Respiratory Syncytial Virus Vaccines: A Review of the Candidates and the Approved Vaccines
Abstract
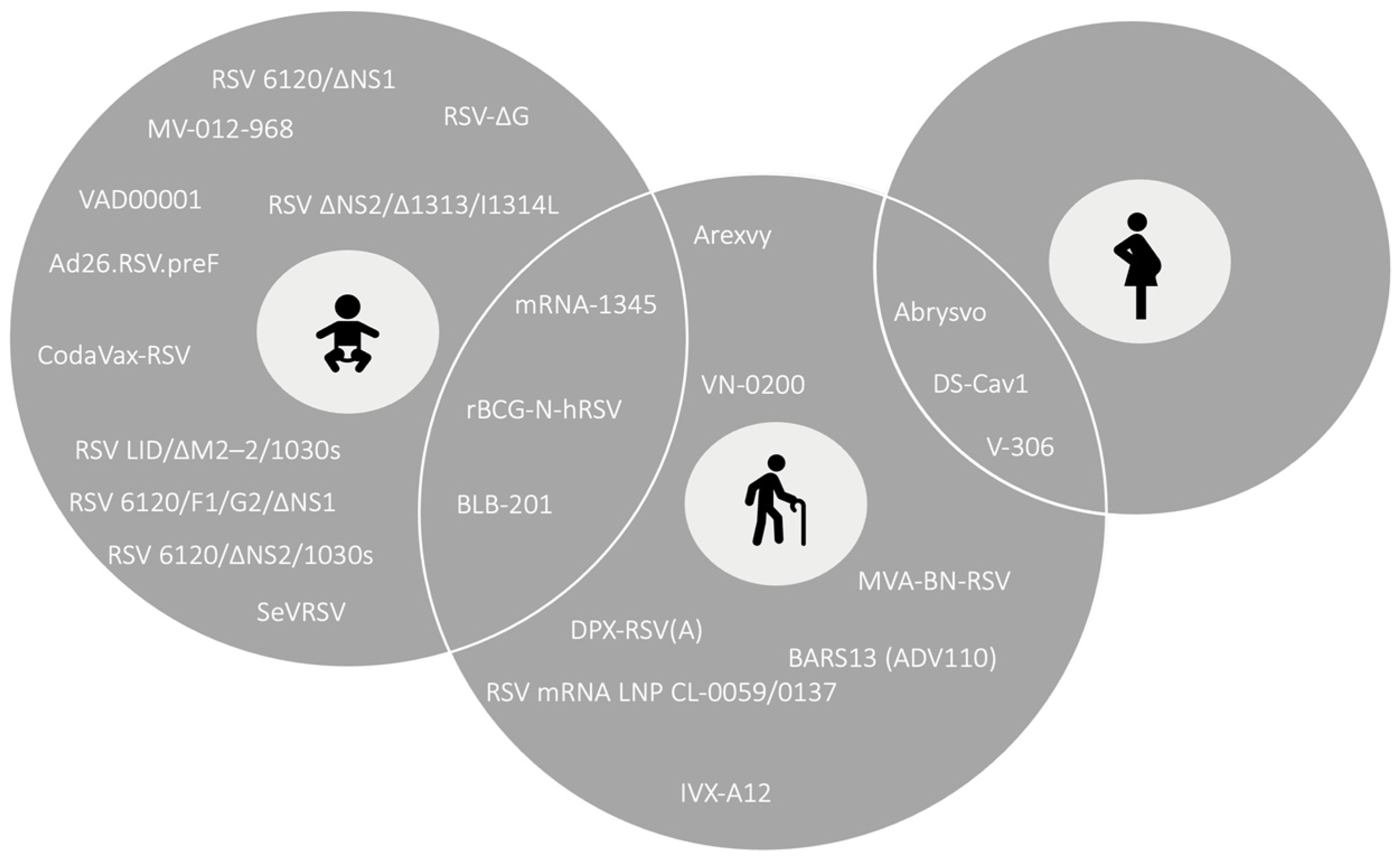
:1. Introduction
2. Materials and Methods
3. Results
3.1. mRNA Vaccines
3.1.1. mRNA-1345
3.1.2. RSV mRNA LNP CL-0059 or RSV mRNA LNP CL-0137
3.2. Live-Attenuated/Chimeric Vaccines (LAVs)
3.2.1. BLB-201
3.2.2. CodaVax-RSV
3.2.3. RSV-ΔG
3.2.4. rBCG-N-hRSV
3.2.5. SeVRSV
3.2.6. MV-012-968
3.2.7. VAD00001
3.2.8. RSV ΔNS2/Δ1313/I1314L
3.2.9. RSV LID/ΔM2–2/1030s
3.2.10. RSV 6120/ΔNS2/1030s
3.2.11. RSV 6120/F1/G2/ΔNS1/RSV 6120/ΔNS1
3.3. Subunit/Virus-Like-Particle (VLP)-Based Vaccines
3.3.1. IVX-A12
3.3.2. V-306
3.3.3. DPX-RSV(A)
3.3.4. VN-0200
3.3.5. BARS13 (ADV110)
3.3.6. DS-Cav1 (VRC-RSVRGP084-00-VP)
3.3.7. Arexvy/RSVPreF3 OA (GSK3844766A)
3.3.8. Abrysvo/RSVpreF
3.4. Recombinant Viral-Vector-Based Vaccines
3.4.1. MVA-BN-RSV
3.4.2. Ad26.RSV.preF
4. Discussion
5. Expert Opinion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Shan, J.; Britton, P.N.; King, C.L.; Booy, R. The Immunogenicity and Safety of Respiratory Syncytial Virus Vaccines in Development: A Systematic Review. Influenza Other Respi. Viruses 2021, 15, 539–551. [Google Scholar] [CrossRef] [PubMed]
- Simoes, E.A.F. Respiratory Syncytial Virus Infection. Lancet 1999, 354, 847–852. [Google Scholar] [CrossRef] [PubMed]
- Battles, M.B.; McLellan, J.S. Respiratory Syncytial Virus Entry and How to Block It. Nat. Rev. Microbiol. 2019, 17, 233–245. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Williams, T.C.; Viboud, C.; Campbell, H.; Chen, J.; Spiro, D.J. RSV Genomic Diversity and the Development of a Globally Effective RSV Intervention. Vaccine 2021, 39, 2811–2820. [Google Scholar] [CrossRef] [PubMed]
- Jain, H.; Schweitzer, J.W.; Justice, N.A. Respiratory Syncytial Virus Infection. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2023. Available online: https://www.ncbi.nlm.nih.gov/books/NBK459215/ (accessed on 20 June 2023).
- Walsh, E.E.; Hall, C.B. Respiratory Syncytial Virus (RSV). In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases; Elsevier: Amsterdam, The Netherlands, 2015; pp. 1948–1960.e3. ISBN 9780128145821. [Google Scholar]
- Villanueva, D.D.H.; Arcega, V.; Rao, M. Review of Respiratory Syncytial Virus Infection among Older Adults and Transplant Recipients. Ther. Adv. Infect. Dis. 2022, 9, 1–13. [Google Scholar] [CrossRef]
- Domachowske, J.B.; Anderson, E.J.; Goldstein, M. The Future of Respiratory Syncytial Virus Disease Prevention and Treatment. Infect. Dis. Ther. 2021, 10, 47–60. [Google Scholar] [CrossRef]
- Mitchell, I. Treatment of RSV Bronchiolitis: Drugs, Antibiotics. Paediatr. Respir. Rev. 2009, 10, 14–15. [Google Scholar] [CrossRef]
- Jenkins, V.A.; Hoet, B.; Hochrein, H.; De Moerlooze, L. The Quest for a Respiratory Syncytial Virus Vaccine for Older Adults: Thinking beyond the F Protein. Vaccines 2023, 11, 382. [Google Scholar] [CrossRef]
- Garegnani, L.; Styrmisdóttir, L.; Roson Rodriguez, P.; Escobar Liquitay, C.M.; Esteban, I.; Franco, J.V.A. Palivizumab for Preventing Severe Respiratory Syncytial Virus (RSV) Infection in Children. Cochrane Database Syst. Rev. 2021, 11, CD013757. [Google Scholar] [CrossRef]
- Griffin, M.P.; Yuan, Y.; Takas, T.; Domachowske, J.B.; Madhi, S.A.; Manzoni, P.; Simões, E.A.F.; Esser, M.T.; Khan, A.A.; Dubovsky, F.; et al. Single-Dose Nirsevimab for Prevention of RSV in Preterm Infants. N. Engl. J. Med. 2020, 383, 415–425. [Google Scholar] [CrossRef]
- Esposito, S.; Abu-Raya, B.; Bonanni, P.; Cahn-Sellem, F.; Flanagan, K.L.; Martinon Torres, F.; Mejias, A.; Nadel, S.; Safadi, M.A.P.; Simon, A. Coadministration of Anti-Viral Monoclonal Antibodies with Routine Pediatric Vaccines and Implications for Nirsevimab Use: A White Paper. Front. Immunol. 2021, 12, 708938. [Google Scholar] [CrossRef] [PubMed]
- Beyfortus Approved in the EU for the Prevention of RSV Lower Respiratory Tract Disease in Infants. Available online: https://www.astrazeneca.com/media-centre/press-releases/2022/beyfortus-approved-in-the-eu-for-the-prevention-of-rsv-lower-respiratory-tract-disease-in-infants.html (accessed on 24 August 2023).
