The Adsorption Kinetic Parameters of Co2+ Ions by α-C2SH
Abstract
:1. Introduction
2. Materials and Methods
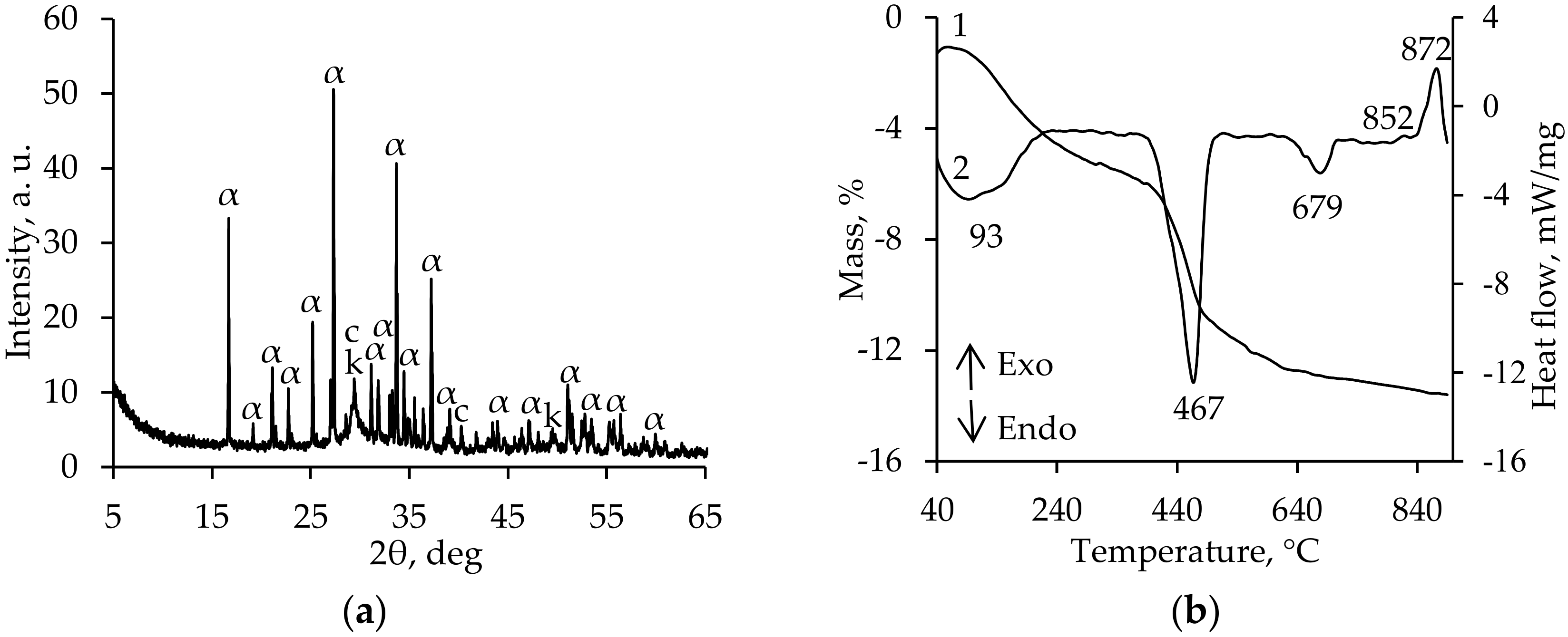
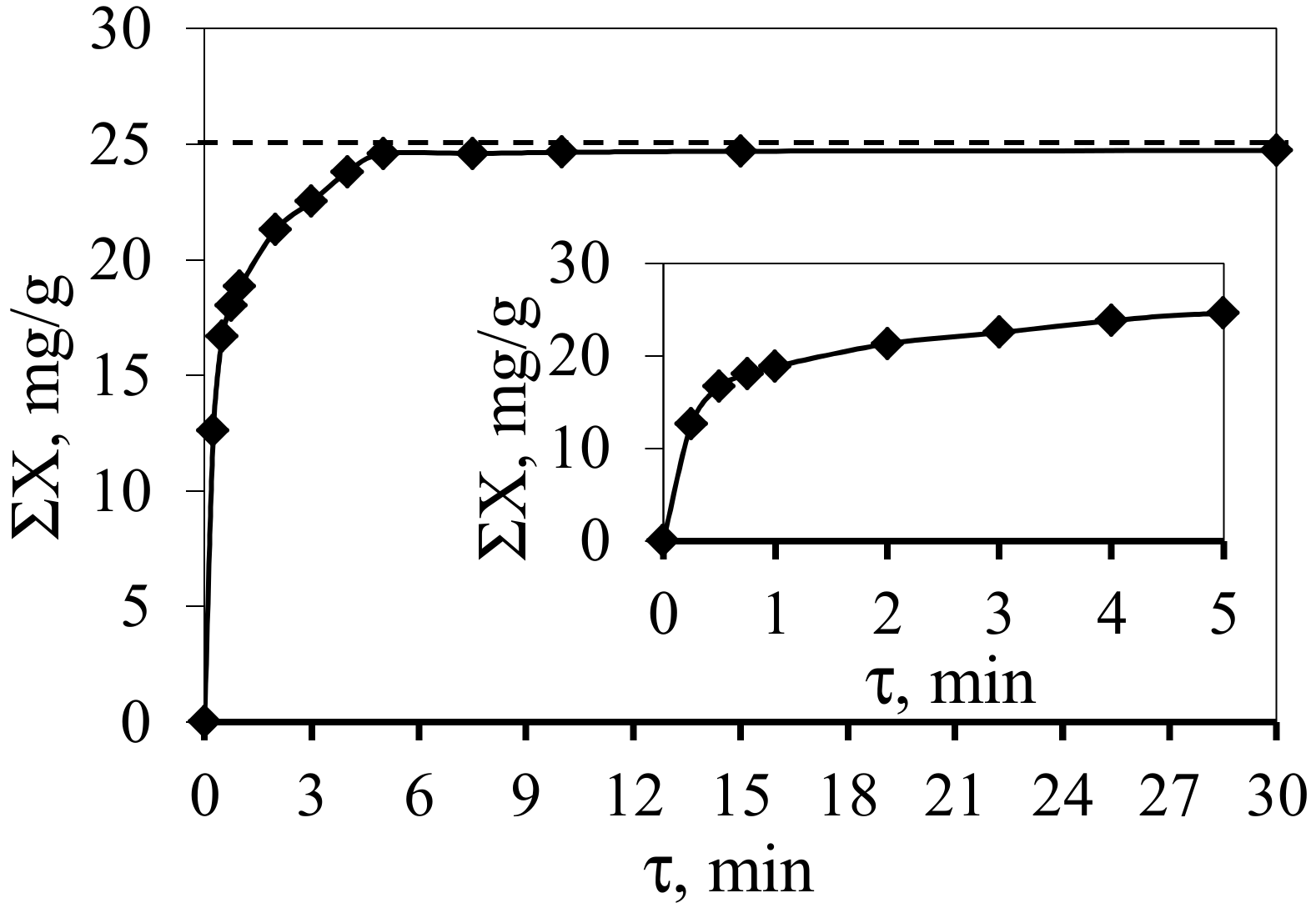
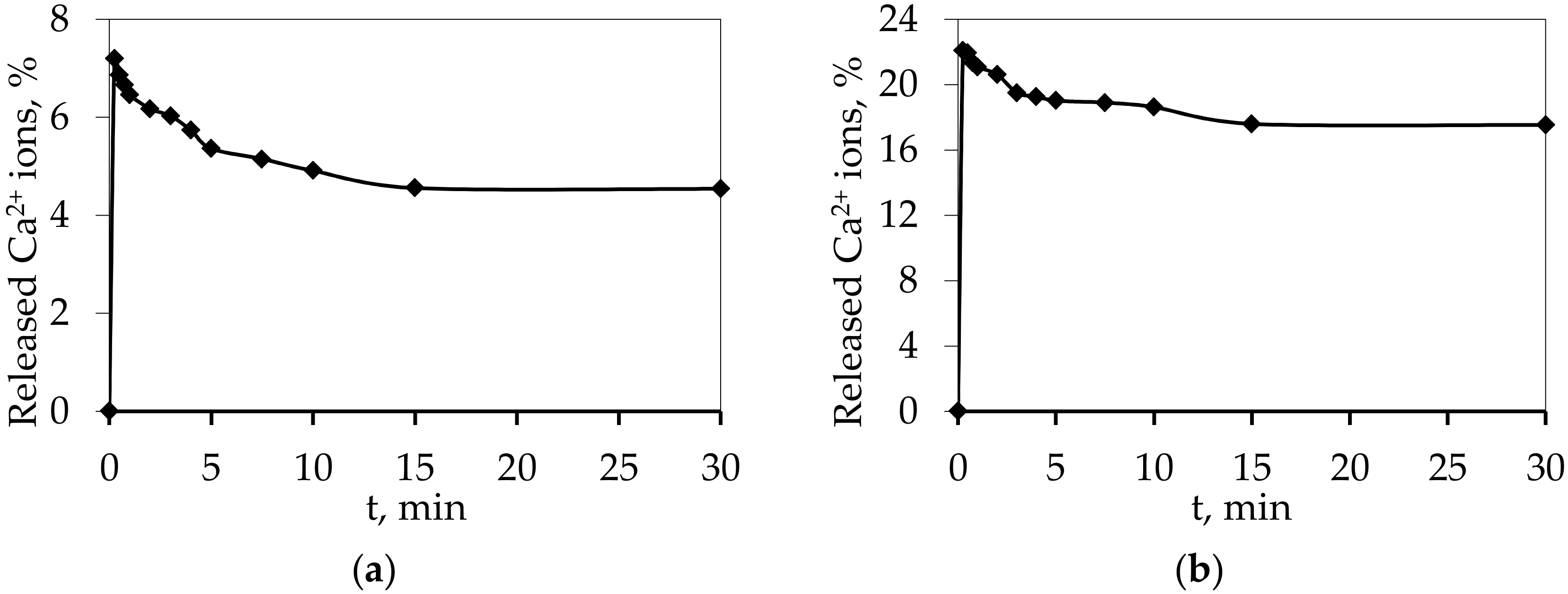
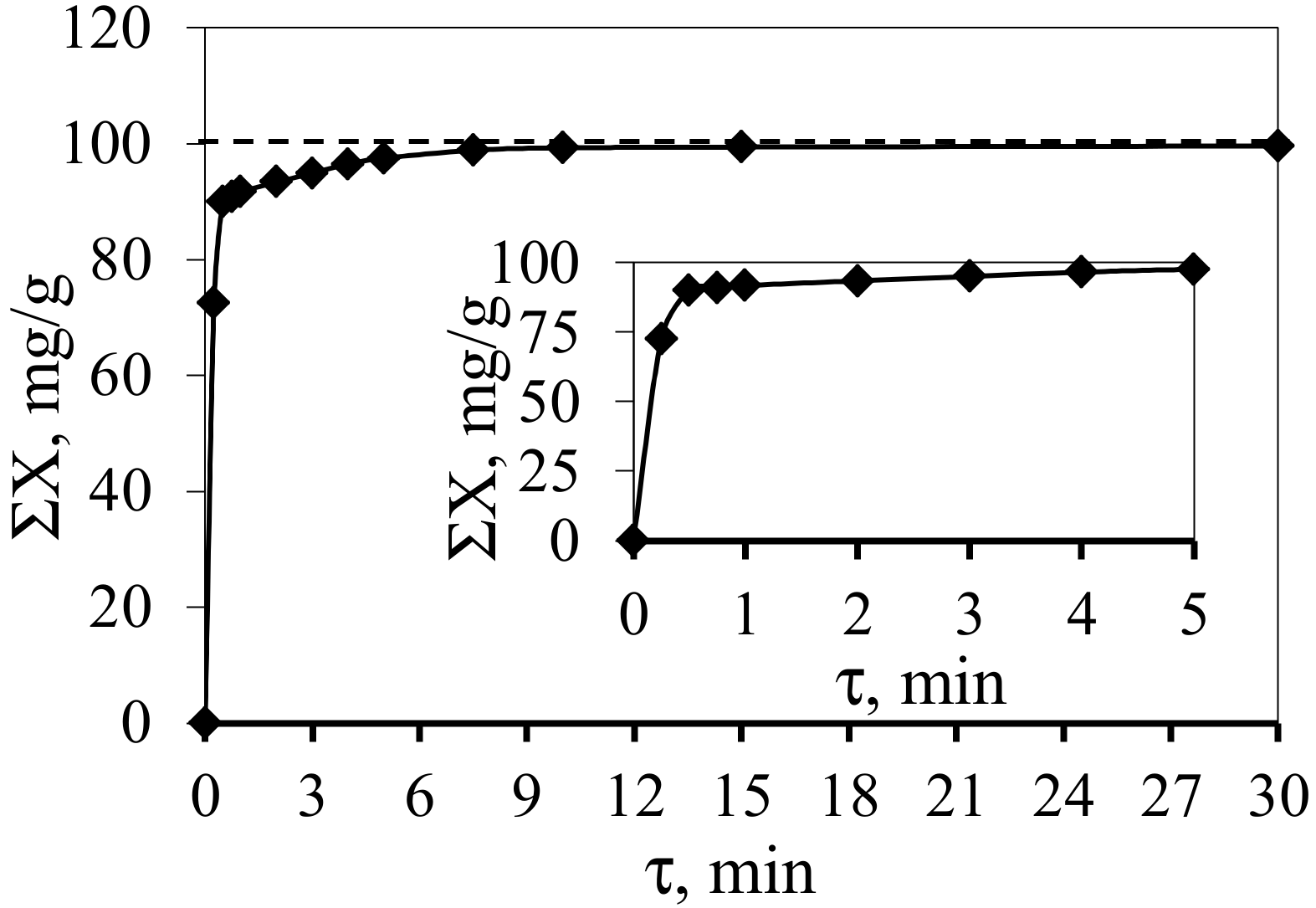
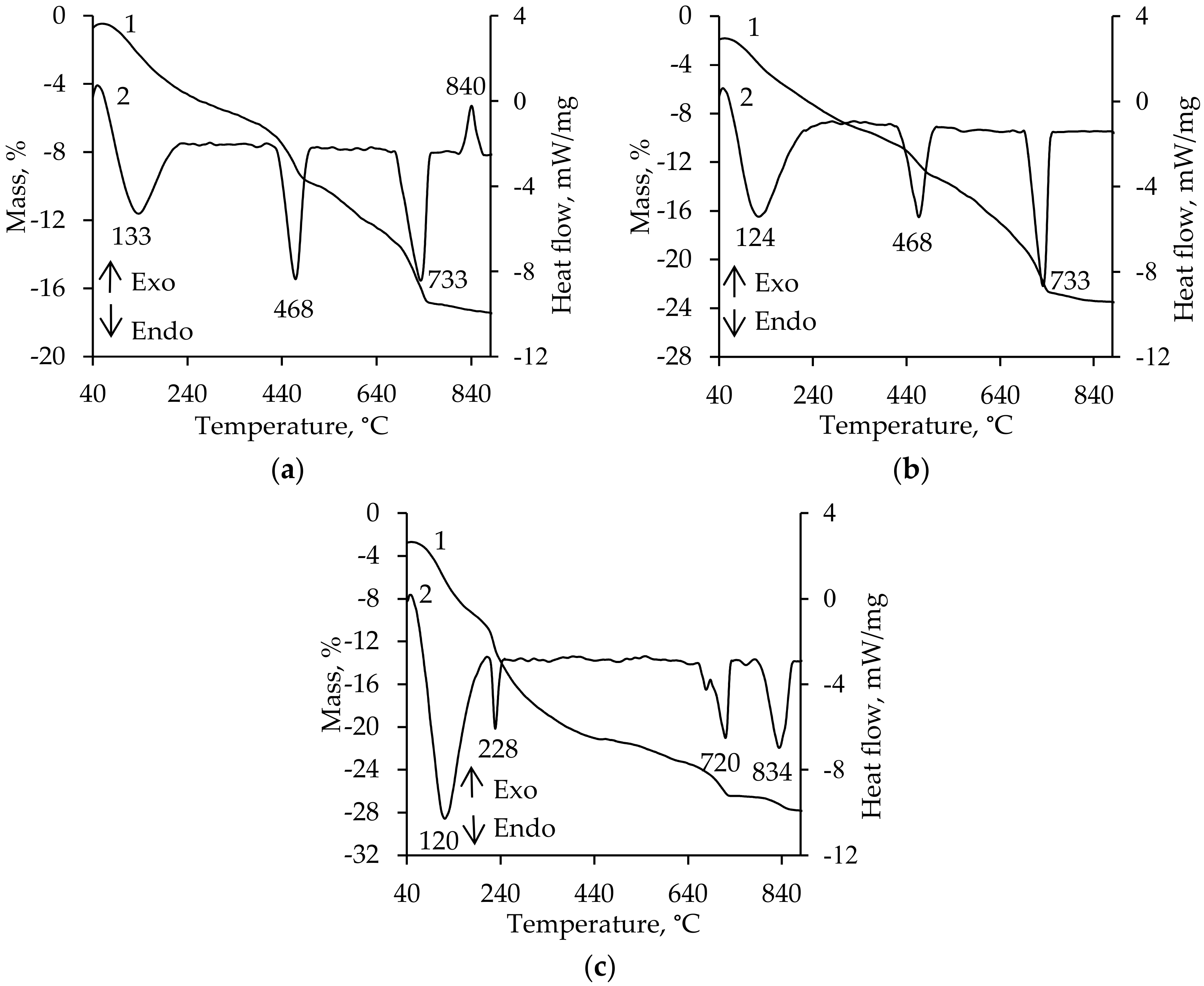
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, J.; Chen, C. Biosorbents for heavy metals removal and their future. Biotechnol. Adv. 2009, 27, 195–226. [Google Scholar] [CrossRef] [PubMed]
- Aguado, J.; Arsuaga, J.A.; Arencibia, A.; Lindo, M.; Gascón, V. Aqueous heavy metals removal by adsorption on amine-functionalized mesoporous silica. J. Hazard. Mater. 2009, 163, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Repo, E.; Warchoł, J.K.; Bhatnagar, A.; Sillanpää, M. Heavy metals adsorption by novel EDTA-modified chitosan–silica hybrid materials. J. Colloid Interface Sci. 2011, 358, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Barakat, M.A. New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 2011, 4, 361–377. [Google Scholar] [CrossRef]
