1. Introduction
In recent years, a growing body of scholarship that empirically examines neurophysiological responses to the built environment has begun to emerge [
1,
2]. These studies provide evidence that visual exposure to biophilic design features, or spatial designs that either integrate live organic material or imitate patterns found in nature (biophilic design [
3]), may reduce physiological stress, a biological response wherein the body’s systems undergo changes to cope with perceived threats or challenges [
4,
5,
6,
7,
8,
9,
10,
11]. Whilst the impact of this emerging research on our understanding of the relationship between the built environment and neurophysiological responses, such as physiological stress, is considerable, when viewed in conjunction with emerging evidence in relation to neuroimmunology, these findings may be of greater significance than previously understood. Research within the domain of neuroimmunology—a specialized field that examines the intricate interplay between the nervous system (responsible for physiological stress responses) and the immune system (the regulator of the body’s inflammatory mechanisms)—suggests a direct linkage between physiological stress and the onset of neuroinflammation [
12]. Neuroinflammation refers to the complex inflammatory response within the central nervous system, and is implicated in the development of various neurological disorders including depression, schizophrenia, autism, obsessive–compulsive disorder, Tourette syndrome [
13,
14,
15], multiple sclerosis [
16], Parkinson’s disease, Alzheimer’s disease, and frontotemporal dementia [
17].
Recent studies in neuroimmunology indicate that physiological reactions to stress result in a bidirectional dialogue between the central nervous system and the immune system [
18,
19]. While acute stress responses play a critical role in survival and healing [
20], chronic exposure to physical and psychological stressors has been found to induce prolonged neuroendocrine responses, which in turn compromise immune function [
21,
22,
23], resulting in systemic proinflammatory responses within the body [
24]. This prolonged elevated neuroinflammatory activity can cause lasting cellular, molecular, and epigenetic changes [
12], resulting in long-term damage to organs and tissues [
25]. Conversely, it has been observed that a decrease in stress levels can mitigate these adverse effects by diminishing systemic inflammation via the same pathways [
26,
27].
Despite these findings, the relationship between visual exposure to biophilic design and neuroinflammation is yet to be directly investigated. This pilot study aimed to develop a research framework through which to specifically test the hypothesis that exposure to architectural designs with increased levels of biophilia may reduce neuroinflammatory activity in the body via their impact on physiological stress responses. Utilizing electroencephalography (EEG) to measure the impact of biophilic design, the study measured frequency band activity across specific neurological markers associated with neuroinflammatory activity. EEG is a method of neurophysiological monitoring that records electrical activity in the brain by placing electrodes on the scalp. Of particular interest were any observable changes in frequency band activity across the relative delta and relative alpha power bands. EEG frequency band activity refers to the categorization of distinct oscillatory patterns, electrical impulses generated by the neurons in the brain, within the electroencephalogram (EEG) signals, and it is indicative of various states of brain function and cognitive processes. Activity in relative delta and relative alpha power bands were of particular interest as these are established proxies for neuroinflammatory microglial and cytokine activity [
28,
29,
30,
31]. Microglial cells are specialized immune cells in the brain that respond to injury, disease, and physiological stress by becoming activated, and cytokines are signalling proteins that are released by microglial cells when activated [
13]. Changes in microglial cell and cytokine activity may indicate inflammation or immune responses within the brain or other tissues [
13]. In EEG readings, “relative delta” and “relative alpha” refer to specific bands of brainwave frequencies that, through noninvasive monitoring, can serve as indirect markers of neuroinflammatory activity within the brain [
28,
29,
30,
31].
Physiological monitoring of participants to assess neuroinflammatory activity while viewing 2D visual material of architectural designs is an innovative approach in the development of evidence-based design outcomes within the built environment. This methodology combines insights from the disparate fields of architectural neuroscience and neuroimmunology to present a new field of scholarship, termed “architectural neuroimmunology”. Architectural neuroimmunology is defined here as the study of the impact of exposure to architectural forms on immune-mediated neurological activity. Whilst this pilot study specifically examines the impact of biophilia on neuroinflammatory responses, architectural neuroimmunology as an emergent field, and the research methodology set out within this pilot study, have broader implications in relation to the evaluation of occupant physiological responses to the built environment. Through this pilot study, this research advocates for the development of evidence-based understandings of how we might design for better health and wellbeing outcomes.
3. Materials and Methods
Despite the evidence of a link between physiological stress and neuroinflammation, the role that the built environment might play in mediating physiological stress and neuroinflammation has yet to be examined. This paper outlines the results from a pilot study established to explore the relationship between the impact of visual exposure to biophilic design and neuroinflammatory responses. A pilot study asks “whether something can be done, should the researchers proceed with it, and if so, how. A pilot study is conducted on a smaller scale than the main or full-scale study and is conducted in order to assess the safety of treatment or interventions and recruitment potentials, examine the randomization and blinding process, increase the researchers’ experience with the study methods or medicine and interventions, and provide estimates for sample size calculation” ([
54], p. 601). The pilot study measured the impact of visual exposure to biophilic design on neuroinflammation. It should be noted that this study does not claim to establish definitive or conclusive findings. Instead, the findings produced by this study serve as an initial pilot study to explore physiological responses to biophilia and test appropriate research methodologies to examine relationships between architectural stimuli and participant neurophysiological responses. Given the complexity of physiological response networks, and the breadth of socioeconomic, environmental, psychosocial, and epigenetic factors that influence neurological responses, significant research needs to be conducted before definitive conclusions can be made concerning the impact of the built environment on neuroimmunological responses. Despite these limitations, it is hoped that this research will serve as a stepping stone for future academic inquiry and developments in the field of architectural neuroimmunology.