- Blanco, J.C.G.; Boukhvalova, M.S.; Morrison, T.G.; Vogel, S.N. A Multifaceted Approach to RSV Vaccination. Hum. Vaccines Immunother. 2018, 14, 1734–1745. [Google Scholar] [CrossRef] [PubMed]
- Graham, B.S.; Modjarrad, K.; McLellan, J.S. Novel Antigens for RSV Vaccines. Curr. Opin. Immunol. 2015, 35, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Beugeling, M.; De Zee, J.; Woerdenbag, H.J.; Frijlink, H.W.; Wilschut, J.C.; Hinrichs, W.L.J. Respiratory Syncytial Virus Subunit Vaccines Based on the Viral Envelope Glycoproteins Intended for Pregnant Women and the Elderly. Expert Rev. Vaccines 2019, 18, 935–950. [Google Scholar] [CrossRef] [PubMed]
- Mejias, A.; Rodríguez-Fernández, R.; Oliva, S.; Peeples, M.E.; Ramilo, O. The Journey to a Respiratory Syncytial Virus Vaccine. Ann. Allergy Asthma Immunol. 2020, 125, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Killikelly, A.M.; Kanekiyo, M.; Graham, B.S. Pre-Fusion F Is Absent on the Surface of Formalin-Inactivated Respiratory Syncytial Virus. Sci. Rep. 2016, 6, 34108. [Google Scholar] [CrossRef]
- Qiu, X.; Xu, S.; Lu, Y.; Luo, Z.; Yan, Y.; Wang, C.; Ji, J. Development of MRNA Vaccines against Respiratory Syncytial Virus (RSV). Cytokine Growth Factor Rev. 2022, 68, 37–53. [Google Scholar] [CrossRef]
- Biagi, C.; Dondi, A.; Scarpini, S.; Rocca, A.; Vandini, S.; Poletti, G.; Lanari, M. Current State and Challenges in Developing Respiratory Syncytial Virus Vaccines. Vaccines 2020, 8, 672. [Google Scholar] [CrossRef]
- Ruckwardt, T.J.; Morabito, K.M.; Graham, B.S. Immunological Lessons from Respiratory Syncytial Virus Vaccine Development. Immunity 2019, 51, 429–442. [Google Scholar] [CrossRef]
- Clark, C.M.; Guerrero-Plata, A. Respiratory Syncytial Virus Vaccine Approaches: A Current Overview. Curr. Clin. Microbiol. Rep. 2017, 4, 202–207. [Google Scholar] [CrossRef]
- Killikelly, A.; Tunis, M.; House, A.; Quach, C.; Vaudry, W.; Moore, D. Overview of the Respiratory Syncytial Virus Vaccine Candidate Pipeline in Canada. Can. Commun. Dis. Rep. 2020, 46, 56–61. [Google Scholar] [CrossRef]
- RSV Vaccine and Mab Snapshot. Available online: https://www.path.org/resources/rsv-vaccine-and-mab-snapshot/ (accessed on 24 August 2023).
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef] [PubMed]
- Moderna Announces Clinical Progress from Its Industry-Leading mRNA Vaccine Franchise and Continues Investments to Accelerate Pipeline Development. Available online: https://investors.modernatx.com/news/news-details/2021/Moderna-Announces-Clinical-Progress-from-its-Industry-Leading-mRNA-Vaccine-Franchise-and-Continues-Investments-to-Accelerate-Pipeline-Development/default.aspx (accessed on 24 August 2023).
- Moderna Announces Significant Advances across Industry-Leading mRNA Portfolio at 2021 R&D Day. Available online: https://investors.modernatx.com/news/news-details/2021/Moderna-Announces-Significant-Advances-Across-Industry-Leading-mRNA-Portfolio-at-2021-RD-Day/default.aspx (accessed on 24 August 2023).
- Moderna Announces Mrna-1345, an Investigational Respiratory Syncytial Virus (RSV) Vaccine, Has Met Primary Efficacy Endpoints in Phase 3 Trial in Older Adults. Available online: https://investors.modernatx.com/news/news-details/2023/Moderna-Announces-mRNA-1345-an-Investigational-Respiratory-Syncytial-Virus-RSV-Vaccine-Has-Met-Primary-Efficacy-Endpoints-in-Phase-3-Trial-in-Older-Adults/default.aspx (accessed on 24 August 2023).
- Moderna Granted FDA Breakthrough Therapy Designation for Mrna-1345, an Investigational Respiratory Syncytial Virus (RSV) Vaccine Candidate. Available online: https://investors.modernatx.com/news/news-details/2023/Moderna-Granted-FDA-Breakthrough-Therapy-Designation-for-mRNA-1345-An-Investigational-Respiratory-Syncytial-Virus-RSV-Vaccine-Candidate/default.aspx (accessed on 24 August 2023).
- Icosavax Announces Positive Topline Interim Phase 1 Results for Bivalent VLP Vaccine Candidate IVX-A12 against RSV and HMPV in Older Adults. Available online: https://investors.icosavax.com/news-releases/news-release-details/icosavax-announces-positive-topline-interim-phase-1-results (accessed on 24 August 2023).
- Leroux-roels, I.; Bruhwyler, J.; Stergiou, L.; Sumeray, M.; Joye, J.; Maes, C.; Lambert, P.; Lerouxroels, G. Evaluating the Safety and Immunogenicity of an Epitope-Specific Chemically Defined Nanoparticle RSV Vaccine. Vaccines 2023, 11, 367. [Google Scholar] [CrossRef] [PubMed]
- Langley, J.M.; Macdonald, L.D.; Weir, G.M.; Mackinnon-Cameron, D.; Ye, L.; Mcneil, S.; Schepens, B.; Saelens, X.; Stanford, M.M.; Halperin, S.A. A Respiratory Syncytial Virus Vaccine Based on the Small Hydrophobic Protein Ectodomain Presented with a Novel Lipid-Based Formulation Is Highly Immunogenic and Safe in Adults: A First-in-Humans Study. J. Infect. Dis. 2018, 218, 378–387. [Google Scholar] [CrossRef]
- Cheng, X.; Zhao, G.; Dong, A.; He, Z.; Wang, J.; Jiang, B.; Wang, B.; Wang, M.; Huai, X.; Zhang, S.; et al. A First in Human Trial to Evaluate the Safety and Immunogenicity of a G Protein Based Recombinant Respiratory Syncytial Virus Vaccine in Healthy Adults 18–45 Years. Vaccines 2022, 11, 999. [Google Scholar] [CrossRef]
- Advaccine Announces First Participants Dosed in Phase 2 Study of ADV110 Evaluating Respiratory Syncytial Virus (RSV) Vaccine Candidate in Australia. Available online: https://www.biospace.com/article/releases/advaccine-announces-first-participants-dosed-in-phase-2-study-of-adv110-evaluating-respiratory-syncytial-virus-rsv-vaccine-candidate-in-australia/ (accessed on 24 August 2023).
- Ruckwardt, T.J.; Morabito, K.M.; Phung, E.; Crank, M.C.; Costner, P.J.; Holman, L.S.A.; Chang, L.A.; Hickman, S.P.; Berkowitz, N.M.; Gordon, I.J.; et al. Safety, Tolerability, and Immunogenicity of the Respiratory Syncytial Virus Prefusion F Subunit Vaccine DS-Cav1: A Phase 1, Randomised, Open-Label, Dose-Escalation Clinical Trial. Lancet Respir. Med. 2021, 9, 1111–1120. [Google Scholar] [CrossRef] [PubMed]
- Leroux-Roels, I.; Davis, M.G.; Steenackers, K.; Essink, B.; Vandermeulen, C.; Fogarty, C.; Andrews, C.P.; Kerwin, E.; David, M.P.; Fissette, L.; et al. Safety and Immunogenicity of a Respiratory Syncytial Virus Prefusion F (RSVPreF3) Candidate Vaccine in Older Adults: Phase 1/2 Randomized Clinical Trial. J. Infect. Dis. 2022, 227, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Kotb, S.; Haranaka, M.; Folschweiller, N.; Nakanwagi, P.; Verheust, C.; De Schrevel, N.; David, M.P.; Mesaros, N.; Hulstrøm, V. Safety and Immunogenicity of a Respiratory Syncytial Virus Prefusion F Protein (RSVPreF3) Candidate Vaccine in Older Japanese Adults: A Phase I, Randomized, Observer-Blind Clinical Trial. Respir. Investig. 2023, 61, 261–269. [Google Scholar] [CrossRef]
- Schwarz, T.F.; Hwang, S.-J.; Ylisastigui, P.P.; Liu, C.-S.; Takazawa, K.; Yono, M.; Ervin, J.E.; Andrews, C.; Fogarty, C.; Eckermann, T.; et al. Safety and Reactogenicity of an Investigational Respiratory Syncytial Virus (RSV) Prefusion F Protein Vaccine for Adults ≥ 60 Years of Age (RSVPreF3 OA): An Interim Analysis at 6 Months after Vaccination. Open Forum Infect. Dis. 2022, 9, ofac492.197. [Google Scholar] [CrossRef]
- Schwarz, T.F.; Hwang, S.-J.; Ylisastigui, P.P.; Liu, C.-S.; Takazawa, K.; Yono, M.; Ervin, J.E.; Andrews, C.; Fogarty, C.; Eckermann, T.; et al. A Candidate Respiratory Syncytial Virus (RSV) Prefusion F Protein Investigational Vaccine (RSVPreF3 OA) Is Immunogenic When Administered in Adults ≥ 60 Years of Age: Results at 6 Months after Vaccination. Open Forum Infect. Dis. 2022, 9, ofac492.174. [Google Scholar] [CrossRef]
- Full Prescribing Information. Available online: https://gskpro.com/content/dam/global/hcpportal/en_US/Prescribing_Information/Arexvy/pdf/AREXVY.PDF (accessed on 24 August 2023).
