Oral Route Infection by Trypanosoma cruzi: From the Beginning to the Present Day
Abstract
1. Introduction
2. The Beginning of Oral Transmission by T. cruzi
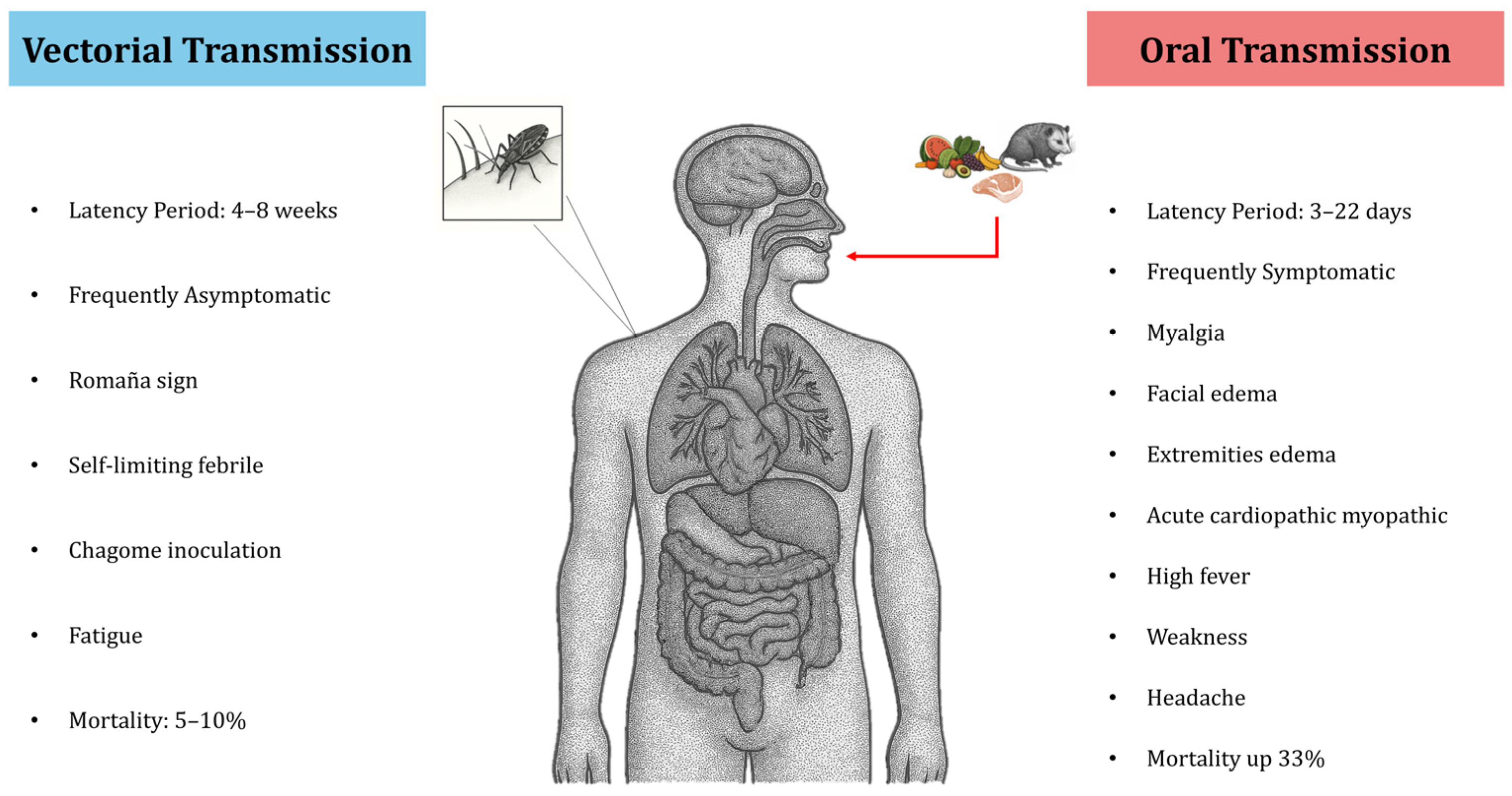
3. The Current Role of the Oral Route in Human T. cruzi Infection
- Tourist or non-local populations visiting tropical areas of countries like Brazil, Colombia, or Venezuela, who may consume local foods, juices, or fruit pulps that are contaminated with infected triatomine feces, or who eat raw/undercooked “exotic” meats (such as armadillo).
- Communities that consume meat from free-ranging domestic or wild animals (e.g., rabbits, hares, or goats) in areas where wild triatomines are present.
4. Clinical Aspects of Orally Acquired Chagas Disease
Clinical and Laboratory Diagnostics
5. Oral Infection by T. cruzi in Animal Models
6. Animals as Reservoirs and Spreading Factors
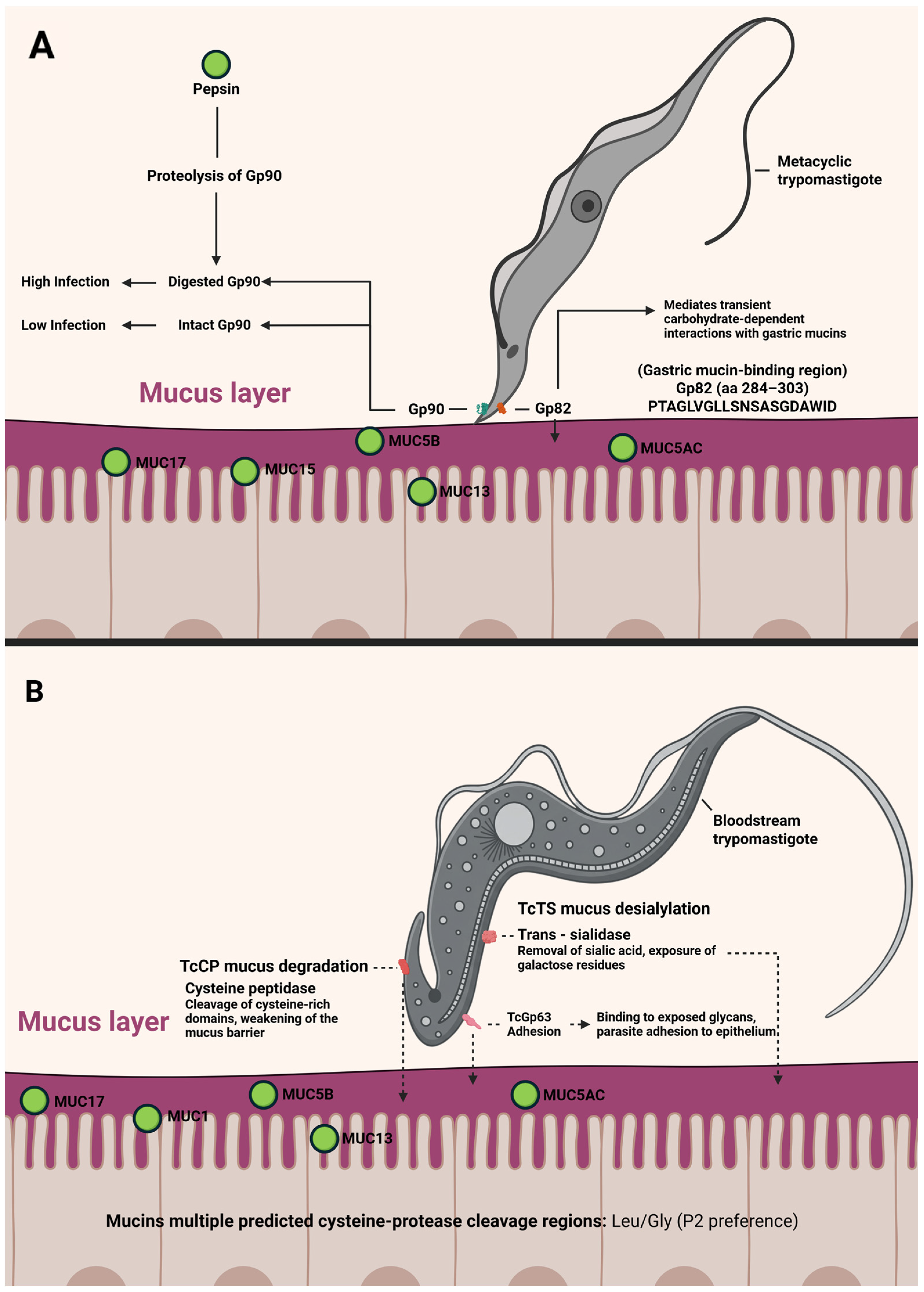
7. What We Know About T. cruzi DTUs and Mechanisms Involved in Oral Infections
8. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Chagas, C. Nova tripanozomiaze humana: Estudos sobre a morfolojia e o ciclo evolutivo do Schizotrypanum cruzi n. gen., n. sp., ajente etiolojico de nova entidade morbida do homem. Mem. Inst. Oswaldo Cruz 1909, 1, 159–218. [Google Scholar] [CrossRef]
- Abril, M.; Abad, F.; Altcheh, J.; Angeleri, P.; Batista, C.; Albajar, P.; Bassani, R.; Bern, C.; Espinal, C.; Chuit, R.; et al. Enfermedad de Chagas en las Américas: Una revisión de la situación actual de salud pública y su visión para el futuro; Organización Mundial de La Salud: Geneva, Switzerland, 2018. [Google Scholar]
- Beatty, N.L.; Hamer, G.L.; Moreno-Peniche, B.; Mayes, B.; Hamer, S.A. Chagas disease, an endemic disease in the United States. Emerg. Infect. Dis. 2025, 31, 1691–1697. [Google Scholar] [CrossRef] [PubMed]
- Coura, J.R.; Vĩas, P.A. Chagas disease: A new worldwide challenge. Nature 2010, 465, S6–S7. [Google Scholar] [CrossRef]
- Bern, C. Chagas’ disease. N. Engl. J. Med. 2015, 373, 456–466. [Google Scholar] [CrossRef]
- Chagas Disease (American Trypanosomiasis). Available online: https://www.who.int/health-topics/chagas-disease#tab=tab_1 (accessed on 29 May 2025).