- Kadirvelu, K.; Thamaraiselvi, K.; Namasivayam, C. Removal of heavy metals from industrial wastewaters by adsorption onto activated carbon prepared from an agricultural solid waste. Bioresour. Technol. 2001, 75, 63–65. [Google Scholar] [CrossRef]
- Fenglian, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Sud, D.; Mahajan, G.; Kaur, M.P. Agricultural waste material as potential adsorbent for sequestering heavy metal ions from aqueous solutions—A review. Bioresour. Technol. 2008, 99, 6017–6027. [Google Scholar] [CrossRef] [PubMed]
- Wan Ngah, W.S.; Hanafiah, M.A.K.M. Removal of heavy metal ions from wastewater by chemically modified plant wastes as adsorbents: A review. Bioresour. Technol. 2008, 99, 3935–3948. [Google Scholar] [CrossRef] [PubMed]
- Ajmal, M.; Rao, R.A.K.; Anwar, S.; Ahmad, J.; Ahmad, R. Adsorption studies on rice husk: Removal and recovery of Cd(II) from wastewater. Bioresour. Technol. 2003, 86, 147–149. [Google Scholar] [CrossRef]
- Argun, M.E.; Dursun, S.; Ozdemir, C.; Karatas, M. Heavy metal adsorption by modified oak sawdust: Thermodynamics and kinetics. J. Hazard. Mater. 2007, 141, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Šćiban, M.; Radetić, B.; Kevrešan, Ž.; Klašnja, M. Adsorption of heavy metals from electroplating wastewater by wood sawdust. Bioresour. Technol. 