1. Introduction
The causative agent of bovine tuberculosis is the acid-fast bacillus
Mycobacterium bovis [
1].
M. bovis, a member of the
Mycobacterium tuberculosis complex, has a broad host range infecting numerous mammalian species from domesticated animals and wildlife to humans, in which it is known as zoonotic tuberculosis [
2]. In the early 1900s, bovine tuberculosis (bTB) was a major threat to the U.S. livestock industry and a significant public health issue mainly due to the consumption of raw dairy products from tuberculous cows [
1]. In response, the U.S. Department of Agriculture (USDA) created the National Tuberculosis Eradication Program in 1917. The program, a collaboration between federal and state governments, has since been very successful, lowering the cattle-level prevalence of bTB from approximately 5% in 1917 to <0.000008% in 2023 [
3]. Despite such a low prevalence in cattle herds, this disease continues to have economic, animal health, and public health implications.
Cattle are commonly infected with
M. bovis via inhalation or ingestion, with calves most commonly exposed via milk [
4,
5]. The disease name is derived from the characteristic formation of tubercles, or granulomas, which are a result of the host’s cell-mediated immune response [
1,
4]. Granuloma formation by the host is a mechanism to contain and eliminate the pathogen by limiting mycobacterial proliferation [
6]. The location and extent of these lesions are determined by several factors, including exposure route and dose, host immune response, and pathogen characteristics [
4,
7].
As bTB prevalence in U.S. cattle is low and clinical signs from advanced disease are rarely seen, the USDA’s National Tuberculosis Eradication Program relies on the detection and submission of gross lesions at slaughter as the primary method for bTB surveillance [
8]. Antemortem diagnostic tests, including skin and blood tests, are also used in both surveillance and disease control. All tests involve stimulation with
M. bovis antigen and measure subsequent host cell-mediated immunity. Because these tests rely on host immune response rather than pathogen detection, results may be affected by the state of disease progression or latency [
9]. The caudal fold tuberculin (CFT) skin test is used for screening, and if positive, is followed by one or both of the official, secondary confirmatory tests, the comparative cervical tuberculin (CCT) skin test and/or the gamma interferon blood test. These tests are used to confirm disease, and if either or both are positive, indicate a need for postmortem testing.
After a herd is determined to be infected with bTB, animals suspected to be infected based on antemortem testing are euthanized to facilitate quick identification and elimination of bTB. The carcasses of these animals are inspected for gross lesions, and samples are submitted to the USDA’s National Veterinary Services Laboratories (NVSL) for confirmatory testing. As the prevalence of bTB continues to decline in U.S. cattle, detection of this pathogen will become increasingly challenging. While experimental studies are incredibly valuable in understanding pathogen characteristics and disease pathogenesis, studies of natural infection may be more relevant for disease detection and management. Studies evaluating data originating from naturally infected herds in the U.S. have been scarce. The objectives of this comparative epidemiological study were to: (1) describe the location of bTB gross lesions from naturally infected cattle, and (2) evaluate the association between bTB gross lesion location and ante-mortem test results.
2. Materials and Methods
Herds selected for inclusion in this retrospective analysis were detected within the last 10 years and completed depopulation or test-and-removal procedures to eliminate bTB. All herds included not only had sufficient reporting of gross lesion data and laboratory results but also underwent epidemiologic investigation into disease source and spread. Data for this study were gathered from multiple USDA and state-level reports and databases. Gross lesion data were obtained from USDA-Animal and Plant Health Inspection Service-Veterinary Services forms 6-35 and 10-7, which report granulomas found at slaughter and lesions found at necropsy of cattle from a bTB-infected herd, respectively. Paper forms were submitted with tissue samples and scanned upon arrival to the NVSL. The digital copies of these forms were transcribed into a Microsoft Excel spreadsheet and imported into SAS 9.4 (SAS Institute Inc., Cary, NC, USA) for data analysis. Animal age and antemortem testing results were also transcribed from these forms. When available, animal age was verified using the date of birth from herd owner records. Additionally, antemortem testing results were verified using federal or state testing records when available. Definitive diagnoses for each animal were obtained via a query of the NVSL STRAND (Searchable Test Results Application for NVSL Diagnostics) database. An animal was classified as infected with bTB if postmortem lab results showed a positive bacteriologic culture or, in the case of a negative culture, the pathogen was detected on both polymerase chain reaction and histopathology analysis.
Gross lesion reporting was variable, with some examiners reporting the specific lesioned tissue and others reporting only the regional location of the lesion. Thus, this analysis focuses on the region of lesion location and discusses specific sites of lesions only where available. Analyses of lesion count and lesion location were conducted on an individual animal basis. Lesion count was defined as the number of distinct specific tissues affected, not the number of lesions within one tissue, as that information was not collected.
Descriptive and statistical analyses were completed using SAS. Statistical tests conducted involving continuous variables included the one-way ANOVA test and the t-test. For associations between categorical variables, the chi-squared test, or Fisher’s exact test when greater than 20% of cell frequencies were less than 5, was used. Figures were created in Microsoft Excel and Adobe Photoshop.
3. Results
Data for analysis were compiled from nine infected U.S. herds, which represent both beef (n = 3) and dairy (n = 6) cattle as well as a variety of geographic locations, herd sizes, sources of introduction, and levels of intra-herd disease prevalence (0.1–16.3%). Details on these herds can be found in
Table 1 below. The location of the herds presented is mapped to the center of their county in
Figure 1 below.
