Development of an Analytical Model for the Extraction of Manganese from Marine Nodules
Abstract
1. Introduction
2. Materials and Methods
2.1. Manganese Nodule Samples
2.2. Tailings Samples
2.3. Reagents and Leaching Parameters
2.4. Experimental Design
2.5. Adjustment of an Analytical Model
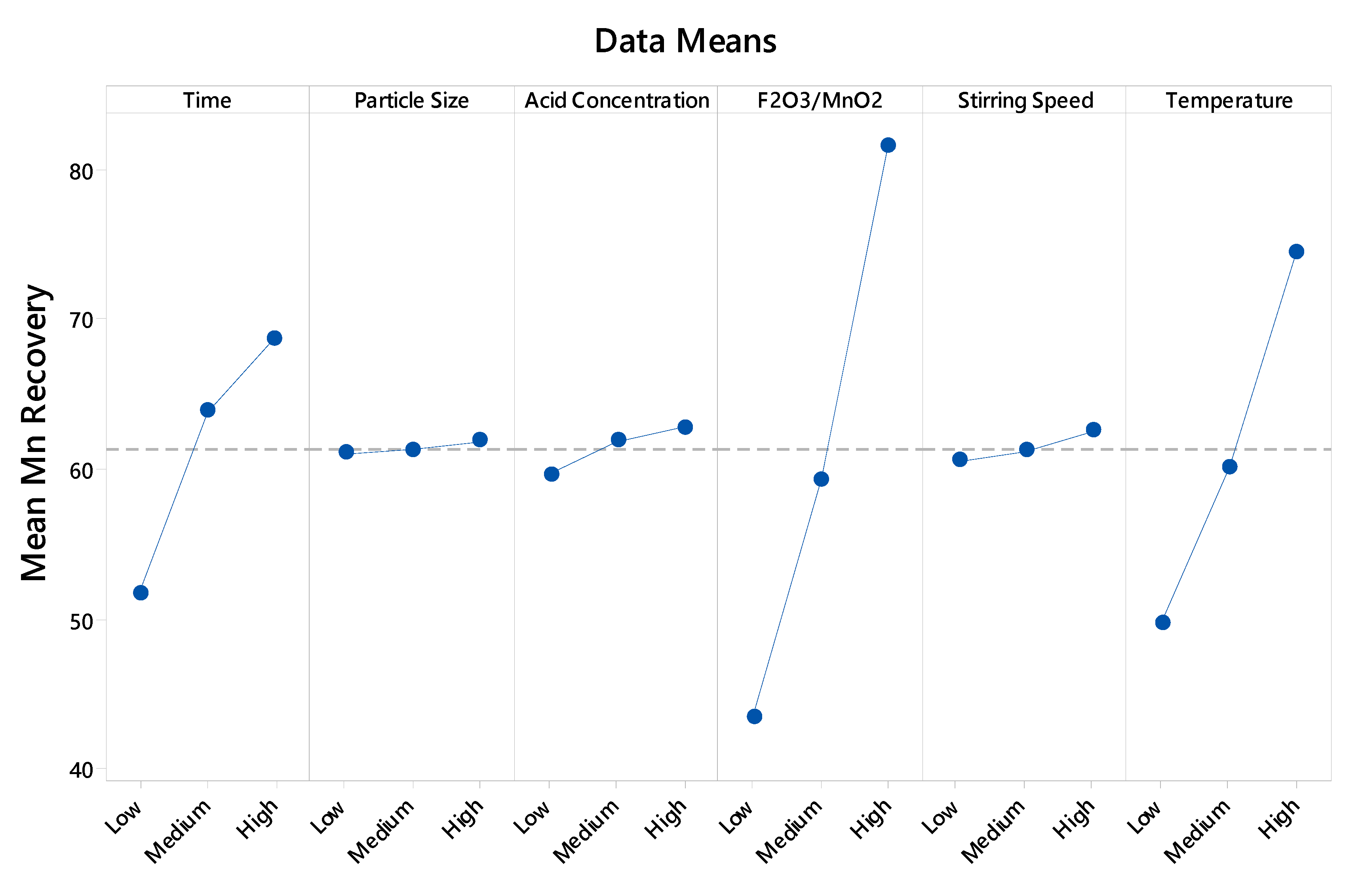
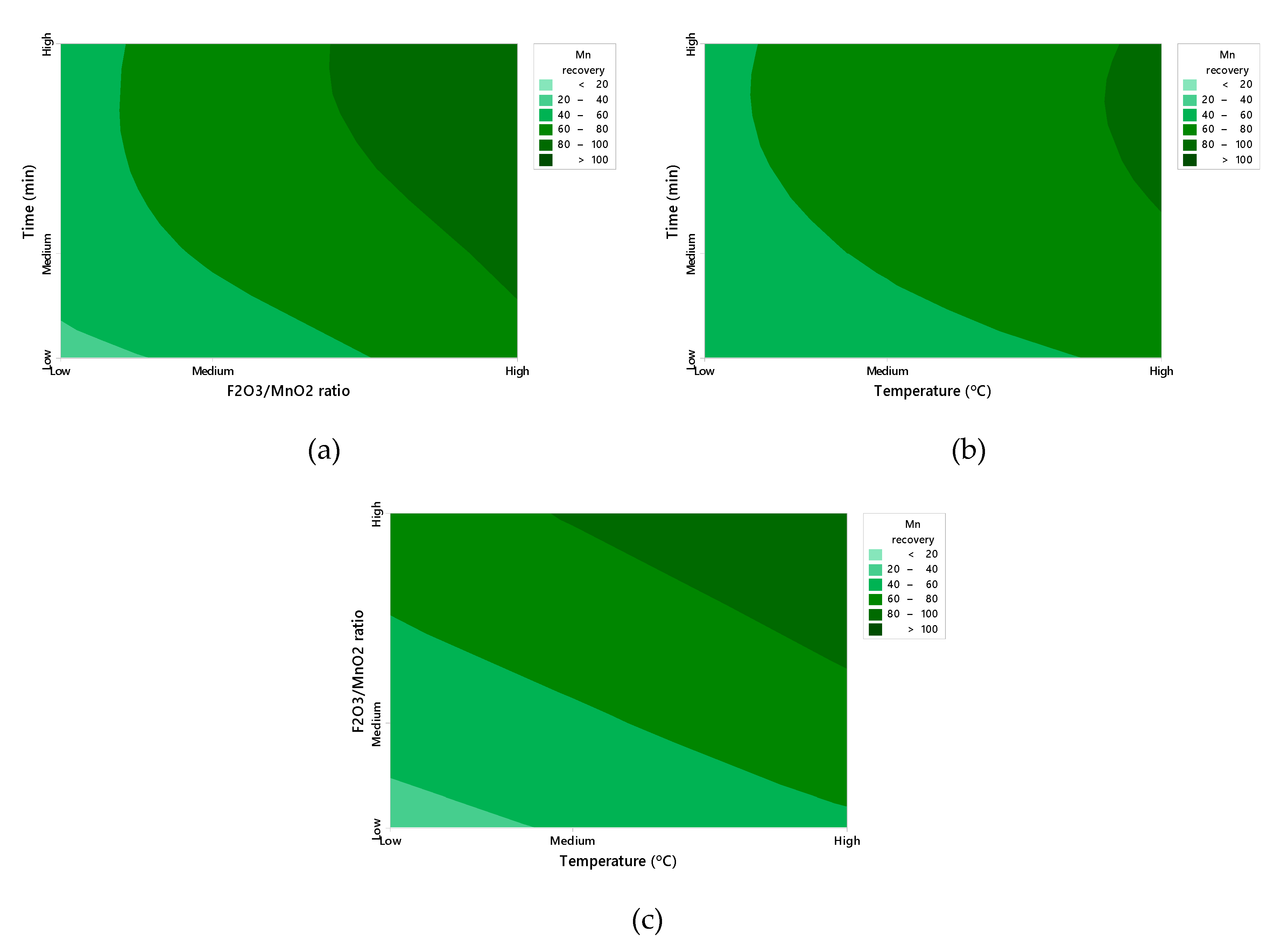
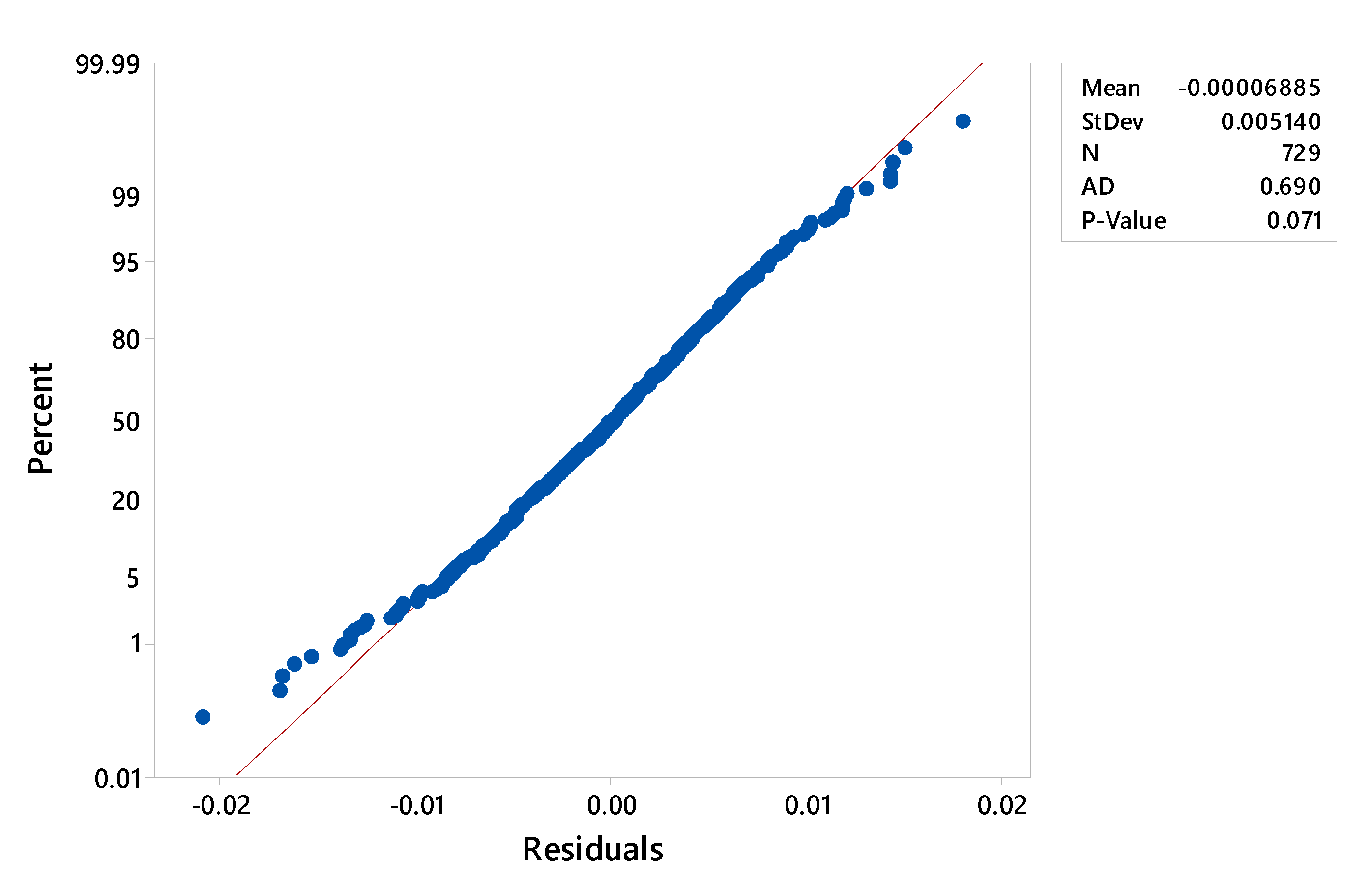
3. Results and Discussion
3.1. Multilinear Regression of Experimental Data
3.2. Fitting of the Exponential Function
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marino, E.; González, F.J.; Somoza, L.; Lunar, R.; Ortega, L.; Vázquez, J.T.; Reyes, J.; Bellido, E. Strategic and rare elements in Cretaceous-Cenozoic cobalt-rich ferromanganese crusts from seamounts in the Canary Island Seamount Province (northeastern tropical Atlantic). Ore Geol. Rev. 2017, 87, 41–61. [Google Scholar] [CrossRef]
- Nishi, K.; Usui, A.; Nakasato, Y.; Yasuda, H. Formation age of the dual structure and environmental change recorded in hydrogenetic ferromanganese crusts from Northwest and Central Paci fi c seamounts. Ore Geol. Rev. 2017, 87, 62–70. [Google Scholar] [CrossRef]
- Lenoble, J.P. Polymetallic Nodules; International Seabed Authority: Kingston, CA, USA, 2000. [Google Scholar]
- Toro, N.; Herrera, N.; Castillo, J.; Torres, C.; Sepúlveda, R. Initial Investigation into the Leaching of Manganese from Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Foundry Slag—Part I. Minerals 2018, 8, 565. [Google Scholar] [CrossRef]
- Premchand, P.; Jana, R.K. Processing of Polymetallic Sea Nodules: An Overview. In Proceedings of the Ocean Mining Symposium, Goa, India, 8–10 November 1999; p. 3. [Google Scholar]
- Hein, J.R.; Cherkashov, G.A. Preface for Ore Geology Reviews Special Issue: Marine mineral deposits: New resources for base, precious, and critical metals. Ore Geol. Rev. 2017, 100, 1–2. [Google Scholar] [CrossRef]