2007, 98, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zhu, Y.J.; Wu, J.; Zheng, J.Q.; Zhao, Z.Y.; Lu, B.Q.; Chen, F. Chitosan-coated mesoporous microspheres of calcium silicate hydrate: Environmentally friendly synthesis and application as a highly efficient adsorbent for heavy metal ions. J. Colloid Interface Sci. 2014, 418, 208–215. [Google Scholar] [CrossRef] [PubMed]
- Belmabkhout, Y.; Serna-Guerrero, R.; Sayari, A. Adsorption of CO2-Containing Gas Mixtures over Amine-Bearing Pore-Expanded MCM-41 Silica: Application for Gas Purification. Ind. Eng. Chem. Res. 2010, 49, 359–365. [Google Scholar] [CrossRef]
- Guo, Y.; Qi, J.; Yang, S.; Yu, K.; Wang, Z.; Xu, H. Adsorption of Cr(VI) on micro- and mesoporous rice husk-based active carbon. Mater. Chem. Phys. 2003, 78, 132–137. [Google Scholar] [CrossRef]
- Cavenati, S.; Grande, C.A.; Rodrigues, A.E. Adsorption Equilibrium of Methane, Carbon Dioxide, and Nitrogen on Zeolite 13X at High Pressures. J. Chem. Eng. Data 2004, 49, 1095–1101. [Google Scholar] [CrossRef]
- Zadaviciute, S.; Bankauskaite, A.; Baltakys, K.; Eisinas, A. The study of CP determination of hydrotalcite intercalated with heavy metal ions. J. Therm. Anal. Calorim. 2017. [Google Scholar] [CrossRef]
