Classification and Anti-Streptococcus mutans Mechanism Summary of Chinese Botanical Products
Abstract
1. Introduction
2. Classifications of Anti-S. mutans CBPs
2.1. Organic Acid-Based CBPs
2.2. Alkaloid-Based CBPs
2.3. Phenol-Based CBPs
2.4. Anthraquinone-Based CBPs
2.5. Other Types
3. Mechanisms of Anti-Caries CBPs
3.1. Biofilm and Insoluble Glucans Synthesis
3.2. Energy and Soluble Glucans Synthesis
3.3. Acidogenicity/Aciduricity
3.4. Cell Integrity and Other Metabolisms
3.5. Demineralization Inhibition and Remineralization Promotion
3.6. Interference with Quorum Sensing
4. Clinical Applications and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Abbreviations | Full name |
| CBP | Chinese botanical product |
| EPS | Extracellular polysaccharides |
| GC | Galla Chinensis |
| GA | Gallic acid |
| MG | Methyl gallate |
| Gtfs | Glucosyltransferases |
| GtfB/C | Glucosyltransferase B/C |
| GtfD | Glucosyltransferase D |
| QS | Quorum sensing |
| MIC | Minimal inhibitory concentration |
| MBC | Minimal bactericidal concentration |
| TP/TPs | Tea polyphenols |
| EGC | Epigallocatechin |
| EGCG | Epigallocatechin gallate |
| PEOME | Propolis essential oil microemulsion |
| CAPE | Caffeic acid phenethyl ester |
| CEO | Clove essential oil |
| PEO | Peppermint essential oil |
| SEM | Scanning electron microscopy |
| TEM | Transmission electron microscopy |
| PEP-PTS | Phosphoenolpyruvate–carbohydrate phosphotransferase system |
| TCA | Tricarboxylic acid |
| GTase | Guanylyl transferase |
| Ftf | Fructosyltransferase |
| Gbp | Glucan binding protein |
| OTE | Oolong tea extract |
| OTF10 | Oolong tea polyphenol 10 |
| AI-2 | Autoinducer-2 |
| LDH | Lactate dehydrogenase |
| AFM | Atomic force microscope |
| XRD | X-ray diffraction |
| SMH | Surface microhardness |
| %SMHR | Surface microhardness recovery |
| CHX | Chlorhexidine |
| HA | Hydroxyapatite |
| RCTs | Randomized controlled trials |
References
- Chen, D.R.; Lin, H.C. Research Updates: Cariogenic Mechanism of Streptococcus mutans. J. Sichuan Univ. (Med. Sci. Ed.) 2022, 53, 208–213. [Google Scholar]
- Lowe, H.; Toyang, N.; Steele, B.; Bryant, J.; Ngwa, W.; Nedamat, K. The Current and Potential Application of Medicinal Cannabis Products in Dentistry. Dent. J. 2021, 9, 106. [Google Scholar] [CrossRef] [PubMed]
- Liljemark, W.F.; Bloomquist, C. Human oral microbial ecology and dental caries and periodontal diseases. Crit. Rev. Oral Biol. Med. 1996, 7, 180–198. [Google Scholar] [CrossRef]
- Zhou, F.; Mu, X.; Li, Z.; Guo, M.; Wang, J.; Long, P.; Wan, Y.; Yuan, T.; Lv, Y. Characteristics of Chinese herbal medicine mouthwash clinical studies: A bibliometric and content analysis. J. Ethnopharmacol. 2023, 307, 116210. [Google Scholar] [CrossRef] [PubMed]
- Amrutha, B.; Sundar, K.; Shetty, P.H. Effect of organic acids on biofilm formation and quorum signaling of pathogens from fresh fruits and vegetables. Microb. Pathog. 2017, 111, 156–162. [Google Scholar] [CrossRef]
- Kim, T.; Silva, J.; Kim, M.; Jung, Y. Enhanced antioxidant capacity and antimicrobial activity of tannic acid by thermal processing. Food Chem. 2010, 118, 740–746. [Google Scholar] [CrossRef]
- Abdel-Azem, H.M.; Elezz, A.F.A.; Safy, R.K. Effect of Galla chinensis on Remineralization of Early Dentin Lesion. Eur. J. Dent. 2020, 14, 651–656. [Google Scholar] [CrossRef]
- Zhang, T.; Chu, J.; Zhou, X. Anti-carious Effects of Galla chinensis: A Systematic Review. Phytother. Res. 2015, 29, 1837–1842. [Google Scholar] [CrossRef]
- Ren, Y.-Y.; Zhang, X.-R.; Li, T.-N.; Zeng, Y.-J.; Wang, J.; Huang, Q.-W. Galla Chinensis, a Traditional Chinese Medicine: Comprehensive review of botany, traditional uses, chemical composition, pharmacology and toxicology. J. Ethnopharmacol. 2021, 278, 114247. [Google Scholar] [CrossRef]
- Djakpo, O.; Yao, W. Rhus chinensis and Galla Chinensis—Folklore to modern evidence: Review. Phytother. Res. 2010, 24, 1739–1747. [Google Scholar] [CrossRef]
- Wu-Yuan, C.; Chen, C.; Wu, R. Gallotannins Inhibit Growth, Water-insoluble Glucan Synthesis, and Aggregation of Mutans Streptococci. J. Dent. Res. 1988, 67, 51–55. [Google Scholar] [CrossRef]
- Tian, F.; Li, B.; Ji, B.; Zhang, G.; Luo, Y. Identification and structure–activity relationship of gallotannins separated from Galla chinensis. LWT-Food Sci. Technol. 2009, 42, 1289–1295. [Google Scholar] [CrossRef]
- Xie, Q.; Li, J.; Zhou, X. Anticaries effect of compounds extracted from Galla Chinensis in a multispecies biofilm model. Oral Microbiol. Immunol. 2008, 23, 459–465. [Google Scholar] [CrossRef]
- Kang, M.-S.; Oh, J.-S.; Kang, I.-C.; Hong, S.-J.; Choi, C.-H. Inhibitory effect of methyl gallate and gallic acid on oral bacteria. J. Microbiol. 2008, 46, 744–750. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Kang, Q.; Zhang, Y.; Chen, M.; Wang, Z.; Wu, Y.; Gao, H.; Zhong, Z.; Tan, W. Glycyrrhizae Radix et Rhizoma: The popular occurrence of herbal medicine applied in classical prescriptions. Phytother. Res. 2023, 37, 3135–3160. [Google Scholar] [CrossRef] [PubMed]
- Rahnama, M.; Mehrabani, D.; Japoni, S.; Edjtehadi, M.; Firoozi, M.S. The healing effect of licorice (Glycyrrhiza glabra) on Helicobacter pylori infected peptic ulcers. J. Res. Med. Sci. Off. J. Isfahan Univ. Med. Sci. 2013, 18, 532–533. [Google Scholar]
- AlDehlawi, H.; Jazzar, A. The Power of Licorice (Radix glycyrrhizae) to Improve Oral Health: A Comprehensive Review of Its Pharmacological Properties and Clinical Implications. Healthcare 2023, 11, 2887. [Google Scholar] [CrossRef]
- Tharakan, A.; Pawar, M.; Kale, S. Effectiveness of licorice in preventing dental caries in children: A systematic review. J. Indian Soc. Pedod. Prev. Dent. 2020, 38, 325–331. [Google Scholar] [CrossRef]
- Sun, N.; Huang, L.-Y.; Yang, S.; Li, J.; Hou, C.-Z.; Liu, Z.-H. Progress on the mining of functional genes of Lonicera japonica. Yi Chuan 2024, 46, 920–936. [Google Scholar]