- Konstantinova, N.; Cherkashov, G.; Hein, J.R.; Mirão, J.; Dias, L.; Madureira, P.; Kuznetsov, V.; Maksimov, F. Composition and characteristics of the ferromanganese crusts from the western Arctic Ocean. Ore Geol. Rev. 2017, 87, 88–99. [Google Scholar] [CrossRef]
- Sharma, R. Environmental Issues of Deep-Sea Mining. Procedia Earth Planet. Sci. 2015, 11, 204–211. [Google Scholar] [CrossRef]
- Senanayake, G. Acid leaching of metals from deep-sea manganese nodules—A critical review of fundamentals and applications. Miner. Eng. 2011, 24, 1379–1396. [Google Scholar] [CrossRef]
- Usui, A.; Nishi, K.; Sato, H.; Nakasato, Y.; Thornton, B.; Kashiwabara, T. Continuous growth of hydrogenetic ferromanganese crusts since 17 Myr ago on Takuyo-Daigo Seamount, NW Pacific, at water depths of 800–5500 m. Ore Geol. Rev. 2017, 87, 71–87. [Google Scholar] [CrossRef]
- Randhawa, N.S.; Hait, J.; Jana, R.K. A brief overview on manganese nodules processing signifying the detail in the Indian context highlighting the international scenario. Hydrometallurgy 2016, 165, 166–181. [Google Scholar] [CrossRef]
- Kanungo, S.B. Rate process of the reduction leaching of manganese nodules in dilute HCl in presence of pyrite. Part I. Dissolution behaviour of iron and sulphur species during leaching. Hydrometallurgy 1999, 52, 313–330. [Google Scholar] [CrossRef]
- Kanungo, S.B. Rate process of the reduction leaching of manganese nodules in dilute HCl in presence of pyrite. Part II: Leaching behavior of manganese Hydrometallurgy 1999, 52, 331–347. [Google Scholar]
- Zakeri, A.; Bafghi, M.S.; Shahriari, S.; Das, S.C.; Sahoo, P.K.; Rao, P.K. Dissolution kinetics of manganese dioxide ore in sulfuric acid in the presence of ferrous ion. Hydrometallurgy 2007, 8, 22–27. [Google Scholar]