- Tyler, K.M.; Engman, D.M. The life cycle of Trypanosoma cruzi revisited. Int. J. Parasitol. 2011, 31, 472–481. [Google Scholar] [CrossRef]
- Tomasina, R.; González, F.C.; Cabrera, A.; Basmadjián, Y.; Robello, C. From Trypomastigotes to Trypomastigotes: Analyzing the one-way intracellular journey of Trypanosoma cruzi by ultrastructure expansion microscopy. Pathogens 2024, 13, 866. [Google Scholar] [CrossRef]
- Coura, J.R. The main sceneries of chagas disease transmission. The vectors, blood and oral transmissions—A comprehensive review. Mem. Inst. Oswaldo Cruz 2015, 110, 277–282. [Google Scholar] [CrossRef]
- de Noya, B.A.; González, O.N. An ecological overview on the factors that drives to Trypanosoma cruzi oral transmission. Acta Trop. 2015, 151, 95–102. [Google Scholar] [CrossRef]
- Coura, J.R. Transmission of chagasic infection by oral route in the natural history of Chagas disease. Rev. Soc. Bras. Med. Trop. 2006, 39, 113–117. [Google Scholar]
- Shikanai-Yasuda, M.A.; Carvalho, N.B. Oral Transmission of Chagas disease. Clin. Infect. Dis. 2012, 54, 845–852. [Google Scholar] [CrossRef] [PubMed]
- Bruneto, E.G.; Fernandes-Silva, M.M.; Toledo-Cornell, C.; Martins, S.; Ferreira, J.M.B.; Corrêa, V.R.; da Costa, J.M.; Pinto, A.Y.D.N.; de Sousa, D.S.M.; Pinto, M.C.G.; et al. Case-fatality from orally transmitted acute Chagas disease: A systematic review and meta-analysis. Clin. Infect. Dis. 2021, 72, 1084–1092. [Google Scholar] [CrossRef]
- Doença de Chagas Aguda—Casos Confirmados Notificados no Sistema de Informação de Agravos de Notificação-Brasil. Available online: http://tabnet.datasus.gov.br/cgi/tabcgi.exe?sinannet/cnv/chagasbr.def (accessed on 29 May 2025).
- Ano. Secretaria de Vigilância em Saúde. Ministério da Saúde Doença de Chaga, n.d. Available online: https://www.saude.gov.br/saude-de-a-z/d/doenca-de-chagas (accessed on 25 December 2025).
- Velásquez-Ortiz, N.; Herrera, G.; Hernández, C.; Muñoz, M.; Ramírez, J.D. Discrete typing units of Trypanosoma cruzi: Geographical and biological distribution in the Americas. Sci. Data 2022, 9, 360. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, N.M.; Miller, S.M.; Evora, P.R.B. The chronic gastrointestinal manifestations of Chagas disease. Clinics 2009, 64, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- de Noya, B.A.; Díaz-Bello, Z.; Colmenares, C.; Ruiz-Guevara, R.; Mauriello, L.; Muñoz-Calderón, A.; Noya, O. Update on oral Chagas disease outbreaks in Venezuela: Epidemiological, clinical and diagnostic approaches. Mem. Inst. Oswaldo Cruz 2015, 110, 377–386. [Google Scholar] [CrossRef]
- Benítez, J.A.; Araujo, B.; Contreras, K.; Rivas, M.; Ramírez, P.; Guerra, W.; Calderon, N.; Terren, C.A.; Barrera, R.; Rodriguez-Morales, A.J. Urban outbreak of acute orally acquired Chagas disease in Táchira, Venezuela. J. Infect. Dev. Ctries. 2013, 7, 638–641. [Google Scholar] [CrossRef]
- Gonçalves, K.R.; Mazzeti, A.L.; da Silva Nascimento, A.F.; Castro–Lacerda, J.M.; Nogueira-Paiva, N.C.; Mathias, F.A.S.; Reis, A.B.; Caldas, S.; Bahia, M.T. The entrance route: Oral, mucous, cutaneous, or systemic has a marked influence on the outcome of Trypanosoma cruzi experimental infection. Acta Trop. 2022, 234, 106581. [Google Scholar] [CrossRef]
- Barreto-de-Albuquerque, J.; Silva-dos-Santos, D.; Pérez, A.R.; Berbert, L.R.; de Santana-van-Vliet, E.; Farias-de-Oliveira, D.A.; Moreira, O.T.; Roggero, E.; Carvalho-Pinto, C.A.; Jurberg, J.; et al. Trypanosoma cruzi infection through the oral route promotes a severe infection in mice: New disease form from an old infection? PLoS Negl. Trop. Dis. 2015, 9, e0003849. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.R.; Gibson, W.C. The evolution of pathogenic trypanosomes. Cad. Saúde Pública. 1999, 15, 673–684. [Google Scholar] [CrossRef]
- Guhl, F.; Auderheide, A.; Ramírez, J.D. From ancient to contemporary molecular eco-epidemiology of Chagas disease in the Americas. Int. J. Parasitol. 2014, 44, 605–612. [Google Scholar] [CrossRef]
- Briones, M.R.S.; Souto, R.P.; Stolf, B.S.; Zingales, B. The evolution of two Trypanosoma cruzi subgroups inferred from rRNA genes can be correlated with the interchange of American mammalian faunas in the Cenozoic and has implications to pathogenicity and host specificity. Mol. Biochem. Parasitol. 1999, 104, 219–232. [Google Scholar] [CrossRef]
- Hamilton, P.B.; Teixeira, M.M.G.; Stevens, J.R. The evolution of Trypanosoma cruzi: The “bat seeding” hypothesis. Trends Parasitol. 2012, 28, 136–141. [Google Scholar] [CrossRef]
- Villena, F.E.; Gomez-Puerta, L.A.; Jhonston, E.J.; Del Alcazar, O.M.; Maguiña, J.L.; Albujar, C.; Laguna-Torres, V.A.; Recuenco, S.E.; Ballard, S.B.; Ampuero, J.S. First report of Trypanosoma cruzi infection in salivary gland of bats from the Peruvian Amazon. Am. J. Trop. Med. Hyg. 2018, 99, 723–728. [Google Scholar] [CrossRef]
- Henry, J.P. Genetics and origin of Homo sapiens. Medecine/Sciences 2019, 35, 39–45. [Google Scholar] [CrossRef]
- Ferreira, L.F.; Britto, C.; Cardoso, M.A.; Fernandes, O.; Reinhard, K.; Araújo, A. Paleoparasitology of Chagas disease revaled by infected tissues from Chilean mummies. Acta Trop. 2000, 75, 79–84. [Google Scholar] [CrossRef]
- Araújo, A.; Jansen, A.M.; Reinhard, K.; Ferreira, L.F. Paleoparasitology of Chagas disease—A review. Mem. Inst. Oswaldo Cruz 2009, 104, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Reinhard, K.J.; Fink, T.M.; Skiles, J. A case of megacolon in Rio Grande Valley as a possible case of Chagas disease. Mem. Inst. Oswaldo Cruz 2003, 98, 165–172. [Google Scholar] [CrossRef]
- Rothhammer, F.; Allison, M.J.; Núñez, L.; Standen, V.; Arriaza, B. Chagas’ disease in pre-Columbian South America. Am. J. Phys. Anthropol. 1985, 68, 495–498. [Google Scholar] [CrossRef]
- Aufderheide, A.C.; Salo, W.; Madden, M.; Streitz, J.; Buikstra, J.; Guhl, F.; Arriaza, B.; Renier, C.; Wittmers, L.E., Jr.; Fornaciari, G.; et al. A 9,000-year record of Chagas’ disease. Proc. Natl. Acad. Sci. USA 2004, 101, 2034–2039. [Google Scholar] [CrossRef] [PubMed]
- Lima, V.S.; Iniguez, A.M.; Otsuki, K.; Ferreira, L.F.; Araújo, A.; Vicente, A.C.P.; Jansen, A.M. Chagas disease in ancient Hunter-Gatherer population, Brazil. Emerg. Infect. Dis. 2008, 14, 1001–1002. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.; Iñiguez, A.M.; Lima, V.S.; Mendonça De Souza, S.M.F.; Ferreira, L.F.; Vicente, A.C.P.; Jansen, A.M. Pre-Columbian Chagas disease in Brazil: Trypanosoma cruzi I in the archaeological remains of a human in Peruaçu Valley, Minas Gerais, Brazil. Mem. Inst. Oswaldo Cruz 2008, 103, 514–516. [Google Scholar] [CrossRef]