- Yong, S.O.; Yang, J.E.; Zhang, Y.S.; Kim, S.J.; Chung, D.Y. Heavy metal adsorption by a formulated zeolite-Portland cement mixture. J. Hazard. Mater. 2007, 147, 91–96. [Google Scholar] [CrossRef]
- Wu, J.; Zhu, Y.J.; Cao, S.W.; Chen, F. Hierarchically nanostructured mesoporous spheres of calcium silicate hydrate: Surfactant-free sonochemical synthesis and drug-delivery system with ultrahigh drug-loading capacity. Adv. Mater. 2010, 22, 749–753. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Zhu, Y.J.; Chen, F. 45S5 Bioglass-derived glass-ceramic scaffolds for bone tissue engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef]
- Eisinas, A.; Baltakys, K.; Siauciunas, R. Utilisation of gyrolite with impure Cd2+ ions in cement stone. Adv. Cem. Res. 2013, 25, 69–79. [Google Scholar] [CrossRef]
- Eisinas, A.; Baltakys, K.; Siauciunas, R. Removal of Zn(II), Cu(II) and Cd(II) from aqueous solution using gyrolite. J. Sci. Ind. Res. 2012, 71, 566–572. [Google Scholar]
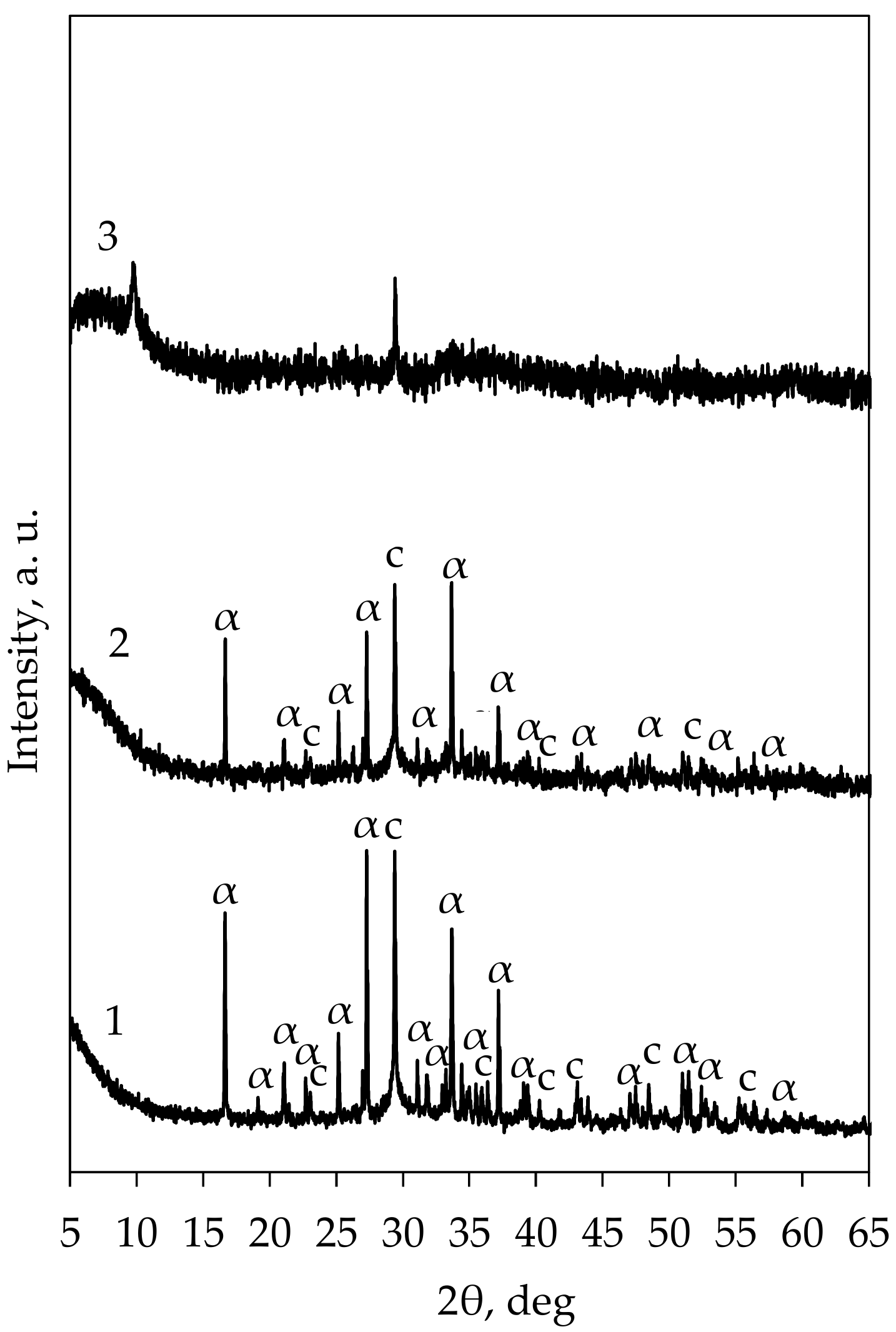
- Ishida, H.; Yamazaki, S.; Sasaki, K.; Okada, Y.; Mitsuda, T. α-Dicalcium Silicate Hydrate—Preparation, Decomposed Phase and Its Hydration. J. Am. Ceram. Soc. 1993, 76, 1707–1712. [Google Scholar] [CrossRef]
