Time Course Characterization of Protective Immune Responses Following BCG Vaccination in BALB/c Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics and Animal Care
2.2. BCG Vaccination, M. tuberculosis Infection, and Sample Collection
2.3. ELISA Analysis
2.4. qPCR Analysis
2.5. Bacterial Culture
2.6. M. tuberculosis H37Rv Infection
2.7. CFU Enumeration
2.8. H&E Staining
2.9. IHC Staining
2.10. Statistical Analysis
3. Results
3.1. IL-2 Surge Was Observed at 8 Weeks Post Vaccination in Serum
3.2. Weekly Cytokine Shifts Elucidated Following BCG Vaccination
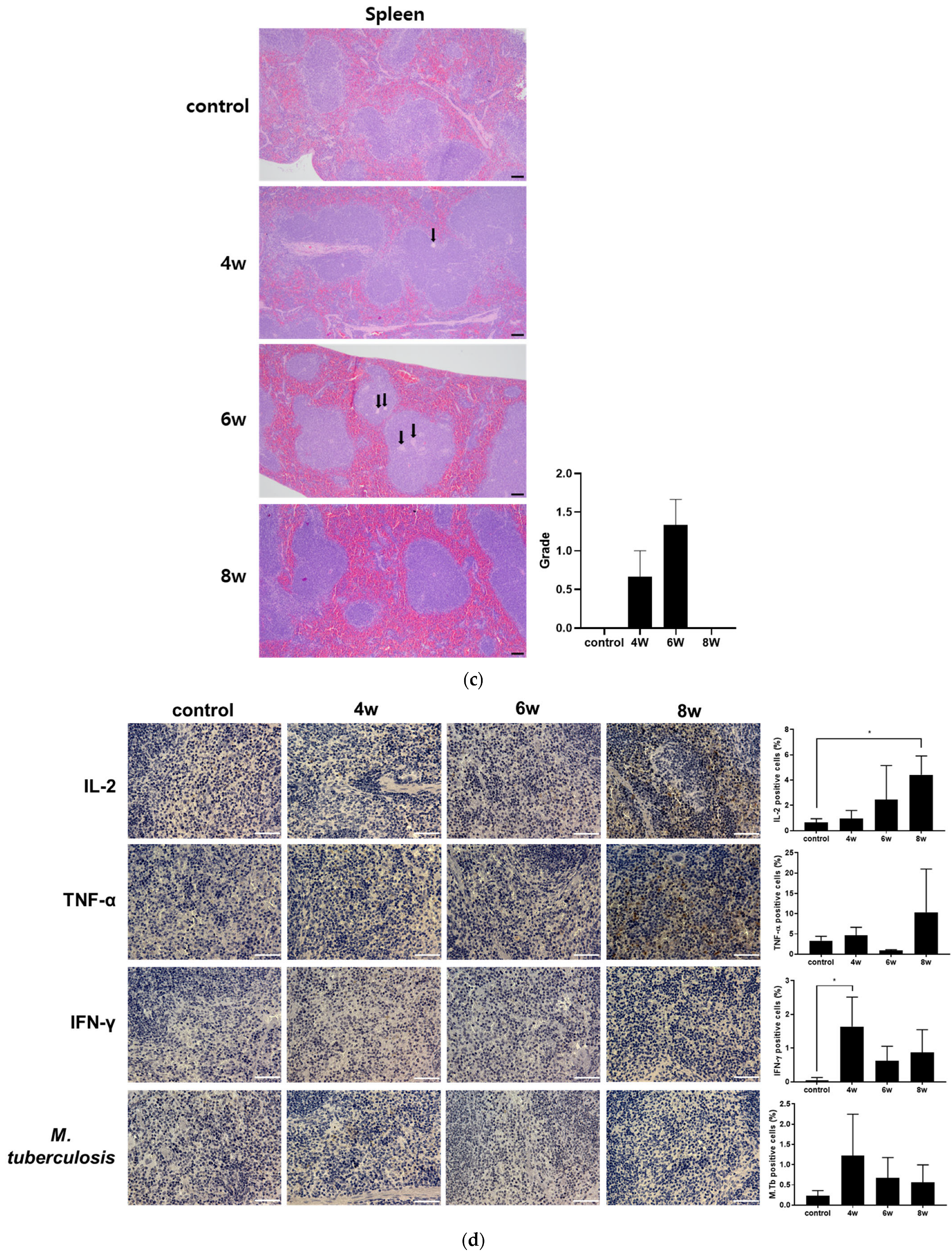
3.3. Tissue-Specific Profiles of IL-2, TNF-α and IFN-γ in the Lung and Spleen for TB Control
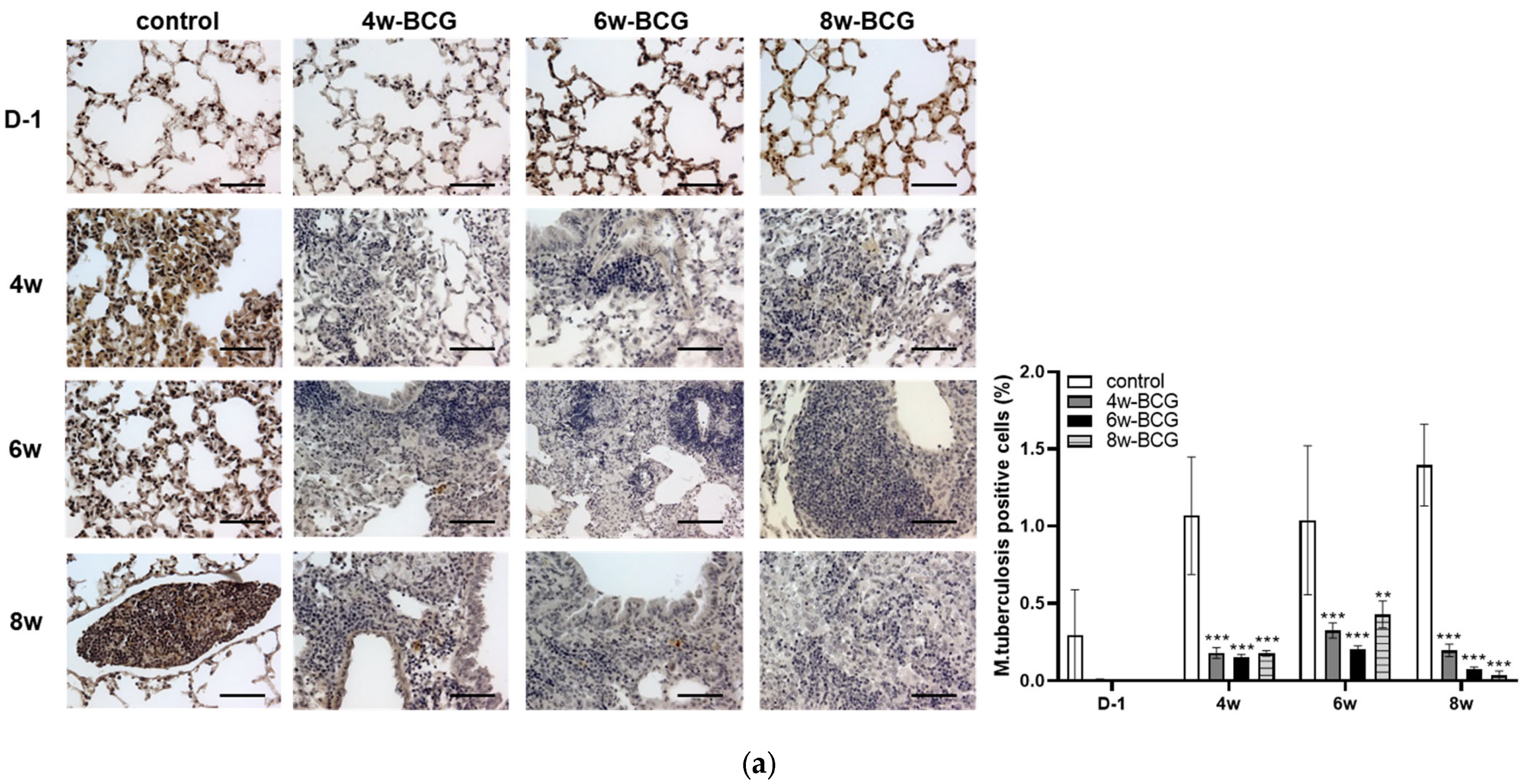
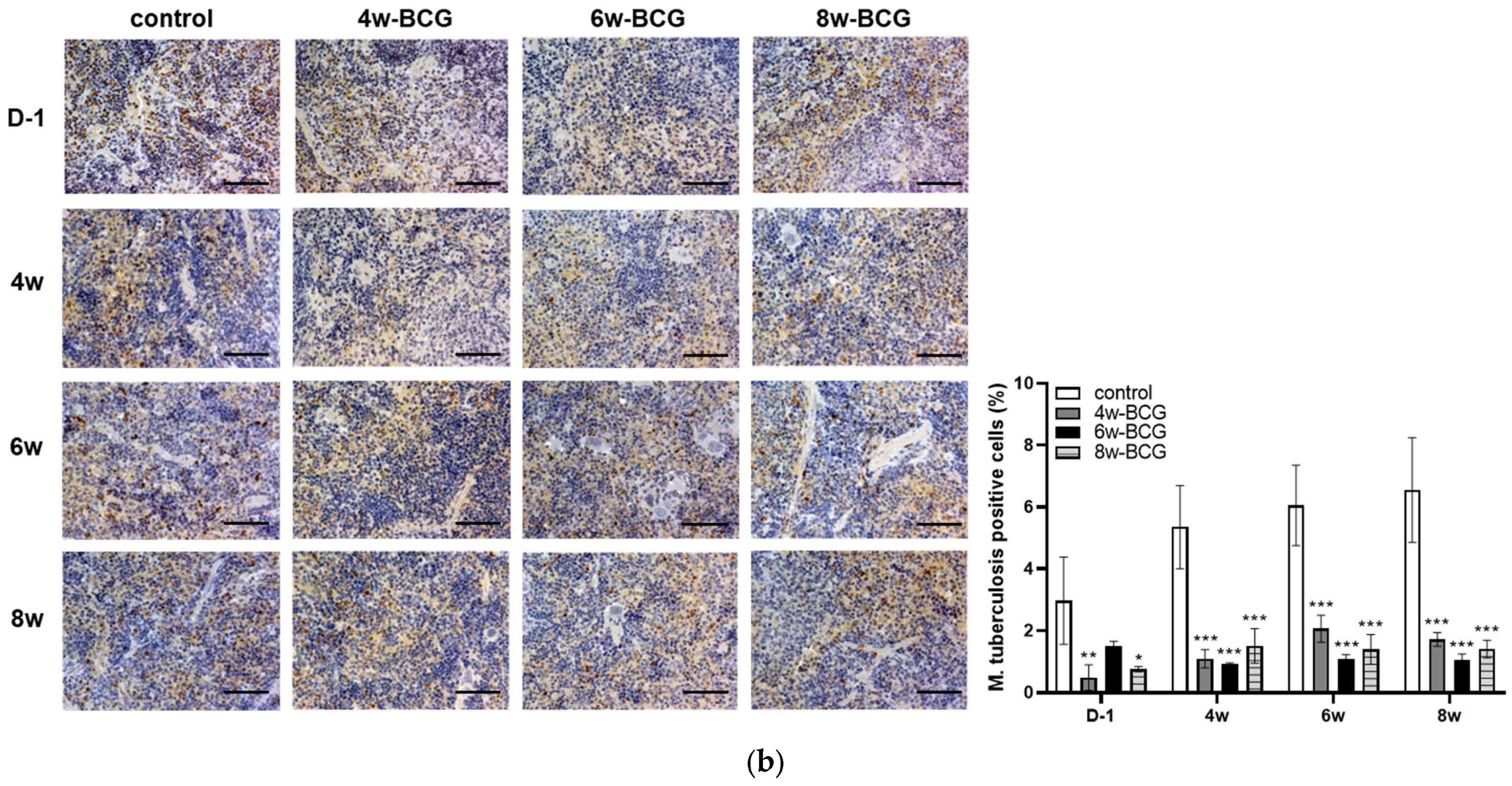
3.4. BCG Vaccination Effectively Suppressed TB Regardless of Vaccination Period
3.5. BCG Efficacy: Consistency Across 4, 6, and 8 Weeks of Immunization
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BCG | Bacillus Calmette–Guérin |
| LTBI | Latent tuberculosis infection |
| MDR-TB | Multidrug-resistant tuberculosis |
| ELISA | Enzyme-Linked Immunosorbent Assay |
| CFUs | Colony-Forming Units |
| H&E | Hematoxylin and Eosin Staining |
| IHC | Immunohistochemistry |
| PBS | Phosphate-Buffered Saline |
| OADC | Oleic acid–Albumin–Dextrose–Catalase |
| TB | tuberculosis |
References
- Mistry, D.; Patil, P.; Beniwal, S.S.; Penugonda, R.; Paila, S.; Deiveegan, D.S.; Tibrewal, C.; Ghazal, K.Y.; Anveshak; Padakanti, S.S.N.; et al. Cachexia in tuberculosis in South-East Asian and African regions: Knowledge gaps and untapped opportunities. Ann. Med. Surg. 2024, 86, 5922–5929. [Google Scholar] [CrossRef]
- Chang, E.; Cavallo, K.; Behar, S.M. CD4 T cell dysfunction is associated with bacterial recrudescence during chronic tuberculosis. Nat. Commun. 2025, 16, 2636. [Google Scholar] [CrossRef]
