Factors Governing the Cross-Species Virulence of Shiga Toxin-Producing Escherichia coli
Abstract
1. Introduction
2. Materials and Methods
2.1. Strains
2.2. Cultivation and Harvesting of A. castellanii
2.3. Amoeba Predation Assay
2.4. Amoeba Killing Assay
2.5. C. elegans Survival Assay
3. Results
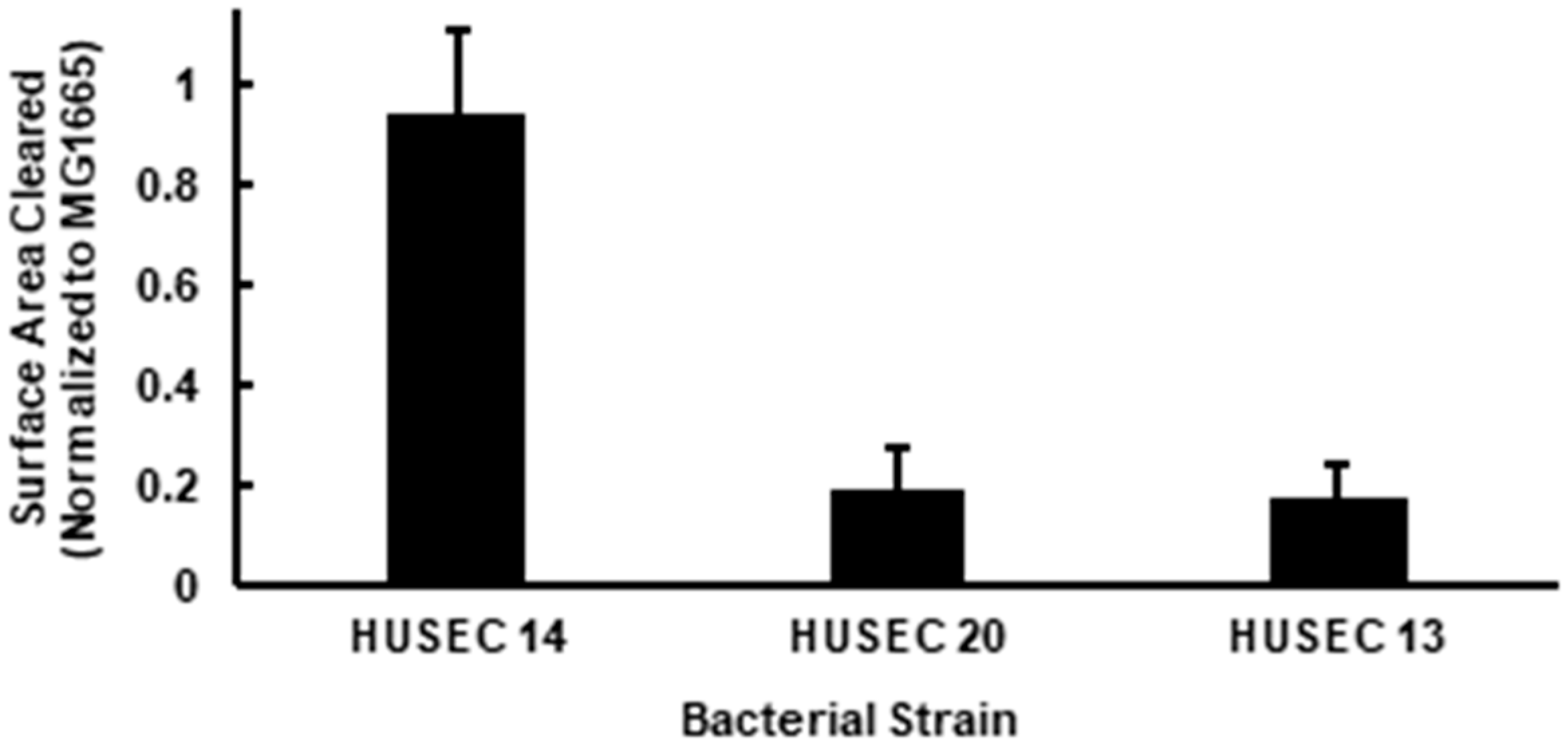
3.1. Effect of Stx Type on A. castellanii Predation
3.2. Effect of O-antigen Type on A. castellanii Predation
3.3. A. castellanii Killing in Liquid Culture
3.4. STEC Killing of C. elegans
4. Discussion
4.1. Shiga Toxin as a Dual-Use Ecological Weapon
4.2. Titer-Fitness Gradient and the Molecular Determinants of High Titer
4.3. Impact of Toxin Type on Predation Resistance
4.4. Role of the Bacterial Cell Surface in Predation Resistance
4.5. STEC Fitness and Virulence
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HUS | Hemolytic uremic syndrome |
| Stx | Shiga toxin |
| STEC | Shiga toxin-producing E. coli |
References
- Majowicz, S.E.; Scallan, E.; Jones-Bitton, A.; Sargeant, J.M.; Stapleton, J.; Angulo, F.J.; Yeung, D.H.; Kirk, M.D. Global Incidence of Human Shiga Toxin–Producing Escherichia coli Infections and Deaths: A Systematic Review and Knowledge Synthesis. Foodborne Pathog. Dis. 2014, 11, 447–455. [Google Scholar] [CrossRef] [PubMed]
- Karch, H.; Tarr, P.I.; Bielaszewska, M. Enterohaemorrhagic Escherichia coli in human medicine. Int. J. Med. Microbiol. 2005, 295, 405–418. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Yu, D.; Chui, L.; Zhou, T.; Feng, Y.; Cao, Y.; Zhi, S. A Comprehensive Review on Shiga Toxin Subtypes and Their Niche-Related Distribution Characteristics in Shiga-Toxin-Producing E. coli and Other Bacterial Hosts. Microorganisms 2024, 12, 687. [Google Scholar] [PubMed]
- WHO. Shiga Toxin-Producing Escherichia coli (STEC) and Food: Attribution, Characterization, and Monitoring: Report; FAO, WHO, Eds.; FAO: Rome, Italy, 2018; p. 174. [Google Scholar]
- Shoaib, M.; Gul, S.; Majeed, S.; He, Z.; Hao, B.; Tang, M.; Zhang, X.; Wu, Z.; Wang, S.; Pu, W. Pathogenomic Characterization of Multidrug-Resistant Escherichia coli Strains Carrying Wide Efflux-Associated and Virulence Genes from the Dairy Farm Environment in Xinjiang, China. Antibiotics 2025, 14, 511. [Google Scholar] [CrossRef]
- Mauro, S.A.; Koudelka, G.B. Shiga Toxin: Expression, Distribution, and Its Role in the Environment. Toxins 2011, 3, 608–625. [Google Scholar] [CrossRef]
- Rahman, A.; Rahman Chowdhury, M.S.; Hossain, H.; Elsaid, F.; Almutairi, L.; Begum, R.; Sabrin, M.S.; Akanda, M.; Hossain, M.M.; Islam, M.R.; et al. Identification of Virulence Genes and Multidrug Resistance in Shiga-Toxin Producing Escherichia coli (STEC) from Migratory and Captive Wild Birds. Pak. Vet. J. 2024, 44, 1120–1130. [Google Scholar] [CrossRef]
- Fadel, H.M.; Afifi, R.; Al-Qabili, D.M. Characterization and zoonotic impact of Shiga toxin producing Escherichia coli in some wild bird species. Vet. World 2017, 10, 1118–1128. [Google Scholar] [CrossRef]