3.1. Beef vs. Dairy Cattle
The average age of infected cattle was 46.6 months of age, with beef cattle being significantly older on average than dairy cattle (
Table 2). One possible reason for the age differences may be attributable to the longevity of beef cattle compared with dairy cattle. The mean age and distribution of beef cattle are older and broader, respectively, compared with dairy cattle. This may be due in part to higher cull rates in dairy cattle when compared to beef cattle [
10,
11]. Modeling results suggest that bTB detection in small- and medium-sized beef herds takes longer than in dairy herds because of the smaller percentage of the adult herd that is removed to slaughter and inspected each year [
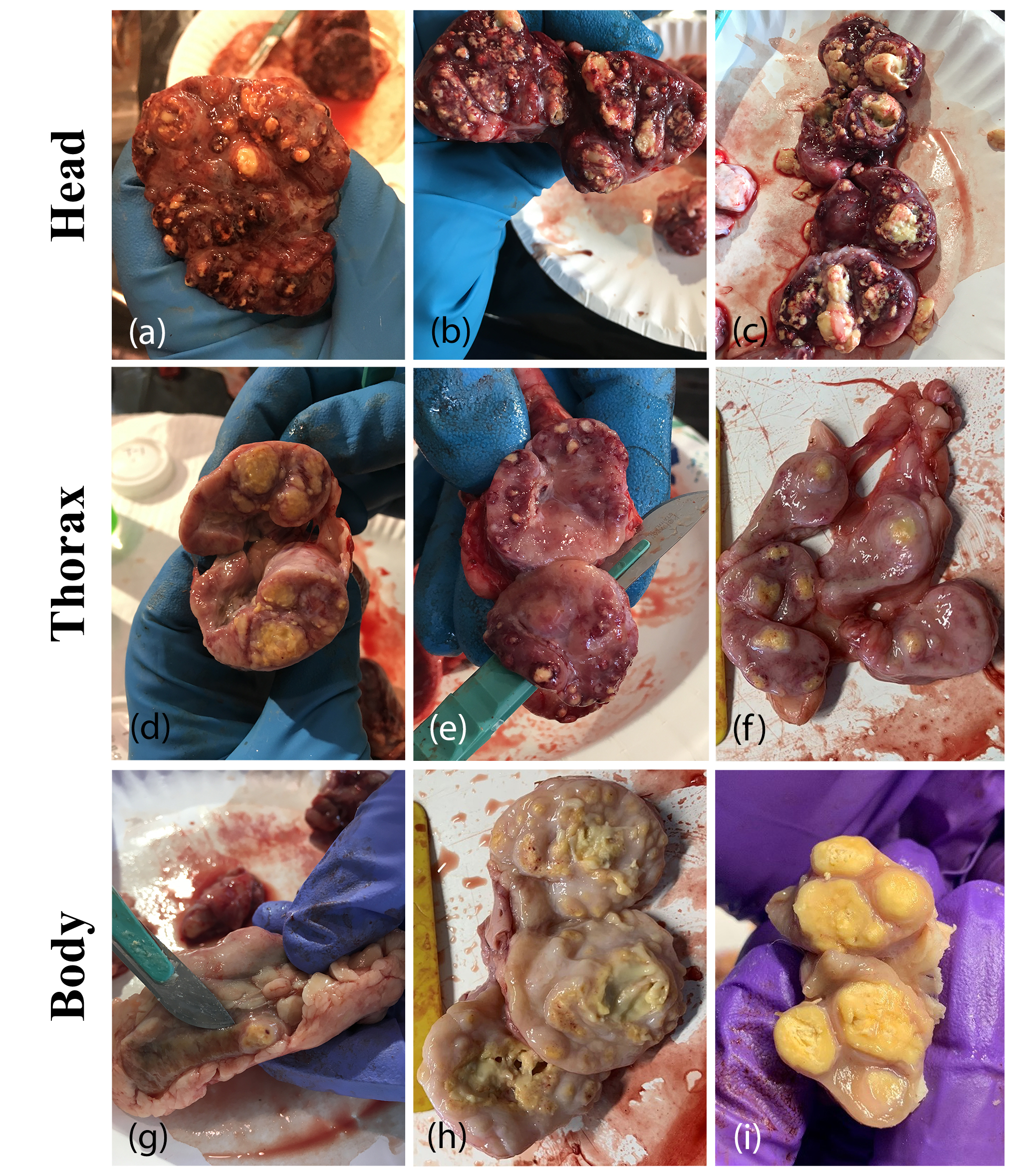
12]. This delay in detection could lead to more within-herd spread and the beef herds in this analysis did have higher herd-level prevalence than dairy herds. Overall, most cattle infected with bTB presented with gross lesions (95.3%) with no difference between beef and dairy. Most often, these cattle had lesions in only one body region, with the majority having lesions only in the thorax, followed by only in the head. Selected lesion photos from Herd D.5 are presented in
Figure 2. Beef and dairy cattle showed similar trends in lesion count and lesion location (
Figure 3). Although a greater number of beef cattle presented with multiple lesions (41.5% vs 30.9%), this finding was not statistically significant. A higher percentage of beef than dairy cattle had any lesions in the body region (26.3% vs 16.1%,
p = 0.019 and only lesions in the body region (11.9% vs 3.9%,
p = 0.002).
3.2. Specific Sites of Gross Lesions in bTB-Infected Cattle
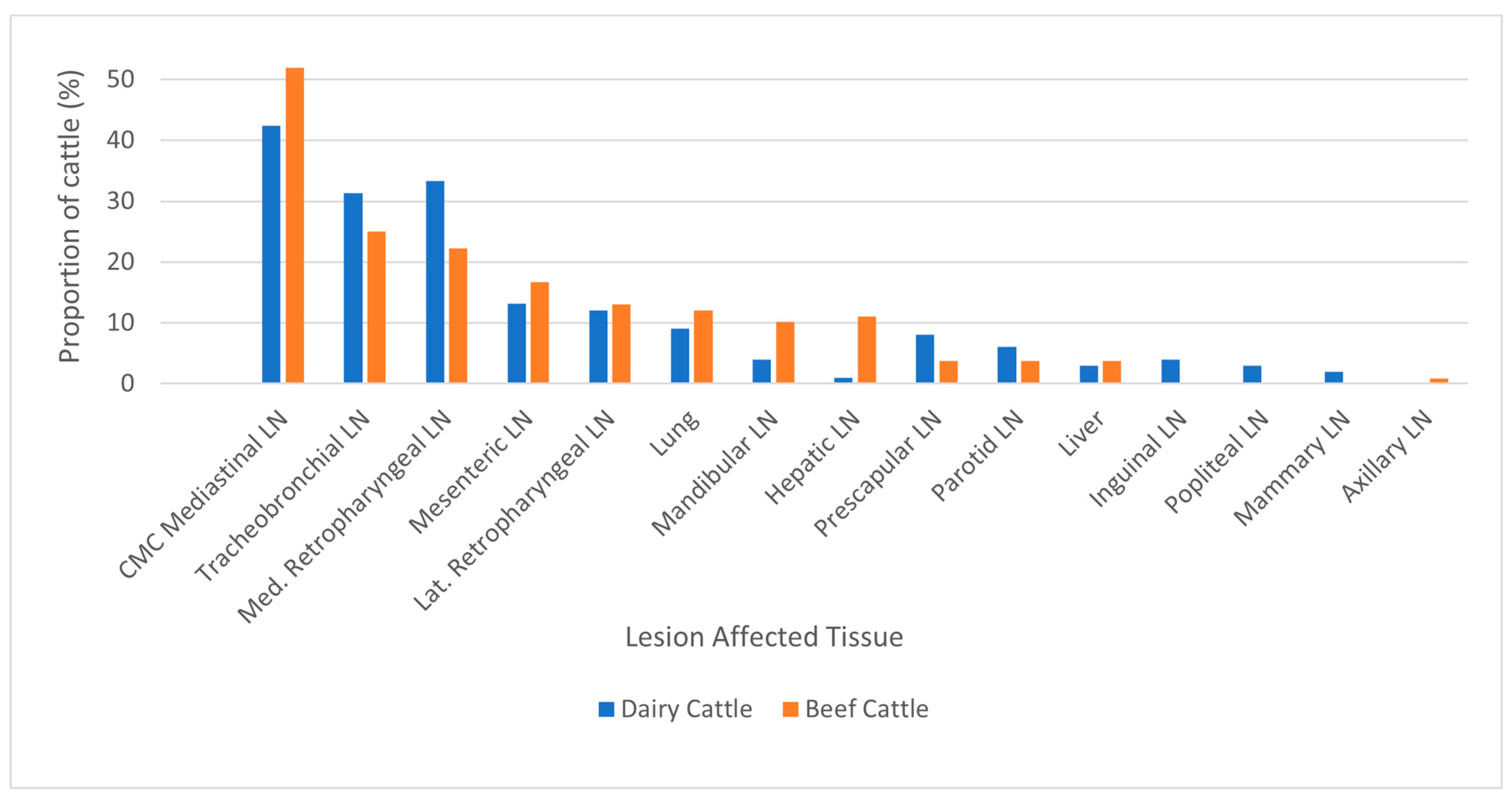
When available, data on the specific tissue affected with gross lesions in bTB-infected animals were analyzed. Overall, the mediastinal lymph nodes were most affected, followed by the tracheobronchial and medial retropharyngeal lymph nodes, being lesioned in 47.3%, 28.0% and 27.5% of all cattle, respectively (
Table 3 and
Figure 4). Beef and dairy cattle showed similar patterns in lesion distribution. Notably, beef cattle were significantly more likely to have lesions in the hepatic lymph node (11.1% and 1.0%, respectively).
