Phenotype-Based Stratification and Early Prediction of Staphylococcal Infective Endocarditis: Development of a Pragmatic Bedside Score
Abstract
1. Introduction
- (1)
- to characterize pathogen-specific clinical phenotypes in IE,
- (2)
- to identify independent predictors of staphylococcal etiology,
- (3)
- to develop and internally validate a pragmatic clinical prediction model, and
- (4)
- to explore platelet kinetics as a potential adjunctive biomarker.
2. Materials and Methods
2.1. Study Design and Population
2.2. Data Collection
2.3. Statistical Analysis
2.4. Ethical Considerations
3. Results
3.1. Staphylococcal IE vs. Non-Staphylococcal IE
3.1.1. Baseline Demographic and Risk Profile
3.1.2. Valvular Substrate and Echocardiographic Characteristics
3.1.3. Clinical Presentation
3.1.4. Laboratory Markers of Inflammation and Organ Dysfunction
3.1.5. Complications and Clinical Outcomes
3.2. Predictors of Staphylococcal Etiology
3.2.1. Multivariable Logistic Regression Model
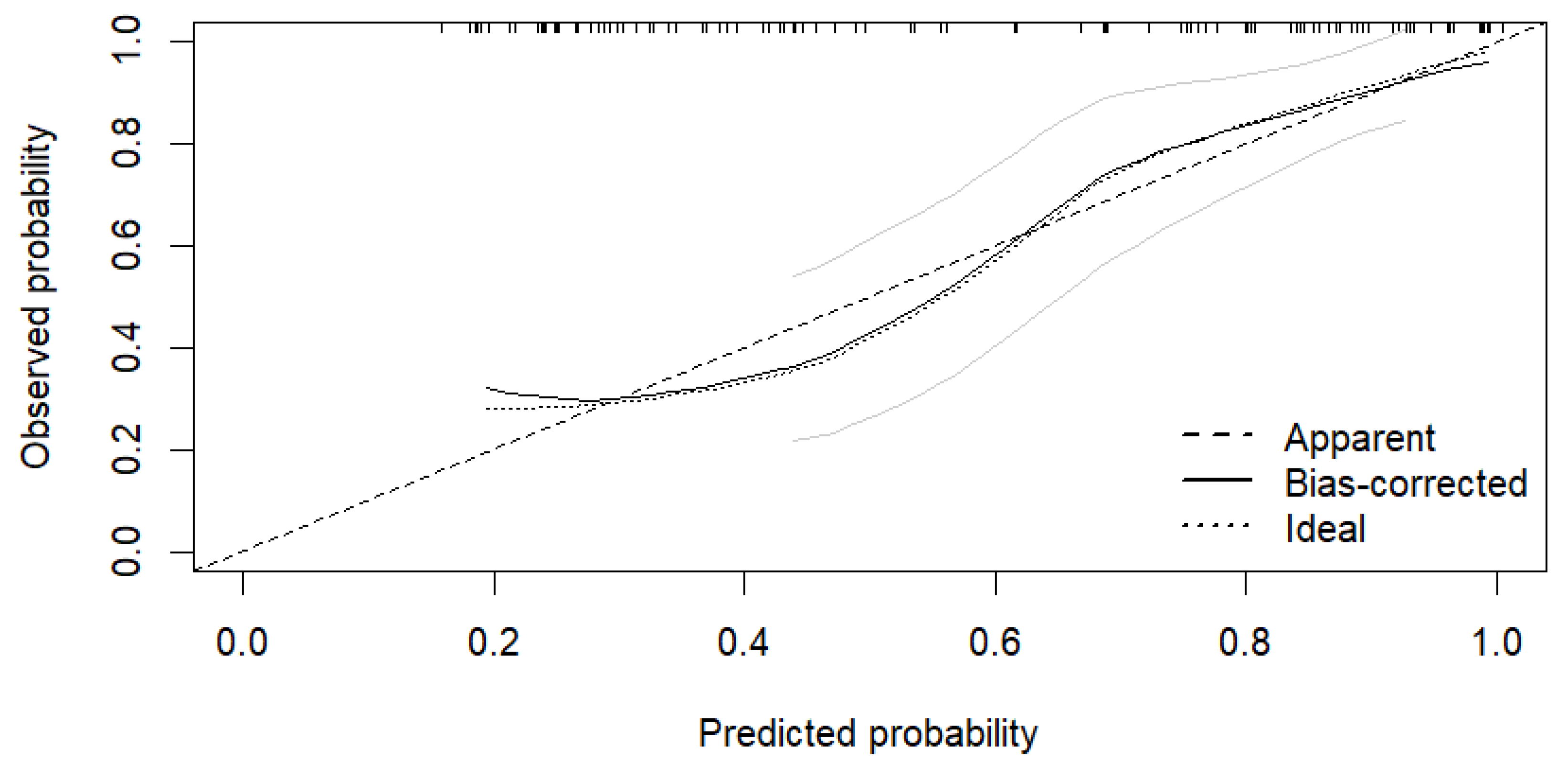
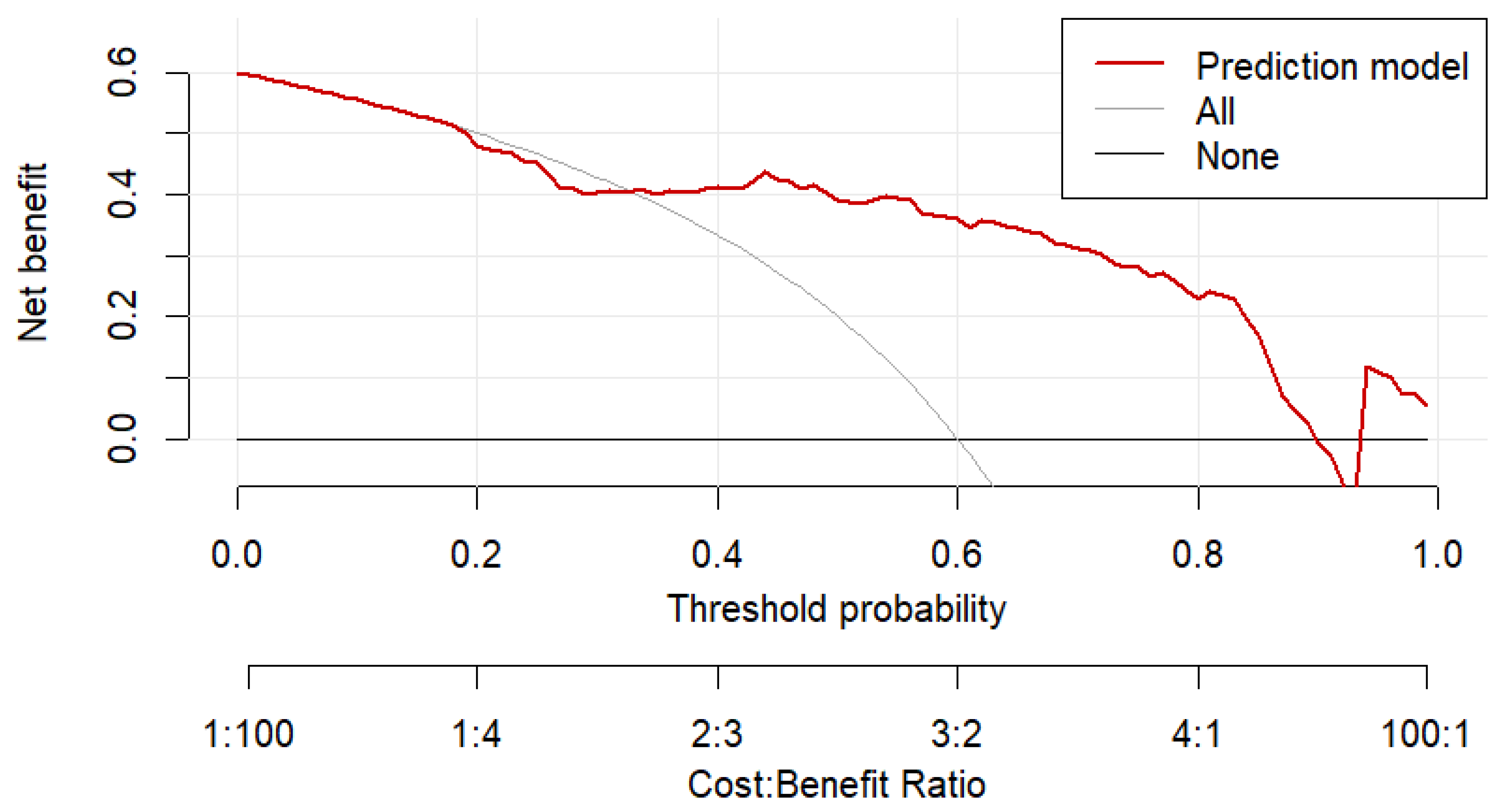
3.2.2. Decision Curve Analysis of Multivariable Logistic Regression Model
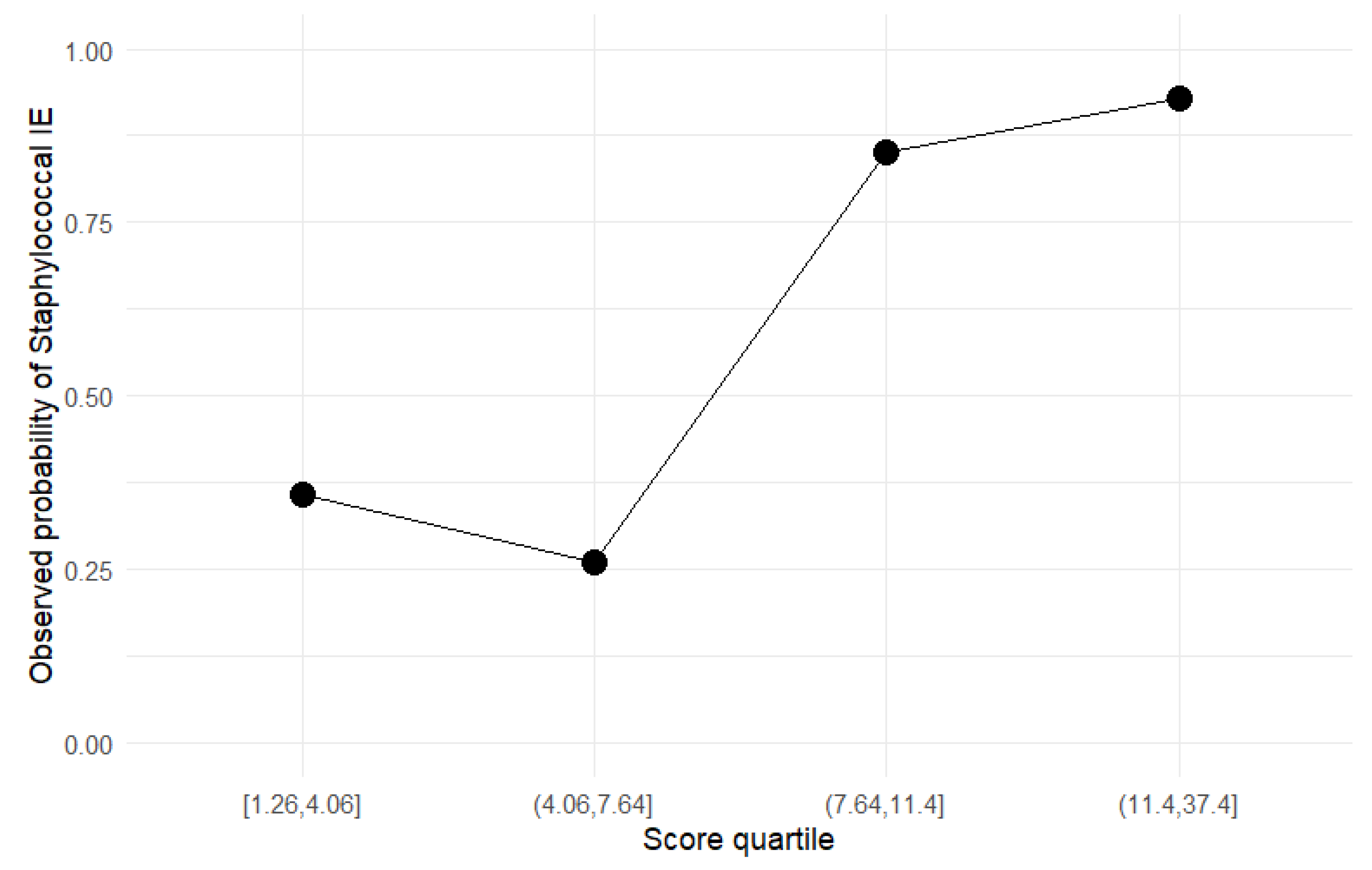
3.2.3. Clinical Risk Score
3.3. Platelet Dynamics During Hospitalization as a Complementary Biomarker
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AUC | Area Under the Curve |
| AV | Aortic Valve |
| BMI | Body Mass Index |
| CCI | Charlson Comorbidity Index |
| CI | Confidence Interval |
| CoNS | Coagulase-negative staphylococci |
| CRP | C-Reactive Protein |
| DCA | Decision Curve Analysis |
| GOT | Glutamic Oxaloacetic Transaminase (Aspartate Aminotransferase, AST) |
| HIV | Human Immunodeficiency Virus |
| HCV | Hepatitis C Virus |
| HBV | Hepatitis B Virus |
| IE | Infective Endocarditis |
| IQR | Interquartile Range |
| IVDU | Intravenous Drug Use |
| LDH | Lactate Dehydrogenase |
| MV | Mitral Valve |
| OR | Odds Ratio |
| ROC | Receiver Operating Characteristic |
| SaO2 | Peripheral Oxygen Saturation |
| SD | Standard Deviation |
| SOFA | Sequential Organ Failure Assessment |
| spp. | Species (plural, used after genus name) |
| TRIPOD | Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis |
| TTE | Transthoracic Echocardiography |
| TEE | Transesophageal Echocardiography |
| TV | Tricuspid Valve |
| PV | Pulmonary Valve |
| Δ | Delta (change or variation) |
References
