Perception of Ticks and Tick-Borne Diseases Worldwide
Abstract
1. Introduction
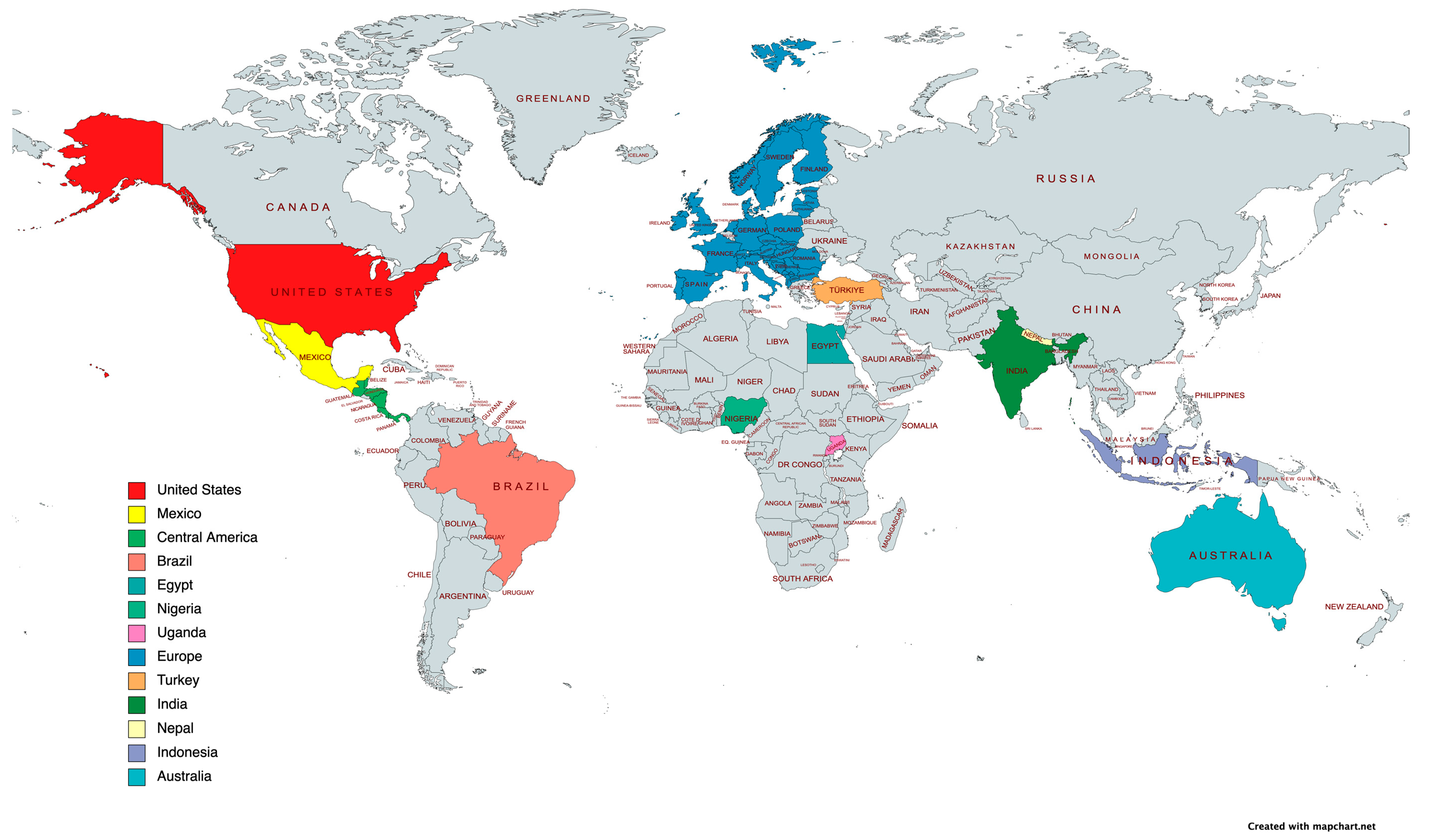
2. Contributions from Different Countries and Regions Worldwide
2.1. United States of America
2.2. Mexico
2.3. Central America
2.4. Brazil
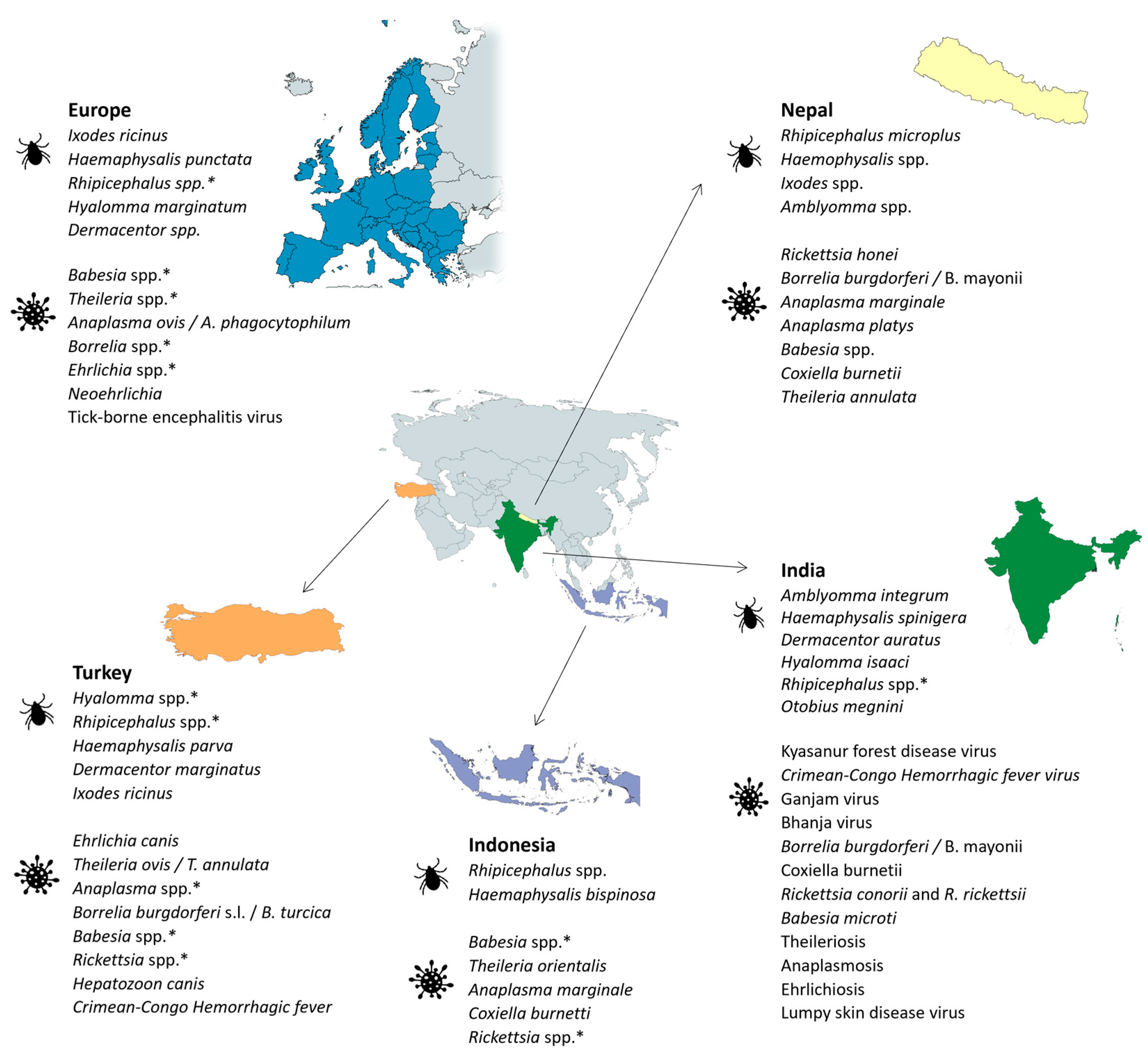
2.5. Europe
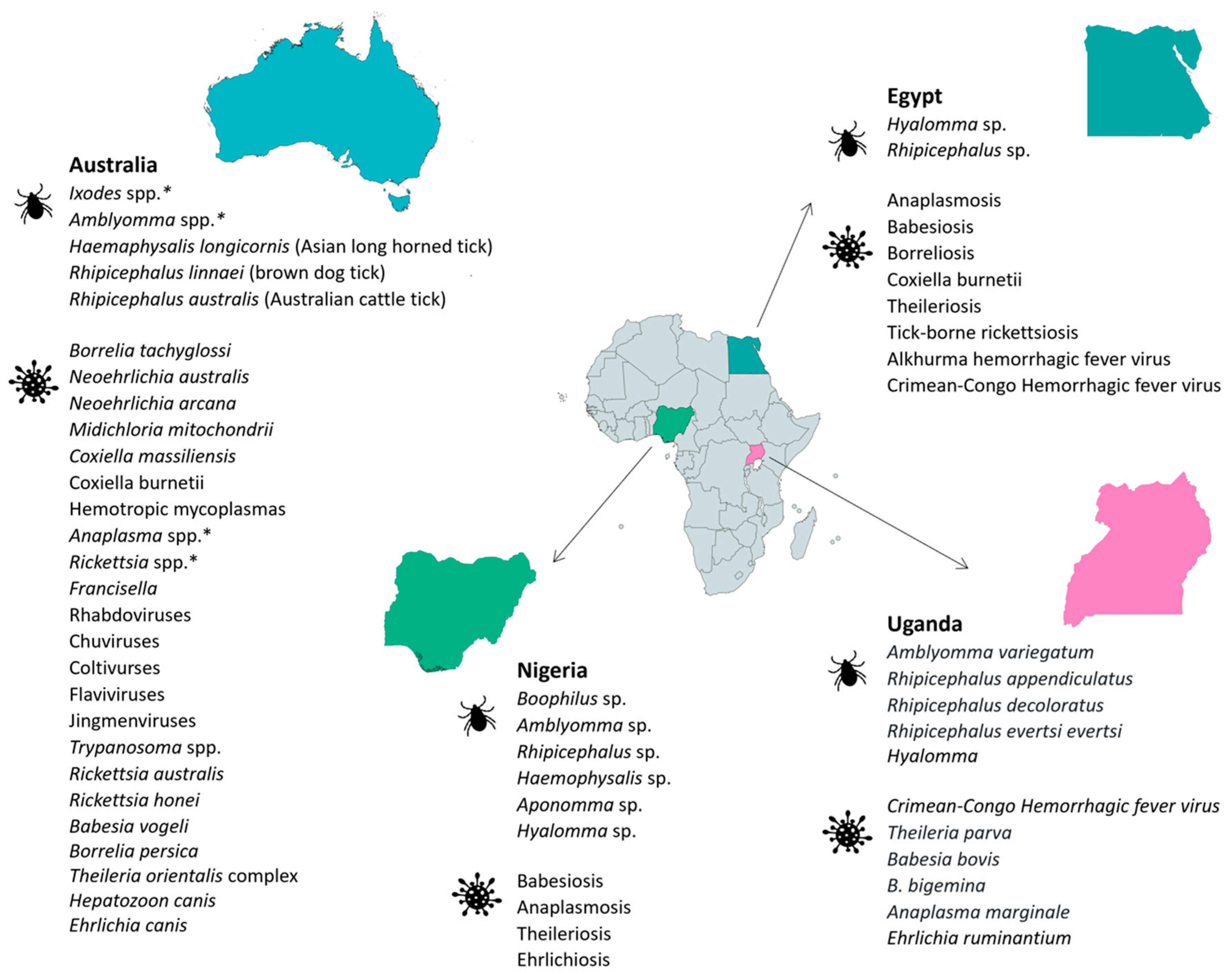
2.6. Egypt
2.7. Uganda
2.8. Nigeria
2.9. India
2.10. Nepal
2.11. Indonesia
2.12. Turkey
2.13. Australia
3. Discussion, Conclusions, and Future Directions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Behzadi, M.Y.; Mostafavi, E.; Rohani, M.; Mohamadi, A.; Ahmadinezhad, M.; Moazzezy, N.; Shams-Ghahfarokhi, M.; Razzaghi-Abyaneh, M. A Review on Important Zoonotic Bacterial Tick-Borne Diseases in the Eastern Mediterranean Region. J. Arthropod-Borne Dis. 2021, 15, 265–277. [Google Scholar] [CrossRef] [PubMed]
- Belobo, J.T.E.; Kenmoe, S.; Kengne-Nde, C.; Emoh, C.P.D.; Bowo-Ngandji, A.; Tchatchouang, S.; Sowe Wobessi, J.N.; Mbongue Mikangue, C.A.; Tazokong, H.R.; Kingue Bebey, S.R.; et al. Worldwide Epidemiology of Crimean-Congo Hemorrhagic Fever Virus in Humans, Ticks and Other Animal Species, a Systematic Review and Meta-Analysis. PLoS Negl. Trop. Dis. 2021, 15, e0009299. [Google Scholar] [CrossRef]
- Jakab, Á.; Kahlig, P.; Kuenzli, E.; Neumayr, A. Tick Borne Relapsing Fever—A Systematic Review and Analysis of the Literature. PLoS Negl. Trop. Dis. 2022, 16, e0010212. [Google Scholar] [CrossRef] [PubMed]
- Karshima, S.N.; Karshima, M.N.; Ahmed, M.I. Animal Reservoirs of Zoonotic Babesia Species: A Global Systematic Review and Meta-Analysis of Their Prevalence, Distribution and Species Diversity. Vet. Parasitol. 2021, 298, 109539. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; O’Bryan, J.; Krause, P.J. The Global Emergence of Human Babesiosis. Pathogens 2021, 10, 1447. [Google Scholar] [CrossRef]
- Tiffin, H.S.; Rajotte, E.G.; Sakamoto, J.M.; Machtinger, E.T. Tick Control in a Connected World: Challenges, Solutions, and Public Policy from a United States Border Perspective. Trop. Med. Infect. Dis. 2022, 7, 388. [Google Scholar] [CrossRef]
- Zhang, Y.-Y.; Sun, Y.-Q.; Chen, J.-J.; Teng, A.-Y.; Wang, T.; Li, H.; Hay, S.I.; Fang, L.-Q.; Yang, Y.; Liu, W. Mapping the Global Distribution of Spotted Fever Group Rickettsiae: A Systematic Review with Modelling Analysis. Lancet Digit. Health 2023, 5, e5–e15. [Google Scholar] [CrossRef]
- Hromníková, D.; Furka, D.; Furka, S.; Santana, J.A.D.; Ravingerová, T.; Klöcklerová, V.; Žitňan, D. Prevention of Tick-Borne Diseases: Challenge to Recent Medicine. Biologia 2022, 77, 1533–1554. [Google Scholar] [CrossRef]
- Monaghan, A.J.; Moore, S.M.; Sampson, K.M.; Beard, C.B.; Eisen, R.J. Climate Change Influences on the Annual Onset of Lyme Disease in the United States. Ticks Tick-Borne Dis. 2015, 6, 615–622. [Google Scholar] [CrossRef]
- Eisen, L. Tick Species Infesting Humans in the United States. Ticks Tick-Borne Dis. 2022, 13, 102025. [Google Scholar] [CrossRef]
- Eisen, L. Rodent-Targeted Approaches to Reduce Acarological Risk of Human Exposure to Pathogen-Infected Ixodes Ticks. Ticks Tick-Borne Dis. 2023, 14, 102119. [Google Scholar] [CrossRef] [PubMed]
- Hart, C.E.; Thangamani, S. Tick-Virus Interactions: Current Understanding and Future Perspectives. Parasite Immunol. 2021, 43, e12815. [Google Scholar] [CrossRef] [PubMed]
- Kong, Y.; Zhang, G.; Jiang, L.; Wang, P.; Zhang, S.; Zheng, X.; Li, Y. Metatranscriptomics Reveals the Diversity of the Tick Virome in Northwest China. Microbiol. Spectr. 2022, 10, e01115-22. [Google Scholar] [CrossRef]
- Kmetiuk, L.B.; Martins, T.F.; Bach, R.V.W.; Martins, C.M.; De Barros-Filho, I.R.; Lipinski, L.C.; Fávero, G.M.; Dos Santos, A.P.; Biondo, A.W. Risk Factors Associated with Ticks and Rickettsia spp. Exposure in Wild Boars (Sus scrofa), Hunting Dogs, and Hunters of Brazil. Vet. World 2021, 14, 2745–2749. [Google Scholar] [CrossRef] [PubMed]
- Chitimia-Dobler, L.; Dunlop, J.A.; Pfeffer, T.; Würzinger, F.; Handschuh, S.; Mans, B.J. Hard Ticks in Burmese Amber with Australasian Affinities. Parasitology 2023, 150, 157–171. [Google Scholar] [CrossRef] [PubMed]
- de la Fuente, J. The Fossil Record and the Origin of Ticks (Acari: Parasitiformes: Ixodida). Exp. Appl. Acarol. 2003, 29, 331–344. [Google Scholar] [CrossRef]
- De la Fuente, J. Fossilized Tick-Borne Diseases. Available online: https://hekint.org/2023/08/31/fossilized-tick-borne-diseases/ (accessed on 6 October 2023).
- De la Fuente, J.; Estrada-Peña, A.; Cabezas-Cruz, A.; Brey, R. Flying Ticks: Anciently Evolved Associations That Constitute a Risk of Infectious Disease Spread. Parasit. Vectors 2015, 8, 538. [Google Scholar] [CrossRef]
- Poinar, G., Jr. Spirochete-like Cells in a Dominican Amber Ambylomma Tick (Arachnida: Ixodidae). Hist. Biol. 2015, 27, 565–570. [Google Scholar] [CrossRef]
- Guglielmone, A.A.; Petney, T.N.; Robbins, R.G. Ixodidae (Acari: Ixodoidea): Descriptions and Redescriptions of All Known Species from 1758 to December 31, 2019. Zootaxa 2020, 4871, zootaxa.4871.1.1. [Google Scholar] [CrossRef]