- Wang, L.; Liu, P.; Wu, Y.; Pei, H.; Cao, X. Inhibitory effect of Lonicera japonica flos on Streptococcus mutans biofilm and mechanism exploration through metabolomic and transcriptomic analyses. Front. Microbiol. 2024, 15, 1435503. [Google Scholar] [CrossRef]
- Bailly, C. Anticancer properties of Prunus mume extracts (Chinese plum, Japanese apricot). J. Ethnopharmacol. 2020, 246, 112215. [Google Scholar] [CrossRef]
- Seneviratne, C.J.; Wong, R.W.K.; Hägg, U.; Chen, Y.; Herath, T.D.K.; Samaranayake, P.L.; Kao, R. Prunus mume extract exhibits antimicrobial activity against pathogenic oral bacteria. Int. J. Paediatr. Dent. 2011, 21, 299–305. [Google Scholar] [CrossRef] [PubMed]
- Zou, Q.; Huang, Y.; Zhang, W.; Lu, C.; Yuan, J. A Comprehensive Review of the Pharmacology, Chemistry, Traditional Uses and Quality Control of Star Anise (Illicium verum Hook. F.): An Aromatic Medicinal Plant. Molecules 2023, 28, 7378. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.; Xiu, Q.; Sun, J.; Hong, E. Anti-platelet and anti-thrombotic effects of triacetylshikimic acid in rats. J. Cardiovasc. Pharmacol. 2002, 39, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Yang, Y.; Sun, Q.; Zeng, W.; Li, Y. Inhibition of Biofilm Formation and Virulence Factors of Cariogenic Oral Pathogen Streptococcus mutans by Shikimic Acid. Microbiol. Spectr. 2022, 10, e0119922. [Google Scholar] [CrossRef] [PubMed]
- Rani, N.; Singh, R.; Kumar, P.; Sharma, P.; Kaur, R.; Arora, R.; Singh, T.G. Alkaloids as Potential Anti-HIV Agents. Curr. HIV Res. 2023, 21, 240–247. [Google Scholar] [CrossRef]
- Zeng, C.; Jiang, W.; Liu, C.; Yu, R.; Li, Y.; Li, P.; Cao, Y. Inhibitory effects of berberine against Streptococcus mutans: An in vitro insight on its anticaries potential. Microbiol. Spectr. 2025, 13, e0070025. [Google Scholar] [CrossRef]
- Zhou, Y.; Liu, Z.; Wen, J.; Lin, H. The inhibitory effect of berberine chloride hydrate on Streptococcus mutans biofilm formation at different pH values. Microbiol. Spectr. 2023, 11, e0217023. [Google Scholar] [CrossRef]
- Fang, S.; Guo, S.; Du, S.; Cao, Z.; Yang, Y.; Su, X.; Wei, W. Efficacy and safety of berberine in preventing recurrence of colorectal adenomas: A systematic review and meta-analysis. J. Ethnopharmacol. 2022, 282, 114617. [Google Scholar] [CrossRef]
- Di Pierro, F.; Bertuccioli, A.; Giuberti, R.; Saponara, M.; Ivaldi, L. Role of a berberine-based nutritional supplement in reducing diarrhea in subjects with functional gastrointestinal disorders. Minerva Gastroenterol. E Dietol. 2020, 66, 29–34. [Google Scholar] [CrossRef]
- Zielińska, S.; Wójciak-Kosior, M.; Dziągwa-Becker, M.; Gleńsk, M.; Sowa, I.; Fijałkowski, K.; Rurańska-Smutnicka, D.; Matkowski, A.; Junka, A. The Activity of Isoquinoline Alkaloids and Extracts from Chelidonium majus against Pathogenic Bacteria and Candida sp. Toxins 2019, 11, 406. [Google Scholar] [CrossRef] [PubMed]
- Gerenčer, M.; Turecek, P.L.; Kistner, O.; Mitterer, A.; Savidis-Dacho, H.; Barrett, N.P. In vitro and in vivo anti-retroviral activity of the substance purified from the aqueous extract of Chelidonium majus L. Antivir. Res. 2006, 72, 153–156. [Google Scholar] [CrossRef] [PubMed]
- Gilca, M.; Gaman, L.; Panait, E.; Stoian, I.; Atanasiu, V. Chelidonium majus—An Integrative Review: Traditional Knowledge versus Modern Findings. Forsch. Komplementarmedizin 2010, 17, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.-B.; Chen, X.; Liu, S.-J.; Zhang, X.-F.; Zhang, G.-H. Experimental study of the inhibitory effects of Chelidonium majus L. extractive on Streptococcus mutans in vitro. Shanghai Kou Qiang Yi Xue = Shanghai J. Stomatol. 2006, 15, 318–320. [Google Scholar]
- Sun, P.; Zhao, W.; Wang, Q.; Chen, L.; Sun, K.; Zhan, Z.; Wang, J. Chemical diversity, biological activities and Traditional uses of and important Chinese herb Sophora. Phytomedicine Int. J. Phytother. Phytopharm. 2022, 100, 154054. [Google Scholar] [CrossRef]
- Sun, J.; Wang, S.; Zhao, Z.; Lu, J.; Zhang, Y.; An, W.; Li, W.; Yang, L.; Tong, X. Oxymatrine Attenuates Ulcerative Colitis through Inhibiting Pyroptosis Mediated by the NLRP3 Inflammasome. Molecules 2024, 29, 2897. [Google Scholar] [CrossRef]
- Kim, C.S.; Park, S.-N.; Ahn, S.-J.; Seo, Y.-W.; Lee, Y.-J.; Lim, Y.K.; Freire, M.O.; Cho, E.; Kook, J.-K. Antimicrobial effect of sophoraflavanone G isolated from Sophora flavescens against mutans streptococci. Anaerobe 2013, 19, 17–21. [Google Scholar] [CrossRef]
- Lee, S.-H. Antimicrobial effects of herbal extracts on Streptococcus mutans and normal oral streptococci. J. Microbiol. 2013, 51, 484–489. [Google Scholar] [CrossRef]
- Sakaue, Y.; Domon, H.; Oda, M.; Takenaka, S.; Kubo, M.; Fukuyama, Y.; Okiji, T.; Terao, Y. Anti-biofilm and bactericidal effects of magnolia bark-derived magnolol and honokiol on Streptococcus mutans. Microbiol. Immunol. 2016, 60, 10–16. [Google Scholar] [CrossRef]
- Lai, X.; Li, Y.; Lan, W.; Zhao, L.; Wang, K.; Hu, Z.; Liu, X. Interactions between proteins and polyphenols in plant-based food: Insight of allergenicity and off-flavor reduction. Food Chem. 2025, 495, 146333. [Google Scholar] [CrossRef]
- Shahidi, F.; Athiyappan, K.D. Polyphenol-polysaccharide interactions: Molecular mechanisms and potential applications in food systems—A comprehensive review. Food Prod. Process. Nutr. 2025, 7, 1–41. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.; Murata, R.; Duarte, S. Antimicrobial Traits of Tea- and Cranberry-Derived Polyphenols against Streptococcus mutans. Caries Res. 2011, 45, 327–335. [Google Scholar] [CrossRef]
- Bai, H.; Liu, T.; Wang, H.; Wang, Z. Antibacterial characteristics and mechanistic insights of combined tea polyphenols, Nisin, and epsilon-polylysine against feline oral pathogens: A comprehensive transcriptomic and metabolomic analysis. J. Appl. Microbiol. 2024, 135, lxae189. [Google Scholar] [CrossRef] [PubMed]
- Mazur, M.; Ndokaj, A.; Jedlinski, M.; Ardan, R.; Bietolini, S.; Ottolenghi, L. Impact of Green Tea (Camellia Sinensis) on periodontitis and caries. Systematic review and meta-analysis. Jpn. Dent. Sci. Rev. 2021, 57, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Liu, Q. Demystifying phytoconstituent-derived nanomedicines in their immunoregulatory and therapeutic roles in inflammatory diseases. Adv. Drug Deliv. Rev. 2022, 186, 114317. [Google Scholar] [CrossRef]
