Development of a Multiplex Real-Time PCR Assay for Predicting Macrolide and Tetracycline Resistance Associated with Bacterial Pathogens of Bovine Respiratory Disease
Abstract
1. Introduction
2. Results
2.1. Characteristics of the Multiplex qPCR Assay
2.2. Phenotypic Antimicrobial Resistance Based on the Gold-Standard Test
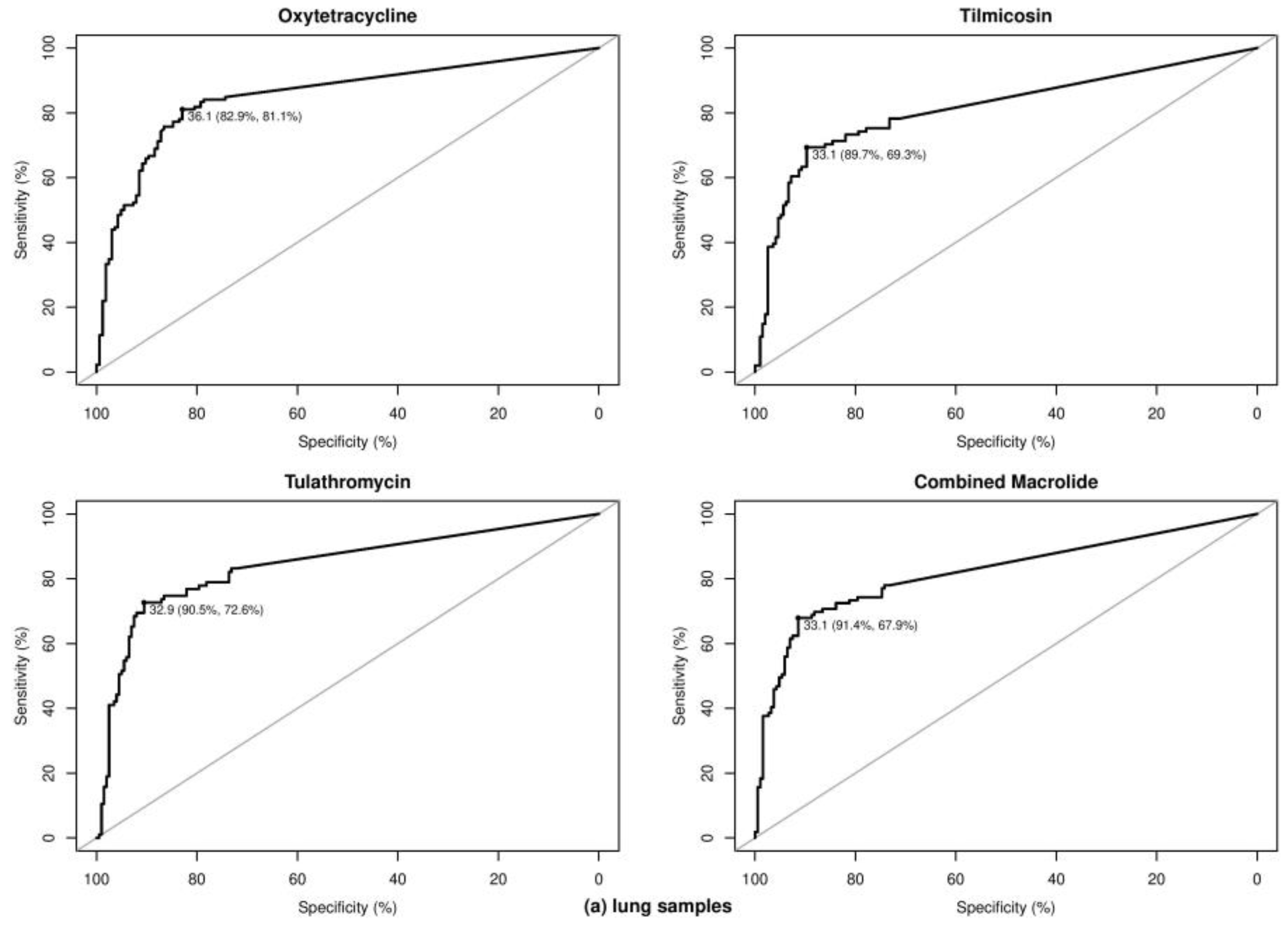
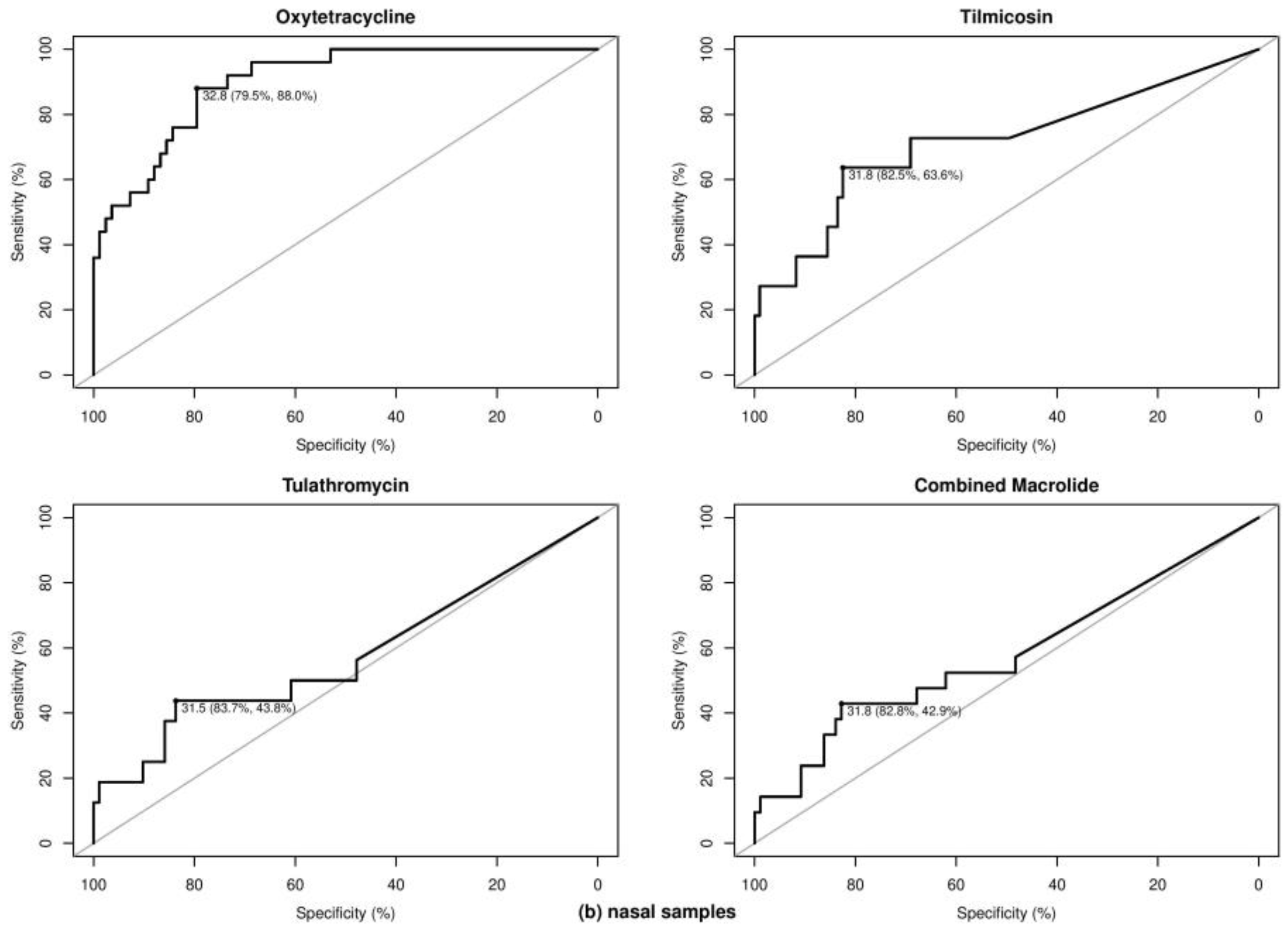
2.3. Optimal Ct Cutoff Value Determination
2.4. Validation of the Computational Approach
3. Discussion
4. Materials and Methods
4.1. Sample Collection and Distribution
4.2. Reference Strains
4.3. Molecular-Based Rapid Detection Assay
4.4. Preliminary Assay Validation and Analytical Sensitivity
4.5. Pathogen Isolation and Determination of Phenotypic Antimicrobial Resistance Characteristics
4.6. Predicting Phenotypic Antimicrobial Resistance
4.6.1. Optimal Ct Cutoff Value Determination
4.6.2. Diagnostic Accuracy Evaluation
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Griffin, D.; Chengappa, M.M.; Kuszak, J.; McVey, D.S. Bacterial pathogens of the bovine respiratory disease complex. Vet. Clin. North Am. Food Anim. Pr. 2010, 26, 381–394. [Google Scholar] [CrossRef] [PubMed]
- Amat, S. Bovine respiratory disease in feedlot cattle: Antimicrobial resistance in bovine respiratory bacterial pathogens and alternative antimicrobial approaches. Bact. Cattle Dis. 2019. [Google Scholar] [CrossRef]
- Faber, R.; Hartwig, N.; Busby, D.; BreDahl, R. The Costs and Predictive Factors of Bovine Respiratory Disease in Standardized Steer Tests. 2000. Available online: https://lib.dr.iastate.edu/beefreports_1999/24 (accessed on 23 September 2020).
