In Vivo Confocal Microscopy as a Prognostic Indicator in Acanthamoeba Keratitis: Insights from a Retrospective Study
Abstract
1. Introduction
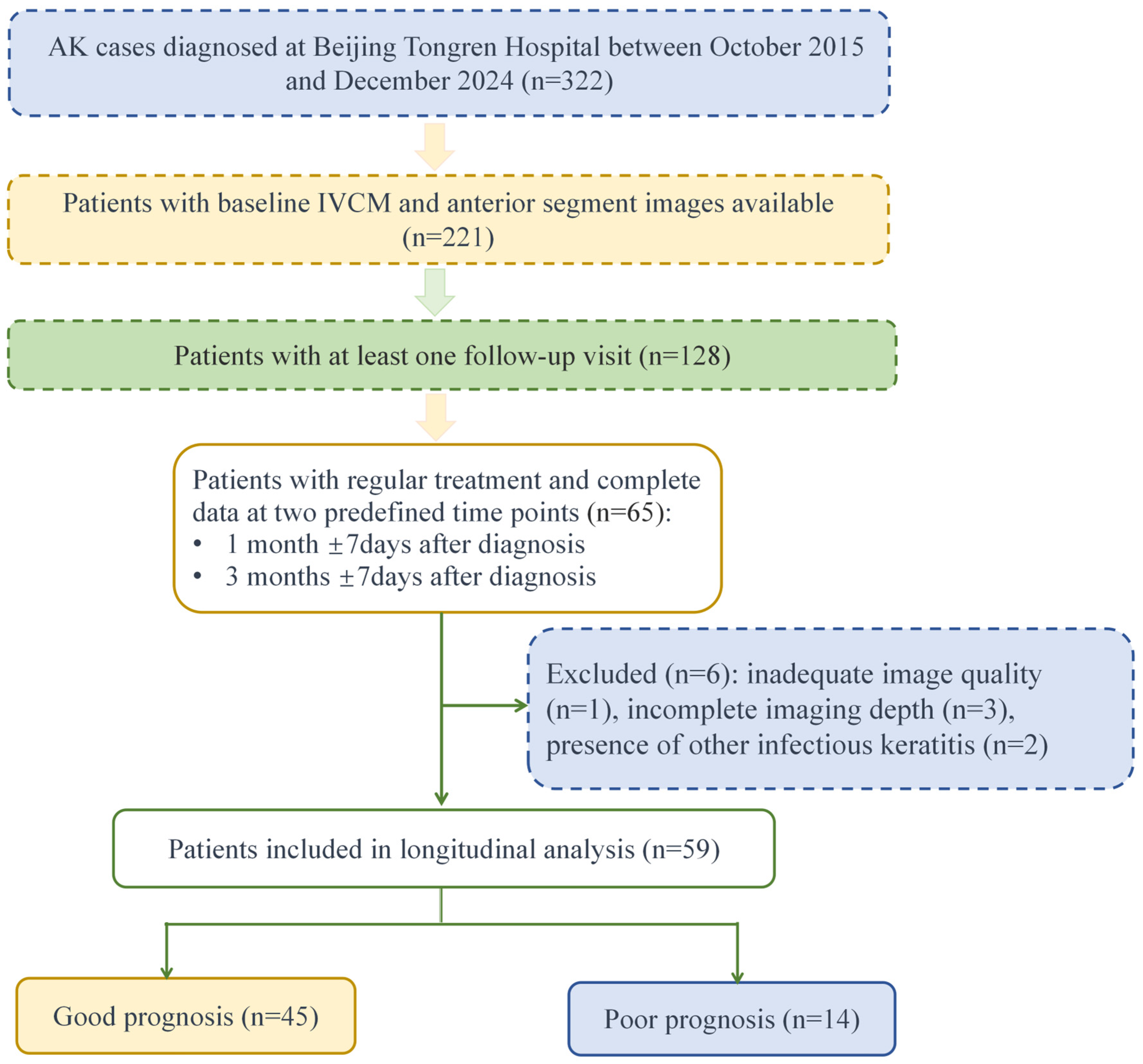
2. Materials and Methods
2.1. Study Design
2.2. Clinical Assessment and Prognostic Grouping
2.3. In Vivo Confocal Microscopy Evaluation
2.4. Feature-Outcome Association Analysis
2.5. Statistical Analysis
3. Results
3.1. Patient Characteristics and Clinical Manifestations
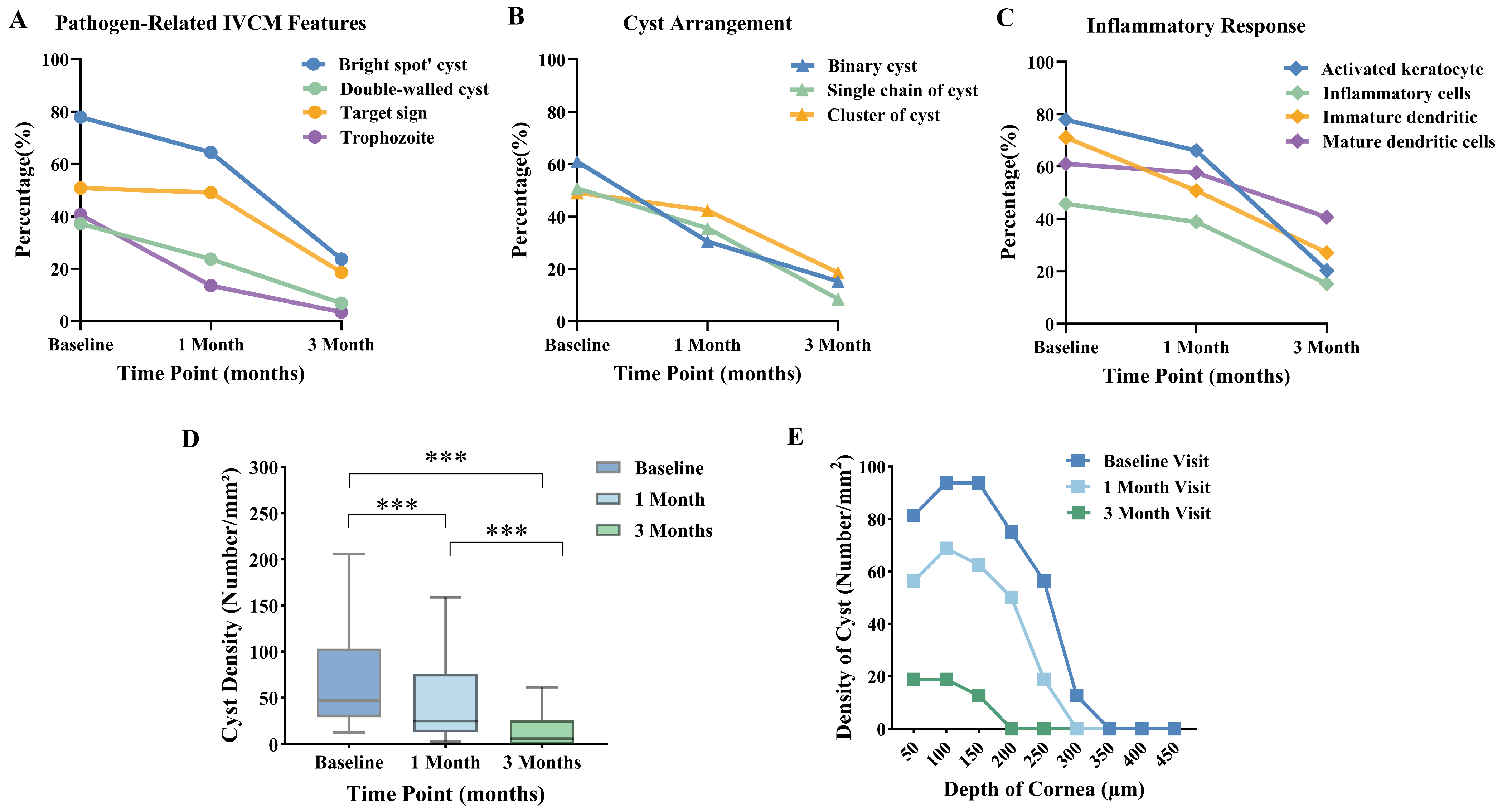
3.2. Quantitative Analysis of IVCM Images
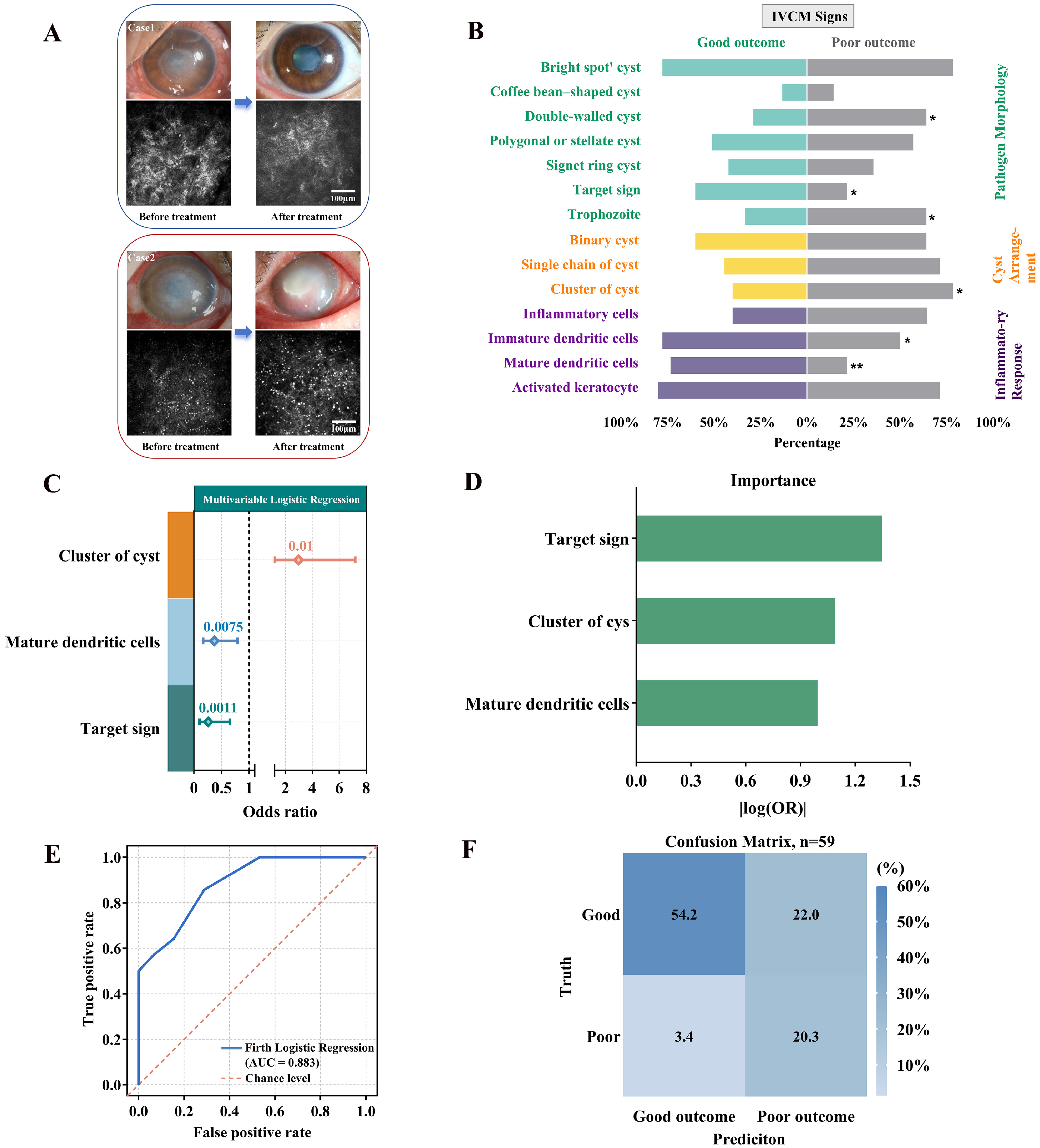
3.3. Prognosis-Associated Differences in Baseline Features
3.4. IVCM Features and Clinical Prognosis
3.5. Multivariable Regression Analysis of IVCM Features and Model Performance Evaluation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AK | Acanthamoeba keratitis |
| IVCM | In vivo confocal microscopy |
| BCVA | Best-corrected visual acuity |
| DCs | Dendritic cells |
References
- Petrillo, F.; Tortori, A.; Vallino, V.; Galdiero, M.; Fea, A.M.; De Sanctis, U.; Reibaldi, M. Understanding Acanthamoeba keratitis: An in-depth review of a sight-threatening eye infection. Microorganisms 2024, 12, 758. [Google Scholar] [CrossRef]
- Garg, D.; Daigavane, S. A comprehensive review on Acanthamoeba keratitis: An overview of epidemiology, risk factors, and therapeutic strategies. Cureus 2024, 16, e67803. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Xu, X.; Wei, Z.; Cao, K.; Zhang, Z.; Liang, Q. The global epidemiology and clinical diagnosis of Acanthamoeba keratitis. J. Infect. Public Health 2023, 16, 841–852. [Google Scholar] [CrossRef]
- Ilyas, M.; Stapleton, F.; Willcox, M.D.P.; Henriquez, F.; Peguda, H.K.; Rayamajhee, B.; Zahid, T.; Petsoglou, C.; Carnt, N.A. Epidemiology of and genetic factors associated with Acanthamoeba keratitis. Pathogens 2024, 13, 142. [Google Scholar] [CrossRef]
- Ross, J.; Roy, S.L.; Mathers, W.D.; Ritterband, D.C.; Yoder, J.S.; Ayers, T.; Shah, R.D.; Samper, M.E.; Shih, C.Y.M.; Schmitz, A.D.; et al. Clinical characteristics of Acanthamoeba keratitis infections in 28 states, 2008 to 2011. Cornea 2014, 33, 161–168. [Google Scholar] [CrossRef]
