4.1. Properties of Mortar with Different Types of Superplasticizers
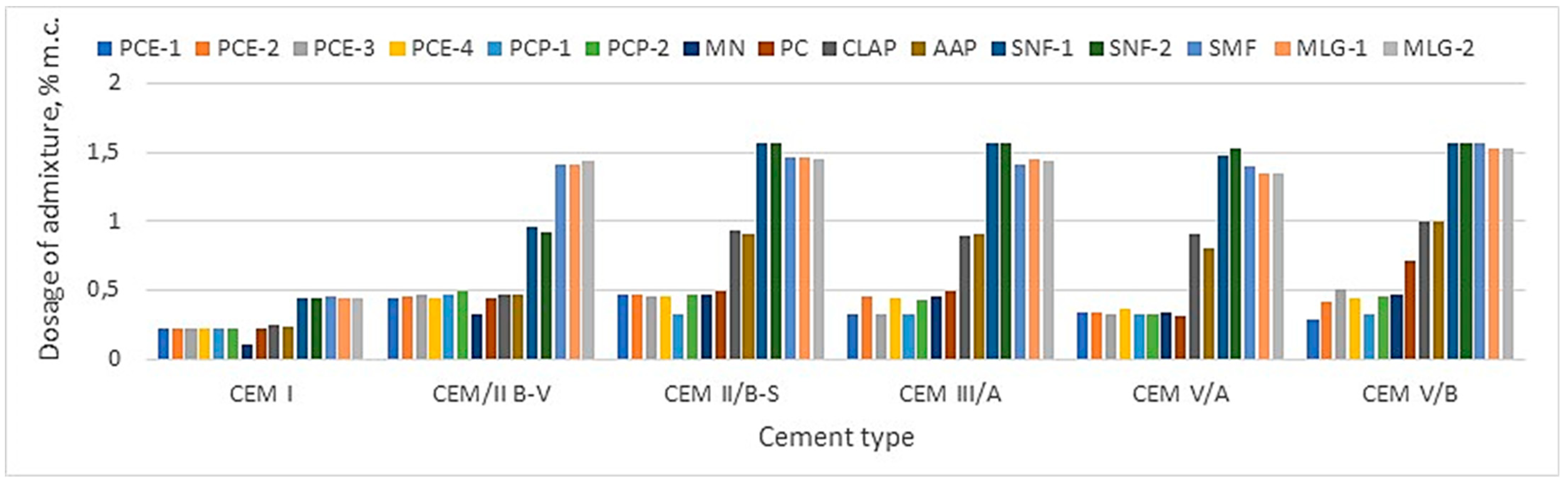
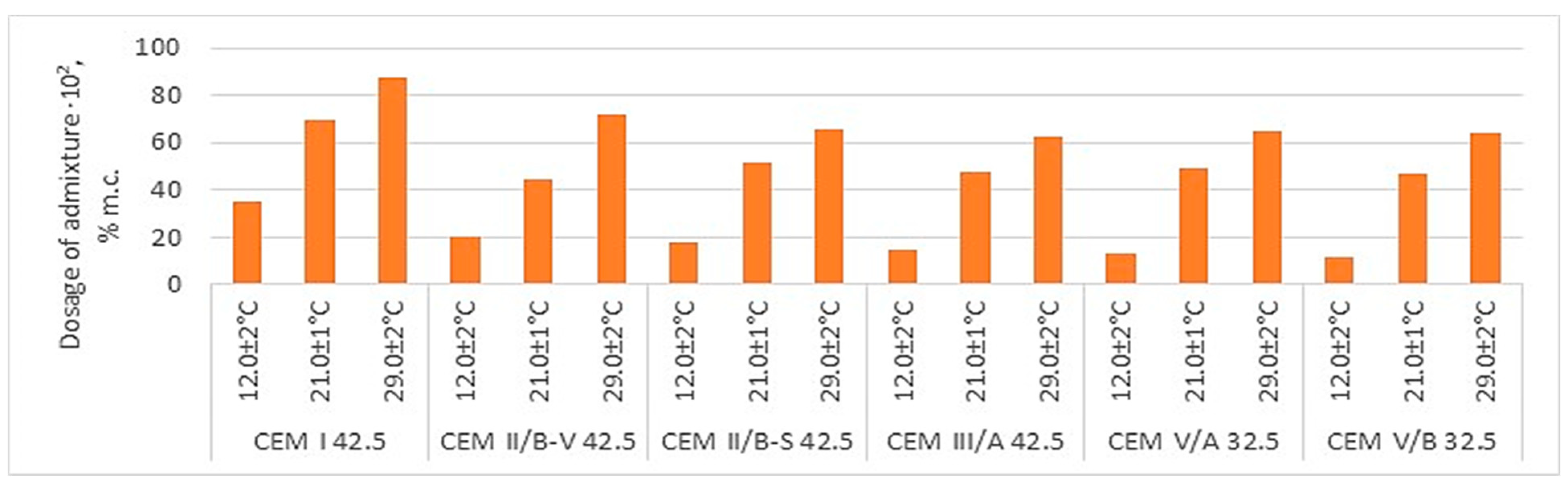
Figure 4 compares the demand for liquefaction or plasticizing