Quantitative Assessment of the Time to End Bainitic Transformation
Abstract
1. Introduction
2. Materials and Methods
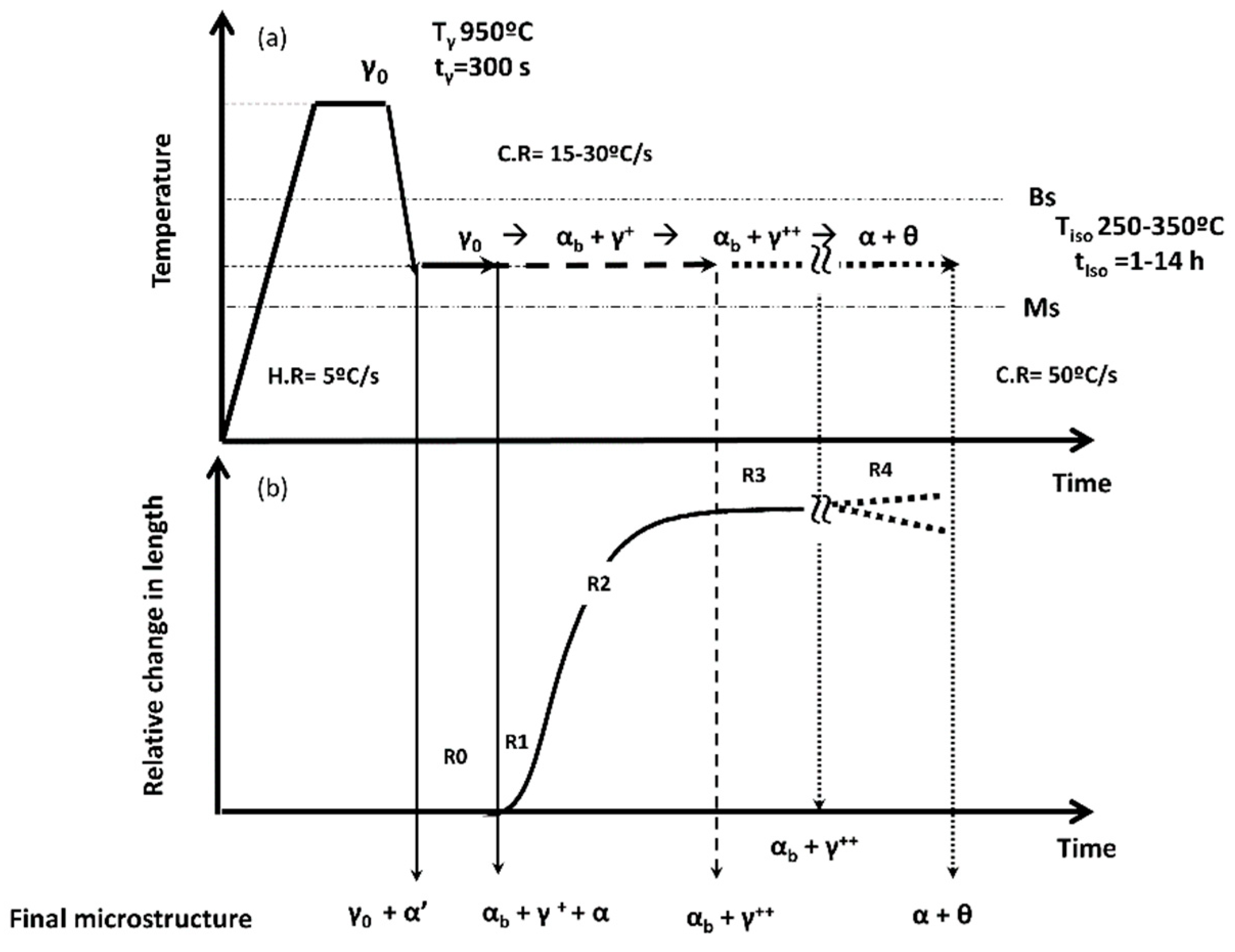
- A first stage, R0, called the incubation period, during which the transformation has not yet started, or it is not yet detectable, and the microstructure remains fully austenitic;
- The transformation period, where nucleation and growth occur, and the microstructure tends to evolve from γ to αb + γ+ (R1 & R2). Differences between R1 and R2 become clear later in the text;
- The last stage in which the signal reaches a steady-state, and transformation does not proceed any further, R3. This region is not always easily identifiable, because in some cases, the RCL signal does not achieve a clear steady-state, but it remains increasing or decreasing with a low but constant slope;
- If treatments are performed for much longer times than necessary, R4, further changes in the microstructure can take place, e.g., autotempering and decomposition of the bainitic microstructure [21,22,23], and discontinuous lines in Figure 1b intend to highlight the fact that the occurrence of those can lead to different outcomes in the dilatometric curve. For example, decomposition of residual austenite into a mixture of ferrite and cementite can lead to a net contraction or expansion depending on the amount of C in solid solution [29], whereas carbon partitioning i.e., further C enrichment of austenite, due to autotempering of bainitic ferrite, will introduce a contraction, and the precipitation of carbides from austenite and bainitic ferrite will induce an expansion and a contraction, respectively.
