Biodegradable Metallic Wires in Dental and Orthopedic Applications: A Review
Abstract
1. Introduction
2. Biological and Mechanical Aspects of Biodegradable Metallic Materials for Bone Regeneration Applications
2.1. Mg and Its Alloys
2.1.1. Advantages of Mg and Its Alloys
2.1.2. Disadvantages of Mg and Its Alloys
2.2. Fe and Its Alloys
2.2.1. Advantages of Fe and Its Alloys
2.2.2. Disadvantages of Fe and Its Alloys
2.3. Zn and Its Alloys
2.3.1. Advantages of Zn and Its Alloys
2.3.2. Disadvantages of Zn and Its Alloys
3. Current Orthopedic and Dental Implant Applications of Metallic Wires
3.1. K-Wires
3.2. Cerclage Wires
3.3. Tension-Band Wires
3.4. Orthodontic Archwires
3.5. Ligature Wires
3.6. Staples
3.7. Sutures
4. The evolution of Metallic Wires in Bone Fixation Devices
4.1. Permanent Metallic Wires
4.2. Biodegradable Polymeric (BP) Wires
5. Recent Developments of BM Wires in Bone Applications
5.1. Medical Sutures and Staples
5.2. Bone Tissue Engineering
6. Future Work
6.1. Alloying Elements
6.1.1. Magnesium
6.1.2. Iron
6.1.3. Zinc
6.2. Surface Modification
6.2.1. Magnesium
6.2.2. Iron
6.3. Fabrication Processes
7. Conclusions
Author Contributions
Conflicts of Interest
References
- Griebel, A.J.; Schaffer, J.E. Absorbable filament technologies: Wire-drawing to enable next-generation medical devices. In Magnesium Technology 2016; Springer: Berlin, Germany, 2016; pp. 323–327. [Google Scholar]
- Sardeshmukh, A.; Reddy, S.; Gautham, B.; Joshi, A.; Panchal, J. A data science approach for analysis of multi-pass wire drawing. In Proceedings of the ASME 2017 International Design Engineering Technical Conferences and Computers and Information in Engineering Conference, Cleveland, OH, USA, 6–9 August 2017; American Society of Mechanical Engineers: New York, NY, USA, 2017; p. V001T002A071. [Google Scholar] [CrossRef]
- Griebel, A.J.; Schaffer, J.E.; Hopkins, T.M.; Alghalayini, A.; Mkorombindo, T.; Ojo, K.O.; Xu, Z.; Little, K.J.; Pixley, S.K. An in vitro and in vivo characterization of fine WE43B magnesium wire with varied thermomechanical processing conditions. J. Biomed. Mater. Res. Part B Appl. Biomater. 2017. [Google Scholar] [CrossRef] [PubMed]
- Duerig, T.; Pelton, A.; Stöckel, D. An overview of nitinol medical applications. Mater. Sci. Eng. A 1999, 273, 149–160. [Google Scholar] [CrossRef]
- Welch, J.M.; Sutton, G. Coronary Guide Catheter. Google Patents WO2017020012 A1, 29 July 2016. [Google Scholar]
- Henkes, H.; Bose, A.; Felber, S.; Miloslavski, E.; Berg-Dammer, E.; Kühne, D. Endovascular coil occlusion of intracranial aneurysms assisted by a novel self-expandable nitinol microstent (neuroform). Int. Neuroradiol. 2002, 8, 107–119. [Google Scholar] [CrossRef] [PubMed]
- Tan, L.; Sun, D.-H.; Yu, T.; Wang, L.; Zhu, D.; Li, Y.-H. Death due to intra-aortic migration of kirschner wire from the clavicle: A case report and review of the literature. Medicine 2016, 95, e3741. [Google Scholar] [CrossRef] [PubMed]
- Bravo, L.A.; de Cabañes, A.G.; Manero, J.M.; Rúperez, E.; Gil, F.J. NiTi superelastic orthodontic archwires with polyamide coating. J. Mater. Sci. Mater. Med. 2014, 25, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Chakravorty, B. Configured ligature wire for quick conventional and overtie lingual archwire ligations. APOS Trends Orthod. 2017, 7, 108–110. [Google Scholar] [CrossRef]
- Iavazzo, C.; Gkegkes, I.D.; Vouloumanou, E.K.; Mamais, I.; Peppas, G.; Falagas, M.E. Sutures versus staples for the management of surgical wounds: A meta-analysis of randomized controlled trials. Am. Surg. 2011, 77, 1206–1221. [Google Scholar] [PubMed]
- Bowen, P.K.; Shearier, E.R.; Zhao, S.; Guillory, R.J.; Zhao, F.; Goldman, J.; Drelich, J.W. Biodegradable metals for cardiovascular stents: From clinical concerns to recent Zn-alloys. Adv. Healthc. Mater. 2016, 5, 1121–1140. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Zheng, Y.; Qin, L. Progress of biodegradable metals. Prog. Natl. Sci. Mater. Int. 2014, 24, 414–422. [Google Scholar] [CrossRef]
- Seitz, J.-M.; Durisin, M.; Goldman, J.; Drelich, J.W. Recent advances in biodegradable metals for medical sutures: A critical review. Adv. Healthc. Mater. 2015, 4, 1915–1936. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Yang, H.T.; Li, X.; Zheng, Y.F. In vitro evaluation of the feasibility of commercial Zn alloys as biodegradable metals. J. Mater. Sci. Technol. 2016, 32, 909–918. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, W.; Maitz, M.F.; Chen, M.; Zhang, H.; Mao, J.; Zhao, Y.; Huang, N.; Wan, G. Comparative corrosion behavior of Zn with Fe and Mg in the course of immersion degradation in phosphate buffered saline. Corros. Sci. 2016, 111, 541–555. [Google Scholar] [CrossRef]
- Li, H.; Yang, H.; Zheng, Y.; Zhou, F.; Qiu, K.; Wang, X. Design and characterizations of novel biodegradable ternary Zn-based alloys with IIA nutrient alloying elements Mg, Ca and Sr. Mater. Des. 2015, 83, 95–102. [Google Scholar] [CrossRef]
- Witte, F. Reprint of: The history of biodegradable magnesium implants: A review. Acta Biomater. 2015, 23, S28–S40. [Google Scholar] [CrossRef] [PubMed]
- Andrews, E.W. Absorbable metal clips as substitutes for ligatures and deep sutures in wound closure. J. Am. Med. Assoc. 1917, 69, 278–281. [Google Scholar] [CrossRef]
- Seelig, M. A study of magnesium wire as an absorbable suture and ligature material. Arch. Surg. 1924, 8, 669–680. [Google Scholar] [CrossRef]
- Hou, L.-D.; Li, Z.; Pan, Y.; Sabir, M.; Zheng, Y.-F.; Li, L. A review on biodegradable materials for cardiovascular stent application. Front. Mater. Sci. 2016, 10, 238–259. [Google Scholar] [CrossRef]