- Campbell, J. Bovine Respiratory Disease Complex. Available online: https://www.merckvetmanual.com/respiratory-system/respiratory-diseases-of-cattle/bovine-respiratory-disease-complex (accessed on 30 November 2020).
- D’Amours, G.H.; Ward, T.I.; Mulvey, M.R.; Read, R.R.; Morck, D.W. Genetic diversity and tetracycline resistance genes of Histophilus somni. Vet. Microbiol. 2011, 150, 362–372. [Google Scholar] [CrossRef] [PubMed]
- Arcangioli, M.A.; Duet, A.; Meyer, G.; Dernburg, A.; Bezille, P.; Poumarat, F.; Le Grand, D. The role of Mycoplasma bovis in bovine respiratory disease outbreaks in veal calf feedlots. Vet. J. 2008, 177, 89–93. [Google Scholar] [CrossRef]
- Anholt, R.M.; Klima, C.; Allan, N.; Matheson-Bird, H.; Schatz, C.; Ajitkumar, P.; Otto, S.J.G.; Peters, D.; Schmid, K.; Olson, M.; et al. Antimicrobial susceptibility of bacteria that cause bovine respiratory disease complex in Alberta, Canada. Front. Vet. Sci. 2017, 4, 207. [Google Scholar] [CrossRef]
- Portis, E.; Lindeman, C.; Johansen, L.; Stoltman, G. A ten-year (2000–2009) study of antimicrobial susceptibility of bacteria that cause bovine respiratory disease complex—Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni—in the United States and Canada. J. Vet. Diagn. Investig. 2012, 24, 932–944. [Google Scholar] [CrossRef]
- USDA. Feedlot 2011 Part IV: Health and Health Management on U.S. Feedlots with a Capacity of 1000 or More Head. Available online: https://www.aphis.usda.gov/animal_health/nahms/feedlot/downloads/feedlot2011/Feed11_dr_PartIV_1.pdf (accessed on 28 August 2020).