3. Methods for the Estimation of the End of the Bainite Transformation (EBT). Results and Discussion
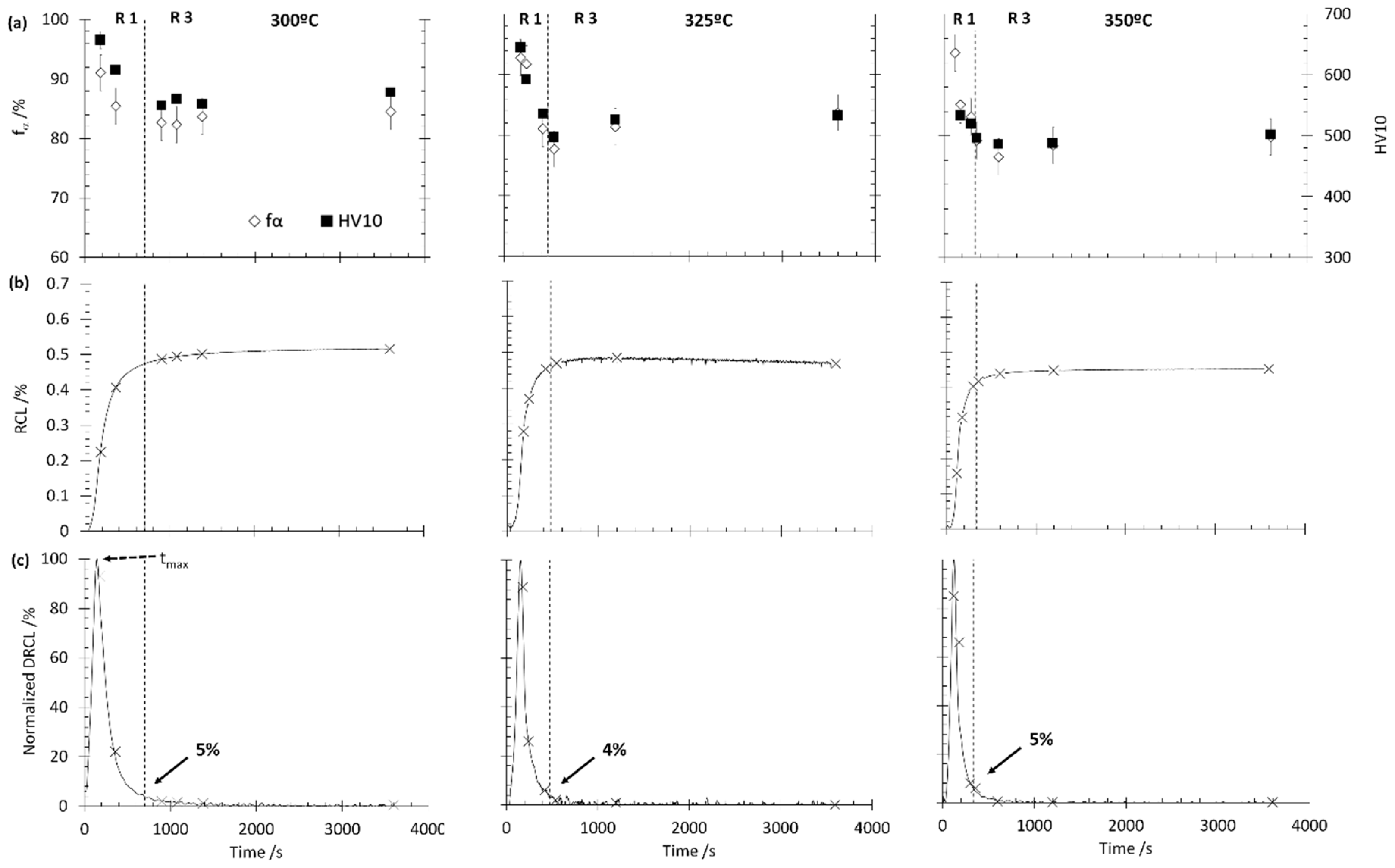
3.1. Method 0. Interrupted Isothermal Tests
3.2. Method I. Graphical Method
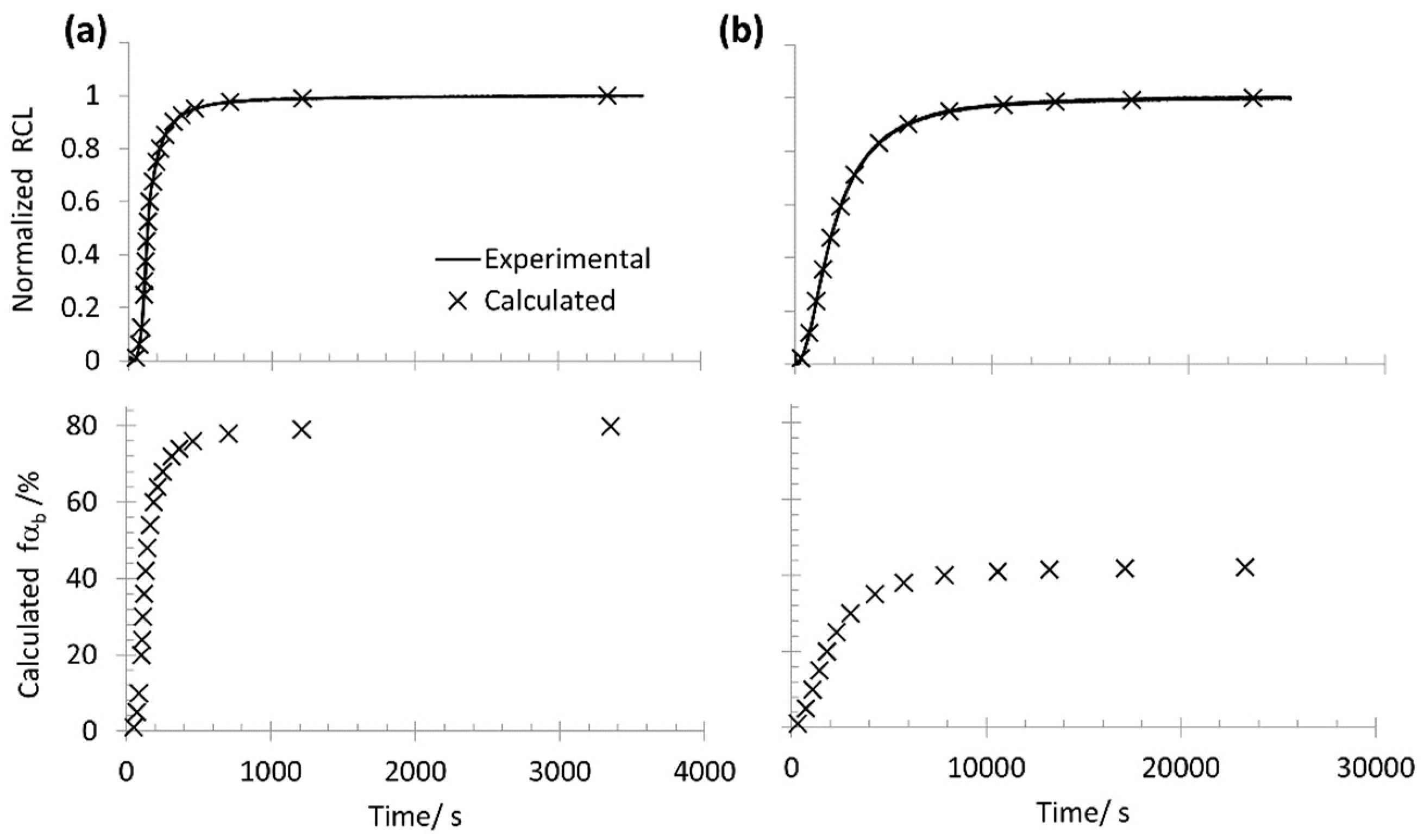
3.3. Method II. Dilatometric Curve Simulation by Atomic Volumes
- From XRD data, bainitic ferrite fraction and lattice parameters, and , are calculated from the lattice parameters, and its C content () is derived using [52]. It has been proven that bainitic ferrite is fairly stable through the whole transformation regardless of the degree of transformation, and therefore, and , i.e., , will remain constant from the beginning to the end of the transformation [24].
- For = 1%, the lever rule is applied to calculate for that amount of transformed bainitic ferrite.
