The Interplay between Salmonella and Intestinal Innate Immune Cells in Chickens
Abstract
1. Introduction
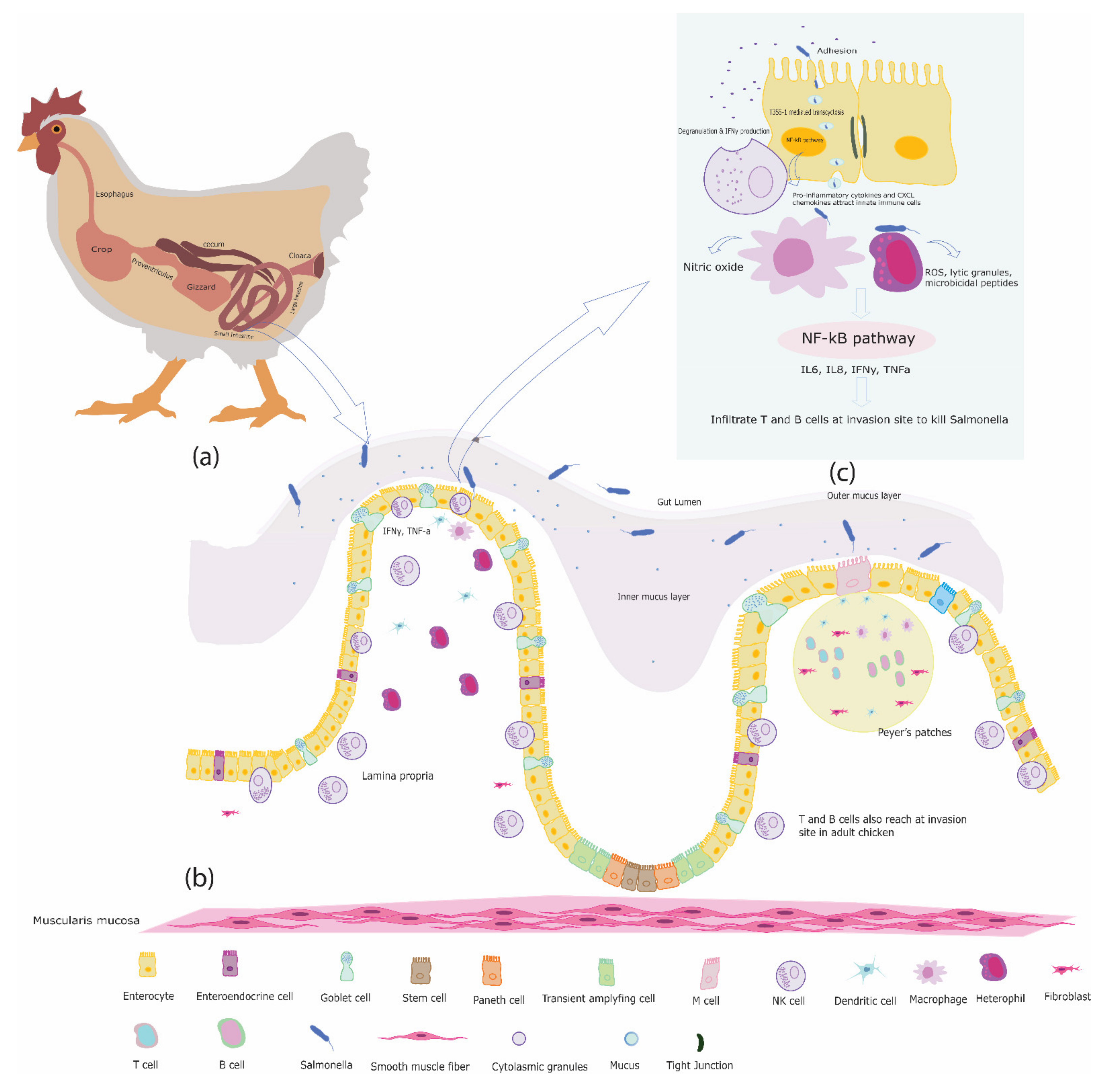
2. The Chicken Intestine
3. The Immune System of the Chicken Intestine
4. Innate Immune Cells in the Chicken Intestine
4.1. Natural Killer (NK) Cells of the Intestinal Immune System
4.2. Heterophils of the Intestinal Immune System
4.3. Dendritic Cells (DCs) of the Intestinal Immune System
4.4. Macrophages of the Intestinal Immune System
4.5. γδ T Cells of the Intestinal Immune System
5. Interactions between Salmonella and Innate Immune Cells in the Chicken Intestine
6. Future Prospects
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Castanheira, S.; García-Del Portillo, F. Salmonella populations inside host cells. Front. Cell. Infect. Microbiol. 2017, 7, 432. [Google Scholar] [CrossRef]
- Shivaprasad, H. Fowl typhoid and pullorum disease. Rev. Sci. Tech. (Int. Off. Epizoot.) 2000, 19, 405–424. [Google Scholar] [CrossRef]
- Snoeyenbos, G. Pullorum disease. In Diseases of Poultry; Iowa State University Press: Ames, IA, USA, 1991; pp. 72–86. [Google Scholar]
- Schleifer, J.; Juven, B.; Beard, C.; Cox, N. The susceptibility of chicks to Salmonella montevideo in artificially contaminated poultry feed. Avian Dis. 1984, 28, 497–503. [Google Scholar] [CrossRef]
- Jones, F.; Axtell, R.; Tarver, F.; Rives, D.; Scheideler, S.; Wineland, M. Environmental Factors Contributing to Salmonella Colonization of Chickens. In Colonization Control of Human Bacterial Enteropathogens in Poultry; Blankenship, L.C., Ed.; Academic Press, Inc.: San Diego, CA, USA, 1991; pp. 3–20. [Google Scholar]
- Authority, E.F.S. The European Union One Health 2019 Zoonoses Report. Efsa J. 2021, 19, e06406. [Google Scholar] [CrossRef]
- Chimalizeni, Y.; Kawaza, K.; Molyneux, E. The epidemiology and management of non typhoidal salmonella infections. In Hot Topics in Infection and Immunity in Children VI; Springer: New York, NY, USA, 2010; pp. 33–46. [Google Scholar] [CrossRef]
- Chen, H.-M.; Wang, Y.; Su, L.-H.; Chiu, C.-H. Nontyphoid Salmonella infection: Microbiology, clinical features, and antimicrobial therapy. Pediatrics Neonatol. 2013, 54, 147–152. [Google Scholar] [CrossRef]
- Suresh, G.; Das, R.K.; Kaur Brar, S.; Rouissi, T.; Avalos Ramirez, A.; Chorfi, Y.; Godbout, S. Alternatives to antibiotics in poultry feed: Molecular perspectives. Crit. Rev. Microbiol. 2018, 44, 318–335. [Google Scholar] [CrossRef]
- Reemers, S.S.; van Leenen, D.; Koerkamp, M.J.G.; van Haarlem, D.; van de Haar, P.; van Eden, W.; Vervelde, L. Early host responses to avian influenza A virus are prolonged and enhanced at transcriptional level depending on maturation of the immune system. Mol. Immunol. 2010, 47, 1675–1685. [Google Scholar] [CrossRef] [PubMed]
- Meijerink, N. Improving Health of Broiler Chickens by Early Life Modulation of Immune Responsiveness and Intestinal Microbiota: Recruiting Natural Killer Cells for Increased Resistance; Utrecht University: Utrecht, The Netherlands, 2021. [Google Scholar]
- Wells, C.A.; Ravasi, T.; Faulkner, G.J.; Carninci, P.; Okazaki, Y.; Hayashizaki, Y.; Sweet, M.; Wainwright, B.J.; Hume, D.A. Genetic control of the innate immune response. BMC Immunol. 2003, 4, 1–18. [Google Scholar] [CrossRef]
- Hofmann, T.; Schmucker, S.S.; Bessei, W.; Grashorn, M.; Stefanski, V. Impact of housing environment on the immune system in chickens: A review. Animals 2020, 10, 1138. [Google Scholar] [CrossRef] [PubMed]
- Svihus, B. Function of the digestive system. J. Appl. Poult. Res. 2014, 23, 306–314. [Google Scholar] [CrossRef]
- Zhu, X.Y.; Zhong, T.; Pandya, Y.; Joerger, R.D. 16S rRNA-based analysis of microbiota from the cecum of broiler chickens. Appl. Environ. Microbiol. 2002, 68, 124–137. [Google Scholar] [CrossRef]
- Richardson, A. The role of the crop in the feeding behaviour of the domestic chicken. Anim. Behav. 1970, 18, 633–639. [Google Scholar] [CrossRef]
- Choi, J.; Kim, G.; Cha, C. Spatial heterogeneity and stability of bacterial community in the gastrointestinal tracts of broiler chickens. Poult. Sci. 2014, 93, 1942–1950. [Google Scholar] [CrossRef] [PubMed]
- Stanley, D.; Hughes, R.J.; Moore, R.J. Microbiota of the chicken gastrointestinal tract: Influence on health, productivity and disease. Appl. Microbiol. Biotechnol. 2014, 98, 4301–4310. [Google Scholar] [CrossRef] [PubMed]
- Konsak, B.M.; Stanley, D.; Haring, V.R.; Geier, M.S.; Hughes, R.J.; Howarth, G.S.; Crowley, T.M.; Moore, R.J. Identification of differential duodenal gene expression levels and microbiota abundance correlated with differences in energy utilisation in chickens. Anim. Prod. Sci. 2013, 53, 1269–1275. [Google Scholar] [CrossRef]
- Clench, M.H.; Mathias, J.R. The avian cecum: A review. Wilson Bull. 1995, 107, 93–121. [Google Scholar]
- Gong, J.; Si, W.; Forster, R.J.; Huang, R.; Yu, H.; Yin, Y.; Yang, C.; Han, Y. 16S rRNA gene-based analysis of mucosa-associated bacterial community and phylogeny in the chicken gastrointestinal tracts: From crops to ceca. FEMS Microbiol. Ecol. 2007, 59, 147–157. [Google Scholar] [CrossRef]
- Owens, B.; Tucker, L.; Collins, M.; McCracken, K. Effects of different feed additives alone or in combination on broiler performance, gut microflora and ileal histology. Br. Poult. Sci. 2008, 49, 202–212. [Google Scholar] [CrossRef]
- Savage, D.C. Microbial ecology of the gastrointestinal tract. Annu. Rev. Microbiol. 1977, 31, 107–133. [Google Scholar] [CrossRef]
- Gaskins, H.R. Intestinal bacteria and their influence on swine growth. In Swine Nutrition, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2000; pp. 585–608. [Google Scholar]
- Apajalahti, J.; Kettunen, A.; Graham, H. Characteristics of the gastrointestinal microbial communities, with special reference to the chicken. World’s Poult. Sci. J. 2004, 60, 223–232. [Google Scholar] [CrossRef]
- Juricova, H.; Videnska, P.; Lukac, M.; Faldynova, M.; Babak, V.; Havlickova, H.; Sisak, F.; Rychlik, I. Influence of Salmonella enterica serovar enteritidis infection on the development of the cecum microbiota in newly hatched chicks. Appl. Environ. Microbiol. 2013, 79, 745–747. [Google Scholar] [CrossRef] [PubMed]
- Varmuzova, K.; Kubasova, T.; Davidova-Gerzova, L.; Sisak, F.; Havlickova, H.; Sebkova, A.; Faldynova, M.; Rychlik, I. Composition of gut microbiota influences resistance of newly hatched chickens to Salmonella Enteritidis infection. Front. Microbiol. 2016, 7, 957. [Google Scholar] [CrossRef] [PubMed]
- Tanikawa, T.; Shoji, N.; Sonohara, N.; Saito, S.; Shimura, Y.; Fukushima, J.; Inamoto, T. Aging transition of the bacterial community structure in the chick ceca. Poult. Sci. 2011, 90, 1004–1008. [Google Scholar] [CrossRef]
- Stanley, D.; Geier, M.S.; Hughes, R.J.; Denman, S.E.; Moore, R.J. Highly variable microbiota development in the chicken gastrointestinal tract. PLoS ONE 2013, 8, e84290. [Google Scholar]
- Mon, K.K.; Saelao, P.; Halstead, M.M.; Chanthavixay, G.; Chang, H.-C.; Garas, L.; Maga, E.A.; Zhou, H. Salmonella enterica serovars Enteritidis infection alters the indigenous microbiota diversity in young layer chicks. Front. Vet. Sci. 2015, 2, 61. [Google Scholar] [CrossRef]