- Garbev, K.; Gasharova, B.; Beuchle, G.; Kreisz, S.; Stemmermann, P. First Observation of α-Ca2[SiO3(OH)](OH)-Ca6[Si2O7][SiO4](OH)2 Phase Transformation upon Thermal Treatment in Air. J. Am. Ceram. Soc. 2008, 91, 263–271. [Google Scholar] [CrossRef]
- Baltakys, K.; Dambrauskas, T.; Eisinas, A. The Synthesis of α-C2S Hydrate Substituted with Al3+ Ions in Mixture with CaO/SiO2 = 1.75. Solid State Phenom. 2015, 244, 26–33. [Google Scholar] [CrossRef]
- Dambrauskas, T.; Baltakys, K.; Eisinas, A.; Siauciunas, R. A study on the thermal stability of kilchoanite synthesized under hydrothermal conditions. J. Therm. Anal. Calorim. 2017, 127, 229–238. [Google Scholar] [CrossRef]
- Dambrauskas, T.; Baltakys, K.; Eisinas, A. Formation and thermal stability of calcium silicate hydrate substituted with Al3+ ions in the mixtures with CaO/SiO2 = 1.5. J. Therm. Anal. Calorim. 2017. [Google Scholar] [CrossRef]
- Sahu, S.; DeCristofaro, N. Part one of a two-part series exploring the chemical properties and performance results of Sustainable Solidia CementTM and Solidia ConcreteTM. Solidia Cem. 2013, 1–12. Available online: http://solidiatech.com/wp-content/uploads/2014/02/Solidia-Cement-White-Paper-12-17-13-FINAL.pdf (accessed on 15 January 2018).