- Reinhard, K.J. Archaeoparasitology in North America. Am. J. Phys. Anthropol. 1990, 82, 145–163. [Google Scholar] [CrossRef]
- Reinhard, K.J.; Geib, P.R.; Callahan, M.M.; Hevly, R.H. Discovery of colon contents in a skeletonized burial: Soil sampling for dietary remains. J. Archaeol. Sci. 1992, 19, 697–705. [Google Scholar] [CrossRef]
- Coimbra, C.E.A. Human settlements, demographic pattern, and epidemiology in lowland Amazonia: The case of Chagas’s disease. Am. Anthropol. 1988, 90, 82–97. [Google Scholar] [CrossRef]
- Reperant, L.A. Putative 14th century outbreak of foodborne Chagas disease, Mexico. Vector-Borne Zoonotic Dis. 2023, 23, 390–392. [Google Scholar] [CrossRef] [PubMed]
- Brumpt, E. Precis de parasitology. J. Parasitol. 1928, 14, 277. [Google Scholar] [CrossRef]
- Mayer, H. Infeccion experimental con Trypanosoma cruzi por via digestiva. An. Inst. Med. Reg. 1961, 5, 43–48. [Google Scholar]
- Da Silva, N.N.; Clausell, D.T.; Nólibos, H.; De Mello, A.L.; Ossanai, J.; Rapone, T.; Snell, T. Epidemic outbreak of Chagas disease probably due to oral contamination. Rev. Inst. Med. Trop. Sao Paulo 1968, 10, 265–276. [Google Scholar]
- Yaeger, R.G. Transmission of Trypanosoma cruzi infection to opossums via the oral route. J. Parasitol. 1971, 57, 1375–1376. [Google Scholar] [CrossRef]
- Barretto, M.P.; Ribeiro, R.D.; Belda Neto, F.M. Reservoirs and wild vectors of Trypanosoma cruzi: Infection of mammals by oral route. Rev. Bras. Biol. 1978, 38, 455–459. [Google Scholar]
- López-García, A.; Gilabert, J.A. Oral transmission of Chagas disease from a One Health approach: A systematic review. Trop. Med. Int. Health 2023, 28, 689–698. [Google Scholar] [CrossRef]
- Coura, J.R.; Junqueira, A.C.V.; Fernandes, O.; Valente, S.A.S.; Miles, M.A. Emerging Chagas disease in Amazonian Brazil. Trends Parasitol. 2002, 18, 171–176. [Google Scholar] [CrossRef]
- Sánchez, L.V.; Ramírez, J.D. Congenital and oral transmission of American trypanosomiasis: An overview of physiopathogenic aspects. Parasitology 2013, 140, 147–159. [Google Scholar] [CrossRef]
- Alarcón de Noya, B.; Colmenares, C.; Díaz-Bello, Z.; Ruiz-Guevara, R.; Medina, K.; Muñoz-Calderón, A.; Mauriello, L.; Cabrera, E.; Montiel, L.; Losada, S. Orally-transmitted Chagas disease: Epidemiological, clinical, serological and molecular outcomes of a school microepidemic in Chichiriviche de la Costa, Venezuela. Parasite Epidemiol. Control. 2016, 1, 188–198. [Google Scholar] [CrossRef]
- Benchimol Barbosa, P.R. The oral transmission of Chagas’ disease: An acute form of infection responsible for regional outbreaks. Int. J. Cardiol. 2006, 112, 132–133. [Google Scholar] [CrossRef] [PubMed]
- Dias, J.P.; Bastos, C.; Araújo, E.; Mascarenhas, A.V.; Netto, E.M.; Grassi, F.; Silva, M.; Tatto, E.; Mendoça, L.; Araujo, R.F.; et al. Acute Chagas disease outbreak associated with oral transmission. Rev. Soc. Bras. Med. Trop. 2008, 41, 296–300. [Google Scholar] [CrossRef]
- Ramírez, J.D.; Montilla, M.; Cucunubá, Z.M.; Floréz, A.C.; Zambrano, P.; Guhl, F. Molecular epidemiology of human oral Chagas disease outbreaks in Colombia. PLoS Negl. Trop. Dis. 2013, 7, e2041. [Google Scholar] [CrossRef]
- Blanchet, D.; Brenière, S.F.; Schijman, A.G.; Bisio, M.; Simon, S.; Véron, V.; Mayence, C.; Demar-Pierre, M.; Djossou, F.; Aznar, C. First report of a family outbreak of Chagas disease in French Guiana and posttreatment follow-up. Infect. Gen. Evol. 2014, 28, 245–250. [Google Scholar] [CrossRef]
- Ferreira, C.S.; Martinho, P.C.; Neto, V.A.; Cruz, R.R.B. Pasteurization of human milk to prevent transmission of Chagas disease. Rev. Inst. Med. Trop. Sao Paulo 2001, 43, 161–162. [Google Scholar] [CrossRef] [PubMed]
- Pereira, K.S.; Schmidt, F.L.; Guaraldo, A.M.A.; Franco, R.M.B.; Dias, V.L.; Passos, L.A.C. Chagas’ disease as a foodborne illness. J. Food Prot. 2009, 72, 441–446. [Google Scholar] [CrossRef]
- Barbosa-Ferreira, J.M.; Guerra, J.A.D.O.; De Santana Filho, F.S.; Magalhães, B.M.L.; Coelho, L.I.A.R.C.; Barbosa, M.D.G.V. Cardiac involvement in acute Chagas’ disease cases in the Amazon region. Arq. Bras. Cardiol. 2010, 94, 147–149. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Simões-Neto, E.A.; Santos, D.W.d.C.L.; Bomfim, M.R.Q.; Costa, J.M.L.; Simões, A.F.; Vasconcelos, L.D.; Sodré, D.C.; Costa, A.C.M.; Dumont, S.V.R.; de Melo, B.O.; et al. Oral Chagas disease outbreak by bacaba juice ingestion: A century after Carlos Chagas’ discovery, the disease is still hard to manage. PLoS Negl. Trop. Dis. 2024, 18, e0012225. [Google Scholar] [CrossRef]
- Santalla Vargas, J.; Carrasco, P.O.; Espinoza, E.; Rios, T.; Brutus, L. First reported outbreak of Chagas disease in the Bolivian Amazonean zone: A report of 14 cases of oral transmission of acute Trypanosoma cruzi in Guayaramerín, Beni-Bolivia. Biofarbo 2011, 19, 52–58. [Google Scholar]
- Neves da Silva, N.; Clausell, D.T.; Nólib, H.; Leite de Mello, A.; Üssanai, J.; Rapone, T.; Snell, T. Surto epidêmico de doença de Chagas com provável contaminação oral. Rev. Inst. Med. Trop. Sao Paulo 1968, 10, 265–276. [Google Scholar]
- Belém, E.M.; Shaw, J.; Lainson, R. Considerações sôbre a epidemiología dos primeiros casos autóctones de doença de Chagas registrados em Belém, Pará, Brasil. Rev. Saude Publica 1969, 3, 153–157. [Google Scholar]
- Rodrigues, I.D.C.; Souza, A.A.D.; Terceros, R.; Valente, S. Doença de Chagas na amazónia: I. Registro de oito casos autóctones em macapá. Rev. Soc. Bras. Med. Trop. 1988, 21, 193–197. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Shikanai-Yasuda, M.A.; Marcondes, C.B.; Guedes, L.A.; Siqueira, G.S.; Barone, A.A.; Dias, J.C.; Neto, V.A.; Tolezano, J.E.; Peres, B.A.; Arrauda, E.R., Jr.; et al. Possible oral transmission of acute Chagas disease in Brazil. Rev. Inst. Med. Trop. São Paulo 1991, 33, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Viana, S.; Farias, E.; Lima, F.; Batista, L.; Vieira, A.; Silva, L.; Lobato, C.; Nascimento, S.; Chalub, S. Doença de Chagas no Estado do Acre, registro de 3 casos de miocardiopatia chagásica aguda autóctone no município de Rio Branco, 1993. Rev. Soc. Bras. Med. Trop. 1994, 27, 77. [Google Scholar]
- Valente, S.A.d.S.; da Costa Valente, V.; das Neves Pinto, A.Y.; de Jesus Barbosa César, M.; dos Santos, M.P.; Miranda, C.O.S.; Cuervo, P.; Fernandes, O. Analysis of an acute Chagas disease outbreak in the Brazilian Amazon: Human cases, triatomines, reservoir mammals and parasites. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 291–297. [Google Scholar] [CrossRef]