- Awad, W.A.; Hess, C.; Hess, M. Enteric pathogens and their toxin-induced disruption of the intestinal barrier through alteration of tight junctions in chickens. Toxins 2017, 9, 60. [Google Scholar] [CrossRef]
- Johansson, M.E.; Hansson, G.C. Immunological aspects of intestinal mucus and mucins. Nat. Rev. Immunol. 2016, 16, 639–649. [Google Scholar] [CrossRef]
- Bansil, R.; Turner, B.S. Mucin structure, aggregation, physiological functions and biomedical applications. Curr. Opin. Colloid Interface Sci. 2006, 11, 164–170. [Google Scholar] [CrossRef]
- Sellers, L.A.; Allen, A.; Morris, E.R.; Ross-Murphy, S.B. Mucus glycoprotein gels. Role of glycoprotein polymeric structure and carbohydrate side-chains in gel-formation. Carbohydr. Res. 1988, 178, 93–110. [Google Scholar] [CrossRef]
- Schumacher, G.; Kim, M.; Hosseinian, A.; Dupon, C. Immunoglobulins, proteinase inhibitors, albumin, and lysozyme in human cervical mucus: I. Communication: Hormonal profiles and cervical mucus changes—Methods and results. Am. J. Obstet. Gynecol. 1977, 129, 629–636. [Google Scholar] [CrossRef]
- Jones, D.E.; Bevins, C.L. Paneth cells of the human small intestine express an antimicrobial peptide gene. J. Biol. Chem. 1992, 267, 23216–23225. [Google Scholar] [CrossRef]
- Mukherjee, S.; Zheng, H.; Derebe, M.G.; Callenberg, K.M.; Partch, C.L.; Rollins, D.; Propheter, D.C.; Rizo, J.; Grabe, M.; Jiang, Q.-X. Antibacterial membrane attack by a pore-forming intestinal C-type lectin. Nature 2014, 505, 103–107. [Google Scholar] [CrossRef]
- Allen, A.; Bell, A.; Mantle, M.; Pearson, J.P. The structure and physiology of gastrointestinal mucus. In Mucus Health and Disease II; Springer: Boston, MA, USA, 1982; pp. 115–133. [Google Scholar]
- Lang, T.; Hansson, G.C.; Samuelsson, T. An inventory of mucin genes in the chicken genome shows that the mucin domain of Muc13 is encoded by multiple exons and that ovomucin is part of a locus of related gel-forming mucins. BMC Genom. 2006, 7, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yegani, M.; Korver, D. Factors affecting intestinal health in poultry. Poult. Sci. 2008, 87, 2052–2063. [Google Scholar] [CrossRef]
- Cheng, H.; Leblond, C. Origin, differentiation and renewal of the four main epithelial cell types in the mouse small intestine I. Columnar cell. Am. J. Anat. 1974, 141, 461–479. [Google Scholar] [CrossRef]
- Uni, Z.; Smirnov, A.; Sklan, D. Pre-and posthatch development of goblet cells in the broiler small intestine: Effect of delayed access to feed. Poult. Sci. 2003, 82, 320–327. [Google Scholar] [CrossRef]
- Rehfeld, J. A centenary of gastrointestinal endocrinology. Horm. Metab. Res. 2004, 36, 735–741. [Google Scholar] [CrossRef]
- Wang, L.; Li, J.; Li, J., Jr.; Li, R.; Lv, C.; Li, S.; Mi, Y.; Zhang, C. Identification of the Paneth cells in chicken small intestine. Poult. Sci. 2016, 95, 1631–1635. [Google Scholar] [CrossRef] [PubMed]
- Nile, C.; Townes, C.; Michailidis, G.; Hirst, B.; Hall, J. Identification of chicken lysozyme g 2 and its expression in the intestine. Cell. Mol. Life Sci. 2004, 61, 2760–2766. [Google Scholar] [CrossRef] [PubMed]
- Nakano, T.; Graf, T. Goose-type lysozyme gene of the chicken: Sequence, genomic organization and expression reveals major differences to chicken-type lysozyme gene. Biochim. Biophys. Acta 1991, 1090, 273–276. [Google Scholar] [CrossRef]
- Smith, A.L.; Powers, C.; Beal, R.K. The avian enteric immune system in health and disease. In Avian Immunology, 2nd ed.; Elsevier: London, UK, 2014; pp. 227–250. [Google Scholar]
- Liebler-Tenorio, E.M.; Pabst, R. MALT structure and function in farm animals. Vet. Res. 2006, 37, 257–280. [Google Scholar] [CrossRef]
- Befus, A.D.; Johnston, N.; Leslie, G.; Bienenstock, J. Gut-associated lymphoid tissue in the chicken. I. Morphology, ontogeny, and some functional characteristics of Peyer’s patches. J. Immunol. 1980, 125, 2626–2632. [Google Scholar]
- Vervelde, L.; Jeurissen, S. Postnatal development of intra-epithelial leukocytes in the chicken digestive tract: Phenotypical characterization in situ. Cell Tissue Res. 1993, 274, 295–301. [Google Scholar] [CrossRef]
- Bockman, D.E.; Cooper, M.D. Pinocytosis by epithelium associated with lymphoid follicles in the bursa of Fabricius, appendix, and Peyer’s patches. An electron microscopic study. Am. J. Anat. 1973, 136, 455–477. [Google Scholar] [CrossRef] [PubMed]
- Jiménez-Dalmaroni, M.J.; Gerswhin, M.E.; Adamopoulos, I.E. The critical role of toll-like receptors—from microbial recognition to autoimmunity: A comprehensive review. Autoimmun. Rev. 2016, 15, 1–8. [Google Scholar] [CrossRef]
- Kogut, M.H.; Iqbal, M.; He, H.; Philbin, V.; Kaiser, P.; Smith, A. Expression and function of Toll-like receptors in chicken heterophils. Dev. Comp. Immunol. 2005, 29, 791–807. [Google Scholar] [CrossRef]
- Kaiser, P.; Rothwell, L.; Galyov, E.E.; Barrow, P.A.; Burnside, J.; Wigley, P. Differential cytokine expression in avian cells in response to invasion by Salmonella typhimurium, Salmonella enteritidis and Salmonella gallinarum The GenBank accession numbers for the sequences reported in this paper are AI982185 for chicken IL-6 cDNA and AJ250838 for the partial chicken IL-6 genomic sequence, respectively. Microbiology 2000, 146, 3217–3226. [Google Scholar]
- Dil, N.; Qureshi, M. Differential expression of inducible nitric oxide synthase is associated with differential Toll-like receptor-4 expression in chicken macrophages from different genetic backgrounds. Vet. Immunol. Immunopathol. 2002, 84, 191–207. [Google Scholar] [CrossRef]
- Kogut, M.H.; Swaggerty, C.; He, H.; Pevzner, I.; Kaiser, P. Toll-like receptor agonists stimulate differential functional activation and cytokine and chemokine gene expression in heterophils isolated from chickens with differential innate responses. Microbes Infect. 2006, 8, 1866–1874. [Google Scholar] [CrossRef]
- Kogut, M.; Genovese, K.; He, H. Flagellin and lipopolysaccharide stimulate the MEK-ERK signaling pathway in chicken heterophils through differential activation of the small GTPases, Ras and Rap1. Mol. Immunol. 2007, 44, 1729–1736. [Google Scholar] [CrossRef] [PubMed]
- Lillehoj, H. Analysis of Eimeria acervulina-induced changes in the intestinal T lymphocyte subpopulations in two chicken strains showing different levels of susceptibility to coccidiosis. Res. Vet. Sci. 1994, 56, 1–7. [Google Scholar] [CrossRef]
- Göbel, T.W.; Kaspers, B.; Stangassinger, M. NK and T cells constitute two major, functionally distinct intestinal epithelial lymphocyte subsets in the chicken. Int. Immunol. 2001, 13, 757–762. [Google Scholar] [CrossRef] [PubMed]
- Van Kaer, L.; Olivares-Villagómez, D. Development, homeostasis, and functions of intestinal intraepithelial lymphocytes. J. Immunol. 2018, 200, 2235–2244. [Google Scholar] [CrossRef]
- Morvan, M.G.; Lanier, L.L. NK cells and cancer: You can teach innate cells new tricks. Nat. Rev. Cancer 2016, 16, 7–19. [Google Scholar] [CrossRef]
- Lodoen, M.B.; Lanier, L.L. Natural killer cells as an initial defense against pathogens. Curr. Opin. Immunol. 2006, 18, 391–398. [Google Scholar] [CrossRef]
