Anaplasma phagocytophilum Ecotype Analysis in Cattle from Great Britain
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection, Polymerase Chain Reaction (PCR) and Sequencing
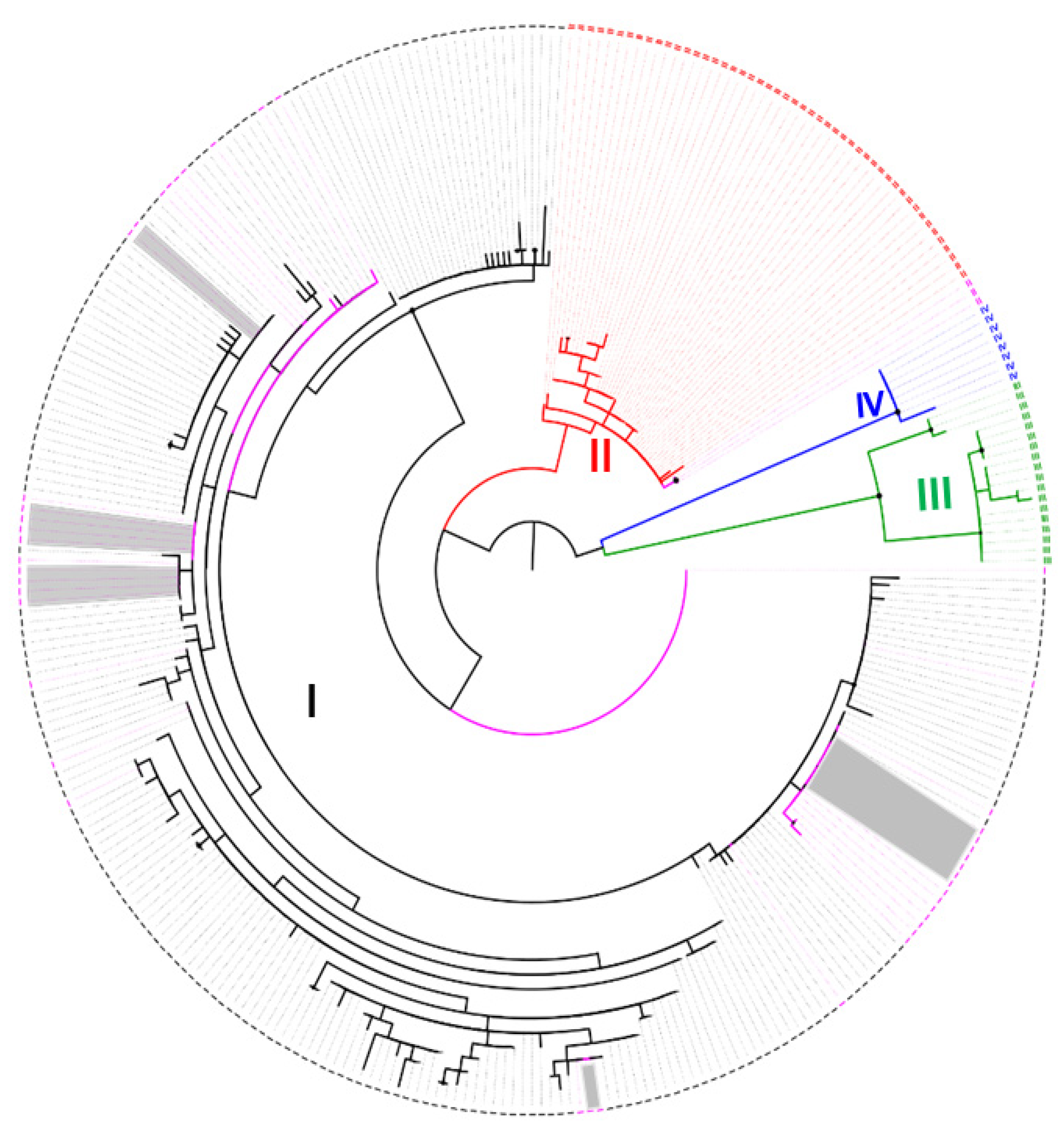
2.2. Sequence Editing, Assembly and Phylogenetic Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dumler, J.S.; Barbet, A.F.; Bekker, C.; Dasch, G.A.; Palmer, G.H.; Ray, S.C.; Rikihisa, Y.; Rurangirwa, F.R. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: Unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and ‘HGE agent’ as subjective synonyms of Ehrlichia phagocytophila. Int. J. Syst. Evol. Microbiol. 2001, 51, 2145–2165. [Google Scholar]
- Woldehiwet, Z. The natural history of Anaplasma phagocytophilum. Vet. Parasitol. 2010, 167, 108–122. [Google Scholar] [CrossRef]
- Beugnet, F.; Marié, J.-L. Emerging arthropod-borne diseases of companion animals in Europe. Vet. Parasitol. 2009, 163, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Blanco, J.R.; Oteo, J.A. Human granulocytic ehrlichiosis in Europe. Clin. Microbiol. Infect. 2002, 8, 763–772. [Google Scholar] [CrossRef] [PubMed]
- Tomanovic, S.; Chochlakis, D.; Radulovic, Z.; Milutinovic, M.; Cakic, S.; Mihaljica, D.; Tselentis, Y.; Psaroulaki, A. Analysis of pathogen co-occurrence in host-seeking adult hard ticks from Serbia. Exp. Appl. Acarol. 2013, 59, 367–376. [Google Scholar] [CrossRef]
- Bonnet, S.; de la Fuente, J.; Nicollet, P.; Liu, X.; Madani, N.; Blanchard, B.; Maingourd, C.; Alongi, A.; Torina, A.; Fernandez de Mera, I.G.; et al. Prevalence of tick-borne pathogens in adult Dermacentor spp. ticks from nine collection sites in France. Vector Borne Zoonotic Dis. 2013, 13, 226–236. [Google Scholar] [CrossRef]
- Palomar, A.; Portillo, A.; Santibáñez, P.; Mazuelas, D.; Roncero, L.; García-Álvarez, L.; Santibáñez, S.; Gutiérrez, Ó.; Oteo, J. Detection of tick-borne Anaplasma bovis, Anaplasma phagocytophilum and Anaplasma centrale in Spain. Med. Vet. Entomol. 2015, 29, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Keyte, S.; Abdullah, S.; James, K.; Newbury, H.; Helps, C.; Tasker, S.; Wall, R. Prevalence and distribution of Anaplasma phagocytophilum in ticks collected from dogs in the United Kingdom. Vet. Rec. 2021, 188, e12. [Google Scholar] [CrossRef] [PubMed]
- Rar, V.A.; Epikhina, T.I.; Yakimenko, V.V.; Malkova, M.G.; Tancev, A.K.; Bondarenko, E.I.; Ivanov, M.K.; Tikunova, N.V. Genetic variability of Anaplasma phagocytophilum in ticks and voles from Ixodes persulcatus/Ixodes trianguliceps sympatric areas from Western Siberia, Russia. Ticks Tick Borne Dis. 2014, 5, 854–863. [Google Scholar] [CrossRef]
