Nano-TiO2 in Hydraulic Lime–Metakaolin Mortars for Restoration Projects: Physicochemical and Mechanical Assessment
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mortar Mixes Design
2.2. Evaluation of Mixtures
3. Results and Discussion
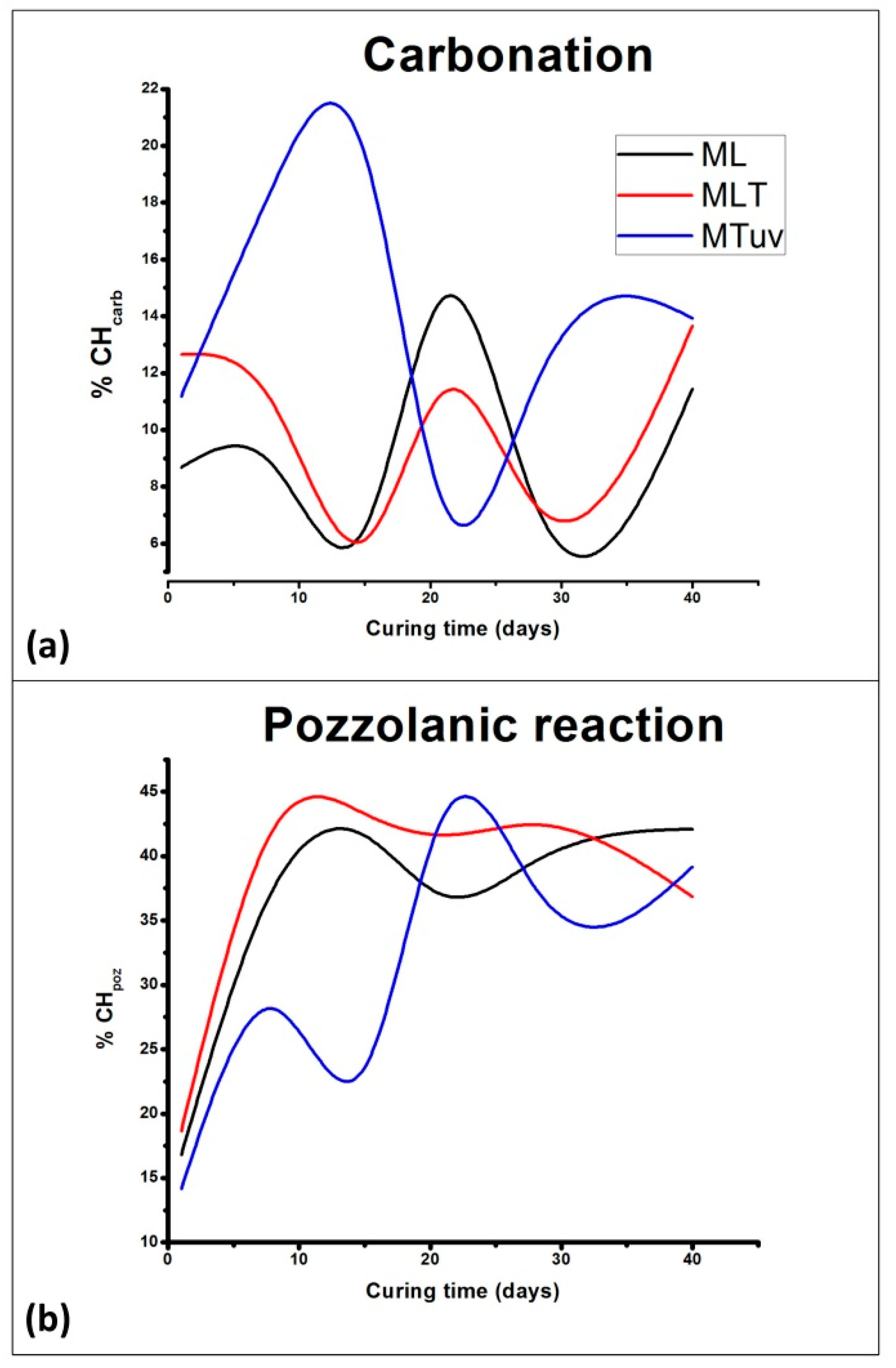
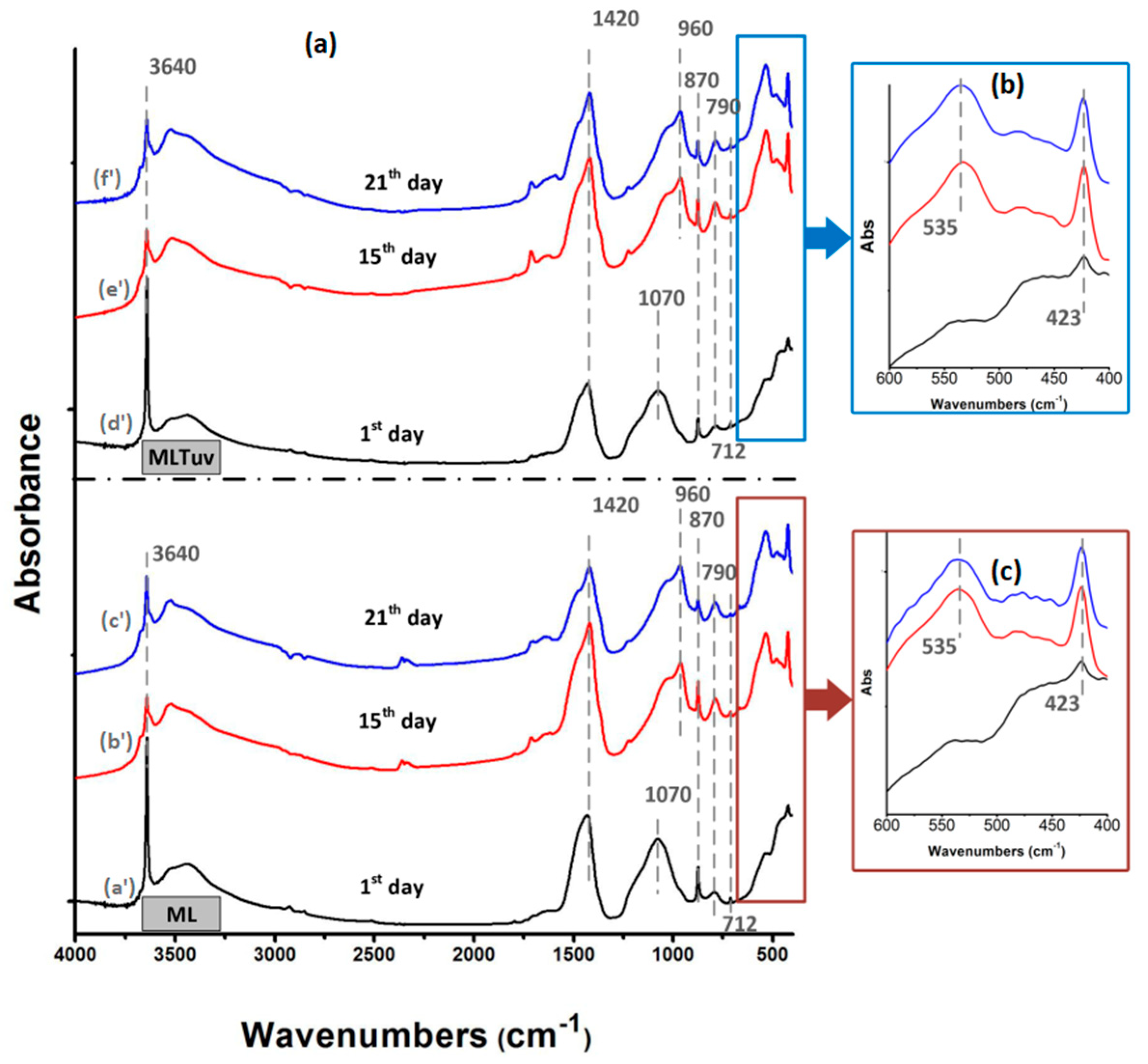
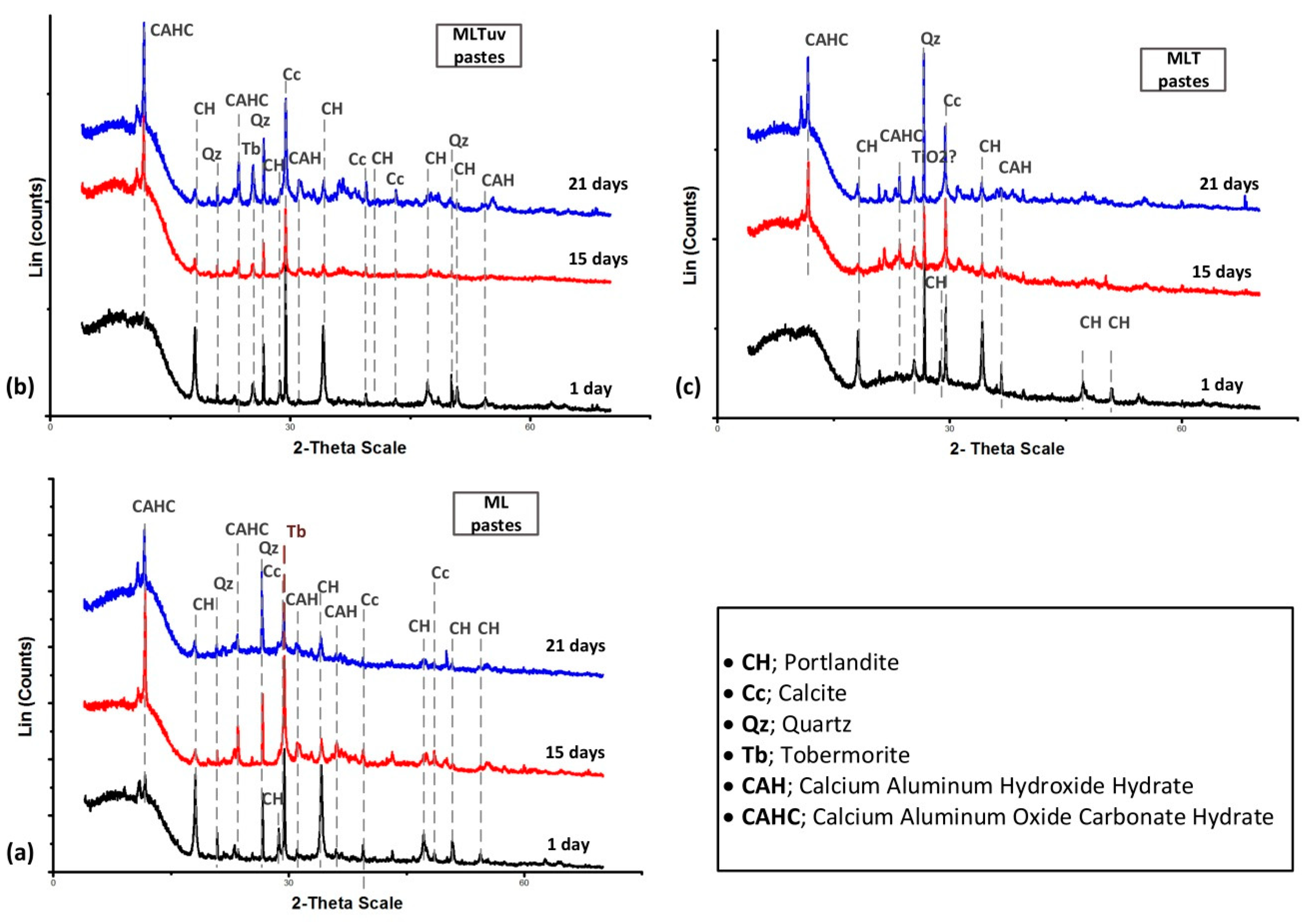
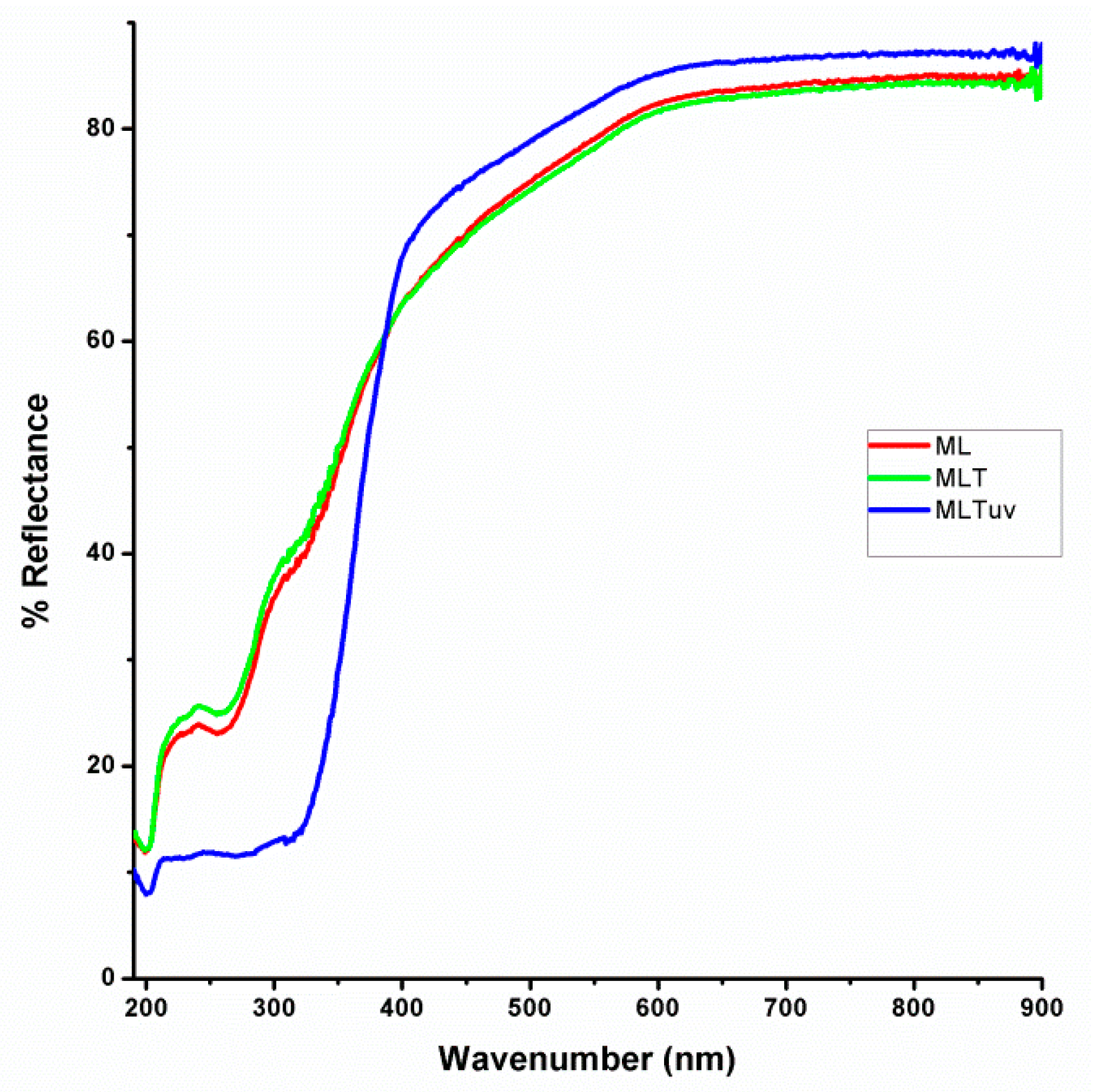
3.1. Physico-Chemical Study
3.1.1. Thermal Analysis (DTA)
3.1.2. Infrared Spectroscopy (FTIR)
3.1.3. Mineralogical Analysis (XRD)
3.1.4. UV-Vis Measurements
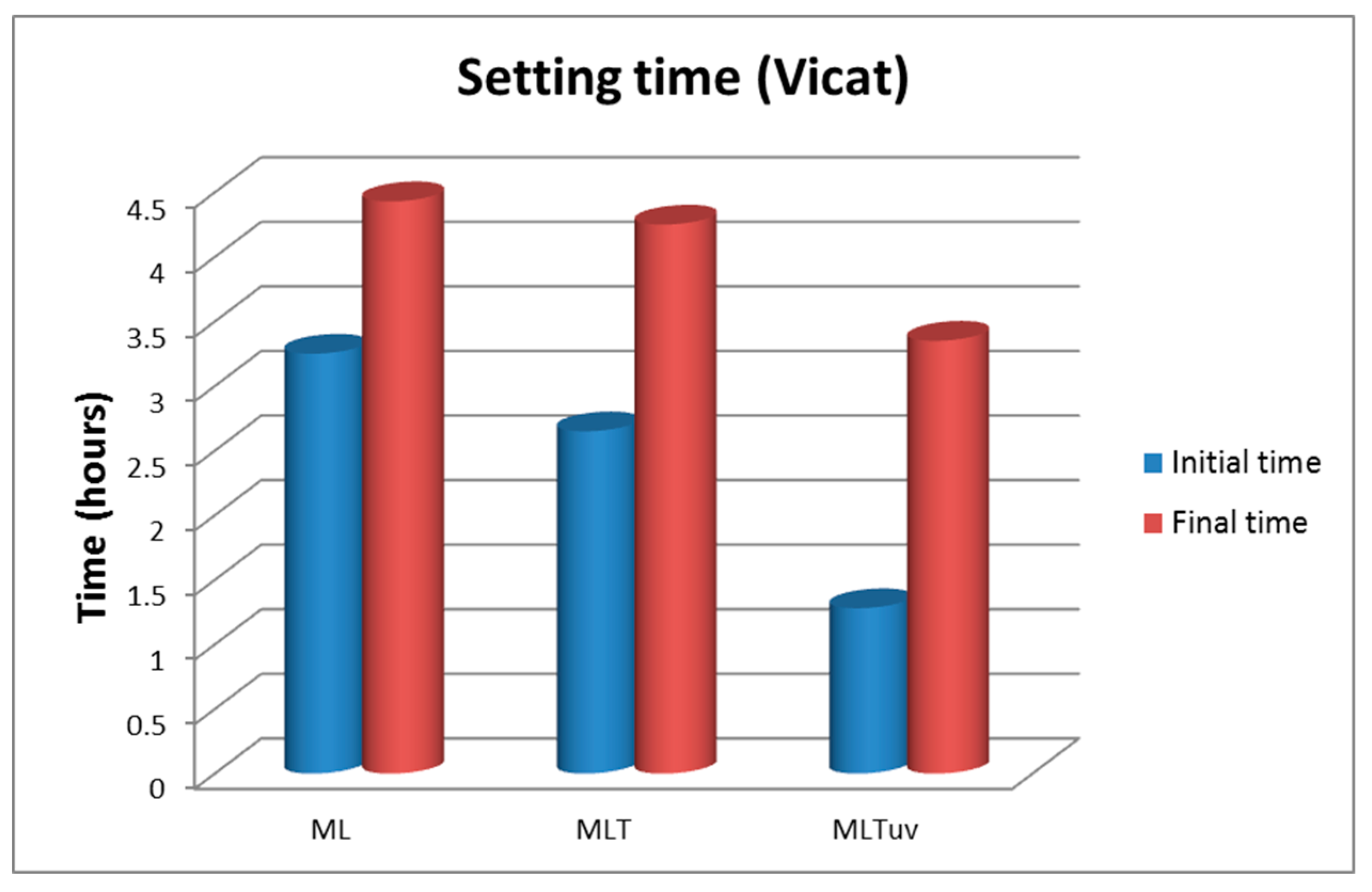
3.1.5. Setting Time
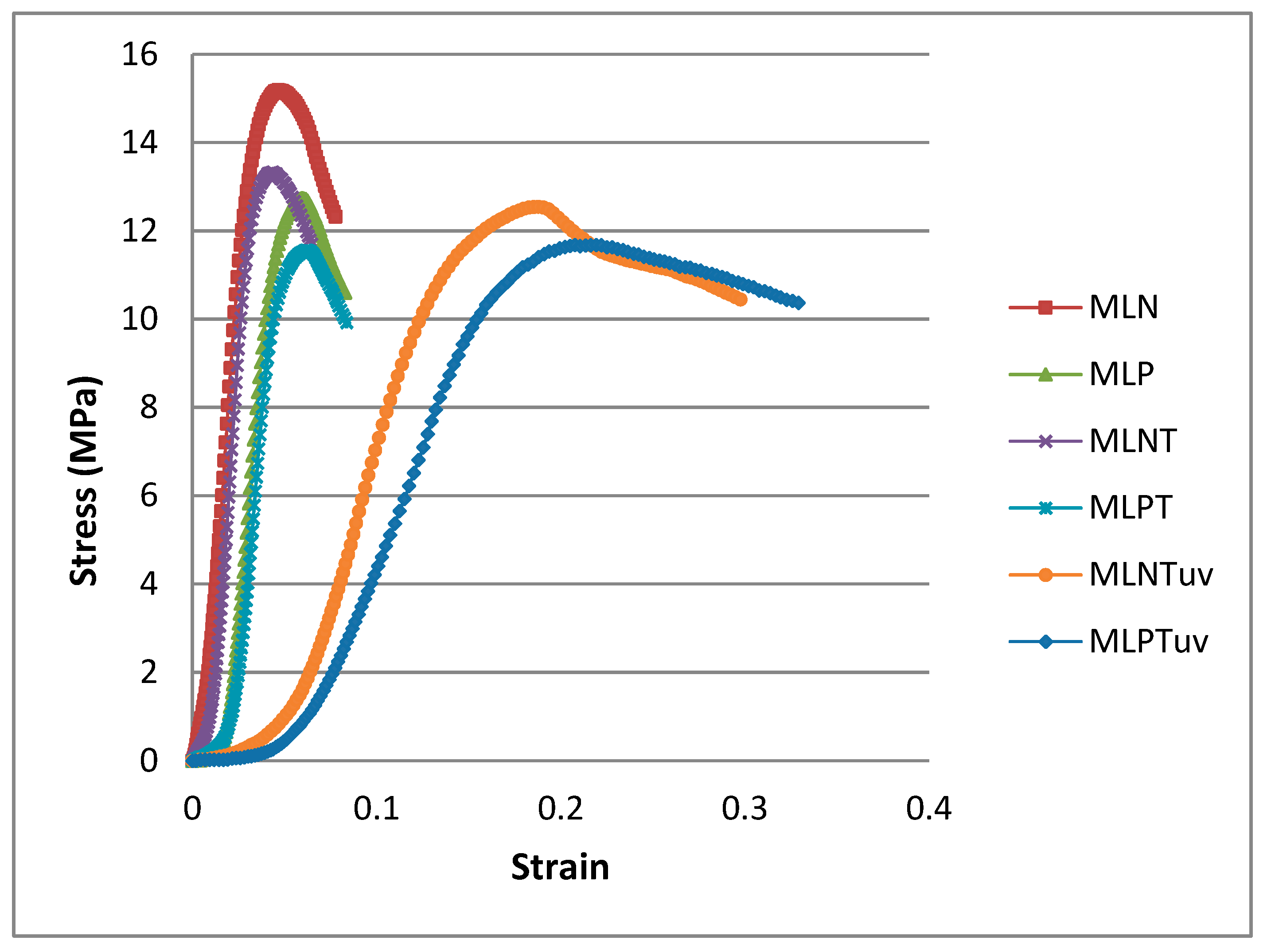
3.2. Mechanical Study
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Maravelaki-Kalaitzaki, P.; Bakolas, A.; Moropoulou, A. Physico-chemical study of cretan ancient mortars. Cem. Concr. Res. 2003, 33, 651–661. [Google Scholar] [CrossRef]
- Moropoulou, A.; Bakolas, A.; Anagnostopoulou, S. Composite materials in ancient structures. Cem. Concr. Compos. 2005, 27, 295–300. [Google Scholar] [CrossRef]
- Ergenc, D.; Fort, R.; Silva, A.S.; Veiga, R.; Arauz, D.S. The effects of DiloCarB as carbonation accelerator on the properties of lime mortars. Mater. Struct. 2018, 51. [Google Scholar] [CrossRef]