- Hughes, J.M.; Wilson, M.E.; Johnson, K.E.; Thorpe, C.M.; Sears, C.L. The Emerging Clinical Importance of Non-O157 Shiga Toxin—Producing Escherichia coli. Clin. Infect. Dis. 2006, 43, 1587–1595. [Google Scholar] [CrossRef]
- Luna-Gierke, R.E.; Griffin, P.M.; Gould, L.H.; Herman, K.; Bopp, C.A.; Strockbine, N.; Mody, R.K. Outbreaks of non-O157 Shiga toxin-producing Escherichia coli infection: USA. Epidemiol. Infect. 2014, 142, 2270–2280. [Google Scholar] [CrossRef]
- Eichhorn, I.; Heidemanns, K.; Semmler, T.; Kinnemann, B.; Mellmann, A.; Harmsen, D.; Anjum, M.F.; Schmidt, H.; Fruth, A.; Valentin-Weigand, P.; et al. Highly Virulent Non-O157 Enterohemorrhagic Escherichia coli (EHEC) Serotypes Reflect Similar Phylogenetic Lineages, Providing New Insights into the Evolution of EHEC. Appl. Environ. Microbiol. 2015, 81, 7041–7047. [Google Scholar] [CrossRef]
- Werber, D.; Beutin, L.; Pichner, R.; Stark, K.; Fruth, A. Shiga toxin-producing Escherichia coli serogroups in food and patients, Germany. Emerg. Infect. Dis. 2008, 14, 1803–1806. [Google Scholar] [CrossRef]
- Whittam, T.S.; Wolfe, M.L.; Wachsmuth, I.K.; Orskov, F.; Orskov, I.; Wilson, R.A. Clonal relationships among Escherichia coli strains that cause hemorrhagic colitis and infantile diarrhea. Infect. Immun. 1993, 61, 1619–1629. [Google Scholar] [CrossRef] [PubMed]
- Roldgaard, B.B.; Scheutz, F.; Boel, J.; Aabo, S.; Schultz, A.C.; Cheasty, T.; Nielsen, E.M.; Olsen, K.E.; Christensen, B.B. VTEC O157 subtypes associated with the most severe clinical symptoms in humans constitute a minor part of VTEC O157 isolates from Danish cattle. Int. J. Med. Microbiol. 2004, 294, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Vanaja, S.K.; Springman, A.C.; Besser, T.E.; Whittam, T.S.; Manning, S.D. Differential expression of virulence and stress fitness genes between Escherichia coli O157:H7 strains with clinical or bovine-biased genotypes. Appl. Environ. Microbiol. 2010, 76, 60–68. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Coombes, B.K.; Gilmour, M.W.; Goodman, C.D. The evolution of virulence in non-o157 shiga toxin-producing Escherichia coli. Front. Microbiol. 2011, 2, 90. [Google Scholar] [CrossRef]
- Gould, L.H.; Demma, L.; Jones, T.F.; Hurd, S.; Vugia, D.J.; Smith, K.; Shiferaw, B.; Segler, S.; Palmer, A.; Zansky, S.; et al. Hemolytic uremic syndrome and death in persons with Escherichia coli O157:H7 infection, foodborne diseases active surveillance network sites, 2000-2006. Clin. Infect. Dis. 2009, 49, 1480–1485. [Google Scholar] [CrossRef]
- Karmali, M.A.; Mascarenhas, M.; Petric, M.; Dutil, L.; Rahn, K.; Ludwig, K.; Arbus, G.S.; Michel, P.; Sherman, P.M.; Wilson, J.; et al. Age-specific frequencies of antibodies to Escherichia coli verocytotoxins (Shiga toxins) 1 and 2 among urban and rural populations in southern Ontario. J. Infect. Dis. 2003, 188, 1724–1729. [Google Scholar] [CrossRef]