- Bafghi, M.S.; Zakeri, A.; Ghasemi, Z.; Adeli, M. Reductive dissolution of manganese ore in sulfuric acid in the presence of iron metal. Hydrometallurgy 2008, 90, 207–212. [Google Scholar] [CrossRef]
- Toro, N.; Saldaña, M.; Castillo, J.; Higuera, F.; Acosta, R. Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings. Minerals 2019, 9, 289. [Google Scholar] [CrossRef]
- Toro, N.; Saldaña, M.; Gálvez, E.; Cánovas, M.; Trigueros, E.; Castillo, J.; Hernández, P.C. Optimization of Parameters for the Dissolution of Mn from Manganese Nodules with the Use of Tailings in An Acid Medium. Minerals 2019, 9, 387. [Google Scholar] [CrossRef]
- Mellado, M.; Cisternas, L.; Lucay, F.; Gálvez, E.; Sepúlveda, F. A Posteriori Analysis of Analytical Models for Heap Leaching Using Uncertainty and Global Sensitivity Analyses. Minerals 2018, 8, 44. [Google Scholar] [CrossRef]
- Mellado, M.E.; Cisternas, L.A.; Gálvez, E.D. An analytical model approach to heap leaching. Hydrometallurgy 2009, 95, 33–38. [Google Scholar] [CrossRef]
- Mellado, M.E.; Casanova, M.P.; Cisternas, L.A.; Gálvez, E.D. On scalable analytical models for heap leaching. Comput. Chem. Eng. 2011, 35, 220–225. [Google Scholar] [CrossRef]
- Mellado, M.E.; Gálvez, E.D.; Cisternas, L.A. On the optimization of flow rates on copper heap leaching operations. Int. J. Miner. Process. 2011, 101, 75–80. [Google Scholar] [CrossRef]
- Moghaddam, M.Y.; Tonkaboni, S.Z.S.; Noaparast, M.; Ardejani, F.D. A mathematical model to simulate Heap (bio)-leaching process: An exact conceptual model, Homotopy theory and comparative insights with conventional methods. Int. J. Model. Simul Sci. Comput. 2017, 8, 1750018. [Google Scholar] [CrossRef]
- Hernández, I.F.; Ordóñez, J.I.; Robles, P.A.; Gálvez, E.D.; Cisternas, L.A. A Methodology for Design and Operation of Heap Leaching Systems. Miner. Process. Extr. Metall. Rev. 2017, 38, 180–192. [Google Scholar] [CrossRef]
- Fekete, V.; Deconinck, E.; Bolle, F.; van Loco, J. Modelling aluminium leaching into food from different foodware materials with multi-level factorial design of experiments. Food Addit. Contam. Part A Chem. Anal. Control. Exp. Risk Assess. 2012, 29, 1322–1333. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Wu, A.X.; Yin, S.H.; Chen, X. Nonlinear chaotic characteristic in leaching process and prediction of leaching cycle period. J. Cent. South Univ. 2016, 23, 2935–2940. [Google Scholar] [CrossRef]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Çetintaş, S.; Bingöl, D. Response surface methodology approach to leaching of nickel laterite and evaluation of different analytical techniques used for the analysis of leached solutions. Anal. Methods 2016, 8, 3075–3087. [Google Scholar] [CrossRef]
- Botane, P.; Brochot, S.; D’Hugues, P.; Spolaore, P. Material size distribution in concurrent bio-leaching and precipitation: Experimental procedure and modelling. Hydrometallurgy 2013, 133, 7–14. [Google Scholar] [CrossRef]
- Mellado, M.E.; Gálvez, E.D.; Cisternas, L.A. Stochastic analysis of heap leaching process via analytical models. Miner. Eng. 2012, 33, 93–98. [Google Scholar] [CrossRef]
- Komnitsas, K.; Manousaki, K.; Zaharaki, D. Assessment of reactivity of sulphidic tailings and river sludges. Geochem. Explor. Environ. Anal. 2009, 9, 313–318. [Google Scholar] [CrossRef]
- Dixon, D.G.; Hendrix, J.L. A general model for leaching of one or more solid reactants from porous ore particles. Metall. Trans. B 1993, 24, 157–169. [Google Scholar] [CrossRef]
- Schlesinger, M.; King, M.; Sole, K.; Davenport, W. Extractive Metallurgy of Copper, 5th ed.; Elsevier: Amsterdam, The Netherlands, 2011; ISBN 978-0-08-096789-9. [Google Scholar]
- Dixon, D.G.; Hendrix, J.L. A mathematical model for heap leaching of one or more solid reactants from porous ore pellets. Metall. Trans. B 1993, 24, 1087–1102. [Google Scholar] [CrossRef]
- Free, M. Hidrometallurgy: Fundamentals and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2013; ISBN 978-1-118-23077-0. [Google Scholar]
- Devore, J. Probability & Statistics for Engineering and the Sciences, 8th ed.; Cengage Learning: Boston, MA, USA, 2010; ISBN 0-538-73352-7. [Google Scholar]
- R Core Team. R: The R Project for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Minitab, LLC. Getting Started with Minitab 18; Minitab Inc.: State College, PA, USA, 2017; p. 73. [Google Scholar]
- Saldaña, M.; Toro, N.; Castillo, J.; Hernández, P.; Navarra, A. Optimization of the Heap Leaching Process through Changes in Modes of Operation and Discrete Event Simulation. Minerals 2019, 9, 421. [Google Scholar] [CrossRef]
- Riquelme, R.; Tapia, M.; Campos, E.; Mpodozis, C.; Carretier, S.; González, R.; Muñoz, S.; Fernández-Mort, A.; Sanchez, C.; Marquardt, C. Supergene and exotic Cu mineralization occur during periods of landscape stability in the Centinela Mining District, Atacama Desert. Basin Res. 2018, 30, 395–425. [Google Scholar] [CrossRef]
- Benavente, O.; Hernández, M.C.; Melo, E.; Núñez, D.; Quezada, V.; Zepeda, Y. Copper Dissolution from Black Copper Ore under Oxidizing and Reducing Conditions. Metals 2019, 9, 799. [Google Scholar] [CrossRef]
- Helle, S.; Pincheira, M.; Jerez, O.; Kelm, U. Sequential extraction to predict the leaching potential of refractory. In Proceedings of the Mineral Processing Congress, Sozopol, Bulgaria, 12–16 June 2013; pp. 109–111. [Google Scholar]


| Mineral | Amount% (w/w) |
|---|---|
| Chalcopyrite/Bornite CuFeS2/Cu5FeS4 | 0.47 |
| Tennantite/Tetrahedrite (Cu12As4S13/Cu12Sb4S13) | 0.03 |
| Other Cu Minerals | 0.63 |
| Cu–Fe Hydroxides | 0.94 |
| Pyrite (FeS2) | 0.12 |
| Magnetite (Fe3O4) | 58.52 |
| Specular Hematite (Fe2O3) | 0.89 |
| Hematite (Fe2O3) | 4.47 |