- Kohn, B.; Galke, D.; Beelitz, P.; Pfister, K. Clinical features of canine granulocytic anaplasmosis in 18 naturally infected dogs. J. Vet. Intern. Med. 2008, 22, 1289–1295. [Google Scholar] [CrossRef]
- Bakken, J.S.; Dumler, J.S. Human Granulocytic Ehrlichiosis. Clin. Infect. Dis. 2000, 31, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Granquist, E.G.; Aleksandersen, M.; Bergström, K.; Dumler, S.J.; Torsteinbø, W.O.; Stuen, S. A morphological and molecular study of Anaplasma phagocytophilum transmission events at the time of Ixodes ricinus tick bite. Acta Vet. Scand. 2010, 52, 43. [Google Scholar] [CrossRef] [PubMed]
- Rar, V.A.; Livanova, N.N.; Panov, V.V.; Doroschenko, E.K.; Pukhovskaya, N.M.; Vysochina, N.P.; Ivanov, L.I. Genetic diversity of Anaplasma and Ehrlichia in the Asian part of Russia. Ticks Tick Borne Dis. 2010, 1, 57–65. [Google Scholar] [CrossRef]
- Horowitz, H.W.; Kilchevsky, E.; Haber, S.; Aguero-Rosenfeld, M.; Kranwinkel, R.; James, E.K.; Wong, S.J.; Chu, F.; Liveris, D.; Schwartz, I. Perinatal transmission of the agent of human granulocytic ehrlichiosis. N. Engl. J. Med. 1998, 339, 375–378. [Google Scholar] [CrossRef] [PubMed]
- Pusterla, N.; Braun, U.; Wolfensberger, C.; Lutz, H. Intrauterine infection with Ehrlichia phagocytophila in a cow. Vet. Rec. 1997, 141, 101–102. [Google Scholar] [CrossRef] [PubMed]
- Johnson, N.; Paul Phipps, L.; McFadzean, H.; Barlow, A.M. An outbreak of bovine babesiosis in February, 2019, triggered by above average winter temperatures in southern England and co-infection with Babesia divergens and Anaplasma phagocytophilum. Parasites Vectors 2020, 13, 305. [Google Scholar] [CrossRef]
- Gandy, S.; Hansford, K.; McGinley, L.; Cull, B.; Smith, R.; Semper, A.; Brooks, T.; Fonville, M.; Sprong, H.; Phipps, P.; et al. Prevalence of Anaplasma phagocytophilum in questing Ixodes ricinus nymphs across twenty recreational areas in England and Wales. Ticks Tick Borne Dis. 2022, 13, 101965. [Google Scholar] [CrossRef]
- Bianchessi, L.; Rocchi, M.S.; Maley, M.; Allen, K.; Ballingall, K.; Turin, L. Presence of Anaplasma phagocytophilum Ecotype I in UK Ruminants and Associated Zoonotic Risk. Pathogens 2023, 12, 216. [Google Scholar] [CrossRef]
- Flattery, A.; McKiernan, F.; Browne, J.; Gray, J.; Zaid, T.; O’Connor, J.; Zintl, A. The prevalence and distribution of Anaplasma phagocytophilum genotypes in Ixodes ricinus nymphs collected from farm- and woodland sites in Ireland. Ticks Tick Borne Dis. 2022, 13, 101928. [Google Scholar] [CrossRef]
- Hoepler, W.; Markowicz, M.; Schoetta, A.-M.; Zoufaly, A.; Stanek, G.; Wenisch, C. Molecular diagnosis of autochthonous human anaplasmosis in Austria—An infectious diseases case report. BMC Infect. Dis. 2020, 20, 288. [Google Scholar] [CrossRef]
