Independent Predictors of 28-Day Mortality and the Critical Role of Source Control in Stenotrophomonas maltophilia Bacteremia in the ICU
Abstract
1. Introduction
2. Methods
2.1. Design and Patients
2.2. Definitions
2.3. Microbiological Characterization
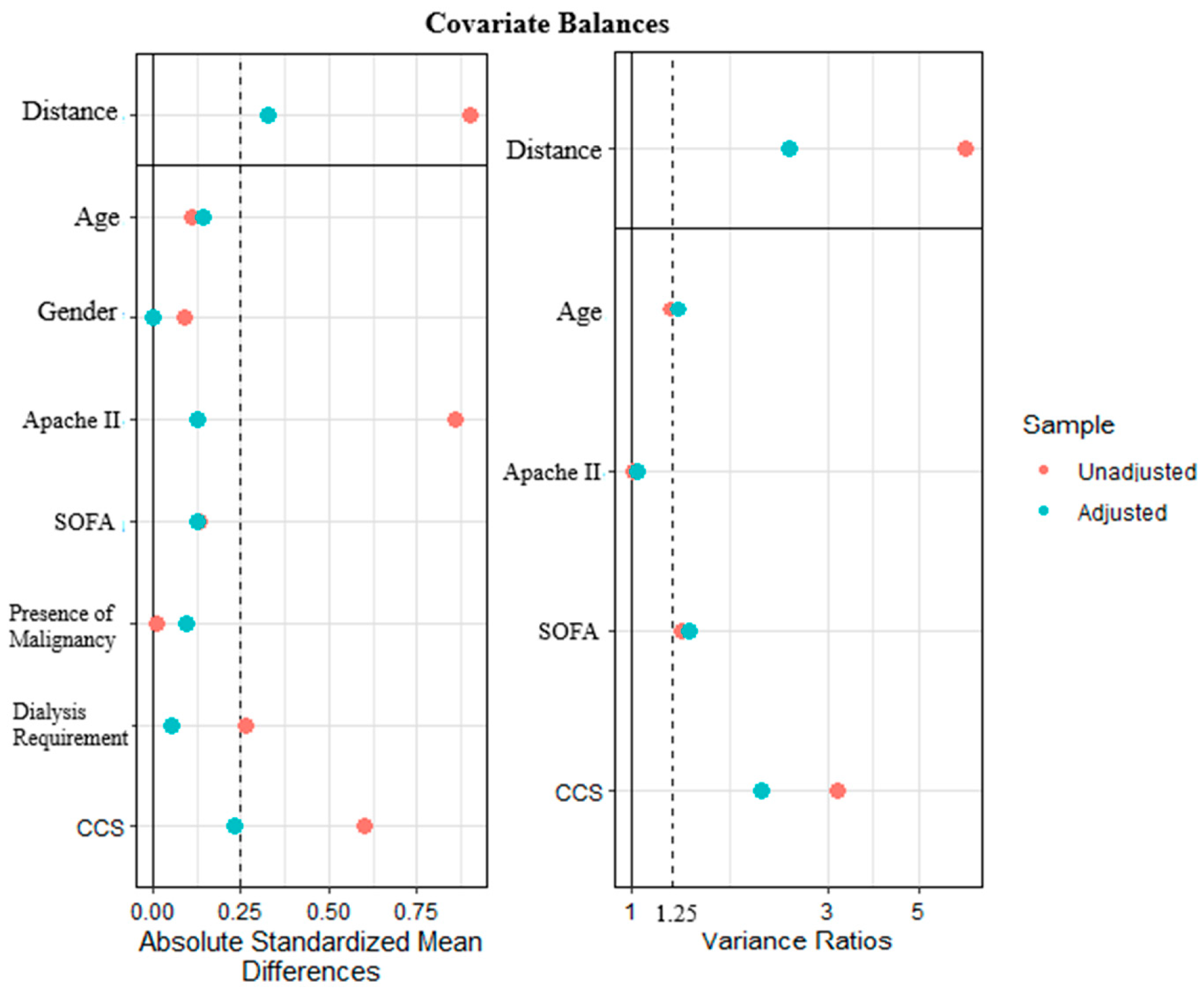
2.4. Statistics
3. Results
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Vincent, J.-L.; Rello, J.; Marshall, J.; Silva, E.; Anzueto, A.; Martin, C.D.; Moreno, R.; Lipman, J.; Gomersall, C.; Sakr, Y.; et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA 2009, 302, 2323–2329. [Google Scholar] [CrossRef] [PubMed]
- Tacconelli, E.; Carrara, E.; Savoldi, A.; Harbarth, S.; Mendelson, M.; Monnet, D.L.; Pulcini, C.; Kahlmeter, G.; Kluytmans, J.; Carmeli, Y.; et al. Discovery, research, and development of new antibiotics: The WHO priority list of antibiotic-resistant bacteria and tuberculosis. Lancet Infect. Dis. 2018, 18, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Brooke, J.S. Stenotrophomonas maltophilia: An Emerging Global Opportunistic Pathogen. Clin. Microbiol. Rev. 2012, 25, 2–41. [Google Scholar] [CrossRef] [PubMed]
- Sumida, K.; Chong, Y.; Miyake, N.; Akahoshi, T.; Yasuda, M.; Shimono, N.; Shimoda, S.; Maehara, Y.; Akashi, K. Risk Factors Associated with Stenotrophomonas maltophilia Bacteremia: A Matched Case-Control Study. PLoS ONE 2015, 10, e0133731. [Google Scholar] [CrossRef] [PubMed]
- Gajdács, M.; Matuz, M.; Ria, B.; Pető, Z.; Hajdú, E. Correlation between Stenotrophomonas maltophilia incidence and systemic antibiotic use: A 10-year retrospective, observational study in Hungary. Eur. J. Microbiol. Immunol. 2024, 14, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Carbonell, N.; Oltra, M.R.; Clari, M.Á. Stenotrophomonas maltophilia: The Landscape in Critically Ill Patients and Optimising Management Approaches. Antibiotics 2024, 13, 577. [Google Scholar] [CrossRef] [PubMed]