- Gaur, S.; Agnihotri, R. Green tea: A novel functional food for the oral health of older adults. Geriatr. Gerontol. Int. 2014, 14, 238–250. [Google Scholar] [CrossRef]
- Musial, C.; Kuban-Jankowska, A.; Gorska-Ponikowska, M. Beneficial Properties of Green Tea Catechins. Int. J. Mol. Sci. 2020, 21, 1744. [Google Scholar] [CrossRef]
- Linke, H.A.; LeGeros, R.Z. Black tea extract and dental caries formation in hamsters. Int.-Al J. Food Sci. Nutr. 2003, 54, 89–95. [Google Scholar] [CrossRef]
- Otake, S.; Makimura, M.; Kuroki, T.; Nishihara, Y.; Hirasawa, M. Anticaries Effects of Polyphenolic Compounds from Japanese Green Tea. Caries Res. 1991, 25, 438–443. [Google Scholar] [CrossRef]
- Sasaki, H.; Matsumoto, M.; Tanaka, T.; Maeda, M.; Nakai, M.; Hamada, S.; Ooshima, T. Antibacterial Activity of Polyphenol Components in Oolong Tea Extract against Streptococcus mutans. Caries Res. 2004, 38, 2–8. [Google Scholar] [CrossRef]
- Moreno, A.P.D.; Marcato, P.D.; Silva, L.B.; de Souza Salvador, S.L.; Del Arco, M.C.G.; de Moraes, J.C.B.; Rossi, A. Antibacterial Activity of Epigallocatechin-3-gallate (EGCG) Loaded Lipid-chitosan Hybrid Nanoparticle against Planktonic Microorganisms. J. Oleo Sci. 2024, 73, 709–716. [Google Scholar] [CrossRef]
- Balasubramaniam, A.K.; Elangovan, A.; Rahman, M.A.; Nayak, S.; Swain, D.; Babu, H.P.; Narasimhan, A.; Monga, V. Propolis: A comprehensive review on the nature’s polyphenolic wonder. Fitoterapia 2025, 183, 106526. [Google Scholar] [CrossRef] [PubMed]
- Przybyłek, I.; Karpiński, T.M. Antibacterial Properties of Propolis. Molecules 2019, 24, 2047. [Google Scholar] [CrossRef] [PubMed]
- Assis, M.A.d.S.; Ramos, L.d.P.; Abu Hasna, A.; de Queiroz, T.S.; Pereira, T.C.; de Lima, P.M.N.; Berretta, A.A.; Marcucci, M.C.; Carvalho, C.A.T.; de Oliveira, L.D. Antimicrobial and Antibiofilm Effect of Brazilian Green Propolis Aqueous Extract against Dental Anaerobic Bacteria. Molecules 2022, 27, 8128. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Yuan, W.; Guo, Y.; Wu, Q.; Wang, F.; Xuan, H. Anti-Biofilm Activities of Chinese Poplar Propolis Essential Oil against Streptococcus mutans. Nutrients 2022, 14, 3290. [Google Scholar] [CrossRef]
- Wang, F.; Yuan, J.; Wang, X.; Xuan, H. Antibacterial and antibiofilm activities of Chinese propolis essential oil microemulsion against Streptococcus mutans. J. Appl. Microbiol. 2023, 134, lxad056. [Google Scholar] [CrossRef]
- Alghutaimel, H.; Matoug-Elwerfelli, M.; Alhaji, M.; Albawardi, F.; Nagendrababu, V.; Dummer, P.M.H. Propolis Use in Dentistry: A Narrative Review of Its Preventive and Therapeutic Applications. Int. Dent. J. 2024, 74, 365–386. [Google Scholar] [CrossRef]
- Liñán-Atero, R.; Aghababaei, F.; García, S.R.; Hasiri, Z.; Ziogkas, D.; Moreno, A.; Hadidi, M. Clove Essential Oil: Chemical Profile, Biological Activities, Encapsulation Strategies, and Food Applications. Antioxidants 2024, 13, 488. [Google Scholar] [CrossRef]
- Sugihartini, N.; Prabandari, R.; Yuwono, T.; Rahmawati, D.R. The anti-inflammatory activity of essential oil of clove (Syzygium aromaticum) in absorption base ointment with addition of oleic acid and propylene glycol as enhancer. Int. J. Appl. Pharm. 2019, 11, 106–109. [Google Scholar] [CrossRef]
- Behbahani, B.A.; Noshad, M.; Falah, F. Study of chemical structure, antimicrobial, cytotoxic and mechanism of action of Syzygium aromaticum essential oil on foodborne pathogens. Potravin. Slovak J. Food Sci. 2019, 13, 875–883. [Google Scholar] [CrossRef]
- Haro-González, J.N.; Castillo-Herrera, G.A.; Martínez-Velázquez, M.; Espinosa-Andrews, H. Clove Essential Oil (Syzygium aromaticum L. Myrtaceae): Extraction, Chemical Composition, Food Applications, and Essential Bioactivity for Human Health. Molecules 2021, 26, 6387. [Google Scholar] [CrossRef] [PubMed]
- Elgamily, H.; Safy, R.; Makharita, R. Influence of Medicinal Plant Extracts on the Growth of Oral Pathogens Streptococcus mutans and Lactobacillus Acidophilus: An In-Vitro Study. Open Access Maced. J. Med. Sci. 2019, 7, 2328–2334. [Google Scholar] [CrossRef] [PubMed]
- Niu, L.; Hou, Y.; Jiang, M.; Bai, G. The rich pharmacological activities of Magnolia officinalis and secondary effects based on significant intestinal contributions. J. Ethnopharmacol. 2021, 281, 114524. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; Li, H.; Ma, R.; Ren, M.; Li, Y.; Li, J.; Chen, H.; Chen, Z.; Gong, D.; Wang, J. Effect of Coptis chinensis franch and Magnolia officinalis on intestinal flora and intestinal barrier in a TNBS-induced ulcerative colitis rats model. Phytomedicine Int. J. Phytother. Phyto-Pharmacol. 2022, 97, 153927. [Google Scholar] [CrossRef]
- Huang, B.; Fan, M.; Wang, S.; Han, D.; Chen, Z.; Bian, Z. The inhibitory effect of magnolol from Magnolia officinalis on glucosyltransferase. Arch. Oral Biol. 2006, 51, 899–905. [Google Scholar] [CrossRef]
- Ghorbani, F.; Haghgoo, R.; Aramjoo, H.; Rakhshandeh, H.; Jamehdar, S.A.; Zare-Bidaki, M. The antibacterial effect of Magnolia mouthwash on the levels of salivary Streptococcus mutans in dental plaque: A randomized, single-blind, placebo-controlled trial. Iran. J. Microbiol. 2021, 13, 104–111. [Google Scholar] [CrossRef]
- Wang, P.; Wei, J.; Hua, X.; Dong, G.; Dziedzic, K.; Wahab, A.; Efferth, T.; Sun, W.; Ma, P. Plant anthraquinones: Classification, distribution, biosynthesis, and regulation. J. Cell. Physiol. 2023, 239, e31063. [Google Scholar] [CrossRef]
- Qun, T.; Zhou, T.; Hao, J.; Wang, C.; Zhang, K.; Xu, J.; Wang, X.; Zhou, W. Antibacterial activities of anthraquinones: Structure–activity relationships and action mechanisms. RSC Med. Chem. 2023, 14, 1446–1471. [Google Scholar] [CrossRef]
- Catalano, A.; Ceramella, J.; Iacopetta, D.; Marra, M.; Conforti, F.; Lupi, F.R.; Gabriele, D.; Borges, F.; Sinicropi, M.S. Aloe vera―An Extensive Review Focused on Recent Studies. Foods 2024, 13, 2155. [Google Scholar] [CrossRef]