- Mascarenhas, J.; Lalitha, P.; Prajna, N.V.; Srinivasan, M.; Das, M.; D’Silva, S.S.; Oldenburg, C.E.; Borkar, D.S.; Esterberg, E.J.; Lietman, T.M.; et al. Acanthamoeba, fungal, and bacterial keratitis: A comparison of risk factors and clinical features. Am. J. Ophthalmol. 2014, 157, 56–62. [Google Scholar] [CrossRef]
- Sharma, S.; Kunimoto, D.Y.; Gopinathan, U.; Athmanathan, S.; Garg, P.; Rao, G.N. Evaluation of corneal scraping smear examination methods in the diagnosis of bacterial and fungal keratitis: A survey of eight years of laboratory experience. Cornea 2002, 21, 643–647. [Google Scholar] [CrossRef]
- Ikeda, Y.; Miyazaki, D.; Yakura, K.; Kawaguchi, A.; Ishikura, R.; Inoue, Y.; Mito, T.; Shiraishi, A.; Ohashi, Y.; Higaki, S.; et al. Assessment of real-time polymerase chain reaction detection of Acanthamoeba and prognosis determinants of Acanthamoeba keratitis. Ophthalmology 2012, 119, 1111–1119. [Google Scholar] [CrossRef]
- Chidambaram, J.D.; Prajna, N.V.; Larke, N.L.; Palepu, S.; Lanjewar, S.; Shah, M.; Elakkiya, S.; Lalitha, P.; Carnt, N.; Vesaluoma, M.H.; et al. Prospective study of the diagnostic accuracy of the s laser scanning confocal microscope for severe microbial keratitis. Ophthalmology 2016, 123, 2285–2293. [Google Scholar] [CrossRef]
- Azzopardi, M.; Chong, Y.J.; Ng, B.; Recchioni, A.; Logeswaran, A.; Ting, D.S.J. Diagnosis of Acanthamoeba keratitis: Past, present and future. Diagnostics 2023, 13, 2655. [Google Scholar] [CrossRef] [PubMed]
- Chidambaram, J.D.; Prajna, N.V.; Palepu, S.; Lanjewar, S.; Shah, M.; Elakkiya, S.; Macleod, D.; Lalitha, P.; Burton, M.J. In vivo confocal microscopy cellular features of host and organism in bacterial, fungal, and Acanthamoeba keratitis. Am. J. Ophthalmol. 2018, 190, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Chopra, R.; Mulholland, P.J.; Hau, S.C. In vivo confocal microscopy morphologic features and cyst density in Acanthamoeba keratitis. Am. J. Ophthalmol. 2020, 217, 38–48. [Google Scholar] [CrossRef] [PubMed]