- Almangour, T.A.; Alali, H.A.; Alkherb, Z.; Alowais, S.A.; Bin Saleh, K.; Almuhisen, S.; Almohaizeie, A.; Alsahli, R.; Alruwaite, S.; Alnashmi, F.; et al. Monotherapy versus combination for the treatment of Stenotrophomonas maltophilia: A multicenter cohort study. Expert. Rev. Anti Infect. Ther. 2024, 22, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Gezer, Y.; Tayşi, M.R.; Tarakçı, A.; Gökçe, Ö.; Danacı, G.; Altunışık Toplu, S.; Erdal Karakaş, E.; Alkan, S.; Kuyugöz Gülbudak, S.; Şahinoğlu, M.S.; et al. Evaluation of clinical outcomes and risk factors associated with mortality in patients with Stenotrophomonas maltophilia bloodstream infection: A multicenter study. BMC Infect. Dis. 2024, 24, 1387. [Google Scholar] [CrossRef] [PubMed]
- Hafiz, T.A.; Aldawood, E.; Albloshi, A.; Alghamdi, S.S.; Mubaraki, M.A.; Alyami, A.S.; Aldriwesh, M.G. Stenotrophomonas maltophilia Epidemiology, Resistance Characteristics, and Clinical Outcomes: Understanding of the Recent Three Years’ Trends. Microorganisms 2022, 10, 2506. [Google Scholar] [CrossRef] [PubMed]
- Banar, M.; Sattari-Maraji, A.; Bayatinejad, G.; Ebrahimi, E.; Jabalameli, L.; Beigverdi, R.; Emaneini, M.; Jabalameli, F. Global prevalence and antibiotic resistance in clinical isolates of Stenotrophomonas maltophilia: A systematic review and meta-analysis. Front. Med. 2023, 10, 1163439. [Google Scholar] [CrossRef] [PubMed]
- Botan, E.; Boyraz, M.; Ulu Botan, E.; Akış, F.; Yüce, S.; Epçaçan, Z. Pseudo-Outbreaks of Stenotrophomonas maltophilia on a Pediatric Intensive Care Unit in Türkiye. J. Pediatr. Inf. 2023, 17, 28–35. [Google Scholar] [CrossRef]
- National Healthcare Safety Network. Bloodstream Infection Event (Central Line-Associated Bloodstream Infection and Non-Central Line Associated Bloodstream Infection) [Internet]. U.S. Centers for Disease Control and Prevention. 2025. Available online: https://www.cdc.gov/nhsn/pdfs/pscmanual/4psc_clabscurrent.pdf (accessed on 11 March 2026).