- Klima, C.L.; Zaheer, R.; Cook, S.R.; Booker, C.W.; Hendrick, S.; Alexander, T.W.; McAllister, T.A. Pathogens of bovine respiratory disease in North American feedlots conferring multidrug resistance via integrative conjugative elements. J. Clin. Microbiol. 2014, 52, 438–448. [Google Scholar] [CrossRef]
- Noyes, N.R.; Benedict, K.M.; Gow, S.P.; Booker, C.W.; Hannon, S.J.; McAllister, T.A.; Morley, P.S. Mannheimia haemolytica in feedlot cattle: Prevalence of recovery and associations with antimicrobial use, resistance, and health outcomes. J. Vet. Intern. Med. 2015, 29, 705–713. [Google Scholar] [CrossRef]
- Timsit, E.; Hallewell, J.; Booker, C.; Tison, N.; Amat, S.; Alexander, T.W. Prevalence and antimicrobial susceptibility of Mannheimia haemolytica, Pasteurella multocida, and Histophilus somni isolated from the lower respiratory tract of healthy feedlot cattle and those diagnosed with bovine respiratory disease. Vet. Microbiol. 2017, 208, 118–125. [Google Scholar] [CrossRef]
- Loy, J.D.; Leger, L.; Workman, A.M.; Clawson, M.L.; Bulut, E.; Wang, B. Development of a multiplex real-time PCR assay using two thermocycling platforms for detection of major bacterial pathogens associated with bovine respiratory disease complex from clinical samples. J. Vet. Diagn. Investig. 2018, 30, 837–847. [Google Scholar] [CrossRef]
- McLain, J.E.; Cytryn, E.; Durso, L.M.; Young, S. Culture-based methods for detection of antibiotic resistance in agroecosystems: Advantages, challenges, and gaps in knowledge. J. Environ. Qual. 2016, 45, 432–440. [Google Scholar] [CrossRef] [PubMed]
- DeDonder, K.D.; Apley, M.D. A literature review of antimicrobial resistance in pathogens associated with bovine respiratory disease. Anim. Health Res. Rev. 2015, 16, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Boolchandani, M.; D’Souza, A.W.; Dantas, G. Sequencing-based methods and resources to study antimicrobial resistance. Nat. Rev. Genet. 2019, 20, 356–370. [Google Scholar] [CrossRef] [PubMed]
- Dennis, E.J. Antimicrobial Used for Metaphylaxis and Implications for Product Diversification in the Animal Health Sector. NebGuide G2319 Animal Agriculture/Beef. 2019. Available online: https://extensionpublications.unl.edu/assets/pdf/g2319.pdf (accessed on 28 August 2020).
- USDA NAHMS. Antimicrobial Use and Stewardship on U.S. Feedlots, 2017. 2019. Available online: https://www.aphis.usda.gov/animal_health/nahms/amr/downloads/amu-feedlots_1.pdf (accessed on 28 August 2020).
- Clawson, M.L.; Murray, R.W.; Sweeney, M.T.; Apley, M.D.; DeDonder, K.D.; Capik, S.F.; Larson, R.L.; Lubbers, B.V.; White, B.J.; Kalbfleisch, T.S.; et al. Genomic signatures of Mannheimia haemolytica that associate with the lungs of cattle with respiratory disease, an integrative conjugative element, and antibiotic resistance genes. BMC Genom. 2016, 17, 982. [Google Scholar] [CrossRef]
- Livermore, D.M.; Winstanley, T.G.; Shannon, K.P. Interpretative reading: Recognizing the unusual and inferring resistance mechanisms from resistance phenotypes. J. Antimicrob. Chemother. 2001, 48 (Suppl. S1), 87–102. [Google Scholar] [CrossRef][Green Version]