admixture, depending on the type of cement, and
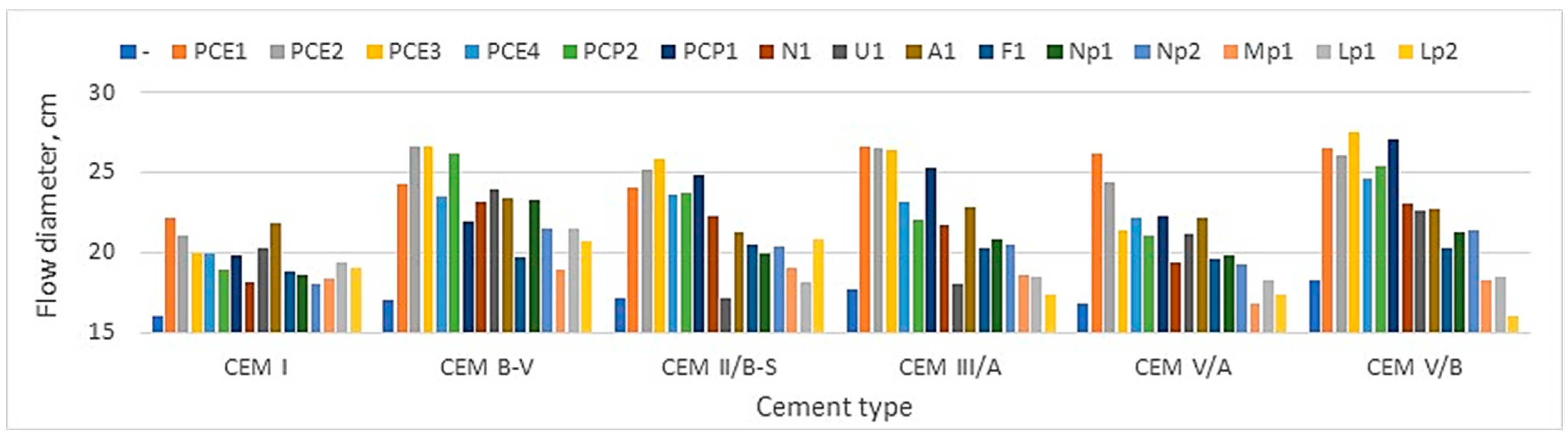
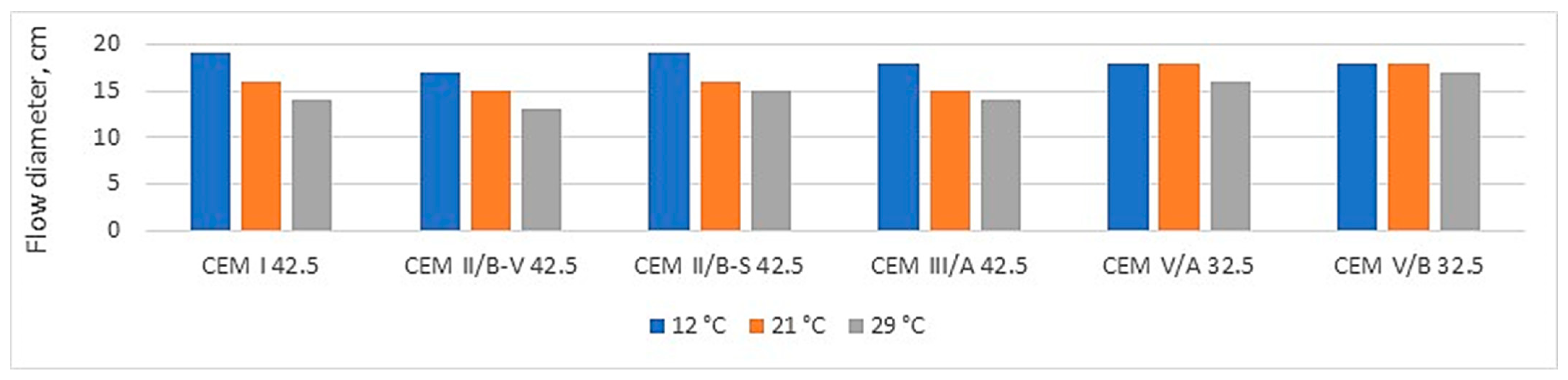
Figure 5 and
Figure 6 compare the air content in mortars with the resulting flow diameter.