- Apostolopoulou, M.; Aggelakopoulou, E.; Bakolas, A.; Moropoulou, A. Compatible mortars for the sustainable conservation of stone in masonries. In Advanced Materials for the Conservation of Stone; Springer: Cham, Switzerland, 2018; pp. 97–123. [Google Scholar]
- Papayianni, I.; Stefanidou, M. Durability aspects of ancient mortars of the archeological site of olynthos. J. Cult. Herit. 2007, 8, 193–196. [Google Scholar] [CrossRef]
- Nežerka, V.; Antoš, J.; Tesárek, P.; Zeman, J. Performance of repair mortars used in bed joints of masonry piers. In Proceedings of the Civil-Comp Proceedings—15th International Conference on Civil, Structural and Environmental Engineering Computing, Prague, Czech Republic, 1–4 September 2015. [Google Scholar]
- Lippiello, M.; Ceraldi, C.; D’Ambra, C.; Lignola, G.P. Mechanical characterization of ancient pozzolanic mortars with additions of brick and tuff dust: A comparative investigation. In Proceedings of the 10th International Conference on Structural Analysis of Historical Constructions (SAHC 2016), Leuven, Belgium, 13–15 September 2016; pp. 558–564. [Google Scholar]
- Aalil, I.; Badreddine, D.; Beck, K.; Brunetaud, X.; Cherkaoui, K.; Chaaba, A.; Al-Mukhtar, M. Valorization of crushed bricks in lime-based mortars. Constr. Build. Mater. 2019, 226, 555–563. [Google Scholar] [CrossRef]
- Karatasios, I.; Amenta, M.; Kilikoglou, V. Hydraulic mortars for joining archaeological stone fragments—A methodological approach. Struct. Integr. Procedia 2018, 10, 211–218. [Google Scholar] [CrossRef]
- Szemerey-Kiss, B.; Torok, A. Failure mechanisms of repair mortar stone interface assessed by pull-off strength tests, failure mechanisms of repair mortar stone interface assessed by pull-off strength tests. Bull. Eng. Geol. Environ. 2017, 76, 159–167. [Google Scholar] [CrossRef]
- Sierra-Fernandez, A.; Gomez-Villalba, L.S.; Rabanal, M.E.; Fort, R. New nanomaterials for applications in conservation and restoration of stony materials: A review. Mater. Construcción 2017, 67, 107. [Google Scholar] [CrossRef]
- Kapridaki, C.; Verganelaki, A.; Dimitriadou, P.; Maravelaki-Kalaitzaki, P. Conservation of monuments by a three-layered compatible treatment of TEOS-Nano-Calcium oxalate consolidant and TEOS-PDMS-TiO2 Hydrophobic/Photoactive hybrid nanomaterials. Materials 2018, 11, 684. [Google Scholar] [CrossRef] [PubMed]