3.1. Participants
Participants were recruited via word of mouth and through calls for research participants, which were distributed via university departmental mail lists and within the university colleges. This study obtained data from 10 participants (n = 10; 4 females and 6 males) following written informed consent. This is in line with findings from a systematic review of the sample sizes used in EEG studies by Larson and Carbine (2017), in which they found that a sample must include between 7 and 26 participants per group to be considered statistically significant [
55]. The participant intake form collected background demographic information on participant age, biological sex assigned at birth, handedness, use of illicit drugs, and general medication intake. Age, sex assigned at birth, and handedness were retained as control variables for statistical analysis (discussed in further detail below). Participant age range was restricted to 18–80 (for legal grounds and validity of sample data). Participants ranged in age from 24 to 76 (mean = 37.6; SD = 19.8).
Gender representation was relatively evenly distributed, with six women and four men participating in the study. Eight of the participants were right-handed, and two were left-handed. All participants reported being healthy and stated that they did not regularly take illicit substances, benzodiazepines, antipsychotics, antidepressants, sedatives, or hypnotic drugs and did not have any known central nervous system disorders. Each participant received GBP 10 as a gesture of acknowledgement of their time—in line with current debate in relation to ethical research frameworks [
56]. The study was approved by the Faculty of Architecture’s Ethics Committee at the University of Cambridge. Additionally, this study was designed in accordance with the World Medical Association’s “Declaration of Helsinki—Ethical Principles for Medical Research Involving Human Participants” [
57].
3.2. Experimental Design
Two buildings were selected to be utilised as visual stimuli as they embodied varying degrees of biophilic architectural features. The level of biophilia for each building was determined by the authors using the Biophilic Healing Index (BHI) [
32]. The BHI is an empirical framework developed by Salingaros (2019) to empirically assess the biophilic content of buildings (see
Appendix A;
Table A1). The BHI was utilised as it provided a mechanism by which to assign a relative biophilic measure to the visual stimuli utilised. The BHI value of a building is calculated by assigning each building a value between 0 and 2 for ten set biophilic criteria. This then produces a final BHI value between 0 and 20 (Equation (
1)).
Although the Biophilic Healing Index (BHI) provides a foundational basis for the empirical examination of biophilic elements, the method involves a subjective and semi-qualitative method of analysis whereby the researcher assigns relative values for each criterion based on personal judgment. As a result, the development of additional tools or computational methods of analysis that limit subjectivity in the assigned values for each variable would help facilitate more robust research into biophilia. These limitations are discussed in greater detail in
Section 5.3.
Sets of 2D images (six images total: three Building 1 and three Building 2) were selected to form the visual stimuli utilised in the study. The image sets were designed to create a high degree of similitude between the images for Building 1 and Building 2 based on image angles, lighting, perspective, and features. Image Set 1 (left) featured a close-up of the building facades, Image Set 2 (centre) featured the exterior hallways, and Image Set 3 (right) featured an exterior perspective of both buildings.
Building 1 (
Figure 1) is a proposed architectural development in Montreal, Canada that incorporates biophilic forms and small-scale agricultural cultivation within the building [
58]. Building 1 is a mid-/high-rise, mixed-use development (residential and commercial) that features semi-enclosed corridors and is situated in an urban centre. Photorealistic renders of the building were used for analysis. The analysis of Building 1 using the BHI determined that the building embodied a moderate degree of biophilia (B = 10)—notably, fractalised design patterns, organic material, organised complexity, and natural materials (
Table 2).
Building 2 (
Figure 2) is situated in The Hague, Netherlands, and was completed in 2011 [
59]. Like Building 1, Building 2 is a mid-/high-rise, mixed-use development (residential and commercial) featuring semi-enclosed corridors and situated in an urban centre. Photos of the completed building were used for analysis. The analysis of Building 2 using the BHI determined that the building embodies a moderate to low degree of biophilia (B = 6). While Building 2 scored similarly to Building 1 on colour, realistic detection of gravity, and level of detail, it lacks the fractalised design patterns, organic material, organised complexity, and natural materials found in Building 1 (
Table 2).
The BHI analysis of the two buildings can be seen in
Table 3 (Building 1 > Building 2;
Table 2).
QEEG was used to assess the participants’ level of neuroinflammation when exposed to images of Building 1 and Building 2. In accordance with current scholarship (see
Table 1), relative alpha and delta power were selected as suitable proxy variables for measuring neuroinflammation using qEEG analysis, namely, decreased relative delta and increased relative alpha power activity are indicative of reduced neuroinflammation and reduced physiological stress responses.
It should be noted that qEEG is a relatively novel method for analysing neuroinflammation. At present, there is no clear understanding of the base level of neuroinflammation present in a subject at any given time. Therefore, researchers using qEEG to analyse neuroinflammation must presume a base level of neuroinflammation in each participant at the outset of analysis. The method then measures changes in relative frequency band activity to estimate a decrease or increase in neuroinflammation when the participant is exposed to a particular stimulus. Despite these limitations, this method of analysis reflects current best practices and enables an assessment of relative changes in neuroinflammatory stress responses.