- Owen, J.R.; Noyes, N.; Young, A.E.; Prince, D.J.; Blanchard, P.C.; Lehenbauer, T.W.; Aly, S.S.; Davis, J.H.; O’Rourke, S.M.; Abdo, Z.; et al. Whole-genome sequencing and concordance between antimicrobial susceptibility genotypes and phenotypes of bacterial isolates associated with bovine respiratory disease. G3 Genes Genomes Genet. 2017, 7, 3059. [Google Scholar] [CrossRef]
- Ruegg, P.L.; Oliveira, L.; Jin, W.; Okwumabua, O. Phenotypic antimicrobial susceptibility and occurrence of selected resistance genes in gram-positive mastitis pathogens isolated from Wisconsin dairy cows. J. Dairy Sci. 2015, 98, 4521–4534. [Google Scholar] [CrossRef]
- Bortolaia, V.; Kaas, R.S.; Ruppe, E.; Roberts, M.C.; Schwarz, S.; Cattoir, V.; Philippon, A.; Allesoe, R.L.; Rebelo, A.R.; Florensa, A.F.; et al. ResFinder 4.0 for predictions of phenotypes from genotypes. J. Antimicrob. Chemother. 2020, 75, 3491–3500. [Google Scholar] [CrossRef]
- Hand, D.J. Measuring classifier performance: A coherent alternative to the area under the ROC curve. Mach. Learn. 2009, 77, 103–123. [Google Scholar] [CrossRef]
- Loy, J.D.; Clawson, M.L. Rapid typing of Mannheimia haemolytica major genotypes 1 and 2 using MALDI-TOF mass spectrometry. J. Microbiol. Methods 2017, 136, 30–33. [Google Scholar] [CrossRef]
- Narayanan, S.; Bates, H.; Confer, A.; Couger, B.; Ramachandran, A. Whole-genome sequence of multidrug-resistant Bibersteinia trehalosi strain OADDL-BT1. Microbiol. Resour. Announc. 2019, 8, e01690-18. [Google Scholar] [CrossRef] [PubMed]
- CLSI. VET01-A4 Performance Standards for Antimicrobial Disk and Dilution Susceptibility Test for Bacteria Isolated from Animals: Approved Standard-Fourth Edition; Watts, J.E.A., Ed.; CLSA: Wayne, PA, USA, 2013; Volume 33. [Google Scholar]
- CLSI. VET01-S2 Performance Standards for Antimicrobial Disk and Dilution Susceptibility Tests for Bacteria Isolated from Animals; Second Information Supplement; Watts, J.E.A., Ed.; CLSA: Wayne, PA, USA, 2013; Volume 33. [Google Scholar]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Robin, X.; Turck, N.; Hainard, A.; Tiberti, N.; Lisacek, F.; Sanchez, J.C.; Muller, M. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinform. 2011, 12, 77. [Google Scholar] [CrossRef] [PubMed]
- Zweig, M.H.; Campbell, G. Receiver-operating characteristic (ROC) plots: A fundamental evaluation tool in clinical medicine. Clin. Chem. 1993, 39, 561–577. [Google Scholar] [CrossRef]
- Melo, F. Area under the ROC Curve. In Encyclopedia of Systems Biology; Dubitzky, W.W.O., Cho, K.H., Yokota, H., Eds.; Springer: New York, NY, USA, 2013. [Google Scholar]
- Dohoo, I.R.; Martin, W.; Stryhn, H.E. Veterinary Epidemiologic Research, 2nd ed.; VER Inc.: Charlottetown, PEI, Canada, 2009; pp. 97–98. [Google Scholar]
- Mabmud, S.M. Cohen’s Kappa. In Encyclopedia of Research Design; Salkind, N.J., Ed.; SAGE Publications, Inc.: Thousand Oaks, CA, USA, 2010. [Google Scholar] [CrossRef]