3.3. Herd Stratification
Gross lesion presentation differed among the herds included in this study. Most infected herds had at least 77% of the cattle with gross lesions (
Table 4). Notably, no gross lesions were found in the three infected cattle from D.1. While most herds predominantly had animals with a single lesion, multiple lesions were observed in at least half of the animals from herds D.3 and D.6. With regard to regional lesion distribution, patterns were variable between herds. In five of the herds, thoracic lesions alone were the most frequent pattern observed. However, in herd D.4, head lesions alone were found more often, and in herd D.3, body lesions predominated both on their own and in conjunction with other regions.
3.4. Antemortem Skin Testing
Antemortem test results presented in
Table 5 were based on the most recent test/series of tests conducted and were therefore those most proximate to the animal’s death. Not all infected animals received any and/or all tests due to the method of detection (slaughter vs herd removal), detection in trace-outs, and differing antemortem testing strategies. The CFT test showed high levels of sensitivity (>90%) and similar performance in both beef and dairy cattle. This sensitivity estimate should be interpreted with caution since cattle with negative CFT results were not generally examined by necropsy. Although many CFT-negative cows from these herds went through slaughter surveillance, this method of detection is not as sensitive as necropsy. The CCT test, however, performed significantly different (
p = 0.003) between beef and dairy cattle, with the CCT positivity rate, defined as those classified as either suspect or reactors, being 81.8% in beef compared to only 58.0% in dairy cattle. Test performance stratified by herd was variable. Although CFT performance was relatively consistent among herds and similar if not greater than established sensitivity levels [
13], CCT results were more variable between breeds (
Figure 5).
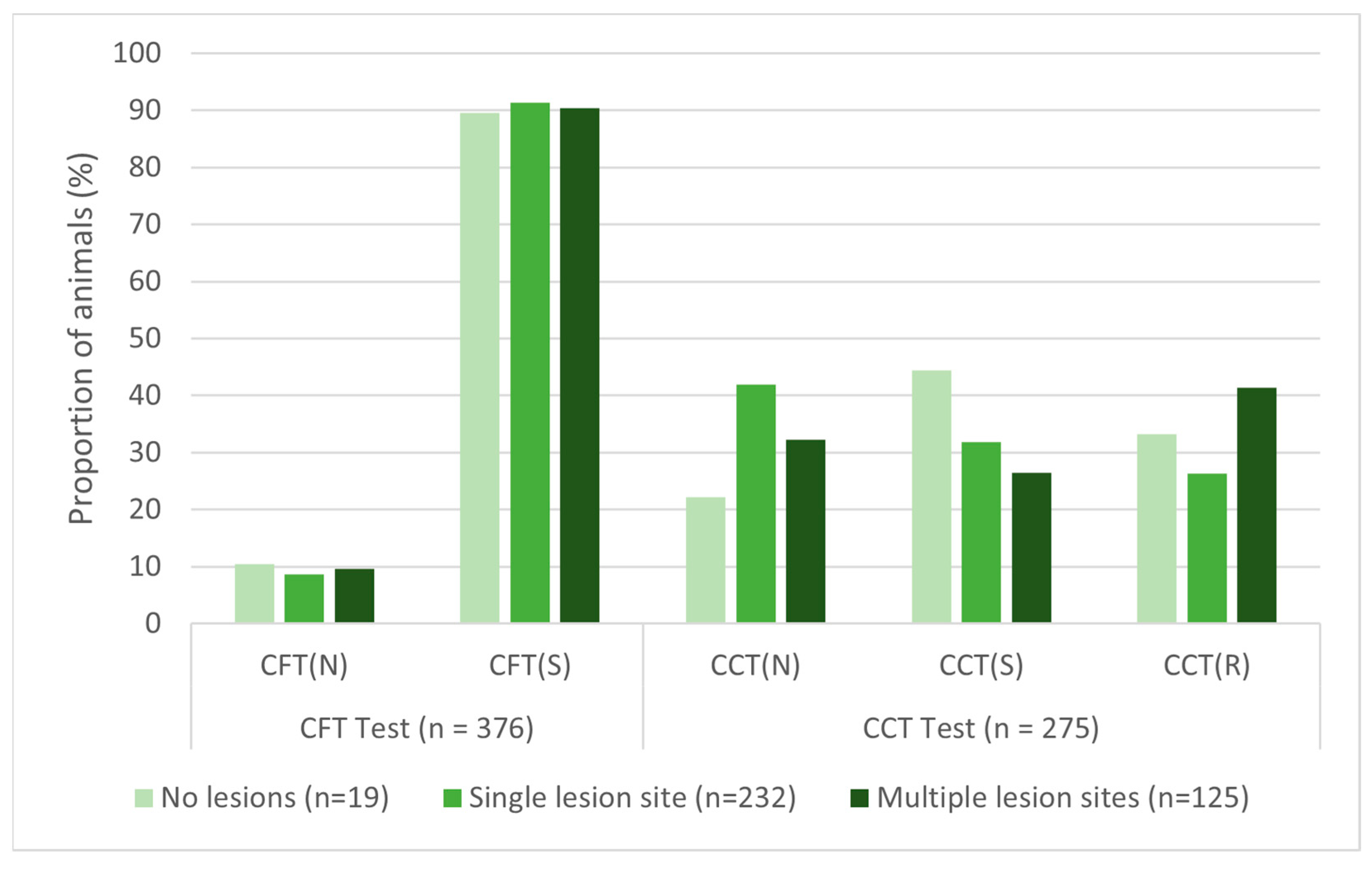
There were no significant associations found between antemortem test results and lesion count for either skin test (
Table 6). The sensitivity of the CFT test was within 2% among animals with zero, one, or multiple lesion sites present, while CCT sensitivity was more variable (
Figure 6). Additionally, there were no associations between skin test results and lesion presence in specific body sites, except that cattle with lesions only in the body region were significantly less likely to show a response to CFT testing than all other lesion location cohorts.
4. Discussion
4.1. Overall Gross Lesion Findings
The sensitivity of gross lesions for the detection of bTB-infected cattle was 95.3%, which was higher than reported from a study evaluating seven herds in Michigan, USA, where the sensitivity of gross lesions was 86.0% [
14]. The presence and extent of gross lesions from bTB are known to vary with route of exposure, pathogen dose and virulence, time between infection and diagnosis, and host immune response [
7]. The predominance of lesions in the thoracic region is consistent with findings from experimental and natural infection with
M. bovis in previous studies [
6,
15,
16]. It is widely accepted that the method of exposure plays a large role in the location and distribution of gross lesions in bTB infection [
7,
17]. Therefore, it is reasonable to speculate that the most common source of transmission to cattle included in this analysis was respiratory. In experimental studies exposing cattle to
M. bovis via aerosol delivery, gross lesions were most often found in the lungs and associated lymph nodes [
18,
19,
20]. In this analysis, lung lesions were only noted in 10.6% of cattle, which may indicate a need for improvement in postmortem lung inspection.
