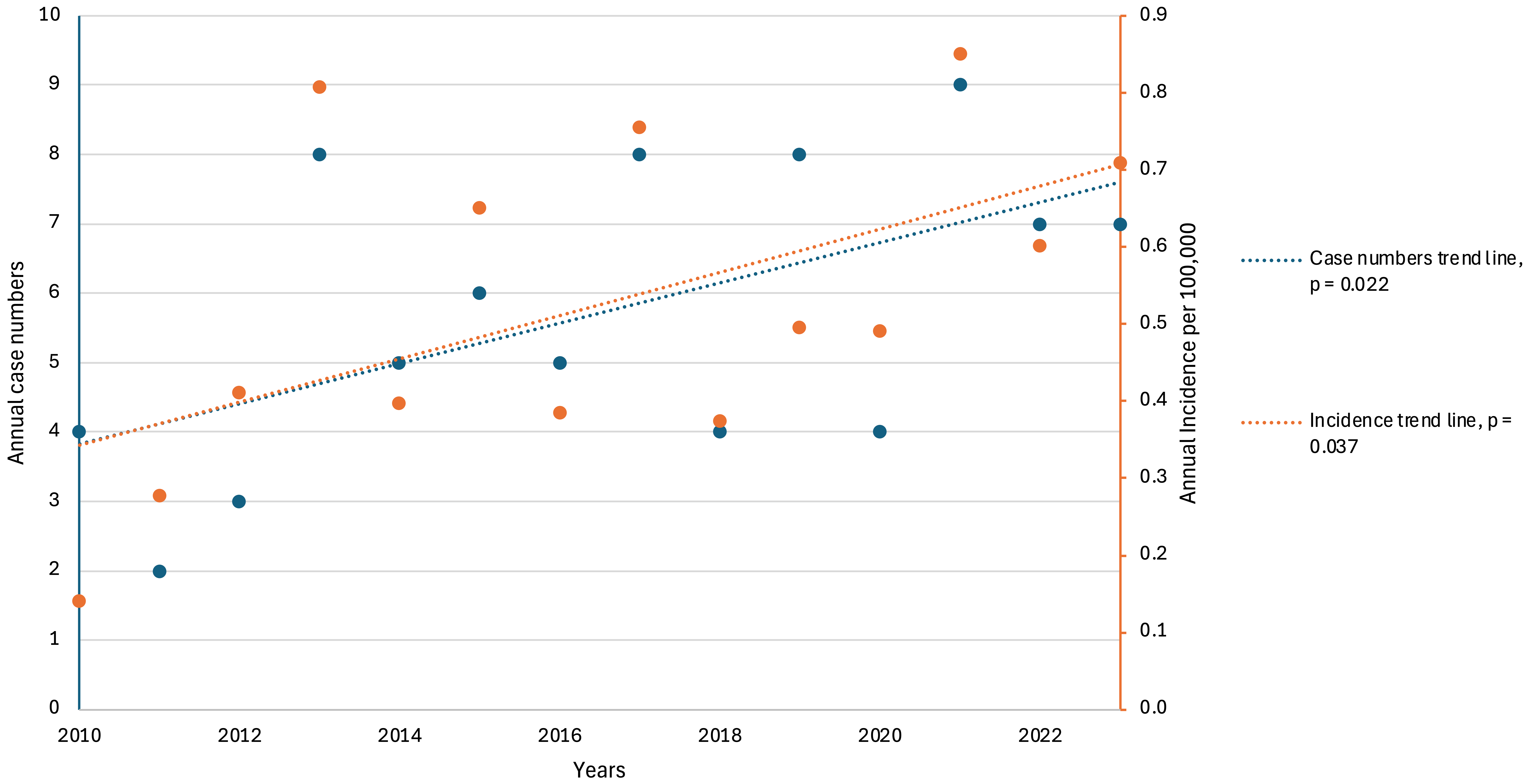
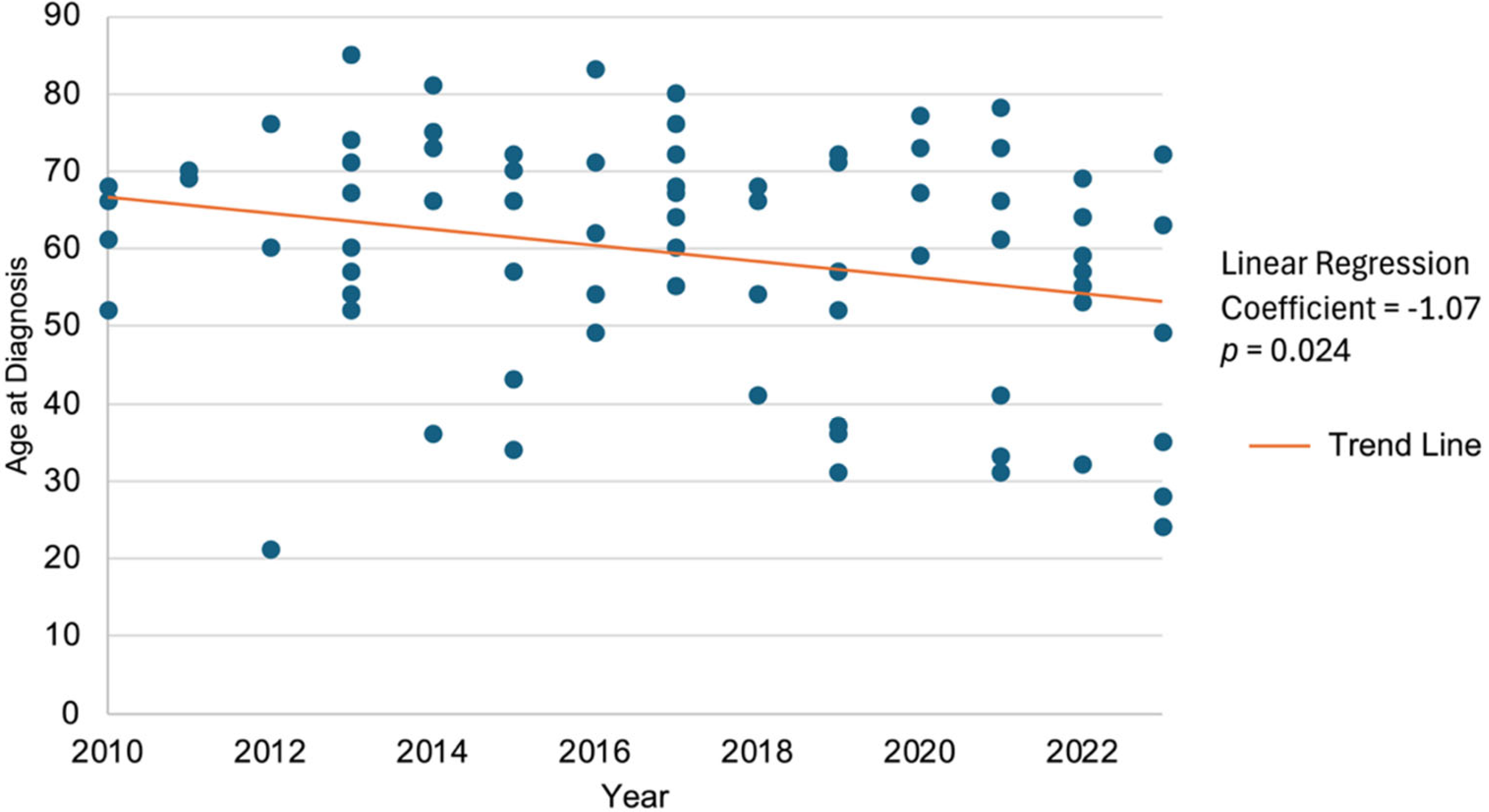
3.1. Baseline Cohort Characteristics
3.2. Clinical Presentation
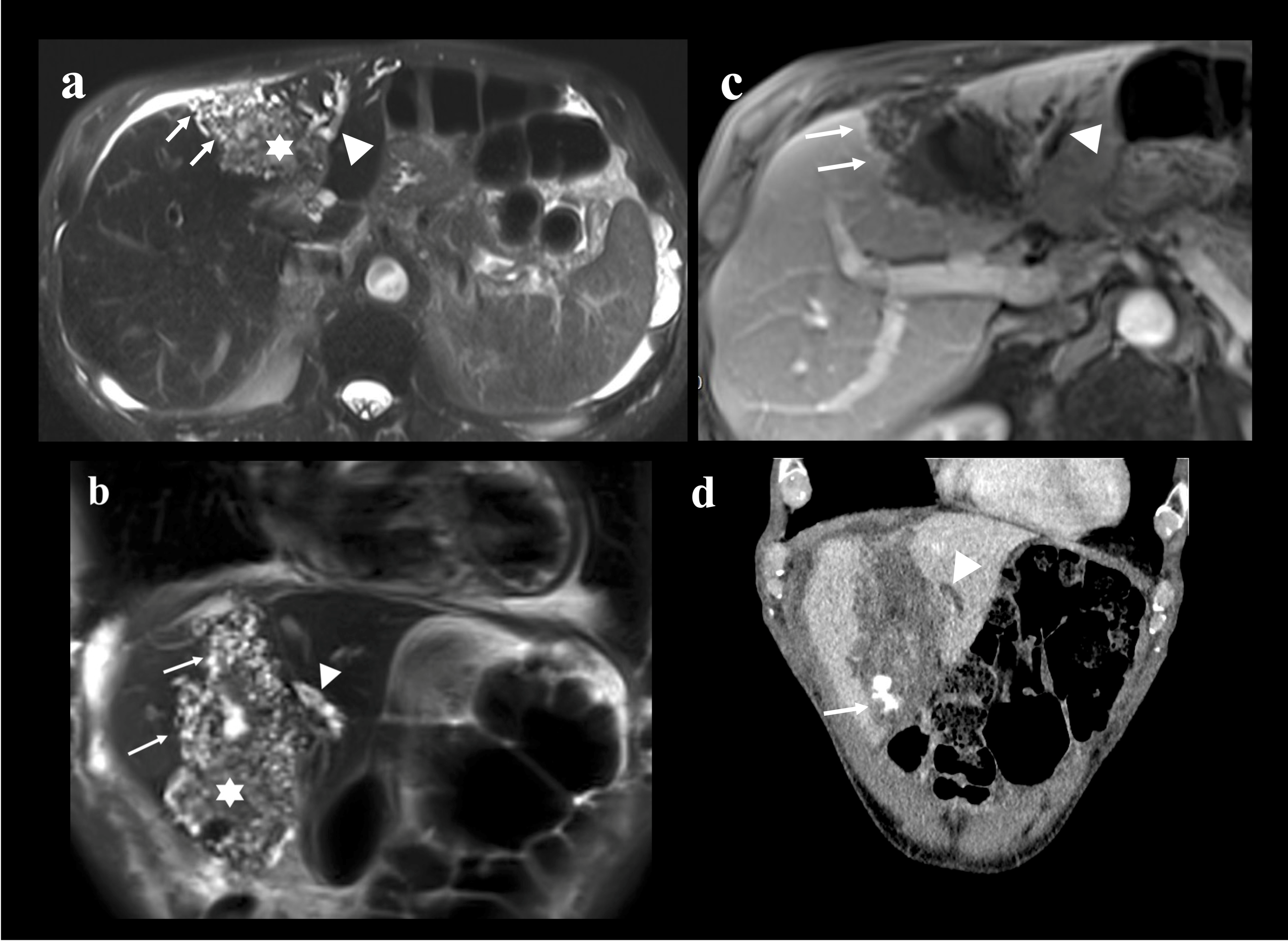
3.3. Radiology
3.4. Serology
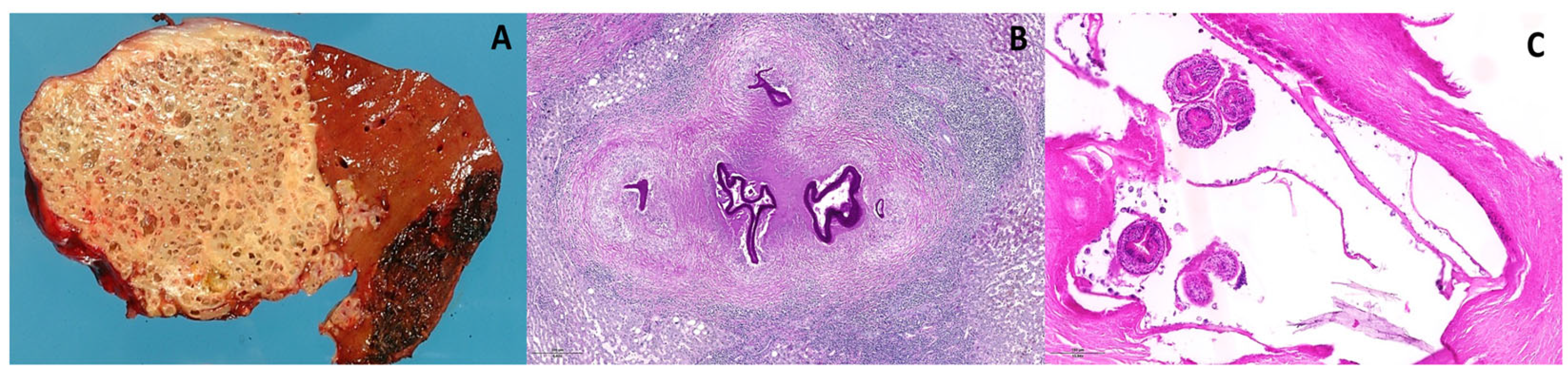
3.5. Histopathology
3.6. Therapy
3.7. Outcome
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AE | Alveolar Echinococcosis |
| ALP | Alkaline Phosphatase |
| ALT | Alanine Aminotransferase |
| AST | Aspartate Aminotransferase |
| CE | Cystic Echinococcosis |
| CRP | C-Reactive Protein |
| CT | Computed Tomography |
| EM | Echinococcus Multilocularis |
| GC | General Consent |
| GGT | Gamma-Glutamyl Transferase |
| HIV | Human Immunodeficiency Virus |
| MRI | Magnetic Resonance Imaging |
| OR | Odds Ratio |
| PET-CT | Positron Emission Tomography-Computed Tomography |
| PT | Prothrombin Time |
| US | Ultrasound |
| WHO | World Health Organization |
References