This study examines whether increased levels of biophilia (Building 1 > Building 2) result in decreased relative delta power activity and increased relative alpha power activity. Whilst this research focused on the assessment of relative alpha and relative delta, all frequency bands were recorded during the participant qEEG process (relative alpha, relative beta, relative theta, relative delta, and relative gamma power).
3.3. Experimental Procedure
The study’s research methodology is outlined below in
Figure 3.
The experiment took place within a controlled university lab setting that hosted the necessary conditions for the qEEG experiment (i.e., consistent indoor lighting with no light flicker, controlled level of humidity, controlled acoustic environment, and constant temperature). Participants were fitted with a 32-Channel BitBrain Semidry EEG (Versatile 32) system (a research-grade wireless semi-dry electrode EEG headset). Once fitted with the EEG, the participants were seated in the laboratory setting in front of an HP M24f 23.8 inch 75 Hz FHD monitor and shown the visual stimuli image sets. Subjects were presented with a continuous stream of images, and each participant was exposed to identical visual stimuli image sets, image order, and image presentation duration. Each of the six images was displayed on the computer monitor for 180 s per image, with a total run time of 1080 s (18 min). This display time was selected to capture a sufficiently significant EEG data sample based on findings from Doudoux et al. (2018, p. 1), who concluded that “92% of the pathological EEG [findings] were provided within the first 10 min of recording” [
60]. The images were displayed consecutively in the following order: Building 2 Image Set, followed by Building 1 Image Set. As this study aimed to examine the impact of biophilic design on neuroinflammatory activity, the visual stimuli were designed such that the moderate biophilic image set (Building 1) followed the moderate to low biophilic image set (Building 2). It should be noted that a follow-up study that employs a randomised stimuli exposure methodology is required to evaluate the impact of image order on participant EEG output so as to test whether or not visual stimuli image order biases results. No audio or haptic stimuli accompanied the visual stimuli. Each experiment took approximately one hour in total.
The EEG electrodes were positioned according to the recommendations for the 10–20 international electrode placement system. EEG activity was measured by the monopolar derivation method from 32 electrodes [
61]. The EEG amplifier featured a 24-bit resolution for analog-to-digital conversion and sampled at 256 Hz, with the EEG signal being band-pass filtered between 0.003 and 150 Hz [
61]. The measured EEG signal (raw data) was collected using BitBrain’s proprietary real-time data collection software BitBrain Data Viewer SDK (version 1.6 BitBrain Technologies, Zaragoza, Spain) and saved in a comma-separated-value (CSV) file format for analysis.
3.4. Data Processing
Following the data collection process, a custom EEGLab protocol established by Uyulan et al. (2023) was applied to process the raw EEG data [
62]. High- and low-pass filters were applied to the data in the EEGLab to remove environmental noise. Low-pass filters (45 Hz) were used to reduce interference, such as muscle tremors [
63], and high-pass filters (0.5 Hz) were used to reduce low-frequency interference from activities such as breathing [
64] and correct for direct current (DC) offset of the signal [
65]. Finally, a 50 Hz notch filter was applied using the CleanLine EEGLab plugin to remove potential interference signals (i.e., remove electrical noise in the environment from standard UK 50 Hz mains electricity) [
66]. The filtered data were then exported as a generic setting (SET) file for EEGLab AMICA
c2_1_runAMICA.m to perform an independent component analysis (ICA) decomposition on the input data. The EEGLab ICLabel [
67] script was then applied to the data, which allowed for the automated identification and removal of independent EEG component artifacts. It should be noted that automated independent component (IC) artifact identifiers have some limitations, namely, increased risk of false artifact identification and the potential removal of legitimate EEG data. To combat this, future research may incorporate a double-blind artifact screening process.
Following the artifact rejection process, the EEGLab trimOutlier (version 0.17) script [
68] was used to eliminate compromised channels and data. Once a “clean” dataset was produced, the LaplaciancleanData.m tool created by Uyulan et al. (2023) was selected to transform the data into a Laplacian montage [
62]. A Laplacian montage was selected for this study based on a literature review of appropriate methodologies that assessed localised neurological activity in architectural contexts [
8]. Given that neuroinflammation is the product of proinflammatory cytokines produced by microglial cells, it was determined that a localised assessment methodology was preferential. It should be noted, however, that there is currently a paucity of research using EEG to measure neuroinflammation. Given this, further research is needed to investigate the most effective montage systems for neuroinflammatory analysis and to determine the limitations of different montage systems.
Multitaper spectral decomposition (mtaper) was then used to transform the time series EEG signals into the corresponding frequency domain. Mtaper converted the raw EEG data from each of the 32 electrodes and extracted the relative delta, relative theta, relative alpha, relative beta, and relative gamma power activity. Mtaper was selected over alternative decomposition methods due to its improved temporal frequency resolution [
69]. Upon completion, the mtaper script was used in MATLAB to export the computed values of frequency power in quarter Hz bins (e.g., delta 0.5–4 Hz and theta 4–8 Hz) and compute descriptive statistics for kurtosis, skewness, maximum frequency, maximum power, median power, and mean power. The data were then exported in .xlsx format. Finally, topographic plots, line plots, and difference plots were generated using the mean mtaper for each subject (n = 10) for both Building 1 and Building 2 samples.