| Ilmenite/Titanite/Rutile (FeTiO3/CaTiSiO3/TiO2) | 0.04 |
| Siderite (FeCO3) | 0.22 |
| Chlorite/Biotite (Mg3Si4O10(OH)2(Mg)3(OH)6/K(Mg)3AlSi3O10(OH)2) | 3.13 |
| Other Phyllosilicates | 11.61 |
| Fayalite (Fe2SiO4) | 4.59 |
| Dicalcium Silicate (Ca2SiO4 | 8.30 |
| Kirschsteinite (CaFeSiO4) | 3.40 |
| Forsterita (Mg2SiO4) | 2.30 |
| Baritine (BaSO4) | 0.08 |
| Zinc Oxide (ZnO) | 0.02 |
| Lead Oxide (PbO) | 0.01 |
| Sulfate (SO4) | 0.20 |
| Others | 0.03 |
| Total | 100.00 |
| Parameter/Value | Low | Medium | High |
|---|---|---|---|
| Time (min) | 5 | 10 | 20 |
| Particle Size (µm) | −150 + 106 | −75 + 53 | −47 + 38 |
| Sulfuric Acid (H2SO4) | 0.1 | 0.3 | 0.5 |
| Fe2O3/MnO2 ratio | 1/2 | 1/1 | 2/1 |
| Stirring Speed (rpm) | 600 | 700 | 800 |
| Temperature (°C) | 25 | 35 | 50 |
| Model/Statistic | MAD | MSE | MAPE |
|---|---|---|---|
| R(t) | 6.19 × 10−5 | 3.57 × 10−7 | 3.88 × 10−4 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saldaña, M.; Toro, N.; Castillo, J.; Hernández, P.; Trigueros, E.; Navarra, A. Development of an Analytical Model for the Extraction of Manganese from Marine Nodules. Metals 2019, 9, 903. https://doi.org/10.3390/met9080903
Saldaña M, Toro N, Castillo J, Hernández P, Trigueros E, Navarra A. Development of an Analytical Model for the Extraction of Manganese from Marine Nodules. Metals. 2019; 9(8):903. https://doi.org/10.3390/met9080903
Chicago/Turabian StyleSaldaña, Manuel, Norman Toro, Jonathan Castillo, Pía Hernández, Emilio Trigueros, and Alessandro Navarra. 2019. "Development of an Analytical Model for the Extraction of Manganese from Marine Nodules" Metals 9, no. 8: 903. https://doi.org/10.3390/met9080903
APA StyleSaldaña, M., Toro, N., Castillo, J., Hernández, P., Trigueros, E., & Navarra, A. (2019). Development of an Analytical Model for the Extraction of Manganese from Marine Nodules. Metals, 9(8), 903. https://doi.org/10.3390/met9080903