The analysis of the test results presented in
Figure 5 and
Figure 6 indicates that reducing the amount of clinkers in cement promotes the consistency of the mortar and the air content in its volume. In the case of CEM V/A (S-V) cement, the largest flow diameters were obtained, with the smallest amount of admixture required among low-emission cement (
Figure 4) and the lowest air compactness. And so, replacing Portland cement with cement with reduced emissions CEM II -CEM V/B (S-V) requires a much larger, more than twice the amount of the plasticizing admixture (SNF, SMF and MGL). However, in the case of more effective liquefaction admixtures, the amount required depends on the type of cement (
Table 9 and
Figure 4). For example, CEM II/B involves doubling liquefaction impurities compared to CEM I, like CEM II/B-S. On the other hand, CEM III/A required a lower requirement for liquefaction admixture than CEM II/B-V and CEM II/B-S. Comparing the demand for liquefying admixture in the case of CEM III/A, it is concluded that in the case of CEM III/A, the amount of liquefying admixture is lower to obtain a similar mortar flow. On the other hand, CEM V/A (S-V) AND CEM V/B (S-V) required a smaller amount of liquefaction admixture than CEM II/B-S and CEM II/B-V but more than CEM I.
The changes mentioned above in demand for the superplasticizing admixture result from the change in the specific surface area of the analyzed cement (
Table 7). The larger the specific surface area of the cement, the more theoretically it requires an increase in the amount of liquefying admixture, with the same w/c of mortar (
Table 13 and
Table 14). As shown in
Table 7, CEM II/B cement has the highest specific surface area; in this case, the required amount of liquefaction admixture was the highest. Therefore, the developed surface of cement can be a determinant for correlating the dosage of liquefying admixtures with the same w/c of mortar or concrete. Omitting this relationship during concrete design, as practice shows, leads to an uncontrolled increase in the amount of water during its construction, especially with low-emission cement with a high content of additives other than clinker. This, in consequence, leads to a significant reduction in the strength of concrete, especially its strength. The increased specific surface area is mainly characterized by low-emission CEM IV/A and CEM IV/B cement, whose specific surface area, according to Blaine, is 4146 and 5200 cm
2/g (!). In their case, the demand for liquefaction is exceptionally high; therefore, they require a very effective selection. As shown in
Figure 4, each type of cement requires an individual selection of the type and amount of liquefaction admixture.
It should be mentioned here that a lower specific density than Portland cement characterizes multi-component cement:
- -
for CEM I: 3.10–3.15 g/cm3,
- -
for CEM II/B: 2.90–3.02 g/cm3,
- -
for CEM III/A: 2.98–3.05 g/cm3,
- -
for CEM V/A (S-V): 2.85–3.00 g/cm3,
- -
for CEM V/B (S-V): 2.80–2.95 g/dm3,
Depending on the degree of clinker milling and other components (
Table 1) and their mutual proportions. Consequently, the lower the clinker content, and thus the lower the CO
2 emission of cement, the more we gain a larger volume of grout with the same cement mass. This promotes the consistency of the mortar (
Figure 5) and concrete. This is undoubtedly a great advantage of using low-emission cement in practice, a fact that this research demonstrates.
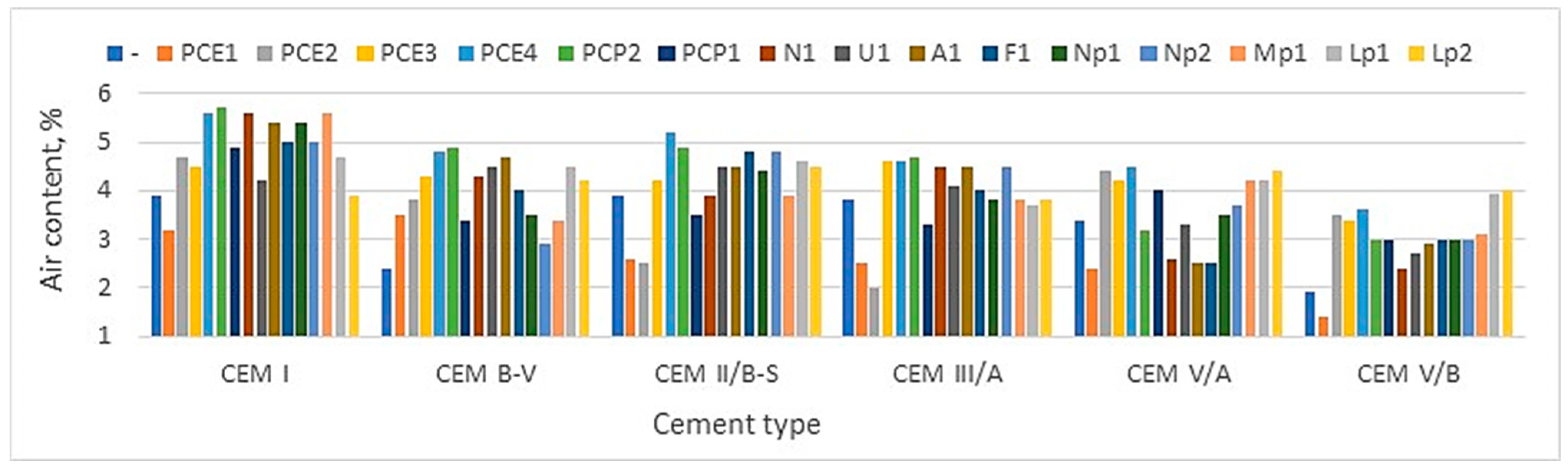
Figure 6 shows the results of measurements of the air content and flow diameter of mortars with CEM I and different admixtures. These results demonstrate that some new generations of superplasticizers can significantly increase the air volume in previously air-entrained cement-based mortars. The latest generation of admixtures substantially increases air content. The air content increases three times compared to previously aerated mortar. Many inorganic electrolytes and polar organic materials affect the foaming ability of surfactants [
22,
33]. The impact of other chemical admixtures on air entrainment is complex. Generally, most organic chemical admixtures can enhance air entrainment. The test results [
33] show that the superplasticizer can reduce water’s surface tension similarly to admixture (AEA). The surface tension of the PCP superplasticizer solution is very low, approaching the level corresponding to the aeration admixture [
33,
34]. It should be noted here that the value of the surface tension of the superplasticizer’s aqueous solution is closely related to its foaming activity.