- Caruso, A.M.; Serbina, N.; Klein, E.; Triebold, K.; Bloom, B.R.; Flynn, J.L. Mice deficient in CD4 T cells have only transiently diminished levels of IFN-γ, yet succumb to tuberculosis. J. Immunol. 1999, 162, 5407–5416. [Google Scholar] [CrossRef]
- Chávez-Galán, L.; Illescas-Eugenio, J.; Alvarez-Sekely, M.; Baez-Saldaña, R.; Chávez, R.; Lascurain, R.J.M. Tuberculosis patients display a high proportion of CD8+ T cells with a high cytotoxic potential. Microbiol. Immunol. 2019, 63, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Subaiya, S.; Dumolard, L.; Lydon, P.; Gacic-Dobo, M.; Eggers, R.; Conklin, L. Global Routine Vaccination Coverage, 2014. Morb. Mortal. Wkly. Rep. 2015, 64, 1252–1255. [Google Scholar] [CrossRef] [PubMed]
- Xu, B.Y.; Yuan, M.J.; Yang, L.S.; Huang, L.; Li, J.X.; Tan, Z.M. Recent Advances in Clinical Research of Prophylactic Vaccines Against Tuberculosis. Vaccines 2025, 13, 959. [Google Scholar] [CrossRef] [PubMed]
- Jo, K.-W.; Yoon, Y.S.; Kim, H.W.; Kim, J.-Y.; Kang, Y.A. Diagnosis and treatment of latent tuberculosis infection in adults in South Korea. Tuberc. Respir. Dis. 2024, 88, 56. [Google Scholar] [CrossRef]
- Kweon, J.; Lee, H.; Kim, S.; Song, G. Environmental antibiotic emissions and their residual impact on livestock farming. Mol. Cell. Toxicol. 2025, 21, 351–362. [Google Scholar] [CrossRef]
- Gebremariam, G.; Kiros, M.; Hagos, S.; Hadush, H.; Gebremichael, A.; Gebrekirstos, G.; Tesfay, A.; Gebrewahid, T.; Berihu, T.; Gebremariam, B. Trend of pulmonary tuberculosis and rifampicin-resistance among tuberculosis presumptive patients in Central Tigray, Ethiopia; 2018–2023: A six-year retrospective study. Trop. Dis. Travel Med. Vaccines 2024, 10, 15. [Google Scholar] [CrossRef]
- Hoelscher, M.; Barros-Aguirre, D.; Dara, M.; Heinrich, N.; Sun, E.G.E.; Lange, C.; Tiberi, S.; Wells, C. Candidate anti-tuberculosis medicines and regimens under clinical evaluation. Clin. Microbiol. Infect. 2024, 30, 1131–1138. [Google Scholar] [CrossRef]