- Bonanni, V.; Sciumè, G.; Santoni, A.; Bernardini, G. Bone marrow NK cells: Origin, distinctive features, and requirements for tissue localization. Front. Immunol. 2019, 10, 1569. [Google Scholar] [CrossRef]
- Göbel, T.W.; Chen, C.L.H.; Shrimpf, J.; Grossi, C.E.; Bernot, A.; Bucy, R.P.; Auffray, C.; Cooper, M.D. Characterization of avian natural killer cells and their intracellular CD3 protein complex. Eur. J. Immunol. 1994, 24, 1685–1691. [Google Scholar] [CrossRef]
- Chai, J.; Lillehoj, H. Isolation and functional characterization of chicken intestinal intra-epithelial lymphocytes showing natural killer cell activity against tumour target cells. Immunology 1988, 63, 111. [Google Scholar]
- Alter, G.; Malenfant, J.M.; Altfeld, M. CD107a as a functional marker for the identification of natural killer cell activity. J. Immunol. Methods 2004, 294, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Jansen, C.A.; van de Haar, P.M.; van Haarlem, D.; van Kooten, P.; de Wit, S.; van Eden, W.; Viertlböck, B.C.; Göbel, T.W.; Vervelde, L. Identification of new populations of chicken natural killer (NK) cells. Dev. Comp. Immunol. 2010, 34, 759–767. [Google Scholar] [CrossRef]
- Jansen, C.A.; De Geus, E.D.; Van Haarlem, D.A.; Van De Haar, P.M.; Löndt, B.Z.; Graham, S.P.; Göbel, T.W.; Van Eden, W.; Brookes, S.M.; Vervelde, L. Differential lung NK cell responses in avian influenza virus infected chickens correlate with pathogenicity. Sci. Rep. 2013, 3, 2478. [Google Scholar] [CrossRef]
- Vervelde, L.; Matthijs, M.; Van Haarlem, D.; de Wit, J.; Jansen, C. Rapid NK-cell activation in chicken after infection with infectious bronchitis virus M41. Vet. Immunol. Immunopathol. 2013, 151, 337–341. [Google Scholar] [CrossRef]
- Meijerink, N.; van Haarlem, D.A.; Velkers, F.C.; Stegeman, A.J.; Rutten, V.P.; Jansen, C.A. Analysis of chicken intestinal natural killer cells, a major IEL subset during embryonic and early life. Dev. Comp. Immunol. 2021, 114, 103857. [Google Scholar] [CrossRef]
- Zhang, L.; Katselis, G.S.; Moore, R.E.; Lekpor, K.; Goto, R.M.; Hunt, H.D.; Lee, T.D.; Miller, M.M. MHC class I target recognition, immunophenotypes and proteomic profiles of natural killer cells within the spleens of day-14 chick embryos. Dev. Comp. Immunol. 2012, 37, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Sivori, S.; Vacca, P.; Del Zotto, G.; Munari, E.; Mingari, M.C.; Moretta, L. Human NK cells: Surface receptors, inhibitory checkpoints, and translational applications. Cell. Mol. Immunol. 2019, 16, 430–441. [Google Scholar] [CrossRef]
- Viertlboeck, B.C.; Habermann, F.A.; Schmitt, R.; Groenen, M.A.; Du Pasquier, L.; Göbel, T.W. The chicken leukocyte receptor complex: A highly diverse multigene family encoding at least six structurally distinct receptor types. J. Immunol. 2005, 175, 385–393. [Google Scholar] [CrossRef]
- Chiang, H.-I.; Zhou, H.; Raudsepp, T.; Jesudhasan, P.R.; Zhu, J.J. Chicken CD69 and CD94/NKG2-like genes in a chromosomal region syntenic to mammalian natural killer gene complex. Immunogenetics 2007, 59, 603–611. [Google Scholar] [CrossRef] [PubMed]
- Viertlboeck, B.C.; Göbel, T.W. The chicken leukocyte receptor cluster. Vet. Immunol. Immunopathol. 2011, 144, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Biassoni, R. Natural killer cell receptors. In Multichain Immune Recognition Receptor Signaling; Springer: New York, NY, USA, 2008; pp. 35–52. [Google Scholar]
- Luci, C.; Reynders, A.; Ivanov, I.I.; Cognet, C.; Chiche, L.; Chasson, L.; Hardwigsen, J.; Anguiano, E.; Banchereau, J.; Chaussabel, D. Influence of the transcription factor RORγt on the development of NKp46+ cell populations in gut and skin. Nat. Immunol. 2009, 10, 75–82. [Google Scholar] [CrossRef]
- Mayassi, T.; Jabri, B. Human intraepithelial lymphocytes. Mucosal Immunol. 2018, 11, 1281–1289. [Google Scholar] [CrossRef]
- Orange, J.S. Formation and function of the lytic NK-cell immunological synapse. Nat. Rev. Immunol. 2008, 8, 713–725. [Google Scholar] [CrossRef] [PubMed]
- Solana, R.; Tarazona, R.; Gayoso, I.; Lesur, O.; Dupuis, G.; Fulop, T. Innate immunosenescence: Effect of aging on cells and receptors of the innate immune system in humans. In Seminars in Immunology; Academic Press: Cambridge, MA, USA, 2012; Volume 24, pp. 331–341. [Google Scholar]
- Jahromi, M.Z.; Bello, M.B.; Abdolmaleki, M.; Yeap, S.K.; Hair-Bejo, M.; Omar, A.R. Differential activation of intraepithelial lymphocyte-natural killer cells in chickens infected with very virulent and vaccine strains of infectious bursal disease virus. Dev. Comp. Immunol. 2018, 87, 116–123. [Google Scholar] [CrossRef]
- Deeg, C.A.; Degroote, R.L.; Giese, I.M.; Hirmer, S.; Amann, B.; Weigand, M.; Wiedemann, C.; Hauck, S.M. CD11d is a novel antigen on chicken leukocytes. J. Proteom. 2020, 225, 103876. [Google Scholar] [CrossRef] [PubMed]
- Bertzbach, L.D.; van Haarlem, D.A.; Härtle, S.; Kaufer, B.B.; Jansen, C.A. Marek’s disease virus infection of natural killer cells. Microorganisms 2019, 7, 588. [Google Scholar] [CrossRef] [PubMed]
- Poggi, A.; Benelli, R.; Venè, R.; Costa, D.; Ferrari, N.; Tosetti, F.; Zocchi, M.R. Human gut-associated natural killer cells in health and disease. Front. Immunol. 2019, 10, 961. [Google Scholar] [CrossRef]
- Wolfe, S.A.; Tracey, D.E.; Henney, C.S. Induction of “natural killer” cells by BCG. Nature 1976, 262, 584–586. [Google Scholar] [CrossRef]
- Baron, E.J.; Proctor, R.A. Inefficient in vitro killing of virulent or nonvirulent Salmonella typhimurium by murine polymorphonuclear neutrophils. Can. J. Microbiol. 1984, 30, 1264–1270. [Google Scholar] [CrossRef]
- Maxwell, M.; Robertson, G. The avian heterophil leucocyte: A review. World’s Poult. Sci. J. 1998, 54, 155–178. [Google Scholar] [CrossRef]
- Kogut, M.; McGruder, E.; Hargis, B.; Corrier, D.; DeLoach, J. Dynamics of avian inflammatory response to Salmonella-immune lymphokines. Inflammation 1994, 18, 373–388. [Google Scholar] [CrossRef]
- Kogut, M.H.; McGruder, E.D.; Hargis, B.M.; Corner, D.E.; DeLoach, J.R. In vivo activation of heterophil function in chickens following injection with Salmonella enteritidis-immune lymphokines. J. Leukoc. Biol. 1995, 57, 56–62. [Google Scholar] [CrossRef]
- Kogut, M.H.; Mcgruder, E.D.; Hargis, B.M.; Corrier, D.E.; Deloach, J.R. Characterization of the pattern of inflammatory cell influx in chicks following the intraperitoneal administration of live Salmonella enteritidis and Salmonella enteritidis-immune lymphokines. Poult. Sci. 1995, 74, 8–17. [Google Scholar] [CrossRef]
- Kogut, M.; Holtzapple, C.; Lowry, V.; Genovese, K.; Stanker, L. Functional responses of neonatal chicken and turkey heterophils following stimulation by inflammatory agonists. Am. J. Vet. Res. 1998, 59, 1404–1408. [Google Scholar] [PubMed]
- Stabler, J.; McCormick, T.; Powell, K.; Kogut, M. Avian heterophils and monocytes: Phagocytic and bactericidal activities against Salmonella enteritidis. Vet. Microbiol. 1994, 38, 293–305. [Google Scholar] [CrossRef]