- Karmali, M.A. Emerging Public Health Challenges of Shiga Toxin-Producing Escherichia coli Related to Changes in the Pathogen, the Population, and the Environment. Clin. Infect. Dis. 2017, 64, 371–376. [Google Scholar] [CrossRef]
- Mody, R.K.; Luna-Gierke, R.E.; Jones, T.F.; Comstock, N.; Hurd, S.; Scheftel, J. Infections in pediatric postdiarrheal hemolytic uremic syndrome: Factors associated with identifying shiga toxin–producing Escherichia coli. Arch. Pediatr. Adolesc. Med. 2012, 166, 902–909. [Google Scholar] [CrossRef][Green Version]
- Endo, Y.; Tsurugi, K.; Yutsudo, T.; Takeda, Y.; Ogasawara, T.; Igarashi, K. Site of action of a Vero toxin (VT2) from Escherichia coli O157:H7 and of Shiga toxin on eukaryotic ribosomes. Eur. J. Biochem. 1988, 171, 45–50. [Google Scholar] [CrossRef]
- Igarashi, K.; Ogasawara, T.; Ito, K.; Yutsudo, T.; Takeda, Y. Inhibition of elongation factor 1-dependent aminoacyl-tRNA binding to ribosomes by Shiga-like toxin I (VT1) from Escherichia coli O157:H7 and by Shiga toxin. FEMS Microbiol. Lett. 1987, 44, 91–94. [Google Scholar] [CrossRef]
- Ogasawara, T.; Ito, K.; Igarashi, K.; Yutsudo, T.; Nakabayashi, N.; Takeda, Y. Inhibition of protein synthesis by a Vero toxin (VT2 or Shiga-like toxin II) produced by Escherichia coli O157:H7 at the level of elongation factor 1-dependent aminoacyl-tRNA binding to ribosomes. Microb. Pathog. 1988, 4, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Obrig, T.G.; Moran, T.P.; Brown, J.E. The mode of action of Shiga toxin on peptide elongation of eukaryotic protein synthesis. Biochem. J. 1987, 244, 287–294. [Google Scholar] [CrossRef] [PubMed]
- Unkmeir, A.; Schmidt, H. Structural analysis of phage-borne stx genes and their flanking sequences in shiga toxin-producing Escherichia coli and Shigella dysenteriae type 1 strains. Infect. Immun. 2000, 68, 4856–4864. [Google Scholar] [CrossRef]
- Zhou, Z.; Li, X.; Liu, B.; Beutin, L.; Xu, J.; Ren, Y.; Feng, L.; Lan, R.; Reeves, P.R.; Wang, L. Derivation of Escherichia coli O157:H7 from Its O55:H7 Precursor. PLoS ONE 2010, 5, e8700. [Google Scholar] [CrossRef]
- Feng, P.; Lampel, K.A.; Karch, H.; Whittam, T.S. Genotypic and Phenotypic Changes in the Emergence of Escherichia coli O157:H7. J. Infect. Dis. 1998, 177, 1750–1753. [Google Scholar] [CrossRef]
- Manning, S.D.; Motiwala, A.S.; Springman, A.C.; Qi, W.; Lacher, D.W.; Ouellette, L.M.; Mladonicky, J.M.; Somsel, P.; Rudrik, J.T.; Dietrich, S.E.; et al. Variation in virulence among clades of Escherichia coli O157:H7 associated with disease outbreaks. Proc. Natl. Acad. Sci. USA 2008, 105, 4868–4873. [Google Scholar] [CrossRef]
- Lacher, D.W. The Evolutionary Model of E. coli O157:H7. In Population Genetics of Bacteria: A Tribute to Thomas S. Whittam; Walk, S.T., Feng, P.C.H., Whittam, T.S., Eds.; ASM Press: Washington, DC, USA, 2011; pp. 227–239. [Google Scholar]
- Melton-Celsa, A.R. Shiga Toxin (Stx) Classification, Structure, and Function. Microbiol. Spectrum. 2014, 2. [Google Scholar] [CrossRef]