- Gottstein, B.; Stojkovic, M.; Vuitton, D.A.; Millon, L.; Marcinkute, A.; Deplazes, P. Threat of alveolar echinococcosis to public health—A challenge for Europe. Trends Parasitol. 2015, 31, 407–412. [Google Scholar] [CrossRef]
- World Health Organization. Echinococcosis; WHO: Geneva, Switzerland, 2024; Available online: https://www.who.int/health-topics/echinococcosis (accessed on 8 May 2025).
- Torgerson, P.R.; Devleesschauwer, B.; Praet, N.; Speybroeck, N.; Willingham, A.L.; Kasuga, F.; Rokni, M.B.; Zhou, X.-N.; Fèvre, E.M.; Sripa, B.; et al. World Health Organization estimates of the global and regional disease burden of 11 foodborne parasitic diseases, 2010: A data synthesis. PLoS Med. 2015, 12, e1001920. [Google Scholar] [CrossRef] [PubMed]
- Kern, P.; Bardonnet, K.; Renner, E.; Auer, H.; Pawlowski, Z.; Ammann, R.W.; Vuitton, D.A.; Kern, P. European echinococcosis registry: Human alveolar echinococcosis, Europe, 1982–2000. Emerg. Infect. Dis. 2003, 9, 343–349. [Google Scholar] [CrossRef]