- Papi, A.; Ison, M.G.; Langley, J.M.; Lee, D.-G.; Leroux-Roels, I.; Martinon-Torres, F.; Schwarz, T.F.; van Zyl-Smit, R.N.; Campora, L.; Dezutter, N.; et al. Respiratory Syncytial Virus Prefusion F Protein Vaccine in Older Adults. N. Engl. J. Med. 2023, 388, 595–608. [Google Scholar] [CrossRef] [PubMed]
- GSK’s Older Adult Respiratory Syncytial Virus (RSV) Vaccine Candidate Shows 94.1% Reduction in Severe RSV Disease and Overall Vaccine Efficacy of 82.6% in Pivotal Trial. Available online: https://www.gsk.com/en-gb/media/press-releases/gsk-s-older-adult-respiratory-syncytial-virus-rsv-vaccine-candidate/ (accessed on 24 August 2023).
- FDA Approves First Respiratory Syncytial Virus (RSV) Vaccine. Available online: https://www.fda.gov/news-events/press-announcements/fda-approves-first-respiratory-syncytial-virus-rsv-vaccine#:~:text=Today%2CtheU.S.Foodand,yearsofageandolder (accessed on 24 August 2023).
- Falsey, A.R.; Walsh, E.E.; Scott, D.A.; Gurtman, A.; Zareba, A.; Jansen, K.U.; Gruber, W.C.; Dormitzer, P.R.; Swanson, K.A.; Jiang, Q.; et al. Phase 1/2 Randomized Study of the Immunogenicity, Safety, and Tolerability of a Respiratory Syncytial Virus Prefusion F Vaccine in Adults with Concomitant Inactivated Influenza Vaccine. J. Infect. Dis. 2022, 225, 2056–2066. [Google Scholar] [CrossRef]
- Baber, J.; Arya, M.; Moodley, Y.; Jaques, A.; Jiang, Q.; Swanson, K.A.; Cooper, D.; Maddur, M.S.; Loschko, J.; Gurtman, A.; et al. A Phase 1/2 Study of a Respiratory Syncytial Virus Prefusion F Vaccine with and Without Adjuvant in Healthy Older Adults. J. Infect. Dis. 2022, 226, 2054–2063. [Google Scholar] [CrossRef]
- Peterson, J.T.; Zareba, A.M.; Fitz-Patrick, D.; Essink, B.J.; Scott, D.A.; Swanson, K.A.; Chelani, D.; Radley, D.; Cooper, D.; Jansen, K.U.; et al. Safety and Immunogenicity of a Respiratory Syncytial Virus Prefusion F Vaccine When Coadministered with a Tetanus, Diphtheria, and Acellular Pertussis Vaccine. J. Infect. Dis. 2022, 225, 2077–2086. [Google Scholar] [CrossRef]
- Simões, E.A.F.; Center, K.J.; Tita, A.T.N.; Swanson, K.A.; Radley, D.; Houghton, J.; McGrory, S.B.; Gomme, E.; Anderson, M.; Roberts, J.P.; et al. Prefusion F Protein–Based Respiratory Syncytial Virus Immunization in Pregnancy. N. Engl. J. Med. 2022, 386, 1615–1626. [Google Scholar] [CrossRef] [PubMed]
- Schmoele-Thoma, B.; Zareba, A.M.; Jiang, Q.; Maddur, M.S.; Danaf, R.; Mann, A.; Eze, K.; Fok-Seang, J.; Kabir, G.; Catchpole, A.; et al. Vaccine Efficacy in Adults in a Respiratory Syncytial Virus Challenge Study. N. Engl. J. Med. 2022, 386, 2377–2386. [Google Scholar] [CrossRef]
- Pfizer Announces Positive Top-Line Data of Phase 3 Global Maternal Immunization Trial for Its Bivalent Respiratory Syncytial Virus (RSV) Vaccine Candidate. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-announces-positive-top-line-data-phase-3-global (accessed on 24 August 2023).
- Kampmann, B.; Madhi, S.A.; Munjal, I.; Simões, E.A.F.; Pahud, B.A.; Llapur, C.; Baker, J.; Pérez Marc, G.; Radley, D.; Shittu, E.; et al. Bivalent Prefusion F Vaccine in Pregnancy to Prevent RSV Illness in Infants. N. Engl. J. Med. 2023, 388, 1451–1464. [Google Scholar] [CrossRef] [PubMed]
- Pfizer Announces Positive Top-Line Data from Phase 3 Trial of Older Adults for Its Bivalent Respiratory Syncytial Virus (RSV) Vaccine Candidate. Available online: https://www.pfizer.com/news/press-release/press-release-detail/pfizer-announces-positive-top-line-data-phase-3-trial-older (accessed on 24 August 2023).
- Respiratory Syncytial Virus Vaccine (Proposed Trade Name: Abrysvo). Available online: https://www.fda.gov/media/165623/download (accessed on 24 August 2023).
- Walsh, E.E.; Pérez Marc, G.; Zareba, A.M.; Falsey, A.R.; Jiang, Q.; Patton, M.; Polack, F.P.; Llapur, C.; Doreski, P.A.; Ilangovan, K.; et al. Efficacy and Safety of a Bivalent RSV Prefusion F Vaccine in Older Adults. N. Engl. J. Med. 2023, 388, 1465–1477. [Google Scholar] [CrossRef]
- Respiratory Syncytial Virus Stabilized Bivalent Prefusion F Subunit Vaccine (rsvpref/abrysvo). Available online: https://www.fda.gov/media/165625/download (accessed on 24 August 2023).
- Blue Lake Biotechnology Announces Positive Interim Phase 1 Data for BLB201 Intranasal RSV Vaccine. Available online: https://www.bluelakebiotechnology.com/news/blue-lake-biotechnology-announces-positive-interim-phase-1-data-for-blb201-intranasal-rsv-vaccine-rnbjl (accessed on 24 August 2023).
- Codagenix Receives FDA Clearance of Investigational New Drug Application for Live-Attenuated, Intranasal RSV Vaccine Candidate CodaVaxTM-RSV. Available online: https://codagenix.com/codagenix-receives-fda-clearance-of-investigational-new-drug-application-for-live-attenuated-intranasal-rsv-vaccine-candidate-codavax-rsv/ (accessed on 24 August 2023).
- Codagenix Inc. Completes Dosing for Phase 1 Trial of Live-Attenuated, Intranasal Vaccine for Respiratory Syncytial Virus (RSV). Available online: https://www.prnewswire.com/news-releases/codagenix-inc-completes-dosing-for-phase-1-trial-of-live-attenuated-intranasal-vaccine-for-respiratory-syncytial-virus-rsv-301182228.html (accessed on 24 August 2023).
- Verdijk, P.; van der Plas, J.L.; van Brummelen, E.M.J.; Jeeninga, R.E.; de Haan, C.A.M.; Roestenberg, M.; Burggraaf, J.; Kamerling, I.M.C. First-in-Human Administration of a Live-Attenuated RSV Vaccine Lacking the G-Protein Assessing Safety, Tolerability, Shedding and Immunogenicity: A Randomized Controlled Trial. Vaccine 2020, 38, 6088–6095. [Google Scholar] [CrossRef]
- Abarca, K.; Rey-Jurado, E.; Muñoz-Durango, N.; Vázquez, Y.; Soto, J.A.; Gálvez, N.M.S.; Valdés-Ferrada, J.; Iturriaga, C.; Urzúa, M.; Borzutzky, A.; et al. Safety and Immunogenicity Evaluation of Recombinant BCG Vaccine against Respiratory Syncytial Virus in a Randomized, Double-Blind, Placebo-Controlled Phase I Clinical Trial. eClinicalMedicine 2020, 27, 100517. [Google Scholar] [CrossRef]
- Scaggs Huang, F.; Bernstein, D.I.; Slobod, K.S.; Portner, A.; Takimoto, T.; Russell, C.J.; Meagher, M.; Jones, B.G.; Sealy, R.E.; Coleclough, C.; et al. Safety and Immunogenicity of an Intranasal Sendai Virus-Based Vaccine for Human Parainfluenza Virus Type I and Respiratory Syncytial Virus (SeVRSV) in Adults. Hum. Vaccines Immunother. 2021, 17, 554–559. [Google Scholar] [CrossRef] [PubMed]
- Medzihradsky, O.F.; Fierro, C.; Schlingmann-molina, B.L.; Cheng, X.; Garg, A.; Blanco, J.C.G.; Tang, R.S.; Slobod, K.S.; Moore, M.L.; City, R.; et al. The Codon Deoptimized, Intranasally Delivered, Live Attenuated RSV Vaccine MV-012-968 Is Well Tolerated and Increases RSV PreF Specific IgA Levels in Healthy Adults. Meissa Vaccines 2016, 1137848. [Google Scholar]
- Meissa Vaccines Provides a Pipeline Update on Vaccine Candidates for COVID-19 and RSV. Available online: https://www.meissavaccines.com/post/meissa-vaccines-provides-a-pipeline-update-on-vaccine-candidates-for-covid-19-and-rsv (accessed on 24 August 2023).
- Meissa Announces 1st Dosing in Phase 2 Study of Intranasal Live Attenuated Vaccine Candidate for RSV. Available online: https://www.meissavaccines.com/post/meissa-announces-1st-dosing-in-phase-2-study-of-intranasal-live-attenuated-vaccine-candidate-for-rsv (accessed on 24 August 2023).
- Meissa’s Positive Interim Clinical Data for Its Intranasal Live Attenuated RSV Vaccine for Infants. Available online: https://www.meissavaccines.com/post/meissa-s-positive-interim-clinical-data-for-its-intranasal-live-attenuated-rsv-vaccine-for-infants (accessed on 24 August 2023).