- Eisen, L.; Eisen, R.J. Changes in the Geographic Distribution of the Blacklegged Tick, Ixodes Scapularis, in the United States. Ticks Tick-Borne Dis. 2023, 14, 102233. [Google Scholar] [CrossRef]
- Eisen, L. Control of Ixodid Ticks and Prevention of Tick-Borne Diseases in the United States: The Prospect of a New Lyme Disease Vaccine and the Continuing Problem with Tick Exposure on Residential Properties. Ticks Tick-Borne Dis. 2021, 12, 101649. [Google Scholar] [CrossRef] [PubMed]
- Paules, C.I.; Marston, H.D.; Bloom, M.E.; Fauci, A.S. Tickborne Diseases—Confronting a Growing Threat. N. Engl. J. Med. 2018, 379, 701–703. [Google Scholar] [CrossRef] [PubMed]
- Hahn, M.B.; Disler, G.; Durden, L.A.; Coburn, S.; Witmer, F.; George, W.; Beckmen, K.; Gerlach, R. Establishing a Baseline for Tick Surveillance in Alaska: Tick Collection Records from 1909–2019. Ticks Tick-Borne Dis. 2020, 11, 101495. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, A.; Drexler, N.A.; McCormick, D.W.; Thompson, J.M.; Kersh, G.; Commins, S.P.; Salzer, J.S. Health Care Provider Knowledge Regarding Alpha-Gal Syndrome—United States, March–May 2022. MMWR Morb. Mortal. Wkly. Rep. 2023, 72, 809–814. [Google Scholar] [CrossRef]
- De la Fuente, J. The Alpha-Gal Syndrome Is Underdiagnosed. Actas Dermosifiliogr. 2023. [Google Scholar] [CrossRef]
- Thompson, J.M.; Carpenter, A.; Kersh, G.J.; Wachs, T.; Commins, S.P.; Salzer, J.S. Geographic Distribution of Suspected Alpha-Gal Syndrome Cases—United States, January 2017–December 2022. MMWR Morb. Mortal. Wkly. Rep. 2023, 72, 815–820. [Google Scholar] [CrossRef]
- Nawrocki, C.C.; Hinckley, A.F. Experiences with Tick Exposure, Lyme Disease, and Use of Personal Prevention Methods for Tick Bites among Members of the U.S. Population, 2013–2015. Ticks Tick-Borne Dis. 2021, 12, 101605. [Google Scholar] [CrossRef]
- Eisen, L. Personal Protection Measures to Prevent Tick Bites in the United States: Knowledge Gaps, Challenges, and Opportunities. Ticks Tick-Borne Dis. 2022, 13, 101944. [Google Scholar] [CrossRef]
- Niesobecki, S.; Hansen, A.; Rutz, H.; Mehta, S.; Feldman, K.; Meek, J.; Niccolai, L.; Hook, S.; Hinckley, A. Knowledge, Attitudes, and Behaviors Regarding Tick-Borne Disease Prevention in Endemic Areas. Ticks Tick-Borne Dis. 2019, 10, 101264. [Google Scholar] [CrossRef]
- Hodo, C.L.; Forgacs, D.; Auckland, L.D.; Bass, K.; Lindsay, C.; Bingaman, M.; Sani, T.; Colwell, K.; Hamer, G.L.; Hamer, S.A. Presence of Diverse Rickettsia spp. and Absence of Borrelia burgdorferi Sensu Lato in Ticks in an East Texas Forest with Reduced Tick Density Associated with Controlled Burns. Ticks Tick-Borne Dis. 2020, 11, 101310. [Google Scholar] [CrossRef]
- Elias, S.P.; Rand, P.W.; Rickard, L.N.; Stone, B.B.; Maasch, K.A.; Lubelczyk, C.B.; Smith, R.P. Support for Deer Herd Reduction on Offshore Islands of Maine, U.S.A. Ticks Tick-Borne Dis. 2021, 12, 101634. [Google Scholar] [CrossRef] [PubMed]
- Omodior, O.; Eze, P.; Anderson, K.R. Using I-Tree Canopy Vegetation Cover Subtype Classification to Predict Peri-Domestic Tick Presence. Ticks Tick-Borne Dis. 2021, 12, 101684. [Google Scholar] [CrossRef] [PubMed]
- Nawrocki, C.C.; Piedmonte, N.; Niesobecki, S.A.; Rowe, A.; Hansen, A.P.; Kaufman, A.; Foster, E.; Meek, J.I.; Niccolai, L.; White, J.; et al. Acceptability of 4-Poster Deer Treatment Devices for Community-Wide Tick Control among Residents of High Lyme Disease Incidence Counties in Connecticut and New York, USA. Ticks Tick-Borne Dis. 2023, 14, 102231. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Eggers, P.; Arana, A.; Kresse, B.; Rios, K.; Brown, L.; Sampson, L.; Kploanyi, M. Knowledge and Preventive Behaviors towards Tick-Borne Diseases in Delaware. Ticks Tick-Borne Dis. 2018, 9, 615–622. [Google Scholar] [CrossRef]
- Bron, G.M.; Fernandez, M.; Larson, S.R.; Maus, A.; Gustafson, D.; Tsao, J.I.; Diuk-Wasser, M.A.; Bartholomay, L.C.; Paskewitz, S.M. Context Matters: Contrasting Behavioral and Residential Risk Factors for Lyme Disease between High-Incidence States in the Northeastern and Midwestern United States. Ticks Tick-Borne Dis. 2020, 11, 101515. [Google Scholar] [CrossRef]
- Beck, A.; Bjork, J.; Biggerstaff, B.J.; Eisen, L.; Eisen, R.; Foster, E.; Signs, K.; Tsao, J.I.; Kough, E.; Peterson, M.; et al. Knowledge, Attitudes, and Behaviors Regarding Tick-Borne Disease Prevention in Lyme Disease-Endemic Areas of the Upper Midwest, United States. Ticks Tick-Borne Dis. 2022, 13, 101925. [Google Scholar] [CrossRef]
- Cuadera, M.K.Q.; Mader, E.M.; Safi, A.G.; Harrington, L.C. Knowledge, Attitudes, and Practices for Tick Bite Prevention and Tick Control among Residents of Long Island, New York, USA. Ticks Tick-Borne Dis. 2023, 14, 102124. [Google Scholar] [CrossRef]
- CDC. Tickborne Disease Surveillance Data Summary. Available online: https://www.cdc.gov/ticks/data-summary/index.html (accessed on 30 September 2023).