- Barcelo, L.; Kline, J.; Walenta, G.; Gartner, E. Cement and carbon emissions. Mater. Struct. 2014, 47, 1055–1065. [Google Scholar] [CrossRef]
- Gartner, E.; Hirao, H. A review of alternative approaches to the reduction of CO2 emissions associated with the manufacture of the binder phase in concrete. Cem. Concr. Res. 2015, 78, 126–142. [Google Scholar] [CrossRef]
- Siauciunas, R.; Baltakys, K.; Gendvilas, R.; Eisinas, A. The influence of Cd-impure gyrolite on the hydration of composite binder material based on α-C2S hydrate. J. Therm. Anal. Calorim. 2014, 118, 857–863. [Google Scholar] [CrossRef]
- Zadaviciute, S.; Baltakys, K.; Eisinas, A.; Bankauskaite, A. Simultaneous adsorption at 25 °C and the peculiarities of gyrolite substituted with heavy metals. J. Therm. Anal. Calorim. 2017, 127, 335–343. [Google Scholar] [CrossRef]
- Garbev, K.; Black, L.; Beuchle, G.; Stemmermann, P. Inorganic Polymers in Cement Based Materials. Wasser Geotechnol. 2002, 1, 19–30. [Google Scholar]
- Garbev, K.; Gasharova, B.; Stemmermann, P. A Modular Concept of Crystal Structure Applied to the Thermal Transformation of α-C2SH. J. Am. Ceram. Soc. 2014, 97, 2286–2297. [Google Scholar] [CrossRef]
- Kasperaviciute, V.; Baltakys, K.; Siauciunas, R. the sorption properties of gyrolite for copper ions. Ceram. Silikáty 2008, 52, 95–101. [Google Scholar]
- Bankauskaite, A.; Eisinas, A.; Baltakys, K.; Zadaviciute, S. A study on the intercalation of heavy metal ions in a wastewater by synthetic layered inorganic adsorbents. Desalin. Water Treat. 2015, 56, 1576–1586. [Google Scholar] [CrossRef]
- Pradhan, N.; Rene, E.R.; Lens, P.N.L.; Dipasquale, L.; D’Ippolito, G.; Fontana, A.; Panico, A.; Esposito, G. Adsorption Behaviour of Lactic Acid on Granular Activated Carbon and Anionic Resins: Thermodynamics, Isotherms and Kinetic Studies. Energies 2017, 10, 665. [Google Scholar] [CrossRef]
- Ho, Y.S.; Wase, D.A.J.; Forster, C.F. Batch Nickel Removal from Aqueous Solution by Sphagnum Moss Peat. Water Res. 1995, 29, 1327–1332. [Google Scholar] [CrossRef]
- Ho, Y.S.; Wase, D.A.J.; Forster, C.F. Kinetic Studies of Competitive Heavy Metal Adsorption by Sphagnum Moss Peat. Environ. Technol. 1996, 17, 71–77. [Google Scholar] [CrossRef]
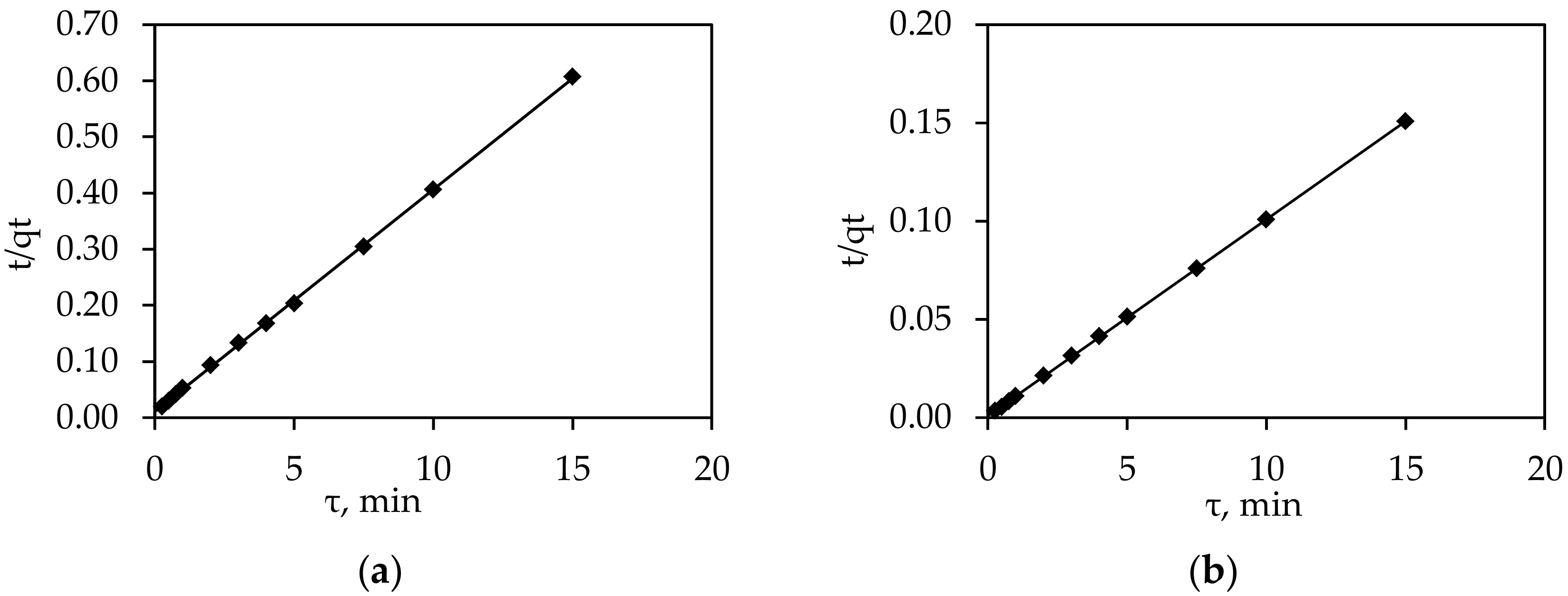