In this analysis, 15% of cattle presented with lesions in the mesenteric Ln. In other studies of gross lesions resulting from natural infection with
M. bovis, the mesenteric Ln. represented 0–10% of lesions [
15,
16,
21]. However, a study from China reported more than 90% of lesions in 151 necropsied cattle were extrapulmonary; more than 70% of lesions were in the gastrointestinal tract, and 43% were in the mesenteric Ln. [
22]. Mesenteric Ln., and others in or adjacent to the gastrointestinal tract, may be affected from exposure to
M. bovis via ingestion [
1,
4]. This method of exposure is generally thought to be most relevant for calves who may consume
M. bovis from contaminated milk or be in close contact with their dams [
4,
22,
23]. Based on this analysis, ingestion may be a more common transmission route in the U.S. than previously thought, occurring in older cohorts of cattle as well as calves. Alternatively, lymph nodes and other organs not associated with the respiratory tract may be affected with gross lesions from disseminated infection. This would likely occur later in the disease process via hematogenous or lymphatic spread, or perhaps secondary to the swallowing of contaminated sputum [
7,
17]. However, of those animals with lesions in the mesenteric lymph nodes (n = 31), 35.5% presented with lesions in the body region alone, indicating that the region may have been the primary site of infection. As we cannot determine the chronological development of these lesions, it is impossible to differentiate respiratory exposure with dissemination to the abdomen from alimentary exposure with dissemination to the thorax.
Of note is that 4.7% of infected animals in this study did not have any gross lesions, and this percentage is likely an underestimate of the infected cattle population. This finding illustrates the importance of testing samples from animals suspected to have bTB, regardless of the presence of gross lesions.
4.2. Beef vs. Dairy
Beef and dairy cattle were compared as these groups involve different breeds and management practices. As noted in
Table 1, the prevalence of disease in beef herds was higher on average than in dairy herds. There are several reasons why the detection of
M. bovis in beef herds may be more challenging, thereby allowing the disease to spread for a greater duration than in dairy herds. The beef industry has a much lower culling (permanent removal) rate than the dairy industry—approximately 13% vs. 30%—meaning that the chance of finding an infected herd via an animal at slaughter is decreased due to sample size alone [
10,
11]. Compounding this is the fact that beef herds, as shown in this analysis, tend to be smaller than dairy herds. The prevalence in beef herds in this study may be exaggerated, as all beef herds were selected for whole-herd depopulation. Therefore, each beef animal underwent either a slaughter or necropsy inspection, which could have resulted in the detection of additional infected animals that may not have been detected by antemortem testing or slaughter surveillance alone. Alternatively, or in conjunction, the management of beef herds may make cow-to-cow transmission more likely, as beef herds tend to comingle animals of different ages, thereby potentially exposing young, vulnerable animals to infected older ones, although this is a risk at some dairies too.
Beef animals were significantly more likely to present with lesions in the body only (11.9% vs. 3.9%, p = 0.002). This suggests beef cattle are exposed to the pathogen more often via ingestion than dairy cattle. Interestingly, beef cattle had a significantly higher proportion of lesions in the hepatic lymph node than dairy cattle, which could be attributed to the filtration of contaminated fluids from the gastrointestinal tract. However, a difference was not found at other related sites such as the liver or mesenteric ln. It is also worth noting that there was a significant difference in the age of infected cattle, with the average age of beef cattle being approximately two years greater than that of dairy cattle, which could have had an impact on lesion development and presentation as disease progresses over time.
4.3. Variation Between Herds
As this analysis includes animals from nine different herds across the country exposed to
M. bovis via natural infection, there were many factors that could account for variation in disease development or presentation among herds. The epidemiologic triad of host, pathogen, and environment provides a framework for these potential factors. Host-related factors include immune status at the time of exposure and duration of illness. Herd-related characteristics could include genetics, nutrition, other comorbidities, and management practices. Environmental factors include stocking density and ventilation, cleanliness, and climate. Pathogen-related factors include the virulence of different strains, the portal of entry, and the dose received. Notably, each herd included in this analysis was predominantly infected by a unique
M. bovis strain, except for Herd D.5. The majority of the cows had the 17B1 strain. In addition, Herd D.5 had a single cow infected with a Group 6A strain similar to Herd B.2, although the strains were not closely related. Herd D.5 also had a single cow infected with Group 13A. However, some studies conclude that livestock management practices may have a greater impact on disease severity than strain [
20]. Future studies should focus on herd-related characteristics to determine the impact of practices on the number and location of bTB lesions.
Although overall trends emerged, there was noticeable variability in lesion count and location, with some herds having unique presentations. In all herds excepting D.1, the majority of cattle presented with gross lesions. Among these eight herds, it was more typical that animals had single lesions, although the proportion with multiple lesions ranged from 27.4 to 58.3%. Variability in the number of lesions between herds may relate to disease chronicity or animal susceptibility. Often, when an infected herd is detected, it is difficult to determine the time since exposure. However, D.1 was confirmed to be infected via human-to-cattle transmission and whole-herd testing was conducted only one month after the human case was detected [
24]. The absence of gross lesions in this herd supports that these animals may have been detected very early in the disease process. By contrast, herd D.3, another confirmed human-to-cattle transmission, was detected three years after presumed exposure and the majority of cattle presented with multiple lesions. Epidemiologic investigation and whole genome sequencing also confirmed cow-to-cow transmission in this herd.
With respect to lesion location, five of eight herds with lesions had the highest proportion of animals present with lesions in the thorax only. Uniquely, animals in D.4 were more often affected with lesions in the head only, while herd D.3 had no predominant presentation. Animals from Hawaiian herds were also variable in presentation, with thoracic lesions only and body lesions only being most common and of equal occurrence. There are many factors that could influence lesion location and dissemination, including pathogen dose, route, disease chronicity, and individual animal characteristics. While the definitive causes of variation cannot be identified in this analysis, perhaps the greater takeaway is the variation alone. As animal health officials seek to identify and eliminate bTB from U.S. herds, recognizing bTB in its variable presentations will be essential.
4.4. Antemortem Skin Testing
Antemortem tests for bTB are known to be imperfect, with the CFT skin test and CCT skin test having a median sensitivity of approximately 84% and 80%, respectively [
13,
25]. The overall sensitivity of the CFT test in this cohort of animals was 91%, which is higher than reported but similar to the 93% reported in a Michigan, USA Study [
14]. This result is biased as animals with a positive CFT test were more likely to undergo postmortem inspection. The performance of the CCT differed significantly between beef and dairy animals. The CCT had a higher sensitivity in beef than in dairy animals (81.8% vs. 58.0%). It is difficult to explain this variation, but it is worth considering the subjectivity of the tuberculin skin tests as well as the potential role of disease chronicity in beef animals. Additionally, there was a large discrepancy between the number of available CCT results for beef vs. dairy animals (44 vs. 231, respectively), which may impact interpretation. Variability in the performance of these tests between herds was similar. While the CFT was consistent among herds with a sensitivity of approximately 80% or higher, CCT performance varied with a sensitivity from 41.2 to 100 addition, test responses may differ by tuberculin dose and purified protein derivative preparations, animal stress level, or stage of disease [
25].
Antemortem tests for bTB do not rely on the detection of the pathogen, but rather on the host’s immune response when stimulated with an antigen. Therefore, it was hypothesized that animals with a greater number of lesions would be more likely to be detected. This assumes that multiple lesion sites indicate more severe disease than a single lesion, as lesion size was not commonly reported. Surprisingly, as seen in
Figure 5 above, there was no significant association or consistent trend between skin test results and the number of lesions. Not finding a difference in immune response in cattle with lesions in multiple sites and those with single or no lesions could suggest that the lack of a robust immune response might lead to more disseminated infection. It is worth noting that animals without gross lesions were a very small proportion of the infected cattle, and therefore, these results must be interpreted with caution. Lastly, there were no notable associations between lesion location and skin test result, except that cattle with lesions in only the body region had lower sensitivity on CFT testing. This has also been reported in experimentally infected calves [
26]. Alternatively, body lesions may not be detected at slaughter because these tissues are not as carefully examined as the thoracic and head tissues [
16].