- With all that information, and using Equation (4), the lattice parameters at the studied temperature are calculated. Then, Equation (5) is used to calculate , associated with the formation of 1% of bainitic ferrite.
- The process is repeated for different values of up to the fraction provided by the XRD experiment, .
- The newly obtained results are then normalized, as it is also the experimental curve.
- From the normalized , a match is found in the—also normalized—dilatometric curve, and therefore, its corresponding experimental time is established.
- Finally, the EBT time would be that at which the calculated , but a less conservative value will be obtained allowing for the XRD uncertainty (±3%), so the EBT can also be calculated as that at which − 1% or that when − 2%.
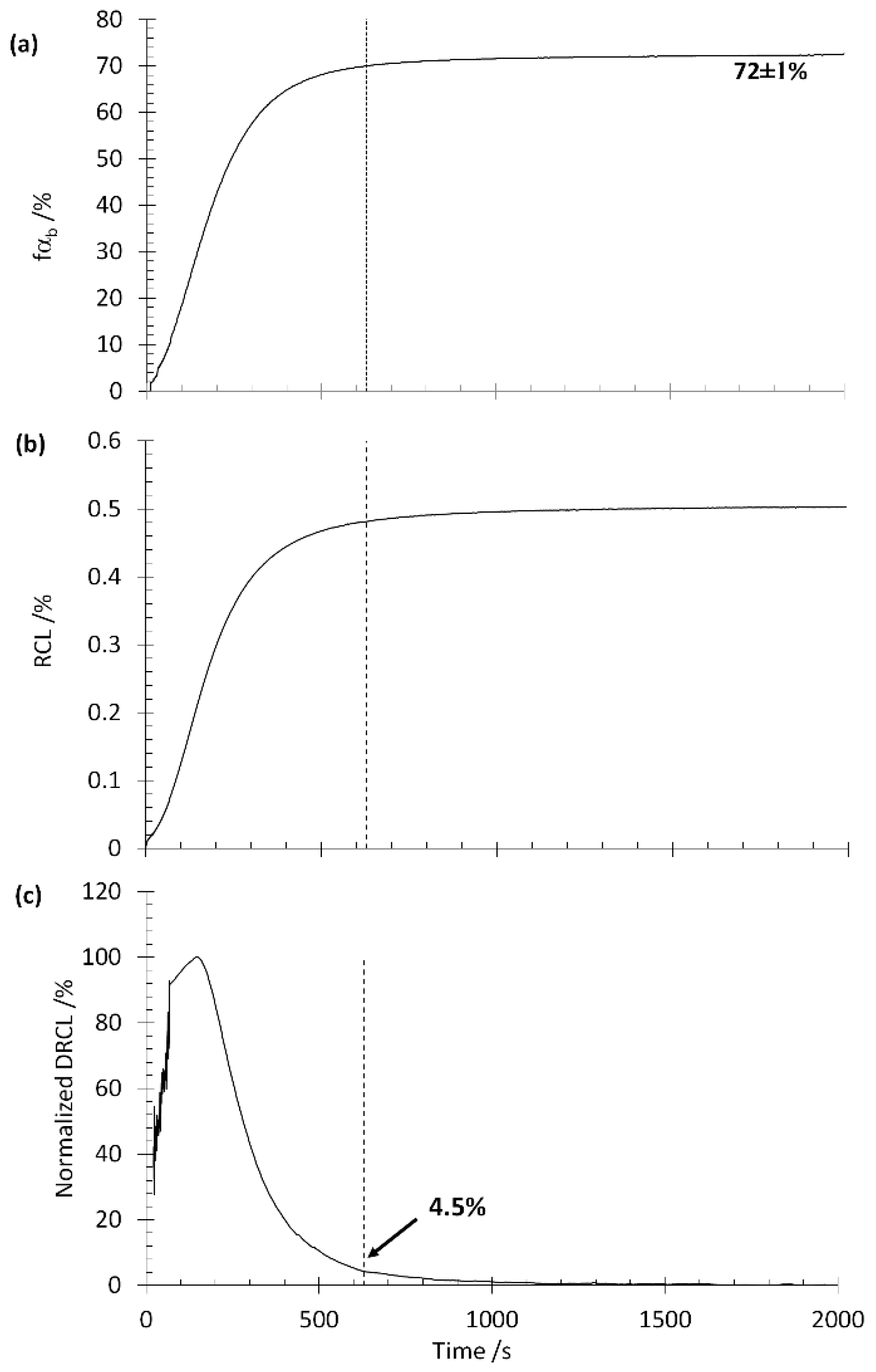
3.4. Method III. Rate of Transformation Approach
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Christian, J.W.; Olson, G.B.; Cohen, M. Classification of displacive transformations: What is a martensitic transformation? J. Phys. IV Colloq. 1995, 05, C8-3–C8-10. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H. Bainite in Steels: Transformations, Microstructure and Properties, 3rd ed.; Institute of Materials, Minerals and Mining: London, UK, 2015. [Google Scholar]
- Bhadeshia, H.K.D.H. Bainite in Steels: Theory and Practice, 3rd ed.; Maney Publishing: Leeds, UK, 2015; p. 616. [Google Scholar]
- Bhadeshia, H.K.D.H.; Honeycombe, R.W.K. Steels: Microstructure and Properties; Butterworths-Heinemann (Elsevier): Oxford, UK, 2006. [Google Scholar]
- Caballero, F.G.; Morales-Rivas, L.; Garcia-Mateo, C. Retained austenite: Stability in a nanostructured bainitic steel. In Encyclopedia of Iron, Steel, and Their Alloys; Taylor & Francis: Abingdon-on-Thames, UK, 2016; pp. 3077–3087. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.G.; Miller, M.K.; Jimenez, J.A. On measurement of carbon content in retained austenite in a nanostructured bainitic steel. J. Mater. Sci. 2012, 47, 1004–1010. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.G. Ultra-high-strength bainitic steels. ISIJ Int. 2005, 45, 1736–1740. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.G. Understanding the mechanical properties of nanostructured bainite. In Handbook of Mechanical Nanostructuring; Aliofkhazraei, M., Ed.; Wiley: Weinheim, Germany, 2015; Volume 1, pp. 35–65. [Google Scholar]
- Rakha, K.; Beladi, H.; Timokhina, I.; Xiong, X.; Kabra, S.; Liss, K.D.; Hodgson, P. On low temperature bainite transformation characteristics using in-situ neutron diffraction and atom probe tomography. Mater. Sci. Eng. A 2014, 589, 303–309. [Google Scholar] [CrossRef]
- Xing, X.L.; Yuan, X.M.; Zhou, Y.F.; Qi, X.W.; Lu, X.; Xing, T.H.; Ren, X.J.; Yang, Q.X. Effect of bainite layer by lsmcit on wear resistance of medium-carbon bainite steel at different temperatures. Surf. Coat. Technol. 2017, 325, 462–472. [Google Scholar] [CrossRef]