- Karron, R.A.; Luongo, C.; Mateo, J.S.; Wanionek, K.; Collins, P.L.; Buchholz, U.J. Safety and Immunogenicity of the Respiratory Syncytial Virus Vaccine RSV/ΔNS2/Δ1313/I1314L in RSVSeronegative Children. J. Infect. Dis. 2020, 222, 82–91. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, C.K.; Karron, R.A.; Muresan, P.; Kelly, M.S.; Mcfarland, E.J.; Perlowski, C.; Libous, J.; Oliva, J.; Jean-philippe, P.; Moyejr, J.; et al. Evaluation of Recombinant Live-Attenuated Respiratory Syncytial Virus (RSV) Vaccines RSV/ΔNS2/Δ1313/I1314L and RSV/276 in RSV-Seronegative Children. J. Infect. Dis. 2022, 226, 92868. [Google Scholar] [CrossRef] [PubMed]
- McFarland, E.J.; Karron, R.A.; Muresan, P.; Cunningham, C.K.; Libous, J.; Perlowski, C.; Thumar, B.; Gnanashanmugam, D.; Moye, J.; Schappell, E.; et al. Live Respiratory Syncytial Virus Attenuated by M2-2 Deletion and Stabilized Temperature Sensitivity Mutation 1030s Is a Promising Vaccine Candidate in Children. J. Infect. Dis. 2020, 221, 534–543. [Google Scholar] [CrossRef]
- Karron, R.A.; Luongo, C.; Woods, S.; Oliva, J.; Collins, P.L.; Buchholz, U.J.; Council-Dibitetto, C.; Gatto, M.; Ghasri, T.; Gormley, A.; et al. Evaluation of the Live-Attenuated Intranasal Respiratory Syncytial Virus (RSV) Vaccine RSV/6120/ΔNS2/1030s in RSV-Seronegative Young Children. J. Infect. Dis. 2023, jiad281. [Google Scholar] [CrossRef]
- Samy, N.; Reichhardt, D.; Schmidt, D.; Chen, L.M.; Silbernagl, G.; Vidojkovic, S.; Meyer, T.P.; Jordan, E.; Adams, T.; Weidenthaler, H.; et al. Safety and Immunogenicity of Novel Modified Vaccinia Ankara-Vectored RSV Vaccine: A Randomized Phase I Clinical Trial. Vaccine 2020, 38, 2608–2619. [Google Scholar] [CrossRef]
- Jordan, E.; Lawrence, S.J.; Meyer, T.P.H.; Schmidt, D.; Schultz, S.; Mueller, J.; Stroukova, D.; Koenen, B.; Gruenert, R.; Silbernagl, G.; et al. Broad Antibody and Cellular Immune Response from a Phase 2 Clinical Trial with a Novel Multivalent Poxvirus-Based Respiratory Syncytial Virus Vaccine. J. Infect. Dis. 2021, 223, 1062–1072. [Google Scholar] [CrossRef]
- MVA-BN® RSV. Available online: https://www.bavarian-nordic.com/what-we-do/pipeline/rsv.aspx (accessed on 24 August 2023).
- Stuart, A.S.V.; Virta, M.; Williams, K.; Seppa, I.; Hartvickson, R.; Omoruyi, E.; Bastian, R.; Haazen, W.; Salisch, N.; Gymnopoulou, E.; et al. Phase 1/2a Safety and Immunogenicity of an Adenovirus 26 Vector Respiratory Syncytial Virus (RSV) Vaccine Encoding Prefusion F in Adults 18–50 Years and RSV-Seropositive Children 12–24 Months. J. Infect. Dis. 2022, 1, 71–82. [Google Scholar] [CrossRef]
- Schlake, T.; Thess, A.; Fotin-mleczek, M.; Kallen, K. Developing MRNA-Vaccine Technologies. RNA Biol. 2012, 9, 1319–1330. [Google Scholar] [CrossRef]
- Barbier, A.J.; Jiang, A.Y.; Zhang, P.; Wooster, R.; Anderson, D.G. The Clinical Progress of MRNA Vaccines and Immunotherapies. Nat. Biotechnol. 2022, 40, 840–854. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.A. A Comparison of Plasmid DNA and MRNA as Vaccine Technologies Margaret. Vaccines 2019, 7, 37. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, L.; Losick, R. Delivering the Message: How a Novel Technology Enabled the Rapid Development of Effective Vaccines. Cell 2020, 184, 5271–5274. [Google Scholar] [CrossRef]
- Zhang, C.; Maruggi, G.; Shan, H.; Li, J. Advances in MRNA Vaccines for Infectious Diseases. Front. Immunol. 2019, 10, 594. [Google Scholar] [CrossRef]
- Deng, Z.; Tian, Y.; Song, J.; An, G.; Yang, P. MRNA Vaccines: The Dawn of a New Era of Cancer Immunotherapy. Front. Immunol. 2022, 13, 887125. [Google Scholar] [CrossRef]
- Moderna Initiates Phase 3 Portion of Pivotal Trial for mRNA Respiratory Syncytial Virus (RSV) Vaccine Candidate, Following Independent Safety Review of Interim Data. Available online: https://investors.modernatx.com/news/news-details/2022/Moderna-Initiates-Phase-3-Portion-of-Pivotal-Trial-for-mRNA-Respiratory-Syncytial-Virus-RSV-Vaccine-Candidate-Following-Independent-Safety-Review-of-Interim-Data/default.aspx (accessed on 24 August 2023).
- Moderna Announces Global Regulatory Submissions for Its Respiratory Syncytial Virus (RSV) Vaccine, mRNA-1345. Available online: https://investors.modernatx.com/news/news-details/2023/Moderna-Announces-Global-Regulatory-Submissions-For-Its-Respiratory-Syncytial-Virus-RSV-Vaccine-MRNA-1345/default.aspx (accessed on 24 August 2023).
- O’connell, A.K.; Douam, F. Humanized Mice for Live-Attenuated Vaccine Research: From Unmet Potential to New Promises. Vaccines 2020, 8, 36. [Google Scholar] [CrossRef]
- Mok, D.Z.L.; Chan, K.R. The Effects of Pre-Existing Antibodies on Live-Attenuated Viral Vaccines. Viruses 2020, 12, 520. [Google Scholar] [CrossRef]
- Ghattas, M.; Dwivedi, G.; Lavertu, M.; Alameh, M.G. Vaccine Technologies and Platforms for Infectious Diseases: Current Progress, Challenges, and Opportunities. Vaccines 2021, 9, 1490. [Google Scholar] [CrossRef]
- Vetter, V.; Denizer, G.; Friedland, L.R.; Krishnan, J.; Shapiro, M. Understanding Modern-Day Vaccines: What You Need to Know. Ann. Med. 2018, 50, 110–120. [Google Scholar] [CrossRef]
- Karron, R.A.; Atwell, J.E.; McFarland, E.J.; Cunningham, C.K.; Muresan, P.; Perlowski, C.; Libous, J.; Spector, S.A.; Yogev, R.; Aziz, M.; et al. Live-Attenuated Vaccines Prevent Respiratory Syncytial Virus-Associated Illness in Young Children. Am. J. Respir. Crit. Care Med. 2021, 203, 594–603. [Google Scholar] [CrossRef]
- Mazur, N.I.; Terstappen, J.; Baral, R.; Bardají, A.; Beutels, P.; Buchholz, U.J.; Cohen, C.; Crowe, J.E., Jr.; Cutland, C.L.; Eckert, L.; et al. Review Respiratory Syncytial Virus Prevention within Reach: The Vaccine and Monoclonal Antibody Landscape. Lancet Infect. Dis. 2023, 1, e2–e21. [Google Scholar] [CrossRef] [PubMed]
- Mazur, N.I.; Higgins, D.; Nunes, M.C.; Melero, J.A.; Langedijk, A.C.; Horsley, N.; Buchholz, U.J.; Openshaw, P.J.; McLellan, J.S.; Englund, J.A.; et al. The Respiratory Syncytial Virus Vaccine Landscape: Lessons from the Graveyard and Promising Candidates. Lancet Infect. Dis. 2018, 18, e295–e311. [Google Scholar] [CrossRef] [PubMed]
- Eichinger, K.M.; Kosanovich, J.L.; Lipp, M.; Empey, K.M.; Petrovsky, N. Strategies for Active and Passive Pediatric RSV Immunization. Ther. Adv. Vaccines Immunother. 2021, 9, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Widjojoatmodjo, M.N.; Boes, J.; van Bers, M.; van Remmerden, Y.; Roholl, P.J.M.; Luytjes, W. A Highly Attenuated Recombinant Human Respiratory Syncytial Virus Lacking the G Protein Induces Long-Lasting Protection in Cotton Rats. Virol. J. 2010, 7, 114. [Google Scholar] [CrossRef]
- Bueno, S.M.; González, P.A.; Cautivo, K.M.; Mora, J.E.; Leiva, E.D.; Tobar, H.E.; Fennelly, G.J.; Eugenin, E.A.; Jacobs, W.R.; Riedel, C.A.; et al. Protective T Cell Immunity against Respiratory Syncytial Virus Is Efficiently Induced by Recombinant BCG. Proc. Natl. Acad. Sci. USA 2008, 105, 20822–20827. [Google Scholar] [CrossRef] [PubMed]
- Cautivo, K.M.; Bueno, S.M.; Cortes, C.M.; Wozniak, A.; Riedel, C.A.; Kalergis, A.M. Efficient Lung Recruitment of Respiratory Syncytial Virus-Specific Th1 Cells Induced by Recombinant Bacillus Calmette-Guérin Promotes Virus Clearance and Protects from Infection. J. Immunol. 2010, 185, 7633–7645. [Google Scholar] [CrossRef]
- Díaz, F.E.; Guerra-Maupome, M.; McDonald, P.O.; Rivera-Pérez, D.; Kalergis, A.M.; McGill, J.L. A Recombinant BCG Vaccine Is Safe and Immunogenic in Neonatal Calves and Reduces the Clinical Disease Caused by the Respiratory Syncytial Virus. Front. Immunol. 2021, 12, 664212. [Google Scholar] [CrossRef]
- Russell, C.J.; Hurwitz, J.L. Sendai Virus-Vectored Vaccines That Express Envelope Glycoproteins of Respiratory Viruses. Viruses 2021, 13, 1023. [Google Scholar] [CrossRef]
- Meissa Vaccines. Available online: https://www.meissavaccines.com/technology (accessed on 24 August 2023).