The results of tests [
33] indicated that the different types of high-range water-reducing admixtures influence surface tension, foaming, and stability of air bubbles differently. The new generation of liquefaction admixtures comprises compounds made from polycarboxylates (PC), copolymers of acrylic acid with acrylates (CAE), or cross-linked acrylic polymers (CLAP). The chemical basis of these superplasticizers is primarily polyacrylates or methacrylates modified with various side chains attached to the main chain [
34]. They operate according to the so-called steric mechanism. Their high efficiency in liquefying the concrete mix results from their characteristic structure. These superplasticizers have a high molecular weight and spatially developed polymer chains, creating a physical barrier between them by adsorbing onto cement grains, thus preventing cement flocculation [
35,
36]. These polymers often have hydrophilic groups in their structure; therefore, in addition to the spatial effect, electrostatic interactions are also observed in the case below. The combined effect of both allows the concrete mix to liquefy by up to 40%. Some types of superplasticizers affect the surface tension of the liquid phase of the cement paste. The presence of functional groups (oxygen in the form of etheric group (–O–), hydroxyl group (–OH) and carboxyl group) produces a decrease in water surface tension, producing flocculation of associated molecules and an increase in moisture of not only grains of cement but also the whole mineral framework [
37]. In the SPs group, some show only dispersion functioning, not decreasing surface tension [
38]. They are hydrocarboxylic acid salts, sulphonic melamine-formaldehyde resins, formaldehyde picodensate salts of beta-naphthalensulphonic acid. The research results [
39,
40]. Demonstrate that the surface tension changes significantly over time, depending on the combination of powder and superplasticizer. The change appears to be caused by sorption, which includes both chemical and physical adsorption and absorption. Among the three types of sorption, the absorption of superplasticizer by the powder hinders the function of the superplasticizer. The tendency was that absorption could occur in the paste, as indicated by the flowability test of the paste.
Plasticizers, on the other hand, are based on sulfonated melamine-formaldehyde polycondensates (SMF) and sulfonated naphthalene-formaldehyde polycondensates (SNF) and work according to a lubricating or electrostatic mechanism. In both cases, the superplasticizer molecules adsorb onto the cement grains, increasing their wetting and causing the cement agglomerates to break down. In the case of more frequent electrostatic interactions, superplasticizers containing SO
3− and COO– COO-functional groups in their chains, when adsorbed on cement grains, impart a negative charge to them and cause the cement grains to repel each other [
34]. In addition, first-generation superplasticizers include modified lignosulfonates (MLS) and other compounds, e.g., sulfonated aromatic amines (AS) [
41]. Lignosulfonate compounds have a bipolar (dipole) structure. The negatively charged pole is hydrophobic and oriented towards the surface of the water molecule, while the positively charged pole connects to the cement particle. Cement conglomerates break down into smaller particles; the surface tension on the surface of grains wetted by mixing water decreases, and refined cement grains move more efficiently relative to one another. This effect will prove beneficial in cases of elevated temperatures, as demonstrated in the following subsection of the article.