- Aranzabe, E.; Blanco, M.; Goitandia, A.M.; Vidal, K.; Casado, M.; Cubillo, J. Preparation and characterisation of photocatalytic pigments for architectural mortar based on ultramarine blue. J. Sol-Gel Sci. Technol. 2019, 1–8. [Google Scholar] [CrossRef]
- Senff, L.; Ascensão, G.; Ferreira, V.M.; Seabra, M.P.; Labrincha, J.A. Development of multifunctional plaster using nano-TiO2 and distinct particle size cellulose fibers. Energy Build. 2018, 158, 721–735. [Google Scholar] [CrossRef]
- Perez-Nicolas, M.; Navarro-Blasco, I.Í.; Duran, A.; Sirera, R.; Fernandez-Alvarez, J.M.; Alvarez, J.I. Obtaining of self-cleaning repair air lime mortars with photocatalysts. In Proceedings of the En: HMC 2016 4th Historic Mortar Conference Scientific Program, Session XV, Santorini, Greece, 10–12 October 2016. [Google Scholar]
- Kapridaki, C.; Xynidis, N.; Vazgiouraki, E.; Kallithrakas-Kontos, N.; Maravelaki-Kalaitzaki, P. Characterization of photoactive Fe-TiO2 lime coatings for building protection: The role of iron content. Materials 2019, 12, 1847. [Google Scholar] [CrossRef] [PubMed]