- Pinto, A.Y.; Harada, G.S.; Valente Vd Abud, J.E.; Gomes Fd Souza, G.C.; Valente, S.A. Cardiac attacks in patients with acute Chagas disease in a family micro-outbreak, in Abaetetuba, Brazilian Amazon. Rev. Soc. Bras. Med. Trop. 2001, 34, 413–419. [Google Scholar] [CrossRef]
- Pinto, A.Y.d.N.; Valente, S.A.d.S.; Valente, V.d.C. Emerging acute Chagas disease in Amazonian Brazil: Case reports with serious cardiac involvement. Braz. J. Infect. Dis. 2004, 8, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Pinto, A.Y.; Farias, J.R.; Marcal, A.S.; Galucio, A.L.; Costi, R.R.; Valente, V.C.; Silva Valente, S.A. Doença de Chagas Aguda grave autóctone na Amazonia brasileira. Rev. Para. Med. 2007, 21, 7–12. [Google Scholar]
- De Medeiros, M.B.; Guerra, J.A.D.O.; De Lacerda, M.V.G. Meningoencephalitis in a patient with acute Chagas disease in the Brazilian Amazon. Rev. Soc. Bras. Med. Trop. 2008, 41, 520–521. [Google Scholar] [CrossRef]
- Pinto, A.Y.D.N.; Valente, S.A.; Valente, V.D.C.; Ferreira, A.G.; Coura, J.R. Acute phase of Chagas disease in the Brazilian Amazon region: Study of 233 cases from Pará, Amapá and Maranhão observed between 1988 and 2005. Rev. Soc. Bras. Med. Trop. 2008, 41, 602–614. [Google Scholar] [CrossRef]
- Borborema, M.; Guerra, J.; Malheiros, R.; Fé, N.; Lacerda, M.; Coelho, L.D. Doença de Chagas urbana em Tefé-AM: Relato de nove casos com suspeita de transmissão oral. Rev. Soc. Bras. Med. Trop. 2005, 38, 491. [Google Scholar]
- Steindel, M.; Kramer Pacheco, L.; Scholl, D.; Soares, M.; de Moraes, M.H.; Eger, I.; Kosmann, C.; Sincero, T.C.M.; Stoco, P.H.; Murta, S.M.F.; et al. Characterization of Trypanosoma cruzi isolated from humans, vectors, and animal reservoirs following an outbreak of acute human Chagas disease in Santa Catarina State, Brazil. Diagn. Microbiol. Infect. Dis. 2008, 60, 25–32. [Google Scholar] [CrossRef]
- Nóbrega, A.A.; Garcia, M.H.; Tatto, E.; Obara, M.T.; Costa, E.; Sobel, J.; Aruajo, W.N. Oral transmission of Chagas disease by consumption of açaí palm fruit, Brazil. Emerg. Infect. Dis. 2009, 15, 653–655. [Google Scholar] [CrossRef] [PubMed]
- de Góes Cavalcanti, L.P.; Rolim, D.A.; Pires Neto, J.A.; Feitosa Vilar, D.C.L. Microepidemia de doença de Chagas aguda por transmissão oral no Ceará. Cad. Saúd Colet. 2009, 17, 911–927. [Google Scholar]
- Bastos, C.J.C.; Aras, R.; Mota, G.; Reis, F.; Dias, J.P.; de Jesus, R.S.; Freire, M.S.; Araújo, E.G.; Prazeres, J.; Grassi, M.F.R. Clinical outcomes of thirteen patients with acute Chagas disease acquired through oral transmission from two urban outbreaks in northeastern Brazil. PLoS Negl Trop. Dis. 2010, 4, e711. [Google Scholar] [CrossRef]
- de Barros Moreira Beltrão, H.; de Paula Cerroni, M.; de Freitas, D.R.C.; Das Neves Pinto, A.Y.; da Costa Valente, V.; Valente, S.A.; Costa, E.G.; Sobel, J. Investigation of two outbreaks of suspected oral transmission of acute Chagas disease in the Amazon region, Para State, Brazil, in 2007. Trop. Dr. 2009, 39, 231–232. [Google Scholar] [CrossRef]
- Monteiro, W.M.; Magalhães, L.K.; Santana Filho, F.S.; Borborema, M.; Silveira, H.; Barbosa, M.D.G.V. Trypanosoma cruzi TcIII/Z3 genotype as agent of an outbreak of Chagas disease in the Brazilian Western Amazonia. Trop. Med. Int. Health 2010, 15, 1049–1051. [Google Scholar] [CrossRef]
- Da Costa Valente, V.; das Neves Pinto, A.Y. Doença de Chagas congênita por infecção aguda maternal por Trypanosoma cruzi transmitida via oral. Rev. Pan-Amaz. Saude 2011, 2, 89–94. [Google Scholar]
- Borba, C.C.; Maués Pereira Moia, J.; de Souza, C.B.; de Barros Barreto, J. Doença de Chagas aguda grave: Relato de caso. Rev. Para. Med. 2010, 24, 39–44. [Google Scholar]
- de Souza-Lima, R.d.C.; Barbosa, M.d.G.V.; Coura, J.R.; Arcanjo, A.R.L.; Nascimento, A.d.S.; Ferreira, J.M.B.B.; Magalhães, L.K.; Alburquerque, B.C.; Araújo, G.A.N.; Guerra, J.A.O. Outbreak of acute Chagas disease associated with oral transmission in the Rio Negro region, Brazilian Amazon. Rev. Soc. Bras. Med. Trop. 2013, 46, 510–514. [Google Scholar] [CrossRef]
- Barbosa, M.d.G.V.; Ferreira, J.M.B.B.; Arcanjo, A.R.L.; Santana, R.A.G.; Magalhães, L.K.C.; Magalhães, L.K.C.; Mota, D.T.; Fé, N.F.; Monteiro, W.M.; Silveira, H.; et al. Chagas disease in the State of Amazonas: History, epidemiological evolution, risks of endemicity and future perspectives. Rev. Soc. Bras. Med. Trop. 2015, 48, 27–33. [Google Scholar] [CrossRef]
- de Góes Costa, E.; dos Santos, S.O.; Sojo-Milano, M.; Amador, E.C.C.; Tatto, E.; Souza, D.S.M.; Costa, F.; Póvoa, R.M.S. Acute Chagas Disease in the Brazilian Amazon: Epidemiological and clinical features. Int. J. Cardiol. 2017, 235, 176–178. [Google Scholar] [CrossRef]
- Vargas, A.; Malta, J.M.A.S.; da Costa, V.M.; Cláudio, L.D.G.; Alves, R.V.; Cordeiro, G.D.S.; Aguiar, L.M.A.; Percio, J. Investigation of an outbreak of acute Chagas disease outside the Amazon Region, in Rio Grande do Norte State, Brazil, 2016. Cad. Saude Publica 2018, 34, e00006517. [Google Scholar] [PubMed]
- de Souza, D.d.S.M.; Araujo, M.T.F.; Garcez, P.d.S.; Furtado, J.C.B.; Figueiredo, M.T.S.; Povoa, R.M.S. Anatomopathological aspects of acute Chagas myocarditis by oral transmission. Arq. Bras. Cardiol. 2016, 107, 77–80. [Google Scholar] [CrossRef] [PubMed]
- de Freitas, V.L.T.; Piotto, M.R.; Esper, H.R.; Nakanishi, E.Y.S.; Fonseca, C.d.A.; Assy, J.G.P.L.; Berreta, O.C.P.; França, F.O.S.; Lopes, M.H. Detection of Trypanosoma cruzi DTUs TcI and TcIV in two outbreaks of orally-transmitted Chagas disease in the Northern region of Brazil. Rev. Inst. Med. Trop. Sao Paulo 2023, 65, e7. [Google Scholar] [CrossRef]
- de Sousa, D.R.T.; de Oliveira Guerra, J.A.; Ortiz, J.V.; do Nascimento Couceiro, K.; da Silva e Silva, M.R.H.; Jorge Brandão, A.R.; Guevara, E.; Arcanjo, A.R.L.; de Oliveira Júnior, E.F.; Smith-Doria, S.; et al. Acute Chagas disease associated with ingestion of contaminated food in Brazilian western Amazon. Trop. Med. Int. Health 2023, 28, 541–550. [Google Scholar] [CrossRef]
- de Brito, A.K.S.B.; de Sousa, D.R.T.; da Silva Junior, E.F.; da Silva Ruiz, H.J.; Arcanjo, A.R.L.; Ortiz, J.V.; de Brito, S.S.; Jesus, D.V.; de Lima, J.R.C.; Couceiro, K.N.; et al. Acute micro-outbreak of Chagas disease in the southeastern Amazon: A report of five cases. Rev. Soc. Bras. Med. Trop. 2022, 55, e0687-2021. [Google Scholar] [CrossRef]
- Hernandez, L.M.; Ramirez, A.N.; Cucunubá, Z.; Bact, P.Z. Brote de Chagas agudo en Lebrija, Santander 2008. Rev. Obs. Salud Pública Santander 2009, 4, 28–36. [Google Scholar]
- Zambrano, P.; Cucunubá, Z.; Montilla, M.; Flórez, C.; Parra, E.; Cortés, L. Brotes de síndrome febril asociado a miocarditis aguda chagásica de posible transmisión oral en el departamento de Santander, diciembre de 2008 a mayo de 2009. Inf. Quinc. Epidemiol. Nac. 2010, 15, 17–32. [Google Scholar]