- Ginsberg, H.S.; Rulison, E.L.; Miller, J.L.; Pang, G.; Arsnoe, I.M.; Hickling, G.J.; Ogden, N.H.; LeBrun, R.A.; Tsao, J.I. Local Abundance of Ixodes scapularis in Forests: Effects of Environmental Moisture, Vegetation Characteristics, and Host Abundance. Ticks Tick-Borne Dis. 2020, 11, 101271. [Google Scholar] [CrossRef]
- de la Fuente, J.; Estrada-Peña, A.; Gortázar, C.; Vaz-Rodrigues, R.; Sánchez, I.; Carrión Tudela, J. Citizen Science on Lyme Borreliosis in Spain Reveals Disease-Associated Risk Factors and Control Interventions. Vector Borne Zoonotic Dis. Larchmt. N 2023, 23, 441–446. [Google Scholar] [CrossRef]
- Foster, E.; Maes, S.A.; Holcomb, K.M.; Eisen, R.J. Prevalence of Five Human Pathogens in Host-Seeking Ixodes scapularis and Ixodes pacificus by Region, State, and County in the Contiguous United States Generated through National Tick Surveillance. Ticks Tick-Borne Dis. 2023, 14, 102250. [Google Scholar] [CrossRef]
- Nesgos, A.T.; Harrington, L.C.; Mader, E.M. Experience and Knowledge of Lyme Disease: A Scoping Review of Patient-Provider Communication. Ticks Tick-Borne Dis. 2021, 12, 101714. [Google Scholar] [CrossRef]
- Pace, E.J.; O’Reilly, M. Tickborne Diseases: Diagnosis and Management. Am. Fam. Physician 2020, 101, 530–540. [Google Scholar] [PubMed]
- Dixon, D.M.; Branda, J.A.; Clark, S.H.; Dumler, J.S.; Horowitz, H.W.; Perdue, S.S.; Pritt, B.S.; Sexton, D.J.; Storch, G.A.; Walker, D.H. Ehrlichiosis and Anaplasmosis Subcommittee Report to the Tick-Borne Disease Working Group. Ticks Tick-Borne Dis. 2021, 12, 101823. [Google Scholar] [CrossRef] [PubMed]
- Almazán, C.; Tipacamu, G.A.; Rodriguez, S.; Mosqueda, J.; de Leon, A.P. Immunological Control of Ticks and Tick-Borne Diseases That Impact Cattle Health and Production. Front. Biosci.-Landmark 2018, 23, 1535–1551. [Google Scholar] [CrossRef] [PubMed]
- Almazán, C.; Reyes de Luna, G.; Tinoco-Gracia, L.; González-Álvarez, V.H.; Zając, Z.; Kulisz, J.; Woźniak, A.; Cabezas-Cruz, A.; Mosqueda, J. Morphological and Molecular Identification of the Brown Dog Tick in Mexico. Vet. Parasitol. Reg. Stud. Rep. 2023, 44, 100908. [Google Scholar] [CrossRef]
- Beristain-Ruiz, D.M.; Garza-Hernández, J.A.; Figueroa-Millán, J.V.; Lira-Amaya, J.J.; Quezada-Casasola, A.; Ordoñez-López, S.; Laredo-Tiscareño, S.V.; Alvarado-Robles, B.; Castillo-Luna, O.R.; Floriano-López, A.; et al. Possible Association between Selected Tick-Borne Pathogen Prevalence and Rhipicephalus sanguineus Sensu Lato Infestation in Dogs from Juarez City (Chihuahua), Northwest Mexico-US Border. Pathogens 2022, 11, 552. [Google Scholar] [CrossRef]
- Zarate-Ramos, J.J.; Nevarez-Garza, A.M.; Zamora-Ávila, D.E.; Rodríguez-Tovar, L.E. Myotonia and Colic Associated with the Spinose Ear Tick, Otobius Megnini, in a Horse in Northern Mexico. Res. J. Parasitol. 2014, 9, 16–20. [Google Scholar] [CrossRef]
- Rodríguez, S.D.; García Ortiz, M.Á.; Jiménez Ocampo, R.; Vega y Murguía, C.A. Molecular Epidemiology of Bovine Anaplasmosis with a Particular Focus in Mexico. Infect. Genet. Evol. 2009, 9, 1092–1101. [Google Scholar] [CrossRef]
- Almazán, C.; Scimeca, R.C.; Reichard, M.V.; Mosqueda, J. Babesiosis and Theileriosis in North America. Pathogens 2022, 11, 168. [Google Scholar] [CrossRef]
- Lira-Amaya, J.J.; Rojas-Martínez, C.; Alvarez-Martinez, A.; Pelaez-Flores, A.; Martinez-Ibañez, F.; Perez-de la Rosa, D.; Figueroa-Millan, J. First Molecular Detection of Babesia canis vogeli in Dogs and Rhipicephalus sanguineus from Mexico. Arch. Palliat. Care 2017, 2, 1013. [Google Scholar]
- Almazán, C.; González-Álvarez, V.H.; Fernández de Mera, I.G.; Cabezas-Cruz, A.; Rodríguez-Martínez, R.; de la Fuente, J. Molecular Identification and Characterization of Anaplasma platys and Ehrlichia canis in Dogs in Mexico. Ticks Tick-Borne Dis. 2016, 7, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Hernández, G.; Candia-Plata, M.D.C.; Delgado-de la Mora, J.; Acuña-Meléndrez, N.H.; Vargas-Ortega, A.P.; Licona-Enríquez, J.D. Rocky Mountain spotted fever in Mexican children: Clinical and mortality factors. Salud Publica Mex. 2016, 58, 385–392. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Colunga-Salas, P.; Sánchez-Montes, S.; Volkow, P.; Ruíz-Remigio, A.; Becker, I. Lyme Disease and Relapsing Fever in Mexico: An Overview of Human and Wildlife Infections. PLoS ONE 2020, 15, e0238496. [Google Scholar] [CrossRef] [PubMed]
- Peniche-Lara, G.; Balmaceda, L.; Perez-Osorio, C.; Munoz-Zanzi, C. Human Babesiosis, Yucatán State, Mexico, 2015. Emerg. Infect. Dis. 2018, 24, 2061–2062. [Google Scholar] [CrossRef]
- Sosa-Gutierrez, C.G.; Cervantes-Castillo, M.A.; Laguna-Gonzalez, R.; Lopez-Echeverria, L.Y.; Ojeda-Ramírez, D.; Oyervides, M. Serological and Molecular Evidence of Patients Infected with Anaplasma phagocytophilum in Mexico. Diseases 2021, 9, 37. [Google Scholar] [CrossRef]
- López González, C.A.; Hernández-Camacho, N.; Aguilar-Tipacamú, G.; Zamora-Ledesma, S.; Olvera-Ramírez, A.M.; Jones, R.W. Gap Analysis of the Habitat Interface of Ticks and Wildlife in Mexico. Pathogens 2021, 10, 1541. [Google Scholar] [CrossRef]
- Soares, J.F.; Labruna, M.B.; de Amorim, D.B.; Baggio-Souza, V.; Fagundes-Moreira, R.; Girotto-Soares, A.; Weck, B.; Nunes, P.H.; Martins, T.F. Description of Amblyomma Monteiroae n. sp. (Acari: Ixodidae), a Parasite of the Great Horned Owl (Strigiformes: Strigidae) in Southern Brazil. Ticks Tick-Borne Dis. 2023, 14, 102239. [Google Scholar] [CrossRef]
- Pereira, M.d.C.; Labruna, M.; Szabó, M.P.J.; Klafke, G.M. Rhipicephalus (Boophilus) Microplus: Biologia, Controle e Resistência; MedVet Livros: São Paulo, Brazil, 2008; p. 169. ISBN 978-85-61461-05-8. [Google Scholar]
- Moraes-Filho, J.; Marcili, A.; Nieri-Bastos, F.A.; Richtzenhain, L.J.; Labruna, M.B. Genetic Analysis of Ticks Belonging to the Rhipicephalus sanguineus Group in Latin America. Acta Trop. 2011, 117, 51–55. [Google Scholar] [CrossRef]
- Szabó, M.P.J.; Mangold, A.J.; João, C.F.; Bechara, G.H.; Guglielmone, A.A. Biological and DNA Evidence of Two Dissimilar Populations of the Rhipicephalus sanguineus Tick Group (Acari: Ixodidae) in South America. Vet. Parasitol. 2005, 130, 131–140. [Google Scholar] [CrossRef]
- Rebanho de Bovinos (Bois e Vacas) No Brasil. Available online: https://www.ibge.gov.br/explica/producao-agropecuaria/bovinos/br (accessed on 4 September 2023).
- Grisi, L.; Leite, R.C.; Martins, J.R.d.S.; de Barros, A.T.M.; Andreotti, R.; Cançado, P.H.D.; de León, A.A.P.; Pereira, J.B.; Villela, H.S. Reassessment of the Potential Economic Impact of Cattle Parasites in Brazil. Rev. Bras. Parasitol. Veterinária 2014, 23, 150–156. [Google Scholar] [CrossRef]
- Ferreira, G.C.M.; Canozzi, M.E.A.; Peripolli, V.; Moura, G.d.P.; Sánchez, J.; Martins, C.E.N. Prevalence of Bovine Babesia spp., Anaplasma marginale, and Their Co-Infections in Latin America: Systematic Review-Meta-Analysis. Ticks Tick-Borne Dis. 2022, 13, 101967. [Google Scholar] [CrossRef] [PubMed]
- Klafke, G.; Webster, A.; Dall Agnol, B.; Pradel, E.; Silva, J.; de La Canal, L.H.; Becker, M.; Osório, M.F.; Mansson, M.; Barreto, R.; et al. Multiple Resistance to Acaricides in Field Populations of Rhipicephalus microplus from Rio Grande Do Sul State, Southern Brazil. Ticks Tick-Borne Dis. 2017, 8, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Vilela, V.L.R.; Feitosa, T.F.; Bezerra, R.A.; Klafke, G.M.; Riet-Correa, F. Multiple Acaricide-Resistant Rhipicephalus microplus in the Semi-Arid Region of Paraíba State, Brazil. Ticks Tick-Borne Dis. 2020, 11, 101413. [Google Scholar] [CrossRef] [PubMed]
- Labruna, M.B.; Kasai, N.; Ferreira, F.; Faccini, J.L.H.; Gennari, S.M. Seasonal Dynamics of Ticks (Acari: Ixodidae) on Horses in the State of São Paulo, Brazil. Vet. Parasitol. 2002, 105, 65–77. [Google Scholar] [CrossRef]
- Barros, E.M.; Braga, Í.A.; Santos, L.G.F.; Ziliani, T.F.; Melo, A.L.T.; Borges, A.M.C.M.; Silva, L.G.; Aguiar, D.M. Detecção de Theileria equi e Babesia caballi e anticorpos anti-Ehrlichia spp. em equídeos do Pantanal Mato-Grossense, Brasil. Arq. Bras. Med. Vet. Zootec. 2015, 67, 716–722. [Google Scholar] [CrossRef][Green Version]
- Peckle, M.; Pires, M.S.; dos Santos, T.M.; Roier, E.C.R.; da Silva, C.B.; Vilela, J.A.R.; Paulino, P.G.; Santos, H.A.; Massard, C.L. Molecular Investigation of Babesia caballi in Horses from the State of Rio de Janeiro, Brazil: Epidemiological Aspects Associated with the Infection. Vet. Parasitol. Reg. Stud. Rep. 2022, 30, 100709. [Google Scholar] [CrossRef]
- Kerber, C.E.; Labruna, M.B.; Ferreira, F.; De Waal, D.T.; Knowles, D.P.; Gennari, S.M. Prevalence of Equine Piroplasmosis and Its Association with Tick Infestation in the State of São Paulo, Brazil. Rev. Bras. Parasitol. Vet. 2009, 18, 1–8. [Google Scholar] [CrossRef]
- Garcia, M.V.; Andreotti, R.; Reis, F.A.; Aguirre, A.d.A.R.; Barros, J.C.; Matias, J.; Koller, W.W. Contributions of the Hair Sheep Breed Santa Ines as a Maintenance Host for Rhipicephalus (boophilus) microplus (Acari: Ixodidae) in Brazil. Parasit. Vectors 2014, 7, 515. [Google Scholar] [CrossRef]
- Šlapeta, J.; Halliday, B.; Chandra, S.; Alanazi, A.D.; Abdel-Shafy, S. Rhipicephalus linnaei (Audouin, 1826) Recognised as the “Tropical Lineage” of the Brown Dog Tick Rhipicephalus sanguineus Sensu Lato: Neotype Designation, Redescription, and Establishment of Morphological and Molecular Reference. Ticks Tick-Borne Dis. 2022, 13, 102024. [Google Scholar] [CrossRef]
- Vieira, R.F.d.C.; Biondo, A.W.; Guimarães, A.M.S.; dos Santos, A.P.; dos Santos, R.P.; Dutra, L.H.; Diniz, P.P.V.d.P.; de Morais, H.A.; Messick, J.B.; Labruna, M.B.; et al. Ehrlichiosis in Brazil. Rev. Bras. Parasitol. Vet. 2011, 20, 1–12. [Google Scholar] [CrossRef]