- De Craene, S.; Knoeri, J.; Georgeon, C.; Kestelyn, P.; Borderie, V.M. Assessment of confocal microscopy for the diagnosis of polymerase chain reaction-positive Acanthamoeba keratitis: A case-control study. Ophthalmology 2018, 125, 161–168. [Google Scholar] [CrossRef]
- Przybek-Skrzypecka, J.; Armstrong, M.; Kim, J.; Walkden, A.; Au, L.; Brahma, A.; Carley, F.; Chidambaram, J.D. Diagnostic features of Acanthamoeba keratitis via in vivo confocal microscopy. Sci. Rep. 2025, 15, 10940. [Google Scholar] [CrossRef]
- Goh, J.W.Y.; Harrison, R.; Hau, S.; Alexander, C.L.; Tole, D.M.; Avadhanam, V.S. Comparison of in vivo confocal microscopy, PCR and culture of corneal scrapes in the diagnosis of Acanthamoeba keratitis. Cornea 2018, 37, 480–485. [Google Scholar] [CrossRef]
- Tabatabaei, S.A.; Soleimani, M.; Tabatabaei, S.M.; Beheshtnejad, A.H.; Valipour, N.; Mahmoudi, S. The use of in vivo confocal microscopy to track treatment success in fungal keratitis and to differentiate between Fusarium and Aspergillus keratitis. Int. Ophthalmol. 2020, 40, 483–491. [Google Scholar] [CrossRef]
- Wei, Z.; Cao, K.; Wang, L.; Baudouin, C.; Labbé, A.; Liang, Q. Corneal changes in Acanthamoeba keratitis at various levels of severity: An in vivo confocal microscopic study. Transl. Vis. Sci. Technol. 2021, 10, 10. [Google Scholar] [CrossRef]
- Essalat, M.; Abolhosseini, M.; Le, T.H.; Moshtaghion, S.M.; Kanavi, M.R. Interpretable deep learning for diagnosis of fungal and acanthamoeba keratitis using in vivo confocal microscopy images. Sci. Rep. 2023, 13, 8953. [Google Scholar] [CrossRef]
- Toba, B.; Lagali, N. Use of in vivo confocal microscopy in suspected Acanthamoeba keratitis: A 12-year real-world data study at a Swedish regional referral center. J. Ophthalmic Inflamm. Infect. 2024, 14, 43. [Google Scholar] [CrossRef]
- Vaddavalli, P.K.; Garg, P.; Sharma, S.; Sangwan, V.S.; Rao, G.N.; Thomas, R. Role of confocal microscopy in the diagnosis of fungal and acanthamoeba keratitis. Ophthalmology 2011, 118, 29–35. [Google Scholar] [CrossRef]
- Shareef, O.; Soleimani, M.; Tu, E.; Jacobs, D.S.; Ciolino, J.B.; Rahdar, A.; Cheraqpour, K.; Ashraf, M.; Habib, N.B.; Greenfield, J.; et al. A novel artificial intelligence model for diagnosing Acanthamoeba keratitis through confocal microscopy. Ocul. Surf. 2024, 34, 159–164. [Google Scholar] [CrossRef]
