Modified Leach Residues from Processing Deep-Sea Nodules as Effective Heavy Metals Adsorbents
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of Adsorbents
2.2. Sorbents Characterization
2.3. Determination of Zero Charge Point (pHpzc)
2.4. Adsorption Tests
3. Results and Discussion
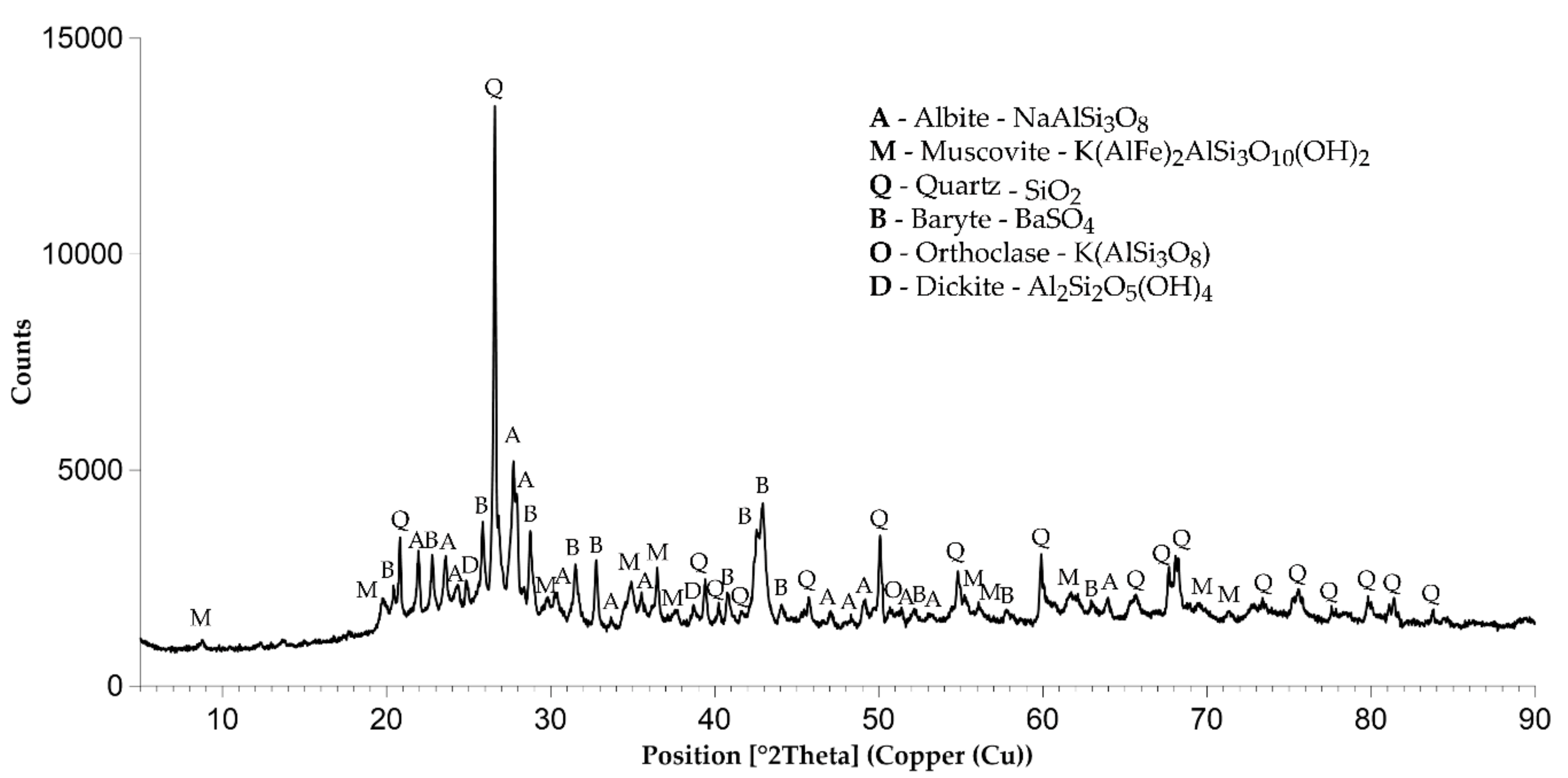
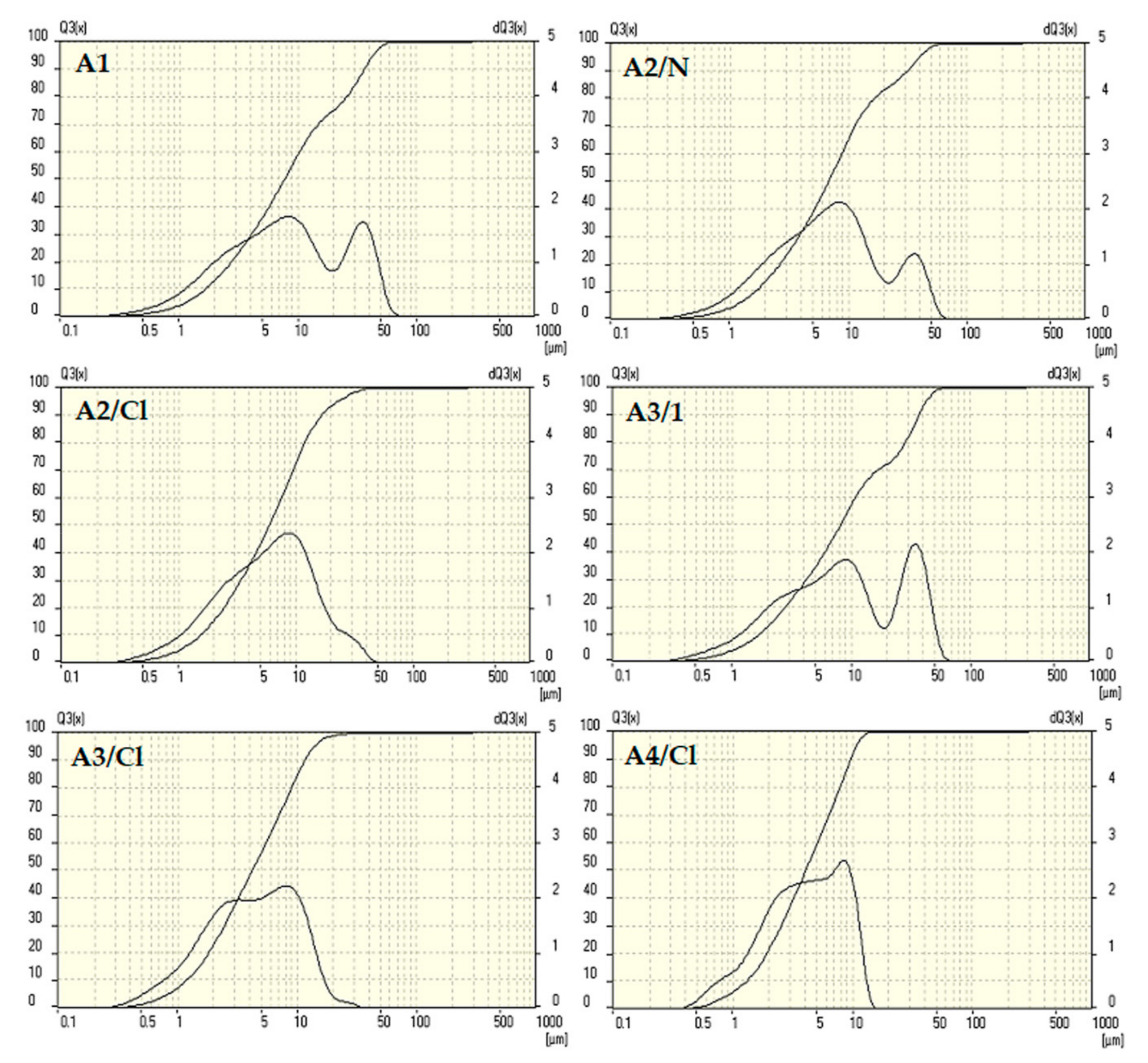
3.1. Characteristics of Prepared Sorbents
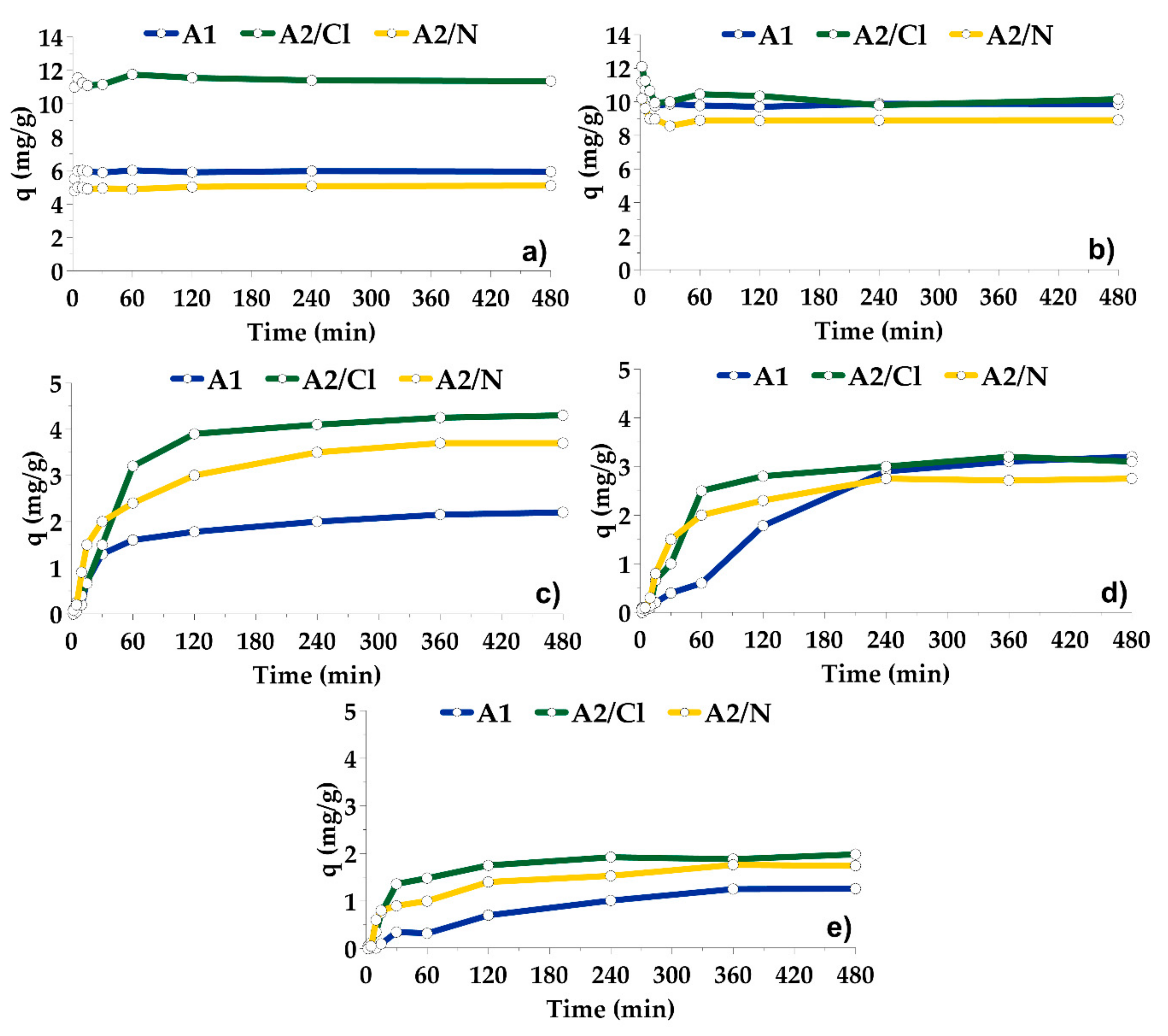
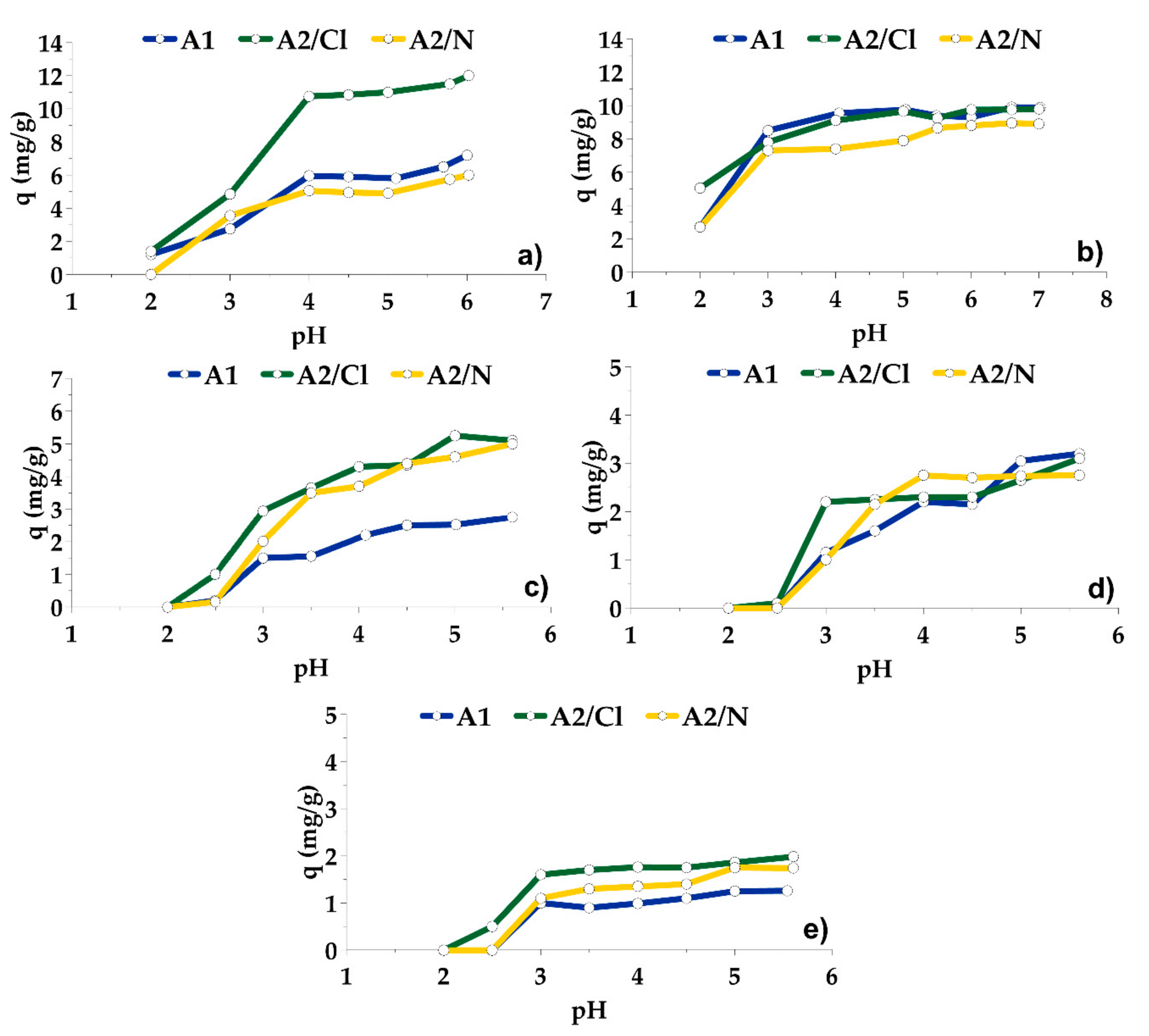
3.2. Adsorption on A1, A2/N and A2/Cl
3.3. Adsorption on A3/1, A3/Cl and A4/Cl
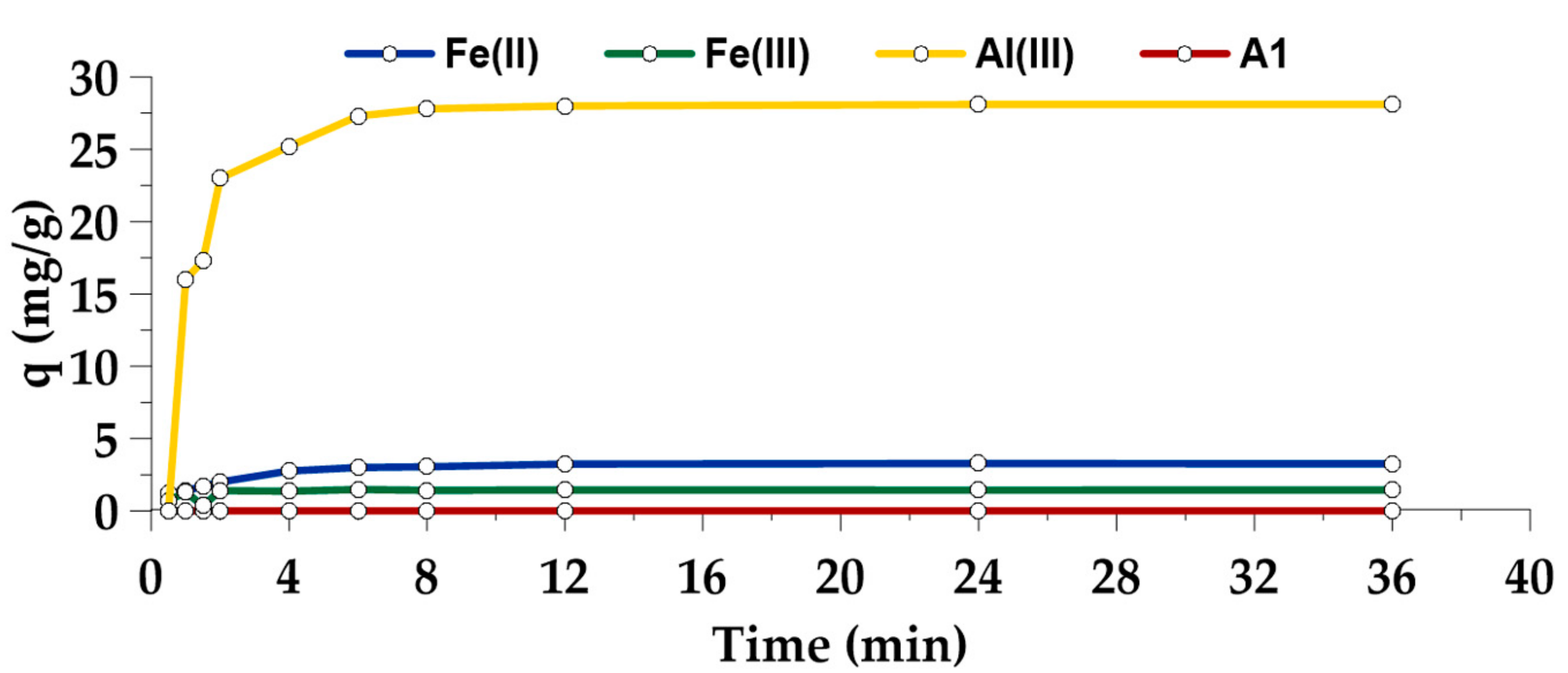
3.4. As(V) adsorption on A1, A5/FeII, A5/FeIII and A5/AlIII
4. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Abramovski, T.; Stefanova, V.P.; Causse, R.; Romanchuk, A. Technologies for the Processing of Polymetallic Nodules from Clarion Clipperton Zone in the Pacific Ocean. J. Chem. Technol. Metall. 2017, 52, 258–269. [Google Scholar]
- Vu, H.; Jandová, J.; Lisá, K.; Vranka, F. Leaching of manganese deep ocean nodules in FeSO4–H2SO4–H2O solutions. Hydrometallurgy 2005, 77, 147–153. [Google Scholar] [CrossRef]
- Das, R.P.; Anand, S. Metallurgical Processing of Polymetallic Ocean Nodules. In Deep-Sea Mining: Resource Potential, Technical and Environmental Considerations; Sharma, R., Ed.; Springer International Publishing: Cham, Switzerland, 2017; pp. 365–394. [Google Scholar] [CrossRef]
- Sahoo, R.N.; Das, S.C.; Reddy, B.R.; Rath, P.C.; Das, R.P. Adsorption of copper on manganese nodule residue obtained from NH3–SO2 leaching. Hydrometallurgy 2001, 62, 185–192. [Google Scholar] [CrossRef]
- Agrawal, A.; Sahu, K.K. Kinetic and isotherm studies of cadmium adsorption on manganese nodule residue. J. Hazard. Mater. 2006, 137, 915–924. [Google Scholar] [CrossRef] [PubMed]
- Randhawa, N.S.; Jana, R.K. Efficient removal of aqueous lead by leached sea nodules residue. Desalin. Water Treat. 2015, 56, 2937–2947. [Google Scholar] [CrossRef]
- El-Ashtoukhy, E.S.Z.; Abdel-Aziz, M.H. Removal of copper from aqueous solutions by cementation in a bubble column reactor fitted with horizontal screens. Int. J. Miner. Process. 2013, 121, 65–69. [Google Scholar] [CrossRef]
- Li, J.-R.; Wang, X.; Yuan, B.; Fu, M.-L. Layered chalcogenide for Cu2+ removal by ion-exchange from wastewater. J. Mol. Liq. 2014, 200, 205–212. [Google Scholar] [CrossRef]
- Abid, M.F.; Zablouk, M.A.; Abid-Alameer, A.M. Experimental study of dye removal from industrial wastewater by membrane technologies of reverse osmosis and nanofiltration. Iran. J. Environ. Health Sci. Eng. 2012, 9, 17. [Google Scholar] [CrossRef]
- Tran, N.; Drogui, P.; Blais, J.-F.; Mercier, G. Phosphorus removal from spiked municipal wastewater using either electrochemical coagulation or chemical coagulation as tertiary treatment. Sep. Purif. Technol. 2012, 95, 16–25. [Google Scholar] [CrossRef]
- Khosa, M.A.; Shah, S.S.; Feng, X. Metal sericin complexation and ultrafiltration of heavy metals from aqueous solution. Chem. Eng. J. 2014, 244, 446–456. [Google Scholar] [CrossRef]
- Rahimi, M.; Schoener, Z.; Zhu, X.; Zhang, F.; Gorski, C.A.; Logan, B.E. Removal of copper from water using a thermally regenerative electrodeposition battery. J. Hazard. Mater. 2017, 322, 551–556. [Google Scholar] [CrossRef]
- Kul, M.; Oskay, K.O. Separation and recovery of valuable metals from real mix electroplating wastewater by solvent extraction. Hydrometallurgy 2015, 155, 153–160. [Google Scholar] [CrossRef]
- Su, Y.-N.; Lin, W.-S.; Hou, C.-H.; Den, W. Performance of integrated membrane filtration and electrodialysis processes for copper recovery from wafer polishing wastewater. J. Water Process Eng. 2014, 4, 149–158. [Google Scholar] [CrossRef]
- Dave, P.N.; Subrahmanyam, N.; Sharma, S. Kinetics and Thermodynamics of Copper Ions Removal from Aqueous Solution by Use of Activated Charcoal. Indian J. Chem. Techn. 2009, 16, 234–239. [Google Scholar]
- Parida, K.M.; Mallick, S.; Mohapatra, B.K.; Misra, V.N. Studies on manganese-nodule leached residues: 1. Physicochemical characterization and its adsorption behavior toward Ni2+ in aqueous system. J. Colloid Interface Sci. 2004, 277, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Mallick, S.; Dash, S.S.; Parida, K.M. Adsorption of hexavalent chromium on manganese nodule leached residue obtained from NH3–SO2 leaching. J. Colloid Interface Sci. 2006, 297, 419–425. [Google Scholar] [CrossRef]
- Das, N.; Jana, R.K. Adsorption of some bivalent heavy metal ions from aqueous solutions by manganese nodule leached residues. J. Colloid Interface Sci. 2006, 293, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Randhawa, N.S.; Dwivedi, D.; Prajapati, S.; Jana, R.K. Application of manganese nodules leaching residue for adsorption of nickel(II) ions from aqueous solution. Int. J. Environ. Sci. Technol. 2015, 12, 857–864. [Google Scholar] [CrossRef][Green Version]
- Randhawa, N.S.; Das, N.N.; Jana, R.K. Selenite adsorption using leached residues generated by reduction roasting–ammonia leaching of manganese nodules. J. Hazard. Mater. 2012, 241, 486–492. [Google Scholar] [CrossRef]
- Dash, S.S.; Parida, K.M. Studies on selenite adsorption using manganese nodule leached residues. J. Colloid Interface Sci. 2007, 307, 333–339. [Google Scholar] [CrossRef]
- Parida, K.M.; Pradhan, A.C. Removal of phenolic compounds from aqueous solutions by adsorption onto manganese nodule leached residue. J. Hazard. Mater. 2010, 173, 758–764. [Google Scholar] [CrossRef] [PubMed]
- Parida, K.M.; Mallick, S.; Dash, S.S. Studies on manganese nodule leached residues: 2. Adsorption of aqueous phosphate on manganese nodule leached residues. J. Colloid Interface Sci. 2005, 290, 22–27. [Google Scholar] [CrossRef]
- Behera, R.K.; Satapathy, P.K.; Randhawa, N.S.; Das, N.N. Adsorptive Removal of Phosphate Ions Using Leached Sea Nodule Residue Generated by the Reduction Roasting Ammoniacal Leaching Process. Adsorpt. Sci. Technol. 2010, 28, 611–627. [Google Scholar] [CrossRef]
- Parida, K.M.; Sahu, S.; Reddy, K.H.; Sahoo, P.C. A Kinetic, Thermodynamic, and Mechanistic Approach toward Adsorption of Methylene Blue over Water-Washed Manganese Nodule Leached Residues. Ind. Eng. Chem. Res. 2011, 50, 843–848. [Google Scholar] [CrossRef]
- Stahl, R.S.; James, B.R. Zinc sorption by manganese-oxide-coated sand as a function of pH. Soil Sci. Soc. Am. J. 1991, 55, 1291–1294. [Google Scholar] [CrossRef]
- Lo, S.-L.; Jeng, H.-T.; Lai, C.-H. Characteristics and adsorption properties of iron-coated sand. Water Sci. Technol. 1997, 35, 63–70. [Google Scholar] [CrossRef]
- Burakov, A.E.; Galunin, E.V.; Burakova, I.V.; Kucherova, A.E.; Agarwal, S.; Tkachev, A.G.; Gupta, V.K. Adsorption of heavy metals on conventional and nanostructured materials for wastewater treatment purposes: A review. Ecotoxicol. Environ. Saf. 2018, 148, 702–712. [Google Scholar] [CrossRef]
- Singh, M.; Thanh, D.N.; Ulbrich, P.; Strnadová, N.; Štěpánek, F. Synthesis, characterization and study of arsenate adsorption from aqueous solution by α- and δ-phase manganese dioxide nanoadsorbents. J. Solid State Chem. 2010, 183, 2979–2986. [Google Scholar] [CrossRef]
- Gong, J.-L.; Wang, X.-Y.; Zeng, G.-M.; Chen, L.; Deng, J.-H.; Zhang, X.-R.; Niu, Q.-Y. Copper (II) removal by pectin–iron oxide magnetic nanocomposite adsorbent. Chem. Eng. J. 2012, 185–186, 100–107. [Google Scholar] [CrossRef]
- Dil, E.A.; Ghaedi, M.; Asfaram, A. The performance of nanorods material as adsorbent for removal of azo dyes and heavy metal ions: Application of ultrasound wave, optimization and modeling. Ultrason. Sonochem. 2017, 34, 792–802. [Google Scholar] [CrossRef]
- Liu, S.; Kang, S.; Wang, G.; Zhao, H.; Cai, W. Micro/nanostructured porous Fe–Ni binary oxide and its enhanced arsenic adsorption performances. J. Colloid Interface Sci. 2015, 458, 94–102. [Google Scholar] [CrossRef]
- Ma, M.-D.; Wu, H.; Deng, Z.-Y.; Zhao, X. Arsenic removal from water by nanometer iron oxide coated single-wall carbon nanotubes. J. Mol. Liq. 2018, 259, 369–375. [Google Scholar] [CrossRef]
- Chen, R.; Zhi, C.; Yang, H.; Bando, Y.; Zhang, Z.; Sugiur, N.; Golberg, D. Arsenic (V) adsorption on Fe3O4 nanoparticle-coated boron nitride nanotubes. J. Colloid Interface Sci. 2011, 359, 261–268. [Google Scholar] [CrossRef]
- Feng, L.; Cao, M.; Ma, X.; Zhu, Y.; Hu, C. Superparamagnetic high-surface-area Fe3O4 nanoparticles as adsorbents for arsenic removal. J. Hazard. Mater. 2012, 217, 439–446. [Google Scholar] [CrossRef]
- Hernández-Flores, H.; Pariona, N.; Herrera-Trejo, M.; Hdz-García, H.M.; Mtz-Enriquez, A.I. Concrete/maghemite nanocomposites as novel adsorbents for arsenic removal. J. Mol. Struct. 2018, 1171, 9–16. [Google Scholar] [CrossRef]


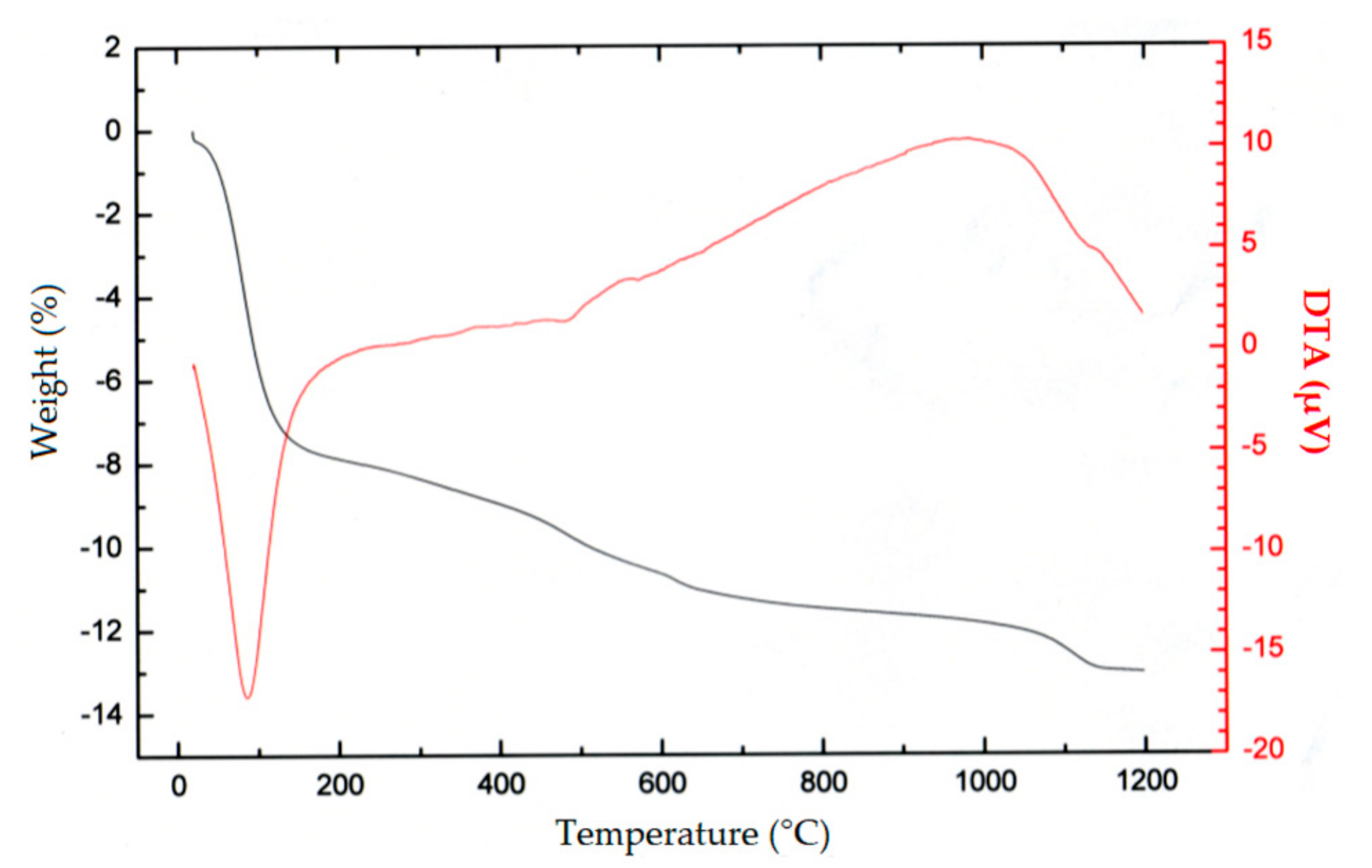
| Element | Mn | Fe | Si | Al | Mg | Ca | Na | Cu | Ni | Ti | Zn | Co |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| wt. % | 30.57 | 4.41 | 3.53 | 2.16 | 1.87 | 1.84 | 1.64 | 1.18 | 1.14 | 0.35 | 0.14 | 0.13 |
| Element | Adsorbent | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| A1 | A2/N | A2/Cl | A3/1 | A3/Cl | A4/Cl | A5/FeII | A5/FeIII | A5/AlIII | |
| Si | 38.71 | 41.87 | 40.24 | 40.30 | 40.18 | 41.04 | 39.16 | 39.73 | 27.78 |
| Al | 5.95 | 6.73 | 6.28 | 6.01 | 6.19 | 6.15 | 6.33 | 6.17 | 13.77 |
| Fe | 1.66 | 1.74 | 1.51 | 1.72 | 1.52 | 1.50 | 3.07 | 4.89 | 1.18 |
| K | 2.39 | 2.48 | 2.36 | 2.50 | 2.41 | 2.34 | 2.37 | 2.48 | 3.21 |
| Ca | 1.42 | 1.34 | 1.29 | 1.35 | 1.28 | 1.28 | 1.40 | 1.39 | 1.14 |
| Na | 0.89 | 1.06 | 0.87 | 0.91 | 0.88 | 0.88 | 1.00 | 1.01 | 0.67 |
| Ba | 3.60 | 3.62 | 4.12 | 4.01 | 4.14 | 3.50 | 3.95 | 3.23 | 2.06 |
| Mg | 0.41 | 0.42 | 0.53 | 0.42 | 0.53 | 0.50 | 0.45 | 0.45 | 0.23 |
| S | 1.10 | 0.92 | 0.62 | 1.14 | 0.62 | 0.54 | 1.43 | 0.89 | 0.68 |
| Sr | 0.29 | 0.15 | 0.14 | 0.30 | 0.14 | 0.15 | 0.27 | 0.18 | 0.09 |
| Ti | 0.67 | 0.63 | 0.52 | 0.69 | 0.53 | 0.52 | 0.64 | 0.66 | 0.46 |
| Pb | 0.24 | 0.14 | 0.16 | 0.29 | 0.15 | 0.15 | 0.25 | 0.14 | 0.15 |
| Cl | 0.03 | 0.03 | 0.03 | 0.04 | 0.03 | 0.03 | 0.03 | 0.02 | 0.79 |
| Phase | SemiQuant % | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| A1 | A2/N | A2/Cl | A3/1 | A3/Cl | A4/Cl | A5/FeII | A5/FeIII | A5/AlIII | |
| Albite | 31 | 28 | 46 | 40 | 39 | 46 | 53 | 40 | 41 |
| Muscovite | 29 | 28 | 21 | 36 | 37 | 21 | 26 | 36 | 36 |
| Quartz | 19 | 19 | 27 | 19 | 18 | 27 | 16 | 20 | 19 |
| Barite | 5 | 6 | 6 | 5 | 6 | 6 | 5 | 4 | 4 |
| Orthoclase | 11 | 13 | - | - | - | - | - | - | - |
| Dickite | 5 | 6 | - | - | - | - | - | - | - |
| Adsorbent | A1 | A2/N | A2/Cl | A3/1 | A3/Cl | A4/Cl | A5/FeII | A5/FeIII | A5/AlIII |
|---|---|---|---|---|---|---|---|---|---|
| Specific surface | 234 | 227 | 229 | 220 | 211 | 216 | 274 | 253 | 298 |
| Adsorbent | pHpzc |
|---|---|
| A1 | 3.12 |
| A2/N | 3.05 |
| A2/Cl | 2.96 |
| A3/1 | 3.10 |
| A3/Cl | 3.01 |
| A4/Cl | 2.97 |
| Adsorbent | q (mg/g) | ||||
|---|---|---|---|---|---|
| Pb | Cd | Cu | Ni | Co | |
| A1 | 5.0 pH 5.0 | 9.8 pH 5.0 | 2.5 pH 5.0 | 2.2 pH 4.0 | 1.1 pH 4.5 |
| A3/1 | 11.0 pH 5.0 | 10.8 pH 5.0 | 3.0 pH 5.0 | 2.5 pH 4 | 0.9 pH 4.5 |
| A3/Cl | 7.7 pH 5.0 | 9.9 pH 5.0 | 5.0 pH 5.0 | 2.4 pH 4.0 | 1.7 pH 4.5 |
| A4/Cl | 9.4 pH 4.5 | 11.5 pH 4.0 | 5.7 pH 4.5 | 2.7 pH 4.0 | 1.7 pH 4.5 |
| Adsorbent | Initial pH | Adsorption Capacity (mg/g) | Specific Surface/Particle Size | Reference |
|---|---|---|---|---|
| Micro-/nanostructured Fe–Ni binary oxides | 7.0 | 90.1 | 245 m2/g | [32] |
| Iron oxide nanoparticle-coated single-wall carbon nanotubes | 4.0 | 49.7 | 2–3 nm | [33] |
| Fe3O4 nanoparticle-coated boron nitride nanotubes | 6.9 | 32.2 | 20–50 nm | [34] |
| α-MnO2 nanorods | 6.5 | 19.4 | 50–100 nm length, 5 nm wide | [29] |
| Ascorbic acid-coated Fe3O4 nanoparticles | 7.0 | 16.7 | 179 m2/g | [35] |
| Concrete/maghemite nanocomposites | 5.0 | 8.8 | nanopore 20–70 nm | [36] |
| AlIII-coated leaching residue | 7.0 | 28.1 | 298 m2/g | Present work |
| FeII-coated leaching residue | 7.0 | 3.3 | 274 m2/g | |
| FeIII-coated leaching residue | 7.0 | 1.5 | 253 m2/g |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vu, N.H.; Kristianová, E.; Dvořák, P.; Abramowski, T.; Dreiseitl, I.; Adrysheva, A. Modified Leach Residues from Processing Deep-Sea Nodules as Effective Heavy Metals Adsorbents. Metals 2019, 9, 472. https://doi.org/10.3390/met9040472
Vu NH, Kristianová E, Dvořák P, Abramowski T, Dreiseitl I, Adrysheva A. Modified Leach Residues from Processing Deep-Sea Nodules as Effective Heavy Metals Adsorbents. Metals. 2019; 9(4):472. https://doi.org/10.3390/met9040472
Chicago/Turabian StyleVu, Nguyen Hong, Eva Kristianová, Petr Dvořák, Tomasz Abramowski, Ivo Dreiseitl, and Aigerim Adrysheva. 2019. "Modified Leach Residues from Processing Deep-Sea Nodules as Effective Heavy Metals Adsorbents" Metals 9, no. 4: 472. https://doi.org/10.3390/met9040472
APA StyleVu, N. H., Kristianová, E., Dvořák, P., Abramowski, T., Dreiseitl, I., & Adrysheva, A. (2019). Modified Leach Residues from Processing Deep-Sea Nodules as Effective Heavy Metals Adsorbents. Metals, 9(4), 472. https://doi.org/10.3390/met9040472