- Steinberg, D.M.; Fine, J.; Chappell, R. Sample size for positive and negative predictive value in diagnostic research using case-control designs. Biostatistics 2009, 10, 94–105. [Google Scholar] [CrossRef]
- Riley, R.D.; Ahmed, I.; Debray, T.P.A.; Willis, B.H.; Noordzij, J.P.; Higgins, J.P.; Deeks, J.J. Summarising and validating test accuracy results across multiple studies for use in clinical practice. Stat. Med. 2015, 34, 2081–2103. [Google Scholar] [CrossRef]

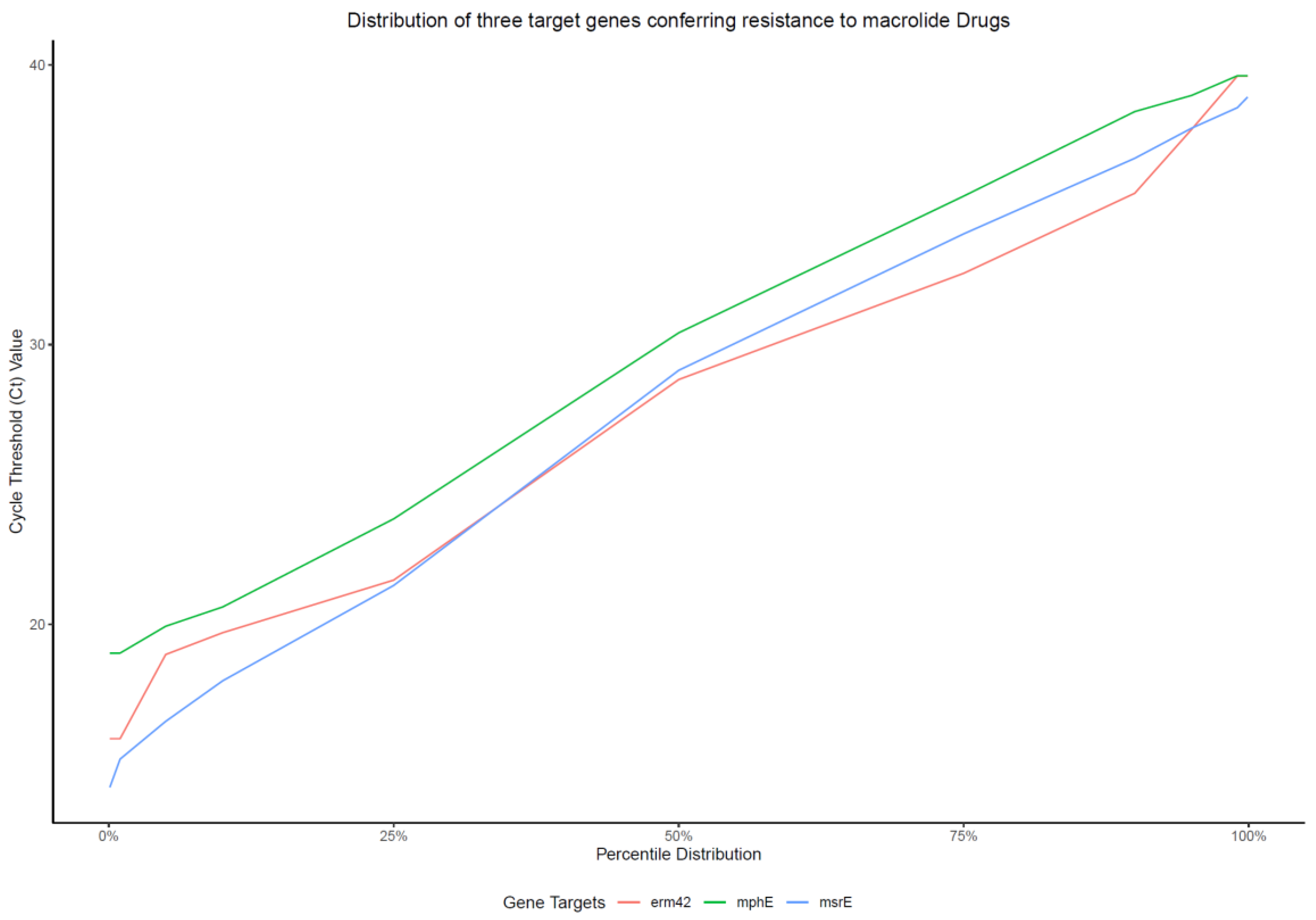
| Target | CFU/rxn | Ct 1 | CD 2 | RE 3 |
|---|---|---|---|---|
| ICETetR | 3200 | 26.78 | ||
| ICETetR | 320 | 30.23 | ||
| ICETetR | 32 | 33.53 | ||
| ICETetR | 3.2 | 37.34 | 0.982 | 0.95 |
| erm42 | 3200 | 26.32 | ||
| erm42 | 320 | 29.85 | ||
| erm42 | 32 | 32.95 | ||
| erm42 | 3.2 | 36.17 | 0.986 | 1.00 |
| mph E | 3200 | 25.61 | ||
| mph E | 320 | 29.05 | ||
| mph E | 32 | 32.30 | ||
| mph E | 3.2 | 36.56 | 0.985 | 0.88 |
| msr E | 3200 | 25.44 | ||
| msr E | 320 | 28.82 | ||
| msrE | 32 | 32.04 | ||
| msr E | 3.2 | 35.70 | 0.985 | 0.98 |
| Sample Type 1 | Total Sample Size 2 | Occurrence of BRD Pathogens No. of Positive Samples (Prevalence, 95% CI) | |||
|---|---|---|---|---|---|
| M. haemolytica | P. multocida | H. somni | Positive for at Least One BRD Pathogen | ||
| Lung sample | 297 | 191 (64.3%, 58.7–69.5%) | 96 (32.3%, 27.3–37.8%) | 93 (31.3%, 26.3–36.8%) | 296 (99.7%, 98.1–100.0%) |
| Nasal sample | 111 | 72 (64.9%, 55.6–73.1%) | 80 (72.1%, 63.1–79.6%) | 29 (26.1%, 18.9–35.0%) | 108 (97.3%, 92.4–99.1%) |
| Others—skin/liver | 4 | 1 (25.0%, 4.6–69.9%) | 0 (0.0%, 0.0%–49.0%) | 1 (25.0%, 4.6–69.9%) | 2 (50%, 15.0–85.0%) |
| Missing | 4 | 1 (25.0%, 4.6–69.9%) | 1 (25.0%, 4.6–69.9%) | 1 (25.0%, 4.6–69.9%) | 1 (25.0%, 4.6–69.9%) |
| Total | 416 | 265 (63.7%, 59.0–68.2%) | 177 (42.5%, 37.9–47.3%) | 123 (29.6%, 25.4–34.1%) | 407 (97.8%, 95.9–98.8%) |
| Pathogen | Sample | Class | Antibiotics | No. of Total Samples | No. (Percentage) of Samples with 1 | |||
|---|---|---|---|---|---|---|---|---|
| R | I | R+I | S | |||||
| M. haemolytica | Lung | Tetracycline | Oxytetracycline | 191 | 78 (40.8%) | 3 (1.6%) | 81 (42.4%) | 110 (57.6%) |
| Macrolide | Tilmicosin | 191 | 69 (36.1%) | 5 (2.6%) | 74 (38.7%) | 117 (61.3%) | ||
| Macrolide | Tulathromycin | 191 | 64 (33.5%) | 3 (1.6%) | 67 (35.1%) | 124 (64.9%) | ||
| Macrolide | Tilmicosin or tulathromycin | 191 | 75 (39.3%) | 0 (0%) | 75 (39.3%) | 116 (60.7%) | ||
| Nasal | Tetracycline | Oxytetracycline | 72 | 3 (4.2%) | 2 (2.7%) | 5 (6.9%) | 67 (93.1%) | |
| Macrolide | Tilmicosin | 72 | 2 (2.8%) | 0 (0%) | 2 (2.8%) | 70 (97.2%) | ||
| Macrolide | Tulathromycin | 72 | 6 (8.3%) | 0 (0%) | 6 (8.3%) | 66 (91.7%) | ||
| Macrolide | Tilmicosin or tulathromycin | 72 | 6 (8.3%) | 0 (0%) | 6 (8.3%) | 66 (91.7%) | ||
| P. multocida | Lung | Tetracycline | Oxytetracycline | 96 | 28 (29.2%) | 2 (2.1%) | 30 (31.3%) | 66 (68.7%) |
| Macrolide | Tilmicosin | 95 | 15 (15.8%) | 1 (1.0%) | 16 (16.8%) | 79 (83.2%) | ||
| Macrolide | Tulathromycin | 96 | 9 (9.4%) | 1 (1.1%) | 10 (10.5%) | 86 (89.5%) | ||
| Macrolide | Tilmicosin or tulathromycin | 95 | 15 (15.8%) | 1 (1.0%) | 16 (16.8%) | 79 (83.2%) | ||