3.5. Data Analysis
The statistical methods employed in this study were selected based on a comprehensive review of research articles that utilized similar methodologies [
6,
8]. Valid EEG signal data from all 10 participants were used for statistical analysis. Statistical analyses were conducted using a combination of Excel and Stata. Among the EEG data collected from the 32 electrodes, changes in the power spectrum of all frequencies (relative delta, relative alpha, relative beta, relative theta, and relative gamma power) were computed for statistically significant deviations in participants’ brain wave activity in response to images of Building 1 and Building 2. However, for the purpose of this study, only relative alpha and relative delta were of primary interest due to their association with neuroinflammatory microglial cells and cytokine activity [
28,
29,
30,
31].
p-values were calculated and the null hypothesis examined based on the following statistical significance parameters: a
p-value of <0.01 was considered highly significant, meaning there is strong evidence against the null hypothesis; <0.05 was considered moderately significant, and <0.10 was considered slightly significant.
Three statistical methods were used to compare the changes in the relative means between subjects when exposed to Building 1 and Building 2. First, a preliminary paired t-test was used to compare the means between Building 1 (i.e., moderate biophilic) and Building 2 (i.e., moderate to low biophilic) for each of the 32 electrodes across all channels (i.e., relative delta, relative alpha, relative beta, relative theta, and relative gamma power). Each electrode was assessed for statistically significant differences in power spectrum changes within each of the five bands using the standard delta t-test, performed in Excel.
Next, a one-way repeated measures ANCOVA regression analysis was conducted in Stata to examine global neurological activity across all bands. In this study, covariates included variables that could reasonably contribute to variations in the dependent variable (age, sex assigned at birth, and handedness). These variables were selected based on their known impact on neurological activity [
70,
71,
72]. For example, left-handed participants appear to elicit increased alpha frequency power during visually induced self-motion perception (i.e., the ability to perceive one’s body moving through space) much earlier than right-handed participants [
72]. Unlike the paired
t-test, the ANCOVA was conducted between averaged frequency band levels (i.e., comparing the difference across all electrodes for each frequency band as opposed to between individual electrodes). This method was applied based on findings that noted that neuroinflammatory activity is most often measured by global changes across the entirety of the brain rather than within individual regions [
73].
Lastly, post hoc ANCOVA was conducted using the Bonferroni correction to account for multiple comparisons between relative delta and all other bands. This post hoc ANCOVA corrected for multiple comparisons and accounted for potential false positives, which can occur when performing a large volume of statistical inferences [
74]. In this instance, the Bonferroni correction allowed for the control of false positives that may have arisen as a result of multiple comparisons between the five bands. However, it should be noted that the Bonferroni correction is the most conservative corrective method for EEG data adjustment and, as a result, may overestimate the degree of false positives [
75].
4. Results
The paired
t-test results (
Table 3) indicate statistically significant differences in relative delta power at electrodes F4 and T7 (
p < 0.05), and FC6 and P8 (
p < 0.10). Relative theta power showed significant differences at electrodes T7 (
p < 0.05) and CP6 (
p < 0.10). Relative alpha power showed significant differences at electrode T7 (
p < 0.05), relative beta power at electrode T7 (
p < 0.05), and relative gamma power at electrodes AF3 (
p < 0.05) and T7 (
p < 0.10). No significant differences were noted between any of the other electrodes. Significant differences occurred primarily in the frontal and temporal lobes, with the electrode T7 (temporal lobe) presenting across multiple bands. Although an examination of this electrode geography is out of the scope of this paper, the recurrent significance of the T7 electrode may be an interesting area for future research. The presence of the T7 electrode across all frequency bands likely indicates physiological artifacting [
76]. This artifacting may be the result of horizontal or vertical head movement, horizontal or vertical eye movement, eye blinking, or jaw movement [
76]. Data artifacts were identified and not included in the final analysis of datasets. Within this research framework, no correction for multiple comparisons was performed as electrodes are interpreted as single occurrences of neural inflammation rather than repeated samples within the same band.
Table 3.
Significant results from paired t-test.
Table 3.
Significant results from paired t-test.
| Frequency | Electrode | Mean Diff | p-Value |
|---|
| DELTA | F4 | −3.378 | 0.024 |
| | FC6 | −2.745 | 0.097 |
| | T7 | −2.555 | 0.044 |
| | P8 | −1.081 | 0.080 |
| THETA | T7 | −2.532 | 0.010 |
| | CP6 | −1.896 | 0.098 |
| ALPHA | T7 | −3.671 | 0.049 |
| BETA | T7 | −2.450 | 0.029 |
| GAMMA | AF3 | −3.887 | 0.021 |
| | T7 | −2.760 | 0.058 |
The ANCOVA results (
Table 4) indicate that the level of biophilia present in visual stimuli had a statistically significant effect on relative delta power compared to all other frequency bands after controlling for age (−0.026,
p-value = 0.000), handedness (−1.40,
p-value = 0.000), and sex as assigned at birth* (−0.31,
p-value = 0.080). This study reveals that the level of biophilia had a larger effect on relative delta power activity compared to relative alpha power (
p
2 = 0.79,
p-value = 0.001), relative beta power (
p
2 = 1.05,
p-value = 0.000), and relative gamma power (
p
2 = 0.73,
p-value = 0.002) activity. The level of biophilia had slightly less (though still statistically significant) effect on relative delta power activity compared to relative theta power activity (
p
2 = 0.60,
p-value = 0.011). From the results, it was found that an increase in biophilia was correlated with a decrease in relative delta power. Additionally, it was found that an increase in biophilia did not result in a statistically significant difference between Building 1 and Building 2 in relative alpha.