- Lundström-Stadelmann, B.; Rostami, A.; Frey, C.F.; Torgerson, P.R.; Riahi, S.M.; Bagheri, K.; Kaethner, M.; Lachenmayer, A.; Beldi, G.; Gasser, R.B.; et al. Human alveolar echinococcosis-global, regional, and national annual incidence and prevalence rates. Clin. Microbiol. Infect. 2025, 31, 1139–1145. [Google Scholar] [CrossRef] [PubMed]
- Deplazes, P.; Rinaldi, L.; Alvarez Rojas, C.A.; Torgerson, P.R.; Harandi, M.F.; Romig, T.; Antolova, D.; Schurer, J.M.; Lahmar, S.; Cringoli, G.; et al. Global distribution of alveolar and cystic echinococcosis. Adv. Parasitol. 2017, 95, 315–493. [Google Scholar] [CrossRef] [PubMed]
- Casulli, A.; Abela, B.; Petrone, D.; Šoba, B.; Dezsényi, B.; Karamon, J.; Millon, L.; Saarma, U.; Antolová, D.; Chappuis, F.; et al. Unveiling the incidences and trends of alveolar echinococcosis in Europe: A systematic review from the KNOW-PATH project. Lancet Infect. Dis. 2026, 26, e49–e61. [Google Scholar] [CrossRef]
- Craig, P.S.; Hegglin, D.; Lightowlers, M.W.; Torgerson, P.R.; Wang, Q. Echinococcosis: Control and prevention. Adv. Parasitol. 2017, 96, 55–158. [Google Scholar] [CrossRef]
- Combes, B.; Comte, S.; Raton, V.; Raoul, F.; Boué, F.; Umhang, G.; Favier, S.; Dunoyer, C.; Woronoff, N.; Giraudoux, P. Westward spread of Echinococcus multilocularis in foxes, France, 2005–2010. Emerg. Infect. Dis. 2012, 18, 2059–2062. [Google Scholar] [CrossRef]
- Liccioli, S.; Giraudoux, P.; Deplazes, P.; Massolo, A. Wilderness in the ‘city’ revisited: Different urbes shape transmission of Echinococcus multilocularis by altering predator and prey communities. Trends Parasitol. 2015, 31, 297–305. [Google Scholar] [CrossRef]
- Hegglin, D.; Bontadina, F.; Deplazes, P. Human–wildlife interactions and zoonotic transmission of Echinococcus multilocularis. Trends Parasitol. 2015, 31, 167–173. [Google Scholar] [CrossRef]
- European Food Safety Authority. E. multilocularis Infections in Animals; EFSA: Parma, Italy, 2015; Available online: https://www.efsa.europa.eu/en/efsajournal/pub/4373 (accessed on 18 August 2025).