- Rios, J.F.; Arboleda, M.; Montoya, A.N.; Alarcon, E.P.; Parra-Henao, G.J. Probable brote de transmisión oral de enfermedad de Chagas en Turbo, Antioquia. Biomedica 2011, 31, 185–195. [Google Scholar] [CrossRef] [PubMed]
- Soto, H.; Tibaduiza, T.; Montilla, M.; Triana, O.; Suárez, D.C.; Torres, M.T.; Arias, M.T.; Lugo, L. Investigación de vectores y reservorios en brote de Chagas agudo por posible transmisión oral en Aguachica, Cesar, Colombia. Cad. Saude Publica 2014, 30, 746–756. [Google Scholar] [CrossRef]
- Zuleta-Dueñas, L.P.; López-Quiroga, Á.J.; Torres-Torres, F.; Castañeda-Porras, O. Possible oral transmission of Chagas disease among hydrocarbons sector workers in Casanare, Colombia, 2014. Biomedica 2017, 37, 218–232. [Google Scholar] [PubMed]
- Segura-Alba, M.L.; Hernandez, C.; Guerra, A.P.; Luna, N.; Cortes, L.J.; Acevedo, C.R.; Ballesteros, N.; Ayala, M.S.; Vera, M.J.; Diaz, R.A.C.; et al. Acute Chagas disease outbreaks in Colombia in 2019. IJID Reg. 2024, 12, 100410. [Google Scholar] [CrossRef] [PubMed]
- Vergara, H.D.; Gómez, C.H.; Faccini-Martínez, Á.A.; Herrera, A.C.; López, M.J.; Camacho, C.; Muñoz, L.; Cruz.Saavedra, L.; Hernández, C.; Ramírez, J.D. Acute Chagas Disease outbreak among military personnel, Colombia, 2021. Emerg. Infect. Dis. 2023, 29, 1882. [Google Scholar] [CrossRef]
- Gutierrez, S.A.; Jaimes-Dueñez, J.; Cruz-Saavedra, L.; Hernandez, C.; Cantillo-Barraza, O.; Alvarez, F.; Blanco, M.; Leal, B.; Marínez, L.; Medina, M.; et al. An Outbreak of acute Chagas disease possibly spread through oral transmission involving animal reservoirs in eastern Colombia. Am. J. Trop. Med. Hyg. 2023, 110, 36. [Google Scholar] [CrossRef]
- Benítez, J.Z.; Díaz, D.C.; Colorado, L.A.; Murillo, L.M.; Orozco, M.T.; Vallecilla, S.; Padilla, J.C.; Olivera, M.J. First report of an acute case of Chagas disease in the municipality of Miraflores, Guaviare, Colombia. Rev. Peru. Med. Exp. Salud Publica 2024, 41, 203. [Google Scholar] [CrossRef]
- Martín, A.; Alarcón de Noya, B.; Montero, R.; Rojas, C.; Garrido, E.; Ruiz-Guevara, R.; Diaz-Bello, Z. Archivos venezolanos de puericultura y pediatría. Arch. Venez. Pueric. Pediatr. 1939, 72, 97–100. [Google Scholar]
- De Noya, B.A.; Díaz-Bello, Z.; Colmenares, C.; Ruiz-Guevara, R.; Mauriello, L.; Zavala-Jaspe, R.; Suarez, J.A.; Abate, T.; Naranjo, L.; Paiva, M.; et al. Large urban outbreak of orally acquired acute Chagas disease at a school in Caracas, Venezuela. J. Infect. Dis. 2010, 201, 1308–1315. [Google Scholar] [CrossRef]
- de Noya, B.; Martinez, J. Transmision oral de la enfermedad de Chagas en Venezuela: Un segundo brote escolar. Rev. Fac. Cienc. Salud 2009, 13, 9. [Google Scholar]
- Añez, N.; Crisante, G.; Rojas, A.; Dávila, D. Boletín de malariología y salud ambiental. Bol. Malariol. Salud Ambient. 2003, 53, 01–10. [Google Scholar]
- Alcaíno, T.; Lorca-Herrera, M.; Issotta, A.; Gorman, T. Chagas’ disease in goats from the metropolitan region, Chile: Seroepidemiological survey and experimental infection. Parasitol. Día 1995, 19, 30–36. [Google Scholar]
- Ulloa, M.; Traslaviña, M.; Alcaíno, H.; Apt, W.; Sandoval, J. Enfermedad de Chagas en caninos y caprinos sinantrópicos de la provincia del Choapa (IV Región), Chile. Parasitol. Día 1989, 13, 120–124. [Google Scholar]
- Garrido, R.; Bacigalupo, A.; Peña-Gómez, F.; Bustamante, R.O.; Cattan, P.E.; Gorla, D.E.; Botto-Mahan, C. Potential impact of climate change on the geographical distribution of two wild vectors of Chagas disease in Chile: Mepraia spinolai and Mepraia gajardoi. Parasit. Vectors 2019, 12, 478. [Google Scholar] [CrossRef] [PubMed]
- Bezerra, C.M.; de Góes Cavalcanti, L.P.; de Souza, R.d.C.M.; Barbosa, S.E.; Xavier, S.C.d.C.; Jansen, A.M.; Ramalho, R.D.; Diotaiut, L. Domestic, Peridomestic and wild hosts in the transmission of Trypanosoma cruzi in the Caatinga area colonised by Triatoma brasiliensis. Mem. Inst. Oswaldo Cruz 2014, 109, 887–898. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, A.M.; Wisnivesky-Coli, C.; Gürtler, R.; Lazzari, J.; Bujas, M.A.; Segura, E.L. Trypanosoma cruzi infection in humans, dogs and goats in rural areas of the province of Córdoba. Medicina 1985, 45, 539–546. [Google Scholar]
- de Araújo-Neto, V.T.; Barbosa-Silva, A.N.; Medeiros Honorato, N.R.; Sales, L.M.L.; de Cassia Pires, R.; do Nascimento Brito, C.R.; da Matta Guedes, P.M.; da Cunha Galvão, L.M.; da Camara, A.C.J. Molecular identification of Trypanosoma cruzi in domestic animals in municipalities of the State of Rio Grande do Norte, Brazil. Parasitol. Res. 2023, 122, 207–215. [Google Scholar] [CrossRef]
- Barbosa, R.L.; Dias, V.L.; Pereira, K.S.; Schmidt, F.L.; Franco, R.M.B.; Guaraldo, A.M.A.; Alves, D.P.; Passos, L.A.C. Survival in vitro and virulence of Trypanosoma cruzi in açaí pulp in experimental acute Chagas disease. J. Food Prot. 2012, 75, 601–606. [Google Scholar] [CrossRef]
- Barbosa, R.L.; Pereira, K.S.; Dias, V.L.; Schmidt, F.L.; Alves, D.P.; Guaraldo, A.M.A.; Passos, L.A.C. Virulence of Trypanosoma cruzi in açai (Euterpe oleraceae Martius) pulp following mild heat treatment. J. Food Prot. 2016, 79, 1807–1812. [Google Scholar] [CrossRef]
- Suzuki, A.F.; de Souza, E.R.; Spadella, M.A.; Chagas, E.F.B.; Hataka, A.; Martins, L.P.A. Oral infection and survival of Trypanosoma cruzi in sugarcane juice conditioned at different temperatures. Acta Parasitol. 2024, 69, 251–259. [Google Scholar] [CrossRef]
- de Sousa, A.S.; Vermeij, D.; Ramos, A.N.; Luquetti, A.O. Chagas disease. Lancet 2024, 403, 203–218. [Google Scholar] [CrossRef]
- Cordeiro da Silva, A.; Calogeropoulou, T.; Costi, M.; Alunda, J. Drugs for vector-borne protozoal diseases in a one health scenario. A european perspective. ACS Infect. Dis. 2024, 10, 3715–3720. [Google Scholar] [CrossRef] [PubMed]
- Filigheddu, M.T.; Górgolas, M.; Ramos, J.M. Orally-transmitted Chagas disease. Med. Clin. 2017, 148, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Añez, N.; Crisante, G.; Rojas, A.; Dávila, D. Acute Chagas disease outbreak of possible oral transmission in Merida, Venezuela[Brote de enfermedad de Chagas agudo de posible transmisión oral en Mérida, Venezuela]. Bol. Malariol. Salud Ambient. 2013, 53, 1–11. [Google Scholar]
- Pittella, J.E.H. Central nervous system involvement in Chagas disease: A hundred-year-old history. Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 973–978. [Google Scholar] [CrossRef]