- Wang, Y.E.; Tepelus, T.C.; Vickers, L.A.; Baghdasaryan, E.; Gui, W.; Huang, P.; Irvine, J.A.; Sadda, S.; Hsu, H.Y.; Lee, O.L. Role of in vivo confocal microscopy in the diagnosis of infectious keratitis. Int. Ophthalmol. 2019, 39, 2865–2874. [Google Scholar] [CrossRef]
- Curro-Tafili, K.; Verbraak, F.D.; de Vries, R.; van Nispen, R.M.A.; Ghyczy, E.A.E. Diagnosing and monitoring the characteristics of Acanthamoeba keratitis using slit scanning and laser scanning in vivo confocal microscopy. Ophthalmic Physiol. Opt. 2024, 44, 131–152. [Google Scholar] [CrossRef]
- Labbé, A.; Khammari, C.; Dupas, B.; Gabison, E.; Brasnu, E.; Labetoulle, M.; Baudouin, C. Contribution of in vivo confocal microscopy to the diagnosis and management of infectious keratitis. Ocul. Surf. 2009, 7, 41–52. [Google Scholar] [CrossRef]
- Wang, Y.E.; Tepelus, T.C.; Gui, W.; Irvine, J.A.; Lee, O.L.; Hsu, H.Y. Reduction of Acanthamoeba cyst density associated with treatment detected by in vivo confocal microscopy in Acanthamoeba keratitis. Cornea 2019, 38, 463–468. [Google Scholar] [CrossRef]
- Chidambaram, J.D.; Prajna, N.V.; Palepu, S.; Lanjewar, S.; Shah, M.; Elakkiya, S.; Lalitha, P.; Macleod, D.; Burton, M.J. Cellular morphological changes detected by laser scanning in vivo confocal microscopy associated with clinical outcome in fungal keratitis. Sci. Rep. 2019, 9, 8334. [Google Scholar] [CrossRef]
- Peng, Y.; Chen, Q.; Wei, Y.; Wang, L.; Zhang, Z.; Wei, Z.; Pang, J.; Peng, B.; Shi, Q.; Wang, Z.; et al. Clinical characteristics and in vivo confocal microscopic study in Candida keratitis. Transl. Vis. Sci. Technol. 2025, 14, 23. [Google Scholar] [CrossRef]
- Lee, H.J.; Alipour, F.; Cruzat, A.; Posarelli, M.; Zheng, L.; Hamrah, P. Utility of in vivo confocal microscopy in diagnosis of Acanthamoeba keratitis: A comparison of patient outcomes. Cornea 2023, 42, 135–140. [Google Scholar] [CrossRef]
- Li, S.; Bian, J.; Wang, Y.; Wang, S.; Wang, X.; Shi, W. Clinical features and serial changes of Acanthamoeba keratitis: An in vivo confocal microscopy study. Eye 2020, 34, 327–334. [Google Scholar] [CrossRef]