| Nasal | Tetracycline | Oxytetracycline | 78 | 15 (19.2%) | 0 (0%) | 15 (19.2%) | 63 (80.8%) | |
| Macrolide | Tilmicosin | 80 | 6 (7.5%) | 0 (0%) | 6 (7.5%) | 74 (92.5%) | ||
| Macrolide | Tulathromycin | 78 | 1 (1.3%) | 3 (3.8%) | 4 (5.1%) | 74 (94.9%) | ||
| Macrolide | Tilmicosin or tulathromycin | 80 | 7 (8.8%) | 0 (0%) | 7 (8.8%) | 73 (91.2%) | ||
| H. somni | Lung | Tetracycline | Oxytetracycline | 93 | 41 (44.1%) | 8 (8.6%) | 49 (52.7%) | 44 (47.3%) |
| Macrolide | Tilmicosin | 93 | 20 (21.5%) | 1 (1.1%) | 21 (22.6%) | 72 (77.4%) | ||
| Macrolide | Tulathromycin | 93 | 20 (21.5%) | 8 (8.6%) | 28 (30.1%) | 65 (69.9%) | ||
| Macrolide | Tilmicosin or tulathromycin | 93 | 26 (28.0%) | 6 (6.4%) | 32 (34.4%) | 61 (65.6%) | ||
| Nasal | Tetracycline | Oxytetracycline | 29 | 13 (44.8%) | 0 (0%) | 13 (44.8%) | 16 (55.2%) | |
| Macrolide | Tilmicosin | 29 | 2 (6.9%) | 1 (3.4%) | 3 (10.3%) | 26 (89.7%) | ||
| Macrolide | Tulathromycin | 28 | 5 (17.9%) | 2 (7.1%) | 7 (25.0%) | 21 (75.0%) | ||
| Macrolide | Tilmicosin or tulathromycin | 28 | 6 (21.4%) | 3 (10.7%) | 9 (32.1%) | 19 (67.9%) | ||
| At least one BRD pathogen | Lung | Tetracycline | Oxytetracycline | 296 | 124 (41.9%) | 8 (2.7%) | 132 (44.6%) | 164 (55.4%) |
| Macrolide | Tilmicosin | 295 | 95 (32.2%) | 6 (2.0%) | 101 (34.2%) | 194 (65.8%) | ||
| Macrolide | Tulathromycin | 296 | 87 (29.4%) | 8 (2.7%) | 95 (32.1%) | 197 (67.9%) | ||
| Macrolide | Tilmicosin or tulathromycin | 295 | 105 (35.6%) | 4 (1.3%) | 109 (36.9%) | 182 (63.1%) | ||
| Nasal | Tetracycline | Oxytetracycline | 108 | 23 (21.3%) | 2 (1.8%) | 25 (23.1%) | 83 (76.9%) | |
| Macrolide | Tilmicosin | 108 | 11 (10.2%) | 0 (0.0%) | 11 (10.2%) | 97 (89.8%) | ||
| Macrolide | Tulathromycin | 108 | 11 (10.2%) | 5 (4.6%) | 16 (14.8%) | 92 (85.2%) | ||
| Macrolide | Tilmicosin or tulathromycin | 108 | 19 (17.6%) | 2 (1.8%) | 21 (19.4%) | 87 (80.6%) | ||
| Class | Antibiotics | Antimicrobial Resistance Classification | |||
|---|---|---|---|---|---|
| Susceptible (S) | Intermediate (I) | Resistant (R) | “Resistant” (R+I) | ||
| Tetracycline | Oxytetracycline | ≤2 | >2 and ≤8 | >8 | >2 |
| Macrolide | Tilmicosin | ≤8 | >8 and ≤32 | >32 | >8 |
| Macrolide | Tulathromycin | ≤16 | >16 and ≤32 | >32 | >16 |
| BRD Pathogen | Sample | Class | Antibiotics | No. of Total Samples | No. of Samples with 1 | Optimal Cycle Threshold (Ct) | Se 2 (%) | Sp 2 (%) | AUC 2 (%) | Kappa (κ) | Prevalence (%) | PPV 2 (%) | NPV 2 (%) | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R+I | S | |||||||||||||
| M. haemolytica | Lung | Tetracycline | Oxytetracycline | 191 | 81 | 110 | 31.00 | 77.78 | 95.45 | 89.99 | 0.75 | 42.41 | 92.65 | 85.37 |
| Macrolide | Tilmicosin | 191 | 74 | 117 | 33.04 | 74.32 | 93.16 | 86.14 | 0.69 | 38.74 | 87.30 | 85.16 | ||
| Macrolide | Tulathromycin | 191 | 67 | 124 | 32.89 | 79.10 | 92.74 | 88.94 | 0.73 | 35.08 | 85.48 | 89.15 | ||
| Macrolide | Tilmicosin or tulathromycin | 191 | 75 | 116 | 33.04 | 73.33 | 93.10 | 85.45 | 0.68 | 39.27 | 87.30 | 84.38 | ||