- Sánchez, M.; González-Burgos, E.; Iglesias, I.; Gómez-Serranillos, M.P. Pharmacological Update Properties of Aloe Vera and its Major Active Constituents. Molecules 2020, 25, 1324. [Google Scholar] [CrossRef]
- Al-Shaibani, M.; Al-Saffar, M.; Mahmood, A. The impact of aloe vera gel on remineralization of the tooth and its effect against enterococcus faecalis: An in vitro study. Georgian Med. News 2023, 338, 63–68. [Google Scholar]
- Al-Abdullah, A.; Edris, S.; Abu Hasna, A.; de Carvalho, L.S.; Al-Nahlawi, T. The Effect of Aloe vera and Chlorhexidine as Disinfectants on the Success of Selective Caries Removal Technique: A Randomized Controlled Trial. Int. J. Dent. 2022, 2022, 9474677. [Google Scholar] [CrossRef] [PubMed]
- Lai, J.-Y.; Fan, X.-L.; Zhang, H.-B.; Wang, S.-C.; Wang, H.; Ma, X.; Zhang, Z.-Q. Polygonum cuspidatum polysaccharide: A review of its extraction and purification, structure analysis, and biological activity. J. Ethnopharmacol. 2024, 331, 118079. [Google Scholar] [CrossRef] [PubMed]
- Lachowicz, S.; Oszmiański, J. Profile of Bioactive Compounds in the Morphological Parts of Wild Fallopia japonica (Houtt) and Fallopia sachalinensis (F. Schmidt) and Their Antioxidative Activity. Molecules 2019, 24, 1436. [Google Scholar] [CrossRef]
- Kwon, Y.-R.; Son, K.-J.; Pandit, S.; Kim, J.-E.; Chang, K.-W.; Jeon, J.-G. Bioactivity-guided separation of anti-acidogenic substances against Streptococcus mutans UA 159 from Polygonum cuspidatum. Oral Dis. 2010, 16, 204–209. [Google Scholar] [CrossRef] [PubMed]
- Bastos, C.F.B.; Gomes-Filho, F.N.; Borges-Grisi, M.H.S.; D’aSsunção, V.C.S.C.; Barros, A.B.C.; Xavier-Júnior, F.H.; Almeida, L.F.D. Antibacterial effect of cinnamaldehyde in a microemulsion system against oral colonizing biofilms. Braz. J. Biol. 2025, 85, e294221. [Google Scholar] [CrossRef]
- Guo, J.; Yan, S.; Jiang, X.; Su, Z.; Zhang, F.; Xie, J.; Hao, E.; Yao, C. Advances in pharmacological effects and mechanism of action of cinnamaldehyde. Front. Pharmacol. 2024, 15, 1365949. [Google Scholar] [CrossRef]
- Aljaafari, M.N.; AlAli, A.O.; Baqais, L.; Alqubaisy, M.; AlAli, M.; Molouki, A.; Ong-Abdullah, J.; Abushelaibi, A.; Lai, K.-S.; Lim, S.-H.E. An Overview of the Potential Therapeutic Applications of Essential Oils. Molecules 2021, 26, 628. [Google Scholar] [CrossRef]
- Herro, E.; Jacob, S.E. Mentha piperita (Peppermint). Dermatitis 2010, 21, 327–329. [Google Scholar] [CrossRef]
- Mahendran, G.; Rahman, L.U. Ethnomedicinal, phytochemical and pharmacological updates on Peppermint (Mentha × piperita L.)—A review. Phytother. Res. 2020, 34, 2088–2139. [Google Scholar] [CrossRef]
- Zhao, H.; Ren, S.; Yang, H.; Tang, S.; Guo, C.; Liu, M.; Tao, Q.; Ming, T.; Xu, H. Peppermint essential oil: Its phytochemistry, biological activity, pharmacological effect and application. Biomed. Pharmacother. 2022, 154, 113559. [Google Scholar] [CrossRef]
- Acevedo, A.M.; Montero, M.; Rojas-Sanchez, F.; Machado, C.; Rivera, L.E.; Wolff, M.; Kleinberg, I. Clinical evaluation of the ability of CaviStat in a mint confection to inhibit the development of dental caries in children. J. Clin. Dent. 2008, 19, 1–8. [Google Scholar] [PubMed]
- Braga, A.S.; Girotti, L.D.; de Melo Simas, L.L.; Pires, J.G.; Pela, V.T.; Buzalaf, M.A.R.; Magalhaes, A.C. Effect of commercial herbal toothpastes and mouth rinses on the prevention of enamel deminerali-zation using a microcosm biofilm model. Biofouling 2019, 35, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Farias, J.M.; Stamford, T.C.M.; Resende, A.H.M.; Aguiar, J.S.; Rufino, R.D.; Luna, J.M.; Sarubbo, L.A. Mouthwash containing a biosurfactant and chitosan: An eco-sustainable option for the control of cariogenic microorganisms. Int. J. Biol. Macromol. 2019, 129, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Cwik, J.; Gonzalez, L.A.; Shi, X.; Spirgel, C.C.; Yankell, S. Plaque reduction and tensile strength evaluations of three dental floss products. Am. J. Dent. 2021, 34, 123–126. [Google Scholar]
- Spence, C. Cinnamon: The historic spice, medicinal uses, and flavour chemistry. Int. J. Gastron. Food Sci. 2023, 35, 100858. [Google Scholar] [CrossRef]
- Guo, J.; Jiang, X.; Tian, Y.; Yan, S.; Liu, J.; Xie, J.; Zhang, F.; Yao, C.; Hao, E. Therapeutic Potential of Cinnamon Oil: Chemical Composition, Pharmacological Actions, and Applications. Pharmaceuticals 2024, 17, 1700. [Google Scholar] [CrossRef]
- Shu, C.; Ge, L.; Li, Z.; Chen, B.; Liao, S.; Lu, L.; Wu, Q.; Jiang, X.; An, Y.; Wang, Z.; et al. Antibacterial activity of cinnamon essential oil and its main component of cinnamaldehyde and the underlying mechanism. Front. Pharmacol. 2024, 15, 1378434. [Google Scholar] [CrossRef]
- Ray, R.R. Dental biofilm: Risks, diagnostics and management. Biocatal. Agric. Biotechnol. 2022, 43, 102381. [Google Scholar] [CrossRef]
- Huang, R.; Li, M.; Gregory, R.L. Bacterial interactions in dental biofilm. Virulence 2011, 2, 435–444. [Google Scholar] [CrossRef]
- Schormann, N.; Patel, M.; Thannickal, L.; Purushotham, S.; Wu, R.; Mieher, J.L.; Wu, H.; Deivanayagam, C. The catalytic domains of Streptococcus mutans glucosyltransferases: A structural analysis. Acta Crystallogr. Sect. F Struct. Biol. Commun. 2023, 79, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Saharan, B.S.; Beniwal, N.; Duhan, J.S. From formulation to function: A detailed review of microbial biofilms and their polymer-based extracellular substances. Microbe 2024, 5, 100194. [Google Scholar] [CrossRef]
- Zhao, J.; Li, J.Y.; Zhu, B.; Zhou, X.D. Effects of traditional Chinese medicine on oral bacteria bio-film. Zhonghua Kou Qiang Yi Xue Za Zhi = Zhonghua Kouqiang Yixue Zazhi = Chin. J. Stomatol. 2007, 42, 585–589. [Google Scholar]
- Edgar, W. Reduction in Enamel Dissolution by Liquorice and Glycyrrhizinic Acid. J. Dent. Res. 1978, 57, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Segal, R.; Pisanty, S.; Wormser, R.; Azaz, E.; Sela, M. Anticariogenic Activity of Licorice and Glycyrrhizine I: Inhibition of In Vitro Plaque Formation by Streptococcus mutans. J. Pharm. Sci. 1985, 74, 79–81. [Google Scholar] [CrossRef]