- Zhang, M.; Wang, T.S.; Wang, Y.H.; Yang, J.; Zhang, F.C. Preparation of nanostructured bainite in medium-carbon alloysteel. Mater. Sci. Eng. A 2013, 568, 123–126. [Google Scholar] [CrossRef]
- Hu, F.; Wu, K.M.; Wan, X.L.; Rodionova, I.; Shirzadi, A.A.; Zhang, F.C. Novel method for refinement of retained austenite in micro/nano-structured bainitic steels. Mater. Sci. Technol. 2017, 33, 1360–1365. [Google Scholar] [CrossRef]
- Zhao, J.; Hou, C.S.; Zhao, G.; Zhao, T.; Zhang, F.C.; Wang, T.S. Microstructures and mechanical properties of bearing steels modified for preparing nanostructured bainite. J. Mater. Eng. Perform. 2016, 25, 4249–4255. [Google Scholar] [CrossRef]
- Nishijima, H.; Tomota, Y.; Su, Y.; Gong, W.; Suzuki, J.I. Monitoring of bainite transformation using in situ neutron scattering. Metals 2016, 6, 16. [Google Scholar] [CrossRef]
- Kabirmohammadi, M.; Avishan, B.; Yazdani, S. Transformation kinetics and microstructural features in low-temperature bainite after ausforming process. Mater. Chem. Phys. 2016, 184, 306–317. [Google Scholar] [CrossRef]
- Jiang, T.; Liu, H.; Sun, J.; Guo, S.; Liu, Y. Effect of austenite grain size on transformation of nanobainite and its mechanical properties. Mater. Sci. Eng. A 2016, 666, 207–213. [Google Scholar] [CrossRef]
- Yakubtsov, I.A.; Purdy, G.R. Analyses of transformation kinetics of carbide-free bainite above and below the athermal martensite-start temperature. Metall. Mater. Trans. A 2011, 43, 437–446. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.G. Nanocrystalline bainitic steels for industrial applications. In Nanotechnology for Energy Sustainability; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2017; pp. 707–724. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.G.; Bhadeshia, H.K.D.H. Acceleration of low-temperature bainite. ISIJ Int. 2003, 43, 1821–1825. [Google Scholar] [CrossRef]
- Eres-Castellanos, A.; Morales-Rivas, L.; Latz, A.; Caballero, F.G.; Garcia-Mateo, C. Effect of ausforming on the anisotropy of low temperature bainitic transformation. Mater. Charact. 2018, 145, 371–380. [Google Scholar] [CrossRef]
- Saha Podder, A.; Bhadeshia, H.K.D.H. Thermal stability of austenite retained in bainitic steels. Mater. Sci. Eng. A 2010, 527, 2121–2128. [Google Scholar] [CrossRef]
- Morales-Rivas, L.; Yen, H.W.; Huang, B.M.; Kuntz, M.; Caballero, F.G.; Yang, J.R.; Garcia-Mateo, C. Tensile response of two nanoscale bainite composite-like structures. JOM 2015, 67, 2223–2235. [Google Scholar] [CrossRef]
- Avishan, B.; Garcia-Mateo, C.; Yazdani, S.; Caballero, F.G. Retained austenite thermal stability in a nanostructured bainitic steel. Mater. Charact. 2013, 81, 105–110. [Google Scholar] [CrossRef]
- Rementeria, R.; Jimenez, J.A.; Allain, S.Y.P.; Geandier, G.; Poplawsky, J.D.; Guo, W.; Urones-Garrote, E.; Garcia-Mateo, C.; Caballero, F.G. Quantitative assessment of carbon allocation anomalies in low temperature bainite. Acta Mater. 2017, 133, 333–345. [Google Scholar] [CrossRef]