4.2. Properties of Mortar at Different Temperatures
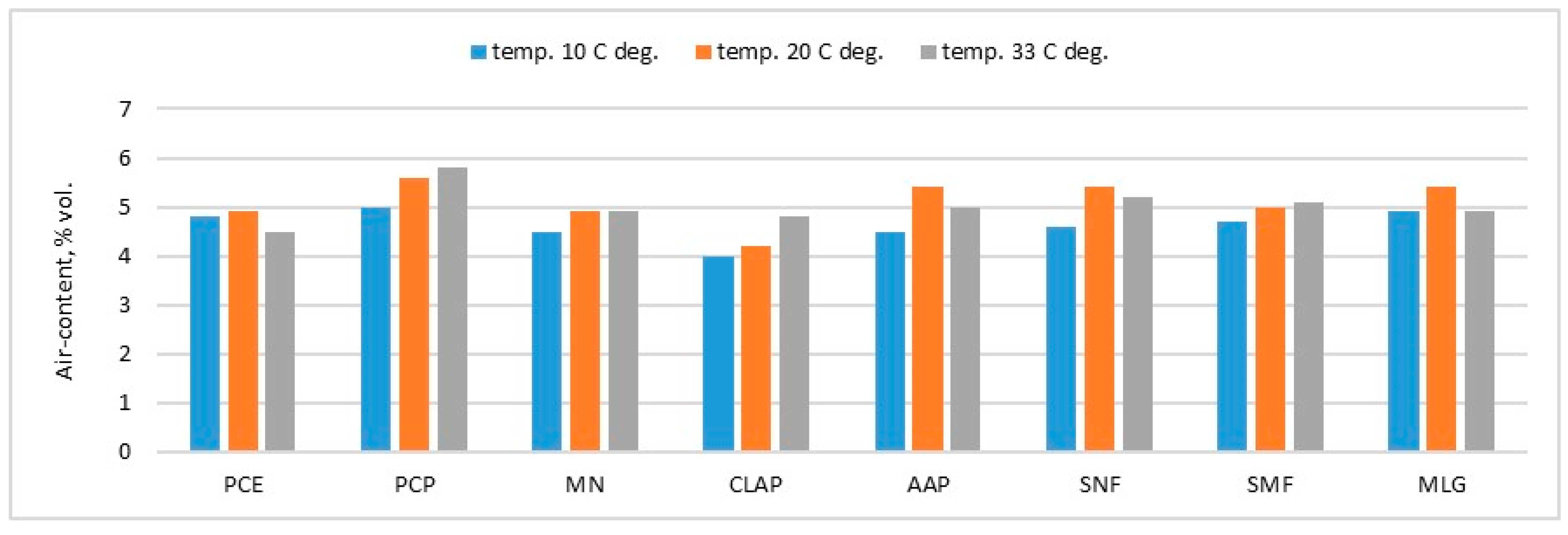
As evidenced by the research results in
Figure 7 and
Figure 8, the type of superplasticizer has a significant impact on the demand for the fluidizing admixture and the consistency of cement mortar at various temperatures. This practical implication of the research is also confirmed by the results of tests on non-aerated mortars conducted by Schmidt [
42]. With the increase in temperature, the required amount of PCE-1 admixture multiplies to the greatest extent in the case of CEM I. The fewer Portland clinkers there were, the less admixture had to be increased, along with reducing the amount of Portland clinker in the cement mass.
As shown by the analysis of the results presented in
Figure 7, the type of cement is crucial for maintaining mortar consistency at different temperatures. CEM V/B (S-V) cement, with the lowest content of Portland clinker, is the least susceptible to changes in consistency. Cement with S slag is more vulnerable to temperature changes than cement with V fly ash. CEM III/A achieved the most significant reduction in mortar fluidity under the influence of elevated temperature, but less than in the case of CEM I.
As shown in the analysis of the test results in
Figure 9, the type of admixture affects the degree of change in mortar flow reduction with increasing temperature. A minor decrease in flow was obtained in the case of lignosulfonate admixtures, which are used in the case of concretes with higher requirements than one would expect, e.g., in the case of self-compacting concretes [
40,
43,
44]. In practice, in the case of elevated temperatures, they are added as consistency precursors before dosing a newer generation of compatible liquefaction admixture.
Reduction of mortar flow under the influence of elevated temperature is more critical in the case of mortars with a superplasticizer than in the case of mortars with a plasticizer (
Figure 9).