4.5. Limitations
There are several limitations to acknowledge in this study, as the data comes from the field and was not originally collected for the purpose of this analysis. Even though not collected for this retrospective study, the data are routinely collected based on guidance in the Bovine Tuberculosis Eradication Uniform Methods and Rules (UMR) [
27]. Official, standardized forms, surveillance guidelines, and testing protocols are outlined in the UMR. Results and conclusions rely on the accurate and thorough inspection and reporting of gross lesions from individuals completing necropsies and slaughter inspections. Data quality and completeness may differ depending on who is completing this task, their skills and knowledge base, and any time constraints they may be working under. For example, slaughter inspection may be less thorough than necropsy due to quick line speeds and logistics regarding the splitting of the carcasses. In addition, a lot of emphasis is placed on head and thoracic evaluation for lesions during the slaughter process. This is because historically, the majority of lesions are in these 2 regions, and because the abdominal organs are removed without the same level of inspection [
14]. Second, the consistency of inspection and reporting may differ as the 403 infected animals included in this study were examined by 35 different individuals across the U.S. However, it is worth noting that 208 necropsies were completed by the same person. Third, animals with gross lesions may be overrepresented, as during certain periods, lesioned samples were prioritized for collection and laboratory testing due to human resource and lab capacity constraints. Therefore, as this sample is biased toward cattle with lesions, the percentage of infected cattle without gross lesions may be understated. Finally, lack of granularity was a limitation in this study, as the specific number or size of lesions in each organ was not recorded.
5. Conclusions
As the National Tuberculosis Eradication Program continues to strive to eliminate bTB, it is worth contemplating how to modify current approaches. bTB lesions in only the body were found in about 6% of cattle and almost 90% of cattle had bTB lesions in lymph nodes of the head and/or thorax. Current necropsy and slaughter sample collection recommendations for bTB control and surveillance should remain and additional emphasis should be placed on more thorough carcass inspections, including the lungs and body lymph nodes. The finding that beef cattle were more likely to have body lesions than dairy cattle could result in more targeted slaughter surveillance based on breed. Furthermore, the fact that not all infected animals had gross lesions, and would be missed on slaughter surveillance, highlights the importance of histopathology of tissues from test-positive animals regardless of the presence of lesions. In the pursuit of disease eradication, it is essential to use information collected from ongoing and past outbreaks to guide future surveillance and control strategies. Despite over 100 years of targeted disease eradication work, there is still much to learn about bovine tuberculosis. To our knowledge, this paper is the first to examine gross lesion data resulting from natural bTB infection across multiple U.S. cattle herds. This analysis provides further insight into bTB control, including exposure and transmission mechanisms, predilection sites, and antemortem testing patterns.
Author Contributions
Conceptualization, J.E.L. and S.R.-A.; methodology, C.C.D. and J.E.L.; validation, C.C.D., K.C.M., C.R., H.M. and J.E.L.; formal analysis, C.C.D. and J.E.L.; investigation, C.C.D., K.C.M., C.R., H.M., S.R.-A. and J.E.L.; resources, C.C.D., K.C.M., C.R., H.M. and J.E.L.; data curation, C.C.D. and J.E.L.; writing—original draft preparation, C.C.D., H.M. and J.E.L.; writing—review and editing, C.C.D., K.C.M., C.R., H.M., S.R.-A. and J.E.L.; visualization, C.C.D., H.M. and J.E.L.; supervision, J.E.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Data were collected during official USDA investigations and are unavailable due to privacy restrictions.
Acknowledgments
The authors thank: State and Federal personnel who performed the on-farm testing, necropsies, and collected the data included in this manuscript, Kate Schinkel for manuscript editing and formatting, and Maddi Funk for creating
Figure 1 and
Figure 2.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations and Acronyms
The following abbreviations and acronyms are used in this manuscript:
| bTB | Bovine tuberculosis |
| USDA | United States Department of Agriculture |
| STRAND | Searchable Test Results Application for NVSL Diagnostics |
| NVSL | National Veterinary Services Laboratories |
| Ln | Lymph node(s) |
Appendix A
Table A1.
Region Description.
Table A1.
Region Description.
| Region | Tissues Included |
|---|
| Head | medial retropharyngeal ln., lateral retropharyngeal ln., parotid ln., mandibular ln. |
| Thorax | mediastinal ln., tracheobronchial ln., lungs, heart |
| Body | mesenteric ln., hepatic ln., mammary ln., prescapular ln., popliteal ln., axillary ln., inguinal ln., liver |
References
- Mycobacterium bovis Infection in Animals and Humans, 1st ed.; Thoen, C.O., Steele, J.H., Eds.; Iowa State University Press: Ames, IA, USA, 1995. [Google Scholar]
- O’Reilly, L.M.; Daborn, C.J. The Epidemiology of Mycobacterium bovis Infections in Animals and Man: A Review. Tuber. Lung Dis. 1995, 76, 1–46. [Google Scholar] [CrossRef] [PubMed]
- USDA National Tuberculosis Eradication Program Update. USAHA Subcommittee on Tuberculosis, USDA APHIS Veterinary Services, National Harbor, MD, USA, October 2023. Available online: https://usaha.org/upload/Committee/CattleBison/USDA_TB_Eradication_Update_Lyons.pdf (accessed on 20 March 2026).
- Zoonotic Tuberculosis: Mycobacterium bovis and Other Pathogenic Mycobacteria, 3rd ed.; Thoen, C.O., Steele, J.H., Kaneene, J.B., Eds.; Wiley Blackwell: Ames, IA, USA, 2014. [Google Scholar]
- Byrne, A.W.; Barrett, D.; Breslin, P.; Fanning, J.; Casey, M.; Madden, J.M.; Lesellier, S.; Gormley, E. Bovine Tuberculosis in Youngstock Cattle: A Narrative Review. Front. Vet. Sci. 2022, 9, 1000124. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.V.; Waters, W.R.; Thacker, T.C. Lesion Development and Immunohistochemical Changes in Granulomas from Cattle Experimentally Infected with Mycobacterium bovis. Vet. Pathol. 2007, 44, 863–874. [Google Scholar] [CrossRef] [PubMed]
- Neill, S.D.; Pollock, J.M.; Bryson, D.B.; Hanna, J. Pathogenesis of Mycobacterium bovis Infection in Cattle. Vet. Microbiol. 1994, 40, 41–52. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture. National Tuberculosis Eradication Program. Available online: https://www.aphis.usda.gov/aphis/ourfocus/animalhealth/animal-disease-information/cattle-disease-information/national-tuberculosis-eradication-program/national-tuberculosis-eradication-program (accessed on 12 May 2023).
- García, J.S.Y.; Bigi, M.M.; Klepp, L.I.; García, E.A.; Blanco, F.C.; Bigi, F. Does Mycobacterium bovis Persist in Cattle in a Non-Replicative Latent State as Mycobacterium tuberculosis in Human Beings? Vet. Microbiol. 2020, 247, 108758. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Agriculture. Beef 2017: Beef Cow-Calf Management Practices in the United States, 2017, Report 1; USDA–APHIS–VS–CEAH–NAHMS #782.0520; U.S. Department of Agriculture: Fort Collins, CO, USA, 2020. Available online: https://www.aphis.usda.gov/sites/default/files/beef2017_dr_parti.pdf (accessed on 20 March 2026).
- U.S. Department of Agriculture. Dairy 2014: Trends in Dairy Cattle Health and Management Practices in the United States, 1991–2014; USDA–APHIS–VS–CEAH-NAHMS #711.0821; U.S. Department of Agriculture: Fort Collins, CO, USA, 2021. Available online: https://www.aphis.usda.gov/sites/default/files/dairy-trends-hlth-mngmnt-1991-2014.pdf (accessed on 20 March 2026).