- Vincent, J.-L.; De Mendonca, A.; Cantraine, F.; Moreno, R.; Takala, J.; Suter, P.M.; Sprung, C.L.; Colardyn, F.; Blecher, S. Use of the SOFA score to assess the incidence of organ dysfunction/failure in intensive care units: Results of a multicenter, prospective study. Crit. Care Med. 1998, 26, 1793–1800. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.D.; Jeong, W.Y.; Kim, M.H.; Jung, I.Y.; Ahn, M.Y.; Ann, H.W.; Ahn, J.Y.; Han, S.H.; Choi, J.Y.; Song, Y.G.; et al. Risk factors for mortality in patients with Stenotrophomonas malto-philia bacteremia. Medicine 2016, 95, e4375. [Google Scholar] [CrossRef] [PubMed]
- Pompilio, A.; Ranalli, M.; Piccirilli, A.; Perilli, M.; Vukovic, D.; Savic, B.; Krutova, M.; Drevinek, P.; Jonas, D.; Fiscarelli, E.V.; et al. Biofilm Formation among Stenotrophomonas maltophilia Isolates Has Clinical Relevance: The ANSELM Prospective Multicenter Study. Microorganisms 2020, 9, 49. [Google Scholar] [CrossRef] [PubMed]
- Boktour, M.; Hanna, H.; Ansari, S.; Bahna, B.; Hachem, R.; Tarrand, J.; Rolston, K.; Safdar, A.; Raad, I. Central venous catheter and Stenotrophomonas maltophilia bacteremia in cancer patients. Cancer 2006, 106, 1967–1973. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Egi, M.; Schneider, A.G.; Bellomo, R.; Hart, G.K.; Hegarty, C. Hypophosphatemia in critically ill patients. J. Crit. Care 2013, 28, 536.e9–536.e19. [Google Scholar] [CrossRef] [PubMed]
- Kollu, K.; Yortanli, B.C.; Cicek, A.N.; Susam, E.; Karakas, N.; Kizilarslanoglu, M.C. Investigation of the Prognostic Value of Novel Laboratory Indices in Patients with Sepsis in an Intensive Care Unit: A Retrospective Observational Study. J. Clin. Med. 2025, 14, 6765. [Google Scholar] [CrossRef] [PubMed]
- Al Harbi, S.A.; Al-Dorzi, H.M.; Al Meshari, A.M.; Tamim, H.; Abdukahil, S.A.I.; Sadat, M.; Arabi, Y. Association between phosphate disturbances and mortality among critically ill patients with sepsis or septic shock. BMC Pharmacol. Toxicol. 2021, 22, 30. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Li, Y.; Zhang, C.; Guo, X.; Liang, X.; Huang, Y.; Zhang, F.; Li, J.; Liu, Q. Prognostic value of serum phosphate levels in sepsis: A systematic review and meta-analysis. PeerJ 2023, 11, e16241. [Google Scholar] [CrossRef] [PubMed]
- Sezen, A.I.; Ozdemir, Y.E.; Yeşilbağ, Z.; Borcak, D.; Canbolat Ünlü, E.; Bayrak Erdem, F.; Çizmeci, Z.; Topcu, E.; Kart Yasar, K. Seven-year evaluation of Stenotrophomonas maltophilia bacteremia in a university-affiliated hospital. J. Infect. Dev. Ctries 2025, 19, 498–503. [Google Scholar] [CrossRef] [PubMed]

| Study Group (n = 148) | Control Group (n = 148) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Median | Min | Max | IQR | Mean | Median | Min | Max | IQR | U | Z | p | |
| Age | 67.77 | 70.5 | 20 | 96 | 17 | 65.58 | 69 | 21 | 99 | 24 | 10,200.5 | −1.021 | 0.307 |
| APACHE II | 13.98 | 12 | 2 | 42 | 11 | 14.94 | 15 | 2 | 40 | 12 | 10,097.5 | −1.164 | 0.244 |
| SOFA | 7.7 | 8 | 0 | 22 | 4 | 7.27 | 6 | 1 | 25 | 3 | 9529.5 | −1.953 | 0.051 |
| CCS | 4.39 | 4 | 0 | 13 | 4 | 3.74 | 4 | 0 | 13 | 2 | 9778 | −1.614 | 0.106 |
| Dialysis duration | 27.59 | 73 | 0 | 150 | 59 | 56.55 | 62 | 0 | 130 | 81 | 100,812 | −1.379 | 0.168 |
| Catheter duration | 5.41 | 10 | 0 | 28 | 12 | 14.16 | 11 | 5 | 44 | 7 | 4805 | −8.484 | <0.001 |
| Culture negative days | 12.86 | 12 | 0 | 35 | 8 | 2.59 | 9 | 3 | 31 | 10 | 2050.5 | −12.246 | <0.001 |
| Study Group (n = 148) | Control Group (n = 148) | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Median | Min | Max | IQR | Mean | Median | Min | Max | IQR | U | Z | p | |
| Albumin 1 | 2.83 | 2.74 | 1.8 | 4.73 | 0.77 | 2.74 | 2.5 | 1.6 | 6 | 1.08 | 9316.5 | −2.222 | 0.126 |
| Protein 1 | 5.64 | 5.51 | 3.33 | 9.9 | 1.52 | 5.69 | 5.6 | 3.9 | 7.7 | 1.1 | 10,306.5 | −0.877 | 0.38 |
| ALT 1 | 45.31 | 24 | 7 | 324 | 38.25 | 101.22 | 21.5 | 7 | 452 | 26 | 9960.5 | −1.349 | 0.177 |
| AST 1 | 53 | 33 | 9 | 531 | 31.25 | 55.45 | 54.94 | 1.34 | 162.1 | 71.35 | 9671.5 | −1.739 | 0.082 |
| CRP 1 | 19.4 | 16.2 | 2.58 | 76 | 13.7 | 20.54 | 19.3 | 0.04 | 62.1 | 19.09 | 10,786 | −0.225 | 0.822 |
| Phosphorus 1 | 39.58 | 36 | 2.67 | 98 | 27.3 | 6.29 | 6 | 2 | 25 | 4 | 475 | −14.257 | <0.001 |
| Calcium 1 | 7.91 | 7.94 | 5.2 | 10 | 0.96 | 7.89 | 7.5 | 5.8 | 9.8 | 1.15 | 9555.5 | −1.899 | 0.058 |
| Creatinine 1 | 2.77 | 1.82 | 0.2 | 11 | 3.53 | 1.94 | 1.45 | 0.27 | 7.29 | 1.67 | 10,067 | −1.202 | 0.229 |
| Lymphocyte 1 | 1338.21 | 1160 | 112 | 8500 | 1081.25 | 1349.03 | 970 | 50 | 11,200 | 950 | 9974 | −1.328 | 0.184 |
| Neutrophil 1 | 6702.3 | 5405 | 680 | 32,510 | 6397.5 | 7625.74 | 6240 | 330 | 31,740 | 8237.5 | 10,220 | −0.994 | 0.32 |
| WBC 1 | 9231.93 | 8165 | 1070 | 38,450 | 6982.5 | 10,046.61 | 8890 | 500 | 38,360 | 8405 | 9943 | −1.37 | 0.171 |
| Procalcitonin 1 | 38.06 | 35.5 | 2.38 | 75 | 25.83 | 16.02 | 2.3 | 0.01 | 128 | 16.69 | 3974 | −9.482 | <0.001 |