- McClellan, K.; Howard, K.; Mayhew, E.; Niederkorn, J.; Alizadeh, H. Adaptive immune responses to Acanthamoeba cysts. Exp. Eye Res. 2002, 75, 285–293. [Google Scholar] [CrossRef]
- Kinnear, F.B. Acanthamoeba pathogenicity for corneal cells. J. Infect. 2004, 49, 310–316. [Google Scholar] [CrossRef]
- Omaña-Molina, M.; González-Robles, A.; Salazar-Villatoro, L.I.; Lorenzo-Morales, J.; Cristóbal-Ramos, A.R.; Hernández-Ramírez, V.I.; Talamás-Rohana, P.; Cruz, A.R.M.; Martínez-Palomo, A. Reevaluating the role of Acanthamoeba proteases in tissue invasion: Observation of cytopathogenic mechanisms on MDCK cell monolayers and hamster corneal cells. BioMed Res. Int. 2013, 2013, 461329. [Google Scholar] [CrossRef]
- Ficker, L.; Seal, D.; Warhurst, D.; Wright, P. Acanthamoeba keratitis—Resistance to medical therapy. Eye 1990, 4, 835–838. [Google Scholar] [CrossRef]
- Pérez-Santonja, J.J.; Kilvington, S.; Hughes, R.; Tufail, A.; Matheson, M.; Dart, J.K. Persistently culture positive Acanthamoeba keratitis: In vivo resistance and in vitro sensitivity. Ophthalmology 2003, 110, 1593–1600. [Google Scholar] [CrossRef]
- Marciano-Cabral, F.; Cabral, G. Acanthamoeba spp. as agents of disease in humans. Clin. Microbiol. Rev. 2003, 16, 273–307. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.C.; Lin, J.L.; Lin-Tan, D.T.; Ma, H.K.; Chen, H.C. Early keratectomy in the treatment of moderate Fusarium keratitis. PLoS ONE 2012, 7, e42126. [Google Scholar] [CrossRef][Green Version]
- Downie, L.E.; Bandlitz, S.; Bergmanson, J.P.G.; Craig, J.P.; Dutta, D.; Maldonado-Codina, C.; Ngo, W.; Siddireddy, J.S.; Wolffsohn, J.S. CLEAR—Anatomy and physiology of the anterior eye. Cont. Lens Anterior Eye 2021, 44, 132–156. [Google Scholar] [CrossRef]
- Polat, H.K.; Kurt, N.; Aytekin, E.; Bozdağ Pehlivan, S.; Çalış, S. Novel drug delivery systems to improve the treatment of keratitis. J. Ocul. Pharmacol. Ther. 2022, 38, 376–395. [Google Scholar] [CrossRef]
- Sarnicola, E.; Sarnicola, C.; Sabatino, F.; Tosi, G.M.; Perri, P.; Sarnicola, V. Early deep anterior lamellar keratoplasty (DALK) for Acanthamoeba keratitis poorly responsive to medical treatment. Cornea 2016, 35, 1–5. [Google Scholar] [CrossRef]