- Bar-Shira, E.; Friedman, A. Development and adaptations of innate immunity in the gastrointestinal tract of the newly hatched chick. Dev. Comp. Immunol. 2006, 30, 930–941. [Google Scholar] [CrossRef] [PubMed]
- Matulova, M.; Varmuzova, K.; Sisak, F.; Havlickova, H.; Babak, V.; Stejskal, K.; Zdrahal, Z.; Rychlik, I. Chicken innate immune response to oral infection with Salmonella enterica serovar Enteritidis. Vet. Res. 2013, 44, 1–11. [Google Scholar] [CrossRef]
- Kogut, M.H.; Rothwell, L.; Kaiser, P. IFN-γ priming of chicken heterophils upregulates the expression of proinflammatory and Th1 cytokine mRNA following receptor-mediated phagocytosis of Salmonella enterica serovar enteritidis. J. Interferon Cytokine Res. 2005, 25, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Radsak, M.P.; Salih, H.R.; Rammensee, H.-G.; Schild, H. Triggering receptor expressed on myeloid cells-1 in neutrophil inflammatory responses: Differential regulation of activation and survival. J. Immunol. 2004, 172, 4956–4963. [Google Scholar] [CrossRef]
- Genovese, K.J.; He, H.; Swaggerty, C.L.; Kogut, M.H. The avian heterophil. Dev. Comp. Immunol. 2013, 41, 334–340. [Google Scholar] [CrossRef]
- Chuammitri, P.; Ostojić, J.; Andreasen, C.B.; Redmond, S.B.; Lamont, S.J.; Palić, D. Chicken heterophil extracellular traps (HETs): Novel defense mechanism of chicken heterophils. Vet. Immunol. Immunopathol. 2009, 129, 126–131. [Google Scholar] [CrossRef]
- Guimaraes-Costa, A.B.; Nascimento, M.T.; Wardini, A.B.; Pinto-da-Silva, L.H.; Saraiva, E.M. ETosis: A microbicidal mechanism beyond cell death. J. Parasitol. Res. 2012, 2012. [Google Scholar] [CrossRef] [PubMed]
- Wells, L.L.; Lowry, V.K.; Deloach, J.R.; Kogut, M.H. Age-dependent phagocytosis and bactericidal activities of the chicken heterophil. Dev. Comp. Immunol. 1998, 22, 103–109. [Google Scholar] [CrossRef]
- Daimon, T.; Caxton-Martins, A. Electron microscopic and enzyme cytochemical studies on granules of mature chicken granular leucocytes. J. Anat. 1977, 123, 553. [Google Scholar]
- Banchereau, J.; Steinman, R.M. Dendritic cells and the control of immunity. Nature 1998, 392, 245–252. [Google Scholar] [CrossRef]
- Farache, J.; Zigmond, E.; Shakhar, G.; Jung, S. Contributions of dendritic cells and macrophages to intestinal homeostasis and immune defense. Immunol. Cell Biol. 2013, 91, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Garceau, V.; Smith, J.; Paton, I.R.; Davey, M.; Fares, M.A.; Sester, D.P.; Burt, D.W.; Hume, D.A. Pivotal Advance: Avian colony-stimulating factor 1 (CSF-1), interleukin-34 (IL-34), and CSF-1 receptor genes and gene products. J. Leukoc. Biol. 2010, 87, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Tamoutounour, S.; Henri, S.; Lelouard, H.; de Bovis, B.; de Haar, C.; van der Woude, C.J.; Woltman, A.M.; Reyal, Y.; Bonnet, D.; Sichien, D. CD 64 distinguishes macrophages from dendritic cells in the gut and reveals the T h1-inducing role of mesenteric lymph node macrophages during colitis. Eur. J. Immunol. 2012, 42, 3150–3166. [Google Scholar] [CrossRef] [PubMed]
- Solano-Gálvez, S.G.; Tovar-Torres, S.M.; Tron-Gómez, M.S.; Weiser-Smeke, A.E.; Álvarez-Hernández, D.A.; Franyuti-Kelly, G.A.; Tapia-Moreno, M.; Ibarra, A.; Gutiérrez-Kobeh, L.; Vázquez-López, R. Human dendritic cells: Ontogeny and their subsets in health and disease. Med Sci. 2018, 6, 88. [Google Scholar] [CrossRef]
- Merad, M.; Sathe, P.; Helft, J.; Miller, J.; Mortha, A. The dendritic cell lineage: Ontogeny and function of dendritic cells and their subsets in the steady state and the inflamed setting. Annu. Rev. Immunol. 2013, 31, 563–604. [Google Scholar] [CrossRef]
- Nagy, N.; Bódi, I.; Oláh, I. Avian dendritic cells: Phenotype and ontogeny in lymphoid organs. Dev. Comp. Immunol. 2016, 58, 47–59. [Google Scholar] [CrossRef]
- Hansell, C.; Zhu, X.W.; Brooks, H.; Sheppard, M.; Withanage, S.; Maskell, D.; McConnell, I. Unique features and distribution of the chicken CD83+ cell. J. Immunol. 2007, 179, 5117–5125. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Rothwell, L.; Young, J.R.; Kaufman, J.; Butter, C.; Kaiser, P. Generation and characterization of chicken bone marrow-derived dendritic cells. Immunology 2010, 129, 133–145. [Google Scholar] [CrossRef] [PubMed]
- Staines, K.; Young, J.R.; Butter, C. Expression of chicken DEC205 reflects the unique structure and function of the avian immune system. PLoS ONE 2013, 8, e51799. [Google Scholar] [CrossRef]
- Manh, T.-P.V.; Marty, H.; Sibille, P.; Le Vern, Y.; Kaspers, B.; Dalod, M.; Schwartz-Cornil, I.; Quéré, P. Existence of conventional dendritic cells in Gallus gallus revealed by comparative gene expression profiling. J. Immunol. 2014, 192, 4510–4517. [Google Scholar] [CrossRef] [PubMed]
- Jeurissen, S.; Janse, E.; Lehrbach, P.; Haddad, E.; Avakian, A.; Whitfill, C. The working mechanism of an immune complex vaccine that protects chickens against infectious bursal disease. Immunology 1998, 95, 494. [Google Scholar] [CrossRef] [PubMed]
- Olah, I.; Glick, B. Structure of the germinal centers in the chicken caecal tonsil: Light and electron microscopic and autoradiographic studies. Poult. Sci. 1979, 58, 195–210. [Google Scholar] [CrossRef]
- Nagy, N.; Oláh, I. Pyloric tonsil as a novel gut-associated lymphoepithelial organ of the chicken. J. Anat. 2007, 211, 407–411. [Google Scholar] [CrossRef]
- Gallego, M.; Del Cacho, E.; Bascuas, J. Antigen-binding cells in the cecal tonsil and Peyer’s patches of the chicken after bovine serum albumin administration. Poult. Sci. 1995, 74, 472–479. [Google Scholar] [CrossRef]
- Abasht, B.; Kaiser, M.; Lamont, S. Toll-like receptor gene expression in cecum and spleen of advanced intercross line chicks infected with Salmonella enterica serovar Enteritidis. Vet. Immunol. Immunopathol. 2008, 123, 314–323. [Google Scholar] [CrossRef]
- Van den Biggelaar, R.H.; Arkesteijn, G.J.; Rutten, V.P.; van Eden, W.; Jansen, C.A. In vitro chicken bone marrow-derived dendritic cells comprise subsets at different states of maturation. Front. Immunol. 2020, 11, 141. [Google Scholar] [CrossRef]
- Zanoni, I.; Ostuni, R.; Marek, L.R.; Barresi, S.; Barbalat, R.; Barton, G.M.; Granucci, F.; Kagan, J.C. CD14 controls the LPS-induced endocytosis of Toll-like receptor 4. Cell 2011, 147, 868–880. [Google Scholar] [CrossRef]
- Yitbarek, A.; Echeverry, H.; Brady, J.; Hernandez-Doria, J.; Camelo-Jaimes, G.; Sharif, S.; Guenter, W.; House, J.; Rodriguez-Lecompte, J. Innate immune response to yeast-derived carbohydrates in broiler chickens fed organic diets and challenged with Clostridium perfringens. Poult. Sci. 2012, 91, 1105–1112. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.; Singh, M.; Chander, V.; Sharma, G.K.; Mahawar, M.; Teeli, A.S.; Goswami, T.K. Differential responses of chicken monocyte-derived dendritic cells infected with Salmonella Gallinarum and Salmonella Typhimurium. Sci. Rep. 2021, 11, 17214. [Google Scholar] [CrossRef]
- Rehman, M.U.; Yoshihisa, Y.; Miyamoto, Y.; Shimizu, T. The anti-inflammatory effects of platinum nanoparticles on the lipopolysaccharide-induced inflammatory response in RAW 264.7 macrophages. Inflamm. Res. 2012, 61, 1177–1185. [Google Scholar] [CrossRef] [PubMed]