- Maravelaki, N.; Kapridaki, C.; Lionakis, E.; Verganelaki, A. Improvement of properties of hydraulic mortars with addition of nano-titania. In Adhesives: Mechanical Properties, Technologies and Economic Importance; Nova Science: Hauppauge, NY, USA, 2014; Chapter 4; pp. 79–93. [Google Scholar]
- Chen, J.; Kou, S.; Poon, C. Hydration and properties of nano-TiO2 blended cement composites. Cem. Concr. Compos. 2012, 34, 642–649. [Google Scholar] [CrossRef]
- Gameiro, A.; Santos Silva, A.; Grilo, J.; Branco, T.; Veiga, R.; Velosa, A. Physical and chemical assessment of lime–metakaolin mortars: Influence of binder: Aggregate ratio. Cem. Concr. Compos. 2014, 45, 264–271. [Google Scholar] [CrossRef]
- Duran, A.; González-Sánchez, J.F.; Fernández, J.M.; Sirera, R.; Navarro-Blasco, Í.; Alvarez, J.I. Influence of two polymer-based Superplasticizers (poly-naphthalene sulfonate, PNS, and lignosulfonate, LS) on compressive and flexural strength, freeze-thaw, and sulphate attack resistance of lime-metakaolin grouts. Polymers 2018, 10, 824. [Google Scholar] [CrossRef] [PubMed]
- Yu, P.; Kirkpatrick, R.J.; Poe, B.; McMillan, P.F.; Cong, X. Structure of calcium silicate hydrate (C-S-H): Near-, mid-, and far-infrared spectroscopy. J. Am. Ceram. Soc. 1999, 82, 742–748. [Google Scholar] [CrossRef]