- de Oliveira, G.M.B.; Muñoz-Leal, S.; Santodomingo, A.; Weck, B.C.; Faccini-Martínez, Á.A.; Horta, M.C.; Labruna, M.B. A Novel Relapsing Fever Group Borrelia Isolated from Ornithodoros Ticks of the Brazilian Caatinga. Microorganisms 2023, 11, 370. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, R.C.; Moraes-Filho, J.; Guedes, E.; Silveira, I.; Richtzenhain, L.J.; Leite, R.C.; Labruna, M.B. Rickettsial Infections of Dogs, Horses and Ticks in Juiz de Fora, Southeastern Brazil, and Isolation of Rickettsia rickettsii from Rhipicephalus sanguineus Ticks. Med. Vet. Entomol. 2011, 25, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Pinter, A.; Dias, R.A.; Gennari, S.M.; Labruna, M.B. Study of the Seasonal Dynamics, Life Cycle, and Host Specificity of Amblyomma aureolatum (Acari: Ixodidae). J. Med. Entomol. 2004, 41, 324–332. [Google Scholar] [CrossRef] [PubMed]
- Reck, J.; Souza, U.; Souza, G.; Kieling, E.; Dall’Agnol, B.; Webster, A.; Michel, T.; Doyle, R.; Martins, T.F.; Labruna, M.B.; et al. Records of Ticks on Humans in Rio Grande Do Sul State, Brazil. Ticks Tick-Borne Dis. 2018, 9, 1296–1301. [Google Scholar] [CrossRef]
- Soares, J.F.; Costa, F.B.; Girotto-Soares, A.; Da Silva, A.S.; França, R.T.; Taniwaki, S.A.; Dall’Agnol, B.; Reck, J.; Hagiwara, M.K.; Labruna, M.B. Evaluation of the Vector Competence of Six Ixodid Tick Species for Rangelia vitalii (Apicomplexa, Piroplasmorida), the Agent of Canine Rangeliosis. Ticks Tick-Borne Dis. 2018, 9, 1221–1234. [Google Scholar] [CrossRef]
- Soares, J.F.; Dall’Agnol, B.; Costa, F.B.; Krawczak, F.S.; Comerlato, A.T.; Rossato, B.C.D.; Linck, C.M.; Sigahi, E.K.O.; Teixeira, R.H.F.; Sonne, L.; et al. Natural Infection of the Wild Canid, Cerdocyon Thous, with the Piroplasmid Rangelia vitalii in Brazil. Vet. Parasitol. 2014, 202, 156–163. [Google Scholar] [CrossRef]
- Paludo, G.R.; Friedmann, H.; Dell’Porto, A.; Macintire, D.K.; Whitley, E.M.; Boudreaux, M.K.; Baneth, G.; Blagburn, B.L.; Dykstra, C.C. Hepatozoon spp.: Pathological and Partial 18S rRNA Sequence Analysis from Three Brazilian Dogs. Parasitol. Res. 2005, 97, 167–170. [Google Scholar] [CrossRef]
- André, M.R.; Calchi, A.C.; Perles, L.; Gonçalves, L.R.; Uccella, L.; Lemes, J.R.B.; Nantes, W.A.G.; Santos, F.M.; Porfírio, G.E.d.O.; Barros-Battesti, D.M.; et al. Novel Ehrlichia and Hepatozoon Genotypes in White-Eared Opossums (Didelphis albiventris) and Associated Ticks from Brazil. Ticks Tick-Borne Dis. 2022, 13, 102022. [Google Scholar] [CrossRef]
- Weck, B.C.; Serpa, M.C.A.; Ramos, V.N.; Luz, H.R.; Costa, F.B.; Ramirez, D.G.; Benatti, H.R.; Piovezan, U.; Szabó, M.P.J.; Marcili, A.; et al. Novel Genotypes of Hepatozoon spp. in Small Mammals, Brazil. Parasit. Vectors 2022, 15, 87. [Google Scholar] [CrossRef]
- Faccini-Martínez, Á.A.; Krawczak, F.d.S.; de Oliveira, S.V.; Labruna, M.B.; Angerami, R.N. Rickettsioses in Brazil: Distinct Diseases and New Paradigms for Epidemiological Surveillance. Rev. Soc. Bras. Med. Trop. 2021, 54, e07322020. [Google Scholar] [CrossRef]
- Weck, B.; Krawczak, F.S.; Costa, F.B.; Dall’Agnol, B.; Marcili, A.; Reck, J.; Labruna, M.B. Rickettsia parkeri in the Pampa Biome of Southern Brazil: Isolation, Molecular Characterization, and Serological Evidence of Canine Infection. Vet. Parasitol. Reg. Stud. Rep. 2020, 22, 100448. [Google Scholar] [CrossRef] [PubMed]
- Guglielmone, A.A.; Nava, S.; Robbins, R.G. Neotropical Hard Ticks (Acari: Ixodida: Ixodidae): A Critical Analysis of Their Taxonomy, Distribution, and Host Relationships; Springer International Publishing: Cham, Switzerland, 2021; ISBN 978-3-030-72352-1. [Google Scholar]
- Binder, L.C.; Ramírez-Hernández, A.; Serpa, M.C.d.A.; Moraes-Filho, J.; Pinter, A.; Scinachi, C.A.; Labruna, M.B. Domestic Dogs as Amplifying Hosts of Rickettsia rickettsii for Amblyomma aureolatum Ticks. Ticks Tick-Borne Dis. 2021, 12, 101824. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.C.; de Castro, M.B.; Labruna, M.B.; Szabó, M.P.J. The Inoculation Eschar of Rickettsia parkeri Rickettsiosis in Brazil: Importance and Cautions. Ticks Tick-Borne Dis. 2023, 14, 102127. [Google Scholar] [CrossRef] [PubMed]
- Ogrzewalska, M.; Saraiva, D.G.; Moraes-Filho, J.; Martins, T.F.; Costa, F.B.; Pinter, A.; Labruna, M.B. Epidemiology of Brazilian Spotted Fever in the Atlantic Forest, State of São Paulo, Brazil. Parasitology 2012, 139, 1283–1300. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, S.V.; Guimarães, J.N.; Reckziegel, G.C.; Neves, B.M.d.C.; de Araújo-Vilges, K.M.; Fonseca, L.X.; Pinna, F.V.; Pereira, S.V.C.; de Caldas, E.P.; Gazeta, G.S.; et al. An Update on the Epidemiological Situation of Spotted Fever in Brazil. J. Venom. Anim. Toxins Trop. Dis. 2016, 22, 22. [Google Scholar] [CrossRef]
- Szabó, M.; Pinter, A.; Labruna, M. Ecology, Biology and Distribution of Spotted-Fever Tick Vectors in Brazil. Front. Cell. Infect. Microbiol. 2013, 3, 27. [Google Scholar] [CrossRef]
- Labruna, M.B. Ecology of Rickettsia in South America. Ann. N. Y. Acad. Sci. 2009, 1166, 156–166. [Google Scholar] [CrossRef]
- Luz, H.R.; Costa, F.B.; Benatti, H.R.; Ramos, V.N.; De, A.; Serpa, M.C.; Martins, T.F.; Acosta, I.C.L.; Ramirez, D.G.; Muñoz-Leal, S.; et al. Epidemiology of Capybara-Associated Brazilian Spotted Fever. PLoS Negl. Trop. Dis. 2019, 13, e0007734. [Google Scholar] [CrossRef]
- de Carvalho Nunes, E.; de Moura-Martiniano, N.O.; de Lima Duré, A.Í.; de Melo Iani, F.C.; de Oliveira, S.V.; de Mello, F.L.; Gazêta, G.S. Spotted Fever in the Morphoclimatic Domains of Minas Gerais State, Brazil. Front. Trop. Dis. 2022, 2, 718047. [Google Scholar] [CrossRef]
- Yoshinari, N.H.; Bonoldi, V.L.N.; Bonin, S.; Falkingham, E.; Trevisan, G. The Current State of Knowledge on Baggio–Yoshinari Syndrome (Brazilian Lyme Disease-like Illness): Chronological Presentation of Historical and Scientific Events Observed over the Last 30 Years. Pathogens 2022, 11, 889. [Google Scholar] [CrossRef]
- de Oliveira, S.V.; Faccini-Martínez, Á.A.; Cerutti Junior, C. Lack of Serological Evidence for Lyme-like Borreliosis in Brazil. Travel Med. Infect. Dis. 2018, 26, 62–63. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, D.D.; Carreira, T.; Nunes, M.; Benitez, A.; Lopes-Mori, F.M.R.; Vidotto, O.; de Freitas, J.C.; Vieira, M.L. First Record of Borrelia Burgdorferi B31 Strain in Dermacentor Nitens Ticks in the Northern Region of Parana (Brazil). Braz. J. Microbiol. Publ. Braz. Soc. Microbiol. 2013, 44, 883–887. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schotthoefer, A.M.; Frost, H.M. Ecology and Epidemiology of Lyme Borreliosis. Clin. Lab. Med. 2015, 35, 723–743. [Google Scholar] [CrossRef] [PubMed]
- Onofrio, V.C.; Guglielmone, A.A.; Barros-Battesti, D.M.; Gianizella, S.L.; Marcili, A.; Quadros, R.M.; Marques, S.; Labruna, M.B. Description of a New Species of Ixodes (Acari: Ixodidae) and First Report of Ixodes lasallei and Ixodes bocatorensis in Brazil. Ticks Tick-Borne Dis. 2020, 11, 101423. [Google Scholar] [CrossRef]
- Faccini-Martínez, Á.A.; Silva-Ramos, C.R.; Santodomingo, A.M.; Ramírez-Hernández, A.; Costa, F.B.; Labruna, M.B.; Muñoz-Leal, S. Historical Overview and Update on Relapsing Fever Group Borrelia in Latin America. Parasit. Vectors 2022, 15, 196. [Google Scholar] [CrossRef]
- Muñoz-Leal, S.; Ramirez, D.G.; Luz, H.R.; Faccini, J.L.H.; Labruna, M.B. “Candidatus Borrelia Ibitipoquensis”, a Borrelia valaisiana–Related Genospecies Characterized from Ixodes Paranaensis in Brazil. Microb. Ecol. 2020, 80, 682–689. [Google Scholar] [CrossRef]
- dos Santos, C.A.; Suzin, A.; Vogliotti, A.; Nunes, P.H.; Barbieri, A.R.M.; Labruna, M.B.; Szabó, M.P.J.; Yokosawa, J. Molecular Detection of a Borrelia sp. in Nymphs of Amblyomma brasiliense Ticks (Acari: Ixodidae) from Iguaçu National Park, Brazil, Genetically Related to Borrelia from Ethiopia and Côte d’Ivoire. Ticks Tick-Borne Dis. 2020, 11, 101519. [Google Scholar] [CrossRef]
- De Figueiredo, G.G.; Amarilla, A.A.; de Souza, W.M.; Fumagalli, M.J.; de Figueiredo, M.L.G.; Szabó, M.P.J.; Badra, S.J.; Setoh, Y.X.; Khromykh, A.A.; Aquino, V.H.; et al. Genetic Characterization of Cacipacoré Virus from Ticks Collected in São Paulo State, Brazil. Arch. Virol. 2017, 162, 1783–1786. [Google Scholar] [CrossRef]
- Pascoal, J.d.O.; de Siqueira, S.M.; Maia, R.d.C.; Juan Szabó, M.P.; Yokosawa, J. Detection and Molecular Characterization of Mogiana Tick Virus (MGTV) in Rhipicephalus microplus Collected from Cattle in a Savannah Area, Uberlândia, Brazil. Ticks Tick-Borne Dis. 2019, 10, 162–165. [Google Scholar] [CrossRef]
- Fletcher, I.K.; Gibb, R.; Lowe, R.; Jones, K.E. Differing Taxonomic Responses of Mosquito Vectors to Anthropogenic Land-Use Change in Latin America and the Caribbean. PLoS Negl. Trop. Dis. 2023, 17, e0011450. [Google Scholar] [CrossRef]
- Rochlin, I.; Toledo, A. Emerging Tick-Borne Pathogens of Public Health Importance: A Mini-Review. J. Med. Microbiol. 2020, 69, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Guglielmone, A.A.; Nava, S.; Robbins, R.G. Geographic Distribution of the Hard Ticks (Acari: Ixodida: Ixodidae) of the World by Countries and Territories. Zootaxa 2023, 5251, 1–274. [Google Scholar] [CrossRef] [PubMed]
- Rubel, F.; Brugger, K.; Monazahian, M.; Habedank, B.; Dautel, H.; Leverenz, S.; Kahl, O. The First German Map of Georeferenced Ixodid Tick Locations. Parasit. Vectors 2014, 7, 477. [Google Scholar] [CrossRef]
- Rubel, F.; Zaenker, S.; Weigand, A.; Weber, D.; Chitimia-Dobler, L.; Kahl, O. Atlas of Ticks (Acari: Argasidae, Ixodidae) in Germany: 1st Data Update. Exp. Appl. Acarol. 2023, 89, 251–274. [Google Scholar] [CrossRef] [PubMed]
- Medlock, J.M.; Hansford, K.M.; Bormane, A.; Derdakova, M.; Estrada-Peña, A.; George, J.-C.; Golovljova, I.; Jaenson, T.G.T.; Jensen, J.-K.; Jensen, P.M.; et al. Driving Forces for Changes in Geographical Distribution of Ixodes Ricinus Ticks in Europe. Parasit. Vectors 2013, 6, 1. [Google Scholar] [CrossRef]