- Martínez-Sellés, M.; Muñoz, P. Epidemiology, Diagnosis, Treatment, and Prognosis of Infective Endocarditis. J. Clin. Med. 2023, 12, 5705. [Google Scholar] [CrossRef] [PubMed]
- Miao, H.; Zhang, Y.; Zhang, Y.; Zhang, J. Update on the epidemiology, diagnosis, and management of infective endocarditis: A review. Trends Cardiovasc. Med. 2024, 34, 499–506. [Google Scholar] [CrossRef]
- Reisinger, M.; Kachel, M.; George, I. Emerging and Re-Emerging Pathogens in Valvular Infective Endocarditis: A Review. Pathogens 2024, 13, 543. [Google Scholar] [CrossRef]
- Borgonovo, F.; Petri, F.; Matsuo, T.; Igwilo-Alaneme, R.; Amin Alavi, S.M.; Mahmoud, O.K.; El Zein, S.; Passerini, M.; Murad, M.H.; DeSimone, D.C.; et al. Infective endocarditis meets native vertebral osteomyelitis: A mortality perspective. J. Bone Jt. Infect. 2025, 10, 425–435. [Google Scholar] [CrossRef]
- Jean, B.; Crolle, M.; Pollani, C.; Le Guilloux, A.; Martin-Blondel, G.; Tattevin, P.; Le Bot, A.; Luque Paz, D.; Guérin, F.; Cattoir, V.; et al. β-Lactam Inoculum Effect in Methicillin-Susceptible Staphylococcus aureus Infective Endocarditis. JAMA Netw. Open 2024, 7, e2451353. [Google Scholar] [CrossRef]
- Lefevre, B.; Habib, G.; Hoen, B.; Selton-Suty, C.; Philip, M.; Issa, N.; Danneels, P.; De La Chapelle, M.; Deschanvres, C.; Erpelding, M.L.; et al. Staphylococcus lugdunensis infective endocarditis: A multicentre international observational study. Infect. Dis. 2025, 58, 189–197. [Google Scholar] [CrossRef]
- Kedsawadevong, P.; Jamnongprasatporn, S.; Ratanasit, N. Streptococcus suis infective endocarditis in patients with Streptococcus suis bacteremia: A retrospective study of prevalence and outcomes. BMC Infect. Dis. 2024, 24, 1304. [Google Scholar] [CrossRef] [PubMed]
- Papadimitriou-Olivgeris, M.; Monney, P.; Frank, M.; Tzimas, G.; Tozzi, P.; Kirsch, M.; Van Hemelrijck, M.; Bauernschmitt, R.; Epprecht, J.; Guery, B.; et al. Evaluation of the 2023 Duke-ISCVID and 2023 Duke-ESC Clinical Criteria for the Diagnosis of Infective Endocarditis in a Multicenter Cohort of Patients With Staphylococcus aureus Bacteremia. Clin. Infect. Dis. 2024, 78, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Strömdahl, M.; Hagman, K.; Hedman, K.; Westman, A.; Hedenstierna, M.; Ursing, J. Time to Staphylococcus aureus Blood Culture Positivity as a Risk Marker of Infective Endocarditis: A Retrospective Cohort Study. Clin. Infect. Dis. 2025, 80, 727–734. [Google Scholar] [CrossRef] [PubMed]