- Sourmail, T.; Smanio, V. Determination of ms temperature: Methods, meaning and influence of ‘slow start’ phenomenon. Mater. Sci. Technol. 2013, 29, 883–888. [Google Scholar] [CrossRef]
- Sourmail, T.; Smanio, V.; Ziegler, C.; Heuer, V.; Kuntz, M.; Caballero, F.G.; Garcia-Mateo, C.; Cornide, J.; Elvira, R.; Leiro, A.; et al. Novel Nanostructured Bainitic Steel Grades to Answer the Need for High-Performance Steel Components (Nanobain); European Commission: Luxembourg, 2013; p. 129. [Google Scholar]
- Garcia-Mateo, C.; Sourmail, T.; Caballero, F.G.; Smanio, V.; Kuntz, M.; Ziegler, C.; Leiro, A.; Vuorinen, E.; Elvira, R.; Teeri, T. Nanostructured steel industrialisation: Plausible reality. Mater. Sci. Technol. 2014, 30, 1071–1078. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H. Thermodynamic analysis of isothermal transformation diagrams. Met. Sci. 1982, 16, 159–165. [Google Scholar] [CrossRef]
- Caballero, F.G.; Garcia-Mateo, C.; de Andres, C.G. Dilatometric study of reaustenitisation of high silicon bainitic steels: Decomposition of retained austenite. Mater. Trans. JIM 2005, 46, 581–586. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Jimenez, J.A.; Yen, H.W.; Miller, M.K.; Morales-Rivas, L.; Kuntz, M.; Ringer, S.P.; Yang, J.R.; Caballero, F.G. Low temperature bainitic ferrite: Evidence of carbon super-saturation and tetragonality. Acta Mater. 2015, 91, 162–173. [Google Scholar] [CrossRef]
- ASTM International. Standard Practice for X-Ray Determination of Retained Austenite in Steel with Near Random Crystallographic Orientation; ASTM International: West Conshohocken, PA, USA, 2013; Volume ASTM E975-13, p. 7. [Google Scholar]
- Jarvinen, M. Texture effect in x-ray analysis of retained austenite in steels. Text. Microstruct. 1996, 26–27, 93–101. [Google Scholar] [CrossRef]
- Dyson, D.J.; Holmes, B. Effect of alloying additions on lattice parameter of austenite. J. Iron Steel Inst. 1970, 208, 469–474. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.G. The role of retained austenite on tensile properties of steels with bainitic microstructures. Mater. Trans. JIM 2005, 46, 1839–1846. [Google Scholar] [CrossRef]
- Arnell, R.D.; Ridal, K.A.; Durnin, J. Determination of retained austenite in steel by x-ray diffraction. J. Iron Steel Inst. 1968, 206, 1035–1036. [Google Scholar]
- Allain, S.Y.P.; Aoued, S.; Quintin-Poulon, A.; Goune, M.; Danoix, F.; Hell, J.C.; Bouzat, M.; Soler, M.; Geandier, G. In situ investigation of the iron carbide precipitation process in a Fe-C-Mn-Si Q&P steel. Materials 2018, 11, 1087. [Google Scholar]
- Allain, S.Y.P.; Gaudez, S.; Geandier, G.; Hell, J.C.; Gouné, M.; Danoix, F.; Soler, M.; Aoued, S.; Poulon-Quintin, A. Internal stresses and carbon enrichment in austenite of quenching and partitioning steels from high energy x-ray diffraction experiments. Mater. Sci. Eng. A 2018, 710, 245–250. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.; Bhadeshia, H. Development of hard bainite. ISIJ Int. 2003, 43, 1238–1243. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.G.; Sourmail, T.; Kuntz, M.; Cornide, J.; Smanio, V.; Elvira, R. Tensile behaviour of a nanocrystalline bainitic steel containing 3 wt% silicon. Mater. Sci. Eng. A 2012, 549, 185–192. [Google Scholar] [CrossRef]