- Perlman, R.L. Mouse models of human disease An evolutionary perspective. Evol. Med. Public Health 2016, 2016, 170–176. [Google Scholar] [CrossRef]
- Nii, T.; Takizawa, S.; Akita, T.; Yamashita, C.; Takeuchi, I.; Makino, K. A Mouse Model for Tuberculosis Combined With Inhalable Imiquimod-PLGA Nanocomposite Particles Based on Macrophage Phenotype. In Vivo 2022, 36, 2166–2172. [Google Scholar] [CrossRef]
- Cho, Y.; Jung, B.; Jeong, S.; Moon, C.; Lee, H.J.; Ang, M.J.C.; Yun, S.P.; Son, Y.; Kim, J.S. Cognitive and emotional dysregulation in T-cell-deficient mice: The role of hippocampal neurogenesis and synaptic plasticity. Mol. Cell. Toxicol. 2025, 1–8. [Google Scholar] [CrossRef]
- Liu, X.Q.; Liu, J.P.; Wan, W.; Guo, L.H.; Zhang, J. The transcription factor ZNF92 suppresses CD8(+) T cell viability and promotes malignant progression in lung adenocarcinoma by elevating PD-L1 expression. Mol. Cell. Toxicol. 2025, 21, 929–940. [Google Scholar] [CrossRef]
- Guo, J.Z.; Jia, Z.X.; Yang, Y.R.; Wang, N.; Xue, Y.; Xiao, L.; Wang, F.H.; Wang, L.; Wang, X.O.; Liu, Y.P.; et al. Bioinformatics analysis, immunogenicity, and therapeutic efficacy evaluation of a novel multi-stage, multi-epitope DNA vaccine for tuberculosis. Int. Immunopharmacol. 2025, 152, 114415. [Google Scholar] [CrossRef] [PubMed]
- Fang, D.A.; Wang, R.H.; Fan, X.T.; Li, M.C.; Qian, C.Y.; Cao, B.; Yu, J.J.; Liu, H.C.; Lou, Y.L.; Wan, K.L. Recombinant BCG vaccine expressing multistage antigens of provides long-term immunity against tuberculosis in BALB/c mice. Hum. Vaccin. Immunother. 2024, 20, 2299607. [Google Scholar] [CrossRef]
- Rukmana, A.; Supardi, L.A.; Sjatha, F.; Nurfadilah, M. Responses of Humoral and Cellular Immune Mediators in BALB/c Mice to LipX (PE11) as Seed Tuberculosis Vaccine Candidates. Genes 2022, 13, 1954. [Google Scholar] [CrossRef]
- Yamaguchi, T.; Samukawa, N.; Matsumoto, S.; Shiota, M.; Matsumoto, M.; Nakao, R.; Hirayama, S.; Yoshida, Y.; Nishiyama, A.; Ozeki, Y.; et al. BCG-derived acellular membrane vesicles elicit antimycobacterial immunity and innate immune memory. Front. Immunol. 2025, 16, 1534615. [Google Scholar] [CrossRef]
- Sowndharya, C.K.; Mehnath, S.; Ponbharathi, A.; Jeyaraj, M. Self-Adjuvanting Adenoviral Nanovaccine for Effective T-Cell-Mediated Immunity and Long-Lasting Memory Cell Activation against Tuberculosis. ACS Infect. Dis. 2024, 10, 3939–3950. [Google Scholar] [CrossRef]
- Ouh, I.-O.; Kim, M.J.; Kim, K.; Lim, H.; Yang, Y.J.; Heo, J.W.; Choi, H.N.; Kim, H.H.; Lee, H.-J.; Koh, P.-O.J.V. Bacillus Calmette–Guérin Vaccination Promotes Efficient and Comprehensive Immune Modulation in Guinea Pig Models. Vaccines 2025, 13, 305. [Google Scholar] [CrossRef]
- Lewis, S.M.; Williams, A.; Eisenbarth, S.C. Structure and function of the immune system in the spleen. Sci. Immunol. 2019, 4, eaau6085. [Google Scholar] [CrossRef]
- Li, Y.; Yang, Z.; Ge, Q.; Zhang, Y.; Gao, M.; Liu, X.; Zhang, L. Specific Cytokines Analysis Incorporating Latency-Associated Antigens Differentiates Mycobacterium tuberculosis Infection Status: An Exploratory Study. Infect. Drug Resist. 2024, 17, 3385–3393. [Google Scholar] [CrossRef]
- Walzl, G.; Ronacher, K.; Hanekom, W.; Scriba, T.J.; Zumla, A. Immunological biomarkers of tuberculosis. Nat. Rev. Immunol. 2011, 11, 343–354. [Google Scholar] [CrossRef] [PubMed]
- O’Garra, A.; Redford, P.S.; McNab, F.W.; Bloom, C.I.; Wilkinson, R.J.; Berry, M.P. The immune response in tuberculosis. Annu. Rev. Immunol. 2013, 31, 475–527. [Google Scholar] [CrossRef] [PubMed]
- Cooper, A.M.; Khader, S.A. The role of cytokines in the initiation, expansion, and control of cellular immunity to tuberculosis. Immunol. Rev. 2008, 226, 191–204. [Google Scholar] [CrossRef] [PubMed]
- Zhai, W.; Wu, F.; Zhang, Y.; Fu, Y.; Liu, Z. The Immune Escape Mechanisms of Mycobacterium Tuberculosis. Int. J. Mol. Sci. 2019, 20, 340. [Google Scholar] [CrossRef]
- Pajuelo, D.; Gonzalez-Juarbe, N.; Tak, U.; Sun, J.; Orihuela, C.J.; Niederweis, M. NAD Depletion Triggers Macrophage Necroptosis, a Cell Death Pathway Exploited by. Cell Rep. 2018, 24, 429–440. [Google Scholar] [CrossRef]
- Flynn, J.L.; Chan, J.; Lin, P.L. Macrophages and control of granulomatous inflammation in tuberculosis. Mucosal Immunol. 2011, 4, 271–278. [Google Scholar] [CrossRef]
- Weeratunga, P.; Moller, D.R.; Ho, L.P. Immune mechanisms of granuloma formation in sarcoidosis and tuberculosis. J. Clin. Investig. 2024, 134, e175264. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, S.T.; Pan, B.W.; Guan, Z.; Yang, Z.J.; Duan, L.F.; Cai, H. A subunit vaccine based on rH-NS induces protection against infection by inducing the Th1 immune response and activating macrophages. Acta Biochim. Biophys. Sin. 2016, 48, 909–922. [Google Scholar]
- Kinjo, Y.; Kawakami, K.; Uezu, K.; Yara, S.; Miyagi, K.; Koguchi, Y.; Hoshino, T.; Okamoto, M.; Kawase, Y.; Yokota, K.; et al. Contribution of IL-18 to Th1 response and host defense against infection by Mycobacterium tuberculosis: A comparative study with IL-12p40. J. Immunol. 2002, 169, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Dong, F.; Shao, L.J.; Gong, R.; Shen, L.H.; Wang, L.M. Histone methyltransferase DOT1L promotes myeloid-derived suppressor cell activation to ameliorate colitis by modulating JAK2/STAT3 signaling. Mol. Cell. Toxicol. 2025, 21, 1333–1344. [Google Scholar] [CrossRef]
- Goenka, A.; Casulli, J.; Hussell, T. Joyrides Alveolar Macrophages into the Pulmonary Interstitium. Cell Host Microbe 2018, 24, 331–333. [Google Scholar] [CrossRef] [PubMed]