- Ham, Y.; Kim, T.-J. Synergistic inhibitory activity of Glycyrrhizae Radix and Rubi Fructus extracts on biofilm formation of Streptococcus mutans. BMC Complement. Med. Ther. 2023, 23, 22. [Google Scholar] [CrossRef]
- Cheng, R.-B.; Chen, X.; Liu, S.-J.; Zhang, X.-F. Effect of Chelerythrine on cell surface hydrophobicity and adherence of Streptococcus mutans. Shanghai Kou Qiang Yi Xue = Shanghai J. Stomatol. 2007, 16, 68–72. [Google Scholar]
- Hattarki, S.A.; Bogar, C.; Bhat, K.G. Green Tea Catechins showed Antibacterial Activity on Streptococcus mutans—An In Vitro Study. Indian J. Dent. Res. 2021, 32, 226–229. [Google Scholar] [CrossRef]
- Goto, I.; Saga, S.; Ichitani, M.; Kimijima, M.; Narisawa, N. Investigation of Components in Roasted Green Tea That Inhibit Streptococcus mutans Biofilm Formation. Foods 2023, 12, 2502. [Google Scholar] [CrossRef]
- Subramaniam, P.; Eswara, U.; Reddy, K.M. Effect of different types of tea on Streptococcus mutans: An in vitro study. Indian J. Dent. Res. 2012, 23, 43–48. [Google Scholar] [CrossRef]
- Uju, D.E.; Obioma, N.P. Anticariogenic potentials of clove, tobacco and bitter kola. Asian Pac. J. Trop. Med. 2011, 4, 814–818. [Google Scholar] [CrossRef]
- Fani, M.; Kohanteb, J. Inhibitory activity of Aloe vera gel on some clinically isolated cariogenic and periodontopathic bacteria. J. Oral Sci. 2012, 54, 15–21. [Google Scholar] [CrossRef]
- Xu, J.-S.; Cui, Y.; Liao, X.-M.; Tan, X.-B.; Cao, X. Effect of emodin on the cariogenic properties of Streptococcus mutans and the development of caries in rats. Exp. Ther. Med. 2014, 8, 1308–1312. [Google Scholar] [CrossRef]
- Jiamboonsri, P.; Kanchanadumkerng, P. Influence of Gallic Acid and Thai Culinary Essential Oils on Antibacterial Activity of Nisin against Streptococcus mutans. Adv. Pharmacol. Pharm. Sci. 2021, 2021, 5539459. [Google Scholar] [CrossRef]
- Balasubramanian, A.R.; Vasudevan, S.; Shanmugam, K.; Lévesque, C.M.; Solomon, A.P.; Neelakantan, P. Combinatorial effects of trans—Cinnamaldehyde with fluoride and chlorhexidine on Streptococcus mutans. J. Appl. Microbiol. 2020, 130, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Jurakova, V.; Farková, V.; Kucera, J.; Dadakova, K.; Zapletalova, M.; Paskova, K.; Reminek, R.; Glatz, Z.; Holla, L.I.; Ruzicka, F.; et al. Gene expression and metabolic activity of Streptococcus mutans during exposure to dietary carbohydrates glucose, sucrose, lactose, and xylitol. Mol. Oral Microbiol. 2023, 38, 424–441. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Jiang, X.; Yang, Q.; Zhang, Y.; Wang, C.; Huang, R. Inhibition of Streptococcus mutans Biofilm Formation by the Joint Action of Oxyresveratrol and Lactobacillus casei. Appl. Environ. Microbiol. 2022, 88, e0243621. [Google Scholar] [CrossRef] [PubMed]
- Bowen, W.H.; Koo, H. Biology of Streptococcus mutans-Derived Glucosyltransferases: Role in Extracellular Matrix Formation of Cariogenic Biofilms. Caries Res. 2011, 45, 69–86. [Google Scholar] [CrossRef]
- Xiang, S.-W.; Shao, J.; He, J.; Wu, X.-Y.; Xu, X.-H.; Zhao, W.-H. A Membrane-Targeted Peptide Inhibiting PtxA of Phosphotransferase System Blocks Streptococcus mutans. Caries Res. 2018, 53, 176–193. [Google Scholar] [CrossRef]
- Passos, M.R.; Almeida, R.S.; Lima, B.O.; de Souza Rodrigues, J.Z.; de Macêdo Neres, N.S.; Pita, L.S.; Marinho, P.D.F.; Santos, I.A.; da Silva, J.P.; Oliveira, M.C.; et al. Anticariogenic activities of Libidibia ferrea, gallic acid and ethyl gallate against Streptococcus mutans in biofilm model. J. Ethnopharmacol. 2021, 274, 114059. [Google Scholar] [CrossRef]
- Sela, M.N.; Steinberg, D.; Segal, R. Inhibition of the activity of glucosyltransferase from Streptococcus mutans by glycyrrhizin. Oral Microbiol. Immunol. 1987, 2, 125–128. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Washio, J.; Abiko, Y.; Zhang, L.; Takahashi, N. Green Tea-Derived Catechins Suppress the Acid Productions of Streptococcus mutans and Enhance the Efficiency of Fluoride. Caries Res. 2023, 57, 255–264. [Google Scholar] [CrossRef] [PubMed]
- Ban, S.-H.; Kwon, Y.-R.; Pandit, S.; Lee, Y.-S.; Yi, H.-K.; Jeon, J.-G. Effects of a bio-assay guided fraction from Polygonum cuspidatum root on the viability, acid production and glucosyltranferase of mutans streptococci. Fitoterapia 2010, 81, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Harper, R.A.; Shelton, R.M.; James, J.D.; Salvati, E.; Besnard, C.; Korsunsky, A.M.; Landini, G. Acid-induced demineralisation of human enamel as a function of time and pH observed using X-ray and polarised light imaging. Acta Biomater. 2021, 120, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Spatafora, G.; Li, Y.; He, X.; Cowan, A.; Tanner, A.C.R. The Evolving Microbiome of Dental Caries. Microorganisms 2024, 12, 121. [Google Scholar] [CrossRef]
- Li, C.; Qi, C.; Yang, S.; Li, Z.; Ren, B.; Li, J.; Zhou, X.; Cai, H.; Xu, X.; Peng, X. F0F1-ATPase Contributes to the Fluoride Tolerance and Cariogenicity of Streptococcus mutans. Front. Microbiol. 2022, 12, 777504. [Google Scholar] [CrossRef]
- Aragão, M.G.B.; Aires, C.P.; Corona, S.A.M. Effects of the green tea catechin epigallocatechin-3-gallate on Streptococcus mutans planktonic cultures and biofilms: Systematic literature review of in vitro studies. Biofouling 2022, 38, 687–695. [Google Scholar] [CrossRef]
- Xu, X.; Zhou, X.D.; Wu, C.D. The Tea Catechin Epigallocatechin Gallate Suppresses Cariogenic Virulence Factors of Streptococcus mutans. Antimicrob. Agents Chemother. 2011, 55, 1229–1236. [Google Scholar] [CrossRef]
- Ren, S.; Yang, Y.; Xia, M.; Deng, Y.; Zuo, Y.; Lei, L.; Hu, T. A Chinese herb preparation, honokiol, inhibits Streptococcus mutans biofilm formation. Arch. Oral Biol. 2022, 147, 105610. [Google Scholar] [CrossRef]
- Duarte, S.; Rosalen, P.L.; Hayacibara, M.F.; Cury, J.A.; Bowen, W.H.; Marquis, R.; Rehder, V.L.; Sartoratto, A.; Ikegaki, M.; Koo, H. The influence of a novel propolis on mutans streptococci biofilms and caries development in rats. Arch. Oral Biol. 2006, 51, 15–22. [Google Scholar] [CrossRef]