- Huang, P.; Tepelus, T.; Vickers, L.A.; Baghdasaryan, E.; Huang, J.; Irvine, J.A.; Hsu, H.Y.; Sadda, S.; Lee, O.L. Quantitative analysis of depth, distribution, and density of cysts in Acanthamoeba keratitis using confocal microscopy. Cornea 2017, 36, 927–932. [Google Scholar] [CrossRef]
- Garajová, M.; Mrva, M.; Vaškovicová, N.; Martinka, M.; Melicherová, J.; Valigurová, A. Cellulose fibrils formation and organisation of cytoskeleton during encystment are essential for Acanthamoeba cyst wall architecture. Sci. Rep. 2019, 9, 4466. [Google Scholar] [CrossRef] [PubMed]
- Campolo, A.; Pifer, R.; Walters, R.; Thomas, M.; Miller, E.; Harris, V.; King, J.; Rice, C.A.; Shannon, P.; Patterson, B.; et al. Acanthamoeba spp. aggregate and encyst on contact lens material increasing resistance to disinfection. Front. Microbiol. 2022, 13, 1089092. [Google Scholar] [CrossRef] [PubMed]
- Puhr, R.; Heinze, G.; Nold, M.; Lusa, L.; Geroldinger, A. Firth’s logistic regression with rare events: Accurate effect estimates and predictions? Stat. Med. 2017, 36, 2302–2317. [Google Scholar] [CrossRef]
- Lincke, A.; Roth, J.; Macedo, A.F.; Bergman, P.; Löwe, W.; Lagali, N.S. AI-based decision-support system for diagnosing Acanthamoeba keratitis using in vivo confocal microscopy images. Transl. Vis. Sci. Technol. 2023, 12, 29. [Google Scholar] [CrossRef]
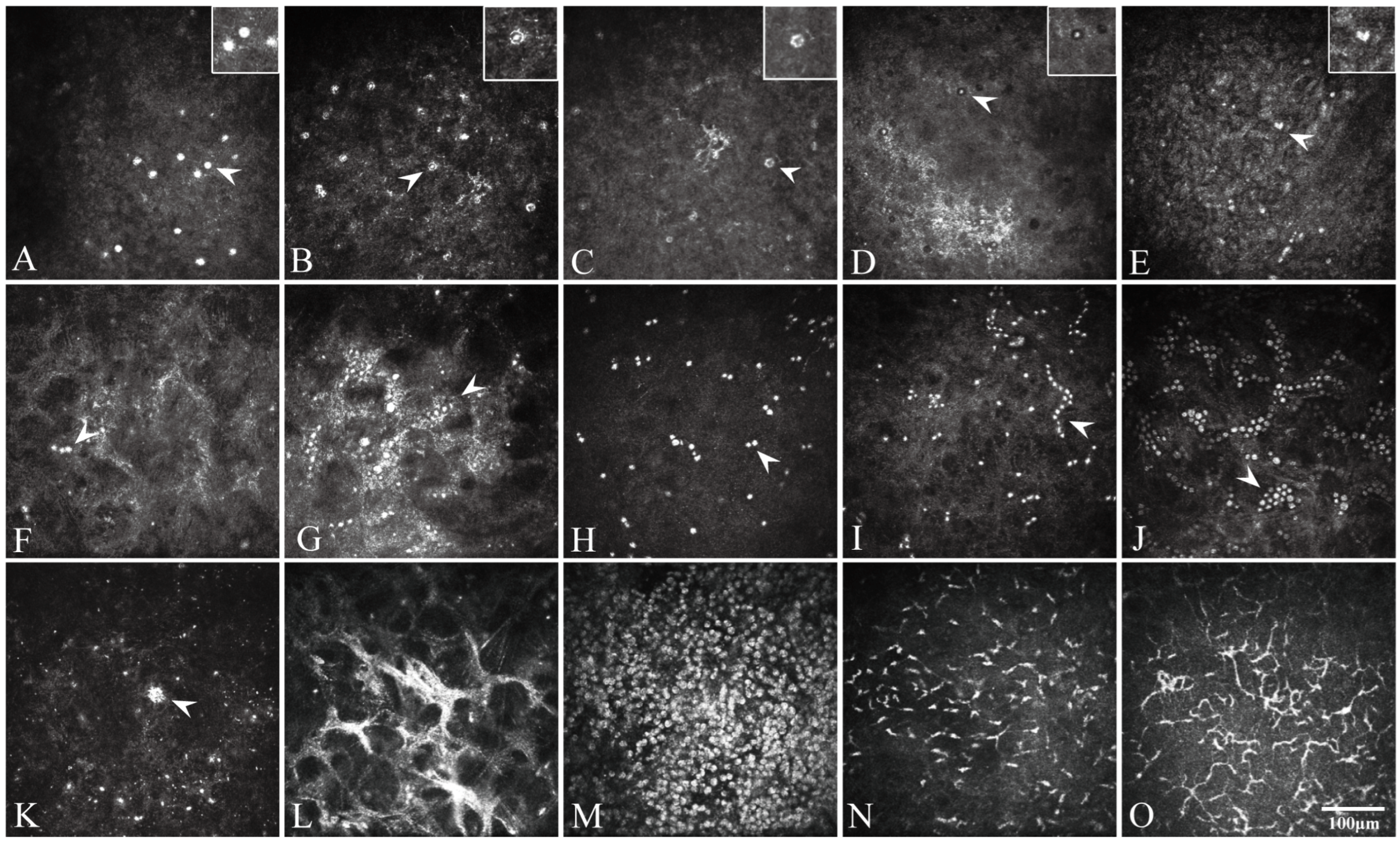

| Classification | Morphological Description | Size (μm) | Reference |
|---|---|---|---|
| Cyst | |||
| Bright spot | Round, oval, hyper-reflective particles or refractile bodies typically measure | 10–30 | [3,6,10] |
| Double wall | Round structures with a bi-layered or double walled hyper-reflective appearance | 10–30 | [20] |
| Signet ring | Hyper-reflective wall or outer ring with a dark center or less reflectivity inside the ring | 5–20 | [3,20] |
| Target sign | Low-refractile outer wall with a halo and a bright center or hyper-reflective nucleus | 5–20 | [6] |
| Coffee bean-shaped | Paired cyst reflections resembling kidney or coffee beans, usually measuring | 5–20 | [5,10] |