The results (
Table 5) illustrate that even after adjustment for multiple comparisons, the change in relative delta power is significantly lower than in all other bands, with the exception of relative theta (relative theta vs. relative delta,
p-value = 0.128). This finding may be due to phase synchronisation between delta and theta oscillations [
77]. Phase synchronization refers to the phenomenon where two or more oscillatory systems (in this case, brain waves) adjust their rhythms due to interaction, leading them to operate at a similar phase [
77]. This phenomenon can indicate that the brain regions producing delta and theta waves are functioning in a coordinated manner due to underlying neurophysiological processes or states that affect these bands in a related way, such as specific types of sleep or rest states where both bands play a significant role [
77]. Further research is required to determine whether the statistical significance noted in relative theta is the result of phase synchronisation. Finally, no statistically significant effect was observed in the difference in relative alpha frequency band between Building 1 and Building 2.
5. Discussion
This pilot study hypothesized that exposure to architecture with increased levels of biophilia may reduce neuroinflammatory activity. In accordance with the current literature [
28,
29,
30,
31], decreased relative delta and increased relative alpha power activity were indicated as suitable proxy variables for reduced neuroinflammation and reduced physiological stress responses.
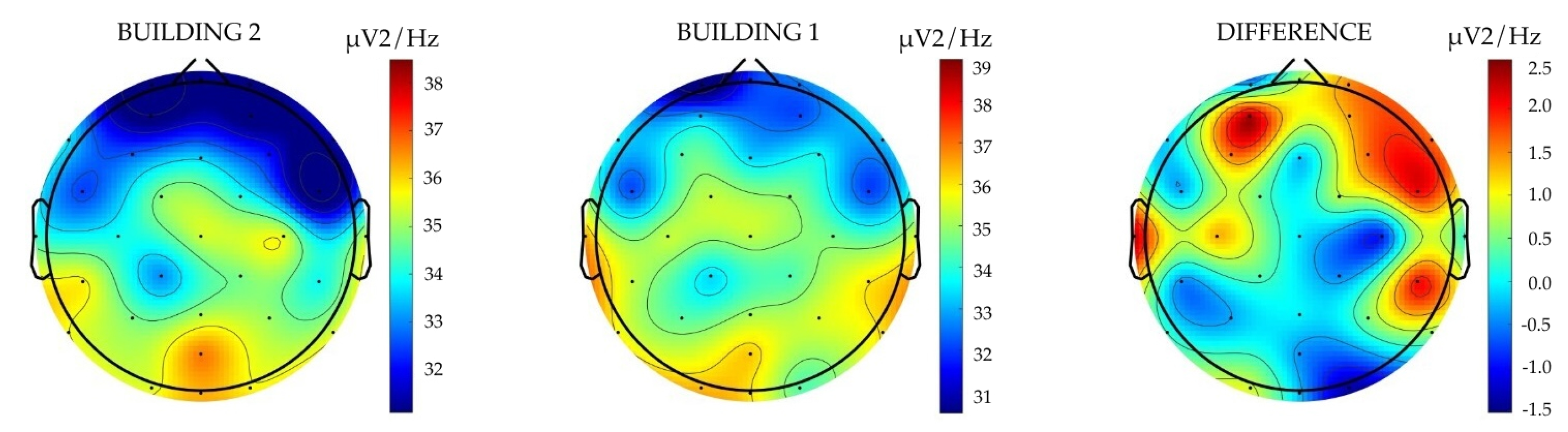
The results revealed different brain oscillation patterns in participants when visually exposed to moderate biophilic and moderate to low biophilic architectural environments. The largest differences were observed in relative delta power—participants exposed to the higher-level biophilic design exhibited a statistically significant decrease in relative delta power compared to when exposed to non-biophilic design. These findings are illustrated below (
Figure 4) by the results from the paired
t-test, ANCOVA, and covariance analysis models, all of which indicate a negative correlation between the level of biophilia and the expression of relative delta power. These findings suggest that exposure to Building 1, with the higher level of biophilia (as measured by the BHI), resulted in a decrease in relative delta power activity, indicating reduced measures of neuroinflammation in participants. The plot on the left illustrates the delta frequency power activity associated with Building 2, whereas the central plot represents the delta frequency power activity for Building 1. The plot on the right delineates the differential activity between the two buildings. The colour scheme employed in these plots signifies variations in the level of activity, measured in microvolts squared per hertz (
V
2/Hz), indicating different intensities of brain wave activity across the spectrum. These findings suggest that exposure to Building 1, with the higher level of biophilia (as measured by the BHI), resulted in decreased relative delta power activity, indicating reduced neuroinflammation.