- Bakken, J.S.; Dumler, J.S.; Chen, S.-M.; Eckman, M.R.; Van Etta, L.L.; Walker, D.H. Human granulocytic ehrlichiosis in the upper Midwest United States: A new species emerging? JAMA 1994, 272, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Petrovec, M.; Lotric Furlan, S.; Zupanc, T.A.; Strle, F.; Brouqui, P.; Roux, V.; Dumler, J.S. Human disease in Europe caused by a granulocytic Ehrlichia species. J. Clin. Microbiol. 1997, 35, 1556–1559. [Google Scholar] [CrossRef]
- Weber, R.; Pusterla, N.; Loy, M.; Lutz, H. Fever, leukopenia, and thrombocytopenia in a patient with acute Lyme borreliosis were due to human granulocytic ehrlichiosis. Clin. Infect. Dis. 1998, 26, 253–254. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Lotrič-Furlan, S.; Petrovec, M.; Zupanc, T.A.; Nicholson, W.L.; Sumner, J.W.; Childs, J.E.; Strle, F. Human granulocytic ehrlichiosis in Europe: Clinical and laboratory findings for four patients from Slovenia. Clin. Infect. Dis. 1998, 27, 424–428. [Google Scholar] [CrossRef]
- Laferl, H.; Hogrefe, W.; Köck, T.; Pichler, H. A further case of acute human granulocytic ehrlichiosis in Slovenia. Eur. J. Clin. Microbiol. Infect. Dis. 1999, 18, 385–386. [Google Scholar] [CrossRef] [PubMed]
- Oteo, J.A.; Blanco, J.R.; Martínez de Artola, V.; Ibarra, V. First report of human granulocytic ehrlichiosis from southern Europe (Spain). Emerg. Infect. Dis. 2000, 6, 430–432. [Google Scholar] [CrossRef]
- Remy, V.; Hansmann, Y.; De Martino, S.; Christmann, D.; Brouqui, P. Human Anaplasmosis Presenting as Atypical Pneumonitis in France. Clin. Infect. Dis. 2003, 37, 846–848. [Google Scholar] [CrossRef]
- Ruscio, M.; Cinco, M. Human granulocytic ehrlichiosis in Italy. Ann. N. Y. Acad. Sci. 2003, 990, 350–352. [Google Scholar] [CrossRef]
- Aktas, M.; Ozubek, S. Bovine anaplasmosis in Turkey: First laboratory confirmed clinical cases caused by Anaplasma phagocytophilum. Vet. Microbiol. 2015, 178, 246–251. [Google Scholar] [CrossRef]
- Matsumoto, K.; Joncour, G.; Davoust, B.; PITEL, P.H.; Chauzy, A.; Collin, E.; Morvan, H.; Vassallo, N.; Brouqui, P. Anaplasma phagocytophilum infection in cattle in France. Ann. N. Y. Acad. Sci. 2006, 1078, 491–494. [Google Scholar] [CrossRef]
- Andersson, M.O.; Vichova, B.; Tolf, C.; Krzyzanowska, S.; Waldenstrom, J.; Karlsson, M.E. Co-infection with Babesia divergens and Anaplasma phagocytophilum in cattle (Bos taurus), Sweden. Ticks Tick Borne Dis. 2017, 8, 933–935. [Google Scholar] [CrossRef] [PubMed]
- McFadzean, H.; Johnson, N.; Phipps, L.P.; Hobbs, R.L. High morbidity associated with an outbreak of tick-borne disease in a dairy herd, Cornwall. Vet. Rec. Case Rep. 2021, 9, e171. [Google Scholar] [CrossRef]