| Nasal | Tetracycline | Oxytetracycline | 72 | 5 | 67 | 32.26 | 100.00 | 79.10 | 92.54 | 0.34 | 6.94 | 21.83 | 98.35 | |
| Macrolide | Tilmicosin | 72 | 2 | 70 | 21.42 | 100.00 | 100.00 | 100.00 | 1.00 | 2.78 | 52.25 | 99.17 | ||
| Macrolide | Tulathromycin | 72 | 6 | 66 | 30.73 | 50.00 | 86.36 | 63.89 | 0.25 | 8.33 | 25.00 | 95.00 | ||
| Macrolide | Tilmicosin or tulathromycin | 72 | 6 | 66 | 30.73 | 50.00 | 86.36 | 63.89 | 0.25 | 8.33 | 25.00 | 95.00 | ||
| P. multocida | Lung | Tetracycline | Oxytetracycline | 96 | 30 | 66 | 36.10 | 83.33 | 86.36 | 86.52 | 0.67 | 31.25 | 73.53 | 91.94 |
| Macrolide | Tilmicosin | 95 | 16 | 79 | 32.91 | 56.25 | 89.87 | 72.23 | 0.45 | 16.84 | 52.94 | 91.03 | ||
| Macrolide | Tulathromycin | 96 | 10 | 86 | 32.91 | 80.00 | 89.53 | 84.94 | 0.53 | 10.42 | 47.06 | 97.47 | ||
| Macrolide | Tilmicosin or tulathromycin | 95 | 16 | 79 | 32.91 | 56.25 | 89.87 | 72.23 | 0.45 | 16.84 | 52.94 | 91.03 | ||
| Nasal | Tetracycline | Oxytetracycline | 78 | 15 | 63 | 29.35 | 66.67 | 92.06 | 88.04 | 0.59 | 19.23 | 66.67 | 92.06 | |
| Macrolide | Tilmicosin | 80 | 6 | 74 | 36.40 | 66.67 | 62.16 | 59.01 | 0.10 | 7.50 | 12.50 | 95.83 | ||
| Macrolide | Tulathromycin | 78 | 4 | 74 | 31.47 | 50.00 | 78.38 | 58.78 | 0.11 | 5.13 | 11.11 | 96.67 | ||
| Macrolide | Tilmicosin or tulathromycin | 80 | 7 | 73 | 32.22 | 42.86 | 76.71 | 56.36 | 0.11 | 8.75 | 15.00 | 93.33 | ||
| H. somni | Lung | Tetracycline | Oxytetracycline | 93 | 49 | 44 | 36.28 | 81.63 | 72.73 | 75.72 | 0.55 | 52.69 | 76.92 | 78.05 |
| Macrolide | Tilmicosin | 93 | 21 | 72 | 33.08 | 61.90 | 79.17 | 67.29 | 0.37 | 22.58 | 46.43 | 87.69 | ||
| Macrolide | Tulathromycin | 93 | 28 | 65 | 31.67 | 60.71 | 87.69 | 75.36 | 0.50 | 30.11 | 68.00 | 83.82 | ||
| Macrolide | Tilmicosin or tulathromycin | 93 | 32 | 61 | 33.08 | 62.50 | 86.89 | 76.95 | 0.51 | 34.41 | 71.43 | 81.54 | ||
| Nasal | Tetracycline | Oxytetracycline | 29 | 13 | 16 | 32.85 | 92.31 | 56.25 | 62.50 | 0.47 | 44.83 | 63.16 | 90.00 | |
| Macrolide | Tilmicosin | 29 | 3 | 26 | 30.88 | 66.67 | 65.38 | 52.56 | 0.15 | 10.34 | 18.18 | 94.44 | ||
| Macrolide | Tulathromycin | 28 | 7 | 21 | 27.19 | 100.00 | 38.10 | 68.03 | -0.36 | 25.00 | 10.54 | 67.85 | ||
| Macrolide | Tilmicosin or tulathromycin | 28 | 9 | 19 | 26.83 | 100.00 | 36.84 | 66.67 | −0.39 | 32.14 | 12.45 | 59.49 | ||
| At least one BRD pathogen | Lung | Tetracycline | Oxytetracycline | 296 | 132 | 164 | 36.06 | 81.06 | 82.93 | 85.29 | 0.64 | 44.59 | 79.26 | 84.47 |
| Macrolide | Tilmicosin | 295 | 101 | 194 | 33.08 | 69.31 | 89.69 | 81.25 | 0.61 | 34.24 | 77.78 | 84.88 | ||
| Macrolide | Tulathromycin | 296 | 95 | 201 | 32.89 | 72.63 | 90.55 | 84.29 | 0.64 | 32.09 | 78.41 | 87.50 | ||
| Macrolide | Tilmicosin or tulathromycin | 295 | 109 | 186 | 33.08 | 67.89 | 91.40 | 81.94 | 0.62 | 36.95 | 82.22 | 82.93 | ||
| Nasal | Tetracycline | Oxytetracycline | 108 | 25 | 83 | 32.81 | 88.00 | 79.52 | 90.07 | 0.56 | 23.15 | 56.41 | 95.65 | |
| Macrolide | Tilmicosin | 108 | 11 | 97 | 31.82 | 63.64 | 82.47 | 71.42 | 0.30 | 10.19 | 29.17 | 95.24 | ||