The proposed association between the level of biophilia and neuroinflammation is further strengthened by findings that indicate that the impact of the level of biophilia on relative delta power is more significant when calculated using a global average (average of all electrodes) as opposed to a local value (individual electrodes). This difference is reflected in the results of the ANCOVA relative to the paired t-test. Namely, the degree of statistical significance was greater in the ANCOVA, which calculated the mean difference in neurological activity between Building 1 and Building 2. This finding is particularly compelling given that the ANCOVA model controlled for significant covariates (i.e., age, sex assigned at birth, and handedness). These findings suggest that the effect of biophilic design on relative delta power is not related to potential confounding variables such as age, sex assigned at birth, or handedness.
However, it should be noted that the possible interference of artifacting cannot be explicitly ruled out. Most notably, changes in relative delta power can occasionally be attributed to a type of biological artifact called a glossokinetic artifact [
78]. The glossokinetic artifact “is a well-known scalp EEG artifact characterised by deflections within the delta to low-theta frequency bands and dynamic polarity” [
79] (p. 481). However, given that the statistically significant relative delta power electrodes are distributed across the frontal, central parietal, and temporal lobes, and there was no observable statistical significance across relative theta power (with the exception of T7, which may be the result of an artifacting error), it can be assumed with a reasonable degree of certainty that these findings accurately represent participant relative band power differences when exposed to the visual stimuli. These preliminary pilot study results suggest that the effect on relative delta power may come from physiologically stress-mediated inflammatory activity in the brain as opposed to environmental artifacting.
Of interest within the results of this pilot study was the finding that no differences were observed in relative alpha power between Building 1 and Building 2 in either the local paired
t-test or global (ANCOVA) analysis. These findings are surprising given that recent research suggests that physiological stress responses may result in neuroinflammation, which can be measured by changes in relative alpha power activity [
28,
29,
30,
31]. Moreover, these findings are incongruent with findings from previous architectural neuroscience research, which established that exposure to biophilic design decreases physiological stress responses, as registered by increased relative alpha power activity [
5,
6,
7,
8].
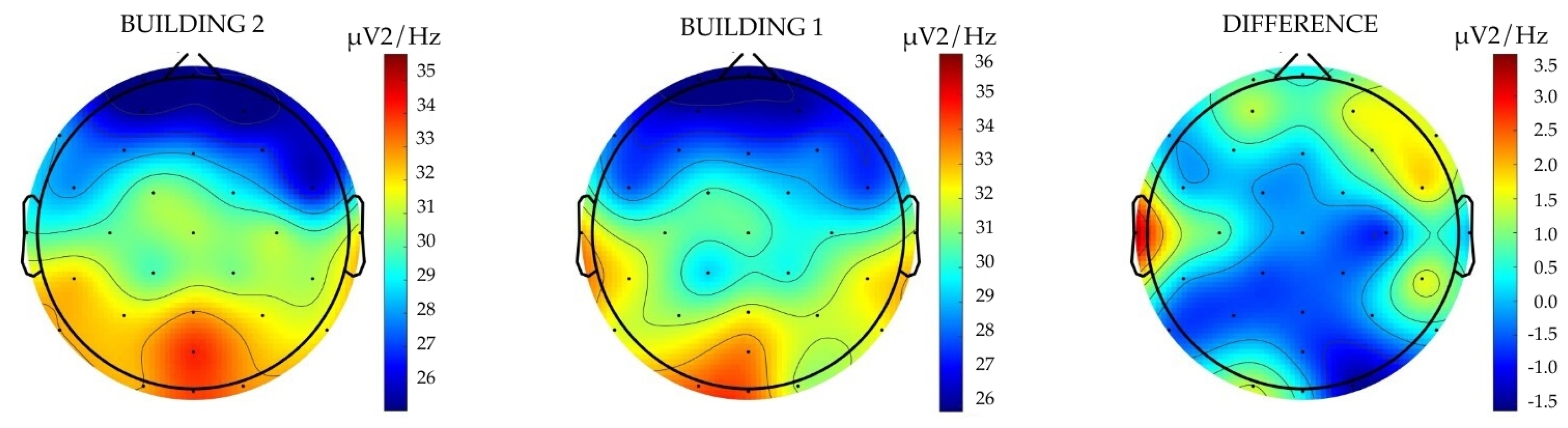
Given the research findings from existing studies, one would expect to see changes in relative alpha power activity when participants are exposed to visual stimuli that embody differential levels of biophilic design. Moreover, if biophilic design impacts neuroinflammatory activity, changes in relative delta power would presumably be accompanied by changes in relative alpha power. The observed lack of statistically significant findings regarding relative alpha power within this pilot study is anomalous in relation to the observed changes in relative delta power and findings from recent studies. One possible explanation for this discrepancy is that the Laplacian montage applied to the EEG data may have abstracted the relative alpha wave activity, thus limiting the observable statistical significance of the impact of increases in biophilia on relative alpha activity. The Laplacian montage is a relative EEG montage, meaning that the value for each electrode is determined in relation to the values of the surrounding electrodes. While this is helpful in calculating changes across local neurological activity (i.e., between specific electrodes/clusters of electrodes), it is less accurate when calculating changes between global regions (i.e., averages of all electrodes). From the observed results within this study, the changes in relative alpha power appear more globalised in nature. As
Figure 5 illustrates, the relative alpha power activity changes between Building 1 (
Figure 5; middle) and Building 2 (
Figure 5; left) appear consistent across the entire brain. These changes are illustrated by a diffuse difference in
V
2/Hz of −0.5 Hz across the brain, with increased activity of 2
V
2/Hz in the prefrontal and dorsolateral cortex (i.e., at the front or anterior of the brain). As a result, the Laplacian montage may not be adequate for measuring relative alpha power activity across the entire brain. Given that this research examines relative delta power and relative alpha power activity across the entire brain (not specific hemispheres or lobes), additional research using bipolar and referential montages is recommended.