- Arslan, M.; Kaleli, İ.; Kutlu, M. Prospective Clinical Follow-Up Results of Infective Endocarditis. Infect. Dis. Clin. Microbiol. 2024, 6, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Fowler, V.G.; Durack, D.T.; Selton-Suty, C.; Athan, E.; Bayer, A.S.; Chamis, A.L.; Dahl, A.; DiBernardo, L.; Durante-Mangoni, E.; Duval, X.; et al. The 2023 Duke-International Society for Cardiovascular Infectious Diseases Criteria for Infective Endocarditis: Updating the Modified Duke Criteria. Clin. Infect. Dis. 2023, 77, 518–526. [Google Scholar] [CrossRef]
- Glasheen, W.P.; Cordier, T.; Gumpina, R.; Haugh, G.; Davis, J.; Renda, A. Charlson Comorbidity Index: ICD-9 Update and ICD-10 Translation. Am. Health Drug Benefits 2019, 12, 188. [Google Scholar]
- Lin, Y.; Liu, F.; Gong, S.; Liao, B.; Liu, H.; Yuan, J.; Yu, D.; Qin, H.; Wu, M.; Dong, S. Validity of SOFA score as a prognostic tool for critically ill elderly patients with acute infective endocarditis. Rev. Cardiovasc. Med. 2021, 22, 967–973. [Google Scholar] [CrossRef]
- Asai, N.; Shiota, A.; Ohashi, W.; Watanabe, H.; Shibata, Y.; Kato, H.; Sakanashi, D.; Hagihara, M.; Koizumi, Y.; Yamagishi, Y.; et al. The SOFA score could predict the severity and prognosis of infective endocarditis. J. Infect. Chemother. 2019, 25, 965–971. [Google Scholar] [CrossRef]
- Bertolino, L.; Unar, A.; Patauner, F.; Gallo, R.; Peluso, A.M.C.; Delle Femine, A.; Infante, O.; Mercadante, S.; Luciano, F.; Manduca, S.; et al. Embolic complications in a large contemporary cohort of infective endocarditis: Do we need score model? Infection 2024, 53, 1101–1113. [Google Scholar] [CrossRef]
- Serban, A.M.; Pepine, D.; Inceu, A.; Dadarlat, A.; Achim, A. Embolic risk management in infective endocarditis: Predicting the ‘embolic roulette’. Open Heart 2025, 12, e003060. [Google Scholar] [CrossRef]
- Bursy, M.; Dudek, A.; Szkudlarek, W.; Linkiewicz, J.; Lizurej, W.; Buda, A.; Kubik, M.; Bogdanski, A.; Bogdanska, K.; Mazur, M.; et al. Infective Endocarditis in Patients with a History of Intravenous Drug Abuse. Cureus 2025, 17, e91270. [Google Scholar] [CrossRef]
- Sidik, A.I.; Chinikov, M.A.; Korjueva, L.S.; Abdi, E.; Seyedamirali, R.; Mobin, A.; Vepa, A.; Hela, K.; Tumaini, S.; Gaudensia, M.; et al. Infective Endocarditis in Special Populations: Epidemiology, Diagnostic Challenges, and Management Strategies. Cureus 2025, 17, e93293. [Google Scholar] [CrossRef]