- Scheutz, F.; Teel, L.D.; Beutin, L.; Piérard, D.; Buvens, G.; Karch, H.; Mellmann, A.; Caprioli, A.; Tozzoli, R.; Morabito, S.; et al. Multicenter Evaluation of a Sequence-Based Protocol for Subtyping Shiga Toxins and Standardizing Stx Nomenclature. J. Clin. Microbiol. 2012, 50, 2951–2963. [Google Scholar] [CrossRef]
- Harada, T.; Iguchi, A.; Iyoda, S.; Seto, K.; Taguchi, M.; Kumeda, Y. Multiplex Real-Time PCR Assays for Screening of Shiga Toxin 1 and 2 Genes, Including All Known Subtypes, and Escherichia coli O26-, O111-, and O157-Specific Genes in Beef and Sprout Enrichment Cultures. J. Food Prot. 2015, 78, 1800–1811. [Google Scholar] [CrossRef]
- De Rauw, K.; Jacobs, S.; Piérard, D. Twenty-seven years of screening for Shiga toxin-producing Escherichia coli in a university hospital. Brussels, Belgium, 1987-2014. PLoS ONE 2018, 13, e0199968. [Google Scholar] [CrossRef]
- Smith, D.L.; Rooks, D.J.; Fogg, P.C.; Darby, A.C.; Thomson, N.R.; McCarthy, A.J.; Allison, H.E. Comparative genomics of Shiga toxin encoding bacteriophages. BMC Genom. 2012, 13, 311. [Google Scholar] [CrossRef] [PubMed]
- Yin, S.; Rusconi, B.; Sanjar, F.; Goswami, K.; Xiaoli, L.; Eppinger, M.; Dudley, E. Escherichia coli O157:H7 strains harbor at least three distinct sequence types of Shiga toxin 2a-converting phages. BMC Genom. 2015, 16, 733. [Google Scholar] [CrossRef]
- Ptashne, M. A Genetic Switch; Blackwell PRESS: Palo Alto, CA, USA, 1986. [Google Scholar]
- Wegrzyn, G.; Wegrzyn, A. Genetic switches during bacteriophage lambda development. Prog. Nucleic Acid. Res. Mol. Biol. 2005, 79, 1–48. [Google Scholar] [CrossRef] [PubMed]
- Casjens, S.R.; Hendrix, R.W. Bacteriophage lambda: Early pioneer and still relevant. Virology 2015, 479–480, 310–330. [Google Scholar] [CrossRef] [PubMed]
- Casas, V.; Sobrepena, G.; Rodriguez-Mueller, B.; AhTye, J.; Maloy, S. Bacteriophage-encoded shiga toxin gene in atypical bacterial host. Gut Pathog. 2011, 3, 10. [Google Scholar] [CrossRef]
- Casas, V.; Miyake, J.; Balsley, H.; Roark, J.; Telles, S.; Leeds, S.; Zurita, I.; Breitbart, M.; Bartlett, D.; Azam, F.; et al. Widespread occurrence of phage-encoded exotoxin genes in terrestrial and aquatic environments in Southern California. FEMS Microbiol. Lett. 2006, 261, 141–149. [Google Scholar] [CrossRef]
- Weinbauer, M.G. Ecology of prokaryotic viruses. FEMS Microbiol. Rev. 2004, 28, 127–181. [Google Scholar] [CrossRef]
- Brown, S.P.; Fredrik Inglis, R.; Taddei, F. SYNTHESIS: Evolutionary ecology of microbial wars: Within-host competition and (incidental) virulence. Evol. Appl. 2009, 2, 32–39. [Google Scholar] [CrossRef]
- Dimitriu, T.; Lotton, C.; Bénard-Capelle, J.; Misevic, D.; Brown, S.P.; Lindner, A.B.; Taddei, F. Genetic information transfer promotes cooperation in bacteria. Proc. Natl. Acad. Sci. USA 2014, 111, 11103–11108. [Google Scholar] [CrossRef]
- Nogueira, T.; Rankin, D.J.; Touchon, M.; Taddei, F.; Brown, S.P.; Rocha, E.P.C. Horizontal Gene Transfer of the Secretome Drives the Evolution of Bacterial Cooperation and Virulence. Curr. Biol. 2009, 19, 1683–1691. [Google Scholar] [CrossRef] [PubMed]
- Los, J.M.; Los, M.; Wegrzyn, A.; Wegrzyn, G. Altruism of Shiga toxin-producing Escherichia coli: Recent hypothesis versus experimental results. Front. Cell. Infect. Microbiol. 2012, 2, 166. [Google Scholar] [CrossRef]
- Jurgens, K.; Matz, C. Predation as a shaping force for the phenotypic and genotypic composition of planktonic bacteria. Antonie Van. Leeuwenhoek 2002, 81, 413–434. [Google Scholar] [CrossRef] [PubMed]
- Matz, C.; Kjelleberg, S. Off the hook-how bacteria survive protozoan grazing. Trends Microbiol. 2005, 13, 302–307. [Google Scholar] [CrossRef]
- Li, X.-P.; Tumer, N.E. Differences in Ribosome Binding and Sarcin/Ricin Loop Depurination by Shiga and Ricin Holotoxins. Toxins 2017, 9, 133. [Google Scholar] [CrossRef]
- Lainhart, W.; Stolfa, G.; Koudelka, G.B. Shiga toxin as a bacterial defense against a eukaryotic predator, Tetrahymena thermophila. J. Bacteriol. 2009, 191, 5116–5122. [Google Scholar] [CrossRef]
- Arnold, J.W.; Spacht, D.; Koudelka, G.B. Determinants that govern the recognition and uptake of Escherichia coli O157 : H7 by Acanthamoeba castellanii. Cell. Microbiol. 2016, 18, 1459–1470. [Google Scholar] [CrossRef]
- Chou, T.C.; Chiu, H.C.; Kuo, C.J.; Wu, C.M.; Syu, W.J.; Chiu, W.T.; Chen, C.S. Enterohaemorrhagic Escherichia coli O157:H7 Shiga-like toxin 1 is required for full pathogenicity and activation of the p38 mitogen-activated protein kinase pathway in Caenorhabditis elegans. Cell. Microbiol. 2013, 15, 82–97. [Google Scholar] [CrossRef]
- Youn, M.; Lee, K.-M.; Kim, S.H.; Lim, J.; Yoon, J.W.; Park, S. Escherichia coli O157:H7 LPS O-side chains and pO157 are required for killing Caenorhabditis elegans. Biochem. Biophys. Res. Commun. 2013, 436, 388–393. [Google Scholar] [CrossRef]
- Lee, K.-M.; Kim, W.-S.; Lim, J.; Nam, S.; Youn, M.; Nam, S.-W.; Kim, Y.; Kim, S.-H.; Park, W.; Park, S. Antipathogenic Properties of Green Tea Polyphenol Epigallocatechin Gallate at Concentrations below the MIC against Enterohemorrhagic Escherichia coli O157:H7. J. Food Prot. 2009, 72, 325–331. [Google Scholar] [CrossRef]
- Blattner, F.R.; Plunkett, G., III; Bloch, C.A.; Perna, N.T.; Burland, V.; Riley, M.; Collado-Vides, J.; Glasner, J.D.; Rode, C.K.; Mayhew, G.F.; et al. The complete genome sequence of Escherichia coli K-12. Science 1997, 277, 1453–1474. [Google Scholar] [CrossRef] [PubMed]
- Brenner, S. The genetics of Caenorhabditis elegans. Genetics 1974, 77, 71–94. [Google Scholar] [CrossRef] [PubMed]
- Arnold, J.W.; Koudelka, G.B. The Trojan Horse of the microbiological arms race: Phage-encoded toxins as a defence against eukaryotic predators. Environ. Microbiol. 2014, 16, 454–466. [Google Scholar] [CrossRef] [PubMed]
- Mellmann, A.; Bielaszewska, M.; Köck, R.; Friedrich, A.W.; Fruth, A.; Middendorf, B.; Harmsen, D.; Schmidt, M.A.; Karch, H. Analysis of collection of hemolytic uremic syndrome-associated enterohemorrhagic Escherichia coli. Emerg. Infect. Dis. 2008, 14, 1287–1290. [Google Scholar] [CrossRef]
- Schmidt, H.; Geitz, C.; Tarr, P.I.; Frosch, M.; Karch, H. Non-O157:H7 pathogenic Shiga toxin-producing Escherichia coli: Phenotypic and genetic profiling of virulence traits and evidence for clonality. J. Infect. Dis. 1999, 179, 115–123. [Google Scholar] [CrossRef]
- Müthing, J.; Meisen, I.; Zhang, W.; Bielaszewska, M.; Mormann, M.; Bauerfeind, R.; Schmidt, M.A.; Friedrich, A.W.; Karch, H. Promiscuous Shiga toxin 2e and its intimate relationship to Forssman. Glycobiology 2012, 22, 849–862. [Google Scholar] [CrossRef]
- Axelsson-Olsson, D.; Olofsson, J.; Ellström, P.; Waldenström, J.; Olsen, B. A simple method for long-term storage of Acanthamoeba species. Parasitol. Res. 2009, 104, 935–937. [Google Scholar] [CrossRef]
- Page, F.C. An Illustrated Key to Freshwater and Soil Amoebae with Notes on Cultivation and Ecology; Freshwater Biological Association: Ambleside, UK, 1976; ISBN 900 386 26 6. [Google Scholar]
- Page, F.C. A New Key to Freshwater and Soil Gymnamoebae: With Instructions for Culture; Freshwater Biological Association: Ambleside, UK, 1988. [Google Scholar]
- Koudelka, G.B.; Arnold, J.W.; Chakraborty, D. Evolution of STEC virulence: Insights from the antipredator activities of Shiga toxin producing E. coli. Int. J. Med. Microbiol. 2018, 308, 956–961. [Google Scholar] [CrossRef]
- Apfeld, J.; Kenyon, C. Regulation of lifespan by sensory perception in Caenorhabditis elegans. Nature 1999, 402, 804–809. [Google Scholar] [CrossRef]
- Hsin, H.; Kenyon, C. Signals from the reproductive system regulate the lifespan of C. elegans. Nature 1999, 399, 362–366. [Google Scholar] [CrossRef]
- Apfeld, J.; Kenyon, C. Cell Nonautonomy of C. elegans daf-2 Function in the Regulation of Diapause and Life Span. Cell 1998, 95, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Royston, P.; Parmar, M.K.B. Restricted mean survival time: An alternative to the hazard ratio for the design and analysis of randomized trials with a time-to-event outcome. BMC Med. Res. Methodol. 2013, 13, 152. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.-S.; Nam, H.-J.; Seo, M.; Han, S.K.; Choi, Y.; Nam, H.G.; Lee, S.-J.; Kim, S. OASIS: Online Application for the Survival Analysis of Lifespan Assays Performed in Aging Research. PLoS ONE 2011, 6, e23525. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Koudelka, G.B. The Oligosaccharide Region of LPS Governs Predation of E. coli by the Bacterivorous Protist, Acanthamoeba castellanii. Microbiol. Spectr. 2023, 11, e02930-22. [Google Scholar] [CrossRef]
- Rodríguez-Rubio, L.; Haarmann, N.; Schwidder, M.; Muniesa, M.; Schmidt, H. Bacteriophages of Shiga Toxin-Producing Escherichia coli and Their Contribution to Pathogenicity. Pathogens 2021, 10, 404. [Google Scholar] [CrossRef]
- Frézal, L.; Félix, M.A. The natural history of model organisms: C. elegans outside the Petri dish. eLife 2015, 4, e05849. [Google Scholar] [CrossRef]