- Estrada-Peña, A. The Climate Niche of the Invasive Tick Species Hyalomma marginatum and Hyalomma rufipes (Ixodidae) with Recommendations for Modeling Exercises. Exp. Appl. Acarol. 2023, 89, 231–250. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Venzal, J.M. Climate Niches of Tick Species in the Mediterranean Region: Modeling of Occurrence Data, Distributional Constraints, and Impact of Climate Change. J. Med. Entomol. 2007, 44, 1130–1138. [Google Scholar] [CrossRef]
- Földvári, G.; Szabó, É.; Tóth, G.E.; Lanszki, Z.; Zana, B.; Varga, Z.; Kemenesi, G. Emergence of Hyalomma marginatum and Hyalomma rufipes Adults Revealed by Citizen Science Tick Monitoring in Hungary. Transbound. Emerg. Dis. 2022, 69, e2240–e2248. [Google Scholar] [CrossRef]
- Vikse, R.; Paulsen, K.M.; Edgar, K.S.; Pettersson, J.H.-O.; Ottesen, P.S.; Okbaldet, Y.B.; Kiran, N.; Lamsal, A.; Lindstedt, H.E.H.; Pedersen, B.N.; et al. Geographical Distribution and Prevalence of Tick-Borne Encephalitis Virus in Questing Ixodes ricinus Ticks and Phylogeographic Structure of the Ixodes ricinus Vector in Norway. Zoonoses Public Health 2020, 67, 370–381. [Google Scholar] [CrossRef]
- Kjellander, P.; Bergvall, U.A.; Chirico, J.; Ullman, K.; Christensson, M.; Lindgren, P.-E. Winter Activity of Ixodes Ricinus in Sweden. Parasit. Vectors 2023, 16, 229. [Google Scholar] [CrossRef]
- Uusitalo, R.; Siljander, M.; Linden, A.; Sormunen, J.J.; Aalto, J.; Hendrickx, G.; Kallio, E.; Vajda, A.; Gregow, H.; Henttonen, H.; et al. Predicting Habitat Suitability for Ixodes ricinus and Ixodes persulcatus Ticks in Finland. Parasit. Vectors 2022, 15, 310. [Google Scholar] [CrossRef]
- Estrada-Peña, A.; Fernández-Ruiz, N. Is Composition of Vertebrates an Indicator of the Prevalence of Tick-Borne Pathogens? Infect. Ecol. Epidemiol. 2022, 12, 2025647. [Google Scholar] [CrossRef] [PubMed]
- Eisen, L.; Eisen, R.J. Benefits and Drawbacks of Citizen Science to Complement Traditional Data Gathering Approaches for Medically Important Hard Ticks (Acari: Ixodidae) in the United States. J. Med. Entomol. 2021, 58, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Abdelbaset, A.E.; Nonaka, N.; Nakao, R. Tick-Borne Diseases in Egypt: A One Health Perspective. One Health 2022, 15, 100443. [Google Scholar] [CrossRef] [PubMed]
- Abdelbaset, A.E.; Kwak, M.L.; Nonaka, N.; Nakao, R. Human-Biting Ticks and Zoonotic Tick-Borne Pathogens in North Africa: Diversity, Distribution, and Trans-Mediterranean Public Health Challenges. One Health 2023, 16, 100547. [Google Scholar] [CrossRef] [PubMed]
- Aboelhadid, S.M.; Arafa, W.M.; Mahrous, L.N.; Fahmy, M.M.; Kamel, A.A. Molecular Detection of Rhipicephalus (boophilus) annulatus Resistance against Deltamethrin in Middle Egypt. Vet. Parasitol. Reg. Stud. Rep. 2018, 13, 198–204. [Google Scholar] [CrossRef]
- El-Ashram, S.; Aboelhadid, S.M.; Kamel, A.A.; Mahrous, L.N.; Fahmy, M.M. First Report of Cattle Tick Rhipicephalus (Boophilus) Annulatus in Egypt Resistant to Ivermectin. Insects 2019, 10, 404. [Google Scholar] [CrossRef]
- Vudriko, P.; Okwee-Acai, J.; Byaruhanga, J.; Tayebwa, D.S.; Okech, S.G.; Tweyongyere, R.; Wampande, E.M.; Okurut, A.R.A.; Mugabi, K.; Muhindo, J.B.; et al. Chemical Tick Control Practices in Southwestern and Northwestern Uganda. Ticks Tick-Borne Dis. 2018, 9, 945–955. [Google Scholar] [CrossRef]
- Kasaija, P.D.; Estrada-Peña, A.; Contreras, M.; Kirunda, H.; de la Fuente, J. Cattle Ticks and Tick-Borne Diseases: A Review of Uganda’s Situation. Ticks Tick-Borne Dis. 2021, 12, 101756. [Google Scholar] [CrossRef]
- Dipeolu, O.O. The Incidence of Ticks of Boophilus Species on Cattle, Sheep and Goats in Nigeria. Trop. Anim. Health Prod. 1975, 7, 35–39. [Google Scholar] [CrossRef]
- Bayer, W.; Maina, J.A. Seasonal Pattern of Tick Load in Bunaji Cattle in the Subhumid Zone of Nigeria. Vet. Parasitol. 1984, 15, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Dipeolu, O.O.; Akinboade, O.A. Studies on Ticks of Veterinary Importance in Nigeria XI. Observations on the Biology of Ticks Detached from the Red-Flanked Duiker (Cephamophys rufulatus) and Parasites Encountered in Their Blood. Vet. Parasitol. 1984, 14, 87–93. [Google Scholar] [CrossRef] [PubMed]
- Akinboade, O.A. Studies on the Bionomics and Biophysiological Constituents of Haemaphysalis leachi leachi (Dog Tick) in Nigeria. Anim. Technol. 1986, 37, 207–209. [Google Scholar]
- Akande, F.; Adebowale, A.; Takeet, M.; Omisile, O. Prevalence of Babesia Species in Hunting Dogs in Ogun State South West Nigeria. Alex. J. Vet. Sci. 2017, 54, 1. [Google Scholar] [CrossRef]
- Akande, F.A.; Oyewusi, I.K.; Ajisafe, M.G.; Idowu, O.A.; Anifowose, I.O. Survey of Cattle Tick Infestation on Farm Herds in Ogun State, Nigeria. Niger. J. Anim. Prod. 2017, 44, 23–30. [Google Scholar] [CrossRef]
- Akande, F.A.; Adebowale, A.F.; Idowu, O.A.; Sofela, O.O. Prevalence of Ticks on Indigenous Breed of Hunting Dogs in Ogun State, Nigeria. Sokoto J. Vet. Sci. 2018, 16, 66–71. [Google Scholar] [CrossRef]
- Obadiah, H.I.; Onah, I.E.; Ugochukwu, J.U.; Gbinde, A.K. Tick Infestation of Cattle in Three Markets in Makurdi, North-Central, Nigeria. Am. J. Entomol. 2017, 1, 6–10. [Google Scholar] [CrossRef]
- Famuyide, I.M.; Takeet, M.I.; Talabi, A.O.; Otesile, E.B. Molecular Detection and Identification of Piroplasms in Semi-Intensively Managed Cattle from Abeokuta, Nigeria. Folia Vet. 2020, 64, 1–8. [Google Scholar] [CrossRef]
- Mamman, A.H.; Lorusso, V.; Adam, B.M.; Dogo, G.A.; Bown, K.J.; Birtles, R.J. First Report of Theileria annulata in Nigeria: Findings from Cattle Ticks in Zamfara and Sokoto States. Parasit. Vectors 2021, 14, 242. [Google Scholar] [CrossRef]
- Daodu, O.B.; Eisenbarth, A.; Schulz, A.; Hartlaub, J.; Olopade, J.O.; Oluwayelu, D.O.; Groschup, M.H. Molecular Detection of Dugbe Orthonairovirus in Cattle and Their Infesting Ticks (Amblyomma and Rhipicephalus (boophilus)) in Nigeria. PLoS Negl. Trop. Dis. 2021, 15, e0009905. [Google Scholar] [CrossRef]
- Madder, M.; Day, M.; Schunack, B.; Fourie, J.; Labuschange, M.; van der Westhuizen, W.; Johnson, S.; Githigia, S.M.; Akande, F.A.; Nzalawahe, J.S.; et al. A Community Approach for Pathogens and Their Arthropod Vectors (Ticks and Fleas) in Cats of Sub-Saharan Africa. Parasit. Vectors 2022, 15, 321. [Google Scholar] [CrossRef] [PubMed]
- Heylen, D.; Day, M.; Schunack, B.; Fourie, J.; Labuschange, M.; Johnson, S.; Githigia, S.M.; Akande, F.A.; Nzalawahe, J.S.; Tayebwa, D.S.; et al. A Community Approach of Pathogens and Their Arthropod Vectors (Ticks and Fleas) in Dogs of African Sub-Sahara. Parasit. Vectors 2021, 14, 576. [Google Scholar] [CrossRef] [PubMed]
- Opara, M.N.; Ezeh, N.O. Ixodid Ticks of Cattle in Borno and Yobe States of Northeastern Nigeria: Breed and Coat Colour Preference. Anim. Res. Int. 2011, 8, 1359–1365. [Google Scholar] [CrossRef]
- Obadiah, H.I.; Shekaro, A. Survey of Tick Infestation in Cattle in Zaria Abattoir, Nigeria. Niger. J. Vet. Adv. 2012, 2, 81–87. [Google Scholar]
- Lorusso, V.; Picozzi, K.; de Bronsvoort, B.M.; Majekodunmi, A.; Dongkum, C.; Balak, G.; Igweh, A.; Welburn, S.C. Ixodid Ticks of Traditionally Managed Cattle in Central Nigeria: Where Rhipicephalus (boophilus) microplus Does Not Dare (Yet?). Parasit. Vectors 2013, 6, 171. [Google Scholar] [CrossRef]
- Musa, H.I.; Jajere, S.M.; Adamu, N.B.; Atsanda, N.N.; Lawal, J.R.; Adamu, S.G.; Lawal, E.K. Prevalence of Tick Infestation in Different Breeds of Cattle in Maiduguri, Northeastern Nigeria. Bangladesh J. Vet. Med. 2014, 12, 161–166. [Google Scholar] [CrossRef]
- Oyewusi, I.K.; Ganiyu, I.A.; Akande, F.A.; Takeet, M.I.; Anifowoshe, I.O.; Famuyide, I.M.; Sogebi, E.a.O.; Adeleke, G.A.; Olugbogi, E.I.; Talabi, A.O. Assessment of Ticks on Cattle Entering Nigeria through a Major Trans-Boundary Animal Route in Ogun State. Bull. Anim. Health Prod. Afr. 2015, 63, 369–377. [Google Scholar] [CrossRef]
- Kamani, J.; Apanaskevich, D.A.; Gutiérrez, R.; Nachum-Biala, Y.; Baneth, G.; Harrus, S. Morphological and Molecular Identification of Rhipicephalus (boophilus) microplus in Nigeria, West Africa: A Threat to Livestock Health. Exp. Appl. Acarol. 2017, 73, 283–296. [Google Scholar] [CrossRef]
- Adedayo, A.; Olukunle, F. First Evidence of an Established Rhipicephalus (boophilus) microplus Population in Nigeria. Alex. J. Vet. Sci. 2018, 56, 182. [Google Scholar] [CrossRef]
- Ikpeze, O.; Eneanya, C.; Onyinye, C.; Aribodor, D.N.; Anyasodor, A. Species Diversity, Distribution and Predilection Sites of Ticks (Acarina: Ixodidae on Trade Cattle at Enugu and Anambra States, Southeastern, Nigeria. Zoologist 2011, 9, 1–8. [Google Scholar] [CrossRef]
- Ameen, S.A.; Odetokun, I.A.; Ghali-Muhammed, L.I.; Azeez, O.M.; Raji, L.O.; Kolapo, T.U. Status of Ticks Infestation in Ruminant Animals in Ogbomoso Area of Oyo State, Nigeria. J. Environ. Issues Agric. Dev. Ctries. 2014, 6, 48–53. [Google Scholar]
- Agboola, B.; Otite, J.; Ayodele, I.; Omonona, A. Tick Infestation of Pangolin (Phataginus Tricuspis) in Omo Forest Reserve Ogun State Nigeria. Int. J. Biodivers. Endanger. Species 2022, 2, 109. [Google Scholar] [CrossRef]
- Akande, F.; Adenubi, O.; Garba, A. In Vitro Analysis of the Efficacy of Selected Commercial Acaricides on The Cattle Tick Rhipicephalus (boophilus) annulatus (Acari: Ixodidae). Egypt. J. Vet. Sci. 2020, 51, 153–161. [Google Scholar] [CrossRef]
- Bishop, R.P.; Githaka, N.W.; Bazarusanga, T.; Bhushan, C.; Biguezoton, A.; Vudriko, P.; Muhanguzi, D.; Tumwebaze, M.; Bosco, T.J.; Shacklock, C.; et al. Control of Ticks and Tick-Borne Diseases in Africa through Improved Diagnosis and Utilisation of Data on Acaricide Resistance. Parasit. Vectors 2023, 16, 224. [Google Scholar] [CrossRef]