- He, Z.; Huang, Z.; Jiang, W.; Zhou, W. Antimicrobial Activity of Cinnamaldehyde on Streptococcus mutans Biofilms. Front. Microbiol. 2019, 10, 2241. [Google Scholar] [CrossRef] [PubMed]
- Kleanthous, C.; Armitage, J.P. The bacterial cell envelope. Philos. Trans. R. Soc. B Biol. Sci. 2015, 370, 20150019. [Google Scholar] [CrossRef]
- Shao, D.; Li, J.; Li, J.; Tang, R.; Liu, L.; Shi, J.; Huang, Q.; Yang, H. Inhibition of Gallic Acid on the Growth and Biofilm Formation of Escherichia coli and Streptococcus mutans. J. Food Sci. 2015, 80, M1299–305. [Google Scholar] [CrossRef]
- Wang, Y.; Lam, A.T. Epigallocatechin gallate and gallic acid affect colonization of abiotic surfaces by oral bacteria. Arch. Oral Biol. 2020, 120, 104922. [Google Scholar] [CrossRef] [PubMed]
- Zulhendri, F.; Felitti, R.; Fearnley, J.; Ravalia, M. The use of propolis in dentistry, oral health, and medicine: A review. J. Oral Biosci. 2021, 63, 23–34. [Google Scholar] [CrossRef]
- El-Darier, S.M.; El-Ahwany, A.M.D.; Elkenany, E.T.; Abdeldaim, A.A. An in vitro study on antimicrobi-al and anticancer potentiality of thyme and clove oils. Rend. Lincei Sci. Fis. E Nat. 2018, 29, 131–139. [Google Scholar] [CrossRef]
- Pitts, N.B.; Zero, D.T.; Marsh, P.D.; Ekstrand, K.; Weintraub, J.A.; Ramos-Gomez, F.; Tagami, J.; Twetman, S.; Tsakos, G.; Ismail, A. Dental caries. Nat. Rev. Dis. Primers 2017, 3, 17030. [Google Scholar] [CrossRef] [PubMed]
- Lawson, N.C. Current Evidence for Caries Prevention and Enamel Remineralization. Compend. Contin. Educ. Dent. 2025, 46, 128–134. [Google Scholar]
- Albutti, A.; Gul, M.S.; Siddiqui, M.F.; Maqbool, F.; Adnan, F.; Ullah, I.; Rahman, Z.; Qayyum, S.; Shah, M.A.; Salman, M. Combating Biofilm by Targeting Its Formation and Dispersal Using Gallic Acid against Single and Multispecies Bacteria Causing Dental Plaque. Pathogens 2021, 10, 1486. [Google Scholar] [CrossRef]
- Schneider-Rayman, M.; Steinberg, D.; Sionov, R.V.; Friedman, M.; Shalish, M. Effect of epigallocatechin gallate on dental biofilm of Streptococcus mutans: An in vitro study. BMC Oral Health 2021, 21, 447. [Google Scholar] [CrossRef]
- Yin, W.; Zhang, Z.; Shuai, X.; Zhou, X.; Yin, D. Caffeic Acid Phenethyl Ester (CAPE) Inhibits Cross-Kingdom Biofilm Formation of Streptococcus mutans and Candida albicans. Microbiol. Spectr. 2022, 10, e0157822. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.; Wang, K.; Zheng, S.; Wang, Y.; Ren, Q.; Li, H.; Ding, L.; Li, W.; Zhang, L. Antibacterial Effect of Caffeic Acid Phenethyl Ester on Cariogenic Bacteria and Streptococcus mutans Biofilms. Antimicrob. Agents Chemother. 2020, 64, 10–1128. [Google Scholar] [CrossRef] [PubMed]
- Muras, A.; Mallo, N.; Otero-Casal, P.; Pose-Rodríguez, J.M.; Otero, A. Quorum sensing systems as a new target to prevent biofilm-related oral diseases. Oral Dis. 2022, 28, 307–313. [Google Scholar] [CrossRef] [PubMed]
- Shanker, E.; Federle, M.J. Quorum Sensing Regulation of Competence and Bacteriocins in Streptococcus pneumoniae and mutans. Genes 2017, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Li, J.; Hao, Y.; Zhou, X. Effect of compounds of Galla chinensis on remineralization of enamel surface in vitro. Arch. Oral Biol. 2010, 55, 435–440. [Google Scholar] [CrossRef]
- Zhang, L.; Li, J.; Zhou, X.; Cui, F.; Li, W. Effects of Galla chinensis on the surface topography of initial enamel carious lesion: An atomic force microscopy study. Scanning 2009, 31, 195–203. [Google Scholar] [CrossRef]
- Li, H.; Deng, M.; Peng, S.C.; Shi, J.H. Effect of Galla chinensis on the Wear Resistance of Dentine. Adv. Mater. Res. 2013, 705, 187–190. [Google Scholar] [CrossRef]
- Zhang, T.-T.; Guo, H.-J.; Liu, X.-J.; Chu, J.-P.; Zhou, X.-D. Galla chinensis Compounds Remineralize Enamel Caries Lesions in a Rat Model. Caries Res. 2016, 50, 159–165. [Google Scholar] [CrossRef]
- Sahin, F.; Oznurhan, F. Antibacterial efficacy and remineralization capacity of glycyrrhizic acid added casein phosphopeptide-amorphous calcium phosphate. Microsc. Res. Technol. 2020, 83, 744–754. [Google Scholar] [CrossRef]
- Zaleh, A.-A.; Salehi-Vaziri, A.; Pourhajibagher, M.; Bahador, A. The synergistic effect of Nano-propolis and curcumin-based photodynamic therapy on remineralization of white spot lesions: An ex vivo study. Photodiagnosis Photodyn. Ther. 2022, 38, 102789. [Google Scholar] [CrossRef]
- Srisomboon, S.; Intharah, T.; Jarujareet, U.; Toneluck, A.; Panpisut, P. The in vitro assessment of rheological properties and dentin remineralization of saliva substitutes containing propolis and aloe vera extracts. PLoS ONE 2024, 19, e0304156. [Google Scholar] [CrossRef]
- Sarialioglu Gungor, A.; Donmez, N. Dentin erosion preventive effects of various plant extracts: An in vitro atomic force microscopy, scanning electron microscopy, and nanoindentation study. Microsc. Res. Technol. 2021, 84, 1042–1052. [Google Scholar] [CrossRef] [PubMed]
- Kiani, S.; Birang, R.; Jamshidian, N. Effect of Propolis mouthwash on clinical periodontal parameters in patients with gingivitis: A double-blinded randomized clinical trial. Int. J. Dent. Hyg. 2022, 20, 434–440. [Google Scholar] [CrossRef] [PubMed]
- Mehta, V.; Mathur, A.; Gopalakrishnan, D.; Rizwan, S.; Shetiya, S.H.; Bagwe, S. Efficacy of green tea-based mouthwashes on dental plaque and gingival inflammation: A systematic review and meta-analysis. Indian J. Dent. Res. 2018, 29, 225–232. [Google Scholar] [CrossRef]
- Ashrafi, B.; Rashidipour, M.; Marzban, A.; Soroush, S.; Azadpour, M.; Delfani, S.; Ramak, P. Mentha piperita essential oils loaded in a chitosan nanogel with inhibitory effect on biofilm formation against S. mutans on the dental surface. Carbohydr. Polym. 2019, 212, 142–149. [Google Scholar] [CrossRef] [PubMed]
- Goultschin, J.; Palmon, S.; Shapira, L.; Brayer, L.; Gedalia, I. Effect of glycyrrhizin-containing toothpaste on dental plaque reduction and gingival health in humans. J. Clin. Periodontol. 1991, 18, 210–212. [Google Scholar] [CrossRef]