- Strockbine, N.A.; Bopp, C.A.; Fields, P.I.; Kaper, J.B.; Nataro, J.P. Escherichia, Shigella, and Salmonella. In Manual of Clinical Microbiology; ASM Press: Washington, DC, USA, 2015; pp. 685–713. [Google Scholar]
- Stolfa, G.; Koudelka, G.B. Entry and Killing of Tetrahymena thermophila by Bacterially Produced Shiga Toxin. mBio 2012, 4, e00416-12. [Google Scholar] [CrossRef]
- Aijaz, I.; Koudelka, G.B. Cheating, facilitation and cooperation regulate the effectiveness of phage-encoded exotoxins as antipredator molecules. MicrobiologyOpen 2018, 8, e00636. [Google Scholar] [CrossRef]
- Chekabab, S.M.; Daigle, F.; Charette, S.J.; Dozois, C.M.; Harel, J. Shiga toxins decrease enterohaemorrhagic Escherichia coli survival within Acanthamoeba castellanii. FEMS Microbiol. Lett. 2013, 344, 86–93. [Google Scholar] [CrossRef]
- Licznerska, K.; Nejman-Faleńczyk, B.; Bloch, S.; Dydecka, A.; Topka, G.; Gąsior, T.; Węgrzyn, A.; Węgrzyn, G. Oxidative Stress in Shiga Toxin Production by Enterohemorrhagic Escherichia coli. Oxid. Med. Cell Longev. 2016, 2016, 3578368. [Google Scholar] [CrossRef]
- Tesh, V.L. Activation of cell stress response pathways by Shiga toxins. Cell. Microbiol. 2012, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Smith, W.E.; Kane, A.V.; Campbell, S.T.; Acheson, D.W.; Cochran, B.H.; Thorpe, C.M. Shiga toxin 1 triggers a ribotoxic stress response leading to p38 and JNK activation and induction of apoptosis in intestinal epithelial cells. Infect. Immun. 2003, 71, 1497–1504. [Google Scholar] [CrossRef] [PubMed]
- Colon, M.P.; Chakraborty, D.; Pevzner, Y.; Koudelka, G.B. Mechanisms that Determine the Differential Stability of Stx(+) and Stx(-) Lysogens. Toxins 2016, 8, 96. [Google Scholar] [CrossRef] [PubMed]
- Gong, C.; Chakraborty, D.; Koudelka, G.B. A prophage encoded ribosomal RNA methyltransferase regulates the virulence of Shiga-toxin-producing Escherichia coli (STEC). Nucleic Acids Res. 2024, 52, 856–871. [Google Scholar] [CrossRef]
- Bullwinkle, T.J.; Samorodnitsky, D.; Rosati, R.C.; Koudelka, G.B. Determinants of bacteriophage 933W repressor DNA binding specificity. PLoS ONE 2012, 7, e34563. [Google Scholar] [CrossRef][Green Version]
- Chakraborty, D.; Clark, E.; Mauro, S.A.; Koudelka, G.B. Molecular Mechanisms Governing “Hair-Trigger” Induction of Shiga Toxin-Encoding Prophages. Viruses 2018, 10, 228. [Google Scholar] [CrossRef]
- Bloch, S.; Nejman-Faleńczyk, B.; Dydecka, A.; Łoś, J.M.; Felczykowska, A.; Węgrzyn, A.; Węgrzyn, G. Different Expression Patterns of Genes from the Exo-Xis Region of Bacteriophage λ and Shiga Toxin-Converting Bacteriophage Φ24B following Infection or Prophage Induction in Escherichia coli. PLoS ONE 2014, 9, e108233. [Google Scholar] [CrossRef]
- Bloch, S.; Nejman-Faleńczyk, B.; Licznerska, K.; Dydecka, A.; Topka-Bielecka, G.; Necel, A.; Węgrzyn, A.; Węgrzyn, G. Complex effects of the exo-xis region of the Shiga toxin-converting bacteriophage Φ24(B) genome on the phage development and the Escherichia coli host physiology. J. Appl. Genet. 2024, 65, 191–211. [Google Scholar] [CrossRef]