- Dos Santos, V.R.C.; Antunes, D.; de Souza, D.D.S.M.; Moreira, O.C.; Lima, I.C.d.A.; Farias-De-oliveira, D.A.; Lobo, J.P.; de Meis, E.; Coura, J.R.; Svino, W.; et al. Human acute Chagas disease: Changes in factor VII, activated protein C and hepatic enzymes from patients of oral outbreaks in Pará state (Brazilian Amazon). Mem. Inst. Oswaldo Cruz 2020, 115, e190364. [Google Scholar] [CrossRef]
- Feilij, H.; Muller, L.; Gonzalez Cappa, S.M. Direct micromethod for diagnosis of acute and congenital Chagas’ disease. J. Clin. Microbiol. 1983, 18, 327–330. [Google Scholar] [CrossRef]
- Luquetti, A.O.; Rassi, A. Diagnóstico laboratorial da infecção pelo Trypanosoma cruzi. In Trypanosoma cruzi e Doença de Chagas, 2nd. ed.; Guanabara Koogan: Rio de Janeiro, Brazil, 2000; pp. 344–378. [Google Scholar]
- Schijman, A.G.; Bisio, M.; Orellana, L.; Sued, M.; Duffy, T.; Mejia-Jaramillo, A.M.; Cura, C.; Auter, F.; Veron, V.; Qvarnstrom, Y.; et al. International study to evaluate PCR methods for detection of Trypanosoma cruzi DNA in blood samples from Chagas disease patients. PLoS Negl. Trop. Dis. 2011, 5, e931. [Google Scholar] [CrossRef]
- Franco-Paredes, C.; Villamil-Gómez, W.E.; Schultz, J.; Henao-Martínez, A.F.; Parra-Henao, G.; Rassi, A.; Rodríguez-Morales, A.J.; Suarez, J.A. A deadly feast: Elucidating the burden of orally acquired acute Chagas disease in Latin America—Public health and travel medicine importance. Travel. Med. Infect. Dis. 2020, 36, 101565. [Google Scholar] [CrossRef]
- Dias, G.B.M.; Gruendling, A.P.; Araújo, S.M.; Gomes, M.Ô.L.; Toledo, M.J.D.O. Evolution of infection in mice inoculated by the oral route with different developmental forms of Trypanosoma cruzi I and II. Exp. Parasitol. 2013, 135, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Silva-dos-Santos, D.; Barreto-de-Albuquerque, J.; Guerra, B.; Moreira, O.C.; Berbert, L.R.; Ramos, M.T.; Mascarenhas, B.A.S.; Britto, C.; Morrot, A.; Villa-Verde, D.M.S.; et al. Unraveling Chagas disease transmission through the oral route: Gateways to Trypanosoma cruzi infection and target tissues. PLoS Negl. Trop. Dis. 2017, 11, e0005507. [Google Scholar] [CrossRef] [PubMed]
- Antunes, D.; Marins-Dos-Santos, A.; Ramos, M.T.; Mascarenhas, B.A.S.; De Carvalho Moreira, C.J.; Farias-De-Oliveira, D.A.; Savino, W.; Monteiro, R.Q.; de Meis, J. Oral route driven acute Trypanosoma cruzi infection unravels an IL-6 dependent hemostatic derangement. Front. Immunol. 2019, 10, 1073. [Google Scholar] [CrossRef]
- Carvalho, L.M.; de Carvalho, T.V.; Ferraz, A.T.; Marques, F.d.S.; Roatt, B.M.; Fonseca, K.D.S.; Reis, L.E.S.; Carneiro, C.M.; de Abreu Vieira, P.M. Histopathological changes in the gastrointestinal tract and systemic alterations triggered by experimental oral infection with Trypanosoma cruzi. Exp. Parasitol. 2020, 218, 108012. [Google Scholar] [CrossRef]
- Marins-Dos-Santos, A.; Ayres-Silva, J.d.P.; Antunes, D.; Moreira, C.J.d.C.; Pelajo-Machado, M.; Alfaro, D.; Zapata, A.G.; Bonomo, A.C.; Savino, W.; de Meis, J.; et al. Oral Trypanosoma cruzi acute infection in mice targets primary lymphoid organs and triggers extramedullary hematopoiesis. Front. Cell Infect. Microbiol. 2022, 12, 800395. [Google Scholar] [CrossRef] [PubMed]
- Covarrubias, C.; Cortez, M.; Ferreira, D.; Yoshida, N. Interaction with host factors exacerbates Trypanosoma cruzi cell invasion capacity upon oral infection. Int. J. Parasitol. 2007, 37, 1609–1616. [Google Scholar] [CrossRef]
- Coffield, D.J.; Spagnuolo, A.M.; Shillor, M.; Mema, E.; Pell, B.; Pruzinsky, A.; Zetye, A. A Model for Chagas disease with oral and congenital transmission. PLoS ONE 2013, 8, e67267. [Google Scholar] [CrossRef]
- Guarner, J. Chagas disease as example of a reemerging parasite. Semin. Diagn. Pathol. 2019, 36, 164–169. [Google Scholar] [CrossRef]
- Muñoz-Calderón, A.; Díaz-Bello, Z.; Valladares, B.; Noya, O.; López, M.C.; Alarcón de Noya, B.; Thomas, M.C. Oral transmission of Chagas disease: Typing of Trypanosoma cruzi from five outbreaks occurred in Venezuela shows multiclonal and common infections in patients, vectors and reservoirs. Infect. Genet. Evol. 2013, 17, 113–122. [Google Scholar] [CrossRef]
- Yefi-Quinteros, E.; Muñoz-San Martín, C.; Bacigalupo, A.; Correa, J.P.; Cattan, P.E. Trypanosoma cruzi load in synanthropic rodents from rural areas in Chile. Parasit. Vectors 2018, 11, 171. [Google Scholar] [CrossRef] [PubMed]
- Coronado, X.; Rozas, M.; Botto-Mahan, C.; Ortíz, S.; Cattan, P.E.; Solari, A. Molecular epidemiology of Chagas disease in the wild transmission cycle: The evaluation in the sylvatic vector Mepraia spinolai from an endemic area of Chile. Am. J. Trop. Med. Hyg. 2009, 81, 656–659. [Google Scholar] [CrossRef] [PubMed]
- Ihle-Soto, C.; Costoya, E.; Correa, J.P.; Bacigalupo, A.; Cornejo-Villar, B.; Estadella, V.; Solari, A.; Ortiz, S.; Hernández, H.J.; Botto-Mahan, C.; et al. Spatio-temporal characterization of Trypanosoma cruzi infection and discrete typing units infecting hosts and vectors from non-domestic foci of Chile. PLoS Negl. Trop. Dis. 2019, 13, e0007170. [Google Scholar] [CrossRef] [PubMed]
- Galuppo, S.; Bacigalupo, A.; García, A.; Ortiz, S.; Coronado, X.; Cattan, P.E.; Solari, A. Predominance of Trypanosoma cruzi genotypes in two reservoirs infected by sylvatic Triatoma infestans of an endemic area of Chile. Acta Trop. 2009, 111, 90–93. [Google Scholar] [CrossRef]
- Rozas, M.; Botto-Mahan, C.; Coronado, X.; Ortiz, S.; Cattan, P.E.; Solari, A. Short report: Trypanosoma cruzi infection in wild mammals from a chagasic area of Chile. Am. J. Tropi Med. Hyg. 2005, 73, 517–519. [Google Scholar] [CrossRef]
- Botto-Mahan, C.; Correa, J.P.; Araya-Donoso, R.; Farías, F.; Juan, E.S.; Quiroga, N.; Campos-Soto, R.; Reyes-Olivares, C.; González-Acuña, D. Lizards as Silent Hosts of Trypanosoma cruzi. Emerg. Infect. Dis. 2022, 28, 1250–1253. [Google Scholar] [CrossRef]
- Cevidanes, A.; Di Cataldo, S.; Muñoz-San Martín, C.; Latrofa, M.S.; Hernández, C.; Cattan, P.E.; Otranto, D.; Millán, J. Co-infection patterns of vector-borne zoonotic pathogens in owned free-ranging dogs in central Chile. Vet. Res. Commun. 2023, 47, 575–588. [Google Scholar] [CrossRef]
- Reyes, C.; González, C.R.; Alvarado, S.; Flores, L.; Martin, C.; Oyarce, A.; Aylwin, M.P.; Canals, M.; Parra, A.; Valderrama, L. Chagas disease in northern Chile: Detection of Trypanosoma cruzi in children, dogs and triatomine bugs. Acta Trop. 2022, 235, 106631. [Google Scholar] [CrossRef]
- Botto-Mahan, C.; Cattan, P.E.; Medel, R. Chagas disease parasite induces behavioural changes in the kissing bug Mepraia spinolai. Acta Trop. 2006, 98, 219–223. [Google Scholar] [CrossRef]
