Progress of the COVID-19: Persistence, Effectiveness, and Immune Escape of the Neutralizing Antibody in Convalescent Serum
Abstract
1. Introduction
2. Materials and Methods
2.1. Ethics Approval Statement
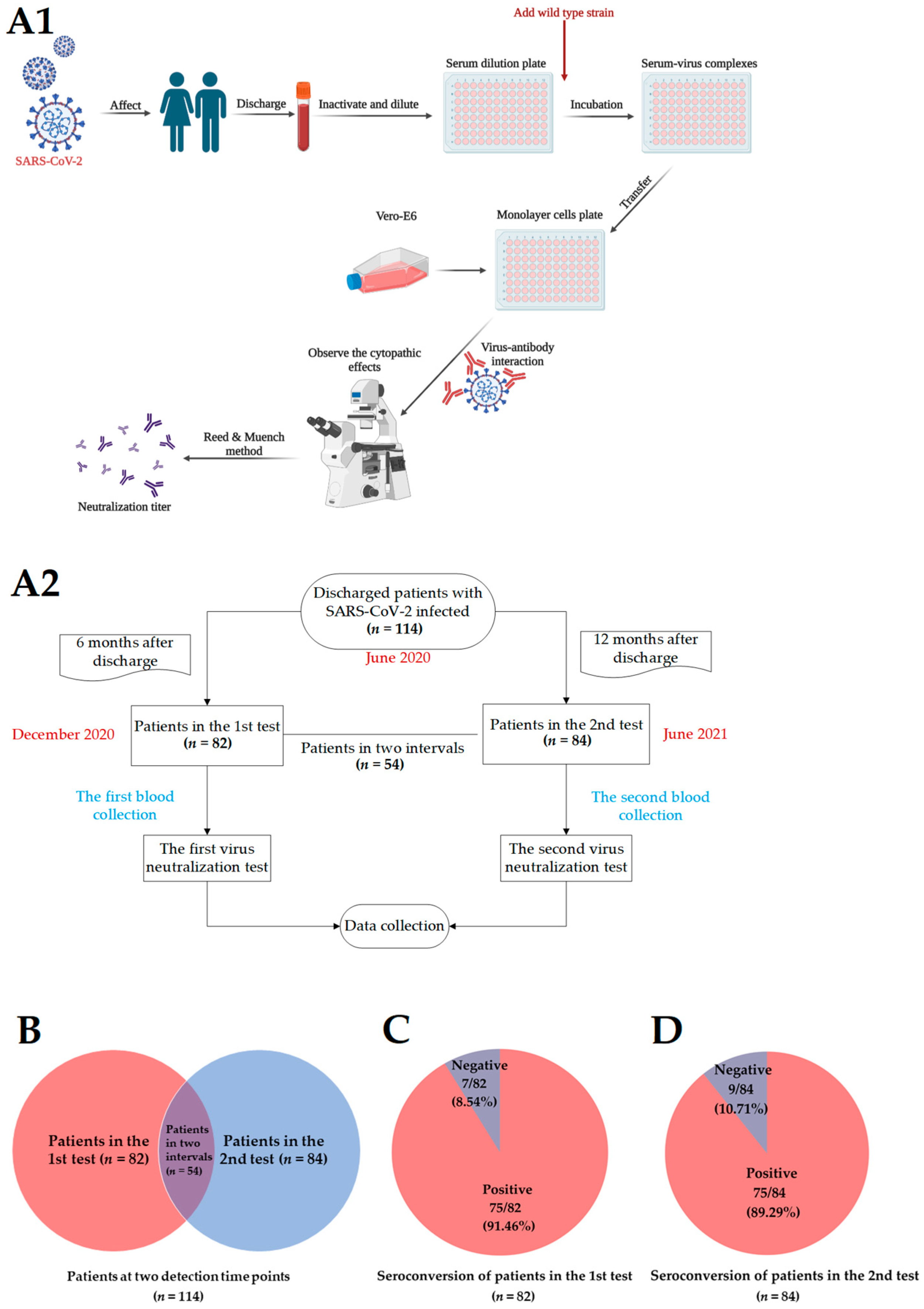
2.2. Serum Specimen
2.3. Vero-E6 Cells
2.4. SARS-CoV-2 Isolates
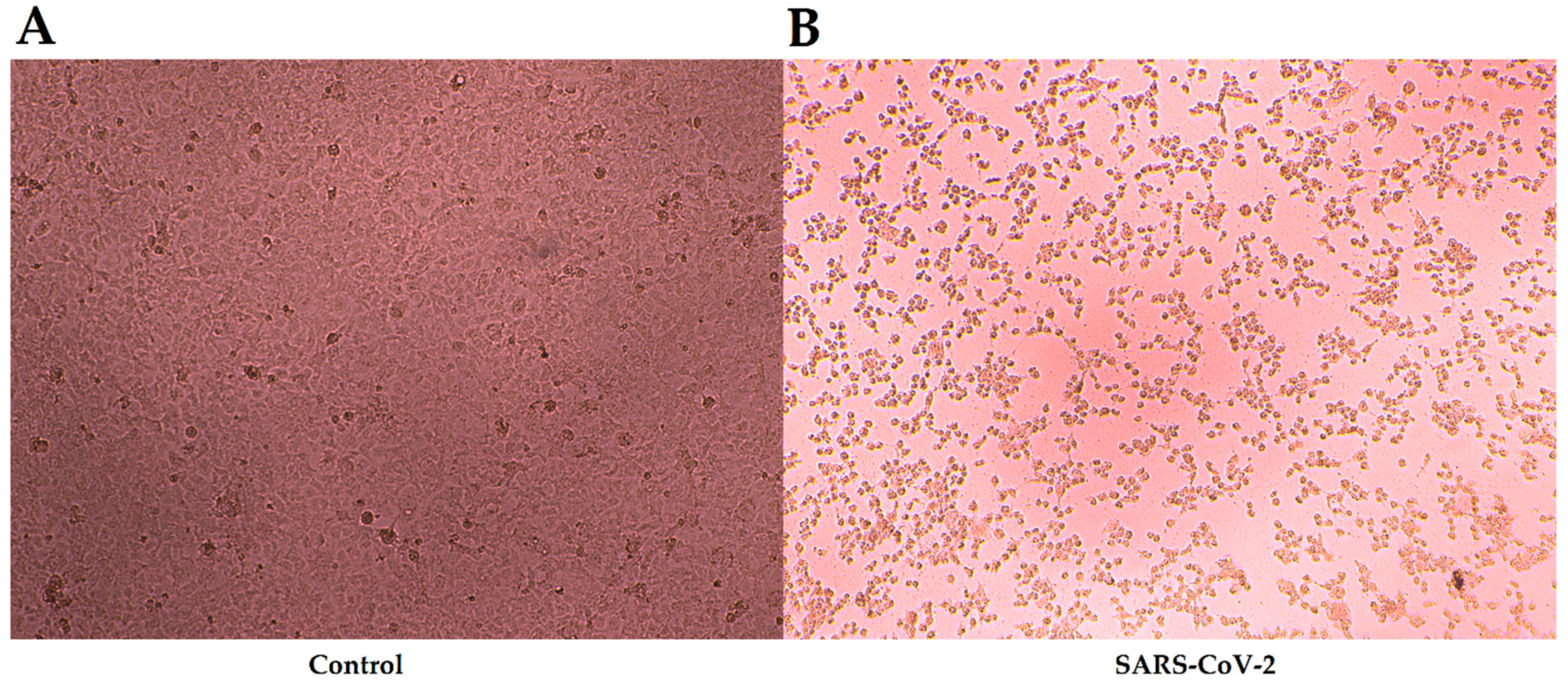
2.5. Virus Neutralization Test
2.6. Statistical Analyses