- Bloch, S.; Nejman-Faleńczyk, B.; Łoś, J.M.; Barańska, S.; Łepek, K.; Felczykowska, A.; Łoś, M.; Węgrzyn, G.; Węgrzyn, A. Genes from the exo-xis region of λ and Shiga toxin-converting bacteriophages influence lysogenization and prophage induction. Arch. Microbiol. 2013, 195, 693–703. [Google Scholar] [CrossRef]
- Berger, P.; Kouzel, I.U.; Berger, M.; Haarmann, N.; Dobrindt, U.; Koudelka, G.B.; Mellmann, A. Carriage of Shiga toxin phage profoundly affects Escherichia coli gene expression and carbon source utilization. BMC Genom. 2019, 20, 504. [Google Scholar] [CrossRef]
- Serra-Moreno, R.; Jofre, J.; Muniesa, M. The CI Repressors of Shiga Toxin-Converting Prophages Are Involved in Coinfection of Escherichia coli Strains, Which Causes a Down Regulation in the Production of Shiga Toxin 2. J. Bacteriol. 2008, 190, 4722–4735. [Google Scholar] [CrossRef]
- Hashuel, R.; Wellins, T.; Rachimi, M.; Gutman, E.; Melki, R.; Taktuk, L.; Wasserzug, P.e.P.; Avissar, R.; Roitman, K.; Litvak, Y. The bully phage: A Shiga toxin-encoding prophage interferes with the induction of co-hosted prophages. bioRxiv 2025. [Google Scholar] [CrossRef]
- Morozov, A.Y.; Adamson, M.W. Evolution of virulence driven by predator-prey interaction: Possible consequences for population dynamics. J. Theor. Biol. 2011, 276, 181–191. [Google Scholar] [CrossRef]




| Strain | Serotype | Stx Type | Stx Titer * |
|---|---|---|---|
| HUSEC 13 | O26:H11 | Stx1 | 724 |
| HUSEC 14 | O26:H- | Stx1 and 2 | 512 |
| HUSEC 19 | OR:H11 | Stx1 | 181 |
| HUSEC 20 | O26:H11 | Stx2 | 512 |
| HUSEC 31 | OR:H- | Stx1 | 724 |
| Bacterial Strain | Restricted Mean Survival Time Hours (± Std Error) |
|---|---|
| OP50 | 239.4 (3.4) |
| H13 | 204.9 (2.3) |
| H14 | 221.2 (2.6) |
| H19 | 225.3 (3.3) |
| H20 | 205.6 (1.9) |
| H31 | 198.2 (2.2) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hart, P.; Bowitch, A.; Mellmann, A.; Ferkey, D.M.; Koudelka, G.B. Factors Governing the Cross-Species Virulence of Shiga Toxin-Producing Escherichia coli. Pathogens 2026, 15, 353. https://doi.org/10.3390/pathogens15040353
Hart P, Bowitch A, Mellmann A, Ferkey DM, Koudelka GB. Factors Governing the Cross-Species Virulence of Shiga Toxin-Producing Escherichia coli. Pathogens. 2026; 15(4):353. https://doi.org/10.3390/pathogens15040353
Chicago/Turabian StyleHart, Paul, Alexander Bowitch, Alexander Mellmann, Denise M. Ferkey, and Gerald B. Koudelka. 2026. "Factors Governing the Cross-Species Virulence of Shiga Toxin-Producing Escherichia coli" Pathogens 15, no. 4: 353. https://doi.org/10.3390/pathogens15040353
APA StyleHart, P., Bowitch, A., Mellmann, A., Ferkey, D. M., & Koudelka, G. B. (2026). Factors Governing the Cross-Species Virulence of Shiga Toxin-Producing Escherichia coli. Pathogens, 15(4), 353. https://doi.org/10.3390/pathogens15040353