This study’s results reveal conflicting findings. The observed decrease in relative delta power activity aligns with the proposed research hypothesis. However, the lack of observed difference in relative alpha power activity is anomalous in relation to observed changes in relative delta power and findings from recent neuroimmunology studies. Such a discrepancy underscores the complexity of neurophysiological responses to environmental stimuli and suggests a nuanced interaction that may not be fully captured by the current scope of the investigation and the Laplacian montage method utilised. The congruence of relative delta changes with the hypothesis suggests that there is a potential link between specific environmental features and neurophysiological markers of stress and relaxation. In contrast, the lack of significant findings in relative alpha frequencies calls into question the uniformity of these responses across different neural mechanisms or possibly suggests variability in individual sensitivities to the studied environmental conditions. It is plausible that this divergence in findings could be attributed to the limitations imposed by the small sample size and the localised nature of the Laplacian montage analysis of relative alpha power. A limited participant pool may not provide a sufficiently diverse representation of responses, thereby obscuring broader trends and potentially significant effects within subgroups or across different conditions. This raises the imperative for further expanded research that employs a larger sample size to examine the intricate dynamics between biophilic design elements and neurophysiological markers of wellbeing.
5.1. Key Findings
To review, this pilot study yielded two primary findings. First, this study found evidence that increases in the level of biophilia resulted in a decrease in the relative delta power in both localised electrode activity and global frequency band activity. This finding supports this study’s hypothesis. Second, no change was observed in relative alpha power between Building 1 and Building 2. This outcome was unexpected, given the association between physiological stress, neuroinflammation, and relative alpha power. The findings in relation to relative alpha are anomalous and deviate from existing findings on biophilic design and physiological stress responses. In this regard, the research yielded mixed outcomes as the observed reduction in relative delta underpins the hypothesis of the study, while the absence of observed statistical significance within the relative alpha parameters does not support the same assessment. It should be noted, however, that whilst the observed lack of statistically significant variance in relative power alpha is incongruous, it does not negate the study findings. Whilst the observable changes in relative power delta support the proposition that the incorporation of biophilic design elements into the built environment may lead to a decrease in stress and neuroinflammation, the mixed and anomalous nature of the findings means that further scholarly investigation is needed to validate the study results and explore further the complex relationship between biophilia and neurophysiological stress responses.
5.2. Research Significance and Implications
While future research is needed to corroborate this study’s findings and develop a more nuanced understanding of neurophysiological responses to the built environment, these findings, in conjunction with emerging neuroimmunological research, highlight that the design of the built environment may have a more profound impact on human health through its effect on neuroimmunology than previously understood. This study further develops the existing body of knowledge in relation to the impact of biophilia in stress response downregulation through the finding that biophilic design may also downregulate neuroinflammatory responses. The field of neuroimmunology has shown that physiological stress has many downstream implications for health, and that exposure to physiological stressors can produce neurochemical signals that activate the body’s fight or flight protection mechanisms [
20,
80]. These adaptations help the person evade threats by triggering a number of physiological changes including an elevated heart rate and increased blood pressure [
38]. Whilst in acute risk settings, these neurochemical responses are beneficial [
39], if exposure to the stressor is chronic, an inflammatory cascade can occur [
41] during which prolonged neuroendocrine responses lead to a dysregulation of the immune system [
25]. Dysregulated activity in the immune system can cause chronic systemic inflammation [
39], which can result in lasting cellular, molecular, and epigenetic changes [
12], long-term damage to organs and tissues [
25], and can play a role in the development of psychiatric and neurodegenerative disorders [
40]. These stress-related diseases account for a significant proportion of global pathological morbidity and mortality [
41].
While this study only measured acute exposure to biophilic design, the findings of this pilot study may have ramifications in relation to longer-term exposure to biophilic design and its potential role in downregulating neuroinflammatory activity and reducing the impact of the situational environment on the development of stress-related diseases. Further research into the impact of long-term exposure to biophilic (and non-biophilic) design on neuroinflammation is needed to develop a better understanding of impact of temporal variations in exposure to biophilia on neuroinflammation. Developing a better understanding of the impact of the built environment on neuroinflammation is particularly pertinent in light of rapid urbanisation. According to a forecast by the United Nations, by the year 2050, urban settings are expected to be home to two-thirds of the global population [
81]. Furthermore, studies have shown that individuals in developed countries typically spend upwards of 90% of their time indoors [
82]. This tendency is markedly pronounced amongst certain vulnerable demographics, such as the elderly, infants, young children, and those with compromised immune systems or mobility issues [
83]. It is predicted that these numbers will increase in the near future due to the emergence of epidemiological conditions such as COVID-19 [
84] and climate-change-related climatic volatility [
83].