- Lovey, A.; Verma, S.; Kaipilyawar, V.; Ribeiro-Rodrigues, R.; Husain, S.; Palaci, M.; Dietze, R.; Ma, S.Y.; Morrison, R.D.; Sherman, D.R.; et al. Early alveolar macrophage response and IL-1R-dependent T cell priming determine transmissibility of strains. Nat. Commun. 2022, 13, 884. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, F.; Jin, M.J.M.; Toxicology, C. Morinda officinalis polysaccharide exerts anti-Parkinson’s disease effect via inhibiting NLRP3 inflammasome and improving pyroptosis of dopaminergic neurons. Mol. Cell. Toxicol. 2025, 21, 1307–1322. [Google Scholar] [CrossRef]
- Cerboni, S.; Gehrmann, U.; Preite, S.; Mitra, S.J.I. Cytokine-regulated Th17 plasticity in human health and diseases. Immunology 2021, 163, 3–18. [Google Scholar] [CrossRef]
- Kim, J.N.; Lee, H.; Abdul-Nasir, S.; Park, Y.S.; Kim, S.K. Immunoredox model for interpretation and prediction of human diseases. Mol. Cell. Toxicol. 2025, 21, 473–480. [Google Scholar] [CrossRef]
- Olsen, A.; Chen, Y.; Ji, Q.Z.; Zhu, G.F.; De Silva, A.D.; Vilchèze, C.; Weisbrod, T.; Li, W.M.; Xu, J.Y.; Larsen, M.; et al. Targeting Tumor Necrosis Factor Alpha-Downregulating Genes for the Development of Antituberculous Vaccines. mBio 2016, 7, e01023-15. [Google Scholar] [CrossRef]
- Allie, N.; Grivennikov, S.I.; Keeton, R.; Hsu, N.J.; Bourigault, M.L.; Court, N.; Fremond, C.; Yeremeev, V.; Shebzukhov, Y.; Ryffel, B.; et al. Prominent role for T cell-derived Tumour Necrosis Factor for sustained control of infection. Sci. Rep. 2013, 3, 1809. [Google Scholar] [CrossRef]
- O’Leary, S.; O’Sullivan, M.P.; Keane, J.J.A. IL-10 blocks phagosome maturation in Mycobacterium tuberculosis–infected human macrophages. Am. J. Respir. Cell Mol. Biol. 2011, 45, 172–180. [Google Scholar] [CrossRef]
- Boussiotis, V.A.; Tsai, E.Y.; Yunis, E.J.; Thim, S.; Delgado, J.C.; Dascher, C.C.; Berezovskaya, A.; Rousset, D.; Reynes, J.-M.; Goldfeld, A.E. IL-10–producing T cells suppress immune responses in anergic tuberculosis patients. J. Clin. Investig. 2000, 105, 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- Bonecini-Almeida, M.G.; Ho, J.L.; Boéchat, N.; Huard, R.C.; Chitale, S.; Doo, H.; Geng, J.; Rego, L.; Lazzarini, L.C.O.; Kritski, A.L.; et al. Down-modulation of lung immune responses by interleukin-10 and transforming growth factor β (TGF-β) and analysis of TGF-β receptors I and II in active tuberculosis. Infect. Immun. 2004, 72, 2628–2634. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, C.S.; Yoneda, T.; Averill, L.; Ellner, J.J.; Toossi, Z. Enhancement of intracellular growth of mycobacterium tuberculosis in human monocytes by transforming growth factor-βl. J. Infect. Dis. 1994, 170, 1229–1237. [Google Scholar] [CrossRef] [PubMed]
- Chandra, P.; Grigsby, S.J.; Philips, J.A. Immune evasion and provocation by Mycobacterium tuberculosis. Nat. Rev. Microbiol. 2022, 20, 750–766. [Google Scholar] [CrossRef]
- Dong, C. Cytokine Regulation and Function in T Cells. Annu. Rev. Immunol. 2021, 39, 51–76. [Google Scholar] [CrossRef]
- Santhi, D.; Raja, A. T cell recall response of two hypothetical proteins (Rv2251 and Rv2721c) from Mycobacterium tuberculosis in healthy household contacts of TB–Possible subunit vaccine candidates. J. Infect. 2016, 73, 455–467. [Google Scholar] [CrossRef]
- Mohan, V.P.; Scanga, C.A.; Yu, K.; Scott, H.M.; Tanaka, K.E.; Tsang, E.; Tsai, M.C.; Flynn, J.L.; Chan, J.J.I. Effects of tumor necrosis factor alpha on host immune response in chronic persistent tuberculosis: Possible role for limiting pathology. Infect. Immun. 2001, 69, 1847–1855. [Google Scholar] [CrossRef]
- Abbas, A. The Surprising Story of IL-2. Am. J. Pathol. 2020, 190, 1776–1781. [Google Scholar] [CrossRef]
- Boyman, O.; Sprent, J. The role of interleukin-2 during homeostasis and activation of the immune system. Nat. Rev. Immunol. 2012, 12, 180–190. [Google Scholar] [CrossRef]
- Ross, S.H.; Cantrell, D.A. Signaling and Function of Interleukin-2 in T Lymphocytes. Annu. Rev. Immunol. 2018, 36, 411–433. [Google Scholar] [CrossRef]





| No. | Stage | Time |
|---|---|---|
| 1 | Pre-heating | 15 min |
| 2 | Nebulizer | 30 min |
| 3 | Cloud decay | 30 min |
| 4 | UV | 15 min |
| 5 | Cool down | 10 min |
| 6 | End | 1 h 40 min |
| Score | Inflammation Characteristic |
|---|---|
| 0 | Normal histological structure |
| 1 | Mild inflammatory cell infiltration in limited areas |
| 2 | Moderate inflammatory infiltration with focal exudate |
| 3 | Marked inflammatory infiltration in extensive areas |
| 4 | Severe inflammation with diffuse infiltration and structural destruction |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Kim, H.H.; Kim, K.; Kim, M.J.; Yang, Y.J.; Choi, E.B.; Heo, J.W.; Moon, S.Y.; Lim, H.; Lee, Y.; Ouh, I.-O.; et al. Time Course Characterization of Protective Immune Responses Following BCG Vaccination in BALB/c Mice. Pathogens 2026, 15, 392. https://doi.org/10.3390/pathogens15040392
Kim HH, Kim K, Kim MJ, Yang YJ, Choi EB, Heo JW, Moon SY, Lim H, Lee Y, Ouh I-O, et al. Time Course Characterization of Protective Immune Responses Following BCG Vaccination in BALB/c Mice. Pathogens. 2026; 15(4):392. https://doi.org/10.3390/pathogens15040392
Chicago/Turabian StyleKim, Hee Ho, Kwangwook Kim, Min Jung Kim, Ye Jin Yang, Eun Bee Choi, Ji Woong Heo, Seo Young Moon, Heeji Lim, Yookyoung Lee, In-Ohk Ouh, and et al. 2026. "Time Course Characterization of Protective Immune Responses Following BCG Vaccination in BALB/c Mice" Pathogens 15, no. 4: 392. https://doi.org/10.3390/pathogens15040392
APA StyleKim, H. H., Kim, K., Kim, M. J., Yang, Y. J., Choi, E. B., Heo, J. W., Moon, S. Y., Lim, H., Lee, Y., Ouh, I.-O., & Park, K. I. (2026). Time Course Characterization of Protective Immune Responses Following BCG Vaccination in BALB/c Mice. Pathogens, 15(4), 392. https://doi.org/10.3390/pathogens15040392