- Moiz, A.; Omar, K.O. Infective Endocarditis with Coronary Artery Embolization and Multisystem Involvement in a Patient with a History of IV Drug Use: A Case Report. J. Community Hosp. Intern. Med. Perspect. 2025, 15, 49. [Google Scholar] [CrossRef]
- Nanu, A.A.; Lazăr DȘtefan Popescu, C.P.; Lazăr, M.I.; Nica, M.; Florescu, S.A. Characteristics of Infective Endocarditis in Intravenous Drug Users vs. Non-Users: A Retrospective Study Conducted in Bucharest, Romania. Medicina 2025, 61, 1785. [Google Scholar] [CrossRef]
- Diego-Yagüe, I.; Ramos-Martínez, A.; Muñoz, P.; Martínez-Sellés, M.; Machado, M.; de Alarcón, A.; Miró, J.M.; Rodríguez-Gacía, R.; Gutierrez-Díez, J.F.; Hidalgo-Tenorio, C.; et al. Clinical features and prognosis of prosthetic valve endocarditis due to Staphylococcus aureus. Eur. J. Clin. Microbiol. Infect. Dis. 2024, 43, 1989–2000. [Google Scholar] [CrossRef] [PubMed]
- Kildahl, H.A.; Brenne, E.L.; Dalen, H.; Wahba, A. Systemic embolization in infective endocarditis. Indian J. Thorac. Cardiovasc. Surg. 2023, 40, 40–46. [Google Scholar] [CrossRef]
- Crosby, H.A.; Kwiecinski, J.; Horswill, A.R. Staphylococcus aureus aggregation and coagulation mechanisms, and their function in host-pathogen interactions. Adv. Appl. Microbiol. 2016, 96, 1–41. [Google Scholar] [PubMed]
- Liesenborghs, L.; Verhamme, P.; Vanassche, T. Staphylococcus aureus, master manipulator of the human hemostatic system. J. Thromb. Haemost. 2018, 16, 441–454. [Google Scholar] [CrossRef]
- Polzin, A.; Dannenberg, L.; M’Pembele, R.; Mourikis, P.; Naguib, D.; Zako, S.; Helten, C.; Petzold, T.; Levkau, B.; Hohlfeld, T.; et al. Staphylococcus aureus increases platelet reactivity in patients with infective endocarditis. Sci. Rep. 2022, 12, 12933. [Google Scholar] [CrossRef] [PubMed]





| Staphylococcus spp. IE (n = 66) | Non-Staphylococcus spp. IE (n = 46) | Test Statistic Value | p-Value | |
|---|---|---|---|---|
| Females, n (%) | 16 (24.2%) | 14 (30.4%) | ꭓ2 = 0.530 | 0.467 a |
| Males, n (%) | 50 (75.8%) | 32 (69.6%) | ||
| Age (years) mean ± SD | 48.7 ± 17.9 | 59.9 ± 16.2 | t = 3.358 | 0.001 |
| BMI—kg/m2, median (IQR) | 19.8 (18.3–25.6) | 23.8 (20.6–29) | U = 708, Z = −2.986 | 0.003 |
| History of IE, n (%) | 6 (9.1%) | 5 (10.9%) | - | 0.758 b |
| Past or active tobacco use, n (%) | 48 (72.7%) | 19 (41.3%) | ꭓ2 = 11.136 | 0.001 a |
| Alcohol consumption, n (%) | 41 (62.1%) | 15 (32.6%) | ꭓ2 = 9.444 | 0.002 a |
| IVDU, n (%) | 36 (54.5%) | 6 (13.0%) | ꭓ2 = 19.921 | <0.001 a |