- U.S. Department of Agriculture, Fort Collins, CO. Analysis of Bovine Tuberculosis Surveillance in Accredited-Free States. 2009; Unpublished work.
- De La Rua-Domenech, R.; Goodchild, A.T.; Vordermeier, H.M.; Hewinson, R.G.; Christiansen, K.H.; Clifton-Hadley, R.S. Ante Mortem Diagnosis of Tuberculosis in Cattle: A Review of the Tuberculin Tests, γ-Interferon Assay and Other Ancillary Diagnostic Techniques. Res. Vet. Sci. 2006, 81, 190–210. [Google Scholar] [CrossRef] [PubMed]
- Norby, B.; Bartlett, P.C.; Fitzgerald, S.D.; Granger, L.M.; Bruning-Fann, C.S.; Whipple, D.L.; Payeur, J.B. The Sensitivity of Gross Necropsy, Caudal Fold and Comparative Cervical Tests for the Diagnosis of Bovine Tuberculosis. J. Vet. Diagn. Investig. 2004, 16, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Liebana, E.; Johnson, L.; Gough, J.; Durr, P.; Jahans, K.; Clifton-Hadley, R.; Spencer, Y.; Hewinson, R.G.; Downs, S.H. Pathology of Naturally Occurring Bovine Tuberculosis in England and Wales. Vet. J. 2008, 176, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Whipple, D.L.; Bolin, C.A.; Miller, J.M. Distribution of Lesions in Cattle Infected with Mycobacterium bovis. J. Vet. Diagn. Investig. 1996, 8, 351–354. [Google Scholar] [CrossRef] [PubMed]
- Domingo, M.; Vidal, E.; Marco, A. Pathology of Bovine Tuberculosis. Res. Vet. Sci. 2014, 97, S20–S29. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.V.; Ray Waters, W.; Whipple, D.L. Aerosol Delivery of Virulent Mycobacterium bovis to Cattle. Tuberculosis 2002, 82, 275–282. [Google Scholar] [CrossRef] [PubMed]
- Palmer, M.V.; Wiarda, J.; Kanipe, C.; Thacker, T.C. Early Pulmonary Lesions in Cattle Infected via Aerosolized Mycobacterium bovis. Vet. Pathol. 2019, 56, 544–554. [Google Scholar] [CrossRef] [PubMed]
- Waters, W.R.; Thacker, T.C.; Nelson, J.T.; DiCarlo, D.M.; Maggioli, M.F.; Greenwald, R.; Esfandiari, J.; Lyashchenko, K.P.; Palmer, M.V. Virulence of Two Strains of Mycobacterium bovis in Cattle Following Aerosol Infection. J. Comp. Pathol. 2014, 151, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Corner, L.; Melville, L.; McCubbin, K.; Small, K.; McCormick, B.; Wood, P.; Rothel, J. Efficiency of Inspection Procedures for the Detection of Tuberculous Lesions in Cattle. Aust. Vet. J. 1990, 67, 389–392. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Tian, L.; Li, Y.; Zhang, X.; Qi, Y.; Jing, Z.; Pan, Y.; Zhang, L.; Fan, X.; Wang, M.; et al. High Prevalence of Extrapulmonary Tuberculosis in Dairy Farms: Evidence for Possible Gastrointestinal Transmission. PLoS ONE 2021, 16, e0249341. [Google Scholar] [CrossRef] [PubMed]
- Fitzgerald, S.D.; Hollinger, C.; Mullaney, T.P.; Bruning-Fann, C.S.; Tilden, J.; Smith, R.; Averill, J.; Kaneene, J.B. Herd Outbreak of Bovine Tuberculosis Illustrates That Route of Infection Correlates with Anatomic Distribution of Lesions in Cattle and Cats. J. Vet. Diagn. Investig. 2016, 28, 129–132. [Google Scholar] [CrossRef] [PubMed]
- Lombard, J.E.; Patton, E.A.; Gibbons-Burgener, S.N.; Klos, R.F.; Tans-Kersten, J.L.; Carlson, B.W.; Keller, S.J.; Pritschet, D.J.; Rollo, S.; Dutcher, T.V.; et al. Human-to-Cattle Mycobacterium Tuberculosis Complex Transmission in the United States. Front. Vet. Sci. 2021, 8, 691192. [Google Scholar] [CrossRef] [PubMed]
- Schiller, I.; Oesch, B.; Vordermeier, H.M.; Palmer, M.V.; Harris, B.N.; Orloski, K.A.; Buddle, B.M.; Thacker, T.C.; Lyashchenko, K.P.; Waters, W.R. Bovine Tuberculosis: A Review of Current and Emerging Diagnostic Techniques in View of Their Relevance for Disease Control and Eradication. Transbound. Emerg. Dis. 2010, 57, 205–220. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.; Sevilla, I.A.; Fuertes, M.; Geijo, M.; Risalde, M.Á.; Ruiz-Fons, J.F.; Gortazar, C.; Juste, R.A.; Domínguez, L.; Elguezabal, N.; et al. Different Lesion Distribution in Calves Orally or Intratracheally Challenged with Mycobacterium bovis: Implications for Diagnosis. Vet. Res. 2018, 49, 74. [Google Scholar] [CrossRef]
- Bovine Tuberculosis Eradication: Uniform Methods and Rules, Effective January 1, 2005; APHIS 91-45-011; Animal and Plant Health Inspection Service, U.S. Department of Agriculture: Washington, DC, USA, 2004. Available online: https://www.aphis.usda.gov/sites/default/files/tb-umr.pdf (accessed on 20 March 2026).
Figure 1.
U.S. map of nine bovine tuberculosis-infected herds mapped to the center of their county location.
Figure 1.
U.S. map of nine bovine tuberculosis-infected herds mapped to the center of their county location.
Figure 2.
Typical bovine tuberculosis lesions detected in the head (a–c), thorax (d–f), and body (g–i), from infected cattle in Herd D.5 (Photos courtesy of Dr. Christa Ray).
Figure 2.
Typical bovine tuberculosis lesions detected in the head (a–c), thorax (d–f), and body (g–i), from infected cattle in Herd D.5 (Photos courtesy of Dr. Christa Ray).
Figure 3.
Lesion distribution in bovine tuberculosis-infected cattle by production type. Lesion categories are not exclusive, i.e., the same cow may be represented in multiple categories if lesions were present in more than one region.
Figure 3.
Lesion distribution in bovine tuberculosis-infected cattle by production type. Lesion categories are not exclusive, i.e., the same cow may be represented in multiple categories if lesions were present in more than one region.
Figure 4.
Lesioned tissues in lymph nodes (LN) and organs from bovine tuberculosis-Infected cattle by production type. 99/272 infected dairy cattle and 108/112 infected beef cattle with lesions had the specific organ of lesion location reported, whereas the remainder had only the region of lesion location(s) (head, thorax, body) and thus were excluded. CMC = Cranial, Middle, Caudal.
Figure 4.
Lesioned tissues in lymph nodes (LN) and organs from bovine tuberculosis-Infected cattle by production type. 99/272 infected dairy cattle and 108/112 infected beef cattle with lesions had the specific organ of lesion location reported, whereas the remainder had only the region of lesion location(s) (head, thorax, body) and thus were excluded. CMC = Cranial, Middle, Caudal.
Figure 5.
Antemortem skin-test results for bovine tuberculosis-infected cattle by breed. CFT = caudal fold tuberculin, CCT = comparative cervical tuberculin, (N) = negative, (S) = suspect, (R) = reactor.