- Schweiger, A.; Ammann, R.W.; Candinas, D.; Clavien, P.-A.; Eckert, J.; Gottstein, B.; Halkic, N.; Muellhaupt, B.; Prinz, B.M.; Reichen, J.; et al. Human alveolar echinococcosis after fox population increase, Switzerland. Emerg. Infect. Dis. 2007, 13, 878–882. [Google Scholar] [CrossRef]
- Gloor, S.; Bontadina, F.; Hegglin, D.; Deplazes, P.; Breitenmoser, U. The rise of urban fox population in Switzerland. Mamm. Biol. 2001, 66, 155–164. [Google Scholar] [CrossRef]
- Piarroux, M.; Piarroux, R.; Giorgi, R.; Knapp, J.; Bardonnet, K.; Sudre, B.; Watelet, J.; Dumortier, J.; Gérard, A.; Beytout, J.; et al. Clinical features and evolution of alveolar echinococcosis in France from 1982 to 2007: Results of a survey in 387 patients. J. Hepatol. 2011, 55, 1025–1033. [Google Scholar] [CrossRef] [PubMed]
- Autier, B.; Gottstein, B.; Millon, L.; Ramharter, M.; Gruener, B.; Bresson-Hadni, S.; Dion, S.; Robert-Gangneux, F. Alveolar echinococcosis in immunocompromised hosts. Clin. Microbiol. Infect. 2023, 29, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Lachenmayer, A.; Gebbers, D.; Gottstein, B.; Candinas, D.; Beldi, G. Elevated incidence of alveolar echinococcosis in immunocompromised patients. Food Waterborne Parasitol. 2019, 16, e00060. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.; Vuitton, L.; Tuxun, T.; Li, J.; Vuitton, D.A.; Zhang, W.; McManus, D.P. Echinococcosis: Advances in the 21st century. Clin. Microbiol. Rev. 2019, 32, e00075-18. [Google Scholar] [CrossRef]
- Brunetti, E.; Kern, P.; Vuitton, D.A. Writing Panel for the WHO-IWGE. Expert consensus for the diagnosis and treatment of cystic and alveolar echinococcosis in humans. Acta Trop. 2010, 114, 1–16. [Google Scholar] [CrossRef]
- Torgerson, P.R.; Schweiger, A.; Deplazes, P.; Pohar, M.; Reichen, J.; Ammann, R.W.; Tarr, P.E.; Halkik, N.; Müllhaupt, B. Alveolar echinococcosis: From a deadly disease to a well-controlled infection. Relative survival and economic analysis in Switzerland over the last 35 years. J. Hepatol. 2008, 49, 72–77. [Google Scholar] [CrossRef]
- Joliat, G.-R.; Roulin, D.; Labgaa, I.; Uldry, E.; Demartines, N.; Halkic, N.; Melloul, E. Nouveautés dans la prise en charge de l’échinococcose alvéolaire. Rev. Médicale Suisse 2023, 19, 1192–1195. [Google Scholar] [CrossRef]
- Pavlidis, E.T.; Galanis, I.N.; Pavlidis, T.E. Current considerations for the management of liver echinococcosis. World J. Gastroenterol. 2025, 31, 103973. [Google Scholar] [CrossRef]
- Ambregna, S.; Koch, S.; Sulz, M.C.; Grüner, B.; Öztürk, S.; Chevaux, J.B.; Sulima, M.; De Gottardi, A.; Napoléon, B.; Abergel, A.; et al. A European survey of perendoscopic treatment of biliary complications in patients with alveolar echinococcosis. Expert Rev. Anti-Infect. Ther. 2017, 15, 79–88. [Google Scholar] [CrossRef]
- Koch, S.; Bresson-Hadni, S.; Miguet, J.-P.; Crumbach, J.-P.; Gillet, M.; Mantion, G.-A.; Heyd, B.; Vuitton, D.-A.; Minello, A.; Kurtz, S.; et al. Experience of liver transplantation for incurable alveolar echinococcosis: A 45-case European collaborative report. Transplantation 2003, 75, 856. [Google Scholar] [CrossRef]
- Universimed. Alveolar Echinococcosis in Switzerland. Available online: https://www.universimed.com/ch/article/gastroenterologie/alveolar-switzerland-265336 (accessed on 26 March 2025).