| CCI score, median (IQR) | 5 (2–7) | 6 (1–7) | U = 1521, Z = 0.021 | 0.983 |
| SOFA score, median (IQR) | 2 (1–4) | 1.5 (0–3) | U = 1876.5, Z = 2.158 | 0.031 |
| HIV infection, n (%) | 19 (28.8%) | 5 (10.9%) | ꭓ2 = 5.169 | 0.023 a |
| Hepatitis B infection, n (%) | 5 (7.6%) | 3 (6.5%) | ꭓ2 = 0.045 | 0.831 a |
| Hepatitis C infection, n (%) | 35 (53.0%) | 7 (15.2%) | ꭓ2 = 16.537 | <0.001 a |
| Staphylococcus aureus IE, n (%) | 55 (83.3%) | - | - | - |
| CoNS, n (%) | 11 (16.6%) | - | - | - |
| Staphylococcus spp. IE (n = 66) | Non-Staphylococcus spp. IE (n = 46) | Test Statistic Value | p-Value | |
|---|---|---|---|---|
| Prosthetic valve, n (%) | 11 (16.7%) | 8 (17.4%) | ꭓ2 = 0.010 | 0.920 a |
| Prior valve stenosis or insufficiency, n (%) | 18 (27.3%) | 25 (54.3%) | ꭓ2 = 8.401 | 0.004 a |
| MV, n (%) | 12 (18.2%) | 15 (32.6%) | ꭓ2 = 3.084 | 0.079 a |
| TV, n (%) | 3 (4.5%) | 3 (6.5%) | ꭓ2 = 0.209 | 0.688 a |
| AV, n (%) | 12 (18.2%) | 18 (39.1%) | ꭓ2 = 6.066 | 0.014 a |
| PV, n (%) | 1 (1.5%) | 1 (2.2%) | - | 0.067 b |
| Staphylococcus spp. IE (n = 66) | Non-Staphylococcus spp. IE (n = 46) | Test Statistic Value | p-Value | |
|---|---|---|---|---|
| Identified vegetation, n (%) | 61 (92.4%) | 46 (100.0%) | - | 0.077 b |
| TTE, n (%) | 49 (74.2%) | 34 (73.9%) | ꭓ2 = 0.002 | 0.969 a |
| TEE, n (%) | 17 (25.8%) | 15 (32.6%) | ꭓ2 = 0.634 | 0.430 a |
| Longitudinal size—mm, median (IQR) | 13.0 (8.0–18.5) | 10.0 (7.0–15.0) | U = 895, Z = 1.910 | 0.056 |
| Transverse width—mm, median (IQR) | 10.0 (5.0–14.5) | 8.0 (5.0–11.5) | U = 838, Z = 1.330 | 0.184 |
| Multiple valve involvement, n (%) | 2 (3.0%) | 2 (4.3%) | - | 1.000 b |
| IE of MV, n (%) | 23 (34.8%) | 27 (58.7%) | ꭓ2 = 6.238 | 0.013 a |
| IE of TV, n (%) | 29 (43.9%) | 8 (17.4%) | ꭓ2 = 8.636 | 0.003 a |
| IE of AV, n (%) | 16 (24.2%) | 18 (39.1%) | ꭓ2 = 2.842 | 0.092 a |
| IE of PV, n (%) | 0 (0%) | 0 (0%) | - | - |
| Prosthetic valve IE, n (%) | 11 (16.7%) | 8 (17.4%) | ꭓ2 = 0.010 | 0.920 a |
| IE affecting valves with prior stenosis or insufficiency, n (%) | 15 (22.7%) | 21 (45.7%) | ꭓ2 = 6.532 | 0.011 a |
| Staphylococcus spp. IE (n = 66) | Non-Staphylococcus spp. IE (n = 46) | Test Statistic Value | p-Value | |
|---|---|---|---|---|
| Fever, n (%) | 60 (90.9%) | 31 (67.4%) | ꭓ2 = 9.841 | 0.002 a |
| Maximum body temperature—°C, median (IQR) | 38.6 (38.0–39.2) | 39.2 (37.4–39.2) | U = 1355, Z = 1.213 | 0.225 |
| Dyspnea, n (%) | 25 (37.9%) | 11 (23.9%) | ꭓ2 = 2.424 | 0.119 a |