Figure 5.
Antemortem skin-test results for bovine tuberculosis-infected cattle by breed. CFT = caudal fold tuberculin, CCT = comparative cervical tuberculin, (N) = negative, (S) = suspect, (R) = reactor.
Figure 6.
Antemortem skin-test results for bovine tuberculosis-infected cattle by lesion count. CFT = caudal fold tuberculin, CCT = comparative cervical tuberculin, (N) = negative, (S) = suspect, (R) = reactor.
Figure 6.
Antemortem skin-test results for bovine tuberculosis-infected cattle by lesion count. CFT = caudal fold tuberculin, CCT = comparative cervical tuberculin, (N) = negative, (S) = suspect, (R) = reactor.
Table 1.
Bovine tuberculosis-infected herd characteristics.
Table 1.
Bovine tuberculosis-infected herd characteristics.
| Herd ID | Year of
Detection | Herd State(s) | Production Type | Breed | Herd Size | Total
Infected | Herd
Prevalence (%) | M. bovis Strain(s)/
Group | Likely Source of Introduction |
|---|
| B.1 | 2017 | South Dakota | Beef | Angus | 656 | 46 | 7.0% | 7B | Unknown |
| B.2 | 2017 | South Dakota | Beef | Angus | 338 | 63 * | 16.3% | 6A | Unknown |
| B.3 † | 2021 | Hawaii | Beef | Angus,
Corriente, Brahman | 1100 | 16 | 1.5% | 5A | Wildlife spillover |
| D.1 | 2013 | North Dakota | Dairy | Holstein | 400 | 3 | 0.8% | 8F | Human-to-cattle |
| D.2 | 2014 | Texas | Dairy | Holstein | 10,000 | 158 | 1.6% | 16C | Unknown |
| D.3 | 2018 | Wisconsin | Dairy | Holstein | 1500 | 12 | 0.8% | 17B3 | Human-to-cattle |
| D.4 | 2019 | New Mexico | Dairy | Holstein | 21,000 | 31 | 0.2% | 24 | Unknown |
| D.5 | 2019 | Texas | Dairy | Holstein | 9000 | 75 | 0.8% | 17B1, 13A, 6A | Unknown |
| D.6 | 2022 | New Mexico and Texas | Dairy | Holstein | 12,000 | 8 | 0.1% | 22 | Unknown |
Table 2.
Summary of lesions in bovine tuberculosis-infected cattle by production type.
Table 2.
Summary of lesions in bovine tuberculosis-infected cattle by production type.
Characteristic
(Number of Infected Cattle) | Beef Cattle
n = 118 | Dairy Cattle
n = 285 | p-Value | Total
n = 403 |
|---|
Age at necropsy (months) *,
mean ± SD | 64.8 ± 29.1 | 39.1 ± 22.4 | <0.001 | 46.6 ± 27.2 |
| Lesion Count | | | 0.104 | |
| No lesion present, n (%) | 6 (5.1) | 13 (4.6) | | 19 (4.7) |
| Any lesion present, n (%) | 112 (94.9) | 272 (95.4) | 384 (95.3) |
| Single site with lesion present, n (%) | 63 (53.4) | 184 (64.6) | 247 (61.3) |
Multiple sites with lesions
present, n (%) | 49 (41.5) | 88 (30.9) | 137 (34.0) |
| Lesion Location(s) ** | | | | |
| Any Head Lesions | 46 (38.9) | 121 (42.5) | 0.520 | 167 (41.4) |
| Any Thorax Lesions | 72 (61.0) | 197 (69.1) | 0.116 | 269 (66.7) |
| Any Body Lesions | 31 (26.3) | 46 (16.1) | 0.019 | 77 (19.1) |
| Head Lesion(s) Only | 21 (17.8) | 53 (18.6) | 0.850 | 74 (18.4) |
| Thorax Lesion(s) Only | 43 (36.4) | 127 (44.6) | 0.133 | 170 (42.4) |
| Body Lesion(s) Only | 14 (11.9) | 11 (3.9) | 0.002 | 25 (6.2) |
| Head and Thorax Lesions | 17 (14.4) | 46 (16.1) | 0.663 | 63 (15.6) |
| Head and Body Lesions | 5 (4.2) | 11 (3.9) | 0.860 | 16 (4.0) |
| Thorax and Body Lesions | 9 (7.6) | 13 (4.6) | 0.218 | 22 (5.5) |
| Head, Thorax, and Body Lesions | 3 (2.5) | 11 (3.9) | 0.511 | 14 (3.5) |
Table 3.
Lesioned tissues in lymph nodes (LN) and organs from bovine tuberculosis-infected cattle by production type *.
Table 3.
Lesioned tissues in lymph nodes (LN) and organs from bovine tuberculosis-infected cattle by production type *.
| Region | Tissue | Beef Cattle
(n = 108 *) | Dairy Cattle
(n = 99 *) | p-Value | Total
(n = 207) |
|---|
| Thorax | Cranial, Middle,
Caudal Mediastinal LN | 56 (51.9) | 42 (42.4) | 0.175 | 98 (47.3) |
| Thorax | Tracheobronchial LN | 27 (25.0) | 31 (31.3) | 0.312 | 58 (28.0) |
| Head | Medial Retropharyngeal LN | 24 (22.2) | 33 (33.3) | 0.074 | 57 (27.5) |
| Body | Mesenteric LN | 18 (16.7) | 13 (13.1) | 0.477 | 31 (15.0) |
| Head | Lateral Retropharyngeal LN | 14 (13.0) | 12 (12.1) | 0.855 | 26 (12.6) |
| Thorax | Lung | 13 (12.0) | 9 (9.1) | 0.492 | 22 (10.6) |
| Head | Mandibular LN | 11 (10.2) | 4 (4.0) | 0.089 | 15 (7.3) |
| Body | Hepatic LN | 12 (11.1) | 1 (1.0) | 0.003 | 13 (6.3) |
| Body | Prescapular LN | 4 (3.7) | 8 (8.1) | 0.237 | 12 (5.8) |
| Head | Parotid LN | 4 (3.7) | 6 (6.1) | 0.430 | 10 (4.8) |
| Body | Liver | 4 (3.7) | 3 (3.0) | 1.0 | 7 (3.4) |
| Body | Inguinal LN | 0 (0.0) | 4 (4.0) | 0.051 | 4 (1.9) |
| Body | Popliteal LN | 0 (0.0) | 3 (3.0) | 0.108 | 3 (1.5) |
| Body | Mammary LN | 0 (0.0) | 2 (2.0) | 0.228 | 2 (1.0) |
| Body | Axillary LN | 1 (0.9) | 0 (0.0) | 1.0 | 1 (0.5) |
Table 4.
Summary of lesion location for bovine tuberculosis-infected cattle by herd.
Table 4.
Summary of lesion location for bovine tuberculosis-infected cattle by herd.