| ESR 1 | 55.66 | 53 | 4 | 122 | 38.75 | 54.27 | 43.5 | 3 | 136 | 48.75 | 9992.5 | −1.303 | 0.192 |
| Lactate 1 | 3.61 | 3.1 | 1.2 | 12.5 | 1.73 | 3.47 | 3 | 0.75 | 13 | 1.6 | 9967.5 | −1.338 | 0.181 |
| Albumin 7 | 2.89 | 2.88 | 1.66 | 4.56 | 0.88 | 2.87 | 2.9 | 1.9 | 4 | 0.7 | 10,743 | −0.284 | 0.776 |
| Protein 7 | 5.64 | 5.64 | 3.56 | 8.5 | 1.29 | 5.56 | 5.71 | 3.32 | 9.41 | 1.13 | 10,466 | −0.66 | 0.509 |
| ALT 7 | 44.63 | 23 | 7 | 1210 | 25.75 | 47.87 | 24 | 4 | 1757 | 27 | 10,130 | −1.117 | 0.264 |
| AST 7 | 83.98 | 31 | 9 | 4596 | 39.5 | 50.13 | 37 | 9 | 704 | 33 | 9487 | −1.99 | 0.047 |
| CRP 7 | 8.58 | 5.05 | 0.2 | 178.9 | 8.23 | 6.08 | 3.7 | 0.6 | 35 | 3.93 | 9445.5 | −2.046 | 0.041 |
| Phosphorus 7 | 46.04 | 36 | 11.8 | 363 | 23 | 30.03 | 28.1 | 1.7 | 100 | 39.8 | 7292 | −4.971 | <0.001 |
| Calcium 7 | 7.98 | 7.97 | 1 | 10.9 | 1.1 | 8.07 | 8.15 | 6.2 | 9.6 | 1.4 | 10,445 | −0.689 | 0.491 |
| Creatinine 7 | 2.73 | 1.37 | 0 | 12.2 | 3.78 | 2.73 | 2.2 | 0.11 | 10.9 | 2.77 | 10,482 | −0.638 | 0.523 |
| Lymphocyte 7 | 1006.48 | 790 | 102 | 3400 | 1083.25 | 796.51 | 745 | 70 | 3300 | 390 | 10,339.5 | −0.832 | 0.405 |
| Neutrophil 7 | 6415.31 | 5560.25 | 820 | 24,290 | 4140.98 | 6605.07 | 6213 | 912 | 15,447 | 5571.75 | 10,187 | −1.039 | 0.299 |
| WBC 7 | 9387.64 | 8685 | 1380 | 25,710 | 5347.5 | 9436 | 8685 | 1380 | 25,710 | 5347 | 10,876 | −0.103 | 0.918 |
| Procalcitonin 7 | 6.47 | 2.15 | 0.04 | 75 | 6.01 | 4.8 | 0.33 | 0.03 | 75 | 0.47 | 6605 | −5.904 | <0.001 |
| ESR 7 | 42.91 | 36 | 1 | 149 | 49.5 | 42.64 | 28 | 4.2 | 161 | 43.05 | 10,461 | −0.667 | 0.505 |
| Lactate 7 | 2.84 | 1.64 | 0.18 | 13.6 | 2.19 | 2.55 | 2.15 | 0.01 | 11 | 2.05 | 9337.5 | −2.193 | 0.028 |
| Study Cases | Control Cases | Total | x2 | p | Cramer’s V | ||
|---|---|---|---|---|---|---|---|
| Presence of malignancy | No | 127 | 141 | 268 | 7.731 | 0.005 | 0.162 |
| Yes | 21 | 7 | 28 | ||||
| Dialysis requirement | No | 91 | 99 | 190 | 0.941 | 0.332 | 0.056 |
| Yes | 57 | 49 | 106 | ||||
| History of catheter infection | No | 130 | 143 | 273 | 7.967 | 0.005 | 0.164 |
| Yes | 18 | 5 | 23 | ||||
| Culture result | No growth | 0 | 113 | 113 | 296 | Not applicable * | 1 |
| S. maltophilia | 148 | 0 | 148 | ||||
| E. coli | 0 | 11 | 11 | ||||
| Enterobacter | 0 | 1 | 1 | ||||
| Klebsiella | 0 | 6 | 6 | ||||
| Pseudomonas | 0 | 2 | 2 | ||||
| S. aureus | 0 | 10 | 10 | ||||
| Other | 0 | 5 | 5 | ||||
| Source of the culture sample | None | 0 | 113 | 113 | 190.766 | <0.001 | 0.803 |
