Enhanced Compressive Property of Al Composite Foams at Elevated Temperatures via Plasma Electrolytic Oxidation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation
2.2. Material Characterization
2.3. Compression Test
3. Results and Discussion
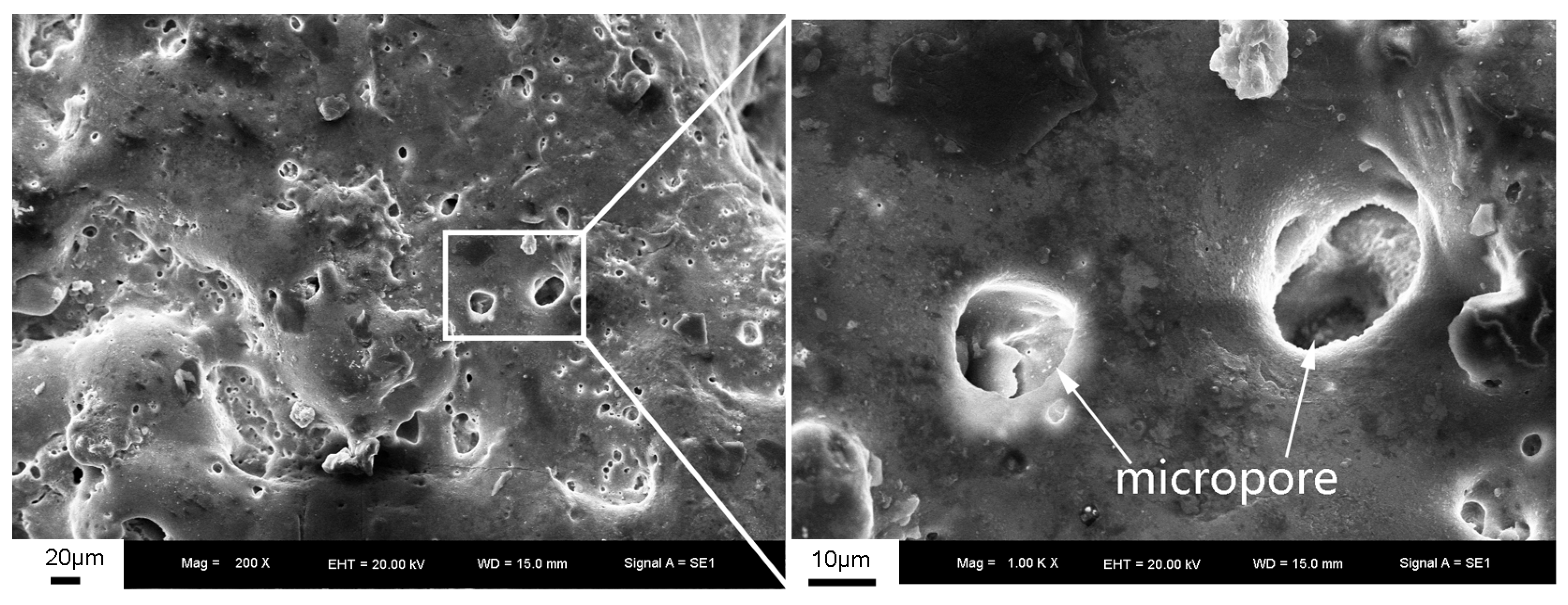
3.1. The Surface Morphology and Thickness of the PEO Coating
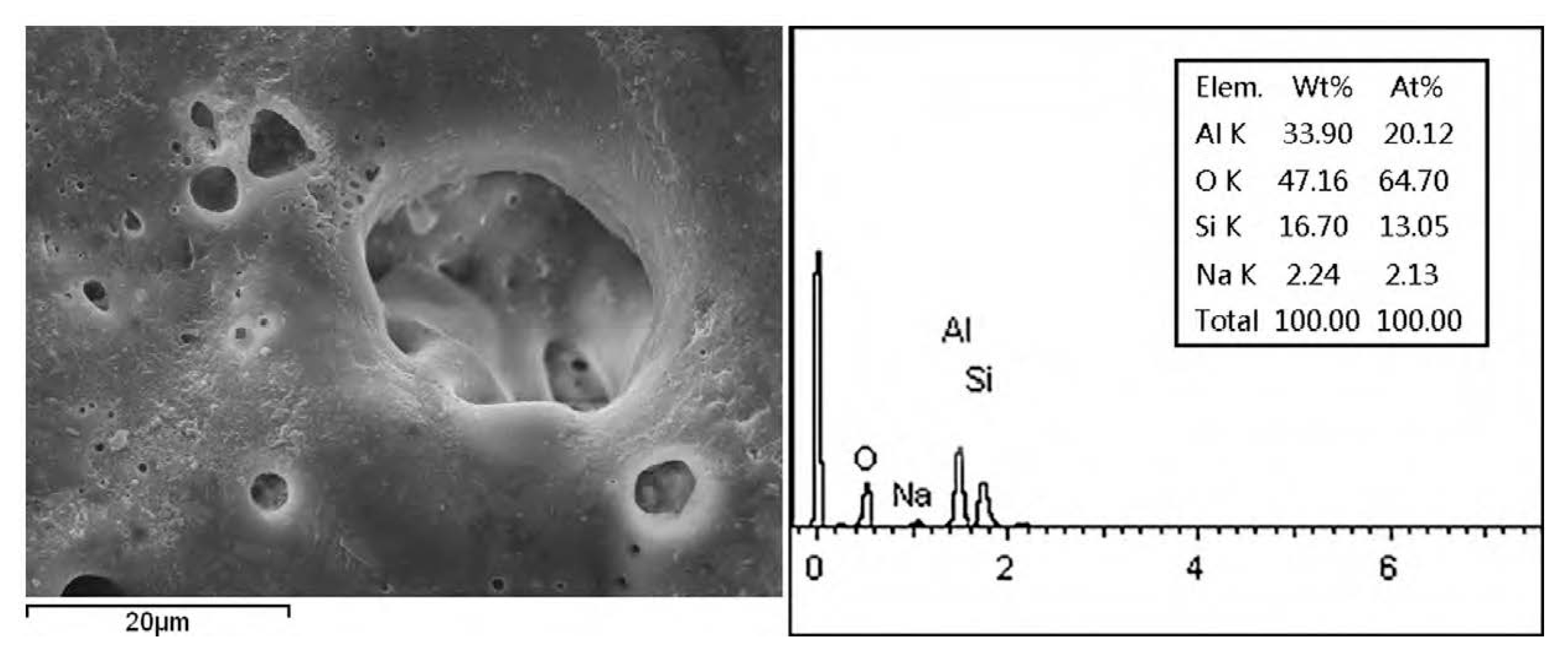
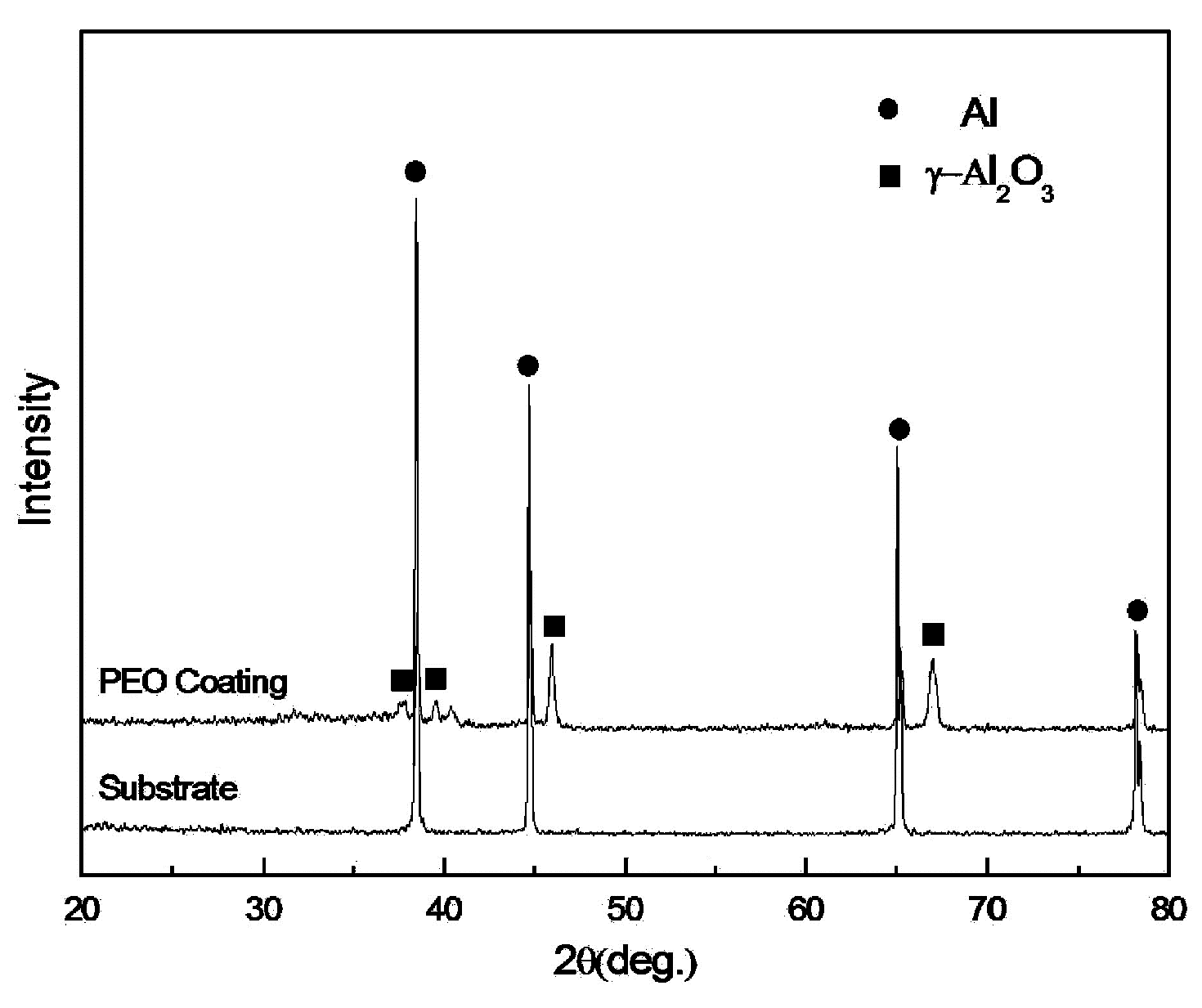
3.2. Element and Phase Composition of the PEO Coating
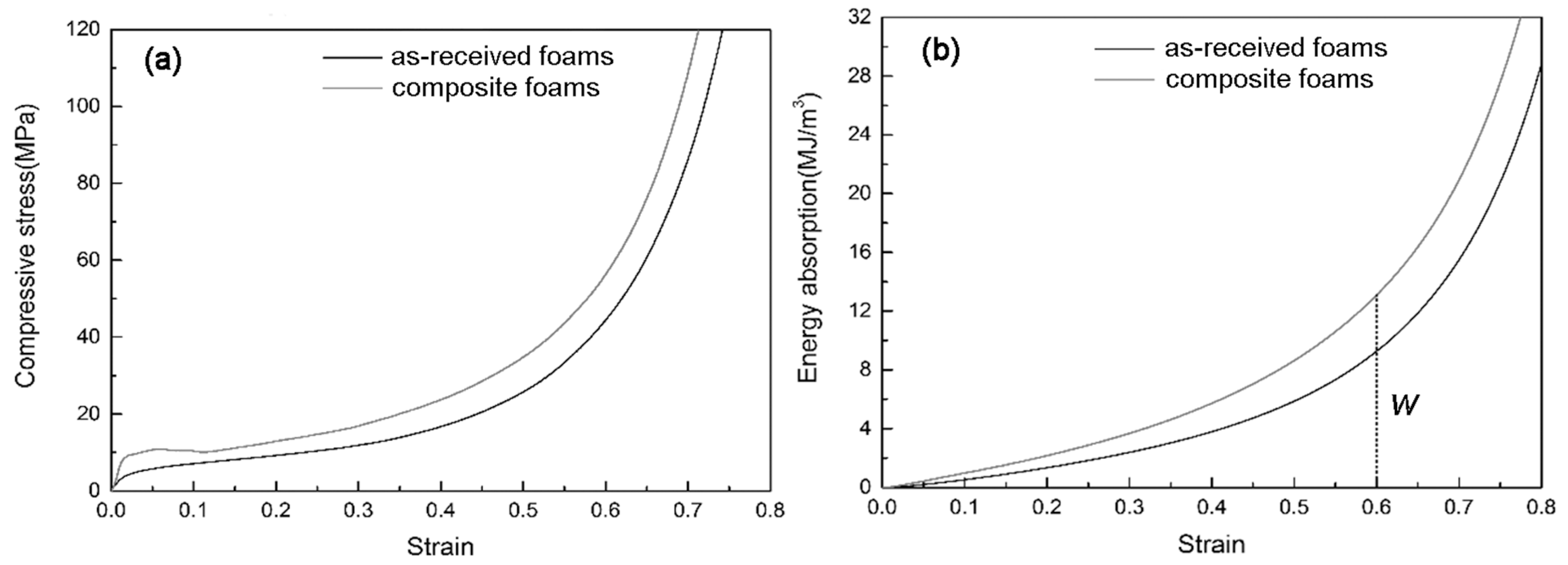
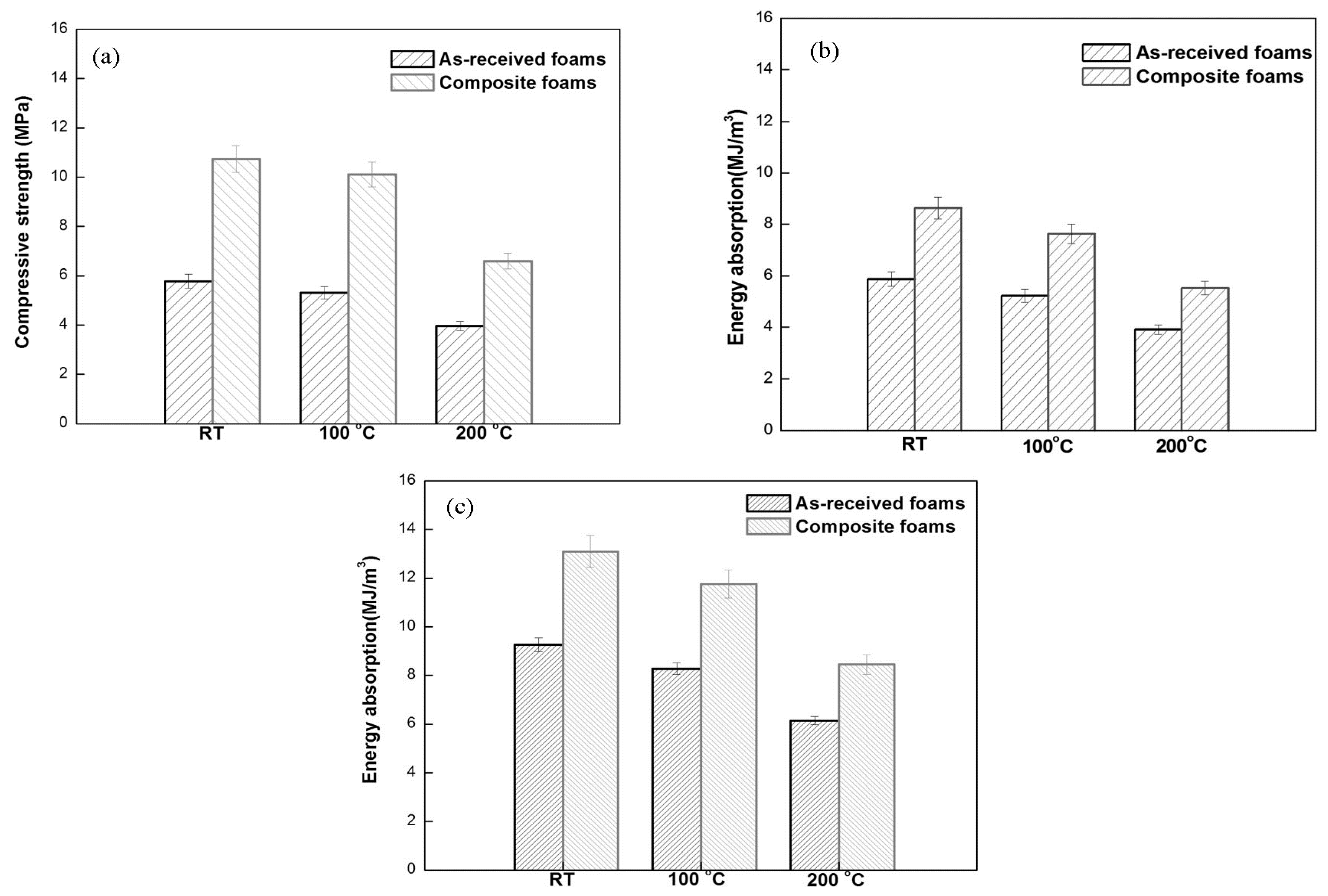
3.3. Compressive Properties of Composite Foams
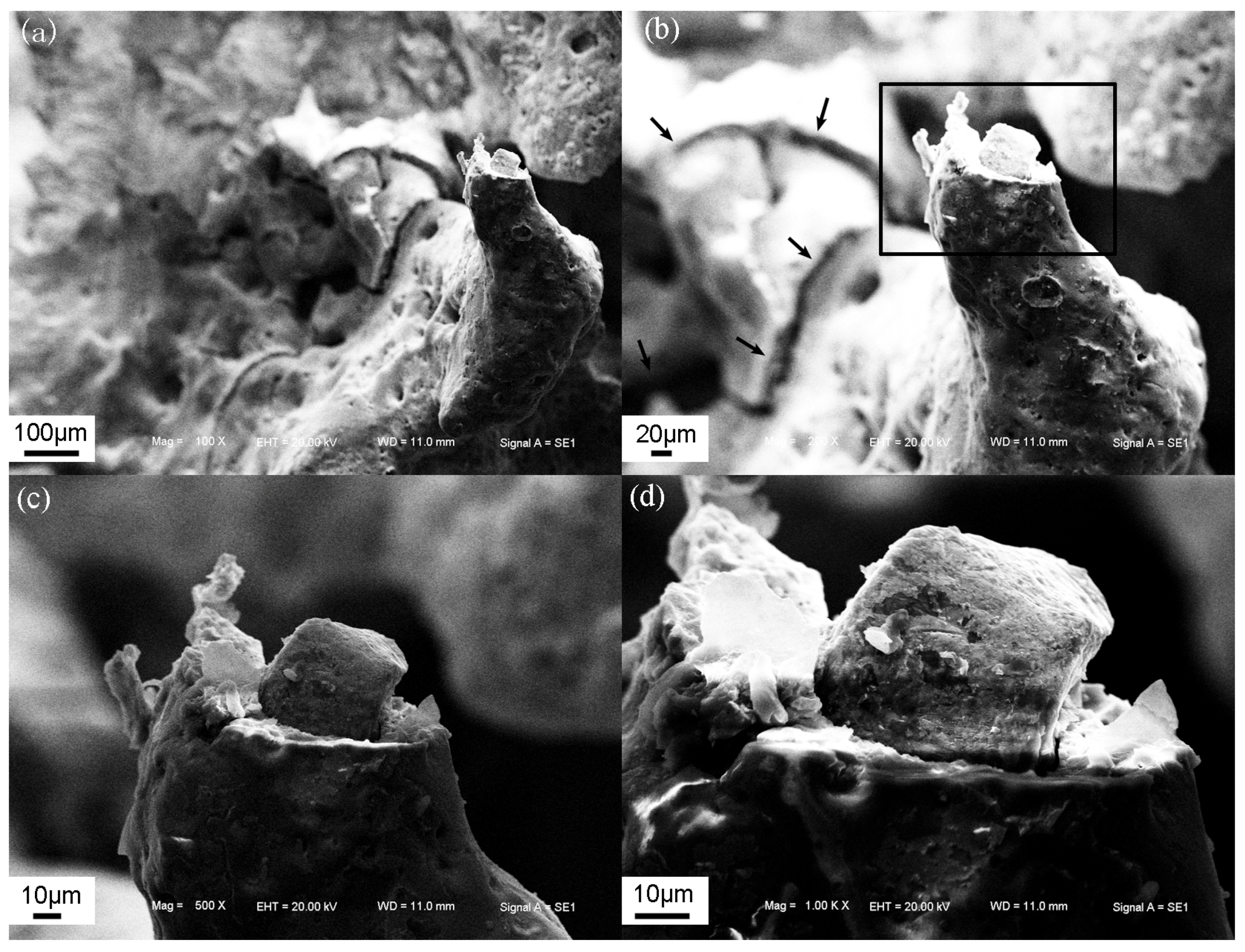
3.4. Failure Mechanism
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ashby, M.F.; Gibson, L. Cellular Solids: Structure and Properties, 2nd ed.; Cambridge University Press: Cambridge, UK, 1997. [Google Scholar]
- Banhart, J. Manufacture, characterisation and application of cellular metals and metal foams. Prog. Mater. Sci. 2001, 46, 559–632. [Google Scholar] [CrossRef]
- Stanev, L.; Kolev, M.; Drenchev, B.; Drenchev, L. Open-cell metallic porous materials obtained through space holders-part II: Structure and properties. A review. J. Manuf. Sci. Eng. 2017, 139, 050802. [Google Scholar] [CrossRef]
- Degischer, H.P.; Brigitte, K. Handbook of Cellular Metals, 1st ed.; WILEY-VCH-Verlag: Weinheim, Germany, 2002. [Google Scholar]
- García-Moreno, F. Commercial applications of metal foams: Their properties and production. Materials 2016, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Aly, M.S. Behavior of closed cell aluminium foams upon compressive testing at elevated temperatures: Experimental results. Mater. Lett. 2007, 61, 3138–3141. [Google Scholar] [CrossRef]
- Wang, P.; Xu, S.; Li, Z.; Yang, J.; Zheng, H.; Hu, S. Temperature effects on the mechanical behavior of aluminum foam under dynamic loading. Mater. Sci. Eng. A 2014, 599, 174–179. [Google Scholar] [CrossRef]
- Liu, J.; Qu, Q.; Liu, Y.; Li, R.; Liu, B. Compressive properties of Al-Si-SiC composite foams at elevated temperatures. J. Alloys Compd. 2016, 676, 239–244. [Google Scholar] [CrossRef]
- Duarte, I.; Ferreira, J. Composite and nanocomposite metal foams. Materials 2016, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Orbulov, I.N. Compressive properties of aluminium matrix syntactic foams. Mater. Sci. Eng. A 2012, 555, 52–56. [Google Scholar] [CrossRef]
- Goel, M.D.; Peroni, M.; Solomos, G.; Mondal, D.P.; Matsagar, V.A.; Gupta, A.K. Dynamic compression behavior of cenosphere aluminum alloy syntactic foam. Mater. Des. 2012, 42, 418–423. [Google Scholar] [CrossRef]
- Dunleavy, C.S.; Curran, J.A.; Clyne, T.W. Plasma electrolytic oxidation of aluminium networks to form a metal-cored ceramic composite hybrid material. Compos. Sci. Technol. 2011, 71, 908–915. [Google Scholar] [CrossRef]
- Abdulla, T.; Yerokhin, A.; Goodall, R. Enhancement in specific strength of open cell aluminium foams through plasma electrolytic oxidation treatment. Scr. Mater. 2014, 75, 38–41. [Google Scholar] [CrossRef]
- Abdulla, T.; Yerokhin, A.; Goodall, R. Effect of plasma electrolytic oxidation coating on the specific strength of open-cell aluminium foams. Mater. Des. 2011, 32, 3742–3749. [Google Scholar] [CrossRef]
- Liu, J.; Zhu, X.; Huang, Z.; Yu, S.; Yang, X. Characterization and property of microarc oxidation coatings on open-cell aluminum foams. J. Coat. Technol. Res. 2011, 9, 357–363. [Google Scholar] [CrossRef]
- Sun, Y.; Burgueño, R.; Wang, W.; Lee, I. Effect of annealing on the mechanical properties of nano-copper reinforced open-cell aluminum foams. Mater. Sci. Eng. A 2014, 613, 340–351. [Google Scholar] [CrossRef]
- Antenucci, A.; Guarino, S.; Tagliaferri, V.; Ucciardello, N. Improvement of the mechanical and thermal characteristics of open cell aluminum foams by the electrodeposition of Cu. Mater. Des. 2014, 59, 124–129. [Google Scholar] [CrossRef]
- Liu, J.; Gao, F.; Rao, Y.; Wu, C.; Liu, Y. Compressive properties of aluminum foams produced by replication route using spheroidal calcium chloride as space holder. Mater. Trans. 2014, 55, 1906–1908. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, N.; Sun, K. Facile ammonia-induced fabrication of nanoporous NiO films with enhanced lithium-storage properties. Electrochem. Commun. 2012, 20, 137–140. [Google Scholar] [CrossRef]
- Duarte, I.; Vesenjak, M.; Krstulović-Opara, L. Variation of quasi-static and dynamic compressive properties in a single aluminium foam block. Mater. Sci. Eng. A 2014, 616, 171–182. [Google Scholar] [CrossRef]
- Sundararajan, G.; Krishna, L.R. Mechanisms underlying the formation of thick alumina coatings through the MAO coating technology. Surf. Coat. Technol. 2003, 167, 269–277. [Google Scholar] [CrossRef]
- Khan, R.H.U.; Yerokhin, A.; Li, X.; Dong, H.; Matthews, A. Surface characterisation of DC plasma electrolytic oxidation treated 6082 aluminium alloy: Effect of current density and electrolyte concentration. Surf. Coat. Technol. 2010, 205, 1679–1688. [Google Scholar] [CrossRef]
- Liu, J.; Si, F.; Zhu, X.; Liu, Y.; Zhang, J.; Liu, Y. Compressive properties of open-cell Al hybrid foams at different temperatures. Materials 2017, 10, 98. [Google Scholar] [CrossRef] [PubMed]










© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, H.; Pan, W.; Si, F.; Huang, K.; Liu, Y.; Liu, J. Enhanced Compressive Property of Al Composite Foams at Elevated Temperatures via Plasma Electrolytic Oxidation. Metals 2018, 8, 118. https://doi.org/10.3390/met8020118
Liu H, Pan W, Si F, Huang K, Liu Y, Liu J. Enhanced Compressive Property of Al Composite Foams at Elevated Temperatures via Plasma Electrolytic Oxidation. Metals. 2018; 8(2):118. https://doi.org/10.3390/met8020118
Chicago/Turabian StyleLiu, Huamin, Wenchi Pan, Fujian Si, Kuo Huang, Yan Liu, and Jiaan Liu. 2018. "Enhanced Compressive Property of Al Composite Foams at Elevated Temperatures via Plasma Electrolytic Oxidation" Metals 8, no. 2: 118. https://doi.org/10.3390/met8020118
APA StyleLiu, H., Pan, W., Si, F., Huang, K., Liu, Y., & Liu, J. (2018). Enhanced Compressive Property of Al Composite Foams at Elevated Temperatures via Plasma Electrolytic Oxidation. Metals, 8(2), 118. https://doi.org/10.3390/met8020118



