Improved Compressive, Damping and Coefficient of Thermal Expansion Response of Mg–3Al–2.5La Alloy Using Y2O3 Nano Reinforcement
Abstract
:1. Introduction
2. Materials and Characterizations
2.1. Materials
2.2. Processing
2.3. Characterizations
2.3.1. Microstructural Characterization
2.3.2. Physical Characterization
2.3.3. Mechanical Characterization
3. Results and Discussion
3.1. Microstructural Characterization
3.2. Physical Characterization
3.2.1. Density and Porosity
3.2.2. The Coefficient of Thermal Expansion
3.2.3. Damping
3.3. Mechanical Characterization
4. Conclusions
- With the addition of the Y2O3 reinforcement, an even finer grain structure can be realized (~3.6 μm for Mg–3Al–2.5La–1.9 Y2O3 alloy, 43% less than that of Mg–3Al–2.5La at ~6.26 μm).
- The microstructural characterization concluded that all intermetallic phases and and were still present in dispersed form, but the sizes of these phases were refined by the addition of nanosize Y2O3 in the Mg–3Al–2.5La alloy.
- The compressive results concluded that the addition of Y2O3 to Mg–3Al–2.5La significantly improved the compressive yield strength and the ultimate compressive strength (CYS from ~141 MPa to ~156 MPa and UCS from ~456 MPa to ~520 MPa), which are even better than those of the Mg–3Al alloy (CYS, ~154 MPa and UCS, ~481 MPa). There was no adverse effect on the fracture strain value recorded for Mg–3Al–2.5La with the addition of Y2O3.
- The damping results concluded that the addition of nanosize Y2O3 to Mg–3Al–2.5La improved the damping capacity. The addition of the Y2O3 reinforcement also improved the CTE value of the Mg–3Al–2.5La alloy.
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Khomamizadeh, F.; Nami, B.; Khoshkhooei, S. Effect of rare-earth element additions on high-temperature mechanical properties of AZ91 magnesium alloy. Metall. Mater. Trans. A 2005, 36, 3489–3494. [Google Scholar] [CrossRef]
- Braszczyńska-Malik, K.N. Precipitates of r–Mg17Al12 Phase in AZ91 Alloy. Available online: http://cdn.intechweb.org/pdfs/12741.pdf. (accessed on 14 March 2017).
- Rokhlin, L.L. Magnesium Alloys Containing Rare Earth Metals: Structure and Properties; Crc Press: Florida, FL, USA, 2003. [Google Scholar]
- Gupta, M.; Sharon, N.M.L. Magnesium, Magnesium Alloys, and Magnesium Composites; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Mirzadeh, H. Constitutive analysis of Mg–Al–Zn magnesium alloys during hot deformation. Mech. Mater. 2014, 77, 80–85. [Google Scholar] [CrossRef]
- Mirzadeh, H. Quantification of the strengthening effect of rare earth elements during hot deformation of Mg–Gd–Y–Zr magnesium alloy. J. Mater. Res. Technol. 2016, 5, 1–4. [Google Scholar] [CrossRef]
- Hihara, L.H.; Adler, R.P.; Latanision, R.M. Environmental Degradation of Advanced and Traditional Engineering Materials; CRC Press: Florida, FL, USA, 2013. [Google Scholar]
- Kumar, A.; Meenashisundaram, G.K.; Manakari, V.; Parande, G.; Gupta, M. Lanthanum effect on improving CTE, damping, hardness and tensile response of Mg–3Al alloy. J. Alloy. Compd. 2017, 695, 3612–3620. [Google Scholar] [CrossRef]
- Hassan, S.; Gupta, M. Development of high performance magnesium nanocomposites using solidification processing route. Mater. Sci. Technol. 2004, 20, 1383–1388. [Google Scholar] [CrossRef]
- Hassan, S.; Gupta, M. Effect of length scale of Al2O3 particulates on microstructural and tensile properties of elemental Mg. Mater. Sci. Eng. A 2006, 425, 22–27. [Google Scholar] [CrossRef]