- Luongo, C.; Winter, C.C.; Collins, P.L.; Buchholz, U.J. Respiratory Syncytial Virus Modified by Deletions of the NS2 Gene and Amino Acid S1313 of the L Polymerase Protein Is a Temperature-Sensitive, Live-Attenuated Vaccine Candidate That Is Phenotypically Stable at Physiological Temperature. J. Virol. 2013, 87, 1985–1996. [Google Scholar] [CrossRef]
- Liang, B.; Matsuoka, Y.; Le Nouën, C.; Liu, X.; Herbert, R.; Swerczek, J.; Santos, C.; Paneru, M.; Collins, P.L.; Buchholz, U.J.; et al. A Parainfluenza Virus Vector Expressing the Respiratory Syncytial Virus (RSV) Prefusion F Protein Is More Effective than RSV for Boosting a Primary Immunization with RSV. J. Virol. 2020, 95, e01512-20. [Google Scholar] [CrossRef]
- Young, A.; Isaacs, A.; Scott, C.A.P.; Modhiran, N.; Mcmillan, C.L.D.; Cheung, S.T.M.; Barr, J.; Marsh, G.; Thakur, N.; Bailey, D.; et al. A Platform Technology for Generating Subunit Vaccines against Diverse Viral Pathogens. Front. Immunol. 2022, 13, 963023. [Google Scholar] [CrossRef] [PubMed]
- Cid, R.; Bol, J. Platforms for Production of Protein-Based Vaccines: From Classical to Next-Generation Strategies. Biomolecules 2021, 11, 1027. [Google Scholar] [CrossRef] [PubMed]
- Vartak, A.; Sucheck, S.J. Recent Advances in Subunit Vaccine Carriers. Vaccines 2016, 4, 12. [Google Scholar] [CrossRef]
- Baxter, D. Active and Passive Immunity, Vaccine Types, Excipients and Licensing. Occup. Med. 2007, 57, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.J.; Dormitzer, P.R.; Nokes, D.J.; Rappuoli, R.; Roca, A.; Graham, B.S. Strategic Priorities for Respiratory Syncytial Virus (RSV) Vaccine Development. Vaccine 2013, 31, B209–B215. [Google Scholar] [CrossRef] [PubMed]
- Moyle, P.M.; Toth, I. Modern Subunit Vaccines: Development, Components, and Research Opportunities. ChemMedChem 2013, 8, 360–376. [Google Scholar] [CrossRef]
- Donaldson, B.; Lateef, Z.; Walker, G.F.; Young, S.L.; Ward, V.K. Virus-like Particle Vaccines: Immunology and Formulation for Clinical Translation. Expert Rev. Vaccines 2018, 17, 833–849. [Google Scholar] [CrossRef]
- Fuenmayor, J.; Gòdia, F.; Cervera, L. Production of Virus-like Particles for Vaccines. N. Biotechnol. 2017, 39, 174–180. [Google Scholar] [CrossRef]
- Nooraei, S.; Bahrulolum, H.; Hoseini, Z.S.; Katalani, C.; Hajizade, A. Virus-like Particles: Preparation, Immunogenicity and Their Roles as Nanovaccines and Drug Nanocarriers. J. Nanobiotechnol. 2021, 19, 59. [Google Scholar] [CrossRef]
- Advaccine. Available online: http://dev.vn.euroland.com:8129/en/pipeline-page/pipeline/#ADV110 (accessed on 24 August 2023).
- Li, C.; Zhou, X.; Zhong, Y.; Li, C.; Dong, A.; He, Z.; Zhang, S.; Wang, B. A Recombinant G Protein Plus Cyclosporine A–Based Respiratory Syncytial Virus Vaccine Elicits Humoral and Regulatory T Cell Responses against Infection without Vaccine-Enhanced Disease. J. Immunol. 2016, 196, 1721–1731. [Google Scholar] [CrossRef]
- Crank, M.C.; Ruckwardt, T.J.; Chen, M.; Morabito, K.M.; Phung, E.; Costner, P.J.; Holman, L.A.; Hickman, S.P.; Berkowitz, N.M.; Gordon, I.J.; et al. A Proof of Concept for Structure-Based Vaccine Design Targeting RSV in Humans. Science 2019, 365, 505–509. [Google Scholar] [CrossRef] [PubMed]
- Flynn, J.A.; Durr, E.; Swoyer, R.; Cejas, P.J.; Horton, M.S.; Galli, J.D.; Cosmi, S.A.; Espeseth, A.S.; Bett, A.J.; Zhang, L. Stability Characterization of a Vaccine Antigen Based on the Respiratory Syncytial Virus Fusion Glycoprotein. PLoS ONE 2016, 11, e0164789. [Google Scholar] [CrossRef] [PubMed]
- McLellan, J.S. Structure-Based Design of a Fusion Glycoprotein Vaccine for Respiratory Syncytial Virus (Science (592)). Science 2013, 342, 931. [Google Scholar] [CrossRef]
- Patel, N.; Tian, J.-H.; Flores, R.; Jacobson, K.; Walker, M.; Portnoff, A.; Gueber-Xabier, M.; Massare, M.J.; Glenn, G.; Ellingsworth, L.; et al. Flexible RSV Prefusogenic Fusion Glycoprotein Exposes Multiple Neutralizing Epitopes That May Collectively Contribute to Protective Immunity. Vaccines 2020, 8, 607. [Google Scholar] [CrossRef] [PubMed]
- McLellan, J.S.; Chen, M.; Leung, S.; Graepel, K.W.; Du, X.; Yang, Y.; Zhou, T.; Baxa, U.; Yasuda, E.; Beaumont, T.; et al. Structure of RSV Fusion Glycoprotein Trimer Bound to a Prefusion-Specific Neutralizing Antibody. Science 2013, 340, 1113–1117. [Google Scholar] [CrossRef]
- Joyce, M.G.; Bao, A.; Chen, M.; Georgiev, I.S.; Ou, L.; Bylund, T.; Druz, A.; Kong, W.P.; Peng, D.; Rundlet, E.J.; et al. Crystal Structure and Immunogenicity of the Ds-Cav1-Stabilized Fusion Glycoprotein from Respiratory Syncytial Virus Subtype b Authors. Pathog. Immun. 2019, 4, 294–323. [Google Scholar] [CrossRef]
- Stewart-Jones, G.B.E.; Thomas, P.V.; Chen, M.; Druz, A.; Joyce, M.G.; Kong, W.P.; Sastry, M.; Soto, C.; Yang, Y.; Zhang, B.; et al. A Cysteine Zipper Stabilizes a Pre-Fusion F Glycoprotein Vaccine for Respiratory Syncytial Virus. PLoS ONE 2015, 10, e0128779. [Google Scholar] [CrossRef]
- Joyce, M.G.; Zhang, B.; Ou, L.; Chen, M.; Chuang, G.-Y.; Druz, A.; Kong, W.-P.; Lai, Y.-T.; Rundlet, E.J.; Tsybovsky, Y.; et al. Iterative Structure-Based Improvement of a Fusion-Glycoprotein Vaccine against RSV. Nat. Struct. Mol. Biol. 2016, 23, 811–820. [Google Scholar] [CrossRef]
- Zhang, L.; Durr, E.; Galli, J.D.; Cosmi, S.; Cejas, P.J.; Luo, B.; Touch, S.; Parmet, P.; Fridman, A.; Espeseth, A.S.; et al. Design and Characterization of a Fusion Glycoprotein Vaccine for Respiratory Syncytial Virus with Improved Stability. Vaccine 2018, 36, 8119–8130. [Google Scholar] [CrossRef]
- Chen, P.; Chen, M.; Menon, A.; Hussain, A.I.; Carey, E.; Lee, C.; Horwitz, J.; O’Connell, S.; Cooper, J.W.; Schwartz, R.; et al. Development of a High Yielding Bioprocess for a Pre-Fusion RSV Subunit Vaccine. J. Biotechnol. 2021, 325, 261–270. [Google Scholar] [CrossRef]
- Summary Basis for Regulatory Action-AREXVY. Available online: https://www.fda.gov/media/168519/download?attachment (accessed on 24 August 2023).
- US FDA Approves GSK’s Arexvy, the World’s First Respiratory Syncytial Virus (RSV) Vaccine for Older Adults. Available online: https://www.gsk.com/en-gb/media/press-releases/us-fda-approves-gsk-s-arexvy-the-world-s-first-respiratory-syncytial-virus-rsv-vaccine-for-older-adults/ (accessed on 24 August 2023).
- European Commission Authorises GSK’s Arexvy, the First Respiratory Syncytial Virus (RSV) Vaccine for Older Adults. Available online: https://www.gsk.com/en-gb/media/press-releases/european-commission-authorises-gsk-s-arexvy-the-first-respiratory-syncytial-virus-rsv-vaccine-for-older-adults/ (accessed on 24 August 2023).
- GSK Provides Further Update on Phase III RSV Maternal Vaccine Candidate Programme. Available online: https://www.gsk.com/en-gb/media/press-releases/gsk-provides-further-update-on-phase-iii-rsv-maternal-vaccine-candidate-programme/ (accessed on 24 August 2023).
- GSK Provides Update on Phase III RSV Maternal Vaccine Candidate Programme. Available online: https://www.gsk.com/en-gb/media/press-releases/gsk-provides-update-on-phase-iii-rsv-maternal-vaccine-candidate-programme/ (accessed on 24 August 2023).
- Full Prescribing Information-Arexvy. Available online: https://www.fda.gov/media/167805/download (accessed on 24 August 2023).