- Kaklis, K.; Maurigiannakis, S.; Agioutantis, Z.; Maravelaki-Kalaitzaki, P. Characterization of pozzolanic lime mortars used as filling material in shaped grooves for restoring member connections in ancient monuments. Int. J. Archit. Herit. 2018, 12, 75–90. [Google Scholar] [CrossRef]
- Maravelaki-Kalaitzaki, P.; Agioutantis, Z.; Lionakis, E.; Stavroulaki, M.; Perdikatsis, V. Physico-chemical and mechanical characterization of hydraulic mortars containing nano-titania for restoration applications. Cem. Concr. Compos. 2013, 36, 33–41. [Google Scholar] [CrossRef]
- Amenta, M.; Karatasios, I.; Maravelaki, P.; Kilikoglou, P. Monitoring of self-healing phenomena towards enhanced sustainability of historic mortars. Appl. Phys. 2016, 122, 554. [Google Scholar] [CrossRef]
| Code | Binder | TiO2 | Aggregates | Consistency (mm) | |||||
|---|---|---|---|---|---|---|---|---|---|
| L | M | Carbonaceous | Silicate | B/A | W/B | ||||
| MORTARS | MLN | 60 | 40 | - | 100 | - | 1 | 1.25 | 136.96 |
| MLNT | 60 | 40 | 6 | 100 | - | 1 | 1.19 | 136.52 | |
| MLNTuv | 60 | 40 | 6 | 100 | - | 1 | 1.19 | 135.94 | |
| MLP | 60 | 40 | - | - | 100 | 1 | 1.35 | 148.12 | |