- Deplazes, P.; Hegglin, D.; Gloor, S.; Romig, T. Wilderness in the city: The urbanization of Echinococcus multilocularis. Trends Parasitol. 2004, 20, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Deibel, A.; Kindler, Y.; Mita, R.; Ghafoor, S.; Meyer Zu Schwabedissen, C.; Brunner-Geissmann, B.; Schweiger, A.; Grimm, F.; Reinehr, M.; Weber, A.; et al. Comprehensive survival analysis of alveolar echinococcosis patients, University Hospital Zurich, Zurich, Switzerland, 1973–2022. Emerg. Infect. Dis. 2025, 31, 906–916. [Google Scholar] [CrossRef] [PubMed]
- Ollagnon, M.; Bresson-Hadni, S.; Spahr, L.; Rubbia-Brandt, L.; Toso, C.; Chappuis, F. Alveolar echinococcosis in the canton of Geneva between 2010 and 2021: A descriptive analysis. Swiss Med. Wkly. 2025, 155, 3863. [Google Scholar] [CrossRef] [PubMed]
- Bresson-Hadni, S.; Delabrousse, E.; Blagosklonov, O.; Bartholomot, B.; Koch, S.; Miguet, J.-P.; André Mantion, G.; Angèle Vuitton, D. Imaging aspects and non-surgical interventional treatment in human alveolar echinococcosis. Parasitol. Int. 2006, 55, S267–S272. [Google Scholar] [CrossRef]
- Bebezov, B.; Mamashev, N.; Umetaliev, T.; Ziadinov, I.; Craig, P.S.; Joekel, D.E.; Deplazes, P.; Grimm, F.; Torgerson, P.R. Intense focus of alveolar echinococcosis, South Kyrgyzstan. Emerg. Infect. Dis. 2018, 24, 1119–1122. [Google Scholar] [CrossRef]
- Grüner, B.; Kern, P.; Mayer, B.; Gräter, T.; Hillenbrand, A.; Barth, T.E.F.; Muche, R.; Henne-Bruns, D.; Kratzer, W.; Kern, P. Comprehensive diagnosis and treatment of alveolar echinococcosis: A single-center, long-term observational study of 312 patients in Germany. GMS Infect. Dis. 2017, 5, Doc01. [Google Scholar] [CrossRef]
- Chauchet, A.; Grenouillet, F.; Knapp, J.; Richou, C.; Delabrousse, E.; Dentan, C.; Millon, L.; Di Martino, V.; Contreras, R.; Deconinck, E.; et al. Increased incidence and characteristics of alveolar echinococcosis in patients with immunosuppression-associated conditions. Clin. Infect. Dis. 2014, 59, 1095–1104. [Google Scholar] [CrossRef]
- Vuitton, D.A.; Gottstein, B. Echinococcus multilocularis and its intermediate host: A model of parasite-host interplay. BioMed Res. Int. 2010, 2010, 923193. [Google Scholar] [CrossRef]