- Ghosh, S.; Azhahianambi, P.; Yadav, M.P. Upcoming and Future Strategies of Tick Control: A Review. J. Vector Borne Dis. 2007, 44, 79–89. [Google Scholar]
- Ranganathan, K.; Renu, G.; Ayyanar, E.; Veeramanoharan, R.; Paulraj, P.S. Species Composition of Hard Ticks (Acari: Ixodidae) on Domestic Animals and Their Public Health Importance in Tamil Nadu, South India. Acarol. Stud. 2021, 3, 16–21. [Google Scholar] [CrossRef]
- Palavesam, A.; Ramakrishnan, R.N.; Harikrishnan, T.J.; Sagar, S.V.; Rajeswaran, A.; Raj, G.D.; Latha, B.R. A Field Study on the Relationship between Tick Burden and Deltamethrin Resistance in Rhipicephalus microplus Isolates of Indian Household Cattle. Syst. Appl. Acarol. 2021, 26, 289–303. [Google Scholar] [CrossRef]
- Soundararajan, C.; Nagarajan, K.; Arul Prakash, M. Tick Infestation in Human Beings in the Nilgiris and Kancheepuram District of Tamil Nadu, India. J. Parasit. Dis. 2018, 42, 50–54. [Google Scholar] [CrossRef]
- Gudkar, A.I.; Koka, K.; Palavesam, A. A Ticking “Time-Bomb”. TNOA J. Ophthalmic Sci. Res. 2020, 58, 130. [Google Scholar] [CrossRef]
- Parthasarathi, B.C.; Kumar, B.; Nagar, G.; Manjunathachar, H.V.; de la Fuente, J.; Ghosh, S. Analysis of Genetic Diversity in Indian Isolates of Rhipicephalus microplus Based on Bm86 Gene Sequence. Vaccines 2021, 9, 194. [Google Scholar] [CrossRef]
- Ghosh, S.; Kumar, B.; Tripathi, A.K.; Jha, A.; Singh, S.K.; Sharma, A.; Singh, R.K.; Mitra, A. Tick Management with One Health Approach. Indian J. Comp. Microb. Infect. Dis. 2023, 43, 16–25. [Google Scholar] [CrossRef]
- Fular, A.; Sharma, A.K.; Kumar, S.; Nagar, G.; Chigure, G.; Ray, D.D.; Ghosh, S. Establishment of a Multi-Acaricide Resistant Reference Tick Strain (IVRI-V) of Rhipicephalus microplus. Ticks Tick-Borne Dis. 2018, 9, 1184–1191. [Google Scholar] [CrossRef] [PubMed]
- Negi, T.; Kandari, L.S.; Arunachalam, K. Update on Prevalence and Distribution Pattern of Tick-Borne Diseases among Humans in India: A Review. Parasitol. Res. 2021, 120, 1523–1539. [Google Scholar] [CrossRef]
- Bohara, T.; Shrestha, S. A Study on Cattle Tick and Tick Borne Pathogens of Midwestern Nepal. Nepal. Vet. J. 2016, 33, 23–27. [Google Scholar]
- Dhital, B.; Shrestha, S.; Kaphle, K.; Pudasaini, R. Distribution of the Cattle Ticks from Mid Hills to Plains of Nepal. J. Agric. Nat. Resour. 2018, 1, 197–205. [Google Scholar] [CrossRef]
- Kunwar, A.; Shakya, S.R.; Ghimire, T.R. Diversity and Prevalence of Ticks in the Goats in Lowland Nepal. Ann. Parasitol. 2022, 68, 287–296. [Google Scholar] [CrossRef]
- Pun, S.K.; Guglielmone, A.A.; Tarragona, E.L.; Nava, S.; Maharjan, M. Ticks (Acari: Ixodidae) of Nepal: First Record of Amblyomma varanense (Supino), with an Update of Species List. Ticks Tick-Borne Dis. 2018, 9, 526–534. [Google Scholar] [CrossRef]
- Panth, Y.; Shrestha, S.; Bastola, R. Demonstration of Circulating Antibodies of Coxiella burnetii in Dairy Cattle of Rupandehi District, Nepal. Int. J. Innov. Res. Multidiscip. Field 2017, 3, 46–49. [Google Scholar]
- Gupta, V.; Gupta, R.; Shrestha, S.P. Infectivity of Theileria annulata in Hyalomma Ticks of Eastern Terai Districts, Nepal. Nepal. J. Zool. 2013, 1, 15–23. [Google Scholar]
- Pandey, G.; Acharya, M.P.; Rana, H.B.; Sadaula, A.; Pandeya, Y.R.; Pathak, C.R.; Pathak, L.R. Molecular Detection of Babesia spp. Infectivity in Ticks from Cattle of Terai, Nepal. In Proceedings of the 11th National Workshop on Livestock and Fisheries Research in Nepal, Lalitpur, Nepal, 16–17 June 2019. [Google Scholar]
- Murphy, H.; Renvoise, A.; Pandey, P.; Parola, P.; Raoult, D. Rickettsia honei Infection in Human, Nepal, 2009. Emerg. Infect. Dis. 2011, 17, 1865–1867. [Google Scholar] [CrossRef] [PubMed]
- Pun, S.B.; Agrawal, S.; Jha, S.; Bhandari, L.N.; Chalise, B.S.; Mishra, A.; Shah, R. First Report of Lyme Disease in Nepal. JMM Case Rep. 2018, 5, e005128. [Google Scholar] [CrossRef]
- Khatri, P.; Sah, C.M.; Piryani, R.M.; Chaudhary, S.; Dhakal, P.R.; Shahi, A.; Karki, M.; Shah, P.; Sapkota, S.; Neupane, A. Lyme Disease, An Emerging Infection in Nepal: A Case Report. J. Univers. Coll. Med. Sci. 2020, 8, 90–92. [Google Scholar] [CrossRef]
- Díaz-Regañón, D.; Agulla, B.; Piya, B.; Fernández-Ruiz, N.; Villaescusa, A.; García-Sancho, M.; Rodríguez-Franco, F.; Sainz, Á. Stray Dogs in Nepal Have High Prevalence of Vector-Borne Pathogens: A Molecular Survey. Parasit. Vectors 2020, 13, 174. [Google Scholar] [CrossRef] [PubMed]
- Zannou, O.M.; Ouedraogo, A.S.; Biguezoton, A.S.; Abatih, E.; Coral-Almeida, M.; Farougou, S.; Yao, K.P.; Lempereur, L.; Saegerman, C. Models for Studying the Distribution of Ticks and Tick-Borne Diseases in Animals: A Systematic Review and a Meta-Analysis with a Focus on Africa. Pathogens 2021, 10, 893. [Google Scholar] [CrossRef] [PubMed]
- Anastos, G. The Scutate Ticks, or Ixodidae, of Indonesia; Business Press Incorporated: Des Moines, IA, USA, 1950. [Google Scholar]
- Hadi, U.K.; Soviana, S.; Pratomo, I.R.C. Prevalence of Ticks and Tick-Borne Diseases in Indonesian Dogs. J. Vet. Sci. Technol. 2016, 7, 330. [Google Scholar] [CrossRef]
- Sahara, A.; Nugraheni, Y.R.; Patra, G.; Prastowo, J.; Priyowidodo, D. Ticks (Acari: Ixodidae) Infestation on Cattle in Various Regions in Indonesia. Vet. World 2019, 12, 1755–1759. [Google Scholar] [CrossRef]
- Hamid, P.H.; Cahyadi, M.; Wardhana, A.H.; Sawitri, D.H.; Setya, N.N.R.; Insyariati, T.; Kurnianto, H.; Hermosilla, C.R. First Autochthonous Report on Cattle Babesia naoakii in Central Java, Indonesia, and Identification of Haemaphysalis bispinosa Ticks in the Investigated Area. Pathogens 2022, 12, 59. [Google Scholar] [CrossRef]
- Payne, R.C.; Ward, D.E.; Usman, M.; Rusli, A.; Djauhari, D.; Husein, A. Prevalence of Bovine Haemoparasites in Aceh Province of Northern Sumatra: Implications for Imported Cattle. Prev. Vet. Med. 1988, 6, 275–283. [Google Scholar] [CrossRef]
- Guswanto, A.; Allamanda, P.; Mariamah, E.S.; Sodirun, S.; Wibowo, P.E.; Indrayani, L.; Nugroho, R.H.; Wirata, I.K.; Jannah, N.; Dias, L.P.; et al. Molecular and Serological Detection of Bovine Babesiosis in Indonesia. Parasit. Vectors 2017, 10, 550. [Google Scholar] [CrossRef]
- Aziz, N.; Maksudi, M.; Prakoso, Y.A. Correlation between Hematological Profile and Theileriosis in Bali Cattle from Muara Bulian, Jambi, Indonesia. Vet. World 2019, 12, 1358–1361. [Google Scholar] [CrossRef]
- Nugroho, E.P.; Setiyono, A.; Hadi, U.K.; Winarsih, W.; Astutid, D. Detection of Coxiella burnetii (Query Fever) DNA by Nested-PCR in Beef Cattle from Ampel Slaughterhouse, Boyolali Regency, Middle Java, Indonesia. Worlds Vet. J. 2021, 11, 267–272. [Google Scholar] [CrossRef]
- Rini, E.P.; Sasaki, M.; Astuti, D.; Juniantito, V.; Wibawan, I.W.T.; Sawa, H.; Setiyono, A. First Molecular Detection of Coxiella Burnetii in Beef Cattle in West Java, Indonesia. Jpn. J. Infect. Dis. 2022, 75, 83–85. [Google Scholar] [CrossRef] [PubMed]
- Nowak, M. Parasitisation and Localisation of Ticks (Acari: Ixodida) on Exotic Reptiles Imported into Poland. Ann. Agric. Environ. Med. AAEM 2010, 17, 237–242. [Google Scholar]
- Supriyono; Takano, A.; Kuwata, R.; Shimoda, H.; Hadi, U.K.; Setiyono, A.; Agungpriyono, S.; Maeda, K. Detection and Isolation of Tick-Borne Bacteria (Anaplasma spp., Rickettsia spp., and Borrelia spp.) in Amblyomma varanense Ticks on Lizard (Varanus salvator). Microbiol. Immunol. 2019, 63, 328–333. [Google Scholar] [CrossRef]
- Murray-Dickson, G.; Ghazali, M.; Ogden, R.; Brown, R.; Auliya, M. Phylogeography of the Reticulated Python (Malayopython reticulatus ssp.): Conservation Implications for the Worlds’ Most Traded Snake Species. PLoS ONE 2017, 12, e0182049. [Google Scholar] [CrossRef]
- Ibrahim, I.N.; Okabayashi, T.; Ristiyanto; Lestari, E.W.; Yanase, T.; Muramatsu, Y.; Ueno, H.; Morita, C. Serosurvey of Wild Rodents for Rickettsioses (Spotted Fever, Murine Typhus and Q Fever) in Java Island, Indonesia. Eur. J. Epidemiol. 1999, 15, 89–93. [Google Scholar] [CrossRef]
- Widjaja, S.; Williams, M.; Winoto, I.; Farzeli, A.; Stoops, C.A.; Barbara, K.A.; Richards, A.L.; Blair, P.J. Geographical Assessment of Rickettsioses in Indonesia. Vector-Borne Zoonotic Dis. 2016, 16, 20–25. [Google Scholar] [CrossRef]
- Faizal, M.D.; Haryanto, A.; Tjahajati, I. Diagnosis and Molecular Characterization of Anaplasma Platys in Dog Patients in Yogyakarta Area, Indonesia. Indones. J. Biotechnol. 2019, 24, 43–50. [Google Scholar] [CrossRef]
- Nguyen, V.-L.; Colella, V.; Greco, G.; Fang, F.; Nurcahyo, W.; Hadi, U.K.; Venturina, V.; Tong, K.B.Y.; Tsai, Y.-L.; Taweethavonsawat, P.; et al. Molecular Detection of Pathogens in Ticks and Fleas Collected from Companion Dogs and Cats in East and Southeast Asia. Parasit. Vectors 2020, 13, 420. [Google Scholar] [CrossRef]
- Lokida, D.; Hadi, U.; Lau, C.-Y.; Kosasih, H.; Liang, C.J.; Rusli, M.; Sudarmono, P.; Lukman, N.; Laras, K.; Asdie, R.H.; et al. Underdiagnoses of Rickettsia in Patients Hospitalized with Acute Fever in Indonesia: Observational Study Results. BMC Infect. Dis. 2020, 20, 364. [Google Scholar] [CrossRef]
- Gasem, M.H.; Kosasih, H.; Tjitra, E.; Alisjahbana, B.; Karyana, M.; Lokida, D.; Neal, A.; Liang, C.J.; Aman, A.T.; Arif, M.; et al. An Observational Prospective Cohort Study of the Epidemiology of Hospitalized Patients with Acute Febrile Illness in Indonesia. PLoS Negl. Trop. Dis. 2020, 14, e0007927. [Google Scholar] [CrossRef] [PubMed]
- Leblebicioglu, H.; Eroglu, C.; Erciyas-Yavuz, K.; Hokelek, M.; Acici, M.; Yilmaz, H. Role of Migratory Birds in Spreading Crimean-Congo Hemorrhagic Fever, Turkey. Emerg. Infect. Dis. 2014, 20, 1331–1334. [Google Scholar] [CrossRef] [PubMed]