| MLPT | 60 | 40 | 6 | - | 100 | 1 | 1.20 | 138.25 | |
| MLPTuv | 60 | 40 | 6 | - | 100 | 1 | 1.22 | 137.16 | |
| PASTES | ML | 60 | 40 | - | - | - | - | 1.75 | - |
| MLT | 60 | 40 | 6 | - | - | - | 1.65 | - | |
| MLTuv | 60 | 40 | 6 | - | - | - | 1.65 | - | |
| Code | Curing Time | Fc (MPa) a | Fb (MPa) b | E (GPa) c | Max Strain d % | T (J m·−3) e |
|---|---|---|---|---|---|---|
| MLN | 28 days | 15.03 (±0.36) | 2.96 (±0.28) | 0.67 | 7.93 | 0.8 |
| 3 months | 9.85 (±0.13) | 2.63 (±0.5) | ||||
| 6 months | 12.53 (±2.2) | 3.85 (±0.41) | ||||
| MLNT | 28 days | 13.54 (±0.45) | 3.82 (±0.28) | 0.66 | 7.12 | 0.59 |
| 3 months | 8.68 (±0.4) | 2.48 (±0.33) | ||||
| 6 months | 11.67 (±0.05) | 3.34 (±0.1) | ||||
| MLNTuv | 28 days | 12.57 (±0.2) | 3.29 (±0.36) | 0.16 | 29.58 | 2.39 |
| 3 months | 11.35 (±0.03) | 1.8 (±0.12) | ||||
| 6 months | 17.18 (±0.57) | 2.53 (±0.35) | ||||
| MLP | 28 days | 12.82 (±0.23) | 2.44 (±0.13) | 0.53 | 7.9 | 0.66 |
| 3 months | 10.26 (±2.43) | 2.85 (±0.27) | ||||
| 6 months | 15.29 (±0.22) | 3.34 (±0.17) | ||||
| MLPT | 28 days | 11.56 (±0.32) | 2.55 (±0.24) | 0.43 | 9.64 | 0.66 |
| 3 months | 10.12 (±0.33) | 1.14 (±0.26) | ||||
| 6 months | 11.06 (±0.36) | 3.19 (±0.08) | ||||