- Okamura, M.; Lillehoj, H.S.; Raybourne, R.B.; Babu, U.S.; Heckert, R.A.; Tani, H.; Sasai, K.; Baba, E.; Lillehoj, E.P. Differential responses of macrophages to Salmonella enterica serovars Enteritidis and Typhimurium. Vet. Immunol. Immunopathol. 2005, 107, 327–335. [Google Scholar] [CrossRef] [PubMed]
- Withanage, G.; Mastroeni, P.; Brooks, H.; Maskell, D.; McConnell, I. Oxidative and nitrosative responses of the chicken macrophage cell line MQ-NCSU to experimental Salmonella infection. Br. Poult. Sci. 2005, 46, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Babu, U.S.; Gaines, D.W.; Lillehoj, H.; Raybourne, R.B. Differential reactive oxygen and nitrogen production and clearance of Salmonella serovars by chicken and mouse macrophages. Dev. Comp. Immunol. 2006, 30, 942–953. [Google Scholar] [CrossRef]
- He, H.; Genovese, K.J.; Nisbet, D.J.; Kogut, M.H. Profile of Toll-like receptor expressions and induction of nitric oxide synthesis by Toll-like receptor agonists in chicken monocytes. Mol. Immunol. 2006, 43, 783–789. [Google Scholar] [CrossRef] [PubMed]
- He, H.; MacKinnon, K.M.; Genovese, K.J.; Kogut, M.H. CpG oligodeoxynucleotide and double-stranded RNA synergize to enhance nitric oxide production and mRNA expression of inducible nitric oxide synthase, pro-inflammatory cytokines and chemokines in chicken monocytes. Innate Immun. 2011, 17, 137–144. [Google Scholar]
- Martinez, F.O.; Gordon, S. The M1 and M2 paradigm of macrophage activation: Time for reassessment. F1000prime Rep. 2014, 6, 6–13. [Google Scholar] [CrossRef]
- Novak, M.L.; Koh, T.J. Macrophage phenotypes during tissue repair. J. Leukoc. Biol. 2013, 93, 875–881. [Google Scholar] [CrossRef]
- Sica, A.; Mantovani, A. Macrophage plasticity and polarization: In vivo veritas. J. Clin. Investig. 2012, 122, 787–795. [Google Scholar] [CrossRef]
- Ma, J.; Chen, T.; Mandelin, J.; Ceponis, A.; Miller, N.; Hukkanen, M.; Ma, G.; Konttinen, Y. Regulation of macrophage activation. Cell. Mol. Life Sci. 2003, 60, 2334–2346. [Google Scholar] [CrossRef] [PubMed]
- Gordon, S.; Martinez, F.O. Alternative activation of macrophages: Mechanism and functions. Immunity 2010, 32, 593–604. [Google Scholar] [CrossRef] [PubMed]
- Luzina, I.G.; Keegan, A.D.; Heller, N.M.; Rook, G.A.; Shea-Donohue, T.; Atamas, S.P. Regulation of inflammation by interleukin-4: A review of “alternatives”. J. Leukoc. Biol. 2012, 92, 753–764. [Google Scholar] [CrossRef] [PubMed]
- Rőszer, T. Understanding the mysterious M2 macrophage through activation markers and effector mechanisms. Mediat. Inflamm. 2015, 2015. [Google Scholar] [CrossRef]
- Peng, L.; van den Biggelaar, R.H.; Jansen, C.A.; Haagsman, H.P.; Veldhuizen, E.J. A method to differentiate chicken monocytes into macrophages with proinflammatory properties. Immunobiology 2020, 225, 152004. [Google Scholar] [CrossRef]
- Chaudhari, A.A.; Kim, W.H.; Lillehoj, H.S. Interleukin-4 (IL-4) may regulate alternative activation of macrophage-like cells in chickens: A sequential study using novel and specific neutralizing monoclonal antibodies against chicken IL-4. Vet. Immunol. Immunopathol. 2018, 205, 72–82. [Google Scholar] [CrossRef]
- Gordon, S. Pattern recognition receptors: Doubling up for the innate immune response. Cell 2002, 111, 927–930. [Google Scholar] [CrossRef]
- Wright, A.E.; Douglas, S.R. An experimental investigation of the role of the blood fluids in connection with phagocytosis. Proc. R. Soc. Lond. 1904, 72, 357–370. [Google Scholar] [CrossRef]
- Janeway, C.A. Approaching the asymptote? Evolution and revolution in immunology. Cold Spring Harb. Symp. Quant. Biol. 1989, 54, 1–13. [Google Scholar] [CrossRef]
- Janeway, C.A., Jr.; Medzhitov, R. Innate immune recognition. Annu. Rev. Immunol. 2002, 20, 197–216. [Google Scholar] [CrossRef] [PubMed]
- Stuart, L.M.; Ezekowitz, R.A.B. Phagocytosis: Elegant complexity. Immunity 2005, 22, 539–550. [Google Scholar] [CrossRef]
- Akira, S.; Yamamoto, M.; Takeda, K. Toll-like receptor family: Receptors essential for microbial recognition and immune responses. Arthritis Res Ther 2003, 5, 1–54. [Google Scholar] [CrossRef]
- He, H.; Genovese, K.J.; Nisbet, D.J.; Kogut, M.H. Synergy of CpG oligodeoxynucleotide and double-stranded RNA (poly I: C) on nitric oxide induction in chicken peripheral blood monocytes. Mol. Immunol. 2007, 44, 3234–3242. [Google Scholar] [CrossRef]
- Hartley, C.; Salisbury, A.-M.; Wigley, P. CpG oligonucleotides and recombinant interferon-γ in combination improve protection in chickens to Salmonella enterica serovar Enteritidis challenge as an adjuvant component, but have no effect in reducing Salmonella carriage in infected chickens. Avian Pathol. 2012, 41, 77–82. [Google Scholar] [CrossRef]
- Zhang, S.; Lillehoj, H.; Kim, C.-H.; Keeler, C., Jr.; Babu, U.; Zhang, M. Transcriptional response of chicken macrophages to Salmonella enterica serovar enteritidis infection. In Animal Genomics for Animal Health; Karger Publishers: Berlin, Germany, 2008; Volume 132, pp. 141–151. [Google Scholar]
- Kim, Y.-J.; Shin, Y.; Lee, K.H.; Kim, T.-J. Anethum graveloens flower extracts inhibited a lipopolysaccharide-induced inflammatory response by blocking iNOS expression and NF-κB activity in macrophages. Biosci. Biotechnol. Biochem. 2012, 76, 1122–1127. [Google Scholar] [CrossRef]
- Gao, J.; Scheenstra, M.R.; van Dijk, A.; Veldhuizen, E.J.; Haagsman, H.P. A new and efficient culture method for porcine bone marrow-derived M1-and M2-polarized macrophages. Vet. Immunol. Immunopathol. 2018, 200, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Djeraba, A.; Musset, E.; van Rooijen, N.; Quéré, P. Resistance and susceptibility to Marek’s disease: Nitric oxide synthase/arginase activity balance. Vet. Microbiol. 2002, 86, 229–244. [Google Scholar] [CrossRef]
- He, H.; Kogut, M.H. CpG-ODN-induced nitric oxide production is mediated through clathrin-dependent endocytosis, endosomal maturation, and activation of PKC, MEK1/2 and p38 MAPK, and NF-κB pathways in avian macrophage cells (HD11). Cell. Signal. 2003, 15, 911–917. [Google Scholar] [CrossRef]
- Khatri, M.; Palmquist, J.M.; Cha, R.M.; Sharma, J.M. Infection and activation of bursal macrophages by virulent infectious bursal disease virus. Virus Res. 2005, 113, 44–50. [Google Scholar] [CrossRef]
- Lillehoj, H.S.; Li, G. Nitric oxide production by macrophages stimulated with coccidia sporozoites, lipopolysaccharide, or interferon-γ, and its dynamic changes in SC and TK strains of chickens infected with Eimeria tenella. Avian Dis. 2004, 48, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, M. Avian macrophage and immune response: An overview. Poult. Sci. 2003, 82, 691–698. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.K.; Kaiser, P.; Rothwell, L.; Humphrey, T.; Barrow, P.A.; Jones, M.A. Campylobacter jejuni-induced cytokine responses in avian cells. Infect. Immun. 2005, 73, 2094–2100. [Google Scholar] [CrossRef] [PubMed]
- Van den Biggelaar, R.H.; Van Eden, W.; Rutten, V.P.; Jansen, C.A. Nitric oxide production and Fc receptor-mediated phagocytosis as functional readouts of macrophage activity upon stimulation with inactivated poultry vaccines in vitro. Vaccines 2020, 8, 332. [Google Scholar] [CrossRef]