3. Results
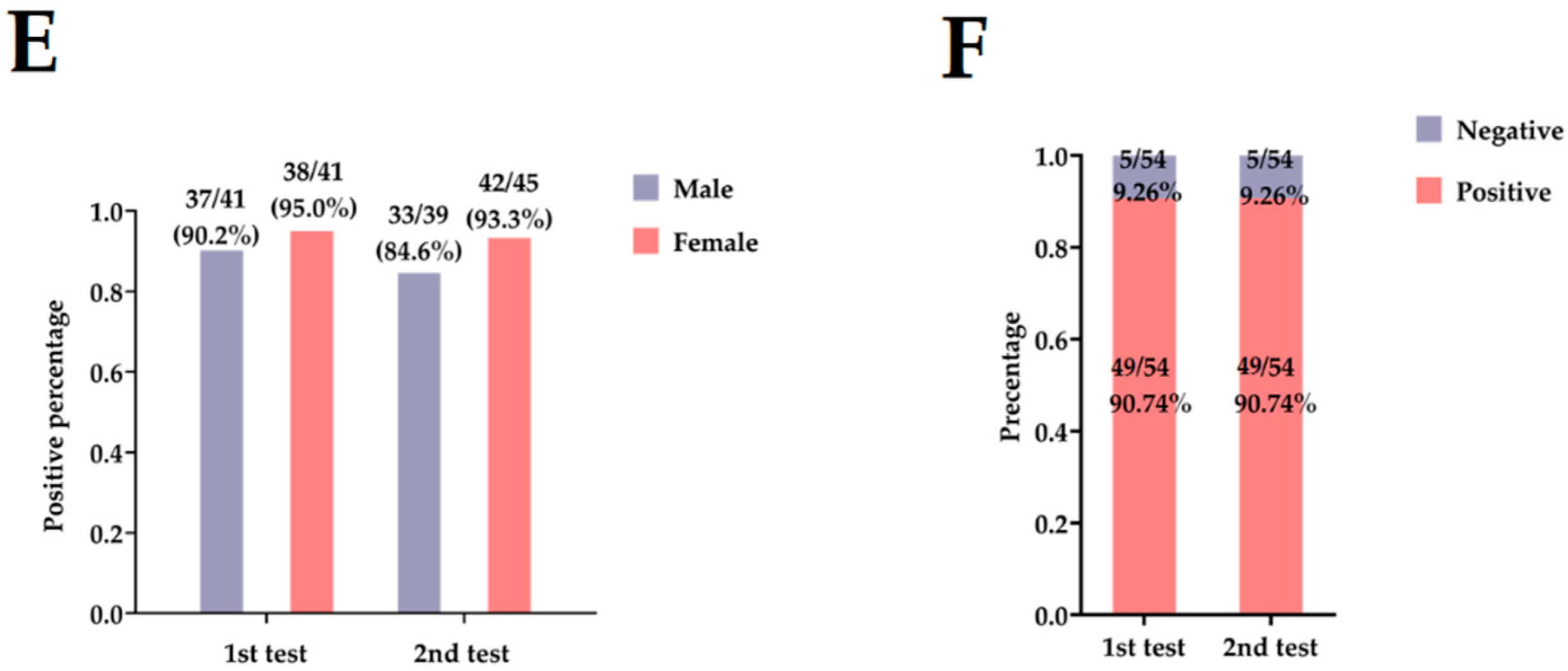
3.1. Basic Information on Patients with COVID-19
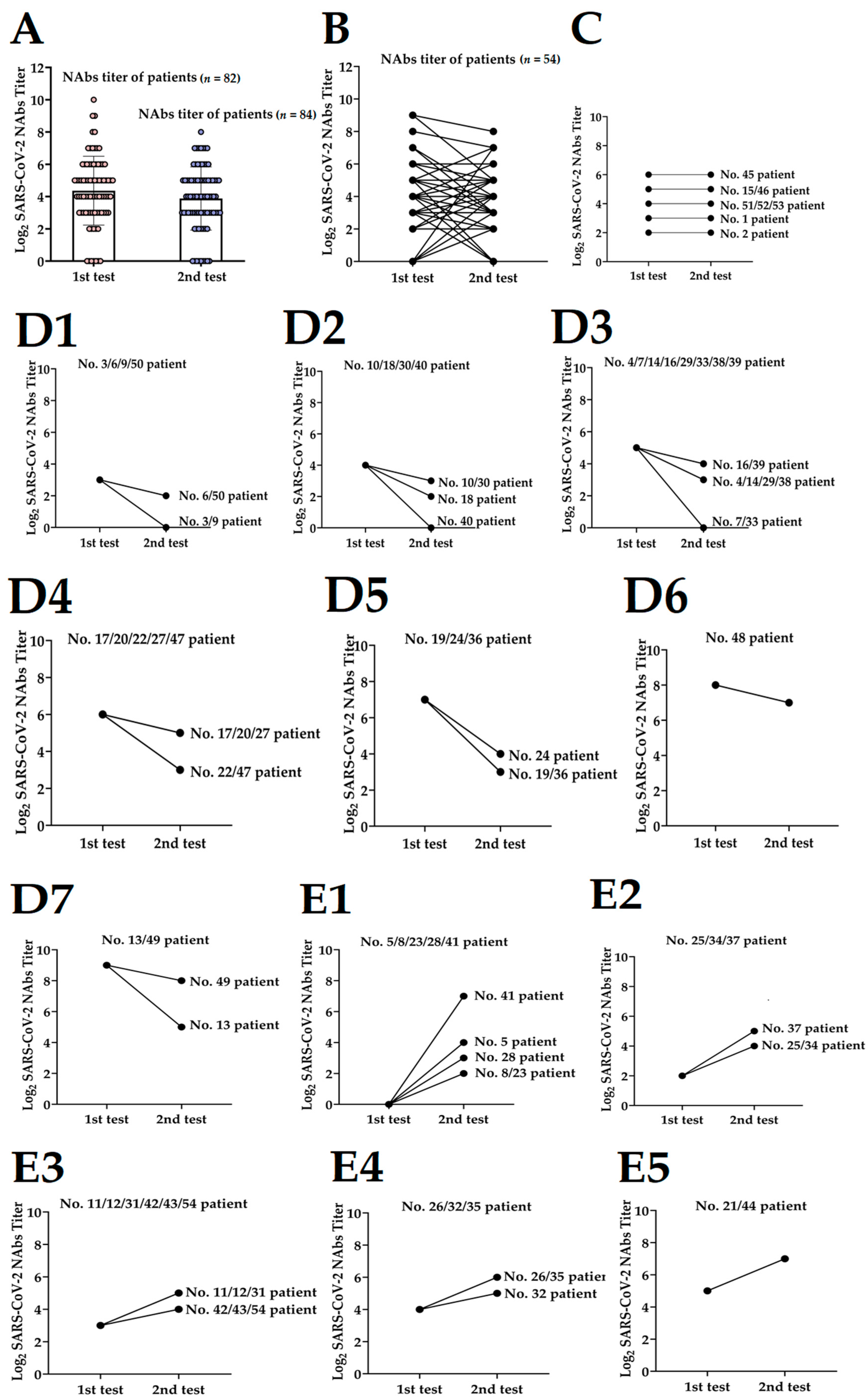
3.2. Dynamics of NAbs Level to Discharged Patients
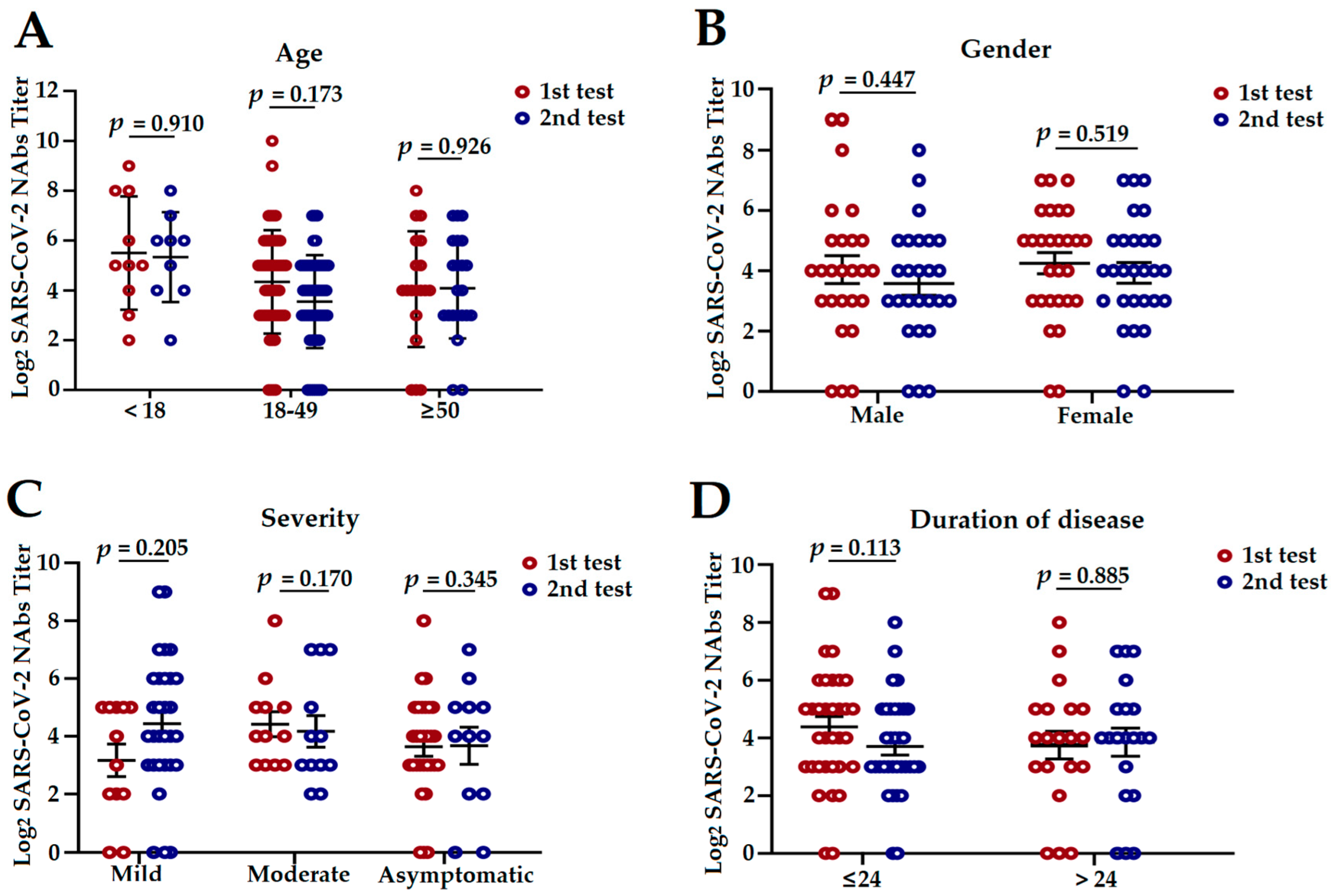
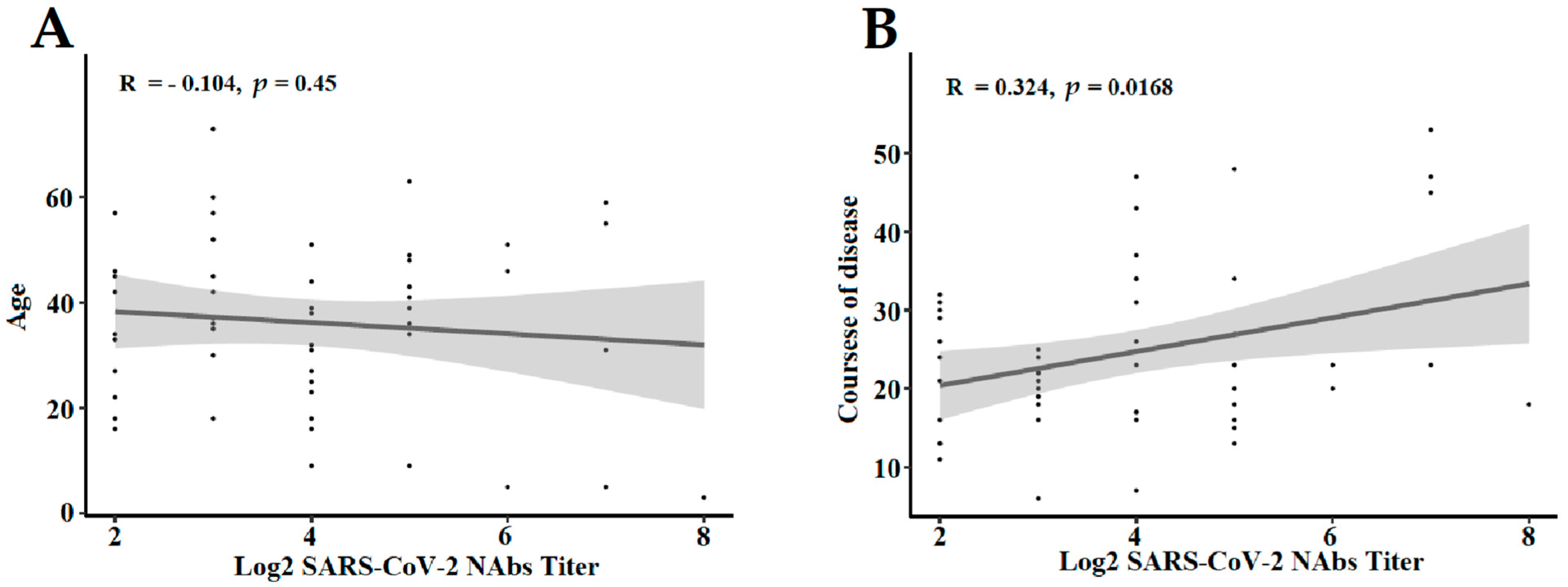
3.3. The Influencing Factors of NAbs Level in COVID-19 Convalescent Patients
3.4. The NAbs against Other Variants in COVID-19 Convalescent Patients
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- V’Kovski, P.; Kratzel, A.; Steiner, S.; Stalder, H.; Thiel, V. Coronavirus biology and replication: Implications for SARS-CoV-2. Nat. Rev. Microbiol. 2021, 19, 155–170. [Google Scholar] [CrossRef] [PubMed]
- Hirabara, S.M.; Serdan, T.D.A.; Gorjao, R.; Masi, L.N.; Pithon-Curi, T.C.; Covas, D.T.; Curi, R.; Durigon, E.L. SARS-CoV-2 Variants: Differences and Potential of Immune Evasion. Front. Cell. Infect. Microbiol. 2021, 11, 781429. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. World Health Organization (WHO)/Home/Diseases/ Coronavirus Disease (COVID-19)/Number at a Glance (last updated: 13 April 2022). Available online: https://www.who.int/emergencies/diseases/novel-coronavirus-2019 (accessed on 14 April 2022).