| MLPTuv | 28 days | 11.78 (±0.53) | 2.70 (±0.19) | 0.15 | 30.48 | 2.19 |
| 3 months | 11.06 (±0.36) | 1.30 (±0.57) | ||||
| 6 months | 15.60 (±0.12) | 2.31 (±0.21) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kapetanaki, K.; Kapridaki, C.; Maravelaki, P.-N. Nano-TiO2 in Hydraulic Lime–Metakaolin Mortars for Restoration Projects: Physicochemical and Mechanical Assessment. Buildings 2019, 9, 236. https://doi.org/10.3390/buildings9110236
Kapetanaki K, Kapridaki C, Maravelaki P-N. Nano-TiO2 in Hydraulic Lime–Metakaolin Mortars for Restoration Projects: Physicochemical and Mechanical Assessment. Buildings. 2019; 9(11):236. https://doi.org/10.3390/buildings9110236
Chicago/Turabian StyleKapetanaki, Kali, Chrysi Kapridaki, and Pagona-Noni Maravelaki. 2019. "Nano-TiO2 in Hydraulic Lime–Metakaolin Mortars for Restoration Projects: Physicochemical and Mechanical Assessment" Buildings 9, no. 11: 236. https://doi.org/10.3390/buildings9110236
APA StyleKapetanaki, K., Kapridaki, C., & Maravelaki, P.-N. (2019). Nano-TiO2 in Hydraulic Lime–Metakaolin Mortars for Restoration Projects: Physicochemical and Mechanical Assessment. Buildings, 9(11), 236. https://doi.org/10.3390/buildings9110236