- Woldehiwet, Z. Anaplasma phagocytophilum in ruminants in Europe. Ann. N. Y. Acad. Sci. 2006, 1078, 446–460. [Google Scholar] [CrossRef]
- Van Loo, H.; Pascottini, O.B.; Hooyberghs, J.; De Bleecker, K.; Ribbens, S.; Opsomer, G.; Pardon, B. Detection of Anaplasma phagocytophilum in fetal and placental tissue of bovine abortions and perinatal mortalities. Vet. Rec. 2023, e2880. [Google Scholar] [CrossRef] [PubMed]
- LEE, C.; Lin, Y.; Tsang, C.; Chung, Y. A loop-mediated isothermal amplification (LAMP) assay for rapid detection of Anaplasma phagocytophilum infection in dogs. Turk. J. Vet. Anim. Sci. 2012, 36, 205–210. [Google Scholar] [CrossRef]
- Kawahara, M.; Rikihisa, Y.; Lin, Q.; Isogai, E.; Tahara, K.; Itagaki, A.; Hiramitsu, Y.; Tajima, T. Novel genetic variants of Anaplasma phagocytophilum, Anaplasma bovis, Anaplasma centrale, and a novel Ehrlichia sp. in wild deer and ticks on two major islands in Japan. Appl. Environ. Microbiol. 2006, 72, 1102–1109. [Google Scholar] [CrossRef] [PubMed]
- Scharf, W.; Schauer, S.; Freyburger, F.; Petrovec, M.; Schaarschmidt-Kiener, D.; Liebisch, G.; Runge, M.; Ganter, M.; Kehl, A.; Dumler, J.S.; et al. Distinct Host Species Correlate with Anaplasma phagocytophilum ankA Gene Clusters. J. Clin. Microbiol. 2011, 49, 790–796. [Google Scholar] [CrossRef]
- Chastagner, A.; Dugat, T.; Vourc’h, G.; Verheyden, H.; Legrand, L.; Bachy, V.; Chabanne, L.; Joncour, G.; Maillard, R.; Boulouis, H.J.; et al. Multilocus sequence analysis of Anaplasma phagocytophilum reveals three distinct lineages with different host ranges in clinically ill French cattle. Vet. Res. 2014, 45, 114. [Google Scholar] [CrossRef]
- Alberti, A.; Addis, M.F.; Sparagano, O.; Zobba, R.; Chessa, B.; Cubeddu, T.; Parpaglia, M.L.P.; Ardu, M.; Pittau, M. Anaplasma phagocytophilum, Sardinia, Italy. Emerg. Infect. Dis. 2005, 11, 1322. [Google Scholar] [CrossRef]
- Chisu, V.; Zobba, R.; Lecis, R.; Sotgiu, F.; Masala, G.; Foxi, C.; Pisu, D.; Alberti, A. GroEL typing and phylogeny of Anaplasma species in ticks from domestic and wild vertebrates. Ticks Tick Borne Dis. 2018, 9, 31–36. [Google Scholar] [CrossRef]
- Courtney, J.W.; Kostelnik, L.M.; Zeidner, N.S.; Massung, R.F. Multiplex real-time PCR for detection of Anaplasma phagocytophilum and Borrelia burgdorferi. J. Clin. Microbiol. 2004, 42, 3164–3168. [Google Scholar] [CrossRef] [PubMed]
- de Marco, M.; Hernandez-Triana, L.M.; Phipps, L.P.; Hansford, K.; Mitchell, E.S.; Cull, B.; Swainsbury, C.S.; Fooks, A.R.; Medlock, J.M.; Johnson, N. Emergence of Babesia canis in southern England. Parasites Vectors 2017, 10, 241. [Google Scholar] [CrossRef] [PubMed]