- U.S. FDA Approves ABRYSVOTM, Pfizer’s Vaccine for the Prevention of Respiratory Syncytial Virus (RSV) in Older Adults. Available online: https://www.pfizer.com/news/press-release/press-release-detail/us-fda-approves-abrysvotm-pfizers-vaccine-prevention (accessed on 24 August 2023).
- Summary Basis for Regulatory Action-ABRYSVO. Available online: https://www.fda.gov/media/172126/download?attachment (accessed on 24 August 2023).
- EMEA-002795-PIP01-20. Available online: https://www.ema.europa.eu/en/medicines/human/paediatric-investigation-plans/emea-002795-pip01-20 (accessed on 24 August 2023).
- Abrysvo. Available online: https://www.ema.europa.eu/en/medicines/human/summaries-opinion/abrysvo (accessed on 24 August 2023).
- U.S. FDA Approves ABRYSVOTM, Pfizer’s Vaccine for the Prevention of Respiratory Syncytial Virus (RSV) in Infants Through Active Immunization of Pregnant Individuals 32–36 Weeks of Gestational Age. Available online: https://www.pfizer.com/news/press-release/press-release-detail/us-fda-approves-abrysvotm-pfizers-vaccine-prevention-0 (accessed on 24 August 2023).
- European Commission Approves Pfizer’s ABRYSVOTM to Help Protect Infants through Maternal Immunization and Older Adults from RSV. Available online: https://www.pfizer.com/news/press-release/press-release-detail/european-commission-approves-pfizers-abrysvotm-help-protect#:~:text=ABRYSVO (accessed on 24 August 2023).
- ABRYSVO-Respiratory Syncytial Virus Vaccine Pfizer Laboratories Div Pfizer Inc. Available online: https://labeling.pfizer.com/ShowLabeling.aspx?id=19589#section-5.1 (accessed on 24 August 2023).
- Boytchev, H. Maternal RSV Vaccine: Further Analysis Is Urged on Preterm Births. BMJ 2023, 381, 1021. [Google Scholar] [CrossRef]
- Jackson, D.A.; Symons, R.H.; Berg, P. Biochemical Method for Inserting New Genetic Information into DNA of Simian Virus 40: Circular SV40 DNA Molecules Containing Lambda Phage Genes and the Galactose Operon of Escherichia Coli. Proc. Natl. Acad. Sci. USA 1972, 69, 2904–2909. [Google Scholar] [CrossRef] [PubMed]
- Hewson, R. Emerging Viruses and Current Strategies for Vaccine Intervention. Clin. Exp. Immunol. 2019, 196, 157–166. [Google Scholar] [CrossRef]
- Souza, A.P.D.; Haut, L.; Reyes-Sandoval, A.; Pinto, A.R. Recombinant Viruses as Vaccines against Viral Diseases. Braz. J. Med. Biol. Res. 2005, 38, 509–522. [Google Scholar] [CrossRef]
- Ramezanpour, B.; Haan, I.; Osterhaus, A.; Claassen, E. Vector-Based Genetically Modified Vaccines: Exploiting Jenner’s Legacy. Vaccine 2020, 34, 6436–6448. [Google Scholar] [CrossRef]
- Robert-Guroff, M. Replicating and Non-Replicating Viral Vectors for Vaccine Development. Curr. Opin. Biotechnol. 2007, 18, 546–556. [Google Scholar] [CrossRef]
- Nuance Pharma Announces Clearance of IND Application for Respiratory Syncytial Virus (RSV) Vaccine in China. Available online: https://www.prnewswire.com/news-releases/nuance-pharma-announces-clearance-of-ind-application-for-respiratory-syncytial-virus-rsv-vaccine-in-china-301676401.html (accessed on 24 August 2023).
- Bavarian Nordic Announces License and Supply Agreement with Nuance Pharma on the Development and Commercialization of Respiratory Syncytial Virus (RSV) Vaccine for Adults in China and Selected Asian Markets. Available online: https://www.bavarian-nordic.com/investor/news/news.aspx?news=6536 (accessed on 24 August 2023).
- Williams, K.; Bastian, A.R.; Feldman, R.A.; Omoruyi, E.; de Paepe, E.; Hendriks, J.; van Zeeburg, H.; Godeaux, O.; Langedijk, J.P.M.; Schuitemaker, H.; et al. Phase 1 Safety and Immunogenicity Study of a Respiratory Syncytial Virus Vaccine with an Adenovirus 26 Vector Encoding Prefusion F (Ad26.RSV.PreF) in Adults Aged ≥ 60 Years. J. Infect. Dis. 2020, 222, 979–988. [Google Scholar] [CrossRef]
- Janssen Provides Portfolio Update. Available online: https://www.jnj.com/janssen-provides-portfolio-update (accessed on 24 August 2023).
- Boyoglu-Barnum, S.; Chirkova, T.; Anderson, L.J. Biology of Infection and Disease Pathogenesis to Guide RSV Vaccine Development. Front. Immunol. 2019, 10, 1675. [Google Scholar] [CrossRef]
- Li, Y.; Wang, X.; Blau, D.M.; Caballero, M.T.; Feikin, D.R.; Gill, C.J.; Madhi, S.A.; Omer, S.B.; Simões, E.A.F.; Campbell, H.; et al. Global, Regional, and National Disease Burden Estimates of Acute Lower Respiratory Infections Due to Respiratory Syncytial Virus in Children Younger than 5 Years in 2019: A Systematic Analysis. Lancet 2022, 399, 2047–2064. [Google Scholar] [CrossRef]
- Herring, W.L.; Zhang, Y.; Shinde, V.; Stoddard, J.; Talbird, S.E.; Rosen, B. Clinical and Economic Outcomes Associated with Respiratory Syncytial Virus Vaccination in Older Adults in the United States. Vaccine 2022, 40, 483–493. [Google Scholar] [CrossRef]
- Zhang, S.; Akmar, L.Z.; Bailey, F.; Rath, B.A.; Alchikh, M.; Schweiger, B.; Lucero, M.G.; Nillos, L.T.; Kyaw, M.H.; Kieffer, A.; et al. Cost of Respiratory Syncytial Virus-Associated Acute Lower Respiratory Infection Management in Young Children at the Regional and Global Level: A Systematic Review and Meta-Analysis. J. Infect. Dis. 2020, 222, S680–S687. [Google Scholar] [CrossRef] [PubMed]
- RSV Vaccines 2023. Available online: https://www.precisionvaccinations.com/vaccines/rsv-vaccines-2023 (accessed on 24 August 2023).
- Vidal Valero, M. ‘A Good Day’: FDA Approves World’s First RSV Vaccine. Nature 2023, 617, 234–235. [Google Scholar] [CrossRef] [PubMed]
- Harris, E. FDA Clears RSV Vaccine for Adults Aged 60 Years or Older. JAMA 2023, 329, 1817. [Google Scholar] [CrossRef] [PubMed]
- Bavarian Nordic Announces Breakthrough Therapy Designation for Its RSV Vaccine Candidate for the Prevention of Respiratory Syncytial Virus in Older Adults. Available online: https://www.bavarian-nordic.com/investor/news/news.aspx?news=6478 (accessed on 24 August 2023).
- Bavarian Nordic Announces Grant of Prime Eligibility from the European Medicines Agency for its RSV Vaccine Candidate for the Prevention of Respiratory Syncytial Virus in Older Adults. Available online: https://www.bavarian-nordic.com/investor/news/news.aspx?news=6589 (accessed on 24 August 2023).
- Moderna Receives FDA Fast Track Designation for Respiratory Syncytial Virus (RSV) Vaccine (mRNA-1345). Available online: https://investors.modernatx.com/news/news-details/2021/Moderna-Receives-FDA-Fast-Track-Designation-for-Respiratory-Syncytial-Virus-RSV-Vaccine-mRNA-1345/default.aspx (accessed on 24 August 2023).