- Hassan, S.; Gupta, M. Effect of type of primary processing on the microstructure, CTE and mechanical properties of magnesium/alumina nanocomposites. Compos. Struct. 2006, 72, 19–26. [Google Scholar] [CrossRef]
- Hassan, S.; Gupta, M. Development of high performance magnesium nano-composites using nano–Al2O3 as reinforcement. Mater. Sci. Eng. A 2005, 392, 163–168. [Google Scholar] [CrossRef]
- Hassan, S.; Gupta, M. Effect of different types of nano-size oxide particulates on microstructural and mechanical properties of elemental Mg. J. Mater. Sci. 2006, 41, 2229–2236. [Google Scholar] [CrossRef]
- Dorri Moghadam, A.; Ferguson, J.B.; Schultz, B.F.; Lopez, H.; Rohatgi, P.K. Direct Synthesis of Nanostructured in Situ Hybrid Aluminum Matrix Nanocomposite. Ind. Eng. Chem. Res. 2016, 55, 6345–6353. [Google Scholar] [CrossRef]
- Moghadam, A.D.; Schultz, B.F.; Ferguson, J.B.; Omrani, E.; Rohatgi, P.K.; Gupta, N. Functional metal matrix composites: Self-lubricating, self-healing, and nanocomposites-an outlook. JOM 2014, 66, 872–881. [Google Scholar] [CrossRef]
- Rohatgi, P.K.; Afsaneh, D.M.; Schultz, B.F.; Ferguson, J.B. Synthesis and Properties of Metal Matrix Nanocomposites (MMNCS), Syntactic Foams, Self Lubricating and Self-Healing Metals. Available online: https://www.researchgate.net/profile/Afsaneh_Dorri_Moghadam/publication/270510257_Synthesis_and_Properties_of_Metal_Matrix_Nanocomposites_MMNCS_Syntactic_Foams_Self_Lubricating_and_Self-Healing_Metals/links/54ac489a0cf2479c2ee7ac9a/Synthesis-and-Properties-of-Metal-Matrix-Nanocomposites-MMNCS-Syntactic-Foams-Self-Lubricating-and-Self-Healing-Metals.pdf (accessed on 18 March 2017).
- Hassan, S.; Gupta, M. Development of nano–Y2O3 containing magnesium nanocomposites using solidification processing. J. Alloy. Compd. 2007, 429, 176–183. [Google Scholar] [CrossRef]
- Sankaranarayanan, S.; Jayalakshmi, S.; Gupta, M. Effect of addition of mutually soluble and insoluble metallic elements on the microstructure, tensile and compressive properties of pure magnesium. Mater. Sci. Eng. A 2011, 530, 149–160. [Google Scholar] [CrossRef]
- Braszczyńska-Malik, K. Some Mechanical Properties of Experimental Mg–Al–RE–Mn Magnesium Alloys. Arch. Foundry Eng. 2014, 14, 13–16. [Google Scholar] [CrossRef]
- Greger, M.; Kocich, R.; Čížek, L.; Dobrzański, L.A.; Juřička, I. Possibilities of mechanical properties and microstructure improvement of magnesium alloys. Arch. Mater. Sci. Eng. 2007, 28, 83–90. [Google Scholar]
- Moosa, A.A. Effect of Lanthanum Addition on the Microstructure of Mg–4Al Alloy. Al-Khwarizmi Eng. J. 2011, 7, 75–82. [Google Scholar]
- Zhang, J.; Yu, P.; Liu, K.; Fang, D.; Meng, J. Effect of substituting cerium-rich mischmetal with lanthanum on microstructure and mechanical properties of die-cast Mg–Al–RE alloys. Mater. Des. 2009, 30, 2372–2378. [Google Scholar] [CrossRef]
- Tekumalla, S.; Seetharaman, S.; Almajid, A.; Gupta, M. Mechanical properties of magnesium-rare earth alloy systems: A review. Metals 2014, 5, 1–39. [Google Scholar] [CrossRef]
- Kainer, K.U.; Kaiser, F. Magnesium Alloys and Technology; John Wiley & Sons: Hoboken, NJ, USA, 2003. [Google Scholar]
- Bai, J.; Sun, Y.; Xue, F. Microstructures and creep properties of Mg–4Al–(1–4) La alloys produced by different casting techniques. Mater. Sci. Eng. A 2012, 552, 472–480. [Google Scholar] [CrossRef]
- Jain, C.C.; Koo, C.H. Creep behavior of extruded sheets of magnesium alloys containing La-rich Mischmetal