| Cough, n (%) | 33 (50.0%) | 10 (21.7%) | ꭓ2 = 9.153 | 0.002 a |
| Peripheral edema, n (%) | 13 (19.7%) | 10 (21.7%) | ꭓ2 = 0.069 | 0.792 a |
| Chest pain, n (%) | 7 (10.6%) | 5 (10.9%) | - | 1.000 b |
| Fatigue, n (%) | 61 (92.4%) | 44 (95.7%) | - | 0.698 b |
| Syncope, n (%) | 5 (7.6%) | 4 (8.7%) | - | 0.830 b |
| Systolic AP < 90 mmHg, n (%) | 6 (9.1%) | 4 (8.7%) | - | 1.000 b |
| RR, median (IQR) | 20 (17–25) | 18 (17–20) | U = 1557, Z = 1.621 | 0.105 |
| SaO2%, median (IQR) | 96.00 (93.00–97.25) | 98.00 (96.00–98.00) | U = 848.5, Z = −3.747 | <0.001 |
| Lung sounds, n (%) | 33 (50.0%) | 8 (17.4%) | ꭓ2 = 12.421 | <0.001 a |
| Murmurs, n (%) | 40 (60.6%) | 34 (73.9%) | ꭓ2 = 2.141 | 0.143 a |
| Lymphadenopathy, n (%) | 20 (30.3%) | 7 (15.2%) | ꭓ2 = 3.372 | 0.066 a |
| Enlarged liver, n (%) | 31 (47.0%) | 9 (19.6%) | ꭓ2 = 8.867 | 0.003 a |
| Staphylococcus spp. IE (n = 66) | Non-Staphylococcus spp. IE (n = 46) | Test Statistic Value | p-Value | |
|---|---|---|---|---|
| Leukocytes × 103/µL, median (IQR) | 12.70 (8.65–17.07) | 10.80 (7.67–13.10) | U = 1814, Z = 1.751 | 0.080 |
| CRP—mg/dL, median (IQR) | 12.85 (5.73–19.52) | 6.71 (4.17–11.07) | U = 2087.5, Z = 3.368 | 0.001 |
| Procalcitonin—ng/mL, median (IQR) | 1.50 (0.23–5.20) | 0.25 (0.12–0.90) | U = 1411.5, Z = 2.619 | 0.009 |
| Fibrinogen—mg/dL, median (IQR) | 513.50 (361.75–652.00) | 534.00 (469.50–641.50) | U = 1299.5, Z = −1.114 | 0.265 |
| Hemoglobin—g/dL, median (IQR) | 9.70 (8.77–11.02) | 10.40 (9.47–11.32) | U = 1201.5, Z = −1.873 | 0.061 |
| Thrombocytes × 103/µL, median (IQR) | 166.50 (74.76–263.65) | 260 (205.50–326.25) | U = 908, Z = −3.608 | <0.001 |
| Ultrasensitive Troponin I—ng/mL, median (IQR) | 48.40 (23.05–111.5) | 29.45 (6.07–56.17) | U = 625, Z = −1.786 | 0.074 |
| D-dimers—ng/mL, median (IQR) | 3.50 (1.82–5.31) | 1.83 (1.01–3.65) | U = 824.5, Z = 2.423 | 0.015 |
| Creatinine—mg/dL, median (IQR) | 1.00 (0.72–1.41) | 1.09 (0.76–1.39) | U = 1492, Z = −0.154 | 0.878 |
| GOT—U/L, median (IQR) | 44.00 (26.00–66.00) | 26.50 (17.25–48.75) | U = 1947.5, Z = 3.197 | 0.001 |
| LDH—U/L, median (IQR) | 310.00 (240.25–460.75) | 226.50 (185.75–279.50) | U = 1486.5, Z = 3.671 | <0.001 |
| Total bilirubin—mg/dL, median (IQR) | 0.88 (0.51–1.32) | 0.54 (0.35–0.95) | U = 1299.5, Z = 2.317 | 0.021 |
| Staphylococcus spp. IE (n = 66) | Non-Staphylococcus spp. IE (n = 46) | Test Statistic Value | p-Value | |
|---|---|---|---|---|