The study’s results are consistent with those of Ghafoori and Diawar [
45], who found that above 20 °C, the flow of the non-aerated mixture decreases with increasing temperature. Additionally, research results [
46] demonstrate that an increase in ambient temperature leads to a decrease in mortar flow, with the degree of reduction depending on the proportion of superplasticizer in the cement mass and the type of superplasticizer or plasticizer used. The results indicate that an increase in temperature leads to an increase in the saturation dose of the mortar with the admixture. The lignosulfonate plasticizer exhibited the most pronounced efficacy reduction with increasing temperature, followed by polycarboxylate and naphthalene, which showed the best effectiveness. The publication draws attention to the fact that under the influence of temperature increase or the presence of ions from salts that easily dissociate in water (for example, as is the case with aluminosilicate binders (geopolymers) [
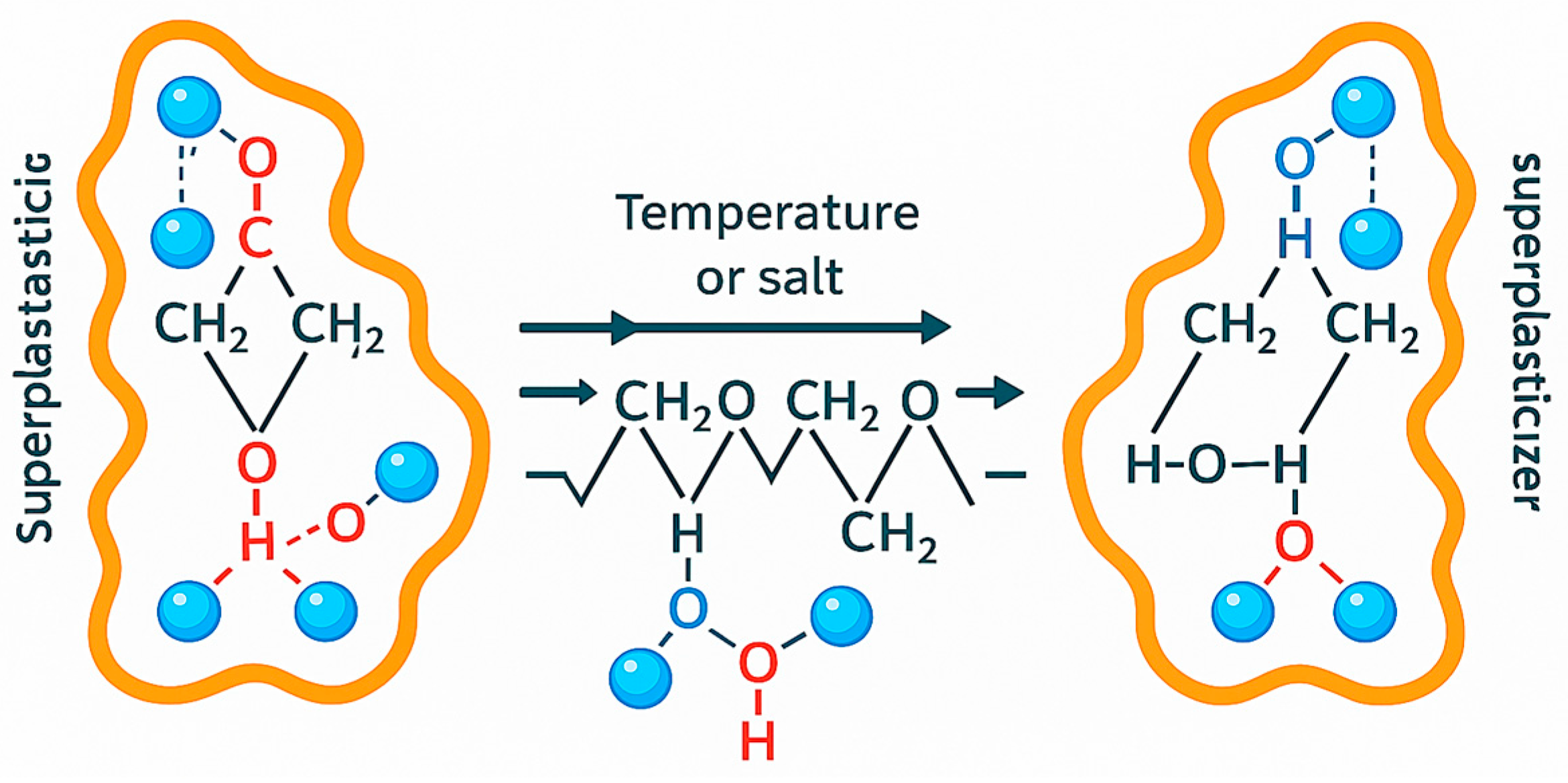
24] hydrogen bonds connecting the polyether chain with water are broken (
Figure 10), as a result of which the chain becomes tangled. The spherical blockade gradually decreases until it disappears completely.
Different performances of these types of superplasticizers can be achieved depending on the type of polymer (MPEG, HPEG, IPEG, etc.), as well as the ionic power and acidity of the cement slurry (pH). Also, depending on the structure of the superplasticizer and, more precisely, on the quantitative content of individual types of polymers in the blocks, it is possible to influence the effectiveness of these superplasticizers in cement. The effect of temperature on the effectiveness of acrylic superplasticizers is related to the change in the degree of hydration of polyether chains within the polymer system of the superplasticizer, as well as the corresponding change in the extent of steric lock on cement grains with increasing temperature. Exceeding the temperature at which the steric blockage on cement grains is insufficient to deflocculated cement grains due to a decrease in the degree of hydration of polyether chains causes a loss of slurry fluidity. According to Plank and Hirsch [
47], ettringite can adsorb significant amounts of negatively charged superplasticizer due to its highly positive ζ-potential, which increases with temperature. When the appropriate number of superplasticizer adsorption fields appears in the pore solution of the cement slurry, the superplasticizer’s efficiency increases, and thus, the diameter of the mixture flows [
48,
49]. According to Yoshioka [
49], most superplasticizers are absorbed by ferrite and aluminum. The primary products of early hydration are Ca(OH)
2, C-S-H, I-phase AFm, and AFph. According to Plank and Hirsch [
47], the amount of superplasticizer absorbed depends strongly on the zeta potential of the hydration products. Ettringite can adsorb large quantities of negatively charged superplasticizers. This results in a more excellent dispersion of mixtures. As the temperature increases, the amount of ettringite increases, thus the mixtures’ fluidity. However, in the analyzed research results, it was not observed that the temperature rise caused an increase in the fluidity of the mortars. Studies [
46] have shown that naphthalene-based admixtures have the best efficiency in increasing the fluidity of mortars at higher temperatures, resulting in a low air content in the volume.