- Van den Biggelaar, R.H.; van Eden, W.; Rutten, V.P.; Jansen, C.A. Macrophage Activation Assays to Evaluate the Immunostimulatory Capacity of Avibacterium paragallinarum in A Multivalent Poultry Vaccine. Vaccines 2020, 8, 671. [Google Scholar] [CrossRef]
- Lillehoj, H.S.; Trout, J.M. Avian gut-associated lymphoid tissues and intestinal immune responses to Eimeria parasites. Clin. Microbiol. Rev. 1996, 9, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Wigley, P.; Hulme, S.; Rothwell, L.; Bumstead, N.; Kaiser, P.; Barrow, P. Macrophages isolated from chickens genetically resistant or susceptible to systemic salmonellosis show magnitudinal and temporal differential expression of cytokines and chemokines following Salmonella enterica challenge. Infect. Immun. 2006, 74, 1425–1430. [Google Scholar] [CrossRef]
- He, H.; Genovese, K.J.; Kogut, M.H. Modulation of chicken macrophage effector function by TH1/TH2 cytokines. Cytokine 2011, 53, 363–369. [Google Scholar] [CrossRef]
- Malkovsky, M.; Bartz, S.R.; Mackenzie, D.; Radtke, B.E.; Wallace, M.; Manning, J.; Pauza, C.D.; Fisch, P. Are γδ T cells important for the elimination of virus-infected cells? J. Med Primatol. 1992, 21, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Hayday, A.C. γδ cells: A right time and a right place for a conserved third way of protection. Annu. Rev. Immunol. 2000, 18, 975–1026. [Google Scholar] [CrossRef] [PubMed]
- Sowder, J.T.; Chen, C.; Ager, L.L.; Chan, M.M.; Cooper, M. A large subpopulation of avian T cells express a homologue of the mammalian T gamma/delta receptor. J. Exp. Med. 1988, 167, 315–322. [Google Scholar] [CrossRef]
- Mackay, C.R.; Hein, W.R. A large proportion of bovine T cells express the γδ T cell receptor and show a distinct tissue distribution and surface phenotype. Int. Immunol. 1989, 1, 540–545. [Google Scholar] [CrossRef]
- Yang, H.; Parkhouse, R. Phenotypic classification of porcine lymphocyte subpopulations in blood and lymphoid tissues. Immunology 1996, 89, 76–83. [Google Scholar] [CrossRef]
- Kubota, T.; Wang, J.-y.; Göbel, T.W.; Hockett, R.D.; Cooper, M.D.; Chen-lo, H.C. Characterization of an avian (Gallus gallus domesticus) TCR αδ gene locus. J. Immunol. 1999, 163, 3858–3866. [Google Scholar]
- Pieper, J.; Methner, U.; Berndt, A. Heterogeneity of avian γδ T cells. Vet. Immunol. Immunopathol. 2008, 124, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Fenzl, L.; Göbel, T.W.; Neulen, M.-L. γδ T cells represent a major spontaneously cytotoxic cell population in the chicken. Dev. Comp. Immunol. 2017, 73, 175–183. [Google Scholar] [CrossRef]
- Holderness, J.; Hedges, J.F.; Ramstead, A.; Jutila, M.A. Comparative biology of γδ T cell function in humans, mice, and domestic animals. Annu. Rev. Anim. Biosci. 2013, 1, 99–124. [Google Scholar] [CrossRef] [PubMed]
- Baldwin, C.L.; Yirsaw, A.; Gillespie, A.; Le Page, L.; Zhang, F.; Damani-Yokota, P.; Telfer, J.C. γδ T cells in livestock: Responses to pathogens and vaccine potential. Transbound. Emerg. Dis. 2020, 67, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Karunakaran, M.M.; Willcox, C.R.; Salim, M.; Paletta, D.; Fichtner, A.S.; Noll, A.; Starick, L.; Nöhren, A.; Begley, C.R.; Berwick, K.A. Butyrophilin-2A1 directly binds germline-encoded regions of the Vγ9Vδ2 TCR and is essential for phosphoantigen sensing. Immunity 2020, 52, 487–498.e486. [Google Scholar] [CrossRef] [PubMed]
- Shibata, K.; Yamada, H.; Nakamura, M.; Hatano, S.; Katsuragi, Y.; Kominami, R.; Yoshikai, Y. IFN-γ–Producing and IL-17–Producing γδ T Cells Differentiate at Distinct Developmental Stages in Murine Fetal Thymus. J. Immunol. 2014, 192, 2210–2218. [Google Scholar] [CrossRef]
- Price, S.J.; Sopp, P.; Howard, C.J.; Hope, J.C. Workshop cluster 1+ γδ T-cell receptor+ T cells from calves express high levels of interferon-γ in response to stimulation with interleukin-12 and-18. Immunology 2007, 120, 57–65. [Google Scholar] [CrossRef]
- Alvarez, A.; Endsley, J.; Werling, D.; Mark Estes, D. WC1+ γδ T Cells Indirectly Regulate Chemokine Production During Mycobacterium bovis Infection in SCID-bo Mice. Transbound. Emerg. Dis. 2009, 56, 275–284. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, M.Z.; Yan, L.; Lillehoj, H.; Pace, L.W.; Zhang, S. Induction of CXC chemokine messenger-RNA expression in chicken oviduct epithelial cells by salmonella enterica serovar enteritidis via the type three secretion system–1. Avian Dis. 2009, 53, 396–404. [Google Scholar] [CrossRef]
- Withanage, G.; Wigley, P.; Kaiser, P.; Mastroeni, P.; Brooks, H.; Powers, C.; Beal, R.; Barrow, P.; Maskell, D.; McConnell, I. Cytokine and chemokine responses associated with clearance of a primary Salmonella enterica serovar Typhimurium infection in the chicken and in protective immunity to rechallenge. Infect. Immun. 2005, 73, 5173–5182. [Google Scholar] [CrossRef]
- Setta, A.; Barrow, P.; Kaiser, P.; Jones, M. Early immune dynamics following infection with Salmonella enterica serovars Enteritidis, Infantis, Pullorum and Gallinarum: Cytokine and chemokine gene expression profile and cellular changes of chicken cecal tonsils. Comp. Immunol. Microbiol. Infect. Dis. 2012, 35, 397–410. [Google Scholar] [CrossRef]
- Withanage, G.; Kaiser, P.; Wigley, P.; Powers, C.; Mastroeni, P.; Brooks, H.; Barrow, P.; Smith, A.; Maskell, D.; McConnell, I. Rapid expression of chemokines and proinflammatory cytokines in newly hatched chickens infected with Salmonella enterica serovar typhimurium. Infect. Immun. 2004, 72, 2152–2159. [Google Scholar] [CrossRef] [PubMed]
- Gart, E.V.; Suchodolski, J.S.; Welsh, T.H., Jr.; Alaniz, R.C.; Randel, R.D.; Lawhon, S.D. Salmonella typhimurium and multidirectional communication in the gut. Front. Microbiol. 2016, 7, 1827. [Google Scholar] [CrossRef]
- Darwin, K.H.; Miller, V.L. Molecular basis of the interaction of Salmonella with the intestinal mucosa. Clin. Microbiol. Rev. 1999, 12, 405–428. [Google Scholar] [CrossRef]
- Van Asten, A.J.; van Dijk, J.E. Distribution of “classic” virulence factors among Salmonella spp. FEMS Immunol. Med Microbiol. 2005, 44, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Marlovits, T.C.; Stebbins, C.E. Type III secretion systems shape up as they ship out. Curr. Opin. Microbiol. 2010, 13, 47–52. [Google Scholar] [CrossRef]
- Foley, S.; Lynne, A.; Nayak, R. Salmonella challenges: Prevalence in swine and poultry and potential pathogenicity of such isolates. J. Anim. Sci. 2008, 86, E149–E162. [Google Scholar] [CrossRef]
- Winnen, B.; Schlumberger, M.C.; Sturm, A.; Schüpbach, K.; Siebenmann, S.; Jenny, P.; Hardt, W.-D. Hierarchical effector protein transport by the Salmonella Typhimurium SPI-1 type III secretion system. PLoS ONE 2008, 3, e2178. [Google Scholar] [CrossRef]
- Schlumberger, M.C.; Hardt, W.-D. Salmonella type III secretion effectors: Pulling the host cell’s strings. Curr. Opin. Microbiol. 2006, 9, 46–54. [Google Scholar] [CrossRef]
- Marlovits, T.C.; Kubori, T.; Sukhan, A.; Thomas, D.R.; Galán, J.E.; Unger, V.M. Structural insights into the assembly of the type III secretion needle complex. Science 2004, 306, 1040–1042. [Google Scholar] [CrossRef] [PubMed]