- Deibel, A.; Meyer zu Schwabedissen, C.; Husmann, L.; Grimm, F.; Deplazes, P.; Reiner, C.S.; Müllhaupt, B. Characteristics and clinical course of alveolar echinococcosis in patients with immunosuppression-associated conditions: A retrospective cohort study. Pathogens 2022, 11, 441. [Google Scholar] [CrossRef] [PubMed]
- Hotz, J.F.; Peters, L.; Kapp-Schwörer, S.; Theis, F.; Eberhardt, N.; Essig, A.; Grüner, B.; Hagemann, J.B. Evaluation of serological markers in alveolar echinococcosis emphasizing the correlation of PET-CTI tracer uptake with RecEm18 and echinococcus-specific IgG. Pathogens 2022, 11, 239. [Google Scholar] [CrossRef] [PubMed]

| Variable | Value |
|---|---|
| Median age at diagnosis in years (range) | 61.5 (21–85) |
| Male, n (%) | 40 (50.0) |
| Symptomatic at diagnosis, n (%) | 35 (43.8) |
| Cholangitis and/or jaundice at presentation, n (%) | 9 (11.3) |
| Immunosuppressed patients, n (%) | 17 (21.3) |
| Liver cirrhosis, n (%) | 4 (5.0) |
| Radiology 1, n (%) | 78 (97.5) |
| Median size of largest lesion in cm (range) | 6.0 (1.0–18) |
| Multifocal liver disease, n (%) | 43 (55.1) |
| Extra-hepatic disease 2, n (%) | 16 (20.5) |
| Histopathological confirmation, n (%) | 69 (86.3) |
| Surgical specimen, n (%) | 48 (60.0) |
| Surgical specimen and liver biopsy, n (%) | 17 (21.3) |
| Liver biopsy alone, n (%) | 3 (3.8) |
| Post-mortem specimen, n (%) | 1 (1.4) |
| Treatments 3, n (%) | 80 (100) |
| Surgery combined with benzimidazole, n (%) | 64 (80.0) |
| Benzimidazole alone, n (%) | 13 (16.3) |
| Surgery alone, n (%) | 1 (1.3) |
| Watch and wait, n (%) | 2 (2.5) |
| Liver transplantation, n (%) | 1 (1.3) |
| Outcomes 4, n (%) | 74 (92.5) |
| Remission, n (%) | 50 (67.6) |
| Stable disease, n (%) | 19 (25.7) |
| Progressive disease, n (%) | 5 (6.8) |
| Variable | Immunocompetent | Immunosuppressed | p-Value (OR 1) | |
|---|---|---|---|---|
| Total number of patients, n (%) | 63 (79) | 17 (21) | ||
| Demography | age in years, median (range) | 64 (21–83) | 60 (28–85) | 0.37 |
| Clinical presentation | Symptomatic disease, n (%) n = 38 | 32 (50.8) | 3 (17.6) | 0.026 (0.21) |