| Concentration (g Co2+/dm3) | R2 | qe(exp) (mg/g) | qe(cal) (mg/g) | k1 (min−1) | k2 (g/(mg·min)) |
|---|---|---|---|---|---|
| Pseudo-First-Order Kinetic Models | |||||
| 0.25 | 0.84 | 24.73 | 8.39 | 0.450 | - |
| 1.00 | 0.85 | 99.62 | 16.93 | 0.356 | - |
| Pseudo-Second-Order Kinetic Models | |||||
| 0.25 | 1.00 | 24.73 | 25.08 | - | 0.139 |
| 1.00 | 1.00 | 99.62 | 100.00 | - | 0.091 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Niuniavaite, D.; Baltakys, K.; Dambrauskas, T. The Adsorption Kinetic Parameters of Co2+ Ions by α-C2SH. Buildings 2018, 8, 10. https://doi.org/10.3390/buildings8010010
Niuniavaite D, Baltakys K, Dambrauskas T. The Adsorption Kinetic Parameters of Co2+ Ions by α-C2SH. Buildings. 2018; 8(1):10. https://doi.org/10.3390/buildings8010010
Chicago/Turabian StyleNiuniavaite, Domante, Kestutis Baltakys, and Tadas Dambrauskas. 2018. "The Adsorption Kinetic Parameters of Co2+ Ions by α-C2SH" Buildings 8, no. 1: 10. https://doi.org/10.3390/buildings8010010
APA StyleNiuniavaite, D., Baltakys, K., & Dambrauskas, T. (2018). The Adsorption Kinetic Parameters of Co2+ Ions by α-C2SH. Buildings, 8(1), 10. https://doi.org/10.3390/buildings8010010