- Galán, J.E. Salmonella interactions with host cells: Type III secretion at work. Annu. Rev. Cell Dev. Biol. 2001, 17, 53–86. [Google Scholar] [CrossRef] [PubMed]
- Lostroh, C.P.; Lee, C.A. The Salmonella pathogenicity island-1 type III secretion system. Microbes Infect. 2001, 3, 1281–1291. [Google Scholar] [CrossRef]
- Zhou, D.; Chen, L.M.; Hernandez, L.; Shears, S.B.; Galán, J.E. A Salmonella inositol polyphosphatase acts in conjunction with other bacterial effectors to promote host cell actin cytoskeleton rearrangements and bacterial internalization. Mol. Microbiol. 2001, 39, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Hardt, W.-D.; Chen, L.-M.; Schuebel, K.E.; Bustelo, X.R.; Galán, J.E.S. typhimurium encodes an activator of Rho GTPases that induces membrane ruffling and nuclear responses in host cells. Cell 1998, 93, 815–826. [Google Scholar] [CrossRef]
- Friebel, A.; Ilchmann, H.; Aepfelbacher, M.; Ehrbar, K.; Machleidt, W.; Hardt, W.-D. SopE and SopE2 from Salmonella typhimurium activate different sets of RhoGTPases of the host cell. J. Biol. Chem. 2001, 276, 34035–34040. [Google Scholar] [CrossRef]
- Iqbal, M.; Philbin, V.J.; Withanage, G.; Wigley, P.; Beal, R.K.; Goodchild, M.J.; Barrow, P.; McConnell, I.; Maskell, D.J.; Young, J. Identification and functional characterization of chicken toll-like receptor 5 reveals a fundamental role in the biology of infection with Salmonella enterica serovar typhimurium. Infect. Immun. 2005, 73, 2344–2350. [Google Scholar] [CrossRef]
- Chappell, L.; Kaiser, P.; Barrow, P.; Jones, M.A.; Johnston, C.; Wigley, P. The immunobiology of avian systemic salmonellosis. Vet. Immunol. Immunopathol. 2009, 128, 53–59. [Google Scholar] [CrossRef]
- Kogut, M.H.; Genovese, K.J.; Lowry, V.K. Differential activation of signal transduction pathways mediating phagocytosis, oxidative burst, and degranulation by chicken heterophils in response to stimulation with opsonized Salmonella enteritidis. Inflammation 2001, 25, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Swaggerty, C.L.; Pevzner, I.Y.; Lowry, V.K.; Farnell, M.B.; Kogut, M.H. Functional comparison of heterophils isolated from commercial broiler chickens. Avian Pathol. 2003, 32, 95–102. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Van Dijk, A.; Tersteeg-Zijderveld, M.H.; Tjeerdsma-van Bokhoven, J.L.; Jansman, A.J.; Veldhuizen, E.J.; Haagsman, H.P. Chicken heterophils are recruited to the site of Salmonella infection and release antibacterial mature Cathelicidin-2 upon stimulation with LPS. Mol. Immunol. 2009, 46, 1517–1526. [Google Scholar] [CrossRef] [PubMed]
- Pieper, J.; Locke, M.; Ruzaike, G.; Voigt, S.; Methner, U.; Berndt, A. In vitro and in vivo generation of heterophil extracellular traps after Salmonella exposure. Vet. Immunol. Immunopathol. 2017, 188, 1–11. [Google Scholar] [CrossRef]
- Steiner, T.S. How flagellin and toll-like receptor 5 contribute to enteric infection. Infect. Immun. 2007, 75, 545–552. [Google Scholar] [CrossRef]
- Park, B.S.; Song, D.H.; Kim, H.M.; Choi, B.-S.; Lee, H.; Lee, J.-O. The structural basis of lipopolysaccharide recognition by the TLR4–MD-2 complex. Nature 2009, 458, 1191–1195. [Google Scholar] [CrossRef]
- Yoon, S.-i.; Kurnasov, O.; Natarajan, V.; Hong, M.; Gudkov, A.V.; Osterman, A.L.; Wilson, I.A. Structural basis of TLR5-flagellin recognition and signaling. Science 2012, 335, 859–864. [Google Scholar] [CrossRef]
- McDermott, P.F.; Ciacci-Woolwine, F.; Snipes, J.A.; Mizel, S.B. High-affinity interaction between gram-negative flagellin and a cell surface polypeptide results in human monocyte activation. Infect. Immun. 2000, 68, 5525–5529. [Google Scholar] [CrossRef] [PubMed]
- Eaves-Pyles, T.; Murthy, K.; Liaudet, L.; Virág, L.; Ross, G.; Soriano, F.G.; Szabó, C.; Salzman, A.L. Flagellin, a novel mediator of Salmonella-induced epithelial activation and systemic inflammation: IκBα degradation, induction of nitric oxide synthase, induction of proinflammatory mediators, and cardiovascular dysfunction. J. Immunol. 2001, 166, 1248–1260. [Google Scholar] [CrossRef]
- Gewirtz, A.T.; Navas, T.A.; Lyons, S.; Godowski, P.J.; Madara, J.L. Cutting edge: Bacterial flagellin activates basolaterally expressed TLR5 to induce epithelial proinflammatory gene expression. J. Immunol. 2001, 167, 1882–1885. [Google Scholar] [CrossRef] [PubMed]
- Means, T.K.; Hayashi, F.; Smith, K.D.; Aderem, A.; Luster, A.D. The Toll-like receptor 5 stimulus bacterial flagellin induces maturation and chemokine production in human dendritic cells. J. Immunol. 2003, 170, 5165–5175. [Google Scholar] [CrossRef]
- Khan, M.A.; Kang, J.; Steiner, T.S. Enteroaggregative Escherichia coli flagellin-induced interleukin-8 secretion requires Toll-like receptor 5-dependent p38 MAP kinase activation. Immunology 2004, 112, 651–660. [Google Scholar] [CrossRef]
- Crippen, T.L. The selective inhibition of nitric oxide production in the avian macrophage cell line HD11. Vet. Immunol. Immunopathol. 2006, 109, 127–137. [Google Scholar] [CrossRef]
- Beal, R.; Wigley, P.; Powers, C.; Hulme, S.; Barrow, P.; Smith, A. Age at primary infection with Salmonella enterica serovar Typhimurium in the chicken influences persistence of infection and subsequent immunity to re-challenge. Vet. Immunol. Immunopathol. 2004, 100, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.; Fresno, A.H.; Skov, S.; Olsen, J.E. Dynamics and outcome of macrophage interaction between Salmonella gallinarum, Salmonella Typhimurium, and Salmonella dublin and macrophages from chicken and cattle. Front. Cell. Infect. Microbiol. 2020, 9, 420. [Google Scholar] [CrossRef]
- Swaggerty, C.L.; Kogut, M.H.; Ferro, P.J.; Rothwell, L.; Pevzner, I.Y.; Kaiser, P. Differential cytokine mRNA expression in heterophils isolated from Salmonella-resistant and-susceptible chickens. Immunology 2004, 113, 139–148. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.; Pan, Q.; Hui, X.; Zhang, B.; Wu, H.; Li, H.; Xu, W.; Zhang, Q.; Li, J.; Deng, X. Stronger in vitro phagocytosis by monocytes-macrophages is indicative of greater pathogen clearance and antibody levels in vivo. Poult. Sci. 2008, 87, 1725–1733. [Google Scholar] [CrossRef]
- Johannessen, M.; Askarian, F.; Sangvik, M.; Sollid, J.E. Bacterial interference with canonical NFκB signalling. Microbiology 2013, 159, 2001. [Google Scholar] [CrossRef]
- Brigelius-Flohé, R.; Flohé, L. Basic principles and emerging concepts in the redox control of transcription factors. Antioxid. Redox Signal. 2011, 15, 2335–2381. [Google Scholar] [CrossRef]
- Bekiaris, V.; Persson, E.K.; Agace, W.W. Intestinal dendritic cells in the regulation of mucosal immunity. Immunol. Rev. 2014, 260, 86–101. [Google Scholar] [CrossRef]
- Vazquez-Torres, A.; Jones-Carson, J.; Bäumler, A.J.; Falkow, S.; Valdivia, R.; Brown, W.; Le, M.; Berggren, R.; Parks, W.T.; Fang, F.C. Extraintestinal dissemination of Salmonella by CD18-expressing phagocytes. Nature 1999, 401, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Swart, A.L.; Hensel, M. Interactions of Salmonella enterica with dendritic cells. Virulence 2012, 3, 660–667. [Google Scholar] [CrossRef] [PubMed]