- Li, X.; Chu, C.; Zhou, L.; Bai, J.; Guo, C.; Xue, F.; Lin, P.; Chu, P.K. Fully degradable PLA-based composite reinforced with 2D-braided Mg wires for orthopedic implants. Compos. Sci. Technol. 2017, 142, 180–188. [Google Scholar] [CrossRef]
- Staiger, M.P.; Pietak, A.M.; Huadmai, J.; Dias, G. Magnesium and its alloys as orthopedic biomaterials: A review. Biomaterials 2006, 27, 1728–1734. [Google Scholar] [CrossRef] [PubMed]
- Zhao, N.; Zhu, D. Application of Mg-based alloys for cardiovascular stents. Int. J. Biomed. Eng. Technol. 2013, 12, 382–398. [Google Scholar] [CrossRef]
- Liu, A.; Sun, M.; Shao, H.; Yang, X.; Ma, C.; He, D.; Gao, Q.; Liu, Y.; Yan, S.; Xu, S. The outstanding mechanical response and bone regeneration capacity of robocast dilute magnesium-doped wollastonite scaffolds in critical size bone defects. J. Mater. Chem. B 2016, 4, 3945–3958. [Google Scholar] [CrossRef]
- Wu, L.; Feyerabend, F.; Schilling, A.F.; Willumeit-Römer, R.; Luthringer, B.J. Effects of extracellular magnesium extract on the proliferation and differentiation of human osteoblasts and osteoclasts in coculture. Acta Biomater. 2015, 27, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Persaud-Sharma, D.; McGoron, A. Biodegradable magnesium alloys: A review of material development and applications. J. Biomim. Biomater. Tissue Eng. 2012, 12, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.F.; Gu, X.N.; Witte, F. Biodegradable metals. Mater. Sci. Eng. R Rep. 2014, 77, 1–34. [Google Scholar] [CrossRef]
- Pilarska, A.A.; Klapiszewski, Ł.; Jesionowski, T. Recent development in the synthesis, modification and application of Mg(OH)2 and MgO: A review. Powder Technol. 2017, 319, 373–407. [Google Scholar] [CrossRef]
- Guo, M.; Muhammad, F.; Wang, A.; Qi, W.; Wang, N.; Guo, Y.; Wei, Y.; Zhu, G. Magnesium hydroxide nanoplates: A pH-responsive platform for hydrophobic anticancer drug delivery. J. Mater. Chem. B 2013, 1, 5273–5278. [Google Scholar] [CrossRef]
- Janning, C.; Willbold, E.; Vogt, C.; Nellesen, J.; Meyer-Lindenberg, A.; Windhagen, H.; Thorey, F.; Witte, F. Magnesium hydroxide temporarily enhancing osteoblast activity and decreasing the osteoclast number in peri-implant bone remodelling. Acta Biomater. 2010, 6, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Weizbauer, A.; Kieke, M.; Rahim, M.I.; Angrisani, G.L.; Willbold, E.; Diekmann, J.; Flörkemeier, T.; Windhagen, H.; Müller, P.P.; Behrens, P.; et al. Magnesium-containing layered double hydroxides as orthopaedic implant coating materials—An in vitro and in vivo study. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 525–531. [Google Scholar] [CrossRef] [PubMed]
- Hickey, D.J.; Ercan, B.; Chung, S.; Webster, T.J.; Sun, L.; Geilich, B. Mgo nanocomposites as new antibacterial materials for orthopedic tissue engineering applications. In Proceedings of the 2014 40th Annual Northeast Bioengineering Conference (NEBEC), Boston, MA, USA, 25–27 April 2014; pp. 1–2. [Google Scholar]
- Huang, L.; Li, D.-Q.; Lin, Y.-J.; Wei, M.; Evans, D.G.; Duan, X. Controllable preparation of Nano-MgO and investigation of its bactericidal properties. J. Inorg. Biochem. 2005, 99, 986–993. [Google Scholar] [CrossRef] [PubMed]
- Haghshenas, M. Mechanical characteristics of biodegradable magnesium matrix composites: A review. J. Magn. Alloys 2017, 5, 189–201. [Google Scholar] [CrossRef]
- Gu, X.-N.; Zheng, Y.-F. A review on magnesium alloys as biodegradable materials. Front. Mater. Sci. China 2010, 4, 111–115. [Google Scholar] [CrossRef]
- Yusop, A.; Bakir, A.; Shaharom, N.; Abdul Kadir, M.; Hermawan, H. Porous biodegradable metals for hard tissue scaffolds: A review. Int. J. Biomater. 2012, 2012, 641430. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Witte, F.; Lu, F.; Wang, J.; Li, J.; Qin, L. Current status on clinical applications of magnesium-based orthopaedic implants: A review from clinical translational perspective. Biomaterials 2017, 112, 287–302. [Google Scholar] [CrossRef] [PubMed]
- Haynes, W.M. CRC Handbook of Chemistry and Physics; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Feyerabend, F.; Wendel, H.-P.; Mihailova, B.; Heidrich, S.; Agha, N.A.; Bismayer, U.; Willumeit-Römer, R. Blood compatibility of magnesium and its alloys. Acta Biomater. 2015, 25, 384–394. [Google Scholar] [CrossRef] [PubMed]
- Harandi, S.E.; Banerjee, P.C.; Easton, C.D.; Singh Raman, R.K. Influence of bovine serum albumin in hanks’ solution on the corrosion and stress corrosion cracking of a magnesium alloy. Mater. Sci. Eng. C 2017, 80, 335–345. [Google Scholar] [CrossRef] [PubMed]
- Noviana, D.; Paramitha, D.; Ulum, M.F.; Hermawan, H. The effect of hydrogen gas evolution of magnesium implant on the postimplantation mortality of rats. J. Orthop. Transl. 2016, 5, 9–15. [Google Scholar] [CrossRef]
- Tang, J.; Wang, J.; Xie, X.; Zhang, P.; Lai, Y.; Li, Y.; Qin, L. Surface coating reduces degradation rate of magnesium alloy developed for orthopaedic applications. J. Orthop. Transl. 2013, 1, 41–48. [Google Scholar] [CrossRef]
- Kraus, T.; Fischerauer, S.F.; Hänzi, A.C.; Uggowitzer, P.J.; Löffler, J.F.; Weinberg, A.M. Magnesium alloys for temporary implants in osteosynthesis: In vivo studies of their degradation and interaction with bone. Acta Biomater. 2012, 8, 1230–1238. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Xu, X.; Xu, Z.; Wang, J.; Cai, H. Development of Fe-based degradable metallic biomaterials. Metallic Biomater. New Direct. Technol. 2017, 113–160. [Google Scholar]