On the other hand, the effect of temperature on the aeration of mortars with various admixtures is not unambiguous [
4,
38,
50,
51,
52]. In this case, it is impossible to form a constant relationship, as was previously the case with PCE 1 (
Figure 10). The air content in the mortar under the influence of temperature depends both on the consistency of the mortar (yield limit and plastic viscosity) [
19,
34,
53,
54,
55,
56], but also on the value of the surface tension of the pore liquid of the cement slurry in the presence of a given type of admixture [
57,
58,
59,
60].
Some of the superplasticizers used in the mortar compositions included accelerating agents intended to enhance early hydration and strength development. It was observed that, under elevated temperature conditions, these admixtures contributed to a slight increase in air content in the mixtures. This effect is likely due to the combined influence of accelerated hydration heat and changes in surface tension, which facilitate the entrapment and stabilization of fine air voids during the mixing process. Although these admixtures are not classified as air-entraining agents, their formulation may produce similar side effects. As such, their impact on the total air content should be considered, especially in systems with low water-to-cement ratios or when evaluating temperature-sensitive performance.
In the case of superplasticizers with an increased side effect associated with excessive “aeration” of the mortar, the aeration of the mortar may increase with an increase in temperature as the superficial tension decreases, which is already relatively low [
33]. The new generation of superplasticizers, which is based, in part, on the mechanism of lowering the surface tension of the liquid (PCP, PCE in
Figure 11) [
20,
21,
56], works more effectively as the temperature of the mortar increases because the surface tension value decreases with the rise in temperature. These superplasticizers and surfactants form an adsorption layer on the water’s surface, reducing surface tension and improving the wetting of the cement grain. Neutral admixtures, resulting from the aeration of the liquefied mortar, include liquefying admixtures F (based on modified phosphonates) and N (based on modified naphthalene), as well as traditional plasticizers, which have a less intense effect on the surface tension of water than PCE or PCP. Naphthalene and melamine plasticizers, as well as MLS-modified calcium or sodium lignosulfonates, and other products, such as formic acid copolymers with naphthalene sulfonic acid or methylnaphthalene sulfonic acid, also reduce the surface tension of water relative to cement and microfilters [
57,
58,
59,
60,
61,
62,
63,
64]. It is associated with more significant adsorption of the PCE superplasticizer on cement particles and cement hydration products.
The analysis of the effect of temperature on the air content in mortars with various liquefying admixtures is not as straightforward as the influence of temperature on consistency.
The dependence of the surface tension of a liquid on temperature is described by the equation [
62] derived by Eötvös:
where:
σ—surface tension of the solution,
M—stands for molar mass,
D—density of the liquid (hence M/d—is the volume of one mole),
Tk—critical temperature,
δ—is a correction of about 6 degrees (it turns out that the disappearance of surface tension occurs at a temperature slightly lower than Tk).
An increase in temperature should reduce the air content in the mortar, as higher temperatures lead to faster water evaporation, thereby reducing the viscosity of the concrete mix. As a result, it is more difficult to keep air bubbles in it, which escape more easily from the concrete [
4]. The viscosity of the cement slurry decreases as the temperature increases, so the air content should be lower. The effect of temperature on the air content of air-entrained mortar matches that observed in the publication [
23]. Analyzing the impact of temperature on the behavior of cement paste, which is contained in its composition SP, can be traced to some analogy in the behavior of the liquid. The air molecules and the pressure can be calculated using equation [
4]:
For the vapour phase, the pressure is described using the Kelvin equation [
63,
64]:
where:
p*—the vapour pressure of the liquid in the absence of additional pressure,
γ—the surface tension,
Vm—the molar volume,
r—the radius of the air bubble,
R—the gas constant,
T—the temperature in Kelvin.
Considering an air bubble in the fresh concrete, with an increase in temperature in concrete, n and T increase in Equation (4). Similarly, γ and T increase in Equation (5). These changes require a concurrent shift in p or V or both. When a liquid heats up, its molecules become excited and move. The energy of this movement is enough to overcome the forces that bind the molecules together, allowing the liquid to become more fluid and decreasing its viscosity. Because the paste pressure balances the internal pressure of air bubbles, it accumulates atmospheric and gravitational forces. It may thus change only slightly due to an increase in the paste’s viscosity [
4,
65,
66,
67,
68,
69]. As a result, the bubbles must expand their volume quite significantly. The outcome is a decrease in entrained air content and larger bubble sizes in fresh concrete whose temperature is higher than when it was initially mixed [
63,
68,
69].