Studies show that urban residents exhibit a higher level of stress than rural residents. In a 2011 study by Lederbogen et al., urban residents were found to have more heightened amygdala (i.e., a part of the brain that governs fear responses) sensitivity and reactivity than those who live in rural environments [
85]. Whilst the volume and complexity of variables influencing urban living make it difficult to isolate any single contributing variable, longitudinal studies indicate that the effects of urbanicity on neurophysiological responses cannot be explained wholly by epidemiological variables alone [
86,
87]. Given that urban dwellers experience higher stress levels than their rural counterparts, the incorporation of higher levels of biophilic design into urban environments as a mechanism by which to reduce neuroinflammation and downregulate stress may have profound health ramifications. The potential of design for reduced neuroinflammation and to serve as a means by which to help create healthier cities and potentially reduce stress-related disease burden is an area of research that should be explored further.
This study’s finding that exposure to biophilic design may elicit beneficial anti-neuroinflammatory responses may present opportunities for architects to contribute to better health and wellbeing outcomes across all spectrums of the built environment. In the face of reported increasing global stress levels, this study offers evidence that even brief passive exposure to biophilic forms may elicit positive stress reduction benefits. Although participants were only exposed to images of the biophilic building for 9 min, statistically significant reductions were observed in neurological activity indicative of neuroinflammation. Moreover, the potential benefits were likely underestimated because of the 2D, static nature of the visual stimuli (these limitations are discussed in greater detail in the following section). Given the large number of people exposed to urban architecture, consideration should be given to the potential impact of the introduction of biophilic design as a cost-effective form of public health intervention.
5.3. Scope and Limitations
Although this study has provided evidence that biophilic design features may work to mediate neuroinflammatory activity, several limitations must be acknowledged. First, this pilot study is limited by its relatively low number of participants. Future research should endeavour to increase the sample size and explore the effects of differences across a range of demographic variables such as gender, age, and educational background.
Second, this study is further limited by the uni-sensory and 2D nature of the visual stimuli. Utilising emerging technologies, such as VR or augmented reality (AR), may enable future research to test more immersive sensory experiences.
Third, while the BHI is highly valuable and lays the groundwork for exploring biophilic elements, its subjective method of feature analysis lacks objective empirical rigour and depth. Future studies that incorporate computational methods would help to bolster analytical precision, and facilitate a more nuanced and thorough investigation of the impact of biophilia on neuroinflammation. Moreover, future research may look to generative AI to produce visual stimuli which embody quantifiable variations in biophilic and biomorphic content.
Fourth, this study is also limited by the use of averaged frequency band levels (i.e., comparing the difference across all electrodes for each frequency band instead of between individual electrodes). Future research would benefit from examining frequency band activity at individual electrodes across the prefrontal, frontal, parietal, and occipital lobes. Furthermore, incorporating additional clinical biosensors, such as eye-tracking, HRV, and GSR, and laboratory metrics, such as salivary cortisol and blood measurements, may also help to examine the relationship between built environment stimuli and observed neuroinflammatory activity.
Lastly, additional consideration must be given to the statistical analysis methods used. Given that frequency bands are not independent of one another (due to phase synchronisation), future researchers are advised to consider using a multivariate analysis of covariance (MANCOVA) as a method of statistical analysis. Despite these limitations, the findings produced by this pilot study serve as a stepping stone to broaden our understanding of architectural neuroimmunology, and help lay the foundation for future academic inquiry in the field.
5.4. Future Research
This study’s findings suggest that incorporating biophilic design into the built environment may contribute to reduced stress and neuroinflammation. The anomalies observed in the results in relation to relative delta and relative alpha power activity underscore the necessity for further investigation. Notably, the unexpected findings regarding relative alpha highlight the need to examine more thoroughly the function of relative alpha power activity within the context of biophilically mediated neuroinflammation. Emphasis should be placed on addressing matters related to the statistical significance of the sample size.
Second, further empirical research is required to gain a comprehensive understanding of the biophilic forms implicated in neuroinflammatory responses. Controlled studies which isolate individual biophilic features and test their respective impacts on neuroinflammatory responses would greatly advance the field of architectural neuroimmunology and provide more practical guidance for architects, planners, and built environment designers.
Third, research should be conducted into the impact of varying degrees of exposure to biophilic design on neuroimmunological activity. For example, does simply driving past biophilic architectural forms provide benefits to the subject, or is long-term exposure required? Does this vary for different demographics, or for subgroups such as those with different types of neurodivergence, or for those exhibiting chronic or acute high stress response conditions such as post-traumatic stress disorder (PTSD)?
Fourth, given the multicausal nature of neuroinflammation, future research could attempt to disentangle how, if at all, individual stressors interact with one another to create cumulative or net effects. This research will inevitably be complicated by the substantial volume and complexity of social, environmental, and physiological factors that mediate physiological stress responses.
Lastly, future research may wish to consider the corollary of this research—namely, are there specific design elements that increase neuroinflammation and physiological stress responses? Research examining the potentially upregulating neuroimmunological impacts of non-biophilic architectural forms is a potential next step for this emerging knowledge domain. If confirmed, these findings may help contribute to insights in relation to the disparate stress levels observed between urban and rural dwellers, and greatly aid our understanding of the ways in which the design of the built environment impacts neurophysiological responses.