- Zimmermann, M.B.; Hurrell, R.F. Nutritional iron deficiency. Lancet 2007, 370, 511–520. [Google Scholar] [CrossRef]
- Toxqui, L.; Vaquero, M.P. Chronic iron deficiency as an emerging risk factor for osteoporosis: A hypothesis. Nutrients 2015, 7, 2324–2344. [Google Scholar] [CrossRef] [PubMed]
- Francis, A.; Yang, Y.; Virtanen, S.; Boccaccini, A.R. Iron and Iron-based alloys for temporary cardiovascular applications. J. Mater. Sci. Mater. Med. 2015, 26, 138. [Google Scholar] [CrossRef] [PubMed]
- Kraus, T.; Moszner, F.; Fischerauer, S.; Fiedler, M.; Martinelli, E.; Eichler, J.; Witte, F.; Willbold, E.; Schinhammer, M.; Meischel, M. Biodegradable Fe-based alloys for use in osteosynthesis: Outcome of an in vivo study after 52 weeks. Acta Biomater. 2014, 10, 3346–3353. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Zheng, Y.; Han, Y. Accelerating degradation rate of pure iron by Zinc Ion implantation. Regener. Biomater. 2016, 3, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Mouzou, E.; Paternoster, C.; Tolouei, R.; Chevallier, P.; Biffi, C.A.; Tuissi, A.; Mantovani, D. CO2-rich atmosphere strongly affects the degradation of Fe-21Mn-1C for biodegradable metallic implants. Mater. Lett. 2016, 181, 362–366. [Google Scholar] [CrossRef]
- Schinhammer, M.; Hanzi, A.C.; Loffler, J.F.; Uggowitzer, P.J. Design strategy for biodegradable Fe-based alloys for medical applications. Acta Biomater. 2010, 6, 1705–1713. [Google Scholar] [CrossRef] [PubMed]
- Keen, C.L.; Zidenberg-Cherr, S. Manganese A2—Caballero, benjamin. In Encyclopedia of Food Sciences and Nutrition, 2nd ed.; Academic Press: Oxford, UK, 2003; pp. 3686–3691. [Google Scholar]
- Dermience, M.; Lognay, G.; Mathieu, F.; Goyens, P. Effects of thirty elements on bone metabolism. J. Trace Elements Med. Biol. 2015, 32, 86–106. [Google Scholar] [CrossRef] [PubMed]
- Sikora-Jasinska, M.; Mostaed, E.; Mostaed, A.; Beanland, R.; Mantovani, D.; Vedani, M. Fabrication, mechanical properties and in vitro degradation behavior of newly developed Zn Ag alloys for degradable implant applications. Mater. Sci. Eng. C 2017, 77, 1170–1181. [Google Scholar] [CrossRef] [PubMed]
- Katarivas Levy, G.; Goldman, J.; Aghion, E. The prospects of zinc as a structural material for biodegradable implants—A review paper. Metals 2017, 7, 402. [Google Scholar] [CrossRef]
- Li, H.F.; Xie, X.H.; Zheng, Y.F.; Cong, Y.; Zhou, F.Y.; Qiu, K.J.; Wang, X.; Chen, S.H.; Huang, L.; Tian, L.; et al. Development of biodegradable Zn-1X binary alloys with nutrient alloying elements Mg, Ca and Sr. Sci. Rep. 2015, 5, 10719. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Wang, C.; Liu, C.; Chen, H.; Wu, Y.; Han, J.; Jia, Z.; Lin, W.; Zhang, D.; Li, W.; et al. Evolution of the degradation mechanism of pure Zinc stent in the one-year study of rabbit abdominal aorta model. Biomaterials 2017, 145, 92–105. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Sun, J.; Yang, Y.; Zhou, F.; Pu, Z.; Li, L.; Zheng, Y. Microstructure, mechanical properties, in vitro degradation behavior and hemocompatibility of novel Zn–Mg–Sr alloys as biodegradable metals. Mater. Lett. 2016, 162, 242–245. [Google Scholar] [CrossRef]
- Zhu, H.; Jin, G.; Cao, H.; Qiao, Y.; Liu, X. Influence of implantation voltage on the biological properties of Zinc-implanted titanium. Surf. Coat. Technol. 2017, 312, 75–80. [Google Scholar] [CrossRef]
- Seo, H.-J.; Cho, Y.-E.; Kim, T.; Shin, H.-I.; Kwun, I.-S. Zinc may increase bone formation through stimulating cell proliferation, alkaline phosphatase activity and collagen synthesis in osteoblastic MC3T3-E1 cells. Nutr. Res. Pract. 2010, 4, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Moonga, B.S.; Dempster, D.W. Zinc is a potent inhibitor of osteoclastic bone resorption in vitro. J. Bone Min. Res. 1995, 10, 453–457. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, P.; Rai, D.V.; Garg, M.L. Zinc inhibits ovariectomy induced microarchitectural changes in the bone tissue. J. Nutr. Intermed. Metab. 2016, 3, 33–40. [Google Scholar] [CrossRef]
- Yamaguchi, M. Role of nutritional zinc in the prevention of osteoporosis. Mol. Cell. Biochem. 2010, 338, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Pasquet, J.; Chevalier, Y.; Pelletier, J.; Couval, E.; Bouvier, D.; Bolzinger, M.-A. The contribution of zinc ions to the antimicrobial activity of zinc oxide. Colloids Surf. A Physicochem. Eng. Asp. 2014, 457, 263–274. [Google Scholar] [CrossRef]
- Boyd, D.; Li, H.; Tanner, D.A.; Towler, M.R.; Wall, J.G. The antibacterial effects of zinc ion migration from Zinc-based glass polyalkenoate cements. J. Mater. Sci. Mater. Med. 2006, 17, 489–494. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, T.J.; Zeelie, J.J.; Krause, D.J. The antimicrobial action of Zinc ion/antioxidant combinations. J. Clin. Pharm. Ther. 1992, 17, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Drelich, A.J.; Bowen, P.K.; LaLonde, L.; Goldman, J.; Drelich, J.W. Importance of oxide film in endovascular biodegradable Zinc stents. Surf. Innov. 2016, 4, 133–140. [Google Scholar] [CrossRef]
- Moon, S.-H.; Choi, W.J.; Choi, S.-W.; Kim, E.H.; Kim, J.; Lee, J.-O.; Kim, S.H. Anti-cancer activity of ZnO chips by sustained Zinc Ion release. Toxicol. Rep. 2016, 3, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-W.; Choi, W.J.; Kim, E.H.; Moon, S.-H.; Park, S.-J.; Lee, J.-O.; Kim, S.H. Inflammatory bone resorption and antiosteosarcoma potentials of Zinc Ion sustained release ZnO chips: Friend or foe? ACS Biomater. Sci. Eng. 2016, 2, 494–500. [Google Scholar] [CrossRef]
- Vojtech, D.; Kubasek, J.; Serak, J.; Novak, P. Mechanical and corrosion properties of newly developed biodegradable Zn-based alloys for bone fixation. Acta Biomater. 2011, 7, 3515–3522. [Google Scholar] [CrossRef] [PubMed]
- Törne, K.; Larsson, M.; Norlin, A.; Weissenrieder, J. Degradation of Zinc in saline solutions, plasma, and whole blood. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 1141–1151. [Google Scholar] [CrossRef] [PubMed]
- Katz, E.P.; Li, S.-T. Structure and function of bone collagen fibrils. J. Mol. Biol. 1973, 80, 1–15. [Google Scholar] [CrossRef]
- Tan, L.; Yu, X.; Wan, P.; Yang, K. Biodegradable materials for bone repairs: A review. J. Mater. Sci. Technol. 2013, 29, 503–513. [Google Scholar] [CrossRef]
- Huber, W. Historical remarks on martin kirschner and the development of the kirschner (K)-wire. Indian J. Plast. Surg. 2008, 41, 89–92. [Google Scholar] [CrossRef] [PubMed]
- Harasen, G. Orthopedic hardware and equipment for the beginner: Part 1. Pins and wires. Can. Vet. J. 2011, 52, 1025–1026. [Google Scholar] [PubMed]
- Ricbard, M.; Slone, M.M.H.; Vander Griend Robert, A.; William, J. Montgomery Orthopaedic fixation devices. RadioGraphics 1991, 11, 823–847. [Google Scholar]
- Rudolph, D.J.; Willes, M.G.; Sameshima, G.T. A finite element model of apical force distribution from orthodontic tooth movement. Angle Orthod. 2001, 71, 127–131. [Google Scholar] [PubMed]
- Henriques, J.F.C.; Higa, R.H.; Semenara, N.T.; Janson, G.; Fernandes, T.M.F.; Sathler, R. Evaluation of deflection forces of orthodontic wires with different ligation types. Braz. Oral Res. 2017, 31, e49. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cowley, L.L. Wire sutures: Braided or monofilament? Am. J. Surg. 1967, 113, 472–474. [Google Scholar] [CrossRef]
- Karmani, S.; Lam, F. The design and function of surgical drills and k-wires. Curr. Orthop. 2004, 18, 484–490. [Google Scholar] [CrossRef]
- Massengill, J.B.; Alexander, H.; Parson, J.R.; Schecter, M.J. Mechanical analysis of kirschner wire fixation in a phalangeal model. J. Hand Surg. 1979, 4, 351–356. [Google Scholar] [CrossRef]
- Viegas, S.F.; Ferren, E.L.; Self, J.; Tencer, A.F. Comparative mechanical properties of various kirschner wire configurations in transverse and oblique phalangeal fractures. J. Hand Surg. 1988, 13, 246–253. [Google Scholar] [CrossRef]
- Stark, H.H.; Rickard, T.; Zemel, N.; Ashworth, C. Treatment of ununited fractures of the scaphoid by iliac bone grafts and kirschner-wire fixation. J. Bone Jt. Surg. Am. 1988, 70, 982–991. [Google Scholar] [CrossRef]
- Hope, P.G.; Williamson, D.M.; Coates, C.J.; Cole, W.G. Biodegradable pin fixation of elbow fractures in children. A randomised trial. J. Bone Jt. Surg. Br. Vol. 1991, 73, 965–968. [Google Scholar] [CrossRef]
- Leppilahti, J.; Jalovaara, P. Migration of kirschner wires following fixation of the clavicle—A report of 2 cases. Acta Orthop. Scand. 1999, 70, 517–519. [Google Scholar] [CrossRef] [PubMed]
- Gbur, J.L.; Lewandowski, J.J. Fatigue and fracture of wires and cables for biomedical applications. Int. Mater. Rev. 2016, 61, 231–314. [Google Scholar] [CrossRef]
- Chen, Q.; Thouas, G.A. Metallic implant biomaterials. Mater. Sci. Eng. R Rep. 2015, 87, 1–57. [Google Scholar] [CrossRef]
- Prasad, K.; Bazaka, O.; Chua, M.; Rochford, M.; Fedrick, L.; Spoor, J.; Symes, R.; Tieppo, M.; Collins, C.; Cao, A.; et al. Metallic biomaterials: Current challenges and opportunities. Materials 2017, 10, 884. [Google Scholar] [CrossRef] [PubMed]
- Clauss, M.; Graf, S.; Gersbach, S.; Hintermann, B.; Ilchmann, T.; Knupp, M. Material and biofilm load of k wires in toe surgery: Titanium versus stainless steel. Clin. Orthop. Relat. Res. 2013, 471, 2312–2317. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.; Jensen, C. Biodegradable pins versus kirschner wires in hand surgery. J. Hand Surg. Br. Eur. Vol. 1996, 21, 507–510. [Google Scholar] [CrossRef]
- Plaga, B.; Royster, R.; Donigian, A.; Wright, G.; Caskey, P. Fixation of osteochondral fractures in rabbit knees. A comparison of kirschner wires, fibrin sealant, and polydioxanone pins. Bone Jt. J. 1992, 74, 292–296. [Google Scholar] [CrossRef]
- Casteleyn, P.P.; Handelberg, F.; Haentjens, P. Biodegradable rods versus kirschner wire fixation of wrist fractures. A randomised trial. J. Bone Jt. Surg. Br. Vol. 1992, 74, 858–861. [Google Scholar] [CrossRef]
- Juutilainen, T.; Patiälä, H.; Rokkanen, P.; Törmälä, P. Biodegradable wire fixation in olecranon and patella fractures combined with biodegradable screws or plugs and compared with metallic fixation. Arch. Orthop. Trauma Surg. 1995, 114, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Lurate, B.; Mukherjee, D.; Kruse, R.; Albright, J. Fixation of osteochondral fractures with absorbable pins. In Proceedings of the 1995 Fourteenth Southern Biomedical Engineering Conference, Shreveport, LA, USA, 7–9 April 1995; pp. 57–58. [Google Scholar]
- Maruyama, T.; Saha, S.; Mongiano, D.O.; Mudge, K. Metacarpal fracture fixation with absorbable polyglycolide rods and stainless steel k wires: A biomechanical comparison. J. Biomed. Mater. Res. Part A 1996, 33, 9–12. [Google Scholar] [CrossRef]
- Tian, P.; Liu, X. Surface modification of biodegradable magnesium and its alloys for biomedical applications. Regen. Biomater. 2015, 2, 135–151. [Google Scholar] [CrossRef] [PubMed]
- Witte, F.; Hort, N.; Vogt, C.; Cohen, S.; Kainer, K.U.; Willumeit, R.; Feyerabend, F. Degradable biomaterials based on magnesium corrosion. Curr. Opin. Solid State Mater. Sci. 2008, 12, 63–72. [Google Scholar] [CrossRef]
- Hanzi, A.C.; Gerber, I.; Schinhammer, M.; Loffler, J.F.; Uggowitzer, P.J. On the in vitro and in vivo degradation performance and biological response of new biodegradable Mg-Y-Zn alloys. Acta Biomater. 2010, 6, 1824–1833. [Google Scholar] [CrossRef] [PubMed]
- KubÁSek, J.; VojtĚCh, D. Structural and corrosion characterization of biodegradable Mg–Re (Re = Gd, Y, Nd) alloys. Trans. Nonferrous Met. Soc. China 2013, 23, 1215–1225. [Google Scholar] [CrossRef]
- Chen, L.; Bin, Y.; Zou, W.; Wang, X.; Li, W. The influence of Sr on the microstructure, degradation and stress corrosion cracking of the Mg alloys—ZK40xSr. J. Mech. Behav. Biomed. Mater. 2017, 66, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Gu, X.; Lou, S.; Zheng, Y. The development of binary Mg-Ca alloys for use as biodegradable materials within bone. Biomaterials 2008, 29, 1329–1344. [Google Scholar] [CrossRef] [PubMed]
- Gu, X.; Zheng, Y.; Cheng, Y.; Zhong, S.; Xi, T. In vitro corrosion and biocompatibility of binary magnesium alloys. Biomaterials 2009, 30, 484–498. [Google Scholar] [CrossRef] [PubMed]
- Lietaert, K.; Weber, L.; Van Humbeeck, J.; Mortensen, A.; Luyten, J.; Schrooten, J. Open cellular magnesium alloys for biodegradable orthopaedic implants. J. Magn. Alloys 2013, 1, 303–311. [Google Scholar] [CrossRef]
- Mutlu, I.; Oktay, E. Influence of fluoride content of artificial saliva on metal release from 17-4 PH stainless steel foam for dental implant applications. J. Mater. Sci. Technol. 2013, 29, 582–588. [Google Scholar] [CrossRef]
- Cui, W.; Beniash, E.; Gawalt, E.; Xu, Z.; Sfeir, C. Biomimetic coating of magnesium alloy for enhanced corrosion resistance and calcium phosphate deposition. Acta Biomater. 2013, 9, 8650–8659. [Google Scholar] [CrossRef] [PubMed]
- Erdmann, N.; Angrisani, N.; Reifenrath, J.; Lucas, A.; Thorey, F.; Bormann, D.; Meyer-Lindenberg, A. Biomechanical testing and degradation analysis of MgCa0.8 alloy screws: A comparative in vivo study in rabbits. Acta Biomater. 2011, 7, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- Kramer, M.; Schilling, M.; Eifler, R.; Hering, B.; Reifenrath, J.; Besdo, S.; Windhagen, H.; Willbold, E.; Weizbauer, A. Corrosion behavior, biocompatibility and biomechanical stability of a prototype magnesium-based biodegradable intramedullary nailing system. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 59, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Chaya, A.; Yoshizawa, S.; Verdelis, K.; Myers, N.; Costello, B.J.; Chou, D.T.; Pal, S.; Maiti, S.; Kumta, P.N.; Sfeir, C. In vivo study of magnesium plate and screw degradation and bone fracture healing. Acta Biomater. 2015, 18, 262–269. [Google Scholar] [CrossRef] [PubMed]
- Mc, B.E. Absorbable metal in bone surgery: A further report on the use of magnesium alloys. J. Am. Med. Assoc. 1938, 111, 2464–2467. [Google Scholar]
- Windhagen, H.; Radtke, K.; Weizbauer, A.; Diekmann, J.; Noll, Y.; Kreimeyer, U.; Schavan, R.; Stukenborg-Colsman, C.; Waizy, H. Biodegradable magnesium-based screw clinically equivalent to titanium screw in hallux valgus surgery: Short term results of the first prospective, randomized, controlled clinical pilot study. Biomed. Eng. Online 2013, 12, 62. [Google Scholar] [CrossRef] [PubMed]
- Ezechieli, M.; Ettinger, M.; Konig, C.; Weizbauer, A.; Helmecke, P.; Schavan, R.; Lucas, A.; Windhagen, H.; Becher, C. Biomechanical characteristics of bioabsorbable magnesium-based (mgyrezr-alloy) interference screws with different threads. Knee Surg. Sports Traumatol. Arthrosc. 2016, 24, 3976–3981. [Google Scholar] [CrossRef] [PubMed]
- Tie, D.; Feyerabend, F.; Muller, W.D.; Schade, R.; Liefeith, K.; Kainer, K.U.; Willumeit, R. Antibacterial biodegradable Mg-Ag alloys. Eur. Cells Mater. 2013, 25, 284–298. [Google Scholar] [CrossRef]
- Jahn, K.; Saito, H.; Taipaleenmaki, H.; Gasser, A.; Hort, N.; Feyerabend, F.; Schluter, H.; Rueger, J.M.; Lehmann, W.; Willumeit-Romer, R.; et al. Intramedullary Mg2Ag nails augment callus formation during fracture healing in mice. Acta Biomater. 2016, 36, 350–360. [Google Scholar] [CrossRef] [PubMed]
- Bian, D.; Zhou, W.; Deng, J.; Liu, Y.; Li, W.; Chu, X.; Xiu, P.; Cai, H.; Kou, Y.; Jiang, B.; et al. Development of magnesium-based biodegradable metals with dietary trace element germanium as orthopaedic implant applications. Acta Biomater. 2017, 64, 421–436. [Google Scholar] [CrossRef] [PubMed]
- Koo, Y.; Lee, H.B.; Dong, Z.; Kotoka, R.; Sankar, J.; Huang, N.; Yun, Y. The effects of static and dynamic loading on biodegradable magnesium pins in vitro and in vivo. Sci. Rep. 2017, 7, 14710. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zheng, Y.F. Effects of alloying elements (Mn, Co, Al, W, Sn, B, C and S) on biodegradability and in vitro biocompatibility of pure iron. Acta Biomater. 2011, 7, 1407–1420. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Zhou, C.; Fan, H.; Fan, Y.; Jiang, Q.; Song, P.; Fan, H.; Chen, Y.; Zhang, X. Bio-functional design, application and trends in metallic biomaterials. Int. J. Mol. Sci. 2017, 19, 24. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Zheng, Y.F.; Ruan, L. In vitro investigation of Fe30Mn6Si shape memory alloy as potential biodegradable metallic material. Mater. Lett. 2011, 65, 540–543. [Google Scholar] [CrossRef]
- Hermawan, H.; Dube, D.; Mantovani, D. Developments in metallic biodegradable stents. Acta Biomater. 2010, 6, 1693–1697. [Google Scholar] [CrossRef] [PubMed]
- Capek, J.; Msallamova, S.; Jablonska, E.; Lipov, J.; Vojtech, D. A novel high-strength and highly corrosive biodegradable Fe-Pd alloy: Structural, mechanical and in vitro corrosion and cytotoxicity study. Mater. Sci. Eng. C Mater. Biol. Appl. 2017, 79, 550–562. [Google Scholar] [CrossRef] [PubMed]
- Hufenbach, J.; Wendrock, H.; Kochta, F.; Kühn, U.; Gebert, A. Novel biodegradable Fe-Mn-C-S alloy with superior mechanical and corrosion properties. Mater. Lett. 2017, 186, 330–333. [Google Scholar] [CrossRef]
- Wang, H.; Zheng, Y.; Liu, J.; Jiang, C.; Li, Y. In vitro corrosion properties and cytocompatibility of Fe-Ga alloys as potential biodegradable metallic materials. Mater. Sci. Eng. C 2017, 71, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Babcock, W.W. Metallic sutures and ligatures. Surg. Clin. N. Am. 1947, 27, 1435–1460. [Google Scholar] [CrossRef]
- Chellamani, K.; Veerasubramanian, D.; Balaji, R. Surgical Sutures: An Overview. J. Acad. Ind. Res. 2013, 1, 778–782. [Google Scholar]
- Seitz, J.-M.; Wulf, E.; Freytag, P.; Bormann, D.; Bach, F.-W. The manufacture of resorbable suture material from magnesium. Adv. Eng. Mater. 2010, 12, 1099–1105. [Google Scholar] [CrossRef]
- Seitz, J.-M.; Utermöhlen, D.; Wulf, E.; Klose, C.; Bach, F.-W. The manufacture of resorbable suture material from magnesium—Drawing and stranding of thin wires. Adv. Eng. Mater. 2011, 13, 1087–1095. [Google Scholar] [CrossRef]
- Bai, J.; Yin, L.; Lu, Y.; Gan, Y.; Xue, F.; Chu, C.; Yan, J.; Yan, K.; Wan, X.; Tang, Z. Preparation, microstructure and degradation performance of biomedical magnesium alloy fine wires. Prog. Natl. Sci. Mater. Int. 2014, 24, 523–530. [Google Scholar] [CrossRef]
- Zhang, S.; Liu, J.; Yan, J.; Chen, Y.; Zhao, C.; Zhang, Y.; Jiang, M.; Xu, H.; Ni, J.; Zhang, X. In vivo degradation and biocompatibility of linear cutter staples made of high purity magnesium. Eur. Cells Mater. 2014, 28, 76. [Google Scholar]
- Wu, H.; Zhao, C.; Ni, J.; Zhang, S.; Liu, J.; Yan, J.; Chen, Y.; Zhang, X. Research of a novel biodegradable surgical staple made of high purity magnesium. Bioact. Mater. 2016, 1, 122–126. [Google Scholar] [CrossRef]
- Cao, J.; Jiang, K.W.; Yang, X.D.; Shen, Z.L.; Guo, P.; Yan, Y.C.; Cui, Y.C.; Han, L.; Lv, Y.; Ye, Y.J.; et al. [animal experimental study of biodegradable magnesium alloy stapler for gastrointestinal anastomosis]. Zhonghua Wei Chang Wai Ke Za Zhi Chin. J. Gastrointest. Surg. 2013, 16, 772–776. [Google Scholar]
- Moore, W.R.; Graves, S.E.; Bain, G.I. Synthetic bone graft substitutes. ANZ J. Surg. 2001, 71, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, W.; Schnitzler, V.; Tancret, F.; Bouler, J.-M. Calcium phosphate cements for bone substitution: Chemistry, handling and mechanical properties. Acta Biomater. 2014, 10, 1035–1049. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, G.; Moghaddam, A. Allograft bone matrix versus synthetic bone graft substitutes. Injury 2011, 42, S16–S21. [Google Scholar] [CrossRef] [PubMed]
- Claes, L.; Hoellen, I.; Ignatius, A. Biodegradable bone cements. Der Orthop. 1997, 26, 459–462. [Google Scholar] [CrossRef] [PubMed]
- Geffers, M.; Groll, J.; Gbureck, U. Reinforcement strategies for load-bearing calcium phosphate biocements. Materials 2015, 8, 2700–2717. [Google Scholar] [CrossRef]
- Vaishya, R.; Chauhan, M.; Vaish, A. Bone cement. J. Clin. Orthop. Trauma 2013, 4, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Bohner, M. Design of ceramic-based cements and putties for bone graft substitution. Eur. Cells Mater. 2010, 20, 3–10. [Google Scholar] [CrossRef]
- Dos Santos, L.A.; Carrodéguas, R.G.; Boschi, A.O.; Fonseca de Arruda, A.C. Fiber-enriched double-setting calcium phosphate bone cement. J. Biomed. Mater. Res. Part A 2003, 65, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Von Gonten, A.; Kelly, J.; Antonucci, J.M. Load-bearing behavior of a simulated craniofacial structure fabricated from a hydroxyapatite cement and bioresorbable fiber-mesh. J. Mater. Sci. Mater. Med. 2000, 11, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.H.K.; Quinn, J.B. Calcium phosphate cement containing resorbable fibers for short-term reinforcement and macroporosity. Biomaterials 2002, 23, 193–202. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, H.H. Effects of synergistic reinforcement and absorbable fiber strength on hydroxyapatite bone cement. J. Biomed. Mater. Res. Part A 2005, 75, 832–840. [Google Scholar] [CrossRef] [PubMed]
- Pan, Z.; Jiang, P.; Fan, Q.; Ma, B.; Cai, H. Mechanical and biocompatible influences of chitosan fiber and gelatin on calcium phosphate cement. J. Biomed. Mater. Res. Part B Appl. Biomater. 2007, 82, 246–252. [Google Scholar] [CrossRef] [PubMed]
- Krüger, R.; Groll, J. Fiber reinforced calcium phosphate cements–on the way to degradable load bearing bone substitutes? Biomaterials 2012, 33, 5887–5900. [Google Scholar] [CrossRef] [PubMed]
- Krüger, R.; Seitz, J.-M.; Ewald, A.; Bach, F.-W.; Groll, J. Strong and tough magnesium wire reinforced phosphate cement composites for load-bearing bone replacement. J. Mech. Behav. Biomed. Mater. 2013, 20, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; He, Y.; Zhou, J.; Tang, S.; Yang, Y.; Wang, X. Effects of scandium addition on biocompatibility of biodegradable Mg–1.5Zn–0.6Zr alloy. Mater. Lett. 2018, 215, 200–202. [Google Scholar] [CrossRef]
- Salleh, E.M.; Ramakrishnan, S.; Hussain, Z. Synthesis of biodegradable Mg-Zn alloy by mechanical alloying: Effect of milling time. Procedia Chem. 2016, 19, 525–530. [Google Scholar] [CrossRef]
- Gui, Z.; Kang, Z.; Li, Y. Mechanical and corrosion properties of Mg-Gd-Zn-Zr-Mn biodegradable alloy by hot extrusion. J. Alloys Compd. 2016, 685, 222–230. [Google Scholar] [CrossRef]
- Dai, J.; Zhang, X.; Yin, Q.; Ni, S.; Ba, Z.; Wang, Z. Friction and wear behaviors of biodegradable Mg-6Gd-0.5Zn-0.4Zr alloy under simulated body fluid condition. J. Magn. Alloys 2017, 5, 448–453. [Google Scholar] [CrossRef]
- Kubásek, J.; Vojtěch, D.; Jablonská, E.; Pospíšilová, I.; Lipov, J.; Ruml, T. Structure, mechanical characteristics and in vitro degradation, cytotoxicity, genotoxicity and mutagenicity of novel biodegradable Zn–Mg alloys. Mater. Sci. Eng. C 2016, 58, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Shen, C.; Liu, X.; Fan, B.; Lan, P.; Zhou, F.; Li, X.; Wang, H.; Xiao, X.; Li, L.; Zhao, S. Mechanical properties, in vitro degradation behavior, hemocompatibility and cytotoxicity evaluation of Zn–1.2Mg alloy for biodegradable implants. RSC Adv. 2016, 6, 86410–86419. [Google Scholar] [CrossRef]
- Dorozhkin, S.V. Calcium orthophosphate coatings on magnesium and its biodegradable alloys. Acta Biomater. 2014, 10, 2919–2934. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.W.; Shan, D.Y.; Han, E.H. Electrodeposition of hydroxyapatite coating on AZ91D magnesium alloy for biomaterial application. Mater. Lett. 2008, 62, 3276–3279. [Google Scholar] [CrossRef]
- Rojaee, R.; Fathi, M.; Raeissi, K. Controlling the degradation rate of AZ91 magnesium alloy via sol–gel derived nanostructured hydroxyapatite coating. Mater. Sci. Eng. C 2013, 33, 3817–3825. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Li, G.; Lian, J. A chemical conversion hydroxyapatite coating on AZ60 magnesium alloy and its electrochemical corrosion behaviour. Int. J. Electrochem. Sci. 2012, 7, 11497–11511. [Google Scholar]
- Zhang, Y.; Ma, Y.; Chen, M.; Wei, J. Effects of anodizing biodegradable Mg–Zn–Zr alloy on the deposition of Ca–P coating. Surf. Coat. Technol. 2013, 228, S111–S115. [Google Scholar] [CrossRef]
- Tsubakino, H.; Yamamoto, A.; Fukumoto, S.; Watanabe, A.; Sugahara, K.; Inoue, H. High-purity magnesium coating on magnesium alloys by vapor deposition technique for improving corrosion resistance. Mater. Trans. 2003, 44, 504–510. [Google Scholar] [CrossRef]
- Johnson, I.; Akari, K.; Liu, H. Nanostructured hydroxyapatite/poly (lactic-co-glycolic acid) composite coating for controlling magnesium degradation in simulated body fluid. Nanotechnology 2013, 24, 375103. [Google Scholar] [CrossRef] [PubMed]
- Butev, E.; Esen, Z.; Bor, S. In vitro bioactivity investigation of alkali treated Ti6Al7Nb alloy foams. Appl. Surf. Sci. 2015, 327, 437–443. [Google Scholar] [CrossRef]
- Tang, H.; Wu, T.; Xu, F.; Tao, W.; Jian, X. Fabrication and characterization of Mg(OH)2 films on AZ31 magnesium alloy by alkali treatment. Int. J. Electrochem. Sci. 2017, 12, 1377–1388. [Google Scholar] [CrossRef]
- Feng, J.; Chen, Y.; Liu, X.; Liu, T.; Zou, L.; Wang, Y.; Ren, Y.; Fan, Z.; Lv, Y.; Zhang, M. In-situ hydrothermal crystallization Mg(Oh)2 films on magnesium alloy AZ91 and their corrosion resistance properties. Mater. Chem. Phys. 2013, 143, 322–329. [Google Scholar] [CrossRef]
- Shadanbaz, S.; Dias, G.J. Calcium phosphate coatings on magnesium alloys for biomedical applications: A review. Acta Biomater. 2012, 8, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Harun, W.S.W.; Asri, R.I.M.; Alias, J.; Zulkifli, F.H.; Kadirgama, K.; Ghani, S.A.C.; Shariffuddin, J.H.M. A comprehensive review of hydroxyapatite-based coatings adhesion on metallic biomaterials. Ceram. Int. 2018, 44, 1250–1268. [Google Scholar] [CrossRef]
- Zhao, N.; Zhu, D. Collagen self-assembly on orthopedic magnesium biomaterials surface and subsequent bone cell attachment. PLoS ONE 2014, 9, e110420. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-L.; Yan, Y.-H.; Wan, T.; Yang, H. Poly (l-lactic acid)/hydroxyapatite/collagen composite coatings on AZ31 magnesium alloy for biomedical application. Proc. Inst. Mech. Eng. Part H J. Eng. Med. 2013, 227, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Z.; Shi, X.-H.; Zhang, P.-C.; Bai, B.; Leng, Y.-X.; Huang, N. The microstructure and properties of commercial pure iron modified by plasma nitriding. Solid State Ion. 2008, 179, 971–974. [Google Scholar] [CrossRef]
- Zhu, S.; Huang, N.; Xu, L.; Zhang, Y.; Liu, H.; Lei, Y.; Sun, H.; Yao, Y. Biocompatibility of Fe–O films synthesized by plasma immersion ion implantation and deposition. Surf. Coat. Technol. 2009, 203, 1523–1529. [Google Scholar] [CrossRef]
- Zhou, J.; Yang, Y.; Alonso Frank, M.; Detsch, R.; Boccaccini, A.R.; Virtanen, S. Accelerated degradation behavior and cytocompatibility of pure iron treated with sandblasting. ACS Appl. Mater. Interfaces 2016, 8, 26482–26492. [Google Scholar] [CrossRef] [PubMed]
- Dobatkin, S.V.; Lukyanova, E.A.; Martynenko, N.S.; Anisimova, N.Y.; Kiselevskiy, M.V.; Gorshenkov, M.V.; Yurchenko, N.Y.; Raab, G.I.; Yusupov, V.S.; Birbilis, N.; et al. Strength, corrosion resistance, and biocompatibility of ultrafine-grained mg alloys after different modes of severe plastic deformation. IOP Conf. Ser. Mater. Sci. Eng. 2017, 194, 012004. [Google Scholar] [CrossRef]
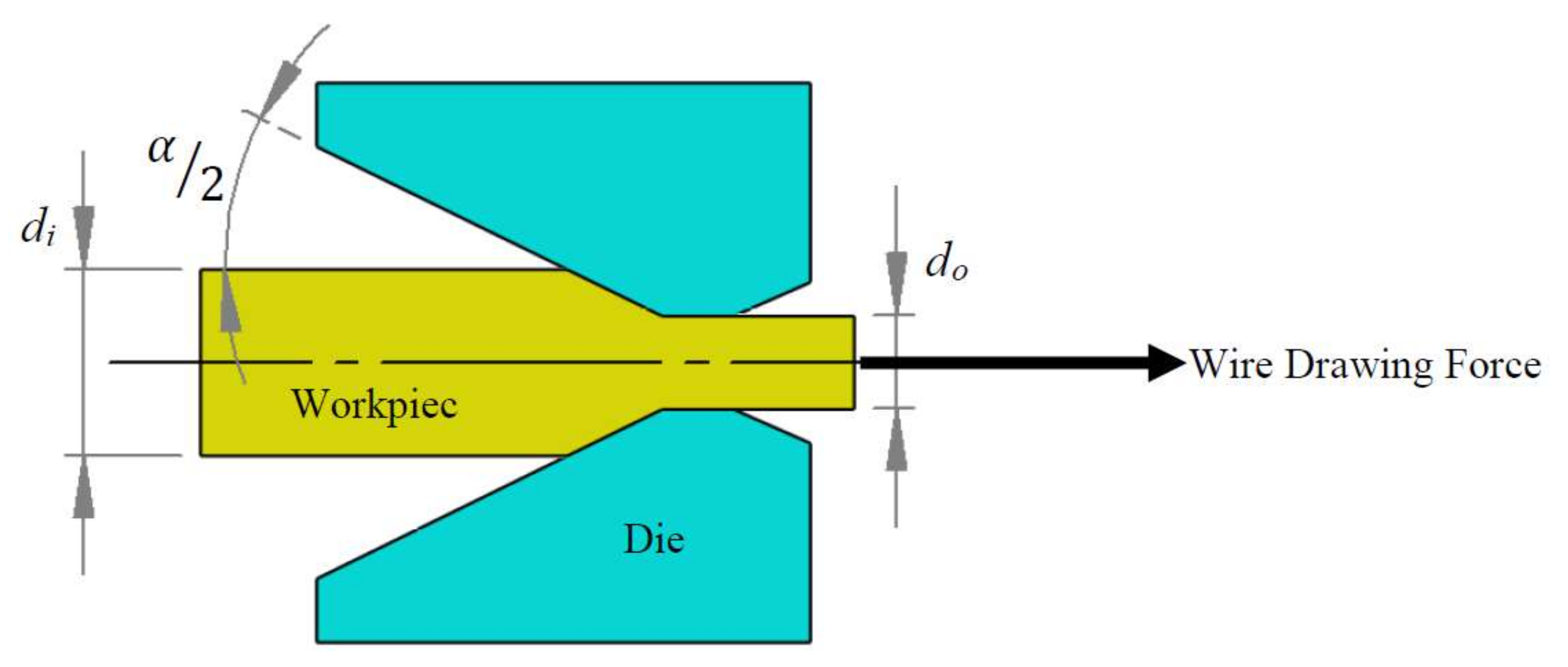
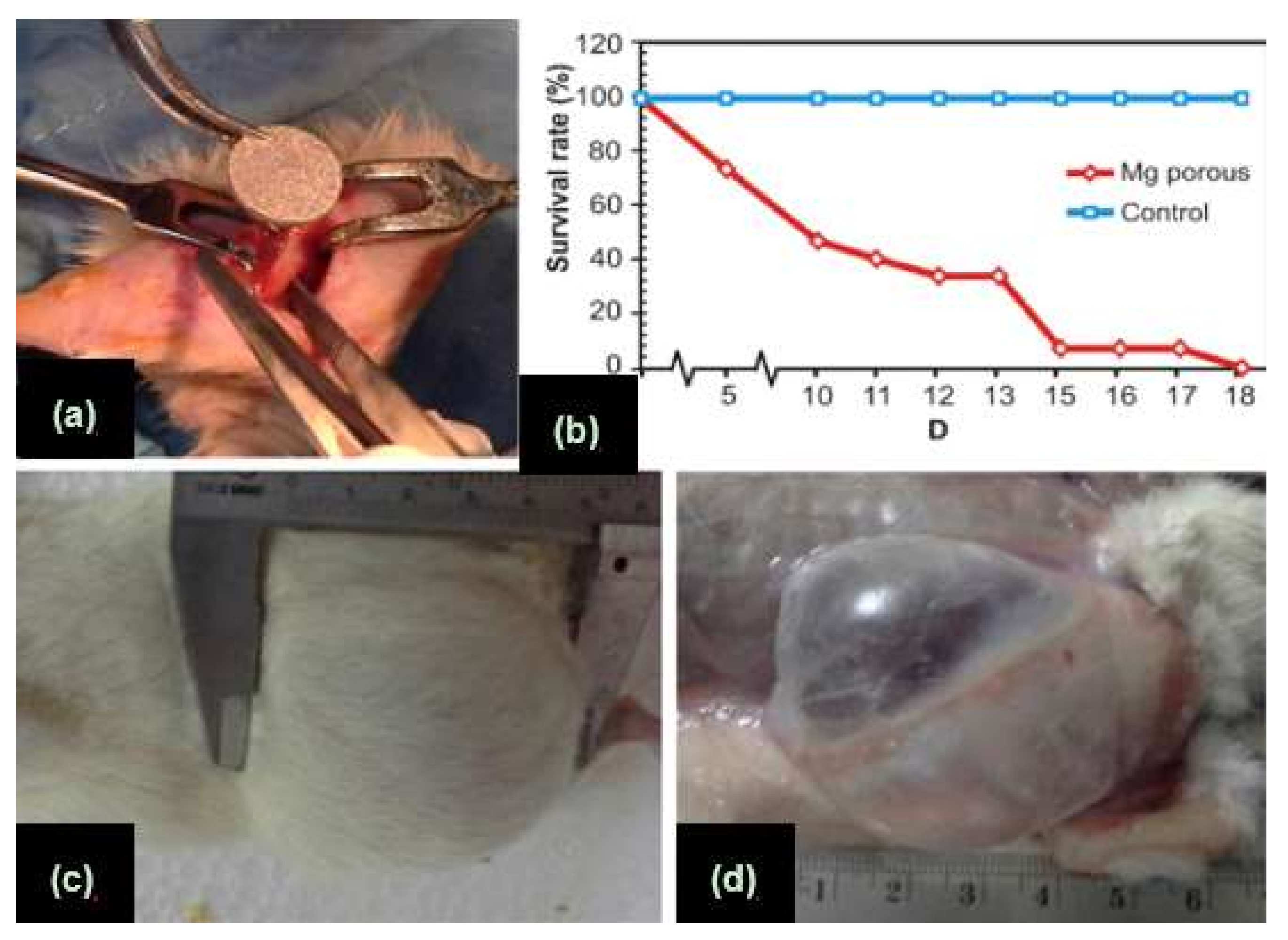
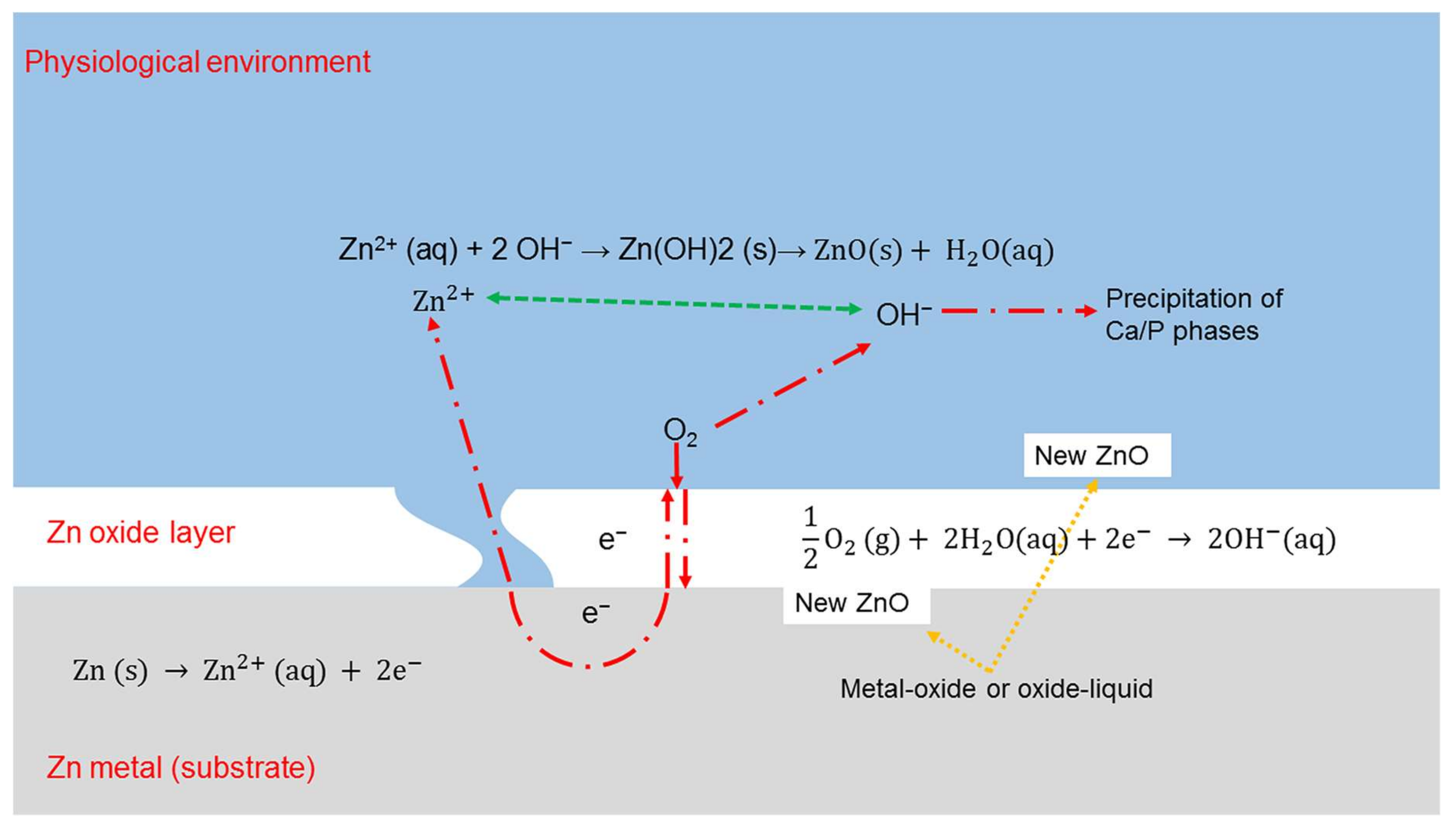
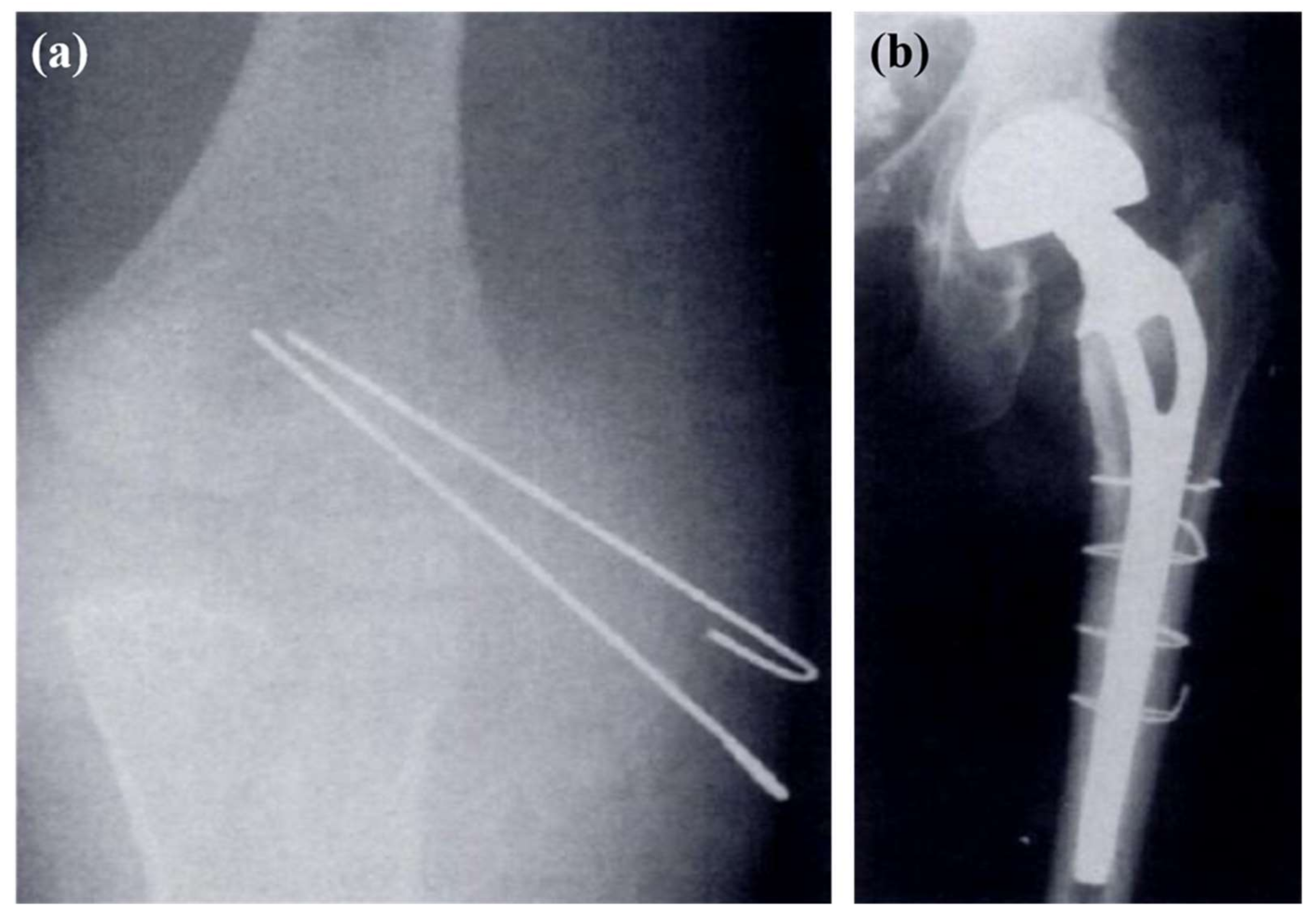
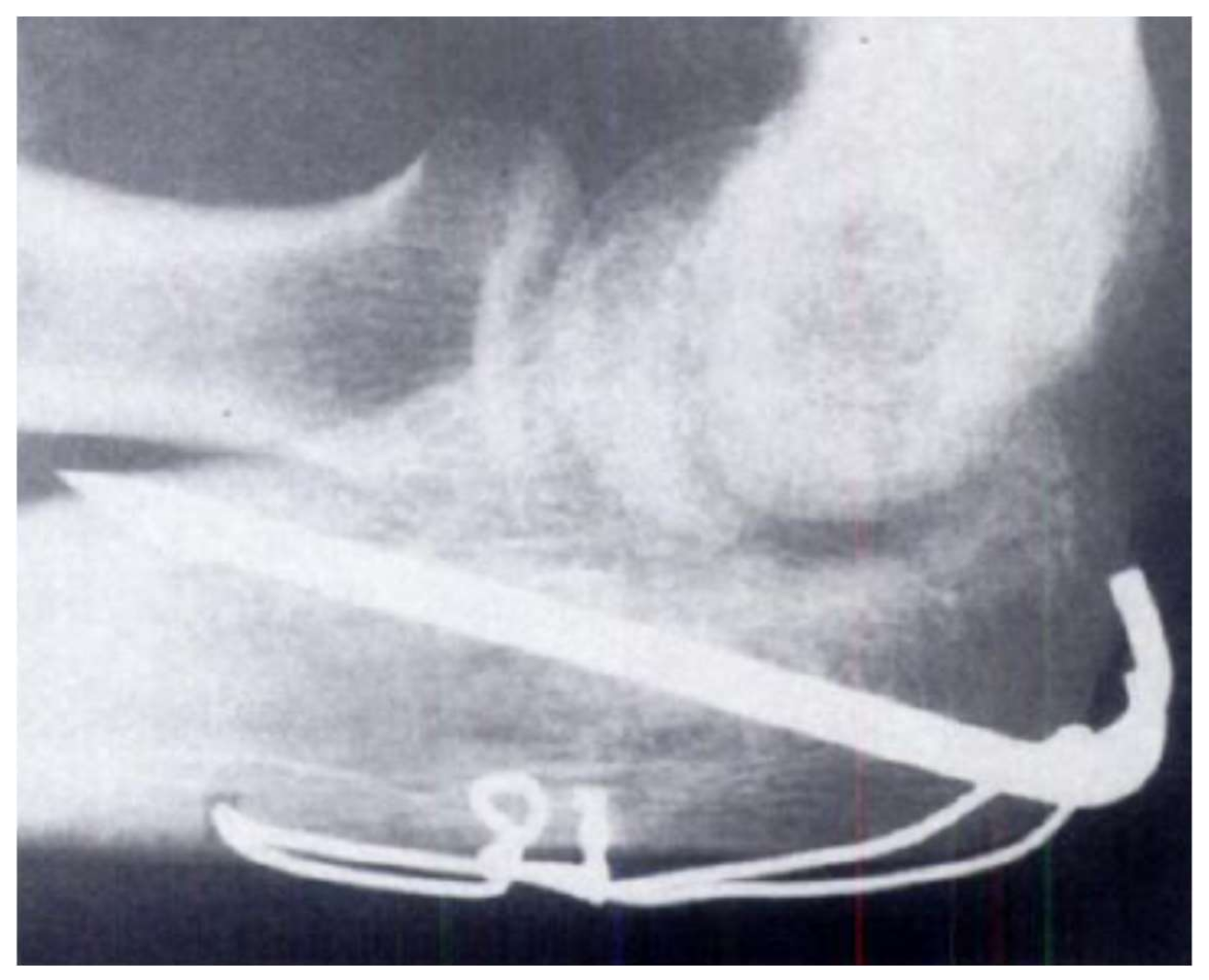
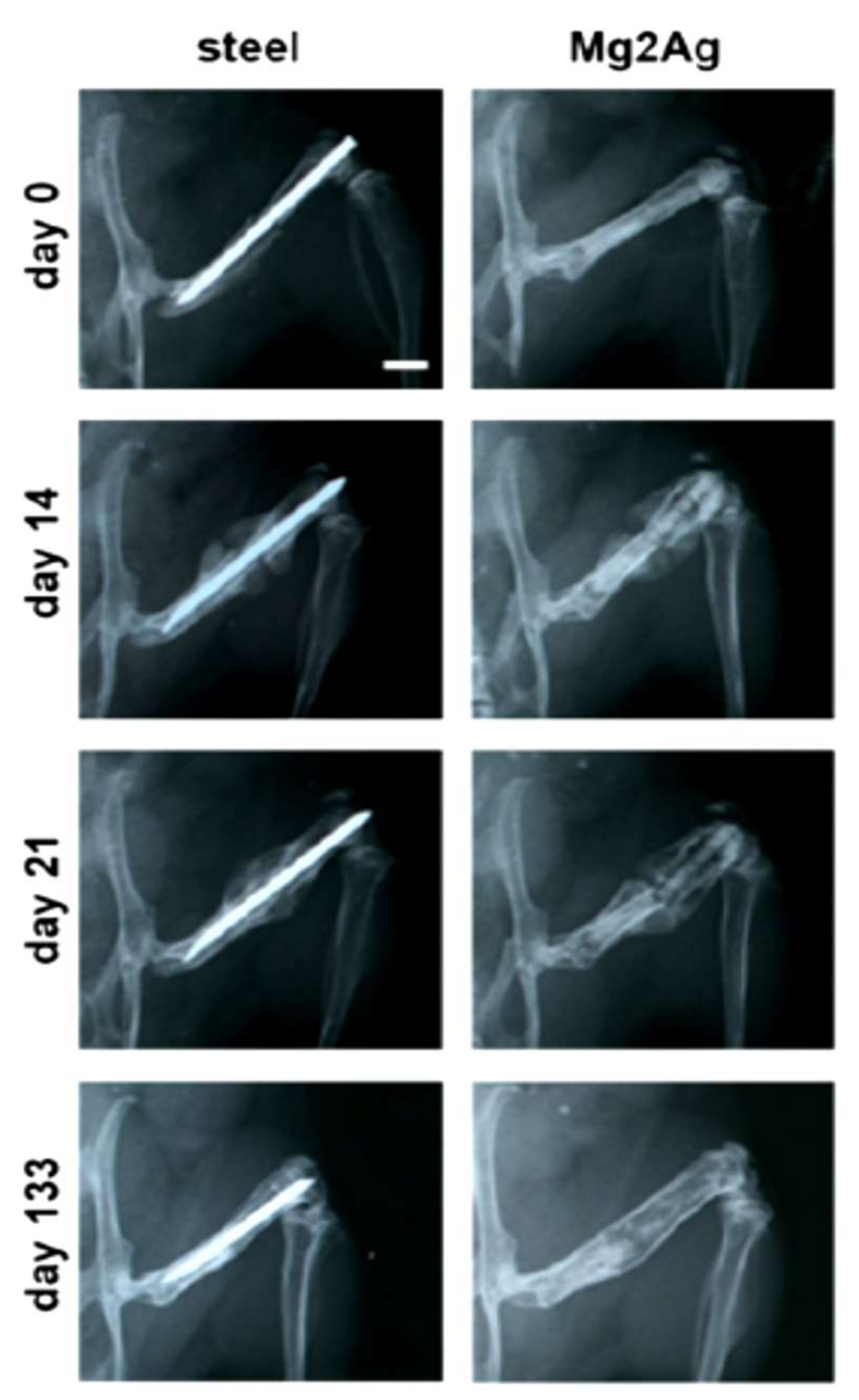
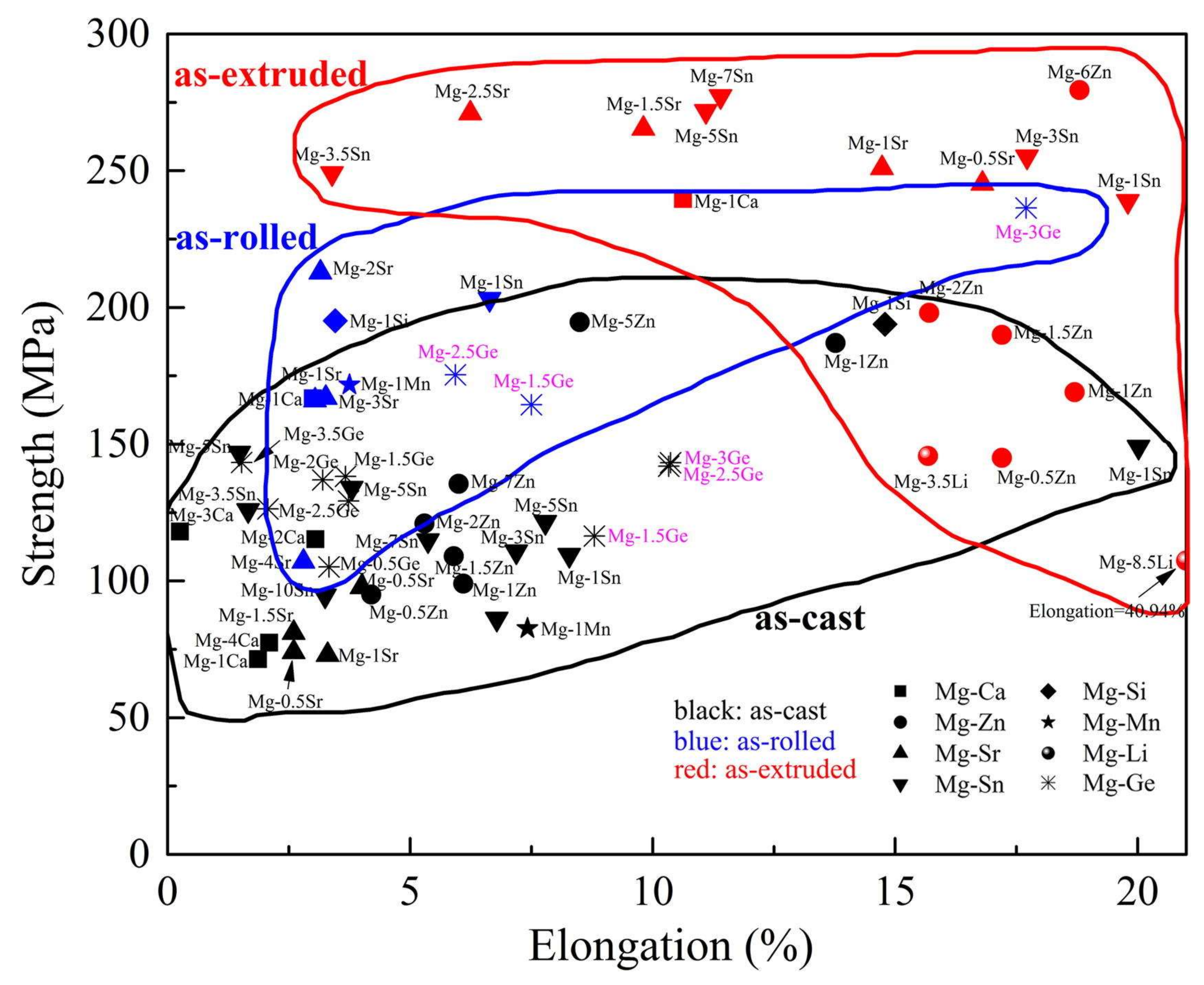


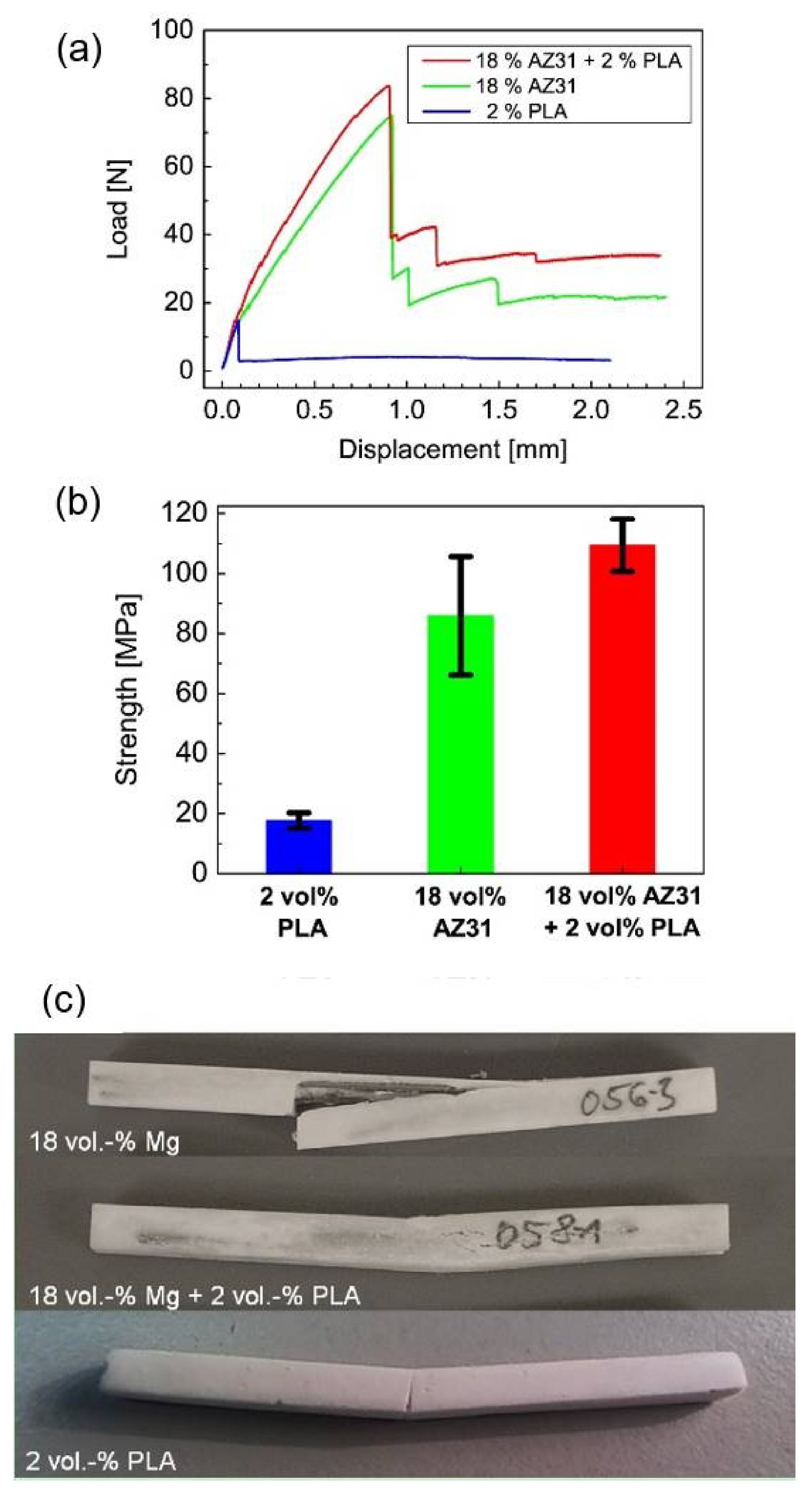
| Tissue/Material | Young’s Modulus, E (GPa) | Yield Tensile Strength (MPa) | Ultimate Tensile Strength (MPa) | Density (g/cm3) |
|---|---|---|---|---|
| Cortical bone | 5–23 | 104.9–114.3 | 35–283 | 1.8–2.0 |
| Pure Mg, wrought | 41–45 | 100 | 180 | 1.74 |
| Pure Zn, as cast | 90 | 10 | 20 | 7.13 |
| Pure Fe | 211.4 | 50 | 540 | 7.87 |
| Ti-6Al-4V, as cast | 114 | 760–880 | 830–1025 | 4.43 |
| 316L stainless steel | 193 | 200–300 | 450–650 | 8 |
| Co-Cr alloy | 240 | 500–1500 | 900–1540 | 8.3 |
| Chemical Composition | Alloy | ASTM Number | Application |
|---|---|---|---|
| Fe-18Cr-14Ni-2.5Mo (C < 0.03) | 316 L | F138-08/F139-08/F1350-08 | Kirschner wires, cerclage wires |
| Fe-19Cr-10Ni-2Mn | 302 | A313/F899 | guidewires |
| Fe-20Cr-10.5Ni-2Mn | - | A313/F899 | Guidewires, orthodontic wires |
| Fe-20Cr-10.5Ni-2Mn (C < 0.03) | - | A580 | Guidewires, Orthodontic wires |
| 35Co-35Ni-20Cr-10Mo (Ti = 0.7) | MP35N | F562 | Orthopedic wires |
| 35Co-35Ni-10Mo (Ti = 0.01) | 35NLT | F562 | Orthopedic wires |
| 40Co-20Cr-16Fe-15Ni-7Mo | Elgiloy | F1058–08 | Surgical wires |
| Ti-6Al-4V | - | F136 | Kirschner-wires |
| Ti-Ni | Nitinol | F2063 | Archwires, Kirschner-wires |
| Base Metal | Alloy | Wire Diameter (mm) | Fabrication Process | Application | Type of Biological Experiments | Published Time |
|---|---|---|---|---|---|---|
| Mg | ZEK100 [125] | 0.3–0.5 | Hot extrusion | Suture | - | 2010 |
| MgCa0.8 [125] | 0.3–0.5 | Hot extrusion | Suture | - | 2010 | |
| AL36 [125] | 0.3–0.5 | Hot extrusion | Suture | - | 2010 | |
| AX30 [125] | 0.3–0.5 | Hot extrusion | Suture | - | 2010 | |
| ZEK100 [126] | 0.274 m–0.822 p | Hot extrusion + Cold drawing + Intermediate annealing | Suture | - | 2011 | |
| MgCa0.8 [126] | 0.274 m–0.822 p | Hot extrusion + Cold drawing + Intermediate annealing | Suture | - | 2011 | |
| AL36 [126] | 0.274 m–0.822 p | Hot extrusion + Cold drawing + Intermediate annealing | Suture | - | 2011 | |
| AX30 [126] | 0.274 m–0.822 p | Hot extrusion + Cold drawing + Intermediate annealing | Suture | - | 2011 | |
| Mg-4Gd-0.4Zn [127] | 0.3 | Hot extrusion + Cold drawing + Intermediate annealing | Suture | in vitro | 2014 | |
| Mg-4Y-0.4Zn [127] | 0.3 | Hot extrusion + Cold drawing + Intermediate annealing | Suture | in vitro | 2014 | |
| Mg-4Nd-0.4Zn [127] | 0.3 | Hot extrusion + Cold drawing + Intermediate annealing | Suture | in vitro | 2014 | |
| Mg2Ag [113] | 0.8 | Hot extrusion + Cold drawing + Intermediate annealing | Guide wire-thin pin | in vitro-in vivo (mouse) | 2016 | |
| Mg3Ge [114] | 2.2 | Hot rolling + Machining | Guide wire-thin pin | in vitro-in vivo (rabbit) | 2017 | |
| Mg-Zn-Mn [115] | 1.6 | Hot extrusion | Guide wire-thin pin | in vitro-in vivo (mouse) | 2017 | |
| Fe | Fe–10Mn–1Pd [48] | 1.6 | Casting + Machining | Guide wire-thin pin | in vitro-in vivo (rat) | 2014 |
| Fe–21Mn–0.7C–1Pd [48] | 1.6 | Casting + Machining | Guide wire-thin pin | in vitro-in vivo (rat) | 2014 |
| Alloying Element | Advantages | Disadvantages |
|---|---|---|
| Al | Increase the strength and ductility and decrease the corrosion rate [26]. | Neurotoxicity and accumulation in bone [87]. |
| Ca | Most abundant mineral and mainly stored in bone and teeth; activator or stabilizer of enzymes [87]. | Calcium metabolism disorder; kidney stones; increasing corrosion rate [97]. |
| Cu | Increase the ductility [97]. | Neurodegenerative diseases including Alzheimer’s, Menkes, and Wilson disease; accelerate corrosion and decrease the strength [26]. |
| Zn | Essential trace element; appear in all enzyme classes; increase the strength and improve corrosion resistance [87]. | Euro toxic and hinder bone development at higher concentration [87]. |
| Mn | Improve corrosion resistance, strength and ductility; essential trace element; activator of enzyme; Mn deficiency is related to osteoporosis, diabetes mellitus, and atherosclerosis [87]. | Excessive Mn results in neurotoxicity [87]. |
| Si | Cross linking agent of connective tissue basement membrane structures; necessary for bone calcification [87]. | Excessive SiO2 causes lung diseases [87]; decreasing the ductility and increasing corrosion rate [97]. |
| Li | Used in the treatment of manic-depressive psychoses [87]. | Decrease the strength and increasing corrosion rate [97]. |
| Zr | Increase the strength and ductility [97]. | High concentration in liver and gall bladder [87]. |
| RE | Compound of drugs for treatment of cancer; improve the mechanical properties and corrosion resistance [87]. | Accumulation in bone and liver [87]. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asgari, M.; Hang, R.; Wang, C.; Yu, Z.; Li, Z.; Xiao, Y. Biodegradable Metallic Wires in Dental and Orthopedic Applications: A Review. Metals 2018, 8, 212. https://doi.org/10.3390/met8040212
Asgari M, Hang R, Wang C, Yu Z, Li Z, Xiao Y. Biodegradable Metallic Wires in Dental and Orthopedic Applications: A Review. Metals. 2018; 8(4):212. https://doi.org/10.3390/met8040212
Chicago/Turabian StyleAsgari, Mohammad, Ruiqiang Hang, Chang Wang, Zhentao Yu, Zhiyong Li, and Yin Xiao. 2018. "Biodegradable Metallic Wires in Dental and Orthopedic Applications: A Review" Metals 8, no. 4: 212. https://doi.org/10.3390/met8040212
APA StyleAsgari, M., Hang, R., Wang, C., Yu, Z., Li, Z., & Xiao, Y. (2018). Biodegradable Metallic Wires in Dental and Orthopedic Applications: A Review. Metals, 8(4), 212. https://doi.org/10.3390/met8040212