| Vaccine Category/ Vaccine Candidate | Vaccine Characteristics (Target Protein, Delivery System, and Adjuvant) | Target Population | Clinical Development Phase/ Clinical Trial Registration Number | Study Population | Results |
|---|---|---|---|---|---|
| mRNA vaccines | |||||
| mRNA-1345 | RSV prefusion form (preF) of the F protein Lipid nanoparticles (LNPs) - | Older Adult Pediatric | Phase I | ||
| NCT04528719 | Adults (18–49 and 65–79 years old), women of child-bearing potential (18–40 years old), and children (12–59 months old) | Interim analysis. No safety concerns. At least 20-fold and 11-fold increases in neutralizing antibody titers against RSV-A and RSV-B, respectively, for the younger adults and 14-fold and 10-fold increases for the older adults, respectively [27,28]. | |||
| NCT05397223 | Adults (18–75 years old) | Ongoing. | |||
| NCT05743881 | Children (5-<24 months old) | Ongoing. | |||
| NCT05585632 | Adults (50–75 years old) | Ongoing. | |||
| Phase II/III | |||||
| ConquerRSV/NCT05127434 | Adults (>60 years old) | Interim analysis. No safety concerns. Report of 9/64 events of RSV lower respiratory tract disease (LRTD) with ≥2 symptoms and 3/20 events with ≥3 symptoms for the vaccinees. Vaccine efficacy (VE): 83.7% for the definition of ≥2 symptoms and 82.4% for the definition of ≥3 symptoms [29,30]. | |||
| Phase III | |||||
| RSVictory/NCT05330975 | Adults (≥50 years old) | Ongoing. | |||
| RSV mRNA LNP CL-0059 RSV mRNA LNP CL-0137 | - LNPs (LNP CL-0059 or LNP CL-0137) - | Older adult | Phase I/II | ||
| NCT05639894 | Adults, non-pregnant or in breastfeeding women (18–50, ≥60 years old) | Ongoing. | |||
| Subunit/Viral-like-particle (VLP)-based vaccines | |||||
| IVX-A12 (VLP-based bivalent vaccine: IVX-121 against RSV and IVX-241 against hMPV) | Stabilized RSV preF protein VLP MF59 (oil-in-water emulsion adjuvant) or none | Older adult | Phase I | ||
| NCT05664334 | Adults (60–75 years old) | Interim analysis (May 2023). No safety concerns with comparable rates of adverse events (AEs) between vaccinees and placebo recipients. Induction of neutralizing antibodies against both strains of RSV and human metapneumovirus (hMPV), 4-fold and 3-fold geometric mean fold rise (GMFR) for RSV-A and RSV-B in seropositive participants, respectively; the combination is applicable [31]. | |||
| Phase II | |||||
| NCT05903183 | Adults (60–85 years old) | Ongoing. | |||
| V-306 (synthetic VLP-based vaccine) | Antigenic site II of RSV F protein (FsII) (preF and post-fusion form of the F protein (postF)) Self-assembling VLP - | Maternal Older adult (as a repeat dose stated from preclinical studies) | Phase I | ||
| NCT04519073 | Non-pregnant women (18–45 years old) | Well-tolerated, transient AEs of mild or moderate severity, no serious AEs (SAEs) or vaccine-associated AEs of special interest (AESIs). FsIIm-specific immunoglobulin G (IgG) antibodies multiplied in the intermediate and high dose groups, RSV-A-neutralizing antibodies (nAbs), and RSV F-specific IgG Abs were not affected; a marginal increase in nAbs against RSV in some participants; improvement needed [32]. | |||
| DPX-RSV(A) | SHe protein DepoVax(DPX) oil-based delivery system None or aluminum hydroxide | Older adult | Phase I | ||
| NCT02472548 | Adults (≥50–64 years old) | No SAEs; no safety concerns; mostly mild or moderate severity of AEs. Geometric mean titer (GMT) rise in SHe-specific antibodies for the unadjuvanted vaccine regimen; maintenance of the response for up to 180 days [33]. Last update: 2020. | |||
| VN-0200 | VAGA-9001a (no further information) - MABH-9002b (no further information) | Older adult | Phase I | ||
| NCT04914520 | Japanese adults (≥20–≤50 years old and ≥65–≤80 years old) | Not available. | |||
| Phase II | |||||
| NCT05547087 | Japanese adults (60–80 years) | Ongoing. | |||
| BARS13 (ADV110) | RSV G protein - None or cyclosporine A (CsA) | Older adult | Phase I | ||
| NCT04851977 | Adults, including non-pregnant women who apply contraceptive measures (18–45 years old) | Safe; most AEs recorded were of mild severity with no SAEs. Increase in geometric mean concentration (GMC) and geometric mean fold increase (GMFI) in RSV G-specific IgG Abs and GMCs of nAbs after each dose remaining until day 60 and supporting a two-dose vaccine schedule. Post hoc analysis suggested that vaccine-enhanced disease (VED) development seems unlikely [34,35]. | |||
| Phase II | |||||
| NCT04681833 | Older adults (60–80 years old) | Ongoing. | |||
| DS-Cav1 (VRC-RSVRGP084-00-VP) | Stabilized RSV preF protein - None or aluminum hydroxide | Older adult Maternal | Phase I | ||
| NCT03049488 | Adults (18–50 years old) | Well-tolerated with no SAEs and AEs of mild or moderate severity among groups. Multiple Ab inductions. nAbs against RSV-A and RSV-B increased, specifically for the high-dose group, and remained until week 44. IgG and immunoglobulin A (IgA) Abs against RSV-A preF, apex-binding, and side-binding epitopes also increased. A two-dose vaccine schedule and alum addition are not recommended [36]. | |||
| RSVPreF3 OA (GSK3844766A) Market-approved Name: Arexvy/AREXVY | Stabilized in trimeric form RSV preF protein - Adjuvant System 01 (AS01E, AS01B) | Older adult Maternal (terminated on February 2022 due to safety signal -association with preterm births) | Phases I/II | ||
| NCT03814590 | Adults (18–40 and 60–80 years old) | Well-tolerated. RSV-A and RSV-B nAbs increased and remained above baseline until month 14. The highest dose (120μg) and AS01E were chosen for further testing [37]. | |||
| Phase I | |||||
| NCT04090658 | Japanese adults (60–80 years old) | Well-tolerated. No SAEs associated with the vaccine. Increasing trend of RSVPreF3-specific IgG GMCs and neutralizing antibodies The two-dose vaccine schedule did not cause further increases [38]. | |||
| Phase II | |||||
| NCT04657198 (extension trial of NCT03814590) | Older adult participants of NCT03814590 | Low rates of SAEs. Similar recorded rates of pain as solicited local AE between groups, higher reported rate of unsolicited AEs, and similar reported rates for low- and medium-dose groups compared to the high-dose group. Humoral immunity activation observed by antibody titers’ increases. | |||
| Phase III | |||||
| NCT04732871 | Older adults (≥60 years old) | Interim analysis. Well-tolerated. One vaccine-associated case of Guillain-Barré syndrome. Increase in RSV-A and RSV-B nAb GMTs and RSVPreF3-specific IgG GMCs until one month and maintenance at higher levels than the baseline until month 6 [39,40]. | |||
| NCT04841577 (co-administration with FLU-QIV) | Older adults (≥60 years old) | Two cases of acute disseminated encephalomyelitis (ADME), one of which was fatal, in the simultaneous co-administration group [41]. | |||
| NCT04886596 | Older adults (≥60 years old) | VE against RSV-LRTD: 82.6%. VE against severe RSV-LRTD: 94.1% VE against RSV-acute respiratory infection (ARI): 71.7%. VE for participants with pre-existing stable diseases and the age group of 70–79 years of this category: 94.6% and 93.8%, respectively. Similar response among RSV-A and RSV-B. Solicited AEs of mild or moderate severity were common. Similar rates of SAEs, potential immune-mediated diseases (pIMDs), and fatal AEs between the groups. Severe-grade atrial fibrillation experienced by 13 vaccinees and 15 placebo recipients [41,42,43,44]. | |||
| NCT05059301 | Older adults (≥60 years old) | Different lots of the vaccine caused similar antibody responses and similar rates of solicited local, systemic, and unsolicited AEs. | |||
| NCT05559476 (co-administration with FLU HD vaccine) | Older adults (≥65 years old) | Ongoing. | |||
| NCT05568797 (co-administration with FLU aQIV vaccine) | Older adults (≥65 years old) | Ongoing. | |||
| NCT05590403 | Adults (50–59 and ≥60 years old) | Ongoing. | |||
| RSVpreF Market-approved Name: Abrysvo/ABRYSVO | Stabilized RSV preF protein of both strains - Aluminium hydroxide (Al(OH)3) | Older adult (Approved) Maternal (Approved) | Phase I/II | ||
| NCT03529773 (alone or co-administration with SIIV) | Adults (18–45 and 50–85 years old) | No SAEs were vaccine-associated. GMTs of RSV-A and RSV-B nAbs increased with a more manifest increase in women and remained at levels above the baseline until 12 months for the younger and older adults. Younger adults experienced more common AEs compared to the older adults. The 120μg unadjuvanted regimen was selected for further assessment [45]. | |||
| NCT03572062 (test of CpG a TLR-9 receptor agonist as adjuvant) | Older adults (65–85 years old) | The study was halted since CpG did not increase immunogenicity [46]. | |||
| NCT05788237 (Phase Ib, co-administration with modRNA qIRV flu vaccine) | Older adults (≥60 years old) | Ongoing. | |||
| Phase II | |||||
| NCT04071158 (Phase IIb, co-administration with Tetanus, Diphtheria and Pertussis (TDAP)) | Non-pregnant women (18–49 years old) | Mild/moderate AEs with similar distribution among groups; no safety concerns. Non-inferiority proof was documented for all the components of the vaccines, except for pertussis for the co-administration compared to the single administration of RSVpreF and TDAP [47]. | |||
| NCT04032093 (Phase IIb) | Pregnant women (18–49 years old) | Interim analysis. Similar or higher increase in the GMTs of RSV-A- and RSV-B-neutralizing antibodies to the non pregnant vaccinees of the previous trial; increased infant titers; no significant safety concerns for pregnant women or infants occurred; preterm births were measured at a rate of 3.7%. Post-hoc analysis showed a VE of 84.7% and 91.5% for medically attended RSV lower respiratory tract infection (MA-RSV-LRTI) and severe RSV lower respiratory tract infection (LRTI), respectively. The 120 μg without adjuvant proceeded to further evaluation [48]. | |||
| NCT04785612 (Phase IIa, RSV challenge study) | Adults (18–50 years old) | Vaccine was deemed safe and protective against RSV challenge; 6% of the vaccinees and 48% of the placebo recipients had symptoms during infection. Low levels of viral load measurements and shorter viral shedding for vaccinees. RSV-A and RSV-B nAb titers showed a 20-fold increase, and RSV preF-IgG titers increased. AEs had a similar pattern of report between groups and were of mild severity with no SAEs [49]. | |||