- Jahfari, S.; Coipan, E.C.; Fonville, M.; Van Leeuwen, A.D.; Hengeveld, P.; Heylen, D.; Heyman, P.; Van Maanen, C.; Butler, C.M.; Földvári, G. Circulation of four Anaplasma phagocytophilum ecotypes in Europe. Parasites Vectors 2014, 7, 365. [Google Scholar] [CrossRef] [PubMed]
- Grassi, L.; Franzo, G.; Martini, M.; Mondin, A.; Cassini, R.; Drigo, M.; Pasotto, D.; Vidorin, E.; Menandro, M.L. Ecotyping of Anaplasma phagocytophilum from Wild Ungulates and Ticks Shows Circulation of Zoonotic Strains in Northeastern Italy. Animals 2021, 11, 310. [Google Scholar] [CrossRef]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New Models and Efficient Methods for Phylogenetic Inference in the Genomic Era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef]
- Hoang, D.T.; Chernomor, O.; Von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the ultrafast bootstrap approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Hasegawa, M.; Kishino, H.; Yano, T.-A. Dating of the human-ape splitting by a molecular clock of mitochondrial DNA. J. Mol. Evol. 1985, 22, 160–174. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.K.F.; von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef]
- Noaman, V. Epidemiological study on Anaplasma phagocytophilum in cattle: Molecular prevalence and risk factors assessment in different ecological zones in Iran. Prev. Vet. Med. 2020, 183, 105118. [Google Scholar] [CrossRef]
- Jahfari, S.; Ruyts, S.C.; Frazer-Mendelewska, E.; Jaarsma, R.; Verheyen, K.; Sprong, H. Melting pot of tick-borne zoonoses: The European hedgehog contributes to the maintenance of various tick-borne diseases in natural cycles urban and suburban areas. Parasites Vectors 2017, 10, 134. [Google Scholar] [CrossRef]
- Hamsikova, Z.; Silaghi, C.; Takumi, K.; Rudolf, I.; Gunar, K.; Sprong, H.; Kazimirova, M. Presence of Roe Deer Affects the Occurrence of Anaplasma phagocytophilum Ecotypes in Questing Ixodes ricinus in Different Habitat Types of Central Europe. Int. J. Environ. Res. Public Health 2019, 16, 4725. [Google Scholar] [CrossRef] [PubMed]
- Razanske, I.; Rosef, O.; Radzijevskaja, J.; Bratchikov, M.; Griciuviene, L.; Paulauskas, A. Prevalence and co-infection with tick-borne Anaplasma phagocytophilum and Babesia spp. in red deer (Cervus elaphus) and roe deer (Capreolus capreolus) in Southern Norway. Int. J. Parasitol. Parasites Wildl. 2019, 8, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Stuen, S.; Pettersen, K.S.; Granquist, E.G.; Bergstrom, K.; Bown, K.J.; Birtles, R.J. Anaplasma phagocytophilum variants in sympatric red deer (Cervus elaphus) and sheep in southern Norway. Ticks Tick Borne Dis. 2013, 4, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Dugat, T.; Zanella, G.; Veran, L.; Lesage, C.; Girault, G.; Durand, B.; Lagree, A.C.; Boulouis, H.J.; Haddad, N. Multiple-locus variable-number tandem repeat analysis potentially reveals the existence of two groups of Anaplasma phagocytophilum circulating in cattle in France with different wild reservoirs. Parasites Vectors 2016, 9, 596. [Google Scholar] [CrossRef]
