Shortcomings of International Standard ISO 9223 for the Classification, Determination, and Estimation of Atmosphere Corrosivities in Subtropical Archipelagic Conditions—The Case of the Canary Islands (Spain)
Abstract
1. Introduction
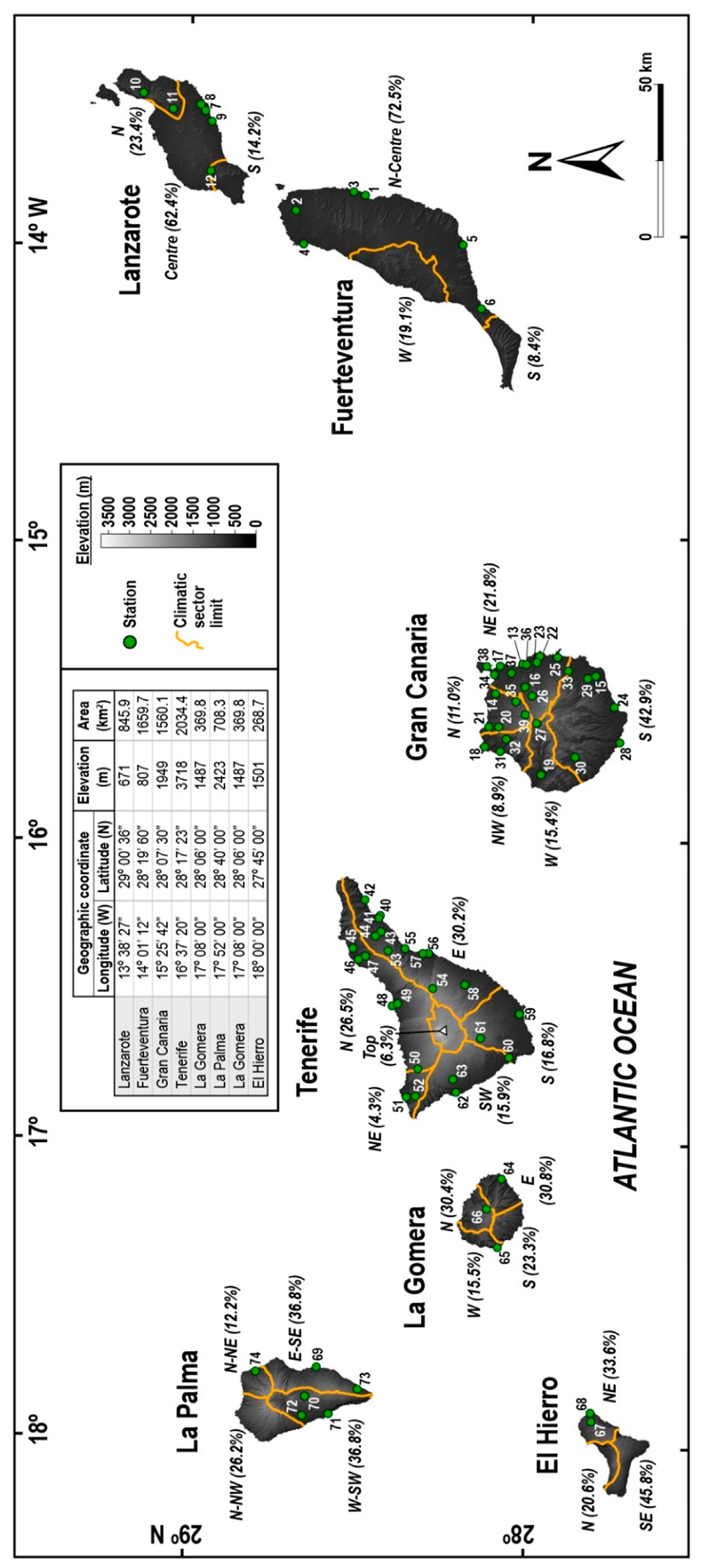
2. Materials and Methods
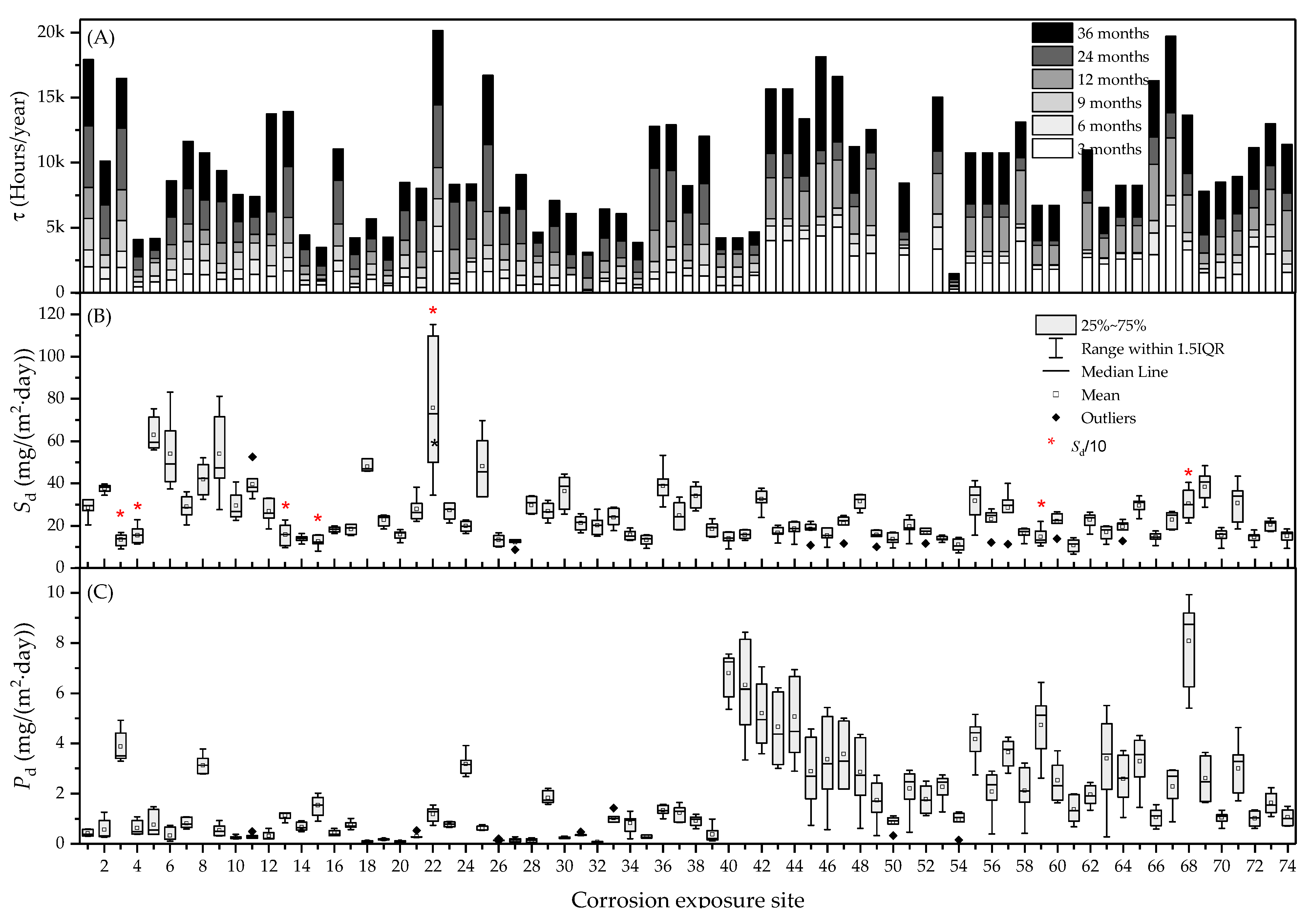