- Keskin, A.; Erciyas-Yavuz, K. Ticks (Acari: Ixodidae) Parasitizing Passerine Birds in Turkey with New Records and New Tick–Host Associations. J. Med. Entomol. 2018, 56, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Bursali, A.; Tekin, S.; Keskin, A.; Ekici, M.; Dundar, E. Species Diversity of Ixodid Ticks Feeding on Humans in Amasya, Turkey: Seasonal Abundance and Presence of Crimean-Congo Hemorrhagic Fever Virus. J. Med. Entomol. 2011, 48, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Dervis, E.; Akın, A.; Ergonul, O.; Gargili, A. Preferences of Different Tick Species for Human Hosts in Turkey. Exp. Appl. Acarol. 2013, 61, 349–355. [Google Scholar] [CrossRef]
- Aktas, M. A Survey of Ixodid Tick Species and Molecular Identification of Tick-Borne Pathogens. Vet. Parasitol. 2014, 200, 276–283. [Google Scholar] [CrossRef]
- Koc, S.; Aydın, L.; Cetin, H. Tick Species (Acari: Ixodida) in Antalya City, Turkey: Species Diversity and Seasonal Activity. Parasitol. Res. 2015, 114, 2581–2586. [Google Scholar] [CrossRef]
- Hekimoglu, O.; Ozer, A.N. Distribution and Phylogeny of Hyalomma Ticks (Acari: Ixodidae) in Turkey. Exp. Appl. Acarol. 2017, 73, 501–519. [Google Scholar] [CrossRef]
- Orkun, Ö. Molecular Characterization Based on 16S rDNA Phylogeny of Some Ixodid Ticks in Turkey. Turk. Parazitol. Derg. 2018, 42, 121–129. [Google Scholar] [CrossRef]
- Aydin, L.; Girişgin, O.; Özüiçli, M.; Girisgin, A.; Coşkunserçe, G. Potential Risk in Public Parks: Investigation of the Tick Species (Acari: Ixodida) in Bursa Metropolitan Area, Turkey. Ank. Üniversitesi Vet. Fakültesi Derg. 2020, 67, 393–397. [Google Scholar] [CrossRef]
- Hekimoglu, O.; Sahin, M.K.; Ergan, G.; Ozer, N. A Molecular Phylogenetic Investigation of Tick Species in Eastern and Southeastern Anatolia. Ticks Tick-Borne Dis. 2021, 12, 101777. [Google Scholar] [CrossRef] [PubMed]
- Güner, E.S.; Hashimoto, N.; Takada, N.; Kaneda, K.; Imai, Y.; Masuzawa, T. First Isolation and Characterization of Borrelia Burgdorferi Sensu Lato Strains from Ixodes Ricinus Ticks in Turkey. J. Med. Microbiol. 2003, 52, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Inci, A.; Yildirim, A.; Duzlu, O.; Doganay, M.; Aksoy, S. Tick-Borne Diseases in Turkey: A Review Based on One Health Perspective. PLoS Negl. Trop. Dis. 2016, 10, e0005021. [Google Scholar] [CrossRef] [PubMed]
- Brinkmann, A.; Hekimoğlu, O.; Dinçer, E.; Hagedorn, P.; Nitsche, A.; Ergünay, K. A Cross-Sectional Screening by next-Generation Sequencing Reveals Rickettsia, Coxiella, Francisella, Borrelia, Babesia, Theileria and Hemolivia Species in Ticks from Anatolia. Parasit. Vectors 2019, 12, 26. [Google Scholar] [CrossRef]
- Ozubek, S.; Bastos, R.G.; Alzan, H.F.; Inci, A.; Aktas, M.; Suarez, C.E. Bovine Babesiosis in Turkey: Impact, Current Gaps, and Opportunities for Intervention. Pathogens 2020, 9, 1041. [Google Scholar] [CrossRef]
- Ceylan, O.; Xuan, X.; Sevinc, F. Primary Tick-Borne Protozoan and Rickettsial Infections of Animals in Turkey. Pathogens 2021, 10, 231. [Google Scholar] [CrossRef]
- Ahrabi, S.Z.; Akyildiz, G.; Kar, S.; Keles, A.G. Detection of the Crimean-Congo Hemorrhagic Fever Virus Genome in Questing Ixodes spp. and Haemaphysalis spp. in the Periurban Forestry Areas of Istanbul: Has a New Biorisk Emerged? Vector Borne Zoonotic Dis. Larchmt. N 2023, 23, 528–536. [Google Scholar] [CrossRef]
- Kirman, R.; Guven, E. Molecular Detection of Babesia and Theileria Species/Genotypes in Sheep and Ixodid Ticks in Erzurum, Northeastern Turkey: First Report of Babesia canis in Sheep. Res. Vet. Sci. 2023, 157, 40–49. [Google Scholar] [CrossRef]
- Brinkmann, A.; Dinçer, E.; Polat, C.; Hekimoğlu, O.; Hacıoğlu, S.; Földes, K.; Özkul, A.; Öktem, İ.M.A.; Nitsche, A.; Ergünay, K. A Metagenomic Survey Identifies Tamdy Orthonairovirus as Well as Divergent Phlebo-, Rhabdo-, Chu- and Flavi-like Viruses in Anatolia, Turkey. Ticks Tick-Borne Dis. 2018, 9, 1173–1183. [Google Scholar] [CrossRef]
- Dinçer, E.; Hacıoğlu, S.; Kar, S.; Emanet, N.; Brinkmann, A.; Nitsche, A.; Özkul, A.; Linton, Y.-M.; Ergünay, K. Survey and Characterization of Jingmen Tick Virus Variants. Viruses 2019, 11, 1071. [Google Scholar] [CrossRef]
- Dinçer, E.; Timurkan, M.Ö.; Oğuz, B.; Şahindokuyucu, İ.; Şahan, A.; Ekinci, M.; Polat, C.; Ergünay, K. Several Tick-Borne Pathogenic Viruses in Circulation in Anatolia, Turkey. Vector Borne Zoonotic Dis. Larchmt. N 2022, 22, 148–158. [Google Scholar] [CrossRef]
- Ergünay, K.; Polat, C.; Özkul, A. Vector-Borne Viruses in Turkey: A Systematic Review and Bibliography. Antivir. Res. 2020, 183, 104934. [Google Scholar] [CrossRef]
- Aktas, M.; Vatansever, Z.; Altay, K.; Aydin, M.F.; Dumanli, N. Molecular Evidence for Anaplasma Phagocytophilum in Ixodes Ricinus from Turkey. Trans. R. Soc. Trop. Med. Hyg. 2010, 104, 10–15. [Google Scholar] [CrossRef]
- Gargili, A.; Midilli, K.; Ergonul, O.; Ergin, S.; Alp, H.G.; Vatansever, Z.; Iyisan, S.; Cerit, C.; Yilmaz, G.; Altas, K.; et al. Crimean-Congo Hemorrhagic Fever in European Part of Turkey: Genetic Analysis of the Virus Strains from Ticks and a Seroepidemiological Study in Humans. Vector-Borne Zoonotic Dis. 2011, 11, 747–752. [Google Scholar] [CrossRef]
- Kar, S.; Yılmazer, N.; Akyıldız, G.; Gargılı, A. The Human Infesting Ticks in the City of Istanbul and Its Vicinity with Reference to a New Species for Turkey. Syst. Appl. Acarol. 2017, 22, 2245–2255. [Google Scholar] [CrossRef]
- Hekimoğlu, O. Phylogenetic Placement of Turkish Populations of Ixodes ricinus and Ixodes inopinatus. Exp. Appl. Acarol. 2022, 88, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Albayrak, H.; Ozan, E.; Kurt, M. Molecular Detection of Crimean-Congo Haemorrhagic Fever Virus (CCHFV) but Not West Nile Virus (WNV) in Hard Ticks from Provinces in Northern Turkey. Zoonoses Public Health 2010, 57, e156–e160. [Google Scholar] [CrossRef] [PubMed]
- Hekimoglu, O.; Ozer, N.; Ergunay, K.; Ozkul, A. Species Distribution and Detection of Crimean Congo Hemorrhagic Fever Virus (CCHFV) in Field-Collected Ticks in Ankara Province, Central Anatolia, Turkey. Exp. Appl. Acarol. 2012, 56, 75–84. [Google Scholar] [CrossRef]
- Ozdarendeli, A.; Aydin, K.; Tonbak, S.; Aktas, M.; Altay, K.; Koksal, I.; Bolat, Y.; Dumanli, N.; Kalkan, A. Genetic Analysis of the M RNA Segment of Crimean-Congo Hemorrhagic Fever Virus Strains in Turkey. Arch. Virol. 2008, 153, 37–44. [Google Scholar] [CrossRef]
- Tekin, S.; Bursali, A.; Mutluay, N.; Keskin, A.; Dundar, E. Crimean-Congo Hemorrhagic Fever Virus in Various Ixodid Tick Species from a Highly Endemic Area. Vet. Parasitol. 2012, 186, 546–552. [Google Scholar] [CrossRef]
- Tonbak, S.; Aktas, M.; Altay, K.; Azkur, A.K.; Kalkan, A.; Bolat, Y.; Dumanli, N.; Ozdarendeli, A. Crimean-Congo Hemorrhagic Fever Virus: Genetic Analysis and Tick Survey in Turkey. J. Clin. Microbiol. 2006, 44, 4120–4124. [Google Scholar] [CrossRef] [PubMed]
- Dinçer, E.; Brinkmann, A.; Hekimoğlu, O.; Hacıoğlu, S.; Földes, K.; Karapınar, Z.; Polat, P.F.; Oğuz, B.; Orunç Kılınç, Ö.; Hagedorn, P.; et al. Generic Amplification and next Generation Sequencing Reveal Crimean-Congo Hemorrhagic Fever Virus AP92-like Strain and Distinct Tick Phleboviruses in Anatolia, Turkey. Parasit. Vectors 2017, 10, 335. [Google Scholar] [CrossRef] [PubMed]
- Ergünay, K.; Dinçer, E.; Kar, S.; Emanet, N.; Yalçınkaya, D.; Polat Dinçer, P.F.; Brinkmann, A.; Hacıoğlu, S.; Nitsche, A.; Özkul, A.; et al. Multiple Orthonairoviruses Including Crimean-Congo Hemorrhagic Fever Virus, Tamdy Virus and the Novel Meram Virus in Anatolia. Ticks Tick-Borne Dis. 2020, 11, 101448. [Google Scholar] [CrossRef] [PubMed]
- İça, A.; Çetīn, H. Molecular investigation of Crimean-Congo Hemorrhagic Fever virus in ticks. Ank. Üniversitesi Vet. Fakültesi Derg. 2016, 63, 251–257. [Google Scholar]
- Kar, S.; Rodriguez, S.E.; Akyildiz, G.; Cajimat, M.N.B.; Bircan, R.; Mears, M.C.; Bente, D.A.; Keles, A.G. Crimean-Congo Hemorrhagic Fever Virus in Tortoises and Hyalomma aegyptium Ticks in East Thrace, Turkey: Potential of a Cryptic Transmission Cycle. Parasit. Vectors 2020, 13, 201. [Google Scholar] [CrossRef]
- Yesilbag, K.; Aydin, L.; Dincer, E.; Alpay, G.; Girisgin, A.O.; Tuncer, P.; Ozkul, A. Tick Survey and Detection of Crimean-Congo Hemorrhagic Fever Virus in Tick Species from a Non-Endemic Area, South Marmara Region, Turkey. Exp. Appl. Acarol. 2013, 60, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Hekimoglu, O.; Elverici, C.; Kuyucu, A.C. Predicting Climate-Driven Distribution Shifts in Hyalomma marginatum (Ixodidae). Parasitology 2023, 150, 883–893. [Google Scholar] [CrossRef] [PubMed]
- Barker, S.C.; Barker, D. Ticks of Australasia: 125 Species of Ticks in and around Australia. Zootaxa 2023, 5253, 1–670. [Google Scholar] [CrossRef]
- Mans, B.J. Paradigms in Tick Evolution. Trends Parasitol. 2023, 39, 475–486. [Google Scholar] [CrossRef]
- Oskam, C.; Ronai, I.; Irwin, P. The Emergence of Tick-Borne Diseases in Domestic Animals in Australia. Clim. Ticks Dis. 2021, 424–429. [Google Scholar] [CrossRef]
- Graves, S.R.; Stenos, J. Tick-Borne Infectious Diseases in Australia. Med. J. Aust. 2017, 206, 320–324. [Google Scholar] [CrossRef] [PubMed]
- Egan, S.L.; Loh, S.-M.; Banks, P.B.; Gillett, A.; Ahlstrom, L.; Ryan, U.M.; Irwin, P.J.; Oskam, C.L. Bacterial Community Profiling Highlights Complex Diversity and Novel Organisms in Wildlife Ticks. Ticks Tick-Borne Dis. 2020, 11, 101407. [Google Scholar] [CrossRef] [PubMed]
- Mackerras, M.J. The Haematozoa of Australian Mammals. Aust. J. Zool. 1959, 7, 105–135. [Google Scholar] [CrossRef]
- Austen, J.M.; Ryan, U.M.; Friend, J.A.; Ditcham, W.G.F.; Reid, S.A. Vector of Trypanosoma copemani Identified as Ixodes sp. Parasitology 2011, 138, 866–872. [Google Scholar] [CrossRef]