- Oluwasina, O.O.; Ezenwosu, I.V.; Ogidi, C.O.; Oyetayo, V.O. Antimicrobial potential of toothpaste formulated from extracts of Syzygium aromaticum, Dennettia tripetala and Jatropha curcas latex against some oral pathogenic microorganisms. AMB Express 2019, 9, 20. [Google Scholar] [CrossRef]
- Campus, G.; Cagetti, M.; Cocco, F.; Sale, S.; Sacco, G.; Strohmenger, L.; Lingström, P. Effect of a Sugar-Free Chewing Gum Containing Magnolia Bark Extract on Different Variables Related to Caries and Gingivitis: A Randomized Controlled Intervention Trial. Caries Res. 2011, 45, 393–399. [Google Scholar] [CrossRef]
- Wessel, S.W.; van der Mei, H.C.; Slomp, A.M.; van de Belt-Gritter, B.; Dodds, M.W.J.; Busscher, H.J. Self-perceived mouthfeel and physico-chemical surface effects after chewing gums containing sorbitol and Magnolia bark extract. Eur. J. Oral Sci. 2017, 125, 379–384. [Google Scholar] [CrossRef]
- Manohar, R.; Ganesh, A.; Abbyramy, N.; Abinaya, R.; Balaji, S.; Priya, S.B. The effect of fennel seeds on pH of saliva—A clinical study. Indian J. Dent. Res. 2020, 31, 921–923. [Google Scholar] [CrossRef]


| Classification | Name | Bioactive Ingredients | Chemical structure | Source |
|---|---|---|---|---|
| Organic acid-based | Galla Chinensis | Gallic acid, methyl gallate, polymeric polyphenols |  Gallic acid | The gall forming when the Chinese sumac aphid Baker (Melaphis chinensis Bell) parasitizes the leaves of Rhus chinensis |
| Radix glycyrrhizae (licorice root) | Glycyrrhizic acid (Glycryrrhizin) | 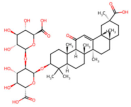 Glycyrrhizin | The dried roots and rhizomes of Glycyrrhiza uralensis Fisch, Glycyrrhiza inflata or Glycyrrhiza glabra | |
| Lonicera japonica (honeysuckle) | Chlorogenic acid, luteolin glycosides | 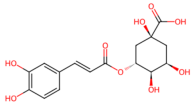 Chlorogenic acid | The dried flower buds or flowers in the early blooming stage of Lonicera japonica | |
| Prunus mume (plum) | Citric acid, mallic acid, chlorogenic acid |  Citric acid | The dried nearly ripe fruit of Prunus mume | |
| Anisum stellatum | Shikimic acid | 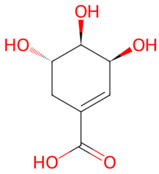 Shikimic acid | The dried ripe fruit of Illicium verum | |
| Alkaloid-based | Coptis chinensis | Berberine | 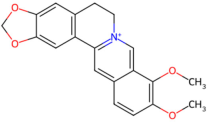 Berberine | The dried rhizome of Coptis chinensis |
| Chelidonium majus | Chelerythrine | 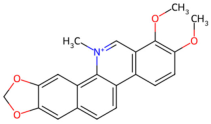 Chelerythrine | The whole herb of Chelidonium majus | |
| Sophora flavescens | Matrine, oxymatrine | 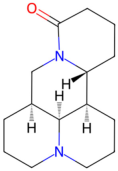 Matrine | The dried roots of Sophora flavescens | |
| Phenol-based | Tea | Tea polyphenols (catechins, EGC, EGCG) |  Catechin | The tender leaves or buds of the plant Camellia sinensis |
| Propolis | Quercetin, rutin, apigenin | 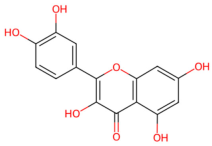 Quercetin | A natural product processed from the resinous substances secreted by honeybees | |
| Clove | Eugenol | 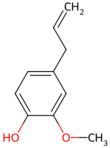 Eugenol | The dried flower buds of Syzygium aromaticum | |
| Magnolia officinalis | Magnolol, honokiol |  Magnolol 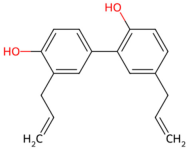 honokiol honokiol | The dried trunk bark, root bark or branch bark of Magnolia officinalis | |
| Anthraquinone-based | Aloe vera | Aloe emodin, aloin |  Aloin | The concentrated and dried juice of the leaves of Aloe vera |
| Polygonum cuspidatum | Polydatin, emodin | 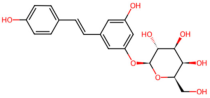 Polydatin | The dried rhizomes and roots of Polygonum cuspidatum | |
| Other types | Peppermint | Menthol | 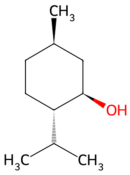 Menthol | The dried aerial parts of Mentha haplocalyx |
| Cinnamon | Cinnamaldehyde, eugenol, linalool | 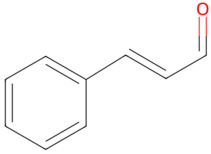 Cinnamaldehyde | The dried bark of Cinnamomum cassia | |
| Shaddock ped | Limonene, naringenin |  Limonene | The pericarp of Citrus maxima |
| Classification (Bioactive Component) | CBP (Representative Extract/Compound) | Effect on GtfB/C Activity | Effect on gtfB/C Expression | Effect on Biofilm Structure/Adhesion | References |
|---|---|---|---|---|---|
| I. Organic Acids | |||||
| 1. Gallotannins/Phenolic acids | Galla chinensis (Aqueous extract, tannic acid) | ↓ (Direct inhibition) | - | ↓ (Complexes with pellicle, reduces affinity) | [13,93] |
| 2. Triterpenoid saponins | Radix Glycyrrhizae (Glycyrrhizic acid) | ↓ | - | ↓ (Surface coating effect) | [94,95,96] |
| 3. Chlorogenic acid | Lonicera japonica (Chlorogenic acid) | ↓ (Via QS inhibition) | - | - | [20] |
| 4. Shikimic acid | Anisum stellatum (Shikimic acid) | - | ↓ | ↓ (Damages cell membrane) | [25] |
| II. Alkaloids | |||||
| 1. Isoquinoline alkaloids | Coptis chinensis (Berberine chloride) | - | - | ↓ (Downregulates srtA, gbpC; inhibits metabolic activity) | [28] |
| 2. Isoquinoline alkaloids | Chelidonium majus (Chelerythrine) | - | - | ↓ (Reduces adhesion ability) | [34,97] |
| III. Phenols | |||||
| 1. Tea polyphenols | Tea (EGCG, catechins) | ↓ (Non-competitive, binds glucan domain) | ↓ | ↓ (Alters hydrophobicity & aggregation) | [42,98,99,100] |
| 2. Flavonoids/Phenolics | Propolis (PEOME, apigenin) | ↓ | - | ↓ (Increases hydrophobicity, damages membrane) | [56,74] |
| 3. Phenolic lignans | Magnolia officinalis (Magnolol) | ↓ (Non-competitive, binds glucan domain) | - | ↓ (Penetrates biofilm) | [39,65] |
| 4. Eugenol derivatives | Clove (Clove essential oil) | - | - | ↓ (Damages cell membrane) | [61,101] |
| IV. Anthraquinones | |||||
| 1. Anthraquinone glycosides | Aloe vera (Gel) | - | - | ↓ (Inhibits growth and adherence) | [102] |
| 2. Emodin, Physcion | Polygonum cuspidatum (Emodin) | - | - | - | [103] - |
| V. Others | |||||
| 1. Cinnamaldehyde, etc. | Cinnamon (Cinnamaldehyde) | - | ↓ | ↓ (Alters hydrophobicity & aggregation) | [104,105] |
| Classification (Bioactive Component) | CBP (Representative Extract/ Compound) | Effect on GtfD/ Soluble Glucan | Effect on Sugar Uptake (PEP-PTS) | Effect on Glycolysis/Energy Metabolism | References |