| Blood | 68 | 22 | 90 | ||||
| Catheter | 38 | 13 | 51 | ||||
| Blood and catheter | 42 | 0 | 42 | ||||
| Hypotension | No | 16 | 12 | 28 | 0.631 | 0.427 | 0.046 |
| Yes | 132 | 136 | 268 | ||||
| Shock | yok | 84 | 20 | 104 | 60.718 | <0.001 | 0.453 |
| var | 64 | 128 | 192 | ||||
| Outcome | Alive | 78 | 74 | 152 | 0.216 | 0.642 | 0.027 |
| Deceased | 70 | 74 | 144 |
| (A) | ||||||
| Median | IQR | U | Z | p | ||
| Age | Deceased | 72 | 16 | 9240.0 | −2.316 | 0.021 |
| Alive | 68 | 21.8 | ||||
| APACHE II | Deceased | 12 | 12 | 10,310.0 | −0.864 | 0.388 |
| Alive | 12 | 11.8 | ||||
| SOFA | Deceased | 8 | 5 | 6560.5 | −6.019 | <0.001 |
| Alive | 6 | 4 | ||||
| CCS | Deceased | 4 | 2 | 10,068.0 | −1.205 | 0.228 |
| Alive | 4 | 3 | ||||
| Dialysis duration | Deceased | 0 | 25.8 | 10,435.0 | −0.806 | 0.420 |
| Alive | 0 | 48 | ||||
| Catheter duration | Deceased | 10 | 14 | 10,785.5 | −0.219 | 0.827 |
| Alive | 10 | 14.8 | ||||
| Culture-negative days | Deceased | 8 | 14 | 10,262.5 | −0.956 | 0.339 |
| Alive | 6 | 12 | ||||
| (B) | ||||||
| Median | IQR | U | Z | p | ||
| Albumin 1 | Deceased | 2.7 | 0.7 | 10,231.0 | −0.969 | 0.333 |
| Alive | 2.6 | 1.02 | ||||
| Protein 1 | Deceased | 5.58 | 1.2 | 10,331.0 | −0.833 | 0.405 |
| Alive | 5.6 | 1.3 | ||||
| ALT 1 | Deceased | 22 | 26 | 10,369.0 | −0.783 | 0.434 |
| Alive | 23 | 32.3 | ||||
| AST 1 | Deceased | 41 | 61.3 | 10,390.5 | −0.752 | 0.452 |
| Alive | 38 | 56.7 | ||||
| Phosphorus 1 | Deceased | 23.3 | 34.5 | 9504 | −0.196 | 0.5 |
| Alive | 13.5 | 30 | ||||
| Calcium 1 | Deceased | 7.7 | 1.22 | 10,341.0 | −0.820 | 0.412 |
| Alive | 7.6 | 0.81 | ||||
| Creatinine 1 | Deceased | 1.48 | 2.8 | 10,892.0 | −0.071 | 0.944 |
| Alive | 1.63 | 2.19 | ||||
| Lactate 1 | Deceased | 3.3 | 2 | 8479.0 | −3.350 | 0.001 |
| Alive | 2.8 | 1.48 | ||||
| Lymphocyte 1 | Deceased | 980 | 850 | 10,449.0 | −0.673 | 0.501 |
| Alive | 1110 | 1234 | ||||
| Neutrophil 1 | Deceased | 6030 | 8423 | 10,498.5 | −0.605 | 0.545 |
| Alive | 5705 | 6153 | ||||
| WBC 1 | Deceased | 8370 | 9070 | 10,743.5 | −0.272 | 0.785 |
| Alive | 8530 | 6583 | ||||
| CRP 1 | Deceased | 17 | 15.9 | 10,817.5 | −0.172 | 0.864 |
| Alive | 16.6 | 15.6 | ||||
| Procalcitonin 1 | Deceased | 26 | 49.3 | 9401.5 | −2.097 | 0.036 |
| Alive | 19 | 37.9 | ||||
| ESR 1 | Deceased | 52 | 46 | 10,407.5 | −0.729 | 0.466 |
| Alive | 47 | 41.8 | ||||
| Albumin 7 | Deceased | 2.84 | 0.73 | 9955.5 | −1.343 | 0.179 |
| Alive | 2.98 | 0.82 | ||||