- Llewellyn, M.S.; Miles, M.A.; Carrasco, H.J.; Lewis, M.D.; Yeo, M.; Vargas, J.; Torrico, F.; Diosque, P.; Valente, V.; Valente, S.A.; et al. Genome-scale multilocus microsatellite typing of Trypanosoma cruzi discrete typing unit I reveals phylogeographic structure and specific genotypes linked to human infection. PLoS Pathog. 2009, 5, e1000410. [Google Scholar] [CrossRef]
- Miles, M.A.; Llewellyn, M.S.; Lewis, M.D.; Yeo, M.; Baleela, R.; Fitzpatrick, S.; Gaunt, M.W.; Mauricio, I.L. The molecular epidemiology and phylogeography of Trypanosoma cruzi and parallel research on Leishmania: Looking back and to the future. Parasitology 2009, 136, 1509–1528. [Google Scholar] [CrossRef]
- O’Connor, O.; Bosseno, M.F.; Barnabé, C.; Douzery, E.J.P.; Brenière, S.F. Genetic clustering of Trypanosoma cruzi I lineage evidenced by intergenic miniexon gene sequencing. Infect. Genet. Evol. 2007, 7, 587–593. [Google Scholar] [CrossRef] [PubMed]
- Bosseno, M.F.; Barnabé, C.; Sierra, M.J.R.; Kengne, P.; Guerrero, S.; Lozano, F.; Ezequiel, K.; Gastélum, M.; Breniere, S.F. Short report: Wild ecotopes and food habits of Triatoma longipennis infected by Trypanosoma cruzi lineages I and II in Mexico. Am. J. Trop. Med. Hyg. 2009, 80, 98–991. [Google Scholar] [CrossRef]
- Cortez, M.; Neira, I.; Ferreira, D.; Luquetti, A.O.; Rassi, A.; Atayde, V.D.; Yoshida, N. Infection by Trypanosoma cruzi metacyclic forms deficient in gp82 but expressing a related surface molecule, gp30. Infect. Immun. 2003, 71, 6184–6191. [Google Scholar] [CrossRef] [PubMed]
- Cortez, M.; Silva, M.R.; Neira, I.; Ferreira, D.; Sasso, G.R.S.; Luquetti, A.O.; Rassi, A.; Yoshida, N. Trypanosoma cruzi surface molecule gp90 downregulates invasion of gastric mucosal epithelium in orally infected mice. Microbes Infect. 2006, 8, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Maeda, F.Y.; Clemente, T.M.; Macedo, S.; Cortez, C.; Yoshida, N. Host cell invasion and oral infection by Trypanosoma cruzi strains of genetic groups TcI and TcIV from chagasic patients. Parasit. Vectors 2016, 9, 189. [Google Scholar] [CrossRef]
- Barbosa, C.G.; Gómez-Hernández, C.; Rezende-Oliveira, K.; Da Silva, M.V.; Ferreira Rodrigues, J.P.; Tiburcio, M.G.S.; Ferreira, T.B.; Rodrigues, V.; Yoshida, N.; Ramirez, L.E. Oral infection of mice and host cell invasion by Trypanosoma cruzi strains from Mexico. Parasitol. Res. 2019, 118, 1493–1500. [Google Scholar] [CrossRef]
- Cortez, C.; Martins, R.M.; Alves, R.M.; Silva, R.C.; Bilches, L.C.; Macedo, S.; Atayde, V.D.; Kawashita, S.Y.; Briones, M.R.S.; Yoshida, N. Differential infectivity by the oral route of Trypanosoma cruzi lineages derived from Y strain. PLoS Negl. Trop. Dis. 2012, 6, e1804. [Google Scholar] [CrossRef]
- Margioto Teston, A.P.; de Abreu, A.P.; Abegg, C.P.; Gomes, M.L.; de Ornelas Toledo, M.J. Outcome of oral infection in mice inoculated with Trypanosoma cruzi IV of the Western Brazilian Amazon. Acta Trop. 2017, 166, 212–217. [Google Scholar] [CrossRef]
- Lewis, M.D.; Francisco, A.F.; Jayawardhana, S.; Langston, H.; Taylor, M.C.; Kelly, J.M. Imaging the development of chronic Chagas disease after oral transmission. Sci. Rep. 2018, 8, 11292. [Google Scholar] [CrossRef]
- Yoshida, N. Molecular basis of mammalian cell invasion by Trypanosoma cruzi. An. Acad. Bras. Cienc. 2006, 78, 87–111. [Google Scholar] [CrossRef]
- Staquicini, D.I.; Martins, R.M.; Macedo, S.; Sasso, G.R.S.; Atayde, V.D.; Juliano, M.A.; Yoshida, N. Role of GP82 in the selective binding to gastric mucin during oral infection with Trypanosoma cruzi. PLoS Negl. Trop. Dis. 2010, 4, e613. [Google Scholar] [CrossRef]
- Maeda, F.Y.; Alves, R.M.; Cortez, C.; Lima, F.M.; Yoshida, N. Characterization of the infective properties of a new genetic group of Trypanosoma cruzi associated with bats. Acta Trop. 2011, 120, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Hoft, D.F. Differential mucosal infectivity of different life stages of Trypanosoma cruzi. Am. J. Trop. Med. Hyg. 1996, 55, 360–364. [Google Scholar] [CrossRef] [PubMed]
- Roellig, D.M.; Ellis, A.E.; Yabsley, M.J. Oral transmission of Trypanosoma cruzi with opposing evidence for the theory of carnivory. J. Parasitol. 2009, 95, 360–364. [Google Scholar] [CrossRef]
- Torres, V.; Contreras, V.; Gutiérrez, B.; San Francisco, J.; Catalán, A.; Vega, J.L.; Moon, K.M.; Foster, L.J.; de Almeida, R.F.; Kalergis, A.M.; et al. Oral infectivity through carnivorism in murine model of Trypanosoma cruzi infection. Front. Cell Infect. Microbiol. 2024, 14, 1297099. [Google Scholar] [CrossRef]
- Guhl, F.; Ramírez, J.D. Trypanosoma cruzi I diversity: Towards the need of genetic subdivision? Acta Trop. 2011, 119, 1–4. [Google Scholar] [CrossRef]
- San Francisco, J.; Barría, I.; Gutiérrez, B.; Neira, I.; Muñoz, C.; Sagua, H.; Araya, J.E.; Andrade, J.C.; Zailberger, A.; Catalán, A.; et al. Decreased cruzipain and gp85/trans-sialidase family protein expression contributes to loss of Trypanosoma cruzi trypomastigote virulence. Microbes Infect. 2017, 19, 55–61. [Google Scholar] [CrossRef]
- San Francisco, J.; Astudillo, C.; Vega, J.L.; Catalán, A.; Gutiérrez, B.; Araya, J.E.; Zailberger, A.; Marina, A.; García, C.; Sanchez, N.; et al. Trypanosoma cruzi pathogenicity involves virulence factor expression and upregulation of bioenergetic and biosynthetic pathways. Virulence 2022, 13, 1827–1848. [Google Scholar] [CrossRef]
- Schaub, G.A. Trypanosoma cruzi: Quantitative studies of development of two strains in small intestine and rectum of the vector Triatoma infestans. Exp. Parasitol. 1989, 68, 260–273. [Google Scholar] [CrossRef]
- Herrera, H.M.; Abreu, U.G.P.; Keuroghlian, A.; Freitas, T.P.; Jansen, A.M. The role played by sympatric collared peccary (Tayassu tajacu), white-lipped peccary (Tayassu pecari), and feral pig (Sus scrofa) as maintenance hosts for Trypanosoma evansi and Trypanosoma cruzi in a sylvatic area of Brazil. Parasitol. Res. 2008, 103, 619–624. [Google Scholar] [CrossRef]
- Muñoz-San Martín, C.; Campo Verde Arbocco, F.; Saavedra, M.; Actis, E.A.; Ríos, T.A.; Abba, A.M.; Morales, M.E.; Cattan, P.E.; Jahn, G.A.; Superina, M. High rates of Trypanosoma cruzi infection in goats from Mendoza province, Argentina: Parasite loads in blood and seasonal variation. Acta Trop. 2020, 208, 105493. [Google Scholar] [CrossRef] [PubMed]
- Pizzi, T. Inmunología de la Enfermedad de Chagas; Editorial Universidad de Chile: Santiago, Chile, 1957. [Google Scholar]
- Yoshida, N. Surface antigens of metacyclic trypomastigotes of Trypanosoma cruzi. Infect. Immun. 1983, 40, 836–839. [Google Scholar] [CrossRef] [PubMed]