- Cornide, J.; Garcia-Mateo, C.; Capdevila, C.; Caballero, F.G. An assessment of the contributing factors to the nanoscale structural refinement of advanced bainitic steels. J. Alloy. Compd. 2013, 577, S43–S47. [Google Scholar] [CrossRef]
- Hehemann, R.F.; Troiano, A.R. Characteristics and Stabilization of the Bainite Reaction; Metal Hydrides Inc: Beverly, MA, USA, 1954. [Google Scholar]
- Chong, S.H. Transformation and Toughness of Iron-9 Percent Nickel Alloy. Ph.D. Thesis, Sheffield Hallam University, Sheffield, UK, 1998. [Google Scholar]
- Garcia-Mateo, C.; Caballero, F.G.; Sourmail, T.; Cornide, J.; Smanio, V.; Elvira, R. Composition design of nanocrystalline bainitic steels by diffusionless solid reaction. Met. Mater. Int. 2014, 20, 405–415. [Google Scholar] [CrossRef]
- Xu, Y.; Xu, G.; Mao, X.; Zhao, G.; Bao, S. Method to evaluate the kinetics of bainite transformation in low-temperature nanobainitic steel using thermal dilatation curve analysis. Metals 2017, 7, 330. [Google Scholar] [CrossRef]
- Garcia-Mateo, C.; Caballero, F.G.; Capdevila, C.; Garcia de Andres, C. Estimation of dislocation density in bainitic microstructures using high-resolution dilatometry. Scr. Mater. 2009, 61, 855–858. [Google Scholar] [CrossRef]
- Santajuana, M.A.; Rementeria, R.; Kuntz, M.; Jimenez, J.A.; Caballero, F.G.; Garcia-Mateo, C. Low-temperature bainite: A thermal stability study. Metall. Mater. Trans. B 2018, 49, 2026–2036. [Google Scholar] [CrossRef]
- Moyer, J.M.; Ansell, G.S. The volume expansion accompanying the martensite transformation in iron-carbon alloys. Metall. Trans. A 1975, 6, 1785. [Google Scholar] [CrossRef]
- Hulme-Smith, C.N.; Peet, M.J.; Lonardelli, I.; Dippel, A.C.; Bhadeshia, H.K.D.H. Further evidence of tetragonality in bainitic ferrite. Mater. Sci. Technol. 2015, 31, 254–256. [Google Scholar] [CrossRef]
- Bhadeshia, H.K.D.H. Carbon in cubic and tetragonal ferrite. Philos. Mag. 2013, 93, 3714–3725. [Google Scholar] [CrossRef]
- Jang, J.H.; Bhadeshia, H.K.D.H.; Suh, D.W. Solubility of carbon in tetragonal ferrite in equilibrium with austenite. Scr. Mater. 2013, 68, 195–198. [Google Scholar] [CrossRef]
- Lee, S.J.; Lusk, M.T.; Lee, Y.K. Conversional model of transformation strain to phase fraction in low alloy steels. Acta Mater. 2007, 55, 875–882. [Google Scholar] [CrossRef]
- Cohen, M. The strengthening of steel. Trans. Metall. AIME 1962, 224, 638–657. [Google Scholar]
- Garcia-Mateo, C.; Peet, M.; Caballero, F.G.; Bhadeshia, H.K.D.H. Tempering of hard mixture of bainitic ferrite and austenite. Mater. Sci. Technol. 2004, 20, 814–818. [Google Scholar] [CrossRef]
- Rementeria, R.; Garcia-Mateo, C.; Caballero, F.G. New insights into carbon distribution in bainitic ferrite. HTM J. Heat Treat. Mater. 2018, 73, 68–79. [Google Scholar] [CrossRef]