Nevertheless, the temperature of the concrete mixture can affect the air content in several ways. First, as it is well known, the higher temperature of water leads to the lower solubility of air in water [
65,
66,
67]. At the same slump value, the mixture with a higher temperature consistently exhibits higher viscosity, and for the mix with higher viscosity, entraining air becomes more complex [
69,
70,
71]. The low temperature decreases the yield stress and viscosity of concrete. Yield stress and viscosity of concrete mixtures act as energy barriers for air bubble formation. The higher the slump, the easier the gas dispersion in the mixture would be, and probably more giant air bubbles would result. The conclusion is consistent with the test results summarized in
Figure 11. In the case of concrete, an increase in a slump from 75 to 150 mm (with all other mixture parameters remaining the same) will increase the air content [
57,
67,
68,
69]; however, above a slump of 150 mm, the large air bubbles become less stable due to buoyancy forces and the air content drops. According to the author, this may explain the difference in the impact of air temperature on the air contents of air-entrained concrete and air-entrained and plasticized concrete. As mentioned earlier, the SP type undoubtedly affects the direction of change in concrete consistency. For other kinds of SP than those analyzed, there may be another change of air entrainment in concrete under the influence of temperature [
66]. Thus, the type of admixture significantly affects the resulting air content and consistency in the mortar at different temperatures; therefore, the resulting air content does not decrease with increasing temperature. This would be the case if the measurements were carried out with the same mortar flow at different temperatures and for the same admixture.
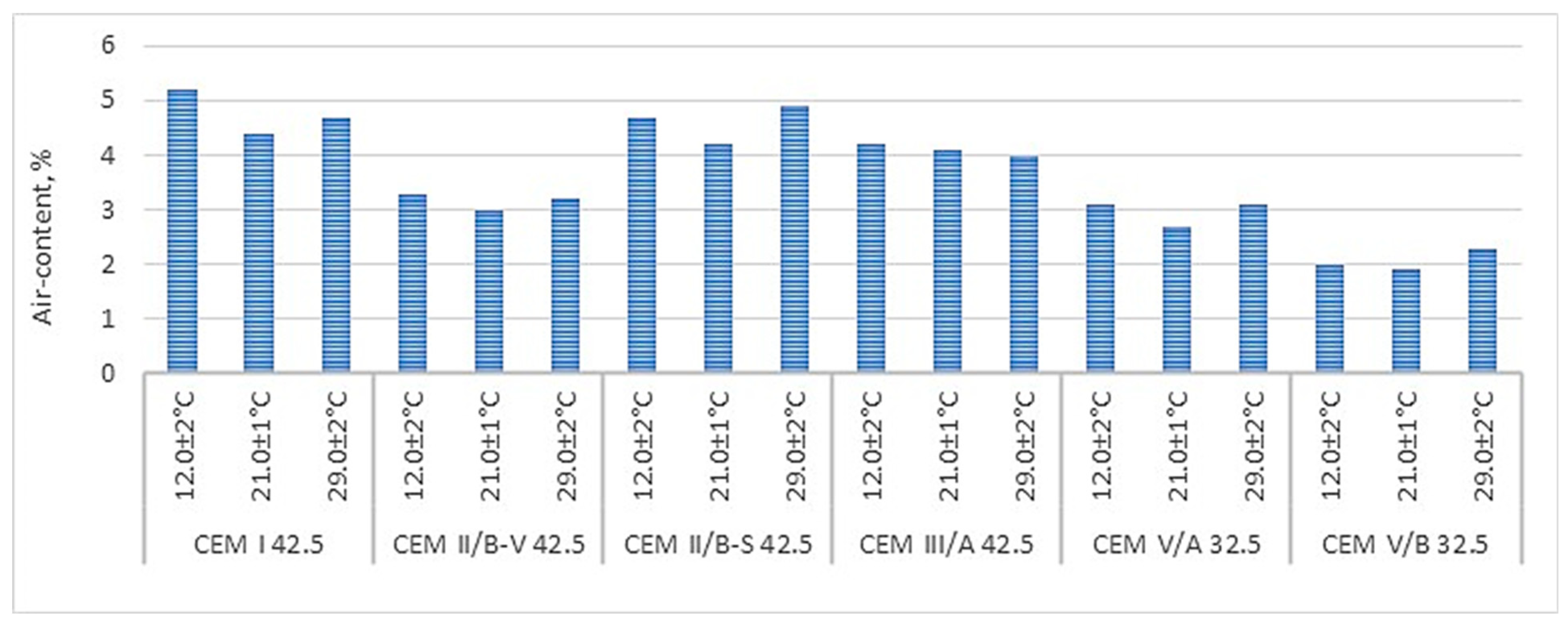
As shown by the analysis of the research results presented in
Figure 12, the lower the clinker and blast furnace slag content in the cement, the lower the air content in the mortar at elevated temperatures (
Figure 12).