- Huhn, C.; Winter, C.; Wolfsperger, T.; Wuppenhorst, N.; Strasek Smrdel, K.; Skuballa, J.; Pfaffle, M.; Petney, T.; Silaghi, C.; Dyachenko, V.; et al. Analysis of the population structure of Anaplasma phagocytophilum using multilocus sequence typing. PLoS ONE 2014, 9, e93725. [Google Scholar] [CrossRef]
- Rar, V.; Tkachev, S.; Tikunova, N. Genetic diversity of Anaplasma bacteria: Twenty years later. Infect. Genet. Evol. 2021, 91, 104833. [Google Scholar] [CrossRef]
| Location | Ecotype | Sequence Variants (n) | Host | Tick Species | Negative (n) | Positive (n) | Total |
|---|---|---|---|---|---|---|---|
| Devon | 1 | 8 | B. taurus | − | 8 | 8 | |
| 1 | 13 | vegetation | I. ricinus * | 2 | 13 | 15 | |
| Cornwall | 1 | 2 | I. ricinus * | − | 2 | 2 | |
| 1 | 5 | B. taurus | − | 5 | 5 | ||
| Norfolk | 1 | 2 | C. elaphus (red deer) | − | 2 | 2 | |
| Cumbria | 1 | 1 | I. ricinus * | − | 1 | 1 | |
| 2 | 1 | C. capreolus (roe deer) | − | 1 | 1 | ||
| Pembrokeshire | 1 | 2 | I. ricinus * | − | 2 | 2 | |
| Dorset | 1 | 1 | O. aries | − | 1 | 1 | |
| 1 | 1 | B. taurus | − | 1 | 1 | ||
| Somerset | 1 | 2 | B. taurus | − | 2 | 2 | |
| 1 | 1 | C. capreolus (roe deer) | − | 1 | 1 | ||
| Powys | 1 | 1 | B. taurus | − | 1 | 1 | |
| Scotland | 1 | 1 | O. aries | − | 1 | 1 | |
| Unknown | 2 | 2 | I. ricinus ** | − | 2 | 2 | |
| 1 | 5 | B. taurus | − | 4 | 4 | ||
| 1 | 1 | O. aries | − | 1 | 2 | ||
| Total | 48 | 2 | 48 | 50 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Apaa, T.T.; McFadzean, H.; Gandy, S.; Hansford, K.; Medlock, J.; Johnson, N. Anaplasma phagocytophilum Ecotype Analysis in Cattle from Great Britain. Pathogens 2023, 12, 1029. https://doi.org/10.3390/pathogens12081029
Apaa TT, McFadzean H, Gandy S, Hansford K, Medlock J, Johnson N. Anaplasma phagocytophilum Ecotype Analysis in Cattle from Great Britain. Pathogens. 2023; 12(8):1029. https://doi.org/10.3390/pathogens12081029
Chicago/Turabian StyleApaa, Ternenge Thaddaeus, Harriet McFadzean, Sara Gandy, Kayleigh Hansford, Jolyon Medlock, and Nicholas Johnson. 2023. "Anaplasma phagocytophilum Ecotype Analysis in Cattle from Great Britain" Pathogens 12, no. 8: 1029. https://doi.org/10.3390/pathogens12081029
APA StyleApaa, T. T., McFadzean, H., Gandy, S., Hansford, K., Medlock, J., & Johnson, N. (2023). Anaplasma phagocytophilum Ecotype Analysis in Cattle from Great Britain. Pathogens, 12(8), 1029. https://doi.org/10.3390/pathogens12081029