- McSorley, S.J.; Asch, S.; Costalonga, M.; Reinhardt, R.L.; Jenkins, M.K. Tracking salmonella-specific CD4 T cells in vivo reveals a local mucosal response to a disseminated infection. Immunity 2002, 16, 365–377. [Google Scholar] [CrossRef]
- Santos, R.L.; Raffatellu, M.; Bevins, C.L.; Adams, L.G.; Tükel, Ç.; Tsolis, R.M.; Bäumler, A.J. Life in the inflamed intestine, Salmonella style. Trends Microbiol. 2009, 17, 498–506. [Google Scholar] [CrossRef]
- Mastroeni, P.; Ménager, N. Development of acquired immunity to Salmonella. J. Med Microbiol. 2003, 52, 453–459. [Google Scholar] [CrossRef]
- Haller, D.; Serrant, P.; Granato, D.; Schiffrin, E.; Blum, S. Activation of human NK cells by staphylococci and lactobacilli requires cell contact-dependent costimulation by autologous monocytes. Clin. Vaccine Immunol. 2002, 9, 649–657. [Google Scholar] [CrossRef]
- Carvajal, B.G.; Methner, U.; Pieper, J.; Berndt, A. Effects of Salmonella enterica serovar Enteritidis on cellular recruitment and cytokine gene expression in caecum of vaccinated chickens. Vaccine 2008, 26, 5423–5433. [Google Scholar] [CrossRef]
- Klasing, K.; Leshchinsky, T. Functions, costs, and benefits of the immune system during development and growth. Ostrich 1999, 69, 32. [Google Scholar]
- Meijerink, N.; van den Biggelaar, R.H.; van Haarlem, D.A.; Stegeman, J.A.; Rutten, V.P.; Jansen, C.A. A detailed analysis of innate and adaptive immune responsiveness upon infection with Salmonella enterica serotype Enteritidis in young broiler chickens. Vet. Res. 2021, 52, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Berndt, A.; Pieper, J.; Methner, U. Circulating γδ T Cells in Response to Salmonella enterica Serovar Enteritidis Exposure in Chickens. Infect. Immun. 2006, 74, 3967–3978. [Google Scholar] [CrossRef]
- Sekelova, Z.; Polansky, O.; Stepanova, H.; Fedr, R.; Faldynova, M.; Rychlik, I.; Vlasatikova, L. Different roles of CD4, CD8 and γδ T-lymphocytes in naive and vaccinated chickens during Salmonella Enteritidis infection. Proteomics 2017, 17, 1700073. [Google Scholar] [CrossRef]
- Schokker, D.; Peters, T.; Hoekman, A.; Rebel, J.; Smits, M. Differences in the early response of hatchlings of different chicken breeding lines to Salmonella enterica serovar Enteritidis infection. Poult. Sci. 2012, 91, 346–353. [Google Scholar] [CrossRef]
- Van Hemert, S.; Hoekman, A.J.; Smits, M.A.; Rebel, J.M. Immunological and gene expression responses to a Salmonella infection in the chicken intestine. Vet. Res. 2007, 38, 51–63. [Google Scholar] [CrossRef]
- Deseke, M.; Prinz, I. Ligand recognition by the γδ TCR and discrimination between homeostasis and stress conditions. Cell. Mol. Immunol. 2020, 17, 914–924. [Google Scholar] [CrossRef]
- Bai, S.; Wu, A.; Ding, X.; Lei, Y.; Bai, J.; Zhang, K.; Chio, J. Effects of probiotic-supplemented diets on growth performance and intestinal immune characteristics of broiler chickens. Poult. Sci. 2013, 92, 663–670. [Google Scholar] [CrossRef]
- Meijerink, N.; de Oliveira, J.E.; van Haarlem, D.A.; Hosotani, G.; Lamot, D.M.; Stegeman, J.A.; Rutten, V.P.; Jansen, C.A. Glucose Oligosaccharide and Long-Chain Glucomannan Feed Additives Induce Enhanced Activation of Intraepithelial NK Cells and Relative Abundance of Commensal Lactic Acid Bacteria in Broiler Chickens. Vet. Sci. 2021, 8, 110. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.H.; Teng, P.Y.; Lee, T.T.; Yu, B. Effects of multi-strain probiotic supplementation on intestinal microbiota, tight junctions, and inflammation in young broiler chickens challenged with Salmonella enterica subsp. enterica. Asian Australas. J. Anim. Sci. 2020, 33, 1797. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Cai, K.; Mishra, R.; Jha, R. In ovo supplementation of chitooligosaccharide and chlorella polysaccharide affects cecal microbial community, metabolic pathways, and fermentation metabolites in broiler chickens. Poult. Sci. 2020, 99, 4776–4785. [Google Scholar] [CrossRef] [PubMed]
- Stefaniak, T.; Madej, J.P.; Graczyk, S.; Siwek, M.; Łukaszewicz, E.; Kowalczyk, A.; Sieńczyk, M.; Bednarczyk, M. Selected prebiotics and synbiotics administered in ovo can modify innate immunity in chicken broilers. BMC Vet. Res. 2019, 15, 1–9. [Google Scholar] [CrossRef]
- Nash, T.J.; Morris, K.M.; Mabbott, N.A.; Vervelde, L. Inside-out chicken enteroids with leukocyte component as a model to study host–pathogen interactions. Commun. Biol. 2021, 4, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Oost, M.J.; Velkers, F.C.; Kraneveld, A.D.; Venema, K. Development of the in vitro Cecal Chicken ALIMEntary tRact mOdel-2 to Study Microbiota Composition and Function. Front. Microbiol. 2021, 12, 726447. [Google Scholar] [CrossRef] [PubMed]
| Host Cell | Mediators | Functions | References |
|---|---|---|---|
| Intestinal epithelial cells | anti-microbial peptides (AMPs), Mucus, CXC chemokines, IL12, IL18, IL1β | Provide diffusive barrier to pathogen; Attract underlying innate immune cells; shape the activity of the immune cells | [173,174,175,176] |
| NK cells | perforins, granzymes, IFNγ, TNFα | Cytolysis of transformed and infected host cells; shape the activity of innate and adaptive immune cells | [79,84] |
| Heterophils | CXCLi, IL6, IL1β, cytoplasmic granules, HET | Direct Killing of invading pathogen by phagocytosis, degranulation, HET; Attract immune cells from peripheral blood and nearby organs | [56,97,98] |
| DCs | CXCLi1, CXCLi2, IL1β, IL10, IL6, IL12p35, TNFα | Antigen presentation to adaptive immune cells to initiate specific immune response; attract and shape the function of innate immune cells | [121] |
| Macrophages | CXCLi1, CXCLi2, IL10, IL12p40, IL12, IL18, TNFα, iNOS | Direct killing of pathogen by producing reactive oxygen species, nitric oxide production; Drive Th1 adaptive immune response | [146,149,152,155,157] |
| γδ T cells | Perforins, granzymes, IL17, IFNγ | Cytolysis of infected host cells; Attract other innate immune cells at site of infection and shape their immune function | [170,171] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ijaz, A.; Veldhuizen, E.J.A.; Broere, F.; Rutten, V.P.M.G.; Jansen, C.A. The Interplay between Salmonella and Intestinal Innate Immune Cells in Chickens. Pathogens 2021, 10, 1512. https://doi.org/10.3390/pathogens10111512
Ijaz A, Veldhuizen EJA, Broere F, Rutten VPMG, Jansen CA. The Interplay between Salmonella and Intestinal Innate Immune Cells in Chickens. Pathogens. 2021; 10(11):1512. https://doi.org/10.3390/pathogens10111512
Chicago/Turabian StyleIjaz, Adil, Edwin J. A. Veldhuizen, Femke Broere, Victor P. M. G. Rutten, and Christine A. Jansen. 2021. "The Interplay between Salmonella and Intestinal Innate Immune Cells in Chickens" Pathogens 10, no. 11: 1512. https://doi.org/10.3390/pathogens10111512
APA StyleIjaz, A., Veldhuizen, E. J. A., Broere, F., Rutten, V. P. M. G., & Jansen, C. A. (2021). The Interplay between Salmonella and Intestinal Innate Immune Cells in Chickens. Pathogens, 10(11), 1512. https://doi.org/10.3390/pathogens10111512