| Leukocytes × 103/µL, median (IQR) | 9.50 (6.72–13.07) | 7.70 (5.50–11.50) | U = 1881, Z = 2.379 | 0.017 |
| CRP—mg/dL, median (IQR) | 2.01 (0.54–8.87) | 1.75 (0.57–7.32) | U = 1567, Z = 0.493 | 0.622 |
| Procalcitonin—ng/mL, median (IQR) | 0.15 (0.05–0.52) | 0.10 (0.02–0.37) | U = 929, Z = 1.082 | 0.279 |
| Fibrinogen—mg/dL, median (IQR) | 392.00 (311.00–491.00) | 494.00 (381.50–544.25) | U = 883, Z = −2.548 | 0.011 |
| Hemoglobin—g/dL, median (IQR) | 9.80 (8.60–10.85) | 10.50 (9.20–11.95) | U = 1179.5, Z = −1.836 | 0.066 |
| Thrombocytes × 103/µL, median (IQR) | 214.50 (136.75–296.50) | 225.00 (168.00–295.50) | U = 1350, Z = −0.811 | 0.417 |
| Ultrasensitive Troponin I—ng/mL, median (IQR) | 32.50 (17.50–49.12) | 29.00 (6.20–36.70) | U = 511, Z = 1.068 | 0.286 |
| D-dimers—ng/mL, median (IQR) | 2.50 (1.55–4.05) | 1.47 (0.60–2.23) | U = 671, Z = 2.734 | 0.006 |
| Creatinine—mg/dL, median (IQR) | 1.00 (0.80–1.69) | 1.00 (0.78–1.77) | U = 1540.5, Z = 0.334 | 0.739 |
| GOT—U/L, median (IQR) | 44.00 (30.25–60.50) | 24.00 (16.75–84.00) | U = 1147.5, Z = 1.538 | 0.124 |
| LDH—U/L, median (IQR) | 308.00 (213.00–388.00) | 222.50 (166.25–298.75) | U = 961, Z = 2.866 | 0.004 |
| Total bilirubin—mg/dL, median (IQR) | 0.87 (0.7–1.1) | 0.8 (0.43–0.90) | U = 603, Z = 1.665 | 0.096 |
| Staphylococcus spp. IE (n = 66) | Non-Staphylococcus spp. IE (n = 46) | Test Statistic Value | p-Value | |
|---|---|---|---|---|
| Any embolic event, n (%) | 36 (54.5%) | 14 (30.4%) | ꭓ2 = 6.376 | 0.012 a |
| Pulmonary embolism, n (%) | 23 (34.8%) | 4 (8.7%) | - | 0.001 b |
| Hepato-splenic embolism, n (%) | 4 (6.1%) | 5 (10.9%) | - | 0.483 b |
| Cerebral/meningeal embolism, n (%) | 13 (19.7%) | 6 (13.0%) | ꭓ2 = 0.852 | 0.356 a |
| ICU admission, n (%) | 17 (25.8%) | 6 (13.0%) | ꭓ2 = 2.685 | 0.101 a |
| Cardiac surgery consult, n (%) | 14 (21.2%) | 10 (21.7%) | ꭓ2 = 0.004 | 0.947 a |
| Cardiac surgery performed, n (%) | 7 (10.6%) | 8 (17.4%) | ꭓ2 = 1.076 | 0.300 a |
| Discharged to other facility, n (%) | 5 (7.6%) | 1 (2.2%) | - | 0.398 b |
| Discharge improved, n (%) | 46 (69.7%) | 33 (71.7%) | ꭓ2 = 0.054 | 0.816 a |
| Hospital stay—days, median (IQR) | 31.5 (14.8–44.3) | 30.0 (14.8–42) | U = 1615.4, Z = 0.571 | 0.568 |
| In-hospital mortality, n (%) | 12 (18.2%) | 4 (8.7%) | ꭓ2 = 1.992 | 0.158 a |
| Odds Ratio (OR) | 95% CI Lower Limit | 95% CI Upper Limit | p-Value | |
|---|---|---|---|---|
| IVDU | 7.099 | 2.446 | 20.605 | <0.001 |
| CRP at admission | 1.077 | 1.009 | 1.148 | 0.025 |