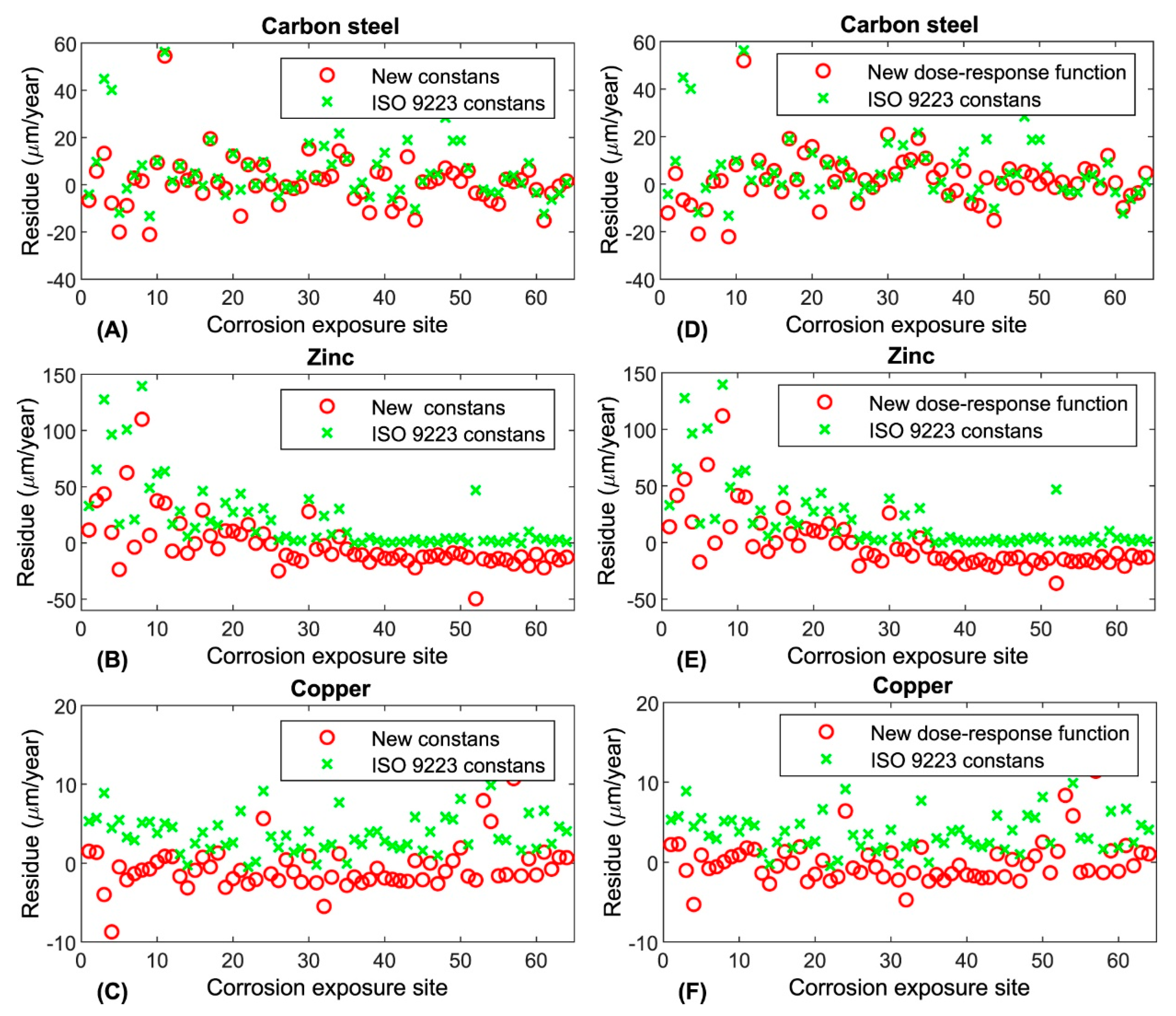
3. Results
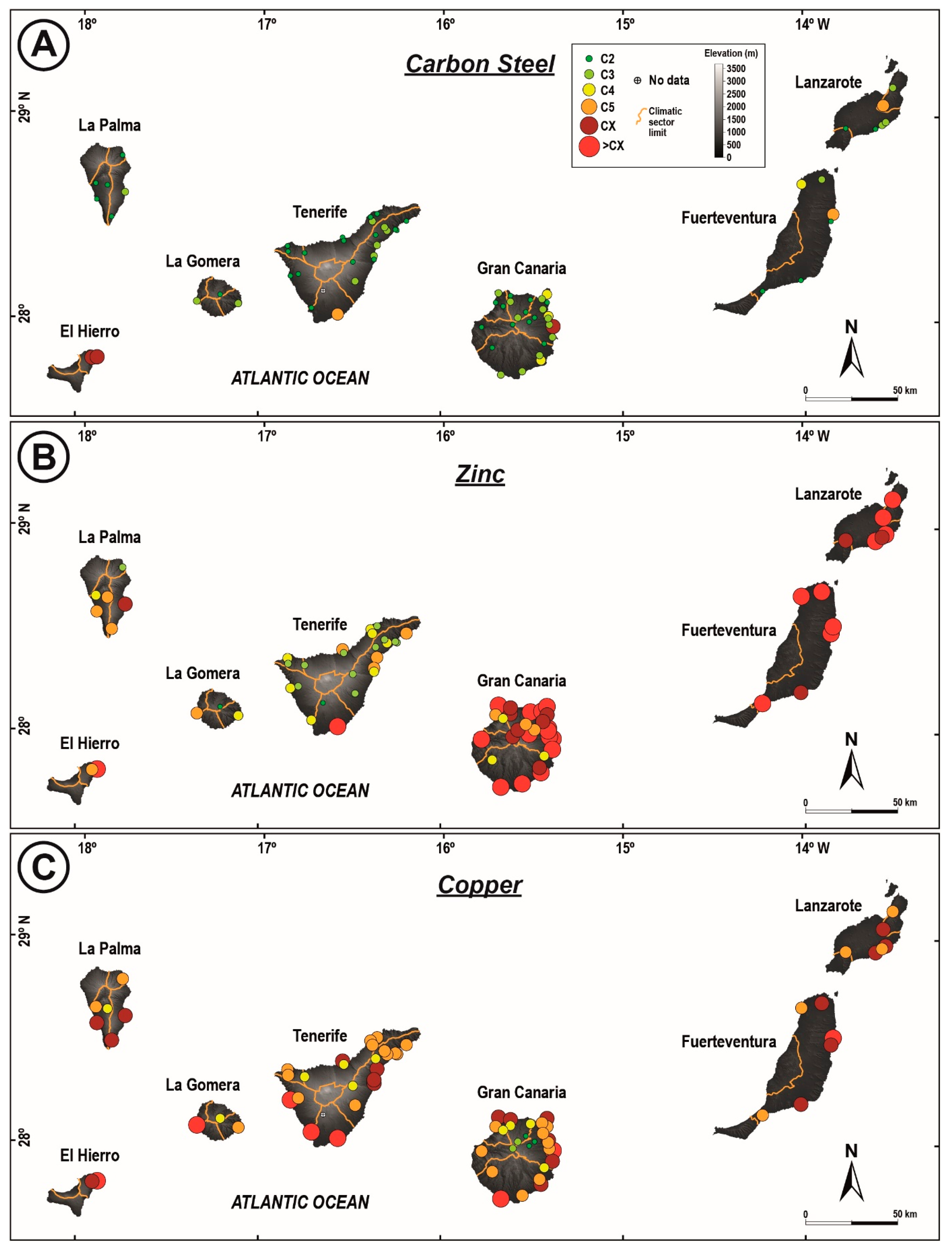
3.1. Classification of Corrosivity of the Atmosphere and General Corrosivity Estimation
3.2. Normative Corrosivity Estimation
4. Discussion
4.1. Corrosivity of the Atmospheres
4.2. ANOVA Analysis
- The corrosion rates, rcorr, showed a strong dependence with Sd for the three metals studied (carbon steel, zinc, and copper), since the p-values were below the level of significance of the study.
- The relative humidity (RH) only had an influence on the corrosion rate of zinc, because this was the only metal with p-value smaller than 5%.
- The deposition of sulfur dioxide (Pd) only showed a weak influence with the corrosion rate of steel, since its p-value slightly exceeded the level of significance 5.8%, being unable to associate such an influence in the cases of zinc and copper.
- Regarding the temperature (T) data, no influence was observed on the corrosion rates for any of the three metal systems.
- The wetting time (τ) exhibited a major effect on the corrosion rate for zinc, whereas its influence was small for steel, and almost negligible for copper.
4.3. Normative Corrosivity Estimation Based on Calculated First-Year Corrosion Losses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Gramberg, U. Korrosionsschutz-Antworten auf eine Herausforderung. Zur Gründung der Gesellschaft für Korrosionsschutz GfKORR. Mater. Corros. 1996, 47, 139–145. [Google Scholar] [CrossRef]
- Dean, S.W.; Hernández-Duque Delgadillo, G.; Bushman, J.B. Marine Corrosion in Tropical Environments, 1st ed.; ASTM Stock Number STP 1399; American Society for Testing Materials: West Conshohocken, PA, USA, 2000; pp. 1–305. [Google Scholar]
- Leygraf, C.; Odnevall Wallinder, I.; Tidblad, J.; Graedel, T. Atmospheric Corrosion, 2nd ed.; The Electrochemical Society, John Wiley & Sons, Inc.: Pennington, NJ, USA, 2016; pp. 1–397. [Google Scholar]
- Lopesino, P.; Alcántara, J.; de la Fuente, D.; Chico, B.; Jiménez, J.A.; Morcillo, M. Corrosion of copper in unpolluted chloride-rich atmospheres. Metals 2018, 8, 866. [Google Scholar] [CrossRef]
- Díaz, I.; Cano, H.; Lopesino, P.; de la Fuente, D.; Chico, B.; Jiménez, J.A.; Medina, S.F.; Morcillo, M. Five-year atmospheric corrosion of Cu, Cr and Ni weathering steels in a wide range of environments. Corros. Sci. 2018, 141, 146–157. [Google Scholar] [CrossRef]
- Díaz, I.; Cano, H.; Crespo, D.; Chico, B.; de la Fuente, D.; Morcillo, M. Atmospheric corrosion of ASTM A-242 and ASTM A-588 weathering steels in different types of atmosphere. Corros. Eng. Sci. Technol. 2018, 53, 449–459. [Google Scholar] [CrossRef]
- LeBozec, N.; Thierry, D.; Persson, D.; Riener, C.K.; Luckeneder, G. Influence of microstructure of zinc-aluminium-magnesium alloy coated Steel on the corrosion behavior in outdoor marine atmosphere. Surf. Coat. Technol. 2019, 374, 897–909. [Google Scholar] [CrossRef]
- Morcillo, M.; Díaz, I.; Cano, H.; Chico, B.; de la Fuente, D. Atmospheric corrosion of weathering steels. Overview for engineers. Part I: Basic concepts. Constr. Build. Mater. 2019, 213, 723–737. [Google Scholar] [CrossRef]
- Morcillo, M.; Díaz, I.; Cano, H.; Chico, B.; de la Fuente, D. Atmospheric corrosion of weathering steels. Overview for engineers. Part II: Testing, inspection, maintenance. Constr. Build. Mater. 2019, 222, 750–765. [Google Scholar] [CrossRef]
- Liu, H.; Cao, F.; Song, G.-L.; Zheng, D.; Shi, Z.; Dargusch, M.S.; Atrens, A. Review of the atmospheric corrosion of magnesium alloys. J. Mater. Sci. Technol. 2019, 35, 2003–2016. [Google Scholar] [CrossRef]
- ISO 9223:2012. Corrosion of Metals and Alloys—Corrosivity of Atmospheres—Clasification, Determination and Estimation, 2nd ed.; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- ISO 9224:2012. Corrosion of Metals and Alloys—Corrosivity of Atmospheres—Guiding Values for the Corrosivity Categories, 2nd ed.; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- ISO 9225:2012. Corrosion of Metals and Alloys—Corrosivity of Atmospheres—Measurement of Environmental Parameters Affecting Corrosivity of Atmospheres, 2nd ed.; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- ISO 9226:2012. Corrosion of Metals and Alloys—Corrosivity of Atmospheres—Determination of Corrosion Rate of Standard Specimens for the Evaluation of Corrosivity, 2nd ed.; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- ISO 9223:1992(E). Corrosion of Metals and Alloys—Corrosivity of Atmospheres—Classification, 1st ed.; International Organization for Standardization: Geneva, Switzerland, 1992. [Google Scholar]
- Ramanauskas, R.; Muleshkova, L.; Maldonado, L.; Dobrovolskis, P. Characterization of the corrosion behaviour of Zn and Zn alloy electrodeposits: Atmospheric and accelerated tests. Corros. Sci. 1998, 40, 401–410. [Google Scholar] [CrossRef]
- Mendoza, A.R.; Corvo, F. Outdoor and indoor atmospheric corrosion of carbon steel. Corros. Sci. 1999, 41, 75–86. [Google Scholar] [CrossRef]
- Santana, J.J.; Santana, J.; González, J.E.; de la Fuente, D.; Chico, B.; Morcillo, M. Atmospheric corrosivity map for steel in Canary Isles. Br. Corros. J. 2001, 36, 266–271. [Google Scholar] [CrossRef]
- Veleva, L.; Kane, R. Atmospheric corrosion. In Corrosion, Fundamentals, Testing and Applications, ASM Handbook Series, 1st ed.; Cramer, S.D., Covino, B.S., Jr., Eds.; American Society for Testing Materials International: Columbus, OH, USA, 2003; Volume 13A, pp. 196–209. [Google Scholar]
- Mikhailov, A.A.; Tidblad, J.; Lucera, V. The classification system of ISO 9223 Standard and the dose-response functions assessing the corrosivity of outdoor atmospheres. Prot. Met. 2004, 40, 541–550. [Google Scholar] [CrossRef]
- Morales, J.; Martín-Krijer, S.; Díaz, F.; Hernández-Borges, J.; González, S. Atmospheric corrosion in subtropical areas: Influences of time of wetness and deficiency of the ISO 9223 norm. Corros. Sci. 2005, 47, 2005–2019. [Google Scholar] [CrossRef]
- Veleva, L.; Acosta, M.; Meraz, E. Atmospheric corrosion of zinc induced by runoff. Corros. Sci. 2009, 51, 2055–2062. [Google Scholar] [CrossRef]
- Sperling, N.F.; Washington, R.; Whittaker, R.J. Future climate change of the subtropical North Atlantic: Implications for the cloud forests of Tenerife. Clim. Change 2004, 65, 103–123. [Google Scholar] [CrossRef]
- Caracterización Climática de las Islas Canarias para la Aplicación del Código Técnico de la Edificaci; CLIMCAN-010 y de su Aplicación Informática, prCTE-DR/CC.AA-008/10; Gobierno de Canarias: Las Palmas, Spain, 2010.
- Real Decreto 314/2006; BOE No. 74; Ministry of Housing of Spain: Madrid, Spain, 2006; pp. 11816–11831.
- Santana Rodríguez, J.J.; Santana Hernández, F.J.; González González, J.E. The effect of environmental and meteorological variables on atmospheric corrosion of carbon Steel, zinc and aluminium in a limited geographic zone with different types of environment. Corros. Sci. 2003, 45, 799–815. [Google Scholar] [CrossRef]
- ASTM G1-90. Standard Practice for Preparing, Cleaning, and Evaluating Corrosion Test Specimens; American Society for Testing Materials: Philadelphia, PA, USA, 1990. [Google Scholar]
- ISO/TC 156 N 250: Corrosion of Metals and Alloys. Aggressivity of Atmospheres. Methods of Measurement of Pollution Data; International Organization for Standardization: Geneva, Switzerland, 1986. [Google Scholar]
- ASTM D 2010-85: Standard Method for Evaluation of Total Sulfation Activity in the Atmosphere by the Lead Dioxide Candle; American Society for Testing Materials: Philadelphia, PA, USA, 1985.
- Nelder, J.A.; Mead, R. A Simplex method for function minimization. Comput. J. 1965, 7, 308–313. [Google Scholar] [CrossRef]
- Cabanelas, I.; Collazo, A.; Izquierdo, M.; Nóvoa, X.R.; Pérez, C. Influence of galvanised surface state on the duplex systems behavior. Corros. Sci. 2007, 49, 1816–1832. [Google Scholar] [CrossRef]
- Pillai, R.; Ackermann, H.; Lucka, K. Predicting the depletion of chromium in two high temperature Ni alloys. Corros. Sci. 2013, 69, 181–190. [Google Scholar] [CrossRef]
| Metal | Element (wt.%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Si | Fe | C | Mn | Zn | Ti | Cu | Mg | Al | Others | |
| Carbon steel | 0.08 | 99.47 | 0.06 | 0.37 | - | - | - | - | - | 0.023 |
| Copper | 0.28 | 0.9 | - | 0.05 | 0.09 | 0.05 | 98.5 | 0.05 | - | 0.09 |
| Zinc | - | - | - | - | 99.77 | 0.02 | 0.2 | - | 0.006 | - |
| Environmental Parameter | Level/Number of Samples per Level | |||||
|---|---|---|---|---|---|---|
| T | T1/10 | T2/14 | T3/40 | - | - | - |
| Pd | P0/49 | P1/15 | P2/0 | P3/0 | - | - |
| Sd | S0/0 | S1/4 | S2/60 | S3/0 | - | - |
| τ | τ1/0 | τ2/0 | τ3/25 | τ4/31 | τ5/8 | - |
| RH | RH1/3 | RH2/0 | RH3/0 | RH4/2 | RH5/46 | RH6/13 |
| Environmental Parameter | Sum sq. | Freedom Degrees | Mean sq. | f-Value | p-Value |
|---|---|---|---|---|---|
| RH | 392.5535 | 3 | 130.8512 | 0.7204 | 0.5442 |
| Pd | 680.9685 | 1 | 680.9685 | 3.7489 | 0.0581 |
| Sd | 4995.7843 | 1 | 4995.7843 | 27.5031 | 0.0000 |
| T | 236.6076 | 2 | 118.3038 | 0.6513 | 0.5254 |
| τ | 1071.0287 | 2 | 535.5143 | 2.9482 | 0.0609 |
| Error | 9808.7825 | 54 | 181.6441 | - | - |
| Total | 20,246.1375 | 63 | - | - | - |
| Environmental Parameter | Sum sq. | Freedom Degrees | Mean sq. | f-Value | p-Value |
|---|---|---|---|---|---|
| RH | 8635.9756 | 3 | 2878.6585 | 5.1955 | 0.0032 |
| Pd | 1302.4139 | 1 | 1302.4139 | 2.3506 | 0.1311 |
| Sd | 7581.5157 | 1 | 7581.5157 | 13.6833 | 0.0005 |
| T | 2580.5849 | 2 | 1290.2925 | 2.3288 | 0.1071 |
| τ | 5884.669 | 2 | 2942.3345 | 5.3104 | 0.0078 |
| Error | 29,919.7938 | 54 | 554.0703 | - | - |
| Total | 60,270.1119 | 63 | - | - | - |
| Environmental Parameter | Sum sq. | Freedom Degrees | Mean sq. | f-Value | p-Value |
|---|---|---|---|---|---|
| RH | 18.194 | 3 | 6.0647 | 0.6768 | 0.57 |
| Pd | 3.7699 | 1 | 3.7699 | 0.4207 | 0.5193 |
| Sd | 77.0076 | 1 | 77.0076 | 8.5939 | 0.0049 |
| T | 45.2614 | 2 | 22.6307 | 2.5255 | 0.0894 |
| τ | 7.9729 | 2 | 3.9865 | 0.4449 | 0.6432 |
| Error | 483.8814 | 54 | 8.9608 | - | - |
| Total | 650.7247 | 63 | - | - | - |
| Equation | Metal | Constants | SSE | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| a | b | c | d | e | f | g | h | |||
| (1) | CS | 1.77 | 0.52 | 0.02 | 0.054 | 0.102 | 0.62 | 0.033 | 0.04 | 116.7368 |
| (1) | Zn | 0.0129 | 0.44 | 0.046 | 0.071 | 0.0175 | 0.57 | 0.008 | 0.085 | 295.5821 |
| (1) | Cu | 0.0053 | 0.26 | 0.059 | 0.08 | 0.01 | 0.27 | 0.036 | 0.049 | 41.4758 |
| (1) * | CS | 4.855 | 0.6 | 0.01589 | 0.1089 | 0.871 | 0.9 | 2.2 × 10−4 | 2.0 × 10−4 | 85.1416 |
| (1) * | Zn | 0.0015 | 0.44 | 0.0359 | 0.371 | 0.4435 | 0.86 | 0.0096 | 0.0189 | 187.3936 |
| (1) * | Cu | 2.6528 | 0.25 | 0.00011 | 0.0048 | 0.09 | 0.9 | 0.0001 | 0.0162 | 22.7040 |
| (3) | CS | 0.3592 | 0.6 | 0.0005 | 1 × 10−6 | 0.8403 | 0.9 | 4 × 10−5 | 0.00163 | 86.7857 |
| (3) | Zn | 6.1796 | 0.44 | 0.0013 | 0.095 | 1.0156 | 0.86 | 0.0001 | 0.1 × 10−6 | 197.68 |
| (3) | Cu | 2.6528 | 0.25 | 1 × 10−6 | 21 × 10−6 | 0.09 | 0.9 | 5.7 × 10−6 | 0.0003 | 21.4148 |
| SO2 deposition rate in mg/(m2 day) | 9223:1992 [15] | 9223:2012 [11] | Level |
| Pd ≤ 10 | Pd ≤ 4 | P0—Rural atmosphere | |
| 10 < Pd ≤ 35 | 4 < Pd ≤ 24 | P1—Urban atmosphere | |
| 35 < Pd ≤ 80 | 24 < Pd ≤ 80 | P2—Industrial atmosphere | |
| 80 < Pd ≤ 200 | 80 < Pd ≤ 200 | P3—Highly polluted industrial atmosphere | |
| New corrosivity category CX (according to ISO 9223:2012). | |||
| Carbon steel | 200 < rcorr ≤ 700 (μm/year) | 1500 < rcorr ≤ 5500 (g/(m2 year)) | |
| Zinc | 8.4 < rcorr ≤ 25 (μm/year) | 60 < rcorr ≤ 180 (g/(m2 year)) | |
| Copper | 5.6 < rcorr ≤ 10 (μm/year) | 50 < rcorr ≤ 90 (g/(m2 year)) | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Santana, J.J.; Ramos, A.; Rodriguez-Gonzalez, A.; Vasconcelos, H.C.; Mena, V.; Fernández-Pérez, B.M.; Souto, R.M. Shortcomings of International Standard ISO 9223 for the Classification, Determination, and Estimation of Atmosphere Corrosivities in Subtropical Archipelagic Conditions—The Case of the Canary Islands (Spain). Metals 2019, 9, 1105. https://doi.org/10.3390/met9101105
Santana JJ, Ramos A, Rodriguez-Gonzalez A, Vasconcelos HC, Mena V, Fernández-Pérez BM, Souto RM. Shortcomings of International Standard ISO 9223 for the Classification, Determination, and Estimation of Atmosphere Corrosivities in Subtropical Archipelagic Conditions—The Case of the Canary Islands (Spain). Metals. 2019; 9(10):1105. https://doi.org/10.3390/met9101105
Chicago/Turabian StyleSantana, Juan J., Alejandro Ramos, Alejandro Rodriguez-Gonzalez, Helena C. Vasconcelos, Vicente Mena, Bibiana M. Fernández-Pérez, and Ricardo M. Souto. 2019. "Shortcomings of International Standard ISO 9223 for the Classification, Determination, and Estimation of Atmosphere Corrosivities in Subtropical Archipelagic Conditions—The Case of the Canary Islands (Spain)" Metals 9, no. 10: 1105. https://doi.org/10.3390/met9101105
APA StyleSantana, J. J., Ramos, A., Rodriguez-Gonzalez, A., Vasconcelos, H. C., Mena, V., Fernández-Pérez, B. M., & Souto, R. M. (2019). Shortcomings of International Standard ISO 9223 for the Classification, Determination, and Estimation of Atmosphere Corrosivities in Subtropical Archipelagic Conditions—The Case of the Canary Islands (Spain). Metals, 9(10), 1105. https://doi.org/10.3390/met9101105