- Rementeria, R.; Capdevila, C.; Dominguez-Reyes, R.; Poplawsky, J.D.; Guo, W.; Urones-Garrote, E.; Garcia-Mateo, C.; Caballero, F.G. Carbon clustering in low-temperature bainite. Metall. Mater. Trans. A 2018, 49, 5277–5287. [Google Scholar] [CrossRef]
- Rementeria, R.; Poplawsky, J.D.; Aranda, M.M.; Guo, W.; Jimenez, J.A.; Garcia-Mateo, C.; Caballero, F.G. Carbon concentration measurements by atom probe tomography in the ferritic phase of high-silicon steels. Acta Mater. 2017, 125, 359–368. [Google Scholar] [CrossRef]



| Alloy | C | Si | Mn | Cr | Mo | Ni | Cu | Ms |
|---|---|---|---|---|---|---|---|---|
| Steel 1 | 0.43 | 3.05 | 0.71 | 0.98 | 0.21 | 0.09 | 0.14 | 280 |
| Steel 2 | 0.99 | 2.47 | 0.74 | 0.98 | 0.02 | 0.12 | 0.19 | 173 |
| Steel 3 | 0.31 | 1.52 | 2.44 | 320 |
| Alloy | TIso/°C | aγ/Å | aα/Å | cα/Å |
|---|---|---|---|---|
| Steel 1 | 300 | 3.612 | 2.852 | 2.876 |
| 325 | 3.614 | 2.853 | 2.875 | |
| 350 | 3.609 | 2.854 | 2.877 | |
| Steel 2 | 250 | 3.627 | 2.852 | 2.877 |
| 300 | 3.626 | 2.850 | 2.876 | |
| 350 | 3.621 | 2.854 | 2.874 |
| Phase (i) | ||
|---|---|---|
| fcc (γ) | 2.065 × 10−5 [21] | |
| bct (a) | 1.490 × 10−5 [51] |
| Alloy | TIso (°C) | Method 0 | Method I | EBT/s | |||||
|---|---|---|---|---|---|---|---|---|---|
| Method II | Method III % of the Maximum DRCL | ||||||||
| 0% | 1% | 2% | 4% | 3% | 2% | ||||
| Steel 1 | 300 | 700 | 1700 | 3600 | 1252 | 1123 | 694 | 777 | 932 |
| 325 | 470 | 500 | 3596 | 3596 | 3596 | 480 | 490 | 530 | |
| 350 | 340 | 1100 | 3347 | 1213 | 705 | 378 | 406 | 475 | |
| Steel 2 | 250 | 21,000 | 42,014 | 50,274 | 37,050 | 27,581 | 25,458 | - | - |
| 300 | 6500 | 12,500 | 28,764 | 15,004 | 11,455 | 8020 | 8690 | - | |
| 350 | 5200 | 11,000 | 23,284 | 10,599 | 7859 | 7503 | 8032 | - | |
| Steel 3 | 400 | 630 | 1264 | 1988 | 870 | 688 | 653 | 724 | 818 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santajuana, M.A.; Eres-Castellanos, A.; Ruiz-Jimenez, V.; Allain, S.; Geandier, G.; Caballero, F.G.; Garcia-Mateo, C. Quantitative Assessment of the Time to End Bainitic Transformation. Metals 2019, 9, 925. https://doi.org/10.3390/met9090925
Santajuana MA, Eres-Castellanos A, Ruiz-Jimenez V, Allain S, Geandier G, Caballero FG, Garcia-Mateo C. Quantitative Assessment of the Time to End Bainitic Transformation. Metals. 2019; 9(9):925. https://doi.org/10.3390/met9090925
Chicago/Turabian StyleSantajuana, Miguel A., Adriana Eres-Castellanos, Victor Ruiz-Jimenez, Sebastien Allain, Guillaume Geandier, Francisca G. Caballero, and Carlos Garcia-Mateo. 2019. "Quantitative Assessment of the Time to End Bainitic Transformation" Metals 9, no. 9: 925. https://doi.org/10.3390/met9090925
APA StyleSantajuana, M. A., Eres-Castellanos, A., Ruiz-Jimenez, V., Allain, S., Geandier, G., Caballero, F. G., & Garcia-Mateo, C. (2019). Quantitative Assessment of the Time to End Bainitic Transformation. Metals, 9(9), 925. https://doi.org/10.3390/met9090925