| Polygonal or stellate | Polygonal or stellate hyper-reflective objects | 10–30 | [7] |
| Binary cysts | Paired or closely adjacent round or ovoid hyper-reflective structures | 5–20 | [20] |
| Single chain | Linear or single-file arrangement of round or ovoid hyper-reflective objects | 5–20 | [20] |
| Cluster of cysts | Clusters of round or ovoid hyper-reflective objects | 5–20 | [20] |
| Trophozoite | Irregular, pear or wedge shaped hyper-reflective structures with acanthopodia or pseudopods | 20–100 | [3,20] |
| Inflammatory cell | |||
| Immature DCs | Round or oval shape with fewer and shorter projections | 10–15 | [5,20] |
| Mature DCs | Bright specular structures with dendriform processes | 15–55 | [5,20] |
| Others | Irregular, oval-shaped hyper-reflective structures, lacking dendritic processes or external walls, resembling leukocytes or neutrophils | 10–40 | [6,20] |
| Activated keratocyte | High reflectivity arranged in a honeycomb pattern | 50–75 | [5,20] |
| Parameters | Baseline | 1 Month | 3 Months |
|---|---|---|---|
| Visual acuity, n (%) | |||
| <0.05 | 14 (23.7) | 14 (23.7) | 13 (22.0) |
| 0.05~0.1 | 12 (20.3) | 5 (8.5) | 3 (5.1) |
| 0.1~0.3 | 28 (47.5) | 32 (54.2) | 27 (45.8) |
| ≥0.3 | 5 (8.5) | 8 (13.6) | 16 (27.1) |
| Clinical signs, n (%) | |||
| Epithelial lesions | 34 (57.6) | 31 (52.5) | 32 (54.2) |
| Deep stromal infiltrates | 28 (47.4) | 25 (42.3) | 15 (25.4) |
| Ring infiltrates | 25 (42.4) | 21 (35.6) | 10 (16.9) |
| Groove-shaped melting | 15 (25.4) | 15 (25.4) | 9 (15.2) |
| Neovascularization | 21 (35.6) | 21 (35.6) | 18 (30.5) |
| Clinical stage, n (%) | |||
| Stage 1 | 6 (10.2) | 14 (23.7) | 30 (50.7) |
| Stage 2 | 28 (47.4) | 24 (40.7) | 19 (32.3) |
| Stage 3 | 25 (42.4) | 21 (35.6) | 10 (17.0) |
| Good Outcome | Poor Outcome | p-Value | |
|---|---|---|---|
| Age [Median (IQR), years] | 27.0 (17.0–50.0) | 58.5 (42.3–63.8) | 0.0008 |
| Gender, n (%) | 0.803 | ||
| Male | 24 (53.3) | 8 (57.1) | |
| Female | 21 (46.7) | 6 (42.9) | |
| IVCM morphological signs, n (%) | |||
| Pathogens | |||
| Bright spot cyst | 35 (77.7) | 11 (78.5) | 1.000 |