- Santos, F.M.; Barreto, W.T.G.; de Macedo, G.C.; Barros, J.H.d.S.; Xavier, S.C.d.C.; Garcia, C.M.; Mourão, G.; de Oliveira, J.; Rimoldi, A.R.; de Oliveira Porfírio, G.E.; et al. The reservoir system for Trypanosoma (Kinetoplastida, Trypanosomatidae) species in large neotropical wetland. Acta Trop. 2019, 199, 105098. [Google Scholar] [CrossRef] [PubMed]



| Outbreaks Places | Year of Occurrence | Cases (N0) | Deaths (No.%) | 95% CI | DTU | Vehicle | References |
|---|---|---|---|---|---|---|---|
| Bolivia | |||||||
| Guayaramerín, Beni | 2010 | 14 | 0 (0%) | [0.00–23.16] | NR | Majo | [56] |
| Brazil | |||||||
| Río Grande Do Norte | 1965 | 17 | 6 (35.2%) | [14.2–61.67] | NR | NR | [57] |
| Belém, Pará | 1968 | 3 | 1 (33.3%) | [0.84–90.57] | NR | NR | [58] |
| Macapá, Ap | 1984 | 8 | 0 (0%) | [0.00–36.94] | NR | NR | [59] |
| Paraíba | 1986 | 26 | 1 (3.8%) | [0.10–19.64] | NR | Cane sugar juice | [60] |
| Río do Branco, Acre | 1993 | 3 | 3 (100%) | [29.4–100.0] | NR | Açaí | [61] |
| Mazagao, Ap | 1996 | 17 | 0 (0%) | [0.00–19.51] | Tc I; TcIIa/TcIIb | Açaí | [62] |
| Abaetetuba, Pará | 1998 | 13 | 0 (0%) | [0.00–24.71] | NR | NR | [63] |
| Cametá, Pará | 1999 | 3 | 2 (66.6%) | [9.43–99.16] | NR | NR | [64] |
| Pará, Macapá, Amapá | 1990–2003 | 20 | 3 (20%) | [3.21–37.89] | NR | NR | [65] |
| Tefé, Amazonas | 2004 | 1 | 0 (0%) | [0.00–97.50] | NR | Açaí | [66] |
| Pará, Amapá, Maranhao | 1998–2005 | 183 | 13 (7.1%) | [3.84–11.84] | TcI | NR | [67] |
| Tefé, Amazonas | 2005 | 9 | 0 (0%) | [0.00–33.63] | NR | Açaí | [68] |
| Santa Catarina | 2005 | 24 | 3 (12.5%) | [2.66–32.36] | TcI; TcII | Cane sugar juice | [69] |
| Bahía | 2006 | 7 | 2 (28.5%) | [3.67–70.96] | NR | Water? | [49] |
| Pará | 2006 | 11 | 0 (0%) | [0.00–28.49] | NR | Açaí | [70] |
| Ceará | 2006 | 8 | 0 (0%) | [0.00–36.94] | NR | Soup | [71] |
| Macaúbas, Bahía | 2006 | 13 | 2 (15.3%) | [1.92–45.45] | NR | Cane sugar juice | [72] |
| Pará | 2007 | 25 | 0 (0%) | [0.00–13.72] | NR | Açaí | [73] |
| Santarem, Manaus, Coarí | 2006–2007 | 4 | 0 (0%) | [0.00–60.24] | NR | Açaí | [54] |
| Coarí, Amazonas | 2007 | 25 | 0 (0%) | [0.00–13.72] | TcIII | NR | [74] |
| Belém, Pará | 2007 | 4 | 0 (0%) | [0.00–60.24] | NR | Açaí | [75] |
| Belém, Pará | 2010 | 1 | 0 (0%) | [0.00–97.50] | NR | Açaí | [76] |
| Río Negro, Aazonas | 2010 | 17 | 0 (0%) | [0.00–19.51] | NR | Açaí | [77] |
| Carauarí, Amazonas (Unpublished) | 2011 | 12 | 0 (0%) | [0.00–26.46] | NR | Açaí | [78] |
| Pará | 2006–2012 | 668 | 20 (2.9%) | [1.84–4.59] | TcI(Zimodeme 1); TcIV (Zimodeme 3) | NR | [79] |
| Tocantis | 2008–2014 | 22 | 1 (4.5%) | [0.12–22.84] | NR | Açaí, bacaba | Tocantis Health Secretary |
| Pará | 2014 | 118 | 1 (0.84%) | [0.02–4.63] | NR | NR | Pará Health Secretary |
| Rio Grande Do Norte | 2015 | 18 | 3 (16.6%) | [3.58–41.42] | NR | NR | [80] |
| Pará | 2015 | 199 | 2 (1%) | [0.12–3.58] | NR | NR | Pará Health secretary |
| Pará | 2016 | 273 | 6 (2.1%) | [0.81–4.72] | NR | NR | Pará Health secretary |
| Pará | 2016 | 1 | 1 (100%) | [2.50–100.0] | NR | [81] | |
| Acre | 2009–2017 | 43 | 4 (9.3%) | [2.59–22.14] | NR | Açaí | Acre Health Secretary |
| Amazonas | 2015–2017 | 24 | 0 (0%) | [0.00–14.25] | NR | Açaí | Amazonas Health Secretary |
| Pará | 2017 | 211 | 1 (0.47%) | [0.01–2.61] | NR | NR | Pará Health Secretary |
| Marimarituba, Cachoeira do Aura, Pará | 2016–2017 | 15 | 1 (6.66%) | [0.17–31.95] * | TcI;TcIV | Bacaba, Pataua | [82] |
| Several locations | 2004–2022 | 147 | 0 (0%) | [0.00–2.48] * | TcIII/TcIV(zimodeme 3); TcIV | Açaí, Pataua | [83] |
| Ipixuna, Pará | 2021 | 6 | 0(0%) | [0.00–45.93] * | NR | Açaí | [84] |
| Colombia | |||||||
| Lebrija, Santander | 2008 | 10 | 2 (20%) | [2.52–55.61] | TcI | Orange juice | [85] |
| Santander | 2008–2009 | 20 | 3 (15%) | [3.21–37.89] | NR | NR | [86] |
| Antioquía | 2010 | 11 | 1 (9.09%) | [0.23–41.28] | NR | NR | [87] |
| Aguachica, Cesar | 2010 | 11 | 0 (0%) | [0.00–28.49] | TcI | NR | [88] |
| Casanare | 2014 | 40 | 2 (0.5%) | [0.61–16.92] | NR | NR | [89] |
| Casanare, sucre, Antioquia, Atlantico, cesar, chocó | 2019 | 124 | 10 (8.24%) | [3.27–12.86] * | TcI;TcII: TcIV | NR | [90] |
| César | 2021 | 9 | 2 (22.2%) | [0.00–49.38] * | NR | NR | [91] |
| Cubará, Boyacá | 2021 | 10 | 0 (0%) | [0.00–30.0] * | TcI; TcII | NR | [92] |
| Miraflores, Boyacá | 2021 | 1 | 0 (0%) | [0.00–97.50] * | NR | NR | [93] |
| French Guiana | |||||||
| Cayena | 2005 | 8 | 0 (0%) | [0.00–36.94] | TcI | Palm juice | [51] |
| Venezuela | |||||||
| Caracas | 2007 | 1 | 0 (0%) | [0.00–97.50] | TcI | NR | [94] |
| Caracas | 2007 | 103 | 1 (0.97%) | [0.02–5.29] | NR | Guava juice | [95] |
| Caracas (unpublished) | 2008 | 3 | 0 (0%) | [0.00–70.76] | NR | NR | [18] |
| Chichiriviche, Caracaya | 2009 | 88 | 4 (4.5%) | [1.25–11.23] | NR | NR | [96] |
| Chichiriviche, Caracaya | 2009 | 89 | 5 (5.6%) | [1.85–12.63] | TcI | Guava juice | [47] |
| Táchira | 2010 | 6 | 1 (16.6%) | [0.42–64.12] | NR | NR | [19] |
| Caracas | 2012 | 4 | 0 (0%) | [0.00–60.24] * | NR | NR | [18] |
| Mérida | 2013 | 5 | 1 (20%) | [0.51–71.64] | NR | NR | [97] |
| Mirimire, Falcón | 2013 | 8 | 1 (12.5%) | [0.32–52.65] | NR | Mango juice | [18] |
| El Guapo, Miranda (unpublished) | 2014 | 3 | 0 (0%) | [0.00–70.76] | NR | Pumarosa juice | [18] |
| Táchira (unpublished) | 2014 | 5 | 0 (0%) | [0.00–52.18] | NR | NR | [18] |
| Contaminated Food | Prevention |
|---|---|
| Açai Pulp | Heating 43 °C × 20 min/Pasteurization |
| Sugar Cane Juice | Pasteurization |
| Tropical Fruits Juice | Pasteurization |
| Palm Juice | Pasteurization |
| Wild Meat | Freezing/Complete cooking (internal temperature > 65 °C) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zambrano, S.; Montoya, K.; Avalos, A.; Gutiérrez, B.; San Francisco, J.; Vega, J.L.; González, J. Oral Route Infection by Trypanosoma cruzi: From the Beginning to the Present Day. Pathogens 2026, 15, 47. https://doi.org/10.3390/pathogens15010047
Zambrano S, Montoya K, Avalos A, Gutiérrez B, San Francisco J, Vega JL, González J. Oral Route Infection by Trypanosoma cruzi: From the Beginning to the Present Day. Pathogens. 2026; 15(1):47. https://doi.org/10.3390/pathogens15010047
Chicago/Turabian StyleZambrano, Sebastián, Kurt Montoya, Alejandro Avalos, Bessy Gutiérrez, Juan San Francisco, José Luis Vega, and Jorge González. 2026. "Oral Route Infection by Trypanosoma cruzi: From the Beginning to the Present Day" Pathogens 15, no. 1: 47. https://doi.org/10.3390/pathogens15010047
APA StyleZambrano, S., Montoya, K., Avalos, A., Gutiérrez, B., San Francisco, J., Vega, J. L., & González, J. (2026). Oral Route Infection by Trypanosoma cruzi: From the Beginning to the Present Day. Pathogens, 15(1), 47. https://doi.org/10.3390/pathogens15010047