- Barbosa, A.D.; Gofton, A.W.; Paparini, A.; Codello, A.; Greay, T.; Gillett, A.; Warren, K.; Irwin, P.; Ryan, U. Increased Genetic Diversity and Prevalence of Co-Infection with Trypanosoma spp. in Koalas (Phascolarctos cinereus) and Their Ticks Identified Using next-Generation Sequencing (NGS). PLoS ONE 2017, 12, e0181279. [Google Scholar] [CrossRef]
- Gofton, A.W.; Doggett, S.; Ratchford, A.; Ryan, U.; Irwin, P. Phylogenetic Characterisation of Two Novel Anaplasmataceae from Australian Ixodes holocyclus Ticks: ‘Candidatus Neoehrlichia Australis’ and ‘Candidatus Neoehrlichia Arcana’. Int. J. Syst. Evol. Microbiol. 2016, 66, 4256–4261. [Google Scholar] [CrossRef] [PubMed]
- Gofton, A.W.; Loh, S.-M.; Barbosa, A.D.; Paparini, A.; Gillett, A.; Macgregor, J.; Oskam, C.L.; Ryan, U.M.; Irwin, P.J. A Novel Ehrlichia Species in Blood and Ixodes ornithorhynchi Ticks from Platypuses (Ornithorhynchus anatinus) in Queensland and Tasmania, Australia. Ticks Tick-Borne Dis. 2018, 9, 435–442. [Google Scholar] [CrossRef]
- Greay, T.L.; Zahedi, A.; Krige, A.-S.; Owens, J.M.; Rees, R.L.; Ryan, U.M.; Oskam, C.L.; Irwin, P.J. Endemic, Exotic and Novel Apicomplexan Parasites Detected during a National Study of Ticks from Companion Animals in Australia. Parasit. Vectors 2018, 11, 197. [Google Scholar] [CrossRef]
- Loh, S.-M.; Paparini, A.; Ryan, U.; Irwin, P.; Oskam, C. Identification of Theileria fuliginosa-like Species in Ixodes australiensis Ticks from Western Grey Kangaroos (Macropus fuliginosus) in Western Australia. Ticks Tick-Borne Dis. 2018, 9, 632–637. [Google Scholar] [CrossRef]
- Oskam, C.; Owens, J.; Codello, A.; Gofton, A.; Greay, T.; Oskam, C.; Owens, J.; Codello, A.; Gofton, A.; Greay, T. Rethinking Coxiella Infections in Australia. Microbiol. Aust. 2018, 39, 223–225. [Google Scholar] [CrossRef]
- Harvey, E.; Rose, K.; Eden, J.-S.; Lo, N.; Abeyasuriya, T.; Shi, M.; Doggett, S.L.; Holmes, E.C. Extensive Diversity of RNA Viruses in Australian Ticks. J. Virol. 2019, 93, e01358-18. [Google Scholar] [CrossRef] [PubMed]
- Egan, S.L.; Ruiz-Aravena, M.; Austen, J.M.; Barton, X.; Comte, S.; Hamilton, D.G.; Hamede, R.K.; Ryan, U.M.; Irwin, P.J.; Jones, M.E.; et al. Blood Parasites in Endangered Wildlife-Trypanosomes Discovered During a Survey of Haemoprotozoa from the Tasmanian Devil. Pathogens 2020, 9, 873. [Google Scholar] [CrossRef] [PubMed]
- O’Brien, C.A.; Huang, B.; Warrilow, D.; Hazlewood, J.E.; Bielefeldt-Ohmann, H.; Hall-Mendelin, S.; Pegg, C.L.; Harrison, J.J.; Paramitha, D.; Newton, N.D.; et al. Extended Characterisation of Five Archival Tick-Borne Viruses Provides Insights for Virus Discovery in Australian Ticks. Parasit. Vectors 2022, 15, 59. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, A.D.; Long, M.; Lee, W.; Austen, J.M.; Cunneen, M.; Ratchford, A.; Burns, B.; Kumarasinghe, P.; Ben-Othman, R.; Kollmann, T.R.; et al. The Troublesome Ticks Research Protocol: Developing a Comprehensive, Multidiscipline Research Plan for Investigating Human Tick-Associated Disease in Australia. Pathogens 2022, 11, 1290. [Google Scholar] [CrossRef] [PubMed]
- Chalada, M.J.; Stenos, J.; Bradbury, R.S. Is There a Lyme-like Disease in Australia? Summary of the Findings to Date. One Health 2016, 2, 42–54. [Google Scholar] [CrossRef]
- Collignon, P.J.; Lum, G.D.; Robson, J.M. Does Lyme Disease Exist in Australia? Med. J. Aust. 2016, 205, 413–417. [Google Scholar] [CrossRef]
- Overseas-Acquired Tick-Borne Diseases: Lyme Disease. Available online: https://www.health.gov.au/resources/publications/overseas-acquired-tick-borne-diseases-lyme-disease-0?language=en (accessed on 10 September 2023).
- Lane, J.; Jubb, T.; Shephard, R.; Webb-Ware, J.; Fordyce, G. Priority List of Endemic Diseases for the Red Meat Industries; Project Report; Meat & Livestock Australia Limited: North Sydney, NSW, Australia, 2015. Available online: https://era.daf.qld.gov.au/id/eprint/5030/1/B.AHE.0010_Final_Report_Priority%20list%20of%20endemic%20diseases%20for%20the%20red%20meat%20industries.pdf (accessed on 6 October 2023).
- Lydecker, H.; Stanfield, E.; Lo, N.; Hochuli, D.; Banks, P. Are Urban Bandicoots Solely to Blame for Tick Concerns? Aust. Zool. 2015, 37, 288–293. [Google Scholar] [CrossRef]
- Taylor, C.L.; Lydecker, H.W.; Hochuli, D.F.; Banks, P.B. Associations between Wildlife Observations, Human-Tick Encounters and Landscape Features in a Peri-Urban Tick Hotspot. Urban Ecosyst. 2023, 26, 1439–1454. [Google Scholar] [CrossRef]
- Taylor, C.L.; Egan, S.L.; Gofton, A.W.; Irwin, P.J.; Oskam, C.L.; Hochuli, D.F.; Banks, P.B. An Invasive Human Commensal and a Native Marsupial Maintain Tick Populations at the Urban Fringe. Med. Vet. Entomol. 2023, 37, 460–471. [Google Scholar] [CrossRef]
- Steele, S.G.; Booy, R.; Manocha, R.; Mor, S.M.; Toribio, J.-A.L.M.L. Towards One Health Clinical Management of Zoonoses: A Parallel Survey of Australian General Medical Practitioners and Veterinarians. Zoonoses Public Health 2021, 68, 88–102. [Google Scholar] [CrossRef]
- Barker, D.; Barker, S.C. Survey of Cases of Tick-Paralysis and the Presence of the Eastern Paralysis Tick, Ixodes holocyclus, and the Southern Paralysis Tick, Ixodes cornuatus, in the Greater Melbourne Area. Aust. Vet. J. 2020, 98, 2–10. [Google Scholar] [CrossRef] [PubMed]
- Teo, E.J.M.; Hailu, S.; Kelava, S.; Zalucki, M.P.; Furlong, M.J.; Nakao, R.; Barker, D.; Barker, S.C. Climatic Requirements of the Southern Paralysis Tick, Ixodes cornuatus, with a Consideration of Its Host, Vombatus ursinus, and the Possible Geographic Range of the Tick up to 2090. Ticks Tick-Borne Dis. 2021, 12, 101758. [Google Scholar] [CrossRef] [PubMed]
- Irwin, P.; Beadle, J. The ‘Other’ Epidemic: Canine Ehrlichiosis in Australia. Microbiol. Aust. 2022, 43, 156–159. [Google Scholar] [CrossRef]
- Lee, W.; Barbosa, A.D.; Irwin, P.J.; Currie, A.; Kollmann, T.R.; Beaman, M.; Lee, A.H.; Oskam, C.L. A Systems Biology Approach to Better Understand Human Tick-Borne Diseases. Trends Parasitol. 2023, 39, 53–69. [Google Scholar] [CrossRef]
- Slunge, D.; Jore, S.; Krogfelt, K.A.; Jepsen, M.T.; Boman, A. Who Is Afraid of Ticks and Tick-Borne Diseases? Results from a Cross-Sectional Survey in Scandinavia. BMC Public Health 2019, 19, 1666. [Google Scholar] [CrossRef]
- Wisely, S.M.; Glass, G.E. Advancing the Science of Tick and Tick-Borne Disease Surveillance in the United States. Insects 2019, 10, 361. [Google Scholar] [CrossRef]
- De la Fuente, J.; Contreras, M. Quantum Vaccinomics Platforms to Advance in Vaccinology. Front. Immunol. 2023, 14, 1172734. [Google Scholar] [CrossRef]
- Garcia-Vozmediano, A.; De Meneghi, D.; Sprong, H.; Portillo, A.; Oteo, J.A.; Tomassone, L. A One Health Evaluation of the Surveillance Systems on Tick-Borne Diseases in the Netherlands, Spain and Italy. Vet. Sci. 2022, 9, 504. [Google Scholar] [CrossRef]
- Mader, E.M.; Ganser, C.; Geiger, A.; Harrington, L.C.; Foley, J.; Smith, R.L.; Mateus-Pinilla, N.; Teel, P.D.; Eisen, R.J. A Survey of Tick Surveillance and Control Practices in the United States. J. Med. Entomol. 2021, 58, 1503–1512. [Google Scholar] [CrossRef]
- Obaid, M.K.; Islam, N.; Alouffi, A.; Khan, A.Z.; da Silva Vaz, I.; Tanaka, T.; Ali, A. Acaricides Resistance in Ticks: Selection, Diagnosis, Mechanisms, and Mitigation. Front. Cell. Infect. Microbiol. 2022, 12, 941831. [Google Scholar] [CrossRef]
- Communication Toolkit on Tick-Borne Diseases and Preventive Measures. Available online: https://www.ecdc.europa.eu/en/publications-data/communication-toolkit-tick-borne-diseases-and-preventive-measures (accessed on 30 September 2023).

| Terms of Search | Number of Publications (%) |
|---|---|
| Tick AND Tick-borne disease | 21,302; corrected as 21,715 |
| Tick AND Tick-borne disease AND Europe | 5194 (23.9%) |
| Tick AND Tick-borne disease AND USA | 4394 (20.2%) |
| Tick AND Tick-borne disease AND Asia | 2950 (13.6%) |
| Tick AND Tick-borne disease AND North America | 2935 (13.5%) |
| Tick AND Tick-borne disease AND Africa | 1967 (9.1%) |
| Tick AND Tick-borne disease AND China | 1119 (5.2%) |
| Tick AND Tick-borne disease AND Russia | 902 (4.2%) |
| Tick AND Tick-borne disease AND South America | 717 (3.3%) |
| Tick AND Tick-borne disease AND Brazil | 703 (3.2%) |
| Tick AND Tick-borne disease AND Australia | 377 (1.7%) |
| Tick AND Tick-borne disease AND Mexico | 267 (1.2%) |
| Tick AND Tick-borne disease AND Central America | 190 (0.9%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de la Fuente, J.; Estrada-Peña, A.; Rafael, M.; Almazán, C.; Bermúdez, S.; Abdelbaset, A.E.; Kasaija, P.D.; Kabi, F.; Akande, F.A.; Ajagbe, D.O.; et al. Perception of Ticks and Tick-Borne Diseases Worldwide. Pathogens 2023, 12, 1258. https://doi.org/10.3390/pathogens12101258
de la Fuente J, Estrada-Peña A, Rafael M, Almazán C, Bermúdez S, Abdelbaset AE, Kasaija PD, Kabi F, Akande FA, Ajagbe DO, et al. Perception of Ticks and Tick-Borne Diseases Worldwide. Pathogens. 2023; 12(10):1258. https://doi.org/10.3390/pathogens12101258
Chicago/Turabian Stylede la Fuente, José, Agustín Estrada-Peña, Marta Rafael, Consuelo Almazán, Sergio Bermúdez, Abdelbaset E. Abdelbaset, Paul D. Kasaija, Fredrick Kabi, Foluke Adedayo Akande, Dorcas Oluwakemi Ajagbe, and et al. 2023. "Perception of Ticks and Tick-Borne Diseases Worldwide" Pathogens 12, no. 10: 1258. https://doi.org/10.3390/pathogens12101258
APA Stylede la Fuente, J., Estrada-Peña, A., Rafael, M., Almazán, C., Bermúdez, S., Abdelbaset, A. E., Kasaija, P. D., Kabi, F., Akande, F. A., Ajagbe, D. O., Bamgbose, T., Ghosh, S., Palavesam, A., Hamid, P. H., Oskam, C. L., Egan, S. L., Duarte-Barbosa, A., Hekimoğlu, O., Szabó, M. P. J., ... Dahal, A. (2023). Perception of Ticks and Tick-Borne Diseases Worldwide. Pathogens, 12(10), 1258. https://doi.org/10.3390/pathogens12101258