Abstract
CuO-ZnO-Al2O3 catalysts are designed for low-temperature conversion in the process of hydrogen and ammonia synthesis gas production. This paper presents the results of research into the recovery of copper and zinc from spent catalysts using pyrometallurgical and hydrometallurgical methods. Under reducing conditions, at high temperature, having appropriately selected the composition of the slag, more than 66% of the copper can be extracted in metallic form, and about 70% of zinc in the form of ZnO from this material. Hydrometallurgical processing of the catalysts was carried out using two leaching solutions: alkaline and acidic. Almost 62% of the zinc contained in the catalysts was leached to the alkaline solution, and about 98% of the copper was leached to the acidic solution. After the hydrometallurgical treatment of the catalysts, an insoluble residue was also obtained in the form of pure ZnAl2O4. This compound can be reused to produce catalysts, or it can be processed under reducing conditions at high temperature to recover zinc. The recovery of zinc and copper from such a material is consistent with the policy of sustainable development, and helps to reduce the environmental load of stored wastes.
1. Introduction
Spent CuO-ZnO-Al2O3 catalysts are very important secondary resources for metal recovery and could be highly usable for copper and zinc recycling [1]. This type of catalyst is used in low-temperature processes of carbon monoxide conversion with steam and to obtain hydrogen as well as a synthesis gas to produce ammonia or methanol [2,3,4,5]. Manufacturers of catalysts define the content of the basic components in new products as follows: CuO—min 50%, ZnO—min 25%. Multiple variations of these are available on the market. They are different in terms of their Cu, Zn and Al oxide content. The copper to zinc mass ratio in industrial catalysts manufactured using the co-precipitation method is usually 7:3 [6]. The presence of spinel structures is a feature specific to the catalysts that are analyzed in the paper. In the literature, CuAl2O4 spinels on the surface of the catalysts and the ZnAl2O4 stoichiometry spinel structures throughout their volume are the most frequently reported [7,8]. X-ray diffraction studies in the literature [9] indicate that the ZnAl2O4 compound is present at a temperature of 1100 °C. Therefore, processing of the spent catalyst with the infusible spinel structures present in them, and the complete recovery of zinc from this type of material, can be difficult using the pyrometallurgical method. Due to the toxic metal content (Zn, Cu), these materials can be considered to pollute the natural environment. In Poland, the volume of CuO-ZnO-Al2O3 waste catalysts is estimated at around 2000 tons per year. The current method for their recycling consists of pyrometallurgical processing together with other waste containing zinc and/or copper. In the literature, there is only quite scarce information related to the processing of this type of catalysts. This is limited to the patents [10,11,12] describing the hydrometallurgical processing of spent CuO-ZnO-Al2O3 catalysts, which consists of leaching the spent catalysts in a solution of nitric acid after high-temperature roasting or leaching in ammonia solutions. From the resulting solutions, copper and zinc compounds can be selectively precipitated and then recycled for the production of catalysts. In this work, it was decided to analyze the use of pyro and hydrometallurgical methods for the processing of catalysts in terms of maximum recovery of metals and minimization of waste.
2. Properties of Examined Material
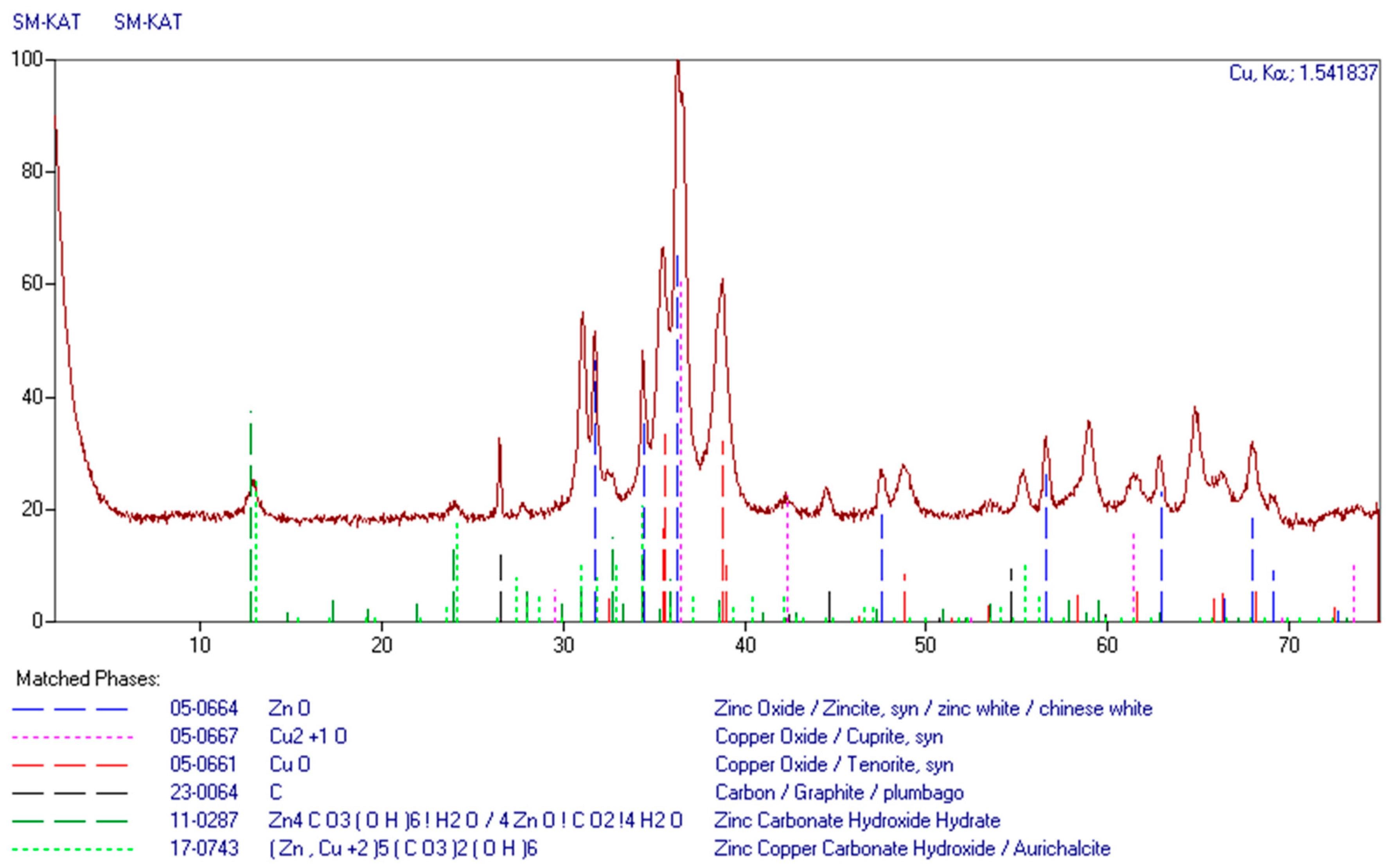
The material tested in this work comprises spent CuO-ZnO-Al2O3 catalysts available on the Polish market. These occur in the form of identical rolls with a diameter of 4.5 mm and a height of 3.5 mm. The chemical analysis (AAS) of the examined samples of spent catalysts indicates different content of copper and zinc in the materials analyzed. With the detailed chemical analysis of the catalyst sample selected to be examined, the following results were obtained: 35.1% of Cu, 29.9% of Zn and 9.2% of Al. A sample of the material was also examined using X-ray diffraction (XRD) and scanning electron microscope observations combined with qualitative energy dispersion chemical analysis (EDS). The results of the phase analysis are shown in Figure 1. CuO and ZnO oxides are the main phase components. Additionally, small quantities of Cu2O and hydroxy-carbonate complexes of zinc and copper are present.
Figure 1.
XRD pattern of the grounded catalyst sample.
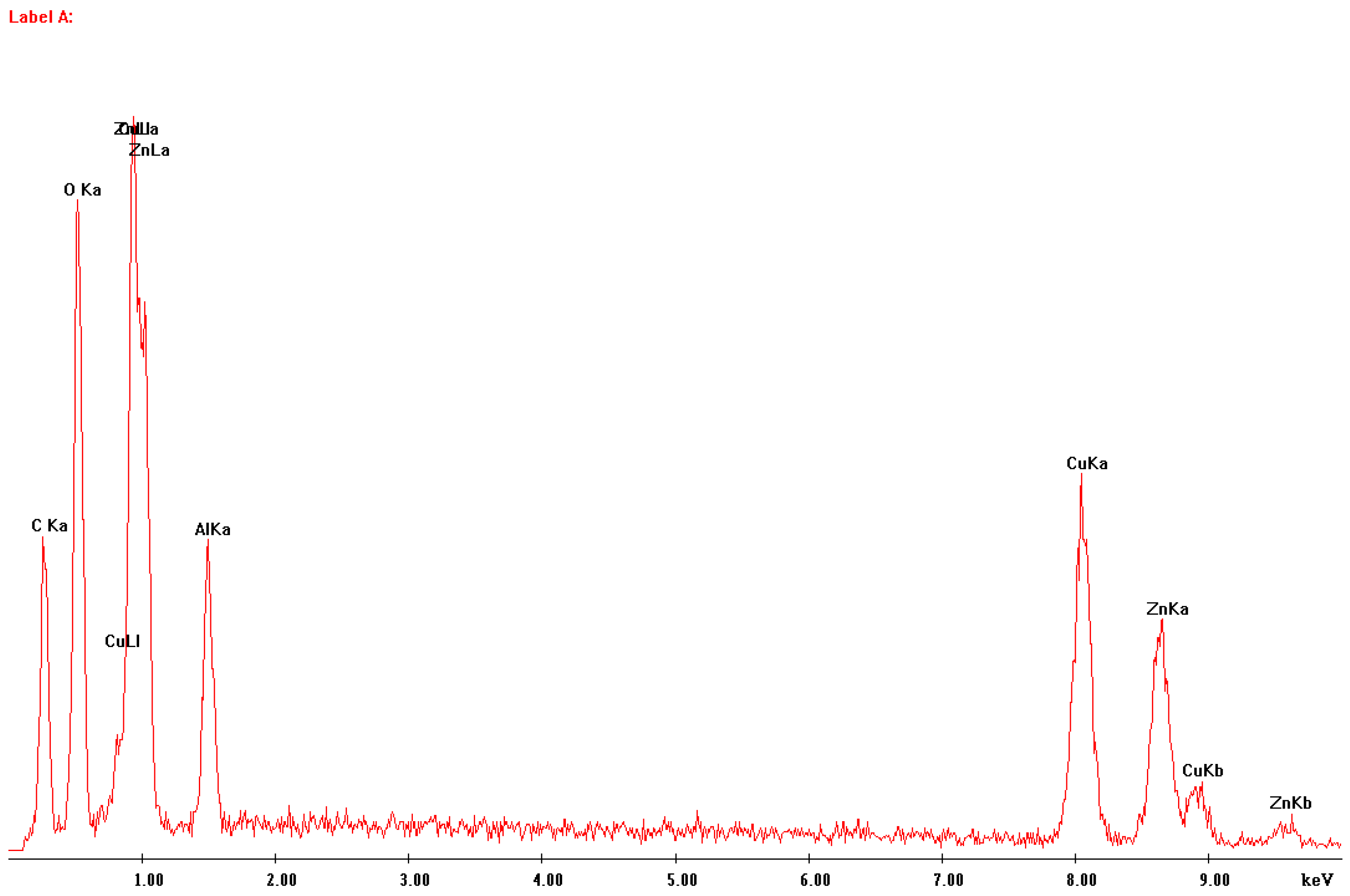
In Figure 2 and Figure 3, respectively, an image of a sample of fragmented catalyst material and the EDS analysis of the area presented are shown. The elements identified are copper, zinc, aluminum, carbon and oxygen. This is only a confirmation of the phase analysis.

Figure 2.
The microscopic image of the sample of the fragmented catalyst material.
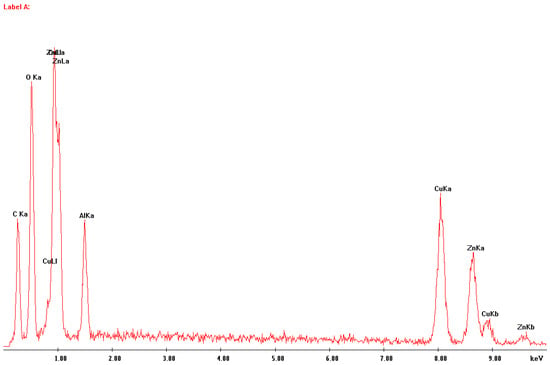
Figure 3.
EDS analysis of the area of the sample seen in Figure 2.
3. Pyrometallurgical Processing of Spent Catalysts
With information about the content of the most valuable components in the material having been obtained, it was decided to perform the test melting in order to extract copper and zinc in the form of zinc oxide from spent catalysts and to optimize the process for maximum yields of Cu and ZnO, as mentioned above. Bearing in mind the oxidic nature of catalysts, in order to lower the melting point (softening point) of the slag created and, if possible, to decrease its viscosity, it was decided to carry out the recovery process under reducing conditions and to apply slag-forming additives (e.g., CaO, SiO2, Na2O). Therefore, in order to determine the amount of additives that would ensure the lowest melting point of the slag, the relevant binary and ternary systems were analyzed. In the case of a binary ZnO-SiO2 system, the liquid phase occurs at a temperature of 1432 °C, with a content of about 52% ZnO by weight [13]; while in the Na2O-SiO2 system, the liquid phase occurs in a wide range of concentrations and temperatures close to 1000 °C [14]. Therefore, it can be assumed that in the Na2O-ZnO-SiO2 ternary system, compositions are likely in which the melting point of the respective ternary phase will be low enough to melt the catalysts processed. The above hypothesis is confirmed by reports in the literature [15], which indicate that once the concentration level of (weight percent) 21.5% Na2O, 12.0% ZnO, 66.5% SiO2 in this system is reached, the minimum melting point is 680 °C. On the basis of the available information about the CaO-ZnO-SiO2 and CaO-ZnO-Al2O3-SiO2 systems [16,17], it was assumed that the lowest melting points could be obtained when the relevant components were at similar concentration levels.
3.1. Experimental Procedure of Pyrometallurgical Processing
The catalysts were processed in an induction furnace, operating at a temperature range between 1100–1300 °C. This temperature was to ensure maximum stripping of zinc and the obtaining of liquid copper by reducing its oxide. The amount of coal for the reduction of CuO and ZnO was determined assuming the formation of CO and CO2. As a result, the amount of reductant added varies between 6 and 12 wt %. For further studies, the mean value was taken and increased by 10% due to the ash content in coal. Initial testing made it possible to determine that at 10% of added reductant, the degree of zinc stripping was at a level of approx. 65%. The remainder of the zinc goes into the slag and metal phase. It should be noted that tests were carried out in a graphite crucible, which additionally improved the reduction conditions. Based on these results, it was possible to determine the test plan to optimize the amount of slag-forming additives.
Four variants of laboratory tests were performed. The tests were different from each other in terms of quantity of slag-forming components added. The optimal process parameters were sought in order to obtain the lowest melting points of slag, and thereby to minimize the loss of metals extracted from the catalysts to slag. 100 g of uncrushed catalysts were used for each of the tests. The weights of copper and zinc contained in these were 35.1 and 29.9 g, respectively. Reducing conditions of the process were secured by adding 10 g of coal and by the fact that the melting was carried out in a graphite crucible. Once the melting was complete, the liquid products contained in the crucible were cooled, separated, and weighed, and the chemical analyses of the materials obtained were performed.
3.2. Results and Discussion of Pyrometallurgical Tests
3.2.1. Test No. 1
100 g of catalysts were mixed with 10 g of carbon, 50 g of SiO2 and 15 g of CaO. The whole feed was placed in a graphite crucible and heated until a temperature of 1300 °C was reached. At the beginning, intensive reduction of ZnO to Zn was observed, and the metal was lifted in the gas phase and re-oxidized. After about 45 min, this process had definitely halted (no emission of ZnO white films), and hence the decision was taken to terminate the test at this time. After cooling the crucible, its contents were separated into three fractions, and 26 g of metal, 62 g of glassy slag and about 17.5 g of unmelted fine fraction were yielded. This phase is, most likely, unreacted slag-forming components and unmelted ZnAl2O4 spinel structures. Additionally, in the experimental system, it was impossible to identify the amount of dust produced. It should be added that in the slag phase, no sedimented tiny metallic copper inclusions were visible.
3.2.2. Test No. 2
100 g of catalysts, 10 g of carbon, 60 g of SiO2 and 20 g of NaOH were the feed for smelting. The temperature of the process was 1300 °C, and the duration was 50 min. Like in the previous test, for the first 45 min of the process, the stripping of the zinc in the form of ZnO was very intense. Upon its completion, after product cooling and separation, 25 g of metal, 95 g of glassy slag and 7.5 g of free-flowing phase (slag-forming components and spinels) were found. Additionally, in this test, metallic copper inclusions were seen in the slag.
3.2.3. Test No. 3
100 g of catalysts, 10 g of carbon, 31 g of Na2CO3 and 55.4 g of SiO2 were the feed for the process. The feed was melted at a temperature of 1250 °C, and the melting time was 90 min. After this period, stripping of zinc drastically decreased, and hence the decision to terminate the smelting was taken. 25 g of metal and 107 g of glassy slag with minor copper inclusions were yielded.
3.2.4. Test No. 4
100 g of catalysts, 10 g of carbon, 16 g of Na2CO3 and 28 g of SiO2 were melted. The melting time was 90 min and the process temperature was 1250 °C. 20 g of metal and 61 g of glassy slag with a certain amount of copper drops were yielded.
The smelting products, namely slag and metallic alloy, were subjects of chemical analysis for the content of copper and zinc. The summary of the test results in Table 1 takes into account the fact that the unbalanced portion of zinc is transferred to the dust phase.

Table 1.
List of parameters for processing the spent catalysts conducted at laboratory scale according to smelting variants.
Slag-forming additives have a significant effect on the recovery of Cu to alloy and Zn to dust. The use of CaO (variant I) results in the best recovery of copper and zinc. The use of other additives (variants II and III) results in similar effects with a much larger quantity of waste slag produced. In the last variant of the remelting (IV), a small addition of Na2CO2 was used and an unsatisfactory degree of copper extraction to metallic alloy was obtained. However, the results of the pyrometallurgical test of catalyst recycling do not provide grounds for optimism. The too-low yield of copper may cause the processing to be less cost-effective. This is due to the problem of obtaining a low-viscosity slag. It is probable that the presence of zinc aluminate is responsible for the high viscosity of the slags. Correction of the slag composition results in the formation of a large amount of slag, and even at a lower copper content, total losses are significant.
4. Processing of Catalysts Using the Hydrometallurgical Method
4.1. Procedure of Hydrometallurgical Tests
The results of using the described pyrometallurgical method to recycle the spent catalysts are not fully satisfactory. Therefore, the decision was taken to use a hydrometallurgical method for their processing. With knowledge about the structure of catalysts, their chemical composition and their phase composition, as well as being familiar with how they are produced [6], an innovative method for processing them was developed. Zinc aluminate (ZnAl2O4), which is present in the catalysts, is a compound highly resistant to both acids and alkalines [18]. Therefore, it has been recognized that after leaching, copper oxides and zinc oxides will be left as insoluble residue. First, catalysts in the form of pellets were fragmented to reach sizes of less than 90 μm. In order to separate zinc and copper, zinc oxide and copper oxide leaching processes were selectively carried out, consisting of two stages:
- Leaching in NaOH solution (temperature 75 °C, process duration 120 min, NaOH concentration = 200 g/dm3, l/s = 10),
- Leaching in H2SO4 solution (temperature 60 °C, process duration 120 min, H2SO4 concentration = 180 g/dm3, l/s = 10).
The process conditions were adjusted based on previous experience in alkaline leaching [19], and pilot tests. After each leaching process, the slurry was filtered (filter Munktell & Filtrak, Stockholm, Sweden, type 392) to separate the deposit. The filtration process is difficult because the deposit consists of very fine grains. During the leaching using NaOH, only zinc was transferred to the solution. In fact, after the acidic leaching, the solution contained just copper. The amount of zinc in the solution was 100 to 150 times less than the amount of copper. Additionally, in order to facilitate transferring the copper to the solution, as acidic leaching was conducted, small amounts of hydrogen peroxide solution were added. A small quantity of this may in fact be present in metallic form.
4.2. Results and Discussion of Hydrometallurgical Tests
The above-described procedure made it possible to obtain the following products from 100 g of recycled catalysts (information based on AAS analysis):
- Zn solution—0.8 dm3 (Zn-23 g/dm3),
- Cu solution—0.8 dm3 (Cu-43 g/dm3),
- ZnAl2O4 deposit in the amount of 33 g.
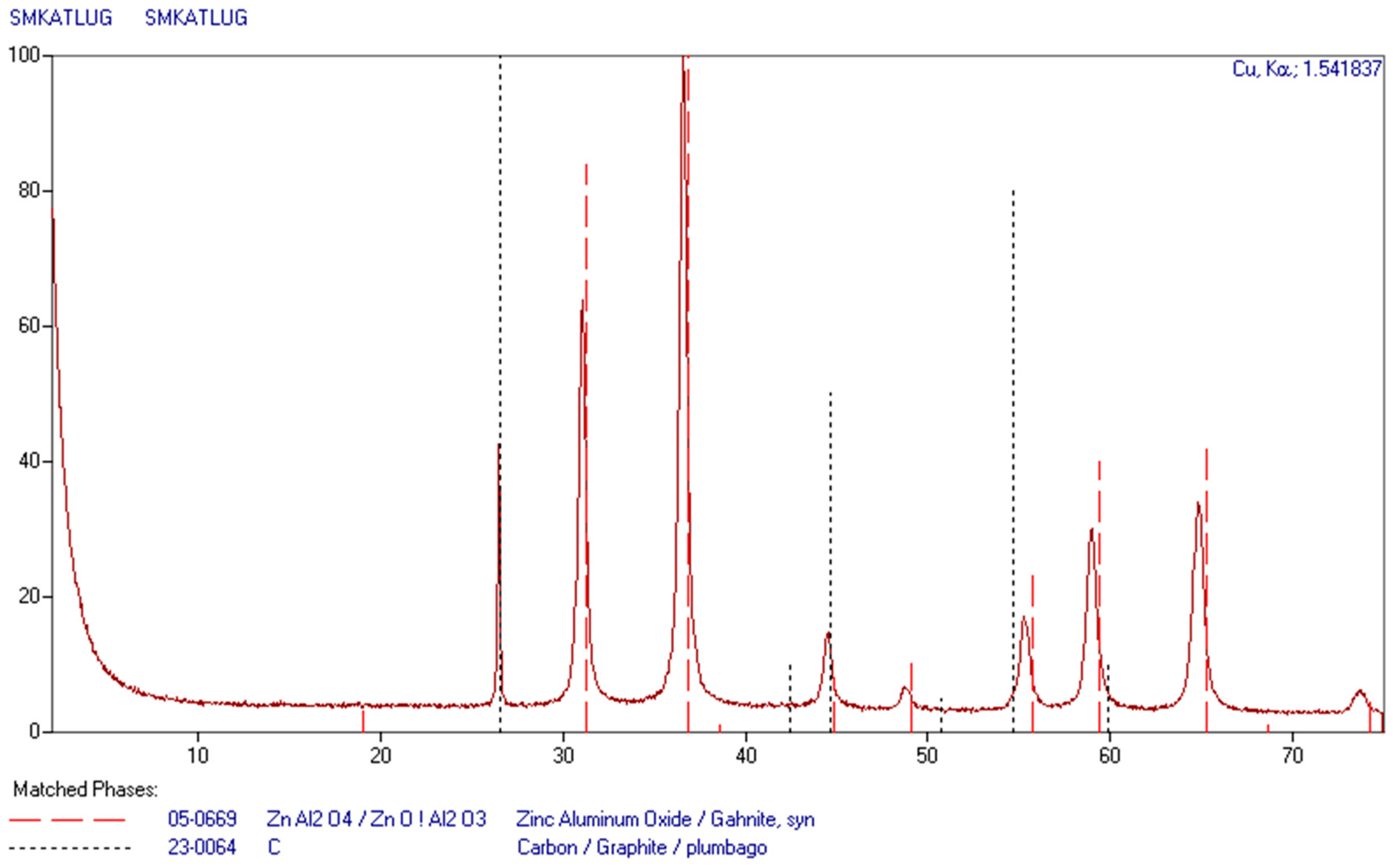
After leaching, the solutions still contain highly concentrated leaching agent, and should be returned to the initial leaching of subsequent batches. In order to recover as much metal as possible, the final leaching must be carried out using highly concentrated leaching agent. The zinc aluminate deposit was of dark gray color, since it contained a certain amount of carbon. An attempt to burn it out (600 °C, air atmosphere) resulted in weight reduction by about 10%, and the color was changed to light gray. The sample obtained in this way was analyzed using X-ray phase analysis. The results are presented in Figure 4.
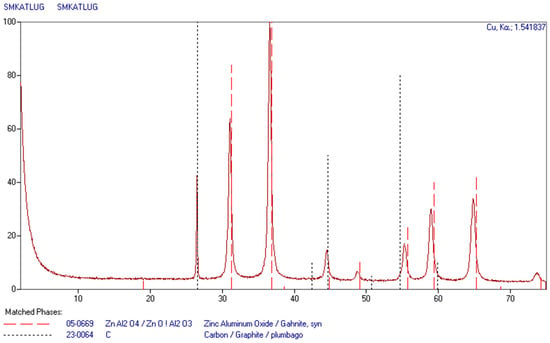
Figure 4.
XRD pattern of zinc aluminate obtained from recycling catalysts.
Results of the phase analysis indicate the presence of only zinc aluminate and carbon. The presence of carbon results from the fact that during leaching, coal does not pass into the solution and accumulates in the residue.
The balance of the processing performed is as follows (100 g of catalysts):
- Amount of zinc in alkaline solution—18.5 g
- Amount of copper in acidic solution—34.5 g
- Zinc aluminate—30 g
- Carbon—3 g
Material balance and analysis of the solutions made it possible to determine the yield of copper in solution at the level of 98%. The yield of zinc to the alkaline solution is 61.9%. Because the residue after leaching is actually pure ZnAl2O4, it can be treated as a product of the process. In this case, zinc yield increases to 97.5%, and the yield of aluminum is 96%.
5. Conclusions
Laboratory tests carried out indicate that the pyrometallurgical method can be used to recover copper and zinc contained in the spent catalysts processed. However, after conducting the tests, some amounts of the alloy in the slag were noted in the form of small. This may be a result of the high viscosity of the slag and a result of the difficult sedimentation conditions under the test conditions. The process should be carried out at temperatures above 1200 °C, and a reductant should be used in the form of coal in an amount of about 10% by weight of catalysts. Liquid copper and ZnO in the form of dust are products of the process. Silica and CaO or NaOH or Na2CO3 are an indispensable technological additive in processing catalysts for slag adjustment. This makes it possible to obtain a glassy slag and to minimize copper losses in the process. Laboratory test conditions made it possible to carry out the process of copper recovery with a yield of up to 66%. The Zn yield for dust was estimated based on the amounts of zinc contained in the alloy obtained and in slag. It can be assumed that under proper process conditions (reducing atmosphere, ~1300 °C, enough time for Zn stripping), transferring to dust more than 70% of zinc contained in the spent catalysts is very possible.
Hydrometallurgical processing based on selective leaching makes it possible to accurately separate the components of spent catalysts. More than 96% degree of metal recovery is reached. In the case of zinc and aluminum, this value is relative to the overall yield. It also includes the content of these metals in the leach residue. This residue may be the product of the process, because it actually contains pure ZnAl2O4. Solutions obtained as a result of applying acidic and alkaline leaching make it possible to perform the selective extraction of the Cu and Zn they contain. These metals can be recovered from solutions in the form of compounds by precipitation or in a metallic form by electrolysis. The residue (mainly ZnAl2O4) left after leaching may be returned to produce new catalysts or may be thermally processed to recover the zinc it contains.
Taking into account the ecological side of the proposed processes, it should be noted that during the pyrometallurgical processing, a waste slag is formed with a relatively high content of zinc and copper. For this reason, the waste slag cannot be considered neutral for the natural environment. Hydrometallurgical conversion of catalysts leads to their waste-free management, and thus is completely environmentally friendly. The optimization of the leaching process parameters is the way of developing the proposed method.
Acknowledgments
This paper is supported by the Ministry of Science and Higher Education (Grant No. 11.11.180.726).
Author Contributions
All parts of the work were done jointly by both authors with the exception of laboratory work. pyrometallurgical tests—Krzysztof Gargul; hydrometallurgical tests—Stanisław Małecki.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Sun, Z.; Xiao, Y.; Agterhuis, H.; Sietsma, J.; Yang, Y. Recycling of metals from urban mines—A strategic evaluation. J. Clean. Prod. 2016, 112, 2977–2987. [Google Scholar] [CrossRef]
- Amphlett, J.; Mann, R.; Weir, R. Hydrogen production by the catalytic steam reforming of methanol: Part 3: Kinetics of methanol decomposition using C18HC catalyst. Can. J. Chem. Eng. 1988, 66, 950–955. [Google Scholar] [CrossRef]
- Agarwal, V.; Patel, S.; Pant, K.K. H2 production by steam reforming of methanol over Cu/ZnO/Al2O3 catalysts: Transient deactivation kinetics modelling. Appl. Catal. A 2005, 279, 155–164. [Google Scholar] [CrossRef]
- Waugh, K.C. Methanol Synthesis. Catal. Lett. 2012, 142, 1153–1166. [Google Scholar] [CrossRef]
- Riaz, A.; Zahedi, G.; Klemes, J.J. A review of cleaner production methods for the manufacture of methanol. J. Clean. Prod. 2013, 57, 19–37. [Google Scholar] [CrossRef]
- Behrens, M.; Studt, F.; Kasatkin, I.; Kühl1, S.; Hävecker, M.; Abild-Pedersen, F.; Zander, S.; Girgsdies, F.; Kurr, P.; Kniep, B.L.; et al. The Active Site of Methanol Synthesis over Cu/ZnO/Al2O3 Industrial Catalysts. Science 2012, 336, 893–897. [Google Scholar] [CrossRef] [PubMed]
- Barroso, M.N.; Gomez, M.F.; Gamboa, J.A.; Arrua, L.A.; Abello, M.C. Preparation and characterization of CuZnAl catalysts by citrate gel process. J. Phys. Chem. Solids 2006, 67, 1583–1589. [Google Scholar] [CrossRef]
- Turco, M.; Bagnasco, G.; Costantino, U.; Marmottini, F.; Montanari, T.; Ramis, G.; Busca, G. Production of hydrogen from oxidative steam reforming of methanol: II. Catalytic activity and reaction mechanism on Cu/ZnO/Al2O3 hydrotalcite-derived catalysts. J. Catal. 2004, 228, 56–65. [Google Scholar] [CrossRef]
- Walerczyk, W.; Zawadzki, M.; Grabowska, H. Glycothermal synthesis and catalytic properties of nanosized Zn1−xCoxAl2O4 (x = 0, 0.5, 1.0) spinels in phenol methylation. Catal. Lett. 2011, 141, 592–601. [Google Scholar] [CrossRef][Green Version]
- Zhou, H.; Huang, Y. Method for Recycling Waste Copper-Based Methanol Waste Catalyst. Patent CN103495426A, 18 January 2014. [Google Scholar]
- Wang, B.; Meng, Y. Recovery Method of Copper and Zinc Oxide from Waste Copper-Zinc Catalyst. Patent CN1258752A, 30 December 1998. [Google Scholar]
- Ling, H.; Liu, J.; Zhang, X.; Xu, X.; Li, Q.; Hu, Z.; Hu, G.; Qiu, C.; Huang, H. Application Method of Waste Copper Based Catalyst to Preparing Catalyst for Preparing Hydrogen from Methanol. Patent CN102125851A, 20 April 2011. [Google Scholar]
- Bunting, E.N. Phase equilibria in the system SiO2-ZnO. J. Am. Ceram. Soc. 1930, 13, 5–10. [Google Scholar] [CrossRef]
- Kracek, F.C. The system sodium oxide-silica. J. Phys. Chem. 1930, 34, 1583–1598. [Google Scholar] [CrossRef]
- Holland, A.E.; Segnit, E.R. The ternary system Na2O-ZnO-SiO2. Aust. J. Chem. 1966, 19, 905–913. [Google Scholar] [CrossRef]
- Segnit, E.R. The System CaO-ZnO-SiO2. J. Am. Ceram. Soc. 1954, 37, 273–277. [Google Scholar] [CrossRef]
- Segnit, E.R. Three planes in the quaternary system CaO-ZnO-Al2O3-SiO2. J. Am. Ceram. Soc. 1962, 45, 600–607. [Google Scholar] [CrossRef]
- Tang, Y.; Shih, K.; Wang, Y.; Chong, T. Zinc stabilization efficiency of aluminate spinel structure and its leaching behaviour. Environ. Sci. Technol. 2011, 45, 10544–10550. [Google Scholar] [CrossRef] [PubMed]
- Gargul, K.; Jarosz, P.; Małecki, S. Alkaline leaching of low zinc content iron-bearing sludges. Arch. Metall. Mater. 2016, 61, 43–50. [Google Scholar] [CrossRef][Green Version]
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).