| SaO2 (%) | 0.747 | 0.604 | 0.924 | 0.007 |
| Variable | β-Coefficient | Scaling | Point Assignment | Rationale |
|---|---|---|---|---|
| Intravenous drug use (IVDU) | 1.96 | binary | +5 points | Strongest independent predictor (highest regression coefficient) |
| C-reactive protein (CRP) | 0.74 | per 10 mg/dL increase | +2 points | Reflects inflammatory burden |
| Oxygen saturation (SaO2) | 0.29 | per 1% decrease | +1 point | Reflects pulmonary involvement (e.g., septic emboli) |
| Score Group | Score Range | Non-Staphylococcal IE | Staphylococcal IE | Observed Risk | Clinical Interpretation |
|---|---|---|---|---|---|
| Low | <7 | 37 (72.5%) | 14 (27.5%) | 28% | Staphylococcus less likely |
| Intermediate | 7–11 | 5 (17.9%) | 23 (82.1%) | 82% | strong suspicion |
| High | >11 | 2 (6.5%) | 29 (93.5%) | 94% | very high probability |
| Variable | Non-Staphylococcal IE (n = 46) | Staphylococcal IE (n = 66) | U Statistic | p-Value |
|---|---|---|---|---|
| Δ Platelet count (cells/µL), median (IQR) | −35,000 (141,000) | 43,000 (143,250) | 1037.5 | 0.007 |
| Platelet recovery, median (IQR) | −0.102 (0.562) | 0.246 (1.57) | 1000.5 | 0.004 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Nanu, A.-A.; Lazăr, M.-I.; Lazăr, D.Ș.; Popescu, C.P.; Nica, M.; Florescu, S.A. Phenotype-Based Stratification and Early Prediction of Staphylococcal Infective Endocarditis: Development of a Pragmatic Bedside Score. Pathogens 2026, 15, 418. https://doi.org/10.3390/pathogens15040418
Nanu A-A, Lazăr M-I, Lazăr DȘ, Popescu CP, Nica M, Florescu SA. Phenotype-Based Stratification and Early Prediction of Staphylococcal Infective Endocarditis: Development of a Pragmatic Bedside Score. Pathogens. 2026; 15(4):418. https://doi.org/10.3390/pathogens15040418
Chicago/Turabian StyleNanu, Adina-Alexandra, Miruna-Ioana Lazăr, Dragoș Ștefan Lazăr, Corneliu Petru Popescu, Maria Nica, and Simin Aysel Florescu. 2026. "Phenotype-Based Stratification and Early Prediction of Staphylococcal Infective Endocarditis: Development of a Pragmatic Bedside Score" Pathogens 15, no. 4: 418. https://doi.org/10.3390/pathogens15040418
APA StyleNanu, A.-A., Lazăr, M.-I., Lazăr, D. Ș., Popescu, C. P., Nica, M., & Florescu, S. A. (2026). Phenotype-Based Stratification and Early Prediction of Staphylococcal Infective Endocarditis: Development of a Pragmatic Bedside Score. Pathogens, 15(4), 418. https://doi.org/10.3390/pathogens15040418