- Lumley, S.F.; O’Donnell, D.; Stoesser, N.E.; Matthews, P.C.; Howarth, A.; Hatch, S.B.; Marsden, B.D.; Cox, S.; James, T.; Warren, F.; et al. Antibody Status and Incidence of SARS-CoV-2 Infection in Health Care Workers. N. Engl. J. Med. 2021, 384, 533–540. [Google Scholar] [CrossRef]
- Khoury, D.S.; Cromer, D.; Reynaldi, A.; Schlub, T.E.; Wheatley, A.K.; Juno, J.A.; Subbarao, K.; Kent, S.J.; Triccas, J.A.; Davenport, M.P. Neutralizing antibody levels are highly predictive of immune protection from symptomatic SARS-CoV-2 infection. Nat. Med. 2021, 27, 1205–1211. [Google Scholar] [CrossRef]
- Muecksch, F.; Weisblum, Y.; Barnes, C.O.; Schmidt, F.; Schaefer-Babajew, D.; Wang, Z.J.; Lorenzi, J.C.C.; Flyak, A.I.; Delaitsch, A.T.; Huey-Tubman, K.E.; et al. Affinity maturation of SARS-CoV-2 neutralizing antibodies confers potency, breadth, and resilience to viral escape mutations. Immunity 2021, 54, 1853–1868.e7. [Google Scholar] [CrossRef] [PubMed]
- Matusali, G.; Colavita, F.; Lapa, D.; Meschi, S.; Bordi, L.; Piselli, P.; Galiardini, R.; Corpolongo, A.; Nicastri, E.; Antinori, A.; et al. SARS-CoV-2 Serum Neutralization Assay: A Traditional Tool for a Brand-New Virus. Viruses 2021, 13, 655. [Google Scholar] [CrossRef] [PubMed]
- Gaebler, C.; Wang, Z.; Lorenzi, J.C.C.; Muecksch, F.; Finkin, S.; Tokuyama, M.; Cho, A.; Jankovic, M.; Schaefer-Babojew, D.; Oliveira, T.Y.; et al. Evolution of antibody immunity to SARS-CoV-2. Nature 2021, 591, 639–644. [Google Scholar] [CrossRef]
- Dan, J.M.; Mateus, J.; Kato, Y.; Hastie, K.M.; Yu, E.D.; Faliti, C.E.; Grifon, A.; Ramirez, S.I.; Haupt, S.; Frazier, A.; et al. Immunological memory to SARS-CoV-2 assessed for up to 8 months after infection. Science 2021, 371, eabf4063. [Google Scholar] [CrossRef]
- Liu, W.; Fontanet, A.; Zhang, P.H.; Zhan, L.; Xin, Z.T.; Baril, L.; Tang, F.; Lv, H.; Cao, W.C. Two-year prospective study of the humoral immune response of patients with severe acute respiratory syndrome. J. Infect. Dis. 2006, 193, 792–795. [Google Scholar] [CrossRef]
- De Giorgi, V.; West, K.A.; Henning, A.N.; Chen, L.N.; Holbrook, M.R.; Gross, R.; Liang, J.; Postnikova, E.; Trenbeath, J.; Pogue, S. Naturally Acquired SARS-CoV-2 Immunity Persists for Up to 11 Months Following Infection. J. Infect. Dis. 2021, 224, 1294–1304. [Google Scholar] [CrossRef]
- Post, N.; Eddy, D.; Huntley, C.; van Schalkwyk, M.C.I.; Shrotri, M.; Leeman, D.; Rigby, S.; Williams, S.V.; Bermingham, W.H.; Kellam, P. Antibody response to SARS-CoV-2 infection in humans: A systematic review. PLoS ONE 2020, 15, e0244126. [Google Scholar] [CrossRef] [PubMed]
- Hou, H.; Zhang, Y.; Tang, G.; Luo, Y.; Liu, W.; Cheng, C.; Jiang, Y.H.; Xiong, Z.G.; Wu, S.J.; Sun, Z.Y. Immunologic memory to SARS-CoV-2 in convalescent COVID-19 patients at 1 year postinfection. J. Allergy Clin. Immunol. 2021, 148, 1481–1492.e2. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Nie, K.; Li, K.; Hu, Y.; Song, Y.; Kang, M.; Zhang, M.; Deng, X.L.; Yuan, J.; Xu, W.B. Genome Characterization of the First Outbreak of COVID-19 Delta Variant B.1.617.2—Guangzhou City, Guangdong Province, China, May 2021. China CDC Wkly. 2021, 3, 587–589. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Zheng, Z.; Zhao, X.; Zeng, Q.; Zhou, T.; Guo, Q.; Hu, Y.; Xu, W.B.; Zhang, Z.B.; Li, B.S. An Imported Case and an Infected Close Contact of the Omicron Variant of SARS-CoV-2—Guangdong Province, China, December 13, 2021. China CDC Wkly. 2022, 4, 96–97. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.Y.; Wang, W.B.; Gao, R.D.; Zhou, A.M. Omicron Variant (B.1.1.529) of SARS-CoV-2: Mutation, infectivity, transmission, and vaccine resistance. World J. Clin. Cases 2022, 10, 1–11. [Google Scholar] [CrossRef]
- University S Coronavirus Antiviral & Resistance Database. Available online: https://covdb.stanford.edu/page/susceptibility-data/ (accessed on 2 December 2022).
- Zhang, M. Omicron Now Account for 35 of US Coronavirus Cases. China Daily Global. Available online: https://global.chinadaily.com.cn/a/202112/15/WS61b95f63a310cdd39bc7b7f8.html (accessed on 2 December 2022).
- Huang, C.; Huang, L.; Wang, Y.; Li, X.; Ren, L.; Gu, X.; Kang, L.; Guo, L.; Liu, M.; Zhou, X.; et al. 6-month consequences of COVID-19 in patients discharged from hospital: A cohort study. Lancet 2021, 397, 220–232. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Battaglia, F.; Terlecky, S.R. Discharged COVID-19 patients testing positive again for SARS-CoV-2 RNA: A minireview of published studies from China. J. Med. Virol. 2021, 93, 262–274. [Google Scholar] [CrossRef]
- Zhang, H.; Zheng, H.Y.; Zou, L.R.; Liu, Z.; Liang, L.J.; Peng, X.F.; Zhang, W.; Ke, C.W.; Wu, J. First Isolation and Identification of SARS-CoV-2 in Guangdong Province, China. Chin. J. Virol. 2020, 36, 155–159. (In Chinese) [Google Scholar] [CrossRef]
- Németh, B.; Fasseeh, A.; Molnár, A.; Bitter, I.; Horváth, M.; Kóczián, K.; Götze, Á.; Nagy, B. A systematic review of health economic models and utility estimation methods in schizophrenia. Expert Rev. Pharm. Outcomes Res. 2018, 18, 267–275. [Google Scholar] [CrossRef]
- Marklund, E.; Leach, S.; Axelsson, H.; Nyström, K.; Norder, H.; Bemark, M.; Aneletti, D.; Lundgren, A.N.; Nilsson, S.; Andersson, L.M.; et al. Serum-IgG responses to SARS-CoV-2 after mild and severe COVID-19 infection and analysis of IgG non-responders. PLoS ONE 2020, 15, e0241104. [Google Scholar] [CrossRef]
- Bongen, E.; Lucian, H.; Khatri, A.; Fragiadakis, G.K.; Bjornson, Z.B.; Nolan, G.P.; Utz, P.J.; Khatri, P. Sex Differences in the Blood Transcriptome Identify Robust Changes in Immune Cell Proportions with Aging and Influenza Infection. Cell Rep. 2019, 29, 1961–1973.e4. [Google Scholar] [CrossRef] [PubMed]
- Schurz, H.; Salie, M.; Tromp, G.; Hoal, E.G.; Kinnear, C.J.; Möller, M. The X chromosome and sex-specific effects in infectious disease susceptibility. Hum. Genom. 2019, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Steeg, L.G.V.; Klein, S.L. SeXX Matters in Infectious Disease Pathogenesis. PLoS Pathog. 2016, 12, e1005374. [Google Scholar]
- Márquez, E.J.; Chung, C.H.; Marches, R.; Rossi, R.J.; Nehar-Belaid, D.; Eroglu, A.; Mellert, D.J.; Kuchel, G.A.; Banchereau, J.; Ucar, D. Sexual-dimorphism in human immune system aging. Nat. Commun. 2020, 11, 751. [Google Scholar] [CrossRef]
- Ober, C.; Loisel, D.A.; Gilad, Y. Sex-specific genetic architecture of human disease. Nat. Rev. Genet. 2008, 9, 911–922. [Google Scholar] [CrossRef]
- Schmiedel, B.J.; Singh, D.; Madrigal, A.; Valdovino-Gonzalez, A.G.; White, B.M.; Zapardiel-Gonzalo, J.; Ha, B.; Altay, G.; Greenbaum, J.A.; McVicker, G.; et al. Impact of Genetic Polymorphisms on Human Immune Cell Gene Expression. Cell. 2018, 175, 1701–1715.e16. [Google Scholar] [CrossRef]
- Piasecka, B.; Duffy, D.; Urrutia, A.; Quach, H.; Patin, E.; Posseme, C.; Bergstedt, J.; Charbit, B.; Rouilly, V.; MacPherson, C.R.; et al. Distinctive roles of age, sex, and genetics in shaping transcriptional variation of human immune responses to microbial challenges. Proc. Natl. Acad. Sci. USA 2018, 115, E488–E497. [Google Scholar] [CrossRef]
- Xia, S.; Zhang, Y.; Wang, Y.; Wang, H.; Yang, Y.; Gao, G.F.; Tan, W.J.; Wu, G.Z.; Xu, M.; Lou, Z.Y.; et al. Safety and immunogenicity of an inactivated SARS-CoV-2 vaccine, BBIBP-CorV: A randomised, double-blind, placebo-controlled, phase 1/2 trial. Lancet Infect. Dis. 2021, 21, 39–51. [Google Scholar] [CrossRef]
- Zhang, J.; Xing, S.; Liang, D.; Hu, W.; Ke, C.; He, J.; Yuan, R.Y.; Huang, Y.L.; Li, Y.Z.; Liu, X.D.; et al. Differential Antibody Response to Inactivated COVID-19 Vaccines in Healthy Subjects. Front. Cell. Infect. Microbiol. 2021, 11, 791660. [Google Scholar] [CrossRef]
- Walsh, E.E.; Frenck, R.W., Jr.; Falsey, A.R.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Mulligan, M.J.; Bailey, R.; et al. Safety and Immunogenicity of Two RNA-Based COVID-19 Vaccine Candidates. N. Engl. J. Med. 2020, 383, 2439–2450. [Google Scholar] [CrossRef]
- Li, X.; Pang, L.; Yin, Y.; Zhang, Y.; Xu, S.; Xu, D.; Shen, T. Patient and Clinical Factors at Admission Affect the Levels of Neutralizing Antibodies Six Months after Recovering from COVID-19. Viruses 2022, 14, 80. [Google Scholar] [CrossRef] [PubMed]
- Tregoning, J.S.; Flight, K.E.; Higham, S.L.; Wang, Z.; Pierce, B.F. Progress of the COVID-19 vaccine effort: Viruses, vaccines and variants versus efficacy, effectiveness and escape. Nat. Rev. Immunol. 2021, 21, 626–636. [Google Scholar] [CrossRef] [PubMed]
- Tang, F.; Quan, Y.; Xin, Z.T.; Wrammert, J.; Ma, M.J.; Lv, H.; Wang, T.B.; Yang, H.; Richardus, J.H.; Liu, W. Lack of peripheral memory B cell responses in recovered patients with severe acute respiratory syndrome: A six-year follow-up study. J. Immunol. 2011, 186, 7264–7268. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.T.; Peng, H.; Zhu, Z.L.; Li, G.; Huang, Z.T.; Zhao, Z.X.; Koup, R.A.; Bailer, R.T.; Wu, C.Y. Long-lived effector/central memory T-cell responses to severe acute respiratory syndrome coronavirus (SARS-CoV) S antigen in recovered SARS patients. Clin. Immunol. 2006, 120, 171–178. [Google Scholar] [CrossRef]
- Long, Q.X.; Liu, B.Z.; Deng, H.J.; Wu, G.C.; Deng, K.; Chen, Y.K.; Liao, P.; Qie, J.F.; Lin, Y.; Cai, X.F. Antibody responses to SARS-CoV-2 in patients with COVID-19. Nat. Med. 2020, 26, 845–848. [Google Scholar] [CrossRef]
- Cohen, K.W.; Linderman, S.L.; Moodie, Z.; Czartoski, J.; Lai, L.; Mantus, G.; Norwood, C.; Nyhoff, L.E.; Edara, V.V.; Floyd, K.; et al. Longitudinal analysis shows durable and broad immune memory after SARS-CoV-2 infection with persisting antibody responses and memory B and T cells. Cell Rep. Med. 2021, 2, 100354. [Google Scholar] [CrossRef]
- Rodda, L.B.; Netland, J.; Shehata, L.; Pruner, K.B.; Morawski, P.A.; Thouvenel, C.D.; Takehara, K.K.; Eggenberger, J.; Hemann, E.A.; Waterman, H.R.; et al. Functional SARS-CoV-2-Specific Immune Memory Persists after Mild COVID-19. Cell 2021, 184, 169–183.e17. [Google Scholar] [CrossRef]
- Xiang, T.; Liang, B.; Fang, Y.; Lu, S.; Li, S.; Wang, H.; Li, H.d.; Yang, X.L.; Shen, S.; Zhu, S.; et al. Declining Levels of Neutralizing Antibodies Against SARS-CoV-2 in Convalescent COVID-19 Patients One Year Post Symptom Onset. Front. Immunol. 2021, 12, 708523. [Google Scholar] [CrossRef]
- Beaudoin-Bussières, G.; Laumaea, A.; Anand, S.P.; Prévost, J.; Gasser, R.; Goyette, G.; Medjahed, H.; Perreaut, J.; Tremblay, T.; Lewin, A.; et al. Decline of Humoral Responses against SARS-CoV-2 Spike in Convalescent Individuals. mBio 2020, 11, e02590-20. [Google Scholar] [CrossRef]
- Seow, J.; Graham, C.; Merrick, B.; Acors, S.; Pickering, S.; Steel, K.J.A.; Hemmings, O.; O’Byrne, A.; Kouphou, N.; Galao, R.P.; et al. Longitudinal observation and decline of neutralizing antibody responses in the three months following SARS-CoV-2 infection in humans. Nat. Microbiol. 2020, 5, 1598–1607. [Google Scholar] [CrossRef]
- Thomopoulos, T.P.; Rosati, M.; Terpos, E.; Stellas, D.; Hu, X.; Karaliota, S.; Bouchla, A.; Katagas, L.; Antoniadou, A.; Mentis, A.; et al. Kinetics of Nucleocapsid, Spike and Neutralizing Antibodies, and Viral Load in Patients with Severe COVID-19 Treated with Convalescent Plasma. Viruses 2021, 13, 1844. [Google Scholar] [CrossRef]
- Addetia, A.; Crawford, K.H.D.; Dingens, A.; Zhu, H.; Roychoudhury, P.; Huang, M.L.; Jerome, K.R.; Bloom, J.D.; Greninger, A.L. Neutralizing Antibodies Correlate with Protection from SARS-CoV-2 in Humans during a Fishery Vessel Outbreak with a High Attack Rate. J. Clin. Microbiol. 2020, 58, e02107-20. [Google Scholar] [CrossRef]
- Guo, Y.; Huang, L.; Zhang, G.; Yao, Y.; Zhou, H.; Shen, S.; Shen, B.Q.; Li, B.; Li, X.; Zhang, Q.; et al. A SARS-CoV-2 neutralizing antibody with extensive Spike binding coverage and modified for optimal therapeutic outcomes. Nat. Commun. 2021, 12, 2623. [Google Scholar] [CrossRef] [PubMed]
- China NHCotPsRo. Trial 8th Edition Diagnosis and Treatment Program of Novel Coronavirus Pneumonia (Last Updated: 15 April 2021). Available online: http://www.nhc.gov.cn/xcs/zhengcwj/202104/7de0b3837c8b4606a0594aeb0105232b.shtml (accessed on 15 April 2022).
- Klein, S.L.; Flanagan, K.L. Sex differences in immune responses. Nat. Rev. Immunol. 2016, 16, 626–638. [Google Scholar] [CrossRef] [PubMed]
- Klein, S.L.; Bird, B.H.; Glass, G.E. Sex differences in immune responses and viral shedding following Seoul virus infection in Norway rats. Am. J. Trop. Med. Hyg. 2001, 65, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Park, M.D. Sex differences in immune responses in COVID-19. Nat. Rev. Immunol. 2020, 20, 461. [Google Scholar] [CrossRef]
- Fathi, A.; Addo, M.M.; Dahlke, C. Sex Differences in Immunity: Implications for the Development of Novel Vaccines Against Emerging Pathogens. Front. Immunol. 2020, 11, 601170. [Google Scholar] [CrossRef]
- Selvi, V. Convalescent Plasma: A Challenging Tool to Treat COVID-19 Patients-A Lesson from the Past and New Perspectives. BioMed Res. Int. 2020, 2020, 2606058. [Google Scholar] [CrossRef]
- Rössler, A.; Riepler, L.; Bante, D.; von Laer, D.; Kimpel, J. SARS-CoV-2 Omicron Variant Neutralization in Serum from Vaccinated and Convalescent Persons. N. Engl. J. Med. 2022, 386, 698–700. [Google Scholar] [CrossRef]
- Khoury, J.; Najjar-Debbiny, R.; Hanna, A.; Jabbour, A.; Abu Ahmad, Y.; Saffuri, A.; Abu-Sinni, M.; Shkeiri, R.; Elemy, A.; Hakim, F. COVID-19 vaccine -Long term immune decline and breakthrough infections. Vaccine 2021, 39, 6984–6989. [Google Scholar] [CrossRef]
- Choi, J.Y.; Smith, D.M. SARS-CoV-2 Variants of Concern. Yonsei Med. J. 2021, 62, 961–968. [Google Scholar] [CrossRef] [PubMed]
- Sanyaolu, A.; Okorie, C.; Marinkovic, A.; Haider, N.; Abbasi, A.F.; Jaferi, U.; Prakash, S.; Balendra, V. The emerging SARS-CoV-2 variants of concern. Ther. Adv. Infect. Dis. 2021, 8, 20499361211024372. [Google Scholar] [CrossRef] [PubMed]
- Campbell, F.; Archer, B.; Laurenson-Schafer, H.; Jinnai, Y.; Konings, F.; Batra, N.; Pavlin, B.; Vandemaele, K.; Kerkhove, M.D.V.; Jombart, T.; et al. Increased transmissibility and global spread of SARS-CoV-2 variants of concern as at June 2021. Eurosurveillance 2021, 26, 2100509. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Li, B.; Hao, P.; Song, Y.; Xu, W.; Liu, N.; Lei, C.L.; Ke, C.W. A Case of New Variant COVID-19 First Emerging in South Africa Detected in Airplane Pilot—Guangdong Province, China, January 6, 2021. China CDC Wkly. 2021, 3, 28–29. [Google Scholar] [CrossRef]
| Male (n = 56) | Female (n = 58) | Total (n = 114) | p-Value | |
|---|---|---|---|---|
| Age, years | 0.192 | |||
| <18 | 10 | 4 | 14 | |
| 18–50 | 33 | 37 | 70 | |
| ≥50 | 13 | 17 | 30 | |
| Median age (IQR) | 39.00 (27.00–48.75) | 39.00 (29.75–52.25) | 39.00 (27.75–50.25) | |
| Clinical classification | 1.000 | |||
| Asymptomatic case | 9 | 9 | 18 | |
| Mild case | 7 | 9 | 16 | |
| Moderate case | 40 | 40 | 80 | |
| NAbs titer | ||||
| 1st test (n = 82) | 41 | 41 | 82 | 1.000 |
| Seronegative (<1:4) | 4 | 3 | 7 | |
| Seropositive (≥1:4) | 37 | 38 | 75 | |
| GMT1st test | 1:23.21 | |||
| 2nd test (n = 84) | 39 | 45 | 84 | 0.292 |
| Seronegative (<1:4) | 6 | 3 | 9 | |
| Seropositive (≥1:4) | 33 | 42 | 75 | |
| GMT2nd test | 1:17.09 | |||
| Average duration of disease (days) | 25.06 | 22.87 | 23.90 | 0.258 |
| Downtrend Curve 1 | Uptrend Curve 2 | Unconverted Curve 3 | p-Value | ||
|---|---|---|---|---|---|
| Total | 10 | 27 | 17 | 0.302 | |
| Male | 7 | 12 | 7 | ||
| Female | 3 | 15 | 10 | ||
| Age (years) | 39.00 (30.00–49.00) | 43.00 (31.50–51.00) | 22.50 (16.00–36.25) | 0.026 a* | |
| Clinical classification | Asymptomatic case | 6 | 5 | 1 | 0.003 b* |
| Mild case | 2 | 3 | 5 | ||
| Moderate case | 1 | 18 | 11 | ||
| Duration of disease | 20.69 | 27.88 | 29.89 | 0.015 c* |
| SAlpha (n = 3) | SBeta (n = 3) | SDelta (n = 31) | SOmicron (n = 25) | |
|---|---|---|---|---|
| Median Age, years | 39 | 47 | 44 | 35 |
| Gender | ||||
| Male | 2 | 3 | 12 | 4 |
| Female | 1 | 0 | 19 | 21 |
| Vaccination history | ||||
| Non-vaccinees | 3 | 3 | 19 | 1 |
| 1st dose | 0 | 0 | 7 | 1 |
| 2nd dose | 0 | 0 | 5 | 9 |
| 3rd dose | 0 | 0 | 0 | 14 |
| GMTs | ||||
| VWild | 50.4 | 5.85 | 25.60 | 263.55 |
| VAlpha | 25.20 | 1.00 | 16.62 | 256.34 |
| VBeta | 31.75 | 7.94 | 12.06 | 164.50 |
| VGamma | 80.00 | 40.00 | 87.48 | 572.82 |
| VDelta | 31.75 | 3.42 | 101.57 | 223.16 |
| VOmicron | 2.15 | 1.71 | 3.19 | 102.67 |
| Vaccination History | GMTs of SDelta (n = 31) | Vaccination History | GMTs of SOmicron (n = 25) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| VWild | VAlpha | VBeta | VGamma | VDelta | VOmicron | VWild | VAlpha | VBeta | VGamma | VDelta | VOmicron | ||
| Non-vaccinees (n = 19) | 18.84 | 24.32 | 19.54 | 115.22 | 160 | 4.78 | Non-vaccinees (n = 1) | 160 | 160 | 20 | 160 | 160 | 40 |
| 1st dose (n = 7) | 63.57 | 5.72 | 3.38 | 24.38 | 23.61 | 1.26 | 1st dose (n = 1) | 10 | 160 | 160 | 320 | 80 | 40 |
| 2nd dose (n = 5) | 22.97 | 17.41 | 11.49 | 183.79 | 139.29 | 2.51 | 2nd dose (n = 9) | 296.28 | 320 | 217.73 | 507.97 | 217.73 | 93.32 |
| 3rd dose (n = 0) | 0 | 0 | 0 | 0 | 0 | 0 | 3rd dose (n = 14) | 320 | 237.76 | 160 | 706.62 | 249.83 | 124.91 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, D.; Zhang, G.; Huang, M.; Wang, L.; Hong, W.; Li, A.; Liang, Y.; Wang, T.; Lu, J.; Ou, M.; et al. Progress of the COVID-19: Persistence, Effectiveness, and Immune Escape of the Neutralizing Antibody in Convalescent Serum. Pathogens 2022, 11, 1531. https://doi.org/10.3390/pathogens11121531
Liang D, Zhang G, Huang M, Wang L, Hong W, Li A, Liang Y, Wang T, Lu J, Ou M, et al. Progress of the COVID-19: Persistence, Effectiveness, and Immune Escape of the Neutralizing Antibody in Convalescent Serum. Pathogens. 2022; 11(12):1531. https://doi.org/10.3390/pathogens11121531
Chicago/Turabian StyleLiang, Dan, Guanting Zhang, Mingxing Huang, Li Wang, Wenshan Hong, An’an Li, Yufeng Liang, Tao Wang, Jiahui Lu, Mengdang Ou, and et al. 2022. "Progress of the COVID-19: Persistence, Effectiveness, and Immune Escape of the Neutralizing Antibody in Convalescent Serum" Pathogens 11, no. 12: 1531. https://doi.org/10.3390/pathogens11121531
APA StyleLiang, D., Zhang, G., Huang, M., Wang, L., Hong, W., Li, A., Liang, Y., Wang, T., Lu, J., Ou, M., Ren, Z., Lu, H., Zheng, R., Cai, X., Pan, X., Xia, J., & Ke, C. (2022). Progress of the COVID-19: Persistence, Effectiveness, and Immune Escape of the Neutralizing Antibody in Convalescent Serum. Pathogens, 11(12), 1531. https://doi.org/10.3390/pathogens11121531