| Double-walled cyst | 13 (28.8) | 9 (64.2) | 0.017 |
| Signet ring cyst | 19 (42.2) | 5 (35.7) | 0.665 |
| Target sign | 27 (60.0) | 3 (21.4) | 0.015 |
| Coffee bean–shaped cyst | 6 (13.3) | 2 (14.3) | 1.000 |
| Polygonal or stellate cyst | 23 (51.1) | 8 (57.1) | 0.693 |
| Trophozoite | 15 (33.3) | 9 (64.2) | 0.039 |
| Cyst Arrangement, n (%) | |||
| Binary cyst | 27 (60.0) | 9 (64.2) | 0.774 |
| Single chain of cysts | 20 (44.4) | 10 (71.4) | 0.125 |
| Cluster of cysts | 18 (40.0) | 11 (78.5) | 0.015 |
| Inflammatory Response, n (%) | |||
| Activated keratocyte | 36 (80.0) | 10 (71.4) | 0.485 |
| Inflammatory cells | 18 (40.0) | 9 (64.3) | 0.314 |
| Immature dendritic cells | 35 (77.7) | 7 (50.0) | 0.045 |
| Mature dendritic cells | 33 (73.3) | 3 (21.4) | 0.001 |
| Variable | Logistic Coefficient | Odds Ratio (95% CI) | p-Value |
|---|---|---|---|
| Target sign | −1.3456 | 0.26 (0.10–0.65) | 0.0011 |
| Cluster of cysts | 1.0934 | 2.98 (1.24–7.19) | 0.0102 |
| Mature dendritic cells | −1.0035 | 0.37 (0.17–0.79) | 0.0075 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Han, Y.; Wei, Y.; Chen, Q.; Pang, J.; Shi, Q.; Liang, Q. In Vivo Confocal Microscopy as a Prognostic Indicator in Acanthamoeba Keratitis: Insights from a Retrospective Study. Pathogens 2026, 15, 262. https://doi.org/10.3390/pathogens15030262
Han Y, Wei Y, Chen Q, Pang J, Shi Q, Liang Q. In Vivo Confocal Microscopy as a Prognostic Indicator in Acanthamoeba Keratitis: Insights from a Retrospective Study. Pathogens. 2026; 15(3):262. https://doi.org/10.3390/pathogens15030262
Chicago/Turabian StyleHan, Yiping, Yuan Wei, Qiankun Chen, Jinding Pang, Qingquan Shi, and Qingfeng Liang. 2026. "In Vivo Confocal Microscopy as a Prognostic Indicator in Acanthamoeba Keratitis: Insights from a Retrospective Study" Pathogens 15, no. 3: 262. https://doi.org/10.3390/pathogens15030262
APA StyleHan, Y., Wei, Y., Chen, Q., Pang, J., Shi, Q., & Liang, Q. (2026). In Vivo Confocal Microscopy as a Prognostic Indicator in Acanthamoeba Keratitis: Insights from a Retrospective Study. Pathogens, 15(3), 262. https://doi.org/10.3390/pathogens15030262