|---|---|---|---|---|---|
| I. Organic acids | |||||
| 1. Gallotannins/ Phenolic acids | Galla chinensis (Gallic acid) | ↓ (Downregulates gtfD) | - | - | [110] |
| 2. Triterpenoid saponins | Radix Glycyrrhizae (Glycyrrhizic acid) | ↓ (Inhibit Gtfs) | - | - | [111] |
| II. Alkaloids | |||||
| 1. Isoquinoline alkaloids | Coptis chinensis (Berberine) | - | - | ↓ (Inhibits biofilm metabolic activity) | [28] |
| III. Phenols | |||||
| 1. Tea polyphenols | Tea (Catechins, TPs) | - | ↓ (Blocks EIIC transporter) | ↓ (Downregulates glycolysis & TCA cycle) | [43,112] |
| IV. Anthraquinones | |||||
| 1. Emodin, Physcion | Polygonum cuspidatum (Bioassay-guided fraction) | - | - | ↓ (Produces anti-acidogenic substances, inhibits glycolytic process) | [75,113] |
| Classification (Bioactive Component) | CBP (Representative Extract/Compound) | Effect on LDH (Acidogenicity) | Effect on F0F1-ATPase (Aciduricity) | References |
|---|---|---|---|---|
| I. Organic acids | Galla chinensis (Extract) | - (Limits acid accumulation) | - | [12] |
| II. Alkaloids | ||||
| 1. Isoquinoline alkaloids | Coptis chinensis (Berberine hydrate) | ↓ (Downregulates ldh expression) | - | [27] |
| III. Phenols | ||||
| 1. Tea polyphenols | Tea (Catechins, EGCG) | ↓ (Inhibits activity; blocks substrate) | ↓ (Inhibits activity & atpD expression) | [112,117,118] |
| 2. Phenolic lignans | Magnolia officinalis (Honokiol) | ↓ (Downregulates ldh expression) | - | [119] |
| 3. Flavonoids/Phenolics | Propolis (Essential oil, PEOME, ethanol extract) | ↓ (Downregulates ldh; inactivates leaked enzyme) | ↓ (Inhibits activity, disrupts pH gradient) | [55,56,120] |
| IV. Anthraquinones | ||||
| 1. Anthraquinone glycosides | Aloe vera (Gel) | - | - | [102] |
| V. Others | ||||
| Cinnamaldehyde, trans-Cinnamaldehyde | Cinnamon | ↓ (Inhibits glycolytic enzymes) | ↓ (Suppresses atpD expression) | [105,121] |
| Classification (Bioactive Component) | CBP (Representative Extract/Compound) | Effect on Cell Membrane | Effect on Ion Homeostasis/Metals | Effect on Cell Wall/Other Metabolism | References |
|---|---|---|---|---|---|
| I. Organic acids | |||||
| 1. Gallotannins/ Phenolic acids | Galla chinensis (Gallic acid) | ↓ (Disrupts bilayer, causes Ca2+ efflux) | ↓ (Iron chelation by tannic acid) | - | [6,104,124] |
| 2. Shikimic acid | Anisum stellatum (Shikimic acid) | ↓ (Alters membrane proteins) | - | - | [25] |
| III. Phenols | |||||
| 1. Tea polyphenols | Tea (EGCG, TPs) | ↓ (Reduces hydrophobicity, impairs permeability) | - | ↓ (Disrupts peptidoglycan cross-linking) | [43,124] |
| 2. Flavonoids/Phenolics | Propolis (Extract) | ↓ (Alters hydrophobicity, forms pores) | - | - | [125] |
| 3. Eugenol derivatives | Clove (Clove essential oil) | ↓ (Penetrates and damages lipids) | - | - | [60,126] |
| V. Others | |||||
| 1.Cinnamaldehyde, etc. | Cinnamon (Essential oil) | ↓ (Damages membrane, reduces ATP) | - | - | [78] |
| Classification (Bioactive Component) | CBP (Representative Extract/Compound) | Effect on ComDE System | Effect on LuxS/AI-2 System | Effect on Other QS-Related Elements | References |
|---|---|---|---|---|---|
| I. Organic acids | |||||
| 1. Gallotannins/ Phenolic acids | Galla chinensis (Polyphenols) | - | - | ↓ (Interacts with QS signals, undermines competence) | [129] |
| 2. Chlorogenic acid | Lonicera japonica (Chlorogenic acid) | - | ↓ (Blocks AI-2 sensing) | ↓ (Downregulates vicK) | [20] |
| II. Alkaloids | |||||
| 1. Isoquinoline alkaloids | Coptis chinensis (Berberine) | ↓ (Suppresses comX expression) | - | - | [28] |
| III. Phenols | |||||
| 1. Tea polyphenols | Tea (EGCG) | - | ↓ (Downregulates luxS expression) | - | [130] |
| 2. Flavonoids/Phenolics | Propolis (CAPE) | - | - | ↓ (Downregulates vicK, vicR, ccpA) | [131,132] |
| V. Others | |||||
| 1. Cinnamaldehyde, etc. | Cinnamon (trans-Cinnamaldehyde) | ↓ (Downregulates comDE) | ↓ (Downregulates luxS) | - | [105] |
| Classification (Bioactive Component) | CBP (Representative Extract/Compound) | Effect on Demineralization | Effect on Remineralization | Proposed Mechanism | References |
|---|---|---|---|---|---|
| I. Organic acids | |||||
| 1. Gallotannins/Phenolic acids | Galla chinensis (GC extract, Gallic Acid, Tannic acid) | ↓ (Inhibits ion diffusion) | ↑↑ (Significant enhancement) | 1. Provides Ca2+ ions. 2. Forms “enamel organic matrix–GC–Ca2+” complex to transport ions. 3. Forms “GC–dentin matrix” complex to stabilize collagen. | [7,135,136,137,138] |
| 2. Triterpenoid saponins | Radix Glycyrrhizae (Glycyrrhizic acid) | ↓ (Reduces enamel dissolution) | - | Surface coating effect that limits acid access. | [139] |
| III. Phenols | |||||
| 1. Flavonoids/Phenolics | Propolis (Extract) | - | ↑ (Potential effect suggested) | May aid in mineral deposition. | [140,141] |
| 2. Eugenol derivatives | Clove (Clove Essential Oil) | - | ↑ (Potential effect suggested) | May operate on de/remineralization balance. | [60,142] |
| IV. Anthraquinones | |||||
| 1. Anthraquinone glycosides | Aloe vera (Gel) | - | ↑ (Improves enamel density/hardness) | Application of gel improves surface microhardness in vitro. | [71,141] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, Y.; Fang, Z.; Huang, R. Classification and Anti-Streptococcus mutans Mechanism Summary of Chinese Botanical Products. Pathogens 2026, 15, 280. https://doi.org/10.3390/pathogens15030280
Li Y, Fang Z, Huang R. Classification and Anti-Streptococcus mutans Mechanism Summary of Chinese Botanical Products. Pathogens. 2026; 15(3):280. https://doi.org/10.3390/pathogens15030280
Chicago/Turabian StyleLi, Yuelin, Zhongyi Fang, and Ruijie Huang. 2026. "Classification and Anti-Streptococcus mutans Mechanism Summary of Chinese Botanical Products" Pathogens 15, no. 3: 280. https://doi.org/10.3390/pathogens15030280
APA StyleLi, Y., Fang, Z., & Huang, R. (2026). Classification and Anti-Streptococcus mutans Mechanism Summary of Chinese Botanical Products. Pathogens, 15(3), 280. https://doi.org/10.3390/pathogens15030280