Herd
(Number of Infected Cattle) | B.1
n = 46 | B.2
n = 56 | B.3
n = 16 | D.1
n = 3 | D.2
n = 158 | D.3
n = 12 | D.4
n = 31 | D.5
n = 73 | D.6
n = 8 |
|---|
Age at necropsy (months) *,
mean ± SD | 62.3 ±
14.8 | 74.5 ±
33.3 | 39.6 ±
30.9 | 38.3 ±
16.7 | 27.8 ±
19.1 | 58.3 ± 11.8 | 36.7 ±
14.9 | 57.5 ±
19.1 | 48.9 ±
4.6 |
| Lesion Count |
| None, n (%) | 1
(2.2) | 5
(8.9) | 0
(0.0) | 3
(100.0) | 0
(0.0) | 2
(16.7) | 7
(22.6) | 0
(0.0) | 1
(12.5) |
Single site,
n (%) | 23
(50.0) | 31
(55.4) | 9
(56.3) | 0
(0.0) | 110
(69.6) | 3
(25.0) | 15
(48.4) | 53
(72.6) | 3
(37.5) |
Multiple sites,
n (%) | 22
(47.8) | 20
(35.7) | 7
(43.8) | 0
(0.0) | 48
(30.4) | 7
(58.3) | 9
(29.0) | 20
(27.4) | 4
(50.0) |
| Lesion Location |
| No lesion present | 1
(2.2) | 5
(8.9) | 0
(0.0) | 3
(100.0) | 0
(0.0) | 2
(16.7) | 7
(22.6) | 0
(0.0) | 1
(12.5) |
| Head Lesion(s) Only | 6
(13.0) | 12
(21.4) | 3
(18.8) | 0
(0.0) | 29
(18.4) | 1
(8.3) | 10
(32.3) | 12
(16.4) | 1
(12.5) |
| Thorax Lesion(s) Only | 22
(47.8) | 17
(30.4) | 4
(25.0) | 0
(0.0) | 78
(49.4) | 1
(8.3) | 8
(25.8) | 35
(48.0) | 5
(62.5) |
| Body Lesion(s) Only | 7
(15.2) | 3
(5.4) | 4
(25.0) | 0
(0.0) | 5
(3.2) | 2
(16.7) | 1
(3.2) | 3
(4.1) | 0
(0.0) |
| Head and Thorax Lesions | 4
(8.7) | 10
(17.9) | 3
(18.8) | 0
(0.0) | 29
(18.4) | 1
(8.3) | 5
(16.1) | 10
(13.7) | 1
(12.5) |
| Head and Body Lesions | 2
(4.4) | 3
(5.4) | 0
(0.0) | 0
(0.0) | 7
(4.4) | 1
(8.3) | 0
(0.0) | 3
(4.1) | 0
(0.0) |
| Thorax and Body Lesions | 4
(8.7) | 3
(5.4) | 2
(12.5) | 0
(0.0) | 7
(4.4) | 2
(16.7) | 0
(0.0) | 4
(5.5) | 0
(0.0) |
Head, Thorax, and Body
Lesions | 0
(0.0) | 3
(5.4) | 0
(0.0) | 0
(0.0) | 3
(1.9) | 2
(16.7) | 0
(0.0) | 6
(8.2) | 0
(0.0) |
Table 5.
Antemortem skin-test results * for bovine tuberculosis-infected cattle by production type and herd.
Table 5.
Antemortem skin-test results * for bovine tuberculosis-infected cattle by production type and herd.
| Test Type | Caudal Fold Tuberculin (CFT) Test * (n = 376) | Comparative Cervical Tuberculin (CCT) Test * (n = 275) |
|---|
Test
Result | Negative | Suspect | p-value | Negative | Suspect | Reactor | p-value ‡ |
All
infected animals, n = 403 | 34
(9.0) | 342
(91.0) | | 105
(38.2) | 84
(30.6) | 86
(31.3) | |
| Production Type | 0.510 | | 0.003 |
| Beef, n = 118 | 7
(7.4) | 88
(92.6) | | 8
(18.2) | 12
(27.3) | 24
(54.5) | |
| Dairy, n = 285 | 27
(9.6) | 254
(90.4) | | 97
(42.0) | 72
(31.2) | 62
(26.8) | |
| Herd |
| B.1, n = 46 | 6
(14.3) | 36
(85.7) | | 8
(21.0) | 6
(15.8) | 24
(63.2) | |
| B.2, n = 56 | 0(0.0) | 46
(100.0) | | NP | NP | NP | |
| B.3 n = 16 | 1
(14.3) | 6
(85.7) | | 0
(0.0) | 0
(0.0) | 6
(100.0) | |
| D.1, n = 3 | 0
(0.0) | 3
(100.0) | | NP | NP | NP | |
| D.2, n = 158 | 22
(14.1) | 134
(85.9) | | 67
(58.8) | 35
(30.7) | 12
(10.5) | |
| D.3, n = 12 | 2
(18.2) | 9
(81.8) | | 3
(33.3) | 4
(44.5) | 2
(22.2) | |
| D.4, n = 31 | 2
(6.7) | 28
(93.3) | | 5
(17.8) | 12
(42.9) | 11
(39.3) | |
| D.5, n = 73 | 1
(1.4) | 72
(98.6) | | 19
(26.0) | 18
(24.7) | 36
(49.3) | |
| D.6, n = 8 | 0
(0.0) | 8
(100.0) | | 3
(42.9) | 3
(42.9) | 1
(14.2) | |
Table 6.
Antemortem skin-test results for bovine tuberculosis-infected cattle by lesion count and location. * Antemortem test results included are those most proximate to the infected animal’s slaughter/necropsy. ‡ p-values for CCT results were calculated based on dichotomous results where CCT(N) = negative and CCT(S) or CCT(R) = positive due to low counts and herd removal practices.
Table 6.
Antemortem skin-test results for bovine tuberculosis-infected cattle by lesion count and location. * Antemortem test results included are those most proximate to the infected animal’s slaughter/necropsy. ‡ p-values for CCT results were calculated based on dichotomous results where CCT(N) = negative and CCT(S) or CCT(R) = positive due to low counts and herd removal practices.
| Test Type | Caudal Fold Tuberculin (CFT) Test *
(n = 376) | Comparative Cervical Tuberculin (CCT) Test * (n = 275) |
|---|
| Test Result | Negative | Suspect | p-value | Negative | Suspect | Reactor | p-value ‡ |
| Lesion Count | 0.929 | | 0.188 |
| None, n (%) | 2
(10.5) | 17
(89.5) | | 2
(22.2) | 4
(44.4) | 3
(33.3) | |
| Single site, n (%) | 20
(8.6) | 212 (91.4) | 75
(41.9) | 57 (31.8) | 47
(26.3) |
| Multiple sites, n (%) | 12
(9.6) | 113 (90.4) | 28
(32.2) | 23 (26.4) | 36
(41.4) |
| Lesion Location |
| No lesion present | 2
(10.5) | 17
(89.5) | 0.686 | 2
(22.2) | 4
(44.5) | 3
(33.3) | 0.490 |
| Head Lesion(s) Only | 6
(8.7) | 63
(91.3) | 1.00 | 20
(40.0) | 20
(40.0) | 10
(20.0) | 0.872 |
Thorax Lesion(s)
Only | 11
(6.8) | 151
(93.2) | 0.207 | 49
(38.6) | 47
(29.1) | 41
(32.3) | 0.902 |
| Body Lesion(s) Only | 5
(14.7) | 18
(78.3) | 0.046 | 6
(40.0) | 4
(26.7) | 5
(33.3) | 1.00 |
| Head and Thorax Lesions | 6
(10.0) | 54
(90.0) | 0.806
| 18
(42.9) | 8
(19.0) | 16
(38.1) | 0.496 |
| Head and Body Lesions | 2
(14.3) | 12
(85.7) | 0.367
| 3
(30.0) | 2
(20.0) | 5
(50.0) | 0.746 |
| Thorax and Body Lesions | 1
(5.6) | 17
(94.4) | 1.00
| 2
(16.7) | 4
(33.3) | 6
(50.0) | 0.140 |
| Head, Thorax, and Body Lesions | 1
(9.1) | 10
(90.9) | 1.00 | 5
(50.0) | 5
(50.0) | 0(0.0) | 01.53 |
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |