Artificial Intelligence in Surveillance, Diagnosis, Drug Discovery and Vaccine Development against COVID-19
Abstract
:1. Introduction
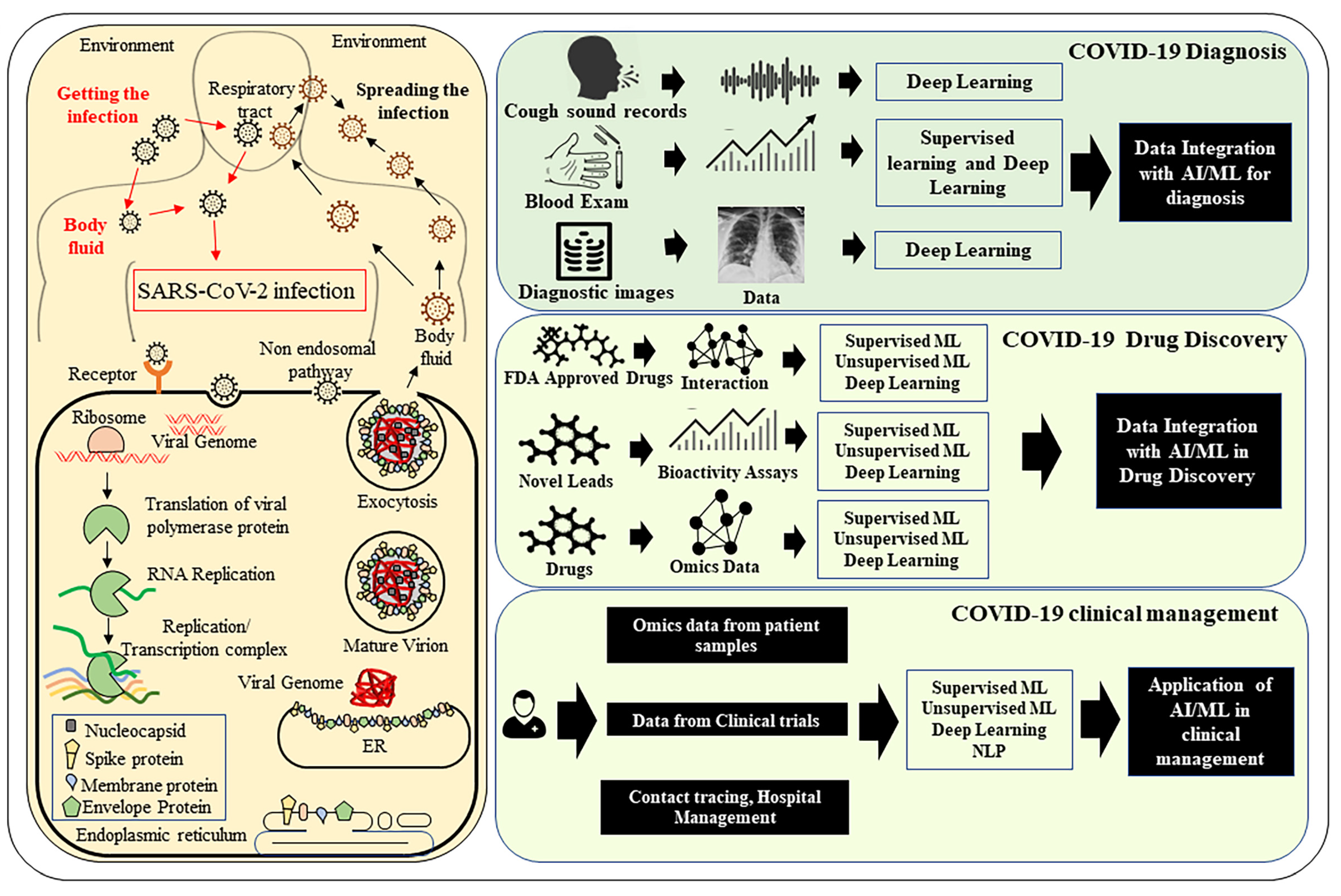
1.1. Pathophysiology of COVID-19
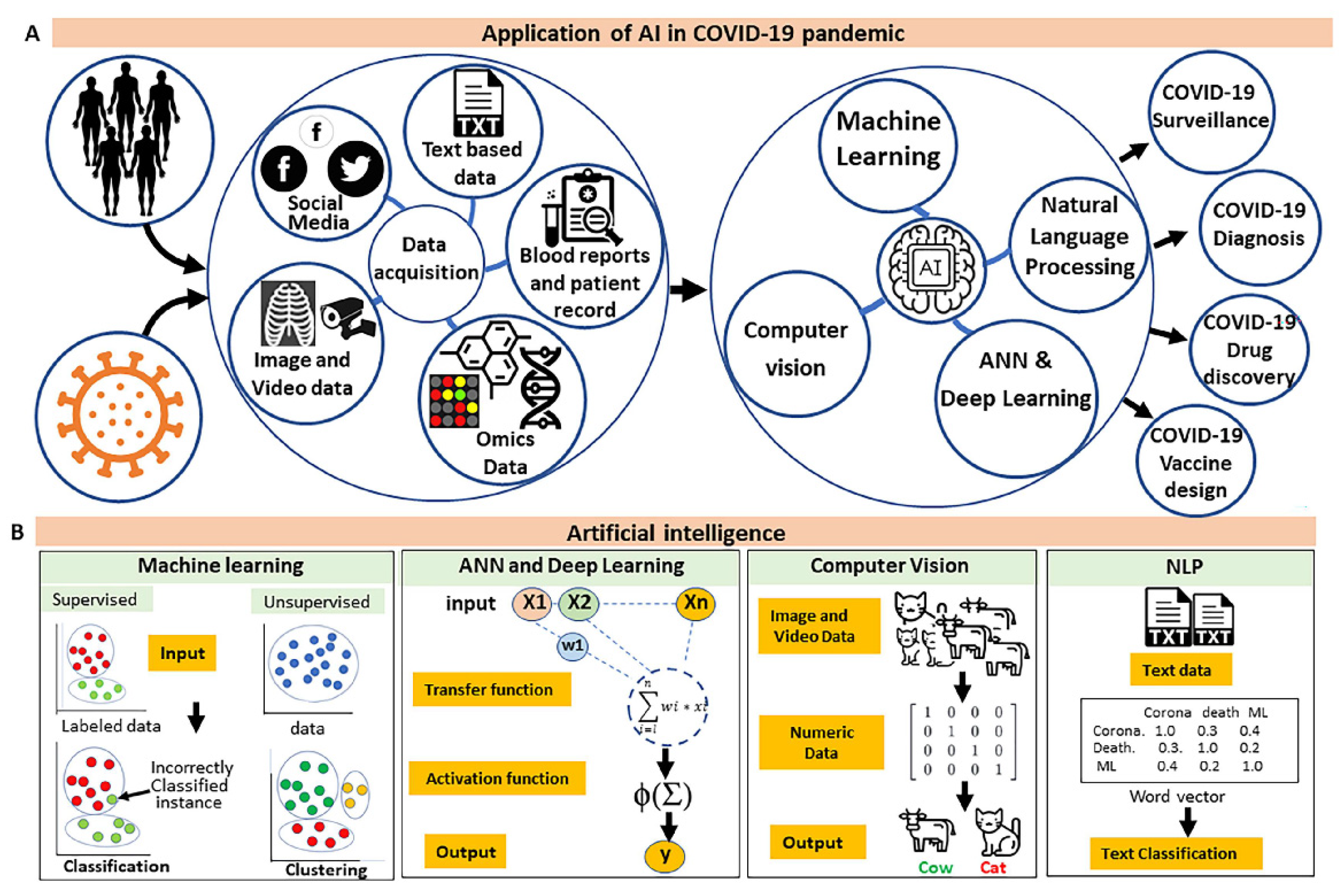
1.2. Advancement of Computational Methods to Combat COVID-19 Pandemic
2. Application of AI in Surveillance of COVID-19
3. Role of AI in the Screening of COVID-19 Infected Patients and Diagnosis
3.1. Imaging-Based Diagnostics
3.2. Blood Analysis Tests
3.3. Analysis of Text and Voice Data
4. Application of AI in Predicting COVID-19 Outcome
5. Application of AI in Drug Discovery
6. Application of AI in Vaccine Development and Delivery
7. Application of AI in Predicting Possible Viral Mutational Landscape
8. Challenges and Limitations Associated with AI
9. Discussion and Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO Coronavirus (COVID-19) Dashboard. Available online: https://covid19.who.int/ (accessed on 7 August 2021).
- Gavas, M.; Pleeck, S. Global Trends in 2021: How COVID-19 Is Transforming International Development. Center for Global Development, 2021; pp. 1–16. Available online: https://www.cgdev.org/publication/global-trends-2021-how-Covid-transforming-international-development (accessed on 7 August 2021).
- Fenizia, C.; Biasin, M.; Cetin, I.; Vergani, P.; Mileto, D.; Spinillo, A.; Gismondo, M.R.; Perotti, F.; Callegari, C.; Mancon, A.; et al. Analysis of SARS-CoV-2 vertical transmission during pregnancy. Nat. Commun. 2020, 11, 5128. [Google Scholar] [CrossRef]
- Greenhalgh, T.; Jimenez, J.L.; Prather, K.A.; Tufekci, Z.; Fisman, D.; Schooley, R. Ten scientific reasons in support of airborne transmission of SARS-CoV-2. Lancet 2021, 397, 1603–1605. [Google Scholar] [CrossRef]
- Sia, S.F.; Yan, L.-M.; Chin, A.W.H.; Fung, K.; Choy, K.-T.; Wong, A.Y.L.; Kaewpreedee, P.; Perera, R.A.P.M.; Poon, L.L.M.; Nicholls, J.M.; et al. Pathogenesis and transmission of SARS-CoV-2 in golden hamsters. Nature 2020, 583, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Mao, Y.; Jones, R.M.; Tan, Q.; Ji, J.S.; Li, N.; Shen, J.; Lv, Y.; Pan, L.; Ding, P.; et al. Aerosol transmission of SARS-CoV-2? Evidence, prevention and control. Environ. Int. 2020, 144, 106039. [Google Scholar] [CrossRef] [PubMed]
- Jayaweera, M.; Perera, H.; Gunawardana, B.; Manatunge, J. Transmission of COVID-19 virus by droplets and aerosols: A critical review on the unresolved dichotomy. Environ. Res. 2020, 188, 109819. [Google Scholar] [CrossRef]
- Stadnytskyi, V.; Bax, C.E.; Bax, A.; Anfinrud, P. The airborne lifetime of small speech droplets and their potential importance in SARS-CoV-2 transmission. Proc. Natl. Acad. Sci. USA 2020, 117, 11875–11877. [Google Scholar] [CrossRef]
- Stadnytskyi, V.; Anfinrud, P.; Bax, A. Breathing, speaking, coughing or sneezing: What drives transmission of SARS-CoV-2? J. Intern. Med. 2021. [Google Scholar] [CrossRef]
- Lewis, D. Superspreading drives the Covid pandemic—And could help to tame it. Nature 2021, 590, 544–546. [Google Scholar] [CrossRef]
- Edwards, D.A.; Ausiello, D.; Salzman, J.; Devlin, T.; Langer, R.; Beddingfield, B.J.; Fears, A.C.; Doyle-Meyers, L.A.; Redmann, R.K.; Killeen, S.Z.; et al. Exhaled aerosol increases with COVID-19 infection, age, and obesity. Proc. Natl. Acad. Sci. USA 2021, 118, e2021830118. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Li, T.; Deng, Y.; Liu, S.; Zhang, D.; Li, H.; Wang, X.; Jia, L.; Han, J.; Bei, Z.; et al. Stability of SARS-CoV-2 on environmental surfaces and in human excreta. J. Hosp. Infect. 2021, 107, 105–107. [Google Scholar] [CrossRef]
- Marques, M.; Domingo, J.L. Contamination of inert surfaces by SARS-CoV-2: Persistence, stability and infectivity. A review. Environ. Res. 2021, 193, 110559. [Google Scholar] [CrossRef] [PubMed]
- Cao, W.; Li, T. COVID-19: Towards understanding of pathogenesis. Cell Res. 2020, 30, 367–369. [Google Scholar] [CrossRef] [PubMed]
- Morawska, L.; Tang, J.W.; Bahnfleth, W.; Bluyssen, P.M.; Boerstra, A.; Buonanno, G.; Cao, J.; Dancer, S.; Floto, A.; Franchimon, F.; et al. How can airborne transmission of COVID-19 indoors be minimised? Environ. Int. 2020, 142, 105832. [Google Scholar] [CrossRef]
- Yuki, K.; Fujiogi, M.; Koutsogiannaki, S. COVID-19 pathophysiology: A review. Clin. Immunol. 2020, 215, 108427. [Google Scholar] [CrossRef]
- Zhai, P.; Ding, Y.; Wu, X.; Long, J.; Zhong, Y.; Li, Y. The epidemiology, diagnosis and treatment of COVID-19. Int. J. Antimicrob. Agents 2020, 55, 105955. [Google Scholar] [CrossRef]
- Lauer, S.A.; Grantz, K.H.; Bi, Q.; Jones, F.K.; Zheng, Q.; Meredith, H.R.; Azman, A.S.; Reich, N.G.; Lessler, J. The incubation period of coronavirus disease 2019 (COVID-19) from publicly reported confirmed cases: Estimation and application. Ann. Intern. Med. 2020, 172, 577–582. [Google Scholar] [CrossRef] [Green Version]
- Shanmugam, C.; Mohammed, A.R.; Ravuri, S.; Luthra, V.; Rajagopal, N.; Karre, S. Covid-2019—A comprehensive pathology insight. Pathol. Res. Pract. 2020, 216, 153222. [Google Scholar] [CrossRef]
- Gao, M.; Yang, L.; Chen, X.; Deng, Y.; Yang, S.; Xu, H.; Chen, Z.; Gao, X. A study on infectivity of asymptomatic SARS-CoV-2 carriers. Respir. Med. 2020, 169, 106026. [Google Scholar] [CrossRef]
- Nikolai, L.A.; Meyer, C.G.; Kremsner, P.G.; Velavan, T.P. Asymptomatic sars coronavirus 2 infection: Invisible yet invincible. Int. J. Infect. Dis. 2020, 100, 112–116. [Google Scholar] [CrossRef]
- Singh, L.; Bajaj, S.; Gadewar, M.; Verma, N.; Ansari, M.N.; Saeedan, A.S.; Kaithwas, G.; Singh, M. Modulation of host immune response is an alternative strategy to combat SARS-CoV-2 pathogenesis. Front. Immunol. 2021, 12, 660632. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Khan, Z.A.; Charles, M.; Pratap, P.; Naeem, A.; Siddiqui, Z.; Naqvi, N.; Srivastava, S. Cytokine storm and mucus hypersecretion in COVID-19: Review of mechanisms. J. Inflamm. Res. 2021, 14, 175–189. [Google Scholar] [CrossRef]
- Davido, B.; Seang, S.; Tubiana, R.; de Truchis, P. Post-COVID-19 chronic symptoms: A postinfectious entity? Clin. Microbiol. Infect. 2020, 26, 1448–1449. [Google Scholar] [CrossRef] [PubMed]
- Romagnoli, S.; Peris, A.; De Gaudio, A.R.; Geppetti, P. SARS-CoV-2 and COVID-19: From the bench to the bedside. Physiol. Rev. 2020, 100, 1455–1466. [Google Scholar] [CrossRef]
- Siddiqi, H.K.; Mehra, M.R. COVID-19 illness in native and immunosuppressed states: A clinical-therapeutic staging proposal. J. Heart Lung Transplant. 2020, 39, 405–407. [Google Scholar] [CrossRef] [Green Version]
- Mason, R.J. Pathogenesis of COVID-19 from a cell biology perspective. Eur. Respir. J. 2020, 55, 2000607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, C.; Lu, W.; Li, K.; Ding, Y.; Wang, J. Ace2 expression in kidney and testis may cause kidney and testis infection in COVID-19 patients. Front. Med. 2020, 7, 563893. [Google Scholar] [CrossRef] [PubMed]
- Strohbehn, I.A.; Zhao, S.; Seethapathy, H.; Lee, M.; Rusibamayila, N.; Allegretti, A.S.; Parada, X.V.; Sise, M.E. Acute kidney injury incidence, recovery, and long-term kidney outcomes among hospitalized patients with COVID-19 and influenza. Kidney Int. Rep. 2021. [Google Scholar] [CrossRef]
- Abd El-Aziz, T.M.; Stockand, J.D. Recent progress and challenges in drug development against COVID-19 coronavirus (SARS-CoV-2)—An update on the status. Infect. Genet. Evol. 2020, 83, 104327. [Google Scholar] [CrossRef] [PubMed]
- Mao, R.; Qiu, Y.; He, J.-S.; Tan, J.-Y.; Li, X.-H.; Liang, J.; Shen, J.; Zhu, L.-R.; Chen, Y.; Iacucci, M.; et al. Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: A systematic review and meta-analysis. Lancet Gastroenterol. Hepatol. 2020, 5, 667–678. [Google Scholar] [CrossRef]
- Ma, C.; Cong, Y.; Zhang, H. COVID-19 and the digestive system. Am. J. Gastroenterol. 2020, 115, 1003–1006. [Google Scholar] [CrossRef]
- Sultan, S.; Altayar, O.; Siddique, S.M.; Davitkov, P.; Feuerstein, J.D.; Lim, J.K.; Falck-Ytter, Y.; El-Serag, H.B.; AGA Institute. Aga institute rapid review of the gastrointestinal and liver manifestations of COVID-19, meta-analysis of international data, and recommendations for the consultative management of patients with COVID-19. Gastroenterology 2020, 159, 320–334.e27. [Google Scholar] [CrossRef] [PubMed]
- Hundt, M.A.; Deng, Y.; Ciarleglio, M.M.; Nathanson, M.H.; Lim, J.K. Abnormal liver tests in COVID-19: A retrospective observational cohort study of 1827 patients in a major U.S. Hospital network. Hepatology 2020, 72, 1169–1176. [Google Scholar] [CrossRef]
- Zhang, C.; Shi, L.; Wang, F.S. Liver injury in COVID-19: Management and challenges. Lancet Gastroenterol. Hepatol. 2020, 5, 428–430. [Google Scholar] [CrossRef]
- Cao, Y.-C.; Deng, Q.-X.; Dai, S.-X. Remdesivir for severe acute respiratory syndrome coronavirus 2 causing COVID-19: An evaluation of the evidence. Travel Med. Infect. Dis. 2020, 35, 101647. [Google Scholar] [CrossRef] [PubMed]
- Feuillet, V.; Canard, B.; Trautmann, A. Combining antivirals and immunomodulators to fight COVID-19. Trends Immunol. 2021, 42, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Wu, R.; Wang, L.; Kuo, H.-C.D.; Shannar, A.; Peter, R.; Chou, P.J.; Li, S.; Hudlikar, R.; Liu, X.; Liu, Z.; et al. An update on current therapeutic drugs treating COVID-19. Curr. Pharmacol. Rep. 2020, 6, 56–70. [Google Scholar] [CrossRef]
- Zhao, M. Cytokine storm and immunomodulatory therapy in COVID-19: Role of chloroquine and anti-il-6 monoclonal antibodies. Int. J. Antimicrob. Agents 2020, 55, 105982. [Google Scholar] [CrossRef]
- Gil, C.; Ginex, T.; Maestro, I.; Nozal, V.; Barrado-Gil, L.; Cuesta-Geijo, M.Á.; Urquiza, J.; Ramírez, D.; Alonso, C.; Campillo, N.E.; et al. COVID-19: Drug targets and potential treatments. J. Med. Chem. 2020, 63, 12359–12386. [Google Scholar] [CrossRef]
- Catanzaro, M.; Fagiani, F.; Racchi, M.; Corsini, E.; Govoni, S.; Lanni, C. Immune response in COVID-19: Addressing a pharmacological challenge by targeting pathways triggered by SARS-CoV-2. Signal Transduct. Target. Ther. 2020, 5, 84. [Google Scholar] [CrossRef]
- Faheem; Kumar, B.K.; Sekhar, K.V.G.C.; Kunjiappan, S.; Jamalis, J.; Balaña-Fouce, R.; Tekwani, B.L.; Sankaranarayanan, M. Druggable targets of SARS-CoV-2 and treatment opportunities for COVID-19. Bioorg. Chem. 2020, 104, 104269. [Google Scholar] [CrossRef]
- Poduri, R.; Joshi, G.; Jagadeesh, G. Drugs targeting various stages of the SARS-CoV-2 life cycle: Exploring promising drugs for the treatment of COVID-19. Cell. Signal. 2020, 74, 109721. [Google Scholar] [CrossRef] [PubMed]
- Saxena, A. Drug targets for COVID-19 therapeutics: Ongoing global efforts. J. Biosci. 2020, 45, 87. [Google Scholar] [CrossRef] [PubMed]
- Taefehshokr, N.; Taefehshokr, S.; Hemmat, N.; Heit, B. COVID-19: Perspectives on innate immune evasion. Front. Immunol. 2020, 11, 580641. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Petitjean, S.J.L.; Koehler, M.; Zhang, Q.; Dumitru, A.C.; Chen, W.; Derclaye, S.; Vincent, S.P.; Soumillion, P.; Alsteens, D. Molecular interaction and inhibition of SARS-CoV-2 binding to the ace2 receptor. Nat. Commun. 2020, 11, 4541. [Google Scholar] [CrossRef] [PubMed]
- Mei, M.; Tan, X. Current strategies of antiviral drug discovery for COVID-19. Front. Mol. Biosci. 2021, 8, 671263. [Google Scholar] [CrossRef]
- Arora, G.; Misra, R.; Sajid, A. Model systems for pulmonary infectious diseases: Paradigms of anthrax and tuberculosis. Curr. Top. Med. Chem. 2017, 17, 2077–2099. [Google Scholar] [CrossRef]
- Dash, S.; Shakyawar, S.K.; Sharma, M.; Kaushik, S. Big data in healthcare: Management, analysis and future prospects. J. Big Data 2019, 6, 54. [Google Scholar] [CrossRef] [Green Version]
- Emmert-Streib, F. From the digital data revolution toward a digital society: Pervasiveness of artificial intelligence. Mach. Learn. Knowl. Extr. 2021, 3, 284–298. [Google Scholar] [CrossRef]
- McGuire, M.F.; Iyengar, M.S.; Mercer, D.W. Computational approaches for translational clinical research in disease progression. J. Investig. Med. 2011, 59, 893–903. [Google Scholar] [CrossRef]
- Yousef, M.; Kumar, A.; Bakir-Gungor, B. Application of biological domain knowledge based feature selection on gene expression data. Entropy 2020, 23, 2. [Google Scholar] [CrossRef]
- Zeng, W.; Gautam, A.; Huson, D.H. On the application of advanced machine learning methods to analyze enhanced, multimodal data from persons infected with COVID-19. Computation 2021, 9, 4. [Google Scholar] [CrossRef]
- Arora, G.; Sajid, A.; Singhal, A.; Joshi, J.; Virmani, R.; Gupta, M.; Verma, N.; Maji, A.; Misra, R.; Baronian, G.; et al. Identification of ser/thr kinase and forkhead associated domains in mycobacterium ulcerans: Characterization of novel association between protein kinase q and mupfha. PLoS Negl. Trop. Dis. 2014, 8, e3315. [Google Scholar] [CrossRef] [Green Version]
- Shah, P.; Kendall, F.; Khozin, S.; Goosen, R.; Hu, J.; Laramie, J.; Ringel, M.; Schork, N. Artificial intelligence and machine learning in clinical development: A translational perspective. NPJ Digit. Med. 2019, 2, 69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dias, R.; Torkamani, A. Artificial intelligence in clinical and genomic diagnostics. Genome Med. 2019, 11, 70. [Google Scholar] [CrossRef] [Green Version]
- Deo, R.C. Machine learning in medicine. Circulation 2015, 132, 1920–1930. [Google Scholar] [CrossRef] [Green Version]
- Bertolaccini, L.; Solli, P.; Pardolesi, A.; Pasini, A. An overview of the use of artificial neural networks in lung cancer research. J. Thorac. Dis. 2017, 9, 924–931. [Google Scholar] [CrossRef] [Green Version]
- Cao, C.; Liu, F.; Tan, H.; Song, D.; Shu, W.; Li, W.; Zhou, Y.; Bo, X.; Xie, Z. Deep learning and its applications in biomedicine. Genom. Proteom. Bioinform. 2018, 16, 17–32. [Google Scholar] [CrossRef]
- Baldominos, A.; Cervantes, A.; Saez, Y.; Isasi, P. A comparison of machine learning and deep learning techniques for activity recognition using mobile devices. Sensors 2019, 19, 521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, D.; Liu, S.; Kingsbury, P.; Sohn, S.; Storlie, C.B.; Habermann, E.B.; Naessens, J.M.; Larson, D.W.; Liu, H. Deep learning and alternative learning strategies for retrospective real-world clinical data. NPJ Digit. Med. 2019, 2, 43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Davenport, T.; Kalakota, R. The potential for artificial intelligence in healthcare. Future Healthc. J. 2019, 6, 94–98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bali, J.; Garg, R.; Bali, R.T. Artificial intelligence (ai) in healthcare and biomedical research: Why a strong computational/ai bioethics framework is required? Indian J. Ophthalmol. 2019, 67, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Vamathevan, J.; Clark, D.; Czodrowski, P.; Dunham, I.; Ferran, E.; Lee, G.; Li, B.; Madabhushi, A.; Shah, P.; Spitzer, M.; et al. Applications of machine learning in drug discovery and development. Nat. Rev. Drug Discov. 2019, 18, 463–477. [Google Scholar] [CrossRef]
- Abbasi, W.A.; Abbas, S.A.; Andleeb, S. Covidx: Computer-aided diagnosis of COVID-19 and its severity prediction with raw digital chest X-ray images. arXiv 2020, arXiv:2012.13605. [Google Scholar]
- Garg, T.; Garg, M.; Mahela, O.P.; Garg, A.R. Convolutional neural networks with transfer learning for recognition of COVID-19: A comparative study of different approaches. AI 2020, 1, 586–606. [Google Scholar] [CrossRef]
- Jain, R.; Gupta, M.; Taneja, S.; Hemanth, D.J. Deep learning based detection and analysis of COVID-19 on chest X-ray images. Appl. Intell. 2020, 51, 1690–1700. [Google Scholar] [CrossRef]
- Barh, D.; Tiwari, S.; Weener, M.E.; Azevedo, V.; Góes-Neto, A.; Gromiha, M.M.; Ghosh, P. Multi-omics-based identification of SARS-CoV-2 infection biology and candidate drugs against COVID-19. Comput. Biol. Med. 2020, 126, 104051. [Google Scholar] [CrossRef]
- Ciliberto, G.; Cardone, L. Boosting the arsenal against COVID-19 through computational drug repurposing. Drug Discov. Today 2020, 25, 946–948. [Google Scholar] [CrossRef]
- Davalbhakta, S.; Advani, S.; Kumar, S.; Agarwal, V.; Bhoyar, S.; Fedirko, E.; Misra, D.P.; Goel, A.; Gupta, L.; Agarwal, V. A systematic review of smartphone applications available for corona virus disease 2019 (COVID19) and the assessment of their quality using the mobile application rating scale (MARS). J. Med. Syst. 2020, 44, 164. [Google Scholar] [CrossRef]
- Naudé, W. Artificial intelligence vs COVID-19: Limitations, constraints and pitfalls. AI Soc. 2020, 35, 761–765. [Google Scholar] [CrossRef]
- Lopez-Rincon, A.; Tonda, A.; Mendoza-Maldonado, L.; Claassen, E.; Garssen, J.; Kraneveld, A.D. Accurate identification of SARS-CoV-2 from viral genome sequences using deep learning. bioRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Khakharia, A.; Shah, V.; Jain, S.; Shah, J.; Tiwari, A.; Daphal, P.; Warang, M.; Mehendale, N. Outbreak prediction of COVID-19 for dense and populated countries using machine learning. Ann. Data Sci. 2021, 8, 1–19. [Google Scholar] [CrossRef]
- Zeng, D.; Cao, Z.; Neill, D.B. Artificial intelligence–enabled public health surveillance—From local detection to global epidemic monitoring and control. In Artificial Intelligence in Medicine; Elsevier: Amsterdam, The Netherlands, 2021; pp. 437–453. Available online: https://www.sciencedirect.com/science/article/pii/B9780128212592000223?via%3Dihub (accessed on 7 August 2021).
- Khan, K.; McNabb, S.J.N.; Memish, Z.A.; Eckhardt, R.; Hu, W.; Kossowsky, D.; Sears, J.; Arino, J.; Johansson, A.; Barbeschi, M.; et al. Infectious disease surveillance and modelling across geographic frontiers and scientific specialties. Lancet Infect. Dis. 2012, 12, 222–230. [Google Scholar] [CrossRef]
- Siettos, C.I.; Russo, L. Mathematical modeling of infectious disease dynamics. Virulence 2013, 4, 295–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malik, Y.S.; Sircar, S.; Bhat, S.; Ansari, M.I.; Pande, T.; Kumar, P.; Mathapati, B.; Balasubramanian, G.; Kaushik, R.; Natesan, S.; et al. How artificial intelligence may help the COVID-19 pandemic: Pitfalls and lessons for the future. Rev. Med. Virol. 2020, e2205. [Google Scholar] [CrossRef]
- Bogoch, I.I.; Watts, A.; Thomas-Bachli, A.; Huber, C.; Kraemer, M.U.G.; Khan, K. Pneumonia of unknown aetiology in wuhan, china: Potential for international spread via commercial air travel. J. Travel Med. 2020, 27, taaa008. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, M.; Pullano, G.; Pinotti, F.; Valdano, E.; Poletto, C.; Boelle, P.Y.; D’Ortenzio, E.; Yazdanpanah, Y.; Eholie, S.P.; Altmann, M.; et al. Preparedness and vulnerability of african countries against importations of COVID-19: A modelling study. Lancet 2020, 395, 871–877. [Google Scholar] [CrossRef] [Green Version]
- Cartaxo, A.N.S.; Barbosa, F.I.C.; de Souza Bermejo, P.H.; Moreira, M.F.; Prata, D.N. The exposure risk to COVID-19 in most affected countries: A vulnerability assessment model. PLoS ONE 2021, 16, e0248075. [Google Scholar] [CrossRef]
- Srinivasa Rao, A.S.R.; Vazquez, J.A. Identification of COVID-19 can be quicker through artificial intelligence framework using a mobile phone-based survey when cities and towns are under quarantine. Infect. Control Hosp. Epidemiol. 2020, 41, 826–830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ardabili, S.F.; Mosavi, A.; Ghamisi, P.; Ferdinand, F.; Varkonyi-Koczy, A.R.; Reuter, U.; Rabczuk, T.; Atkinson, P.M. COVID-19 outbreak prediction with machine learning. Algorithms 2020, 13, 249. [Google Scholar] [CrossRef]
- Pinter, G.; Felde, I.; Mosavi, A.; Ghamisi, P.; Gloaguen, R. COVID-19 pandemic prediction for hungary; a hybrid machine learning approach. Mathematics 2020, 8, 890. [Google Scholar] [CrossRef]
- Punn, N.S.; Sonbhadra, S.K.; Agarwal, S. COVID-19 epidemic analysis using machine learning and deep learning algorithms. medRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Hung, M.; Lauren, E.; Hon, E.S.; Birmingham, W.C.; Xu, J.; Su, S.; Hon, S.D.; Park, J.; Dang, P.; Lipsky, M.S. Social network analysis of COVID-19 sentiments: Application of artificial intelligence. J. Med. Internet Res. 2020, 22, e22590. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Ding, S.; Xiong, L. A new system for surveillance and digital contact tracing for COVID-19: Spatiotemporal reporting over network and gps. JMIR Mhealth Uhealth 2020, 8, e19457. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.A.; Luo, Z.; Watkins, C. Epidemic contact tracing with smartphone sensors. arXiv 2020, arXiv:2006.00046. [Google Scholar] [CrossRef]
- Cresswell, K.; Tahir, A.; Sheikh, Z.; Hussain, Z.; Domínguez Hernández, A.; Harrison, E.; Williams, R.; Sheikh, A.; Hussain, A. Understanding public perceptions of COVID-19 contact tracing apps: Artificial intelligence–enabled social media analysis. J. Med. Internet Res. 2021, 23, e26618. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Hou, S.; Fan, Y.; Zhang, Y.; Qian, Y.; Sun, S.; Peng, Q.; Ju, M.; Song, W.; Loparo, K. A-satellite: An ai-driven system and benchmark datasets for dynamic COVID-19 risk assessment in the united states. IEEE J. Biomed. Health Inform. 2020, 24, 2755–2764. [Google Scholar] [CrossRef]
- Tuli, S.; Tuli, S.; Tuli, R.; Gill, S.S. Predicting the growth and trend of COVID-19 pandemic using machine learning and cloud computing. Internet Things 2020, 11, 100222. [Google Scholar] [CrossRef]
- Atta-ur-Rahman; Sultan, K.; Naseer, I.; Majeed, R.; Musleh, D.; Gollapalli, M.-A.-S.; Chabani, S.; Ibrahim, N.; Siddiqui, S.-Y.; Khan, M.-A. Supervised machine learning-based prediction of COVID-19. Comput. Mater. Contin. 2021, 69, 21–34. [Google Scholar] [CrossRef]
- Bobrow, B.J.; Panczyk, M.J.; Villa, N.W. Emergency medicine: The finest hour in our time. J. Am. Coll. Emerg. Physicians Open 2020, 1, 242–243. [Google Scholar] [CrossRef] [PubMed]
- Tagashira, Y.; Takamatsu, A.; Hasegawa, S.; Uenoyama, Y.; Honda, H. A survey of preparedness against coronavirus disease 2019 (COVID-19) in hospitals in tokyo, japan, with healthcare personnel with COVID-19 and in-facility transmission. Infect. Control Hosp. Epidemiol. 2021, 42, 746–750. [Google Scholar] [CrossRef]
- Hosny, A.; Parmar, C.; Quackenbush, J.; Schwartz, L.H.; Aerts, H.J.W.L. Artificial intelligence in radiology. Nat. Rev. Cancer 2018, 18, 500–510. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Che, Q.; Ji, X.; Meng, X.; Zhang, L.; Jia, R.; Lyu, H.; Bai, W.; Tan, L.; Gao, Y. Correlation between lung infection severity and clinical laboratory indicators in patients with COVID-19: A cross-sectional study based on machine learning. BMC Infect. Dis. 2021, 21, 192. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.; Chen, W.; Cao, Y.; Xu, Z.; Tan, Z.; Zhang, X.; Deng, L.; Zheng, C.; Zhou, J.; Shi, H.; et al. Development and evaluation of an artificial intelligence system for COVID-19 diagnosis. Nat. Commun. 2020, 11, 5088. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lin, Z.Q.; Wong, A. Covid-net: A tailored deep convolutional neural network design for detection of COVID-19 cases from chest X-ray images. Sci. Rep. 2020, 10, 19549. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, R.; Nishio, M.; Do, R.K.G.; Togashi, K. Convolutional neural networks: An overview and application in radiology. Insights Imaging 2018, 9, 611–629. [Google Scholar] [CrossRef] [Green Version]
- Ghaderzadeh, M.; Asadi, F. Deep learning in the detection and diagnosis of COVID-19 using radiology modalities: A systematic review. J. Healthc. Eng. 2021, 2021, 6677314. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zha, Y.; Li, W.; Wu, Q.; Li, X.; Niu, M.; Wang, M.; Qiu, X.; Li, H.; Yu, H.; et al. A fully automatic deep learning system for COVID-19 diagnostic and prognostic analysis. Eur. Respir. J. 2020, 56, 2000775. [Google Scholar] [CrossRef]
- Mei, X.; Lee, H.-C.; Diao, K.-Y.; Huang, M.; Lin, B.; Liu, C.; Xie, Z.; Ma, Y.; Robson, P.M.; Chung, M.; et al. Artificial intelligence-enabled rapid diagnosis of patients with COVID-19. Nat. Med. 2020, 26, 1224–1228. [Google Scholar] [CrossRef]
- Gomes, J.C.; Barbosa, V.A.d.F.; de Santana, M.A.; Bandeira, J.; Valenca, M.J.S.; de Souza, R.E.; Ismael, A.M.; dos Santos, W.P. Ikonos: An intelligent tool to support diagnosis of COVID-19 by texture analysis of x-ray images. Res. Biomed. Eng. 2020, 1–14. [Google Scholar] [CrossRef]
- Belfiore, M.P.; Urraro, F.; Grassi, R.; Giacobbe, G.; Patelli, G.; Cappabianca, S.; Reginelli, A. Artificial intelligence to codify lung ct in COVID-19 patients. Radiol. Med. 2020, 125, 500–504. [Google Scholar] [CrossRef]
- Pan, Y.; Li, X.; Yang, G.; Fan, J.; Tang, Y.; Zhao, J.; Long, X.; Guo, S.; Zhao, Z.; Liu, Y.; et al. Serological immunochromatographic approach in diagnosis with SARS-CoV-2 infected COVID-19 patients. J. Infect. 2020, 81, e28–e32. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.-C.; Chen, J.; Zhang, J.; Lin, J.; Yan, S.; Zhang, S.; Li, X.; Liang, C.; Peng, C.; Lin, Q.; et al. Early cancer detection from multianalyte blood test results. iScience 2019, 15, 332–341. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Syed-Abdul, S.; Firdani, R.-P.; Chung, H.-J.; Uddin, M.; Hur, M.; Park, J.H.; Kim, H.W.; Gradišek, A.; Dovgan, E. Artificial intelligence based models for screening of hematologic malignancies using cell population data. Sci. Rep. 2020, 10, 4583. [Google Scholar] [CrossRef]
- de Freitas Barbosa, V.A.; Gomes, J.C.; de Santana, M.A.; Albuquerque, J.E.d.A.; de Souza, R.G.; de Souza, R.E.; dos Santos, W.P. Heg.IA: An intelligent system to support diagnosis of COVID-19 based on blood tests. Res. Biomed. Eng. 2021, 1–18. [Google Scholar] [CrossRef]
- Alsheref, F.K.; Hassan, W. Blood diseases detection using classical machine learning algorithms. Int. J. Adv. Comput. Sci. Appl. 2019, 10, 77–81. [Google Scholar] [CrossRef] [Green Version]
- Park, D.J.; Park, M.W.; Lee, H.; Kim, Y.-J.; Kim, Y.; Park, Y.H. Development of machine learning model for diagnostic disease prediction based on laboratory tests. Sci. Rep. 2021, 11, 7567. [Google Scholar] [CrossRef]
- Karahan, O.; Yavuz, C.; Kankilic, N.; Demirtas, S.; Tezcan, O.; Caliskan, A.; Mavitas, B. Simple blood tests as predictive markers of disease severity and clinical condition in patients with venous insufficiency. Blood Coagul. Fibrinolysis 2016, 27, 684–690. [Google Scholar] [CrossRef]
- Zeng, F.; Li, L.; Zeng, J.; Deng, Y.; Huang, H.; Chen, B.; Deng, G. Can we predict the severity of coronavirus disease 2019 with a routine blood test? Pol. Arch. Intern. Med. 2020, 130, 400–406. [Google Scholar]
- AlJame, M.; Ahmad, I.; Imtiaz, A.; Mohammed, A. Ensemble learning model for diagnosing COVID-19 from routine blood tests. Inform. Med. Unlocked 2020, 21, 100449. [Google Scholar] [CrossRef]
- Brinati, D.; Campagner, A.; Ferrari, D.; Locatelli, M.; Banfi, G.; Cabitza, F. Detection of COVID-19 infection from routine blood exams with machine learning: A feasibility study. J. Med. Syst. 2020, 44, 135. [Google Scholar] [CrossRef]
- Banerjee, A.; Ray, S.; Vorselaars, B.; Kitson, J.; Mamalakis, M.; Weeks, S.; Baker, M.; Mackenzie, L.S. Use of machine learning and artificial intelligence to predict SARS-CoV-2 infection from full blood counts in a population. Int. Immunopharmacol. 2020, 86, 106705. [Google Scholar] [CrossRef] [PubMed]
- Ko, H.; Chung, H.; Kang, W.S.; Park, C.; Kim, D.W.; Kim, S.E.; Chung, C.R.; Ko, R.E.; Lee, H.; Seo, J.H.; et al. An artificial intelligence model to predict the mortality of COVID-19 patients at hospital admission time using routine blood samples: Development and validation of an ensemble model. J. Med. Internet Res. 2020, 22, e25442. [Google Scholar] [CrossRef]
- Chary, M.; Parikh, S.; Manini, A.F.; Boyer, E.W.; Radeos, M. A review of natural language processing in medical education. West J. Emerg. Med. 2019, 20, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Dreisbach, C.; Koleck, T.A.; Bourne, P.E.; Bakken, S. A systematic review of natural language processing and text mining of symptoms from electronic patient-authored text data. Int. J. Med. Inform. 2019, 125, 37–46. [Google Scholar] [CrossRef]
- Khanday, A.M.U.D.; Rabani, S.T.; Khan, Q.R.; Rouf, N.; Mohi Ud Din, M. Machine learning based approaches for detecting COVID-19 using clinical text data. Int. J. Inf. Technol. 2020, 12, 731–739. [Google Scholar] [CrossRef]
- Imran, A.; Posokhova, I.; Qureshi, H.N.; Masood, U.; Riaz, M.S.; Ali, K.; John, C.N.; Hussain, M.I.; Nabeel, M. Ai4COVID-19: AI enabled preliminary diagnosis for COVID-19 from cough samples via an app. Inform. Med. Unlocked 2020, 20, 100378. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Rophina, M.; Mahajan, S.; Krishnan, B.B.; Sharma, M.; Mandal, S.; Fernandez, T.; Sultanji, S.; Jolly, B.; Mathew, S.; et al. Analysis of the potential impact of genomic variants in global SARS-CoV-2 genomes on molecular diagnostic assays. Int. J. Infect. Dis. 2021, 102, 460–462. [Google Scholar] [CrossRef] [PubMed]
- Pal, R.; Chopra, H.; Awasthi, R.; Bandhey, H.; Nagori, A.; Gulati, A.; Kumaraguru, P.; Sethi, T. Predicting emerging themes in rapidly expanding COVID-19 literature with dynamic word embedding networks and machine learning. medRxiv 2021. [Google Scholar] [CrossRef]
- Heldt, F.S.; Vizcaychipi, M.P.; Peacock, S.; Cinelli, M.; McLachlan, L.; Andreotti, F.; Jovanović, S.; Dürichen, R.; Lipunova, N.; Fletcher, R.A.; et al. Early risk assessment for COVID-19 patients from emergency department data using machine learning. Sci. Rep. 2021, 11, 4200. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.; Liu, Z.; Jiang, Y.; Shi, O.; Zhang, X.; Xu, K.; Suo, C.; Wang, Q.; Song, Y.; Yu, K.; et al. Early prediction of mortality risk among patients with severe COVID-19, using machine learning. Int. J. Epidemiol. 2021, 49, 1918–1929. [Google Scholar] [CrossRef]
- Yan, L.; Zhang, H.-T.; Goncalves, J.; Xiao, Y.; Wang, M.; Guo, Y.; Sun, C.; Tang, X.; Jing, L.; Zhang, M.; et al. An interpretable mortality prediction model for COVID-19 patients. Nat. Mach. Intell. 2020, 2, 283–288. [Google Scholar] [CrossRef]
- Wu, G.; Zhou, S.; Wang, Y.; Lv, W.; Wang, S.; Wang, T.; Li, X. A prediction model of outcome of SARS-CoV-2 pneumonia based on laboratory findings. Sci. Rep. 2020, 10, 14042. [Google Scholar] [CrossRef]
- Chowdhury, M.E.H.; Rahman, T.; Khandakar, A.; Al-Madeed, S.; Zughaier, S.M.; Doi, S.A.R.; Hassen, H.; Islam, M.T. An early warning tool for predicting mortality risk of COVID-19 patients using machine learning. Cognit. Comput. 2021, 1–16. [Google Scholar] [CrossRef]
- van der Schaar, M.; Alaa, A.M.; Floto, A.; Gimson, A.; Scholtes, S.; Wood, A.; McKinney, E.; Jarrett, D.; Lio, P.; Ercole, A. How artificial intelligence and machine learning can help healthcare systems respond to COVID-19. Mach. Learn. 2020, 110, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Iwendi, C.; Bashir, A.K.; Peshkar, A.; Sujatha, R.; Chatterjee, J.M.; Pasupuleti, S.; Mishra, R.; Pillai, S.; Jo, O. COVID-19 patient health prediction using boosted random forest algorithm. Front. Public Health 2020, 8, 357. [Google Scholar] [CrossRef] [PubMed]
- Souza, F.S.H.; Hojo-Souza, N.S.; Santos, E.B.; Silva, C.M.; Guidoni, D.L. Predicting the disease outcome in COVID-19 positive patients through machine learning: A retrospective cohort study with brazilian data. medRxiv 2020. [Google Scholar] [CrossRef]
- Burdick, H.; Lam, C.; Mataraso, S.; Siefkas, A.; Braden, G.; Dellinger, R.P.; McCoy, A.; Vincent, J.-L.; Green-Saxena, A.; Barnes, G.; et al. Prediction of respiratory decompensation in COVID-19 patients using machine learning: The ready trial. Comput. Biol. Med. 2020, 124, 103949. [Google Scholar] [CrossRef]
- Yadaw, A.S.; Li, Y.-C.; Bose, S.; Iyengar, R.; Bunyavanich, S.; Pandey, G. Clinical features of COVID-19 mortality: Development and validation of a clinical prediction model. Lancet Digit. Health 2020, 2, e516–e525. [Google Scholar] [CrossRef]
- Ingraham, N.E.; Lotfi-Emran, S.; Thielen, B.K.; Techar, K.; Morris, R.S.; Holtan, S.G.; Dudley, R.A.; Tignanelli, C.J. Immunomodulation in COVID-19. Lancet Respir. Med. 2020, 8, 544–546. [Google Scholar] [CrossRef]
- Şimşek Yavuz, S.; Ünal, S. Antiviral treatment ofCOVID-19. Turk. J. Med. Sci. 2020, 50, 611–619. [Google Scholar] [CrossRef]
- Sanders, J.M.; Monogue, M.L.; Jodlowski, T.Z.; Cutrell, J.B. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): A review. JAMA 2020, 323, 1824–1836. [Google Scholar] [CrossRef] [PubMed]
- Parvathaneni, V.; Gupta, V. Utilizing drug repurposing against COVID-19—Efficacy, limitations, and challenges. Life Sci. 2020, 259, 118275. [Google Scholar] [CrossRef]
- Benke, K.; Benke, G. Artificial intelligence and big data in public health. Int. J. Environ. Res. Public Health 2018, 15, 2796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sanchez-Pinto, L.N.; Luo, Y.; Churpek, M.M. Big data and data science in critical care. Chest 2018, 154, 1239–1248. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Srivastava, D.; Sahu, M.; Tiwari, S.; Ambasta, R.K.; Kumar, P. Artificial intelligence to deep learning: Machine intelligence approach for drug discovery. Mol. Divers. 2021, 25, 1315–1360. [Google Scholar] [CrossRef]
- Paul, D.; Sanap, G.; Shenoy, S.; Kalyane, D.; Kalia, K.; Tekade, R.K. Artificial intelligence in drug discovery and development. Drug Discov. Today 2021, 26, 80–93. [Google Scholar] [CrossRef]
- Mohapatra, S.; Nath, P.; Chatterjee, M.; Das, N.; Kalita, D.; Roy, P.; Satapathi, S. Repurposing therapeutics for COVID-19: Rapid prediction of commercially available drugs through machine learning and docking. PLoS ONE 2020, 15, e0241543. [Google Scholar] [CrossRef]
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; Doig, A.; Guilliams, T.; Latimer, J.; McNamee, C.; et al. Drug repurposing: Progress, challenges and recommendations. Nat. Rev. Drug Discov. 2019, 18, 41–58. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, F.; Tang, J.; Nussinov, R.; Cheng, F. Artificial intelligence in COVID-19 drug repurposing. Lancet Digit. Health 2020, 2, e667–e676. [Google Scholar] [CrossRef]
- Mohanty, S.; Harun Ai Rashid, M.; Mridul, M.; Mohanty, C.; Swayamsiddha, S. Application of artificial intelligence in COVID-19 drug repurposing. Diabetes Metab. Syndr. 2020, 14, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Zeng, X.; Zhu, S.; Hou, Y.; Zhang, P.; Li, L.; Li, J.; Huang, L.F.; Lewis, S.J.; Nussinov, R.; Cheng, F. Network-based prediction of drug-target interactions using an arbitrary-order proximity embedded deep forest. Bioinformatics 2020, 36, 2805–2812. [Google Scholar] [CrossRef]
- Beck, B.R.; Shin, B.; Choi, Y.; Park, S.; Kang, K. Predicting commercially available antiviral drugs that may act on the novel coronavirus (2019-ncov), wuhan, china through a drug-target interaction deep learning model. Comput. Struct. Biotechnol. J. 2020, 18, 784–790. [Google Scholar] [CrossRef] [PubMed]
- Pham, T.-H.; Qiu, Y.; Zeng, J.; Xie, L.; Zhang, P. A deep learning framework for high-throughput mechanism-driven phenotype compound screening and its application to COVID-19 drug repurposing. Nat. Mach. Intell. 2021, 3, 247–257. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Choi, A.S.; Nam, H. Drug repositioning of herbal compounds via a machine-learning approach. BMC Bioinform. 2019, 20, 247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chakraborty, R.; Parvez, S. COVID-19: An overview of the current pharmacological interventions, vaccines, and clinical trials. Biochem. Pharmacol. 2020, 180, 114184. [Google Scholar] [CrossRef]
- Zhu, Y.; Li, J.; Pang, Z. Recent insights for the emerging COVID-19: Drug discovery, therapeutic options and vaccine development. Asian J. Pharm. Sci. 2021, 16, 4–23. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Luo, X.; Qiu, H.; Mackey, V.; Sun, L.; Ouyang, X. Drug discovery and drug marketing with the critical roles of modern administration. Am. J. Transl. Res. 2018, 10, 4302–4312. [Google Scholar] [PubMed]
- Yildirim, O.; Gottwald, M.; Schüler, P.; Michel, M.C. Opportunities and challenges for drug development: Public-private partnerships, adaptive designs and big data. Front. Pharmacol. 2016, 7, 461. [Google Scholar] [CrossRef] [Green Version]
- Singh, S.B.; Young, K.; Silver, L.L. What is an ”ideal” antibiotic? Discovery challenges and path forward. Biochem. Pharmacol. 2017, 133, 63–73. [Google Scholar] [CrossRef]
- Lo, Y.-C.; Rensi, S.E.; Torng, W.; Altman, R.B. Machine learning in chemoinformatics and drug discovery. Drug Discov. Today 2018, 23, 1538–1546. [Google Scholar] [CrossRef]
- Carpenter, K.A.; Cohen, D.S.; Jarrell, J.T.; Huang, X. Deep learning and virtual drug screening. Future Med. Chem. 2018, 10, 2557–2567. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carpenter, K.A.; Huang, X. Machine learning-based virtual screening and its applications to alzheimer’s drug discovery: A review. Curr. Pharm. Des. 2018, 24, 3347–3358. [Google Scholar] [CrossRef]
- Zhang, H.; Saravanan, K.M.; Yang, Y.; Hossain, M.T.; Li, J.; Ren, X.; Pan, Y.; Wei, Y. Deep learning based drug screening for novel coronavirus 2019-ncov. Interdiscip. Sci. 2020, 12, 368–376. [Google Scholar] [CrossRef]
- Xu, Z.; Yang, L.; Zhang, X.; Zhang, Q.; Yang, Z.; Liu, Y.; Wei, S.; Liu, W. Discovery of potential flavonoid inhibitors against COVID-19 3cl proteinase based on virtual screening strategy. Front. Mol. Biosci. 2020, 7, 556481. [Google Scholar] [CrossRef] [PubMed]
- Kabra, R.; Singh, S. Evolutionary artificial intelligence based peptide discoveries for effective COVID-19 therapeutics. Biochim. Biophys. Acta Mol. Basis Dis. 2021, 1867, 165978. [Google Scholar] [CrossRef]
- Dhiman, R.; Singh, R. Recent advances for identification of new scaffolds and drug targets for mycobacterium tuberculosis. IUBMB Life 2018, 70, 905–916. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rana, A.; Thakur, S.; Kumar, G.; Akhter, Y. Recent trends in system-scale integrative approaches for discovering protective antigens against mycobacterial pathogens. Front. Genet. 2018, 9, 572. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keshavarzi Arshadi, A.; Salem, M.; Collins, J.; Yuan, J.S.; Chakrabarti, D. Deepmalaria: Artificial intelligence driven discovery of potent antiplasmodials. Front. Pharmacol. 2019, 10, 1526. [Google Scholar] [CrossRef] [Green Version]
- Winkler, D.A. Use of artificial intelligence and machine learning for discovery of drugs for neglected tropical diseases. Front. Chem. 2021, 9, 614073. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, Z.; Mohamed, K.; Zeeshan, S.; Dong, X. Artificial intelligence with multi-functional machine learning platform development for better healthcare and precision medicine. Database 2020, 2020, baaa010. [Google Scholar] [CrossRef]
- Toh, T.S.; Dondelinger, F.; Wang, D. Looking beyond the hype: Applied ai and machine learning in translational medicine. EBioMedicine 2019, 47, 607–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Naseem, M.; Akhund, R.; Arshad, H.; Ibrahim, M.T. Exploring the potential of artificial intelligence and machine learning to combat COVID-19 and existing opportunities for lmic: A scoping review. J. Prim. Care Community Health 2020, 11, 2150132720963634. [Google Scholar] [CrossRef] [PubMed]
- Vaishya, R.; Javaid, M.; Khan, I.H.; Haleem, A. Artificial intelligence (ai) applications for COVID-19 pandemic. Diabetes Metab. Syndr. 2020, 14, 337–339. [Google Scholar] [CrossRef] [PubMed]
- Dai, L.; Gao, G.F. Viral targets for vaccines against COVID-19. Nat. Rev. Immunol. 2020, 21, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Dai, T.; Wei, Y.; Zhang, L.; Zheng, M.; Zhou, F. A systematic review of SARS-CoV-2 vaccine candidates. Signal Transduct. Target. Ther. 2020, 5, 237. [Google Scholar] [CrossRef] [PubMed]
- Ong, E.; Wong, M.U.; Huffman, A.; He, Y. COVID-19 coronavirus vaccine design using reverse vaccinology and machine learning. Front. Immunol. 2020, 11, 1581. [Google Scholar] [CrossRef]
- Jia, Q.; Bielefeldt-Ohmann, H.; Maison, R.M.; Maslesa-Galic, S.; Cooper, S.K.; Bowen, R.A.; Horwitz, M.A. Replicating bacterium-vectored vaccine expressing SARS-CoV-2 membrane and nucleocapsid proteins protects against severe COVID-19-like disease in hamsters. NPJ Vaccines 2021, 6, 47. [Google Scholar] [CrossRef] [PubMed]
- Bagcchi, S. The world’s largest COVID-19 vaccination campaign. Lancet Infect. Dis. 2021, 21, 323. [Google Scholar] [CrossRef]
- Callaway, E.; Ledford, H. How to redesign Covid vaccines so they protect against variants. Nature 2021, 590, 15–16. [Google Scholar] [CrossRef]
- Livingston, E.H.; Malani, P.N.; Creech, C.B. The Johnson & Johnson vaccine for COVID-19. JAMA 2021, 325, 1575. [Google Scholar]
- Madkaikar, M.; Gupta, N.; Yadav, R.M.; Bargir, U.A. India’s crusade against COVID-19. Nat. Immunol. 2021, 22, 258–259. [Google Scholar] [CrossRef]
- McMurry, R.; Lenehan, P.; Awasthi, S.; Silvert, E.; Puranik, A.; Pawlowski, C.; Venkatakrishnan, A.J.; Anand, P.; Agarwal, V.; O’Horo, J.C.; et al. Real-time analysis of a mass vaccination effort confirms the safety of fda-authorized mrna vaccines for COVID-19 from Moderna and Pfizer/Biontech. medRxiv 2021. [Google Scholar] [CrossRef]
- Remmel, A. Covid vaccines and safety: What the research says. Nature 2021, 590, 538–540. [Google Scholar] [CrossRef] [PubMed]
- Wise, J. COVID-19: Pfizer biontech vaccine reduced cases by 94% in israel, shows peer reviewed study. BMJ 2021, 372, n567. [Google Scholar] [CrossRef] [PubMed]
- Rawat, K.; Kumari, P.; Saha, L. COVID-19 vaccine: A recent update in pipeline vaccines, their design and development strategies. Eur. J. Pharmacol. 2021, 892, 173751. [Google Scholar] [CrossRef] [PubMed]
- Polack, F.P.; Thomas, S.J.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Perez, J.L.; Pérez Marc, G.; Moreira, E.D.; Zerbini, C.; et al. Safety and efficacy of the bnt162b2 mrna COVID-19 vaccine. N. Engl. J. Med. 2020, 383, 2603–2615. [Google Scholar] [CrossRef]
- Risma, K.A.; Edwards, K.M.; Hummell, D.S.; Little, F.F.; Norton, A.E.; Stallings, A.; Wood, R.A.; Milner, J.D. Potential mechanisms of anaphylaxis to COVID-19 mrna vaccines. J. Allergy Clin. Immunol. 2021, 147, 2075–2082.e2. [Google Scholar] [CrossRef]
- Banerji, A.; Wickner, P.G.; Saff, R.; Stone, C.A., Jr.; Robinson, L.B.; Long, A.A.; Wolfson, A.R.; Williams, P.; Khan, D.A.; Phillips, E.; et al. Mrna vaccines to prevent COVID-19 disease and reported allergic reactions: Current evidence and suggested approach. J. Allergy Clin. Immunol. Pract. 2021, 9, 1423–1437. [Google Scholar] [CrossRef]
- Chen, B.; Khodadoust, M.S.; Olsson, N.; Wagar, L.E.; Fast, E.; Liu, C.L.; Muftuoglu, Y.; Sworder, B.J.; Diehn, M.; Levy, R.; et al. Predicting hla class ii antigen presentation through integrated deep learning. Nat. Biotechnol. 2019, 37, 1332–1343. [Google Scholar] [CrossRef]
- Moore, T.V.; Nishimura, M.I. Improved mhc ii epitope prediction—A step towards personalized medicine. Nat. Rev. Clin. Oncol. 2020, 17, 71–72. [Google Scholar] [CrossRef]
- Racle, J.; Michaux, J.; Rockinger, G.A.; Arnaud, M.; Bobisse, S.; Chong, C.; Guillaume, P.; Coukos, G.; Harari, A.; Jandus, C.; et al. Robust prediction of hla class ii epitopes by deep motif deconvolution of immunopeptidomes. Nat. Biotechnol. 2019, 37, 1283–1286. [Google Scholar] [CrossRef]
- Agerer, B.; Koblischke, M.; Gudipati, V.; Montaño-Gutierrez, L.F.; Smyth, M.; Popa, A.; Genger, J.-W.; Endler, L.; Florian, D.M.; Mühlgrabner, V.; et al. SARS-CoV-2 mutations in mhc-i-restricted epitopes evade CD8+ T cell responses. Sci. Immunol. 2021, 6, eabg6461. [Google Scholar] [CrossRef]
- Castro, A.; Ozturk, K.; Zanetti, M.; Carter, H. In silico analysis suggests less effective mhc-ii presentation of SARS-CoV-2 rbm peptides: Implication for neutralizing antibody responses. PLoS ONE 2021, 16, e0246731. [Google Scholar] [CrossRef] [PubMed]
- Prachar, M.; Justesen, S.; Steen-Jensen, D.B.; Thorgrimsen, S.; Jurgons, E.; Winther, O.; Bagger, F.O. Identification and validation of 174 COVID-19 vaccine candidate epitopes reveals low performance of common epitope prediction tools. Sci. Rep. 2020, 10, 20465. [Google Scholar] [CrossRef]
- He, Y.; Xiang, Z.; Mobley, H.L.T. Vaxign: The first web-based vaccine design program for reverse vaccinology and applications for vaccine development. J. Biomed. Biotechnol. 2010, 2010, 297505. [Google Scholar] [CrossRef] [PubMed]
- Keshavarzi Arshadi, A.; Webb, J.; Salem, M.; Cruz, E.; Calad-Thomson, S.; Ghadirian, N.; Collins, J.; Diez-Cecilia, E.; Kelly, B.; Goodarzi, H.; et al. Artificial intelligence for COVID-19 drug discovery and vaccine development. Front. Artif. Intell. 2020, 3, 65. [Google Scholar] [CrossRef]
- Liu, G.; Carter, B.; Bricken, T.; Jain, S.; Viard, M.; Carrington, M.; Gifford, D.K. Computationally optimized SARS-CoV-2 mhc class i and ii vaccine formulations predicted to target human haplotype distributions. Cell Syst. 2020, 11, 131–144.e6. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Carter, B.; Gifford, D.K. Predicted cellular immunity population coverage gaps for SARS-CoV-2 subunit vaccines and their augmentation by compact peptide sets. Cell Syst. 2021, 12, 102–107.e4. [Google Scholar] [CrossRef] [PubMed]
- Asgary, A.; Valtchev, S.Z.; Chen, M.; Najafabadi, M.M.; Wu, J. Artificial intelligence model of drive-through vaccination simulation. Int. J. Environ. Res. Public Health 2021, 18, 268. [Google Scholar] [CrossRef] [PubMed]
- Bharadwaj, K.K.; Srivastava, A.; Panda, M.K.; Singh, Y.D.; Maharana, R.; Mandal, K.; Manisha Singh, B.S.; Singh, D.; Das, M.; Murmu, D.; et al. Computational intelligence in vaccine design against COVID-19. In Computational Intelligence Methods in COVID-19: Surveillance, Prevention, Prediction and Diagnosis; Raza, K., Ed.; Springer: Singapore, 2021; Volume 923, pp. 311–329. [Google Scholar]
- Chen, T.; Wang, Y.-C.; Wu, H.-C. Analyzing the impact of vaccine availability on alternative supplier selection amid the COVID-19 pandemic: A cfgm-ftopsis-fwi approach. Healthcare 2021, 9, 71. [Google Scholar] [CrossRef]
- De Maio, N.; Walker, C.R.; Turakhia, Y.; Lanfear, R.; Corbett-Detig, R.; Goldman, N. Mutation rates and selection on synonymous mutations in SARS-CoV-2. Genome Biol. Evol. 2021, 13, evab087. [Google Scholar] [CrossRef]
- Adly, A.S.; Adly, A.S.; Adly, M.S. Approaches based on artificial intelligence and the internet of intelligent things to prevent the spread of COVID-19: Scoping review. J. Med. Internet Res. 2020, 22, e19104. [Google Scholar] [CrossRef]
- Hie, B.; Zhong, E.D.; Berger, B.; Bryson, B. Learning the language of viral evolution and escape. Science 2021, 371, 284–288. [Google Scholar] [CrossRef] [PubMed]
- Salama, M.A.; Hassanien, A.E.; Mostafa, A. The prediction of virus mutation using neural networks and rough set techniques. EURASIP J. Bioinform. Syst. Biol. 2016, 2016, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malone, B.; Simovski, B.; Moliné, C.; Cheng, J.; Gheorghe, M.; Fontenelle, H.; Vardaxis, I.; Tennøe, S.; Malmberg, J.-A.; Stratford, R.; et al. Artificial intelligence predicts the immunogenic landscape of SARS-CoV-2 leading to universal blueprints for vaccine designs. Sci. Rep. 2020, 10, 22375. [Google Scholar] [CrossRef] [PubMed]
- Haimed, A.M.A.; Saba, T.; Albasha, A.; Rehman, A.; Kolivand, M. Viral reverse engineering using artificial intelligence and big data COVID-19 infection with long short-term memory (lstm). Environ. Technol. Innov. 2021, 22, 101531. [Google Scholar] [CrossRef]
- Bansal, A.; Padappayil, R.P.; Garg, C.; Singal, A.; Gupta, M.; Klein, A. Utility of artificial intelligence amidst the Covid 19 pandemic: A review. J. Med. Syst. 2020, 44, 156. [Google Scholar] [CrossRef]
- Philomina, J.B.; Jolly, B.; John, N.; Bhoyar, R.C.; Majeed, N.; Senthivel, V.; Cp, F.; Rophina, M.; Vasudevan, B.; Imran, M.; et al. Genomic survey of SARS-CoV-2 vaccine breakthrough infections in healthcare workers from Kerala, India. J. Infect. 2021, 83, 237–279. [Google Scholar] [CrossRef]
- Rodríguez-González, A.; Zanin, M.; Menasalvas-Ruiz, E. Public health and epidemiology informatics: Can artificial intelligence help future global challenges? An overview of antimicrobial resistance and impact of climate change in disease epidemiology. Yearb. Med. Inform. 2019, 28, 224–231. [Google Scholar] [CrossRef] [Green Version]
- Beil, M.; Proft, I.; van Heerden, D.; Sviri, S.; van Heerden, P.V. Ethical considerations about artificial intelligence for prognostication in intensive care. Intensive Care Med. Exp. 2019, 7, 70. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dexter, G.P.; Grannis, S.J.; Dixon, B.E.; Kasthurirathne, S.N. Generalization of machine learning approaches to identify notifiable conditions from a statewide health information exchange. AMIA Summits Transl. Sci. 2020, 2020, 152–161. [Google Scholar]
- Wang, F.; Preininger, A. AI in health: State of the art, challenges, and future directions. Yearb. Med. Inform. 2019, 28, 16–26. [Google Scholar] [CrossRef] [Green Version]
- Futoma, J.; Simons, M.; Panch, T.; Doshi-Velez, F.; Celi, L.A. The myth of generalisability in clinical research and machine learning in health care. Lancet Digit. Health 2020, 2, e489–e492. [Google Scholar] [CrossRef]
- Kuziemski, M.; Misuraca, G. Ai governance in the public sector: Three tales from the frontiers of automated decision-making in democratic settings. Telecomm. Policy 2020, 44, 101976. [Google Scholar] [CrossRef]
- Gerke, S.; Minssen, T.; Cohen, G. Ethical and legal challenges of artificial intelligence-driven healthcare. In Artificial Intelligence in Healthcare; Elsevier: Amsterdam, The Netherlands, 2020; pp. 295–336. [Google Scholar]
- Goodman, K.; Zandi, D.; Reis, A.; Vayena, E. Balancing risks and benefits of artificial intelligence in the health sector. Bull. World Health Organ. 2020, 98, 230–230A. [Google Scholar] [CrossRef] [PubMed]
- Cath, C. Governing artificial intelligence: Ethical, legal and technical opportunities and challenges. Philos. Transact. A Math. Phys. Eng. Sci. 2018, 376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lalmuanawma, S.; Hussain, J.; Chhakchhuak, L. Applications of machine learning and artificial intelligence for COVID-19 (SARS-CoV-2) pandemic: A review. Chaos Solitons Fractals 2020, 139, 110059. [Google Scholar] [CrossRef] [PubMed]
- Alimadadi, A.; Aryal, S.; Manandhar, I.; Munroe, P.B.; Joe, B.; Cheng, X. Artificial intelligence and machine learning to fight COVID-19. Physiol. Genom. 2020, 52, 200–202. [Google Scholar] [CrossRef]
- Gutierrez, G. Artificial intelligence in the intensive care unit. Crit. Care 2020, 24, 101. [Google Scholar] [CrossRef] [Green Version]
- McRae, M.P.; Dapkins, I.P.; Sharif, I.; Anderman, J.; Fenyo, D.; Sinokrot, O.; Kang, S.K.; Christodoulides, N.J.; Vurmaz, D.; Simmons, G.W.; et al. Managing COVID-19 with a clinical decision support tool in a community health network: Algorithm development and validation. J. Med. Internet Res. 2020, 22, e22033. [Google Scholar] [CrossRef]
- Abir, S.M.A.A.; Islam, S.N.; Anwar, A.; Mahmood, A.N.; Oo, A.M.T. Building resilience against COVID-19 pandemic using artificial intelligence, machine learning, and IoT: A survey of recent progress. IoT 2020, 1, 506–528. [Google Scholar] [CrossRef]
- Albahri, O.S.; Zaidan, A.A.; Albahri, A.S.; Zaidan, B.B.; Abdulkareem, K.H.; Al-Qaysi, Z.T.; Alamoodi, A.H.; Aleesa, A.M.; Chyad, M.A.; Alesa, R.M.; et al. Systematic review of artificial intelligence techniques in the detection and classification of COVID-19 medical images in terms of evaluation and benchmarking: Taxonomy analysis, challenges, future solutions and methodological aspects. J. Infect. Public Health 2020, 13, 1381–1396. [Google Scholar] [CrossRef] [PubMed]
- Bachtiger, P.; Peters, N.S.; Walsh, S.L. Machine learning for COVID-19-asking the right questions. Lancet Digit. Health 2020, 2, e391–e392. [Google Scholar] [CrossRef]
- Bullock, J.; Luccioni, A.; Hoffman Pham, K.; Sin Nga Lam, C.; Luengo-Oroz, M. Mapping the landscape of artificial intelligence applications against COVID-19. J. Artif. Intell. Res. 2020, 69, 807–845. [Google Scholar] [CrossRef]
- Kumar, A.; Gupta, P.K.; Srivastava, A. A review of modern technologies for tackling COVID-19 pandemic. Diabetes Metab. Syndr. 2020, 14, 569–573. [Google Scholar] [CrossRef]
- Mohamadou, Y.; Halidou, A.; Kapen, P.T. A review of mathematical modeling, artificial intelligence and datasets used in the study, prediction and management of COVID-19. Appl. Intell. 2020, 50, 3913–3925. [Google Scholar] [CrossRef]
- Raza, K. Artificial intelligence against COVID-19: A meta-analysis of current research. In Big Data Analytics and Artificial Intelligence against COVID-19: Innovation Vision and Approach; Hassanien, A.-E., Dey, N., Elghamrawy, S., Eds.; Springer International Publishing: Cham, Switzerland, 2020; Volume 78, pp. 165–176. [Google Scholar]
- Schultz, M.B.; Vera, D.; Sinclair, D.A. Can artificial intelligence identify effective COVID-19 therapies? EMBO Mol. Med. 2020, 12, e12817. [Google Scholar] [CrossRef]
- Shi, F.; Wang, J.; Shi, J.; Wu, Z.; Wang, Q.; Tang, Z.; He, K.; Shi, Y.; Shen, D. Review of artificial intelligence techniques in imaging data acquisition, segmentation, and diagnosis for COVID-19. IEEE Rev. Biomed. Eng. 2021, 14, 4–15. [Google Scholar] [CrossRef] [Green Version]
- Sujath, R.; Chatterjee, J.M.; Hassanien, A.E. A machine learning forecasting model for COVID-19 pandemic in india. Stoch. Environ. Res. Risk Assess. 2020, 34, 959–972. [Google Scholar] [CrossRef]
- Acharya, A.; Agarwal, R.; Baker, M.B.; Baudry, J.; Bhowmik, D.; Boehm, S.; Byler, K.G.; Chen, S.Y.; Coates, L.; Cooper, C.J.; et al. Supercomputer-based ensemble docking drug discovery pipeline with application to COVID-19. J. Chem. Inf. Model. 2020, 60, 5832–5852. [Google Scholar] [CrossRef]
- Arora, G.; Bothra, A.; Prosser, G.; Arora, K.; Sajid, A. Role of post-translational modifications in the acquisition of drug resistance in mycobacterium tuberculosis. FEBS J. 2021, 288, 3375–3393. [Google Scholar] [CrossRef]
- Arora, G.; Sajid, A.; Gupta, M.; Bhaduri, A.; Kumar, P.; Basu-Modak, S.; Singh, Y. Understanding the role of pknj in mycobacterium tuberculosis: Biochemical characterization and identification of novel substrate pyruvate kinase A. PLoS ONE 2010, 5, e10772. [Google Scholar] [CrossRef]
- Garay, P.G.; Barrera, E.E.; Klein, F.; Machado, M.R.; Soñora, M.; Pantano, S. The Sirah-Cov-2 initiative: A coarse-grained simulations’ dataset of the SARS-CoV-2 proteome. Front. Med. Technol. 2021, 3, 644039. [Google Scholar] [CrossRef]
- Lin, X.; Li, X.; Lin, X. A review on applications of computational methods in drug screening and design. Molecules 2020, 25, 1375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chalapareddy, S.K.; Sajid, A.; Saxena, M.; Arora, K.; Guha, R.; Arora, G. Emerging therapeutic modalities against malaria. In Translational Biotechnology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 267–286. [Google Scholar]
- Sunita; Sajid, A.; Singh, Y.; Shukla, P. Computational tools for modern vaccine development. Hum. Vaccines Immunother. 2020, 16, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Virmani, R.; Sajid, A.; Singhal, A.; Gaur, M.; Joshi, J.; Bothra, A.; Garg, R.; Misra, R.; Singh, V.P.; Molle, V.; et al. The ser/thr protein kinase prkc imprints phenotypic memory in bacillus anthracis spores by phosphorylating the glycolytic enzyme enolase. J. Biol. Chem. 2019, 294, 8930–8941. [Google Scholar] [CrossRef]
- Muehlematter, U.J.; Daniore, P.; Vokinger, K.N. Approval of artificial intelligence and machine learning-based medical devices in the USA and Europe (2015–20): A comparative analysis. Lancet Digit. Health 2021, 3, e195–e203. [Google Scholar] [CrossRef]
- Alsharif, M.H.; Alsharif, Y.H.; Albreem, M.A.; Jahid, A.; Solyman, A.A.A.; Yahya, K.; Alomari, O.A.; Hossain, M.S. Application of machine intelligence technology in the detection of vaccines and medicines for SARS-CoV-2. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 11977–11981. [Google Scholar] [PubMed]
- Omolo, C.A.; Soni, N.; Fasiku, V.O.; Mackraj, I.; Govender, T. Update on therapeutic approaches and emerging therapies for SARS-CoV-2 virus. Eur. J. Pharmacol. 2020, 883, 173348. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arora, G.; Joshi, J.; Mandal, R.S.; Shrivastava, N.; Virmani, R.; Sethi, T. Artificial Intelligence in Surveillance, Diagnosis, Drug Discovery and Vaccine Development against COVID-19. Pathogens 2021, 10, 1048. https://doi.org/10.3390/pathogens10081048
Arora G, Joshi J, Mandal RS, Shrivastava N, Virmani R, Sethi T. Artificial Intelligence in Surveillance, Diagnosis, Drug Discovery and Vaccine Development against COVID-19. Pathogens. 2021; 10(8):1048. https://doi.org/10.3390/pathogens10081048
Chicago/Turabian StyleArora, Gunjan, Jayadev Joshi, Rahul Shubhra Mandal, Nitisha Shrivastava, Richa Virmani, and Tavpritesh Sethi. 2021. "Artificial Intelligence in Surveillance, Diagnosis, Drug Discovery and Vaccine Development against COVID-19" Pathogens 10, no. 8: 1048. https://doi.org/10.3390/pathogens10081048
APA StyleArora, G., Joshi, J., Mandal, R. S., Shrivastava, N., Virmani, R., & Sethi, T. (2021). Artificial Intelligence in Surveillance, Diagnosis, Drug Discovery and Vaccine Development against COVID-19. Pathogens, 10(8), 1048. https://doi.org/10.3390/pathogens10081048