| Protein 7 | Deceased | 5.7 | 1.1 | 9920.0 | −1.392 | 0.164 |
| Alive | 5.71 | 1.26 | ||||
| ALT 7 | Deceased | 24 | 25.8 | 10,623.5 | −0.436 | 0.663 |
| Alive | 22 | 28 | ||||
| AST 7 | Deceased | 35 | 38 | 10,623.0 | −0.436 | 0.663 |
| Alive | 32 | 33.5 | ||||
| Phosphorus 7 | Deceased | 33.1 | 31.4 | 10,821.5 | −0.166 | 0.868 |
| Alive | 34 | 22.1 | ||||
| Calcium 7 | Deceased | 8.1 | 1.14 | 10,662.0 | −0.383 | 0.702 |
| Alive | 8.1 | 1.26 | ||||
| Creatinine 7 | Deceased | 2.47 | 3.8 | 8685.5 | −3.069 | 0.002 |
| Alive | 1.36 | 2.62 | ||||
| Lactate 7 | Deceased | 1.99 | 2.59 | 9389.0 | −2.113 | 0.035 |
| Alive | 1.78 | 2 | ||||
| Lymphocyte 7 | Deceased | 782 | 494 | 10,648.0 | −0.402 | 0.688 |
| Alive | 740 | 585 | ||||
| Neutrophil 7 | Deceased | 5543 | 5301 | 10,258.5 | −0.931 | 0.352 |
| Alive | 5995 | 4886 | ||||
| WBC 7 | Deceased | 8628 | 6450 | 10,363.0 | −0.789 | 0.430 |
| Alive | 8469 | 5935 | ||||
| CRP 7 | Deceased | 4.1 | 8.47 | 9809.5 | −1.541 | 0.123 |
| Alive | 4 | 5.46 | ||||
| Procalcitonin 7 | Deceased | 0.935 | 5.58 | 10,477.5 | −0.634 | 0.526 |
| Alive | 0.475 | 3.81 | ||||
| ESR 7 | Deceased | 32.6 | 45.1 | 9763.0 | −1.605 | 0.109 |
| Alive | 26.6 | 48.5 | ||||
| Age | SOFA | Presence of Malignancy | Shock | Culture-Negative Days | Catheter Duration | Phosphorus 1 | Phosphorus 7 | Lactate 1 | Lactate 7 | CCS | Outcome | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Age | rs | 1.000 | |||||||||||
| p | |||||||||||||
| SOFA | rs | 0.154 ** | 1.000 | ||||||||||
| p | 0.008 | ||||||||||||
| Presence of malignancy | rs | 0.023 | 0.065 | 1.000 | |||||||||
| p | 0.700 | 0.262 | |||||||||||
| Shock | rs | 0.075 | 0.098 | −0.004 | 1.000 | ||||||||
| p | 0.195 | 0.092 | 0.946 | ||||||||||
| Culture-negative days | rs | 0.073 | 0.129 * | 0.117 * | −0.211 ** | 1.000 | |||||||
| p | 0.213 | 0.026 | 0.044 | <0.001 | |||||||||
| Catheter duration | rs | 0.103 | −0.045 | −0.104 | 0.375 ** | −0.315 ** | 1.000 | ||||||
| p | 0.077 | 0.439 | 0.075 | <0.001 | <0.001 | ||||||||
| Phosphorus 1 | rs | 0.048 | 0.142 * | 0.177 ** | −0.335 ** | 0.554 ** | −0.334 ** | 1.000 | |||||
| p | 0.412 | 0.014 | 0.002 | <0.001 | <0.001 | <0.001 | |||||||
| Phosphorus 7 | rs | 0.083 | −0.018 | 0.057 | 0.015 | 0.283 ** | 0.013 | 0.317 ** | 1.000 | ||||
| p | 0.156 | 0.762 | 0.328 | 0.794 | <0.001 | 0.830 | <0.001 | ||||||
| Lactate1 | rs | 0.131 * | 0.166 ** | 0.161 ** | 0.035 | 0.017 | −0.068 | 0.098 | −0.069 | 1.000 | |||
| p | 0.024 | 0.004 | 0.006 | 0.552 | 0.766 | 0.245 | 0.094 | 0.238 | |||||
| Lactate 7 | rs | −0.014 | 0.132 * | 0.068 | 0.205 ** | −0.030 | 0.119 * | −0.027 | −0.206 ** | 0.124 * | 1.000 | ||
| p | 0.805 | 0.023 | 0.240 | 0.000 | 0.606 | 0.041 | 0.645 | <0.001 | 0.034 | ||||
| CCS | rs | 0.101 | 0.134 * | 0.343 ** | −0.016 | 0.030 | 0.144 * | 0.117 * | 0.068 | 0.087 | 0.055 | 1.000 | |
| p | 0.081 | 0.021 | <0.001 | 0.778 | 0.610 | 0.013 | 0.044 | 0.244 | 0.137 | 0.348 | |||
| Outcome | rs | 0.135 * | 0.350 ** | 0.194 ** | 0.334 ** | 0.056 | −0.013 | 0.114 * | 0.010 | 0.195 ** | 0.123 * | 0.070 | 1.000 |
| p | 0.020 | <0.001 | 0.001 | <0.001 | 0.340 | 0.827 | 0.050 | 0.868 | 0.001 | 0.034 | 0.229 |
| B | S.E. | Wald | df | Sig. | Exp(B) | 95% C.I. for EXP(B) | ||
|---|---|---|---|---|---|---|---|---|
| Lower | Upper | |||||||
| Age | 0.010 | 0.009 | 1.215 | 1 | 0.270 | 1.010 | 0.993 | 1.027 |
| Shock (1) | 1.794 | 0.333 | 28.944 | 1 | <0.001 | 6.014 | 3.128 | 11.561 |
| Presence of malignancy (1) | 1.462 | 0.561 | 6.784 | 1 | 0.009 | 4.314 | 1.436 | 12.961 |
| SOFA | 0.244 | 0.051 | 23.112 | 1 | <0.001 | 1.276 | 1.155 | 1.409 |
| Procalcitonin 1 | 0.014 | 0.006 | 5.503 | 1 | 0.019 | 1.014 | 1.002 | 1.026 |
| Phosphorus 1 | 0.013 | 0.007 | 3.212 | 1 | 0.073 | 1.013 | 0.999 | 1.028 |
| Lactate1 | 0.130 | 0.075 | 2.963 | 1 | 0.085 | 1.139 | 0.982 | 1.320 |
| Constant | −4.617 | 0.763 | 36.609 | 1 | <0.001 | 0.010 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Uğuz, M.; Çirkin Doruk, B. Independent Predictors of 28-Day Mortality and the Critical Role of Source Control in Stenotrophomonas maltophilia Bacteremia in the ICU. Pathogens 2026, 15, 364. https://doi.org/10.3390/pathogens15040364
Uğuz M, Çirkin Doruk B. Independent Predictors of 28-Day Mortality and the Critical Role of Source Control in Stenotrophomonas maltophilia Bacteremia in the ICU. Pathogens. 2026; 15(4):364. https://doi.org/10.3390/pathogens15040364
Chicago/Turabian StyleUğuz, Mustafa, and Berfin Çirkin Doruk. 2026. "Independent Predictors of 28-Day Mortality and the Critical Role of Source Control in Stenotrophomonas maltophilia Bacteremia in the ICU" Pathogens 15, no. 4: 364. https://doi.org/10.3390/pathogens15040364
APA StyleUğuz, M., & Çirkin Doruk, B. (2026). Independent Predictors of 28-Day Mortality and the Critical Role of Source Control in Stenotrophomonas maltophilia Bacteremia in the ICU. Pathogens, 15(4), 364. https://doi.org/10.3390/pathogens15040364