| Macrolide | Tulathromycin | 108 | 16 | 92 | 31.47 | 43.75 | 83.70 | 57.54 | 0.24 | 14.81 | 31.82 | 89.53 | ||
| Macrolide | Tilmicosin or tulathromycin | 108 | 21 | 87 | 31.82 | 42.86 | 82.76 | 57.85 | 0.24 | 19.44 | 37.50 | 85.71 | ||
| Class | Antibiotics | Lung Sample | Nasal Sample | ||
|---|---|---|---|---|---|
| Optimal Cycle Threshold (Ct) | Kappa (κ) | Optimal Cycle Threshold (Ct) | Kappa (κ) | ||
| Tetracycline | Oxytetracycline | 35.66 | 0.61 | 33.27 | 0.49 |
| Macrolide | Tilmicosin | 33.12 | 0.61 | - | - |
| Macrolide | Tulathromycin | 32.64 | 0.63 | - | - |
| Macrolide | Tilmicosin or tulathromycin | 33.12 | 0.62 | - | - |
| Target | Primer/Probe | Sequence (5′-3′) | Size | Reference |
|---|---|---|---|---|
| tetR | ICEtetR-F | TTTGGCTTTCTTGATGCTCTTG | 71 | This paper |
| ICEtetR-R | GTGATGCTGGGTTTAGTCTATCT | |||
| ICEtetR-P (CY5/TAO-IAB-RQ) | CGCAATAGAGCTTAATGCATACACGGC | |||
| erm42 | erm42-F | GCCGTTAATGCTATTGAGTTCG | 105 | This paper |
| erm42-R | CGGCTTCAATAATAGACACATTTGA | |||
| erm42-P (FAM/ZEN-IAB-FQ) | AGTGTATTGGCTGATAAGTTGAGCCATGA | |||
| msrE | msrE-F | GGGTGGTTACTCGGATTACTTG | 88 | This paper |
| msrE-R | CTCCCGTTCCTTCATCATCAG | |||
| msrE-P (Texas Red/IAB-RQ) | AGCGACAACACCAAGCCGTAGAAT | |||
| mphE | mphE-F | TTGGAAACCCGCTACAGAAA | 113 | This paper |
| mphE-R | GCTCCATCCTTTGAAGCTAGT | |||
| mphE-P (JOE/ZEN-IAB-FQ) | TGATGTTCTATGGGCAGATTTCACCCA |
| Multiplex qPCR Assay | Culture-Based Gold-Standard Test | ||
|---|---|---|---|
| “R” 1 | S 2 | ||
| “R” 1 | True Positive (TP) | False Positive (FP) | PPV 3 = TP/(TP + FP) |
| S 2 | False Negative (FN) | True Negative (TN) | NPV 3 = T N/(TN + FN) |
| Se 3 = TP/(TP + FN) | Sp 3 = TN/(TN + FP) | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dutta, E.; Loy, J.D.; Deal, C.A.; Wynn, E.L.; Clawson, M.L.; Clarke, J.; Wang, B. Development of a Multiplex Real-Time PCR Assay for Predicting Macrolide and Tetracycline Resistance Associated with Bacterial Pathogens of Bovine Respiratory Disease. Pathogens 2021, 10, 64. https://doi.org/10.3390/pathogens10010064
Dutta E, Loy JD, Deal CA, Wynn EL, Clawson ML, Clarke J, Wang B. Development of a Multiplex Real-Time PCR Assay for Predicting Macrolide and Tetracycline Resistance Associated with Bacterial Pathogens of Bovine Respiratory Disease. Pathogens. 2021; 10(1):64. https://doi.org/10.3390/pathogens10010064
Chicago/Turabian StyleDutta, Enakshy, John Dustin Loy, Caitlyn A. Deal, Emily L. Wynn, Michael L. Clawson, Jennifer Clarke, and Bing Wang. 2021. "Development of a Multiplex Real-Time PCR Assay for Predicting Macrolide and Tetracycline Resistance Associated with Bacterial Pathogens of Bovine Respiratory Disease" Pathogens 10, no. 1: 64. https://doi.org/10.3390/pathogens10010064
APA StyleDutta, E., Loy, J. D., Deal, C. A., Wynn, E. L., Clawson, M. L., Clarke, J., & Wang, B. (2021). Development of a Multiplex Real-Time PCR Assay for Predicting Macrolide and Tetracycline Resistance Associated with Bacterial Pathogens of Bovine Respiratory Disease. Pathogens, 10(1), 64. https://doi.org/10.3390/pathogens10010064