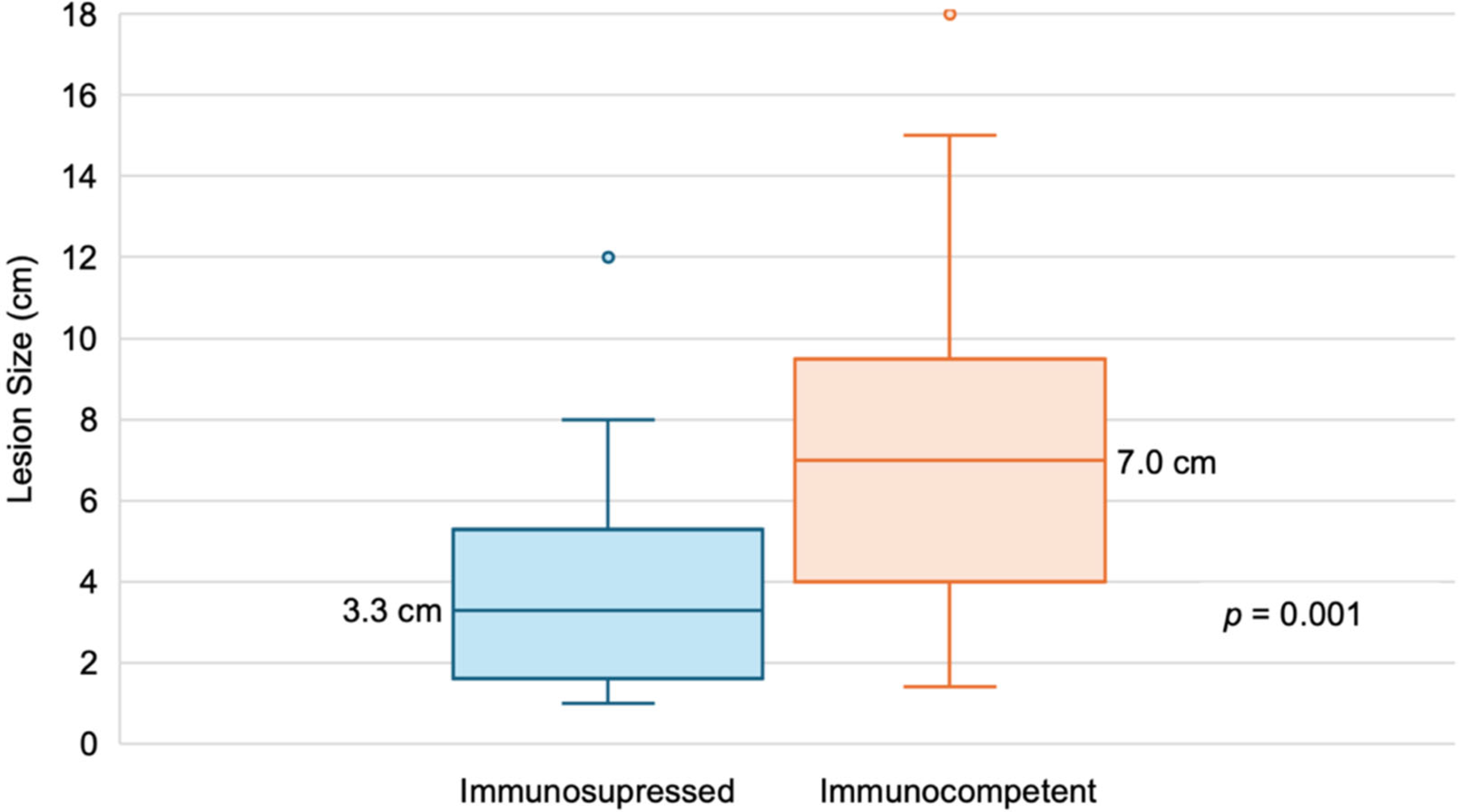
| Radiology 2 | Size of largest lesion in cm, median (range) | 7 (1.4–18.0) | 3.3 (1.0–12.-0) | 0.001 |
| Multifocal liver disease, n (%) n = 43 | 36 (59.0) | 7 (41.2) | 0.27 (0.49) | |
| Extra-hepatic disease, n (%) n = 16 | 15 (24.6) | 1 (5.9) | 0.17 (0.20) | |
| Serology 3 | Negative, n (%) | 1 (1.6) | 2 (14.3) | 0.12 (8.0) |
| Pathology | Biopsy, n (%) n = 20 | 13 (20.6) | 7 (41.2) | 0.11 (2.69) |
| Therapy 4 | Surgery combined with benzimidazole, n (%) n = 64 | 51 (81.0) | 13 (76.5) | 0.74 (0.76) |
| Benzimidazole alone, n (%) n = 14 | 10 (15.9) | 4 (23.5) | 0.48 (1.63) | |
| Outcome 5 | Remission, n (%) n = 50 | 39 (66.1) | 11 (73.3) | 0.34 (2.15) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Weikert, L.; Kasmi, S.; Schneider, M.; Faouzi, M.; Schmidt Kobbe, S.; Sempoux, C.; Cavassini, M.; Gauthiez, E.; Uldry, E.; Fraga, M. Increasing Case Numbers and Earlier Age at Diagnosis of Alveolar Echinococcosis: Insights from a 13-Year Retrospective Swiss Study. Pathogens 2026, 15, 495. https://doi.org/10.3390/pathogens15050495
Weikert L, Kasmi S, Schneider M, Faouzi M, Schmidt Kobbe S, Sempoux C, Cavassini M, Gauthiez E, Uldry E, Fraga M. Increasing Case Numbers and Earlier Age at Diagnosis of Alveolar Echinococcosis: Insights from a 13-Year Retrospective Swiss Study. Pathogens. 2026; 15(5):495. https://doi.org/10.3390/pathogens15050495
Chicago/Turabian StyleWeikert, Lasse, Sophie Kasmi, Michael Schneider, Mohamed Faouzi, Sabine Schmidt Kobbe, Christine Sempoux, Matthias Cavassini, Emeline Gauthiez, Emilie Uldry, and Montserrat Fraga. 2026. "Increasing Case Numbers and Earlier Age at Diagnosis of Alveolar Echinococcosis: Insights from a 13-Year Retrospective Swiss Study" Pathogens 15, no. 5: 495. https://doi.org/10.3390/pathogens15050495
APA StyleWeikert, L., Kasmi, S., Schneider, M., Faouzi, M., Schmidt Kobbe, S., Sempoux, C., Cavassini, M., Gauthiez, E., Uldry, E., & Fraga, M. (2026). Increasing Case Numbers and Earlier Age at Diagnosis of Alveolar Echinococcosis: Insights from a 13-Year Retrospective Swiss Study. Pathogens, 15(5), 495. https://doi.org/10.3390/pathogens15050495