| Phase III | |||||
| NCT04424316 (name: MATISSE) | Pregnant women (≤49 years old) | Interim analysis. Well-tolerated. No safety concerns for pregnant women or infants. VE: 81.8% protection against severe MA-LRTI for infants within 90 days after birth and 69.4% in the 6-month period. VE: 57.1% (not significant) clinically assessable for MA-LRTI and 51.3% in the 6-month period [50,51] | |||
| NCT05035212 (name: RENOIR) | Older adults (≥60 years old) | Interim analysis VE: 66.7% for LRTI with ≥2 symptoms, 85.7% for LRTI with ≥3 symptoms, and 62.1% for RSV-ARI. Acceptable safety profile; fatal SAEs had a similar distribution among groups. AEs were common among vaccinees, especially in the age group of 60–69 years. Ten cases of atrial fibrillation occurred among the vaccine recipients compared to four in placebo recipients, and two cases of Guillain-Barré syndrome (possibly vaccine-associated) were recorded [52,53,54]. | |||
| NCT05096208 (tested 3 different lots) | Adults, including non-pregnant women (18–49 years old) | Well-tolerated, the primary outcome was met [53,55]. | |||
| NCT05301322 (co-administration with SIIV) | Older adults (≥65 years old) | Not available. | |||
| NCT05842967 (name: MONET) | Adults with risk factors for severe RSV-mediated disease (≥18 years old) | Ongoing. | |||
| Live-attenuated/Chimeric vaccines | |||||
| BLB-201 | Full-length RSV F-protein Live-attenuated parainfluenza virus-type 5 (PIV5) - | Pediatric (under 2 years of age) Older adult | Phase I | ||
| NCT05281263 | Adults (18–59 years and 60–75 years old) | Interim analysis. A total of 64% of the vaccine recipients experienced a rise in RSV antibody responses and low replication rate, and there were no major safety concerns [56]. | |||
| Phases I/II | |||||
| NCT05655182 | Seropositive children (18–59 months old) and seropositive or seronegative children (6–24 months old) | Ongoing. | |||
| CodaVax-RSV | Modifications of the genome - - | Pediatric | Phase I | ||
| NCT04295070 | Adults (18–49 and 50–75 years old) | Safe; induction of cellular immunity [57,58]. | |||
| NCT04919109 | Seropositive (2–5 years old) and seronegative (6 months old to <2 years old) children | Ongoing | |||
| RSV-ΔG | Deletion of G-protein - - | Pediatric | Phase I | ||
| NTR7173 (Netherlands Trial Register) | Adults (18–50 years old) | AEs of mild or moderate severity with a similar distribution between groups; no SAEs; and no induction of immunity in the seropositive population. The next step is a trial including seronegative children [59]. | |||
| rBCG-N-hRSV (chimeric) | Live-attenuated Mycobacterium bovis strain Bacillus-Calmette-Guérin (BCG) vaccine modified to express N protein of RSV | Pediatric Older adults | Phase I | ||
| NCT03213405 | Younger adults (18–50 years old) | No evidence of virulence. AEs of mild or moderate severity. Dose-dependent increase trend for IgG antibodies against both N-RSV and BCG; the post-vaccination in vitro challenge of participants’ cells with N-protein activates the production of cytokines [60]. | |||
| SeVRSV (chimeric) | Modified parainfluenza virus-type 1 (PIV-1) that expresses the F-protein of RSV | Pediatric | Phase I | ||
| NCT03473002 | Adults (18–45 years old) | Frequently reported local and systemic AEs of mild or moderate severity; no SAEs. No 4-fold increase in RSV-specific antibodies (pre-exposed patients; information deficit). A further evaluation in seronegative children is planned [61]. | |||
| MV-012-968 | Codon deoptimization of NS1, NS2, and G genes and SH gene remotion for attenuation | Pediatric | Phase I | ||
| NCT04227210 | Adults (18–40 years old) | Confirmed attenuation; mucosal RSV-specific immunity; RSV-specific antibody titers did not increase (seropositive population) [62,63]. | |||
| NCT04444284 (Phase Ib) | Seropositive children (15–59 months old) | Preliminary data. Specific mucosal immunity induction, good safety profile, and no signs of viral replication [64]. | |||
| NCT04909021 (Phase Ic) | Seronegative children (6–36 months old) | Interim data. Acceptable safety profile; mild solicited AEs; no SAEs. Changes in measurements of neutralizing antibody titers in 78% of the population with a rate of 89%, including the mucosal response [65]. | |||
| Phase II | |||||
| NCT04690335 (viral challenge model) | Adults (18–45 years old) | Not available. | |||
| VAD00001 | Live-attenuated | Pediatric | Phase I | ||
| NCT04491877 | Children (6–18 months old) | Not available. | |||
| RSV ΔNS2/Δ1313/I1314L | Deletion of the NS2 gene, Δ1313 codon in the L gene, and stabilizing modification at codon 1314 | Pediatric | Phase I | ||
| NCT01893554 | Seropositive and seronegative children (4–59 months old) | Safe, highly infectious, and attenuated vaccine. Increase in RSV neutralizing antibodies and RSV F IgG antibody titers; no ≥4-fold increase in antibody titers in seropositive participants. A total of 2 of 15 and 4 of 20 children of the low- and high-dose group, respectively, experienced RSV-mediated medically attended acute respiratory infection (MAARIs). Anamnestic antibody response occurred [66]. | |||
| NCT03227029 (assessed also RSV 276, which resulted from a deletion of the M2-2 Protein) | Seronegative children (6–24 months old) | The acceptable safety profile for both vaccines presented, High reported ratio of cough cases with RSV/276; great infectivity for both vaccines; ≥4-fold increase in neutralizing antibody titers were measured in 60% of the RSV ΔNS2/Δ1313/I1314L recipients. Only RSV ΔNS2/Δ1313/I1314L was chosen to be further assessed [67]. | |||
| Phases I/II | |||||
| NCT03916185 (assesses also RSV 6120/ΔNS2/1030s and RSV/276) | Seronegative children (6–24 months old) | Ongoing. | |||
| RSV LID/ΔM2–2/1030s | Attenuation based on deletion of the M2-2 protein and 1030s mutation at polymerase L protein | Pediatric | Phase I | ||
| NCT02794870 and NCT02952339 | Seronegative children (6–24 months old) | No SAEs; mostly mild or moderate AEs. High infectivity rate; stable mutations; 4-fold increase in serum antibody assays in 90% of the vaccinees and ≥4-fold increase in RSV F-specific IgG antibody titers in 85% of the vaccinees. Anamnestic antibody induction [68]. | |||
| NCT04520659 (Phase Ib) | Seronegative children (6–24 months old) | Ongoing. | |||
| RSV 6120/ΔNS2/1030s | Attenuation based on the deletion of NS2 protein and 1030s modification of polymerase L protein | Pediatric | Phase I | ||
| NCT03387137 | Seropositive (12–59 months old) and seronegative (6–24 months old) children | Infectious potential and immunogenicity were proven with greater rates of rhinorrhea in participants who received the candidate vaccine [69]. | |||
| RSV 6120/F1/G2/ΔNS1 RSV 6120/ΔNS1 | Deletion of NS1 for both candidates and RSV 6120/F1/G2/ΔNS1 It is modified regarding the F gene, and F and G genes are transported in other genome positions | Pediatric | Phase I | ||
| NCT03596801 | Seropositive (12–59 months old) and seronegative (6–24 months old) children | Ongoing. | |||
| Recombinant vector-based vaccines | |||||
| MVA-BN-RSV | MVA vector that transfers genetic information for Proteins F, G (both subtypes), N and M2 | Older adult | Phase I | ||
| NCT02419391 | Adults (18–49 years old and 50–65 years old) | No safety concerns. A 2-fold rise in interferon-γ (IFN-γ) GMTs. No increase in cells producing interleukin-4 (IL-4) (cellular immunity activation, protective factor against enhanced respiratory disease (ERD)). Increase to a lesser extent, in RSV-specific antibodies; similar responses between the age groups [70]. | |||
| Phase II | |||||
| NCT02873286 | Adults (≥55 years old) | High specific-antibody GMTs and until week 30 without the effect of a second dose. A 5- to 10-fold increase in T cell immunity titers against various RSV proteins; mucosal immunity is stimulated. All the high-dose group participants experienced immunity induction to, at least, one of three proteins and 65% to all proteins, with similar safety data compared to the previous Phase I trial with no safety concerns [71]. | |||
| NCT04752644 (challenge model) | Adults (18–50 years old) | Reduction in the viral burden in vaccinees; effective in inhibiting the onset of symptoms in RSV infection; similar safety data with regard to the previous trials [72]. | |||
| Phase III | |||||
| NCT05238025 (name: VANIR) | Older adults (≥60 years old) | Ongoing | |||
| Ad26.RSV.preF | Replication of defective Adenovirus 26 as a vector modified to express stabilized pre-F from the RSV-A2 strain | Older adult—Discontinued (trials are not described) Pediatric | Phases I/II | ||
| NCT03303625 | Younger adults (18–50 years old) and seropositive children (12–24 months old) | Safe in children and younger adults (as shown in previous studies in adult participants). High titers of RSV-A2-antibody titers and preF and postF antibodies after two vaccinations that remained until 6 months; RSV-B antibodies also increased in the pediatric group (cross-neutralizing immunity) [73]. | |||
| NCT03606512 | Seronegative children (12–24 months old) | Antibody titers were higher from the baseline on day 85 and remained until the end of the first RSV season, with the occurrence of SAEs in 5% of the vaccinees. | |||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Topalidou, X.; Kalergis, A.M.; Papazisis, G. Respiratory Syncytial Virus Vaccines: A Review of the Candidates and the Approved Vaccines. Pathogens 2023, 12, 1259. https://doi.org/10.3390/pathogens12101259
Topalidou X, Kalergis AM, Papazisis G. Respiratory Syncytial Virus Vaccines: A Review of the Candidates and the Approved Vaccines. Pathogens. 2023; 12(10):1259. https://doi.org/10.3390/pathogens12101259
Chicago/Turabian StyleTopalidou, Xanthippi, Alexis M. Kalergis, and Georgios Papazisis. 2023. "Respiratory Syncytial Virus Vaccines: A Review of the Candidates and the Approved Vaccines" Pathogens 12, no. 10: 1259. https://doi.org/10.3390/pathogens12101259
APA StyleTopalidou, X., Kalergis, A. M., & Papazisis, G. (2023). Respiratory Syncytial Virus Vaccines: A Review of the Candidates and the Approved Vaccines. Pathogens, 12(10), 1259. https://doi.org/10.3390/pathogens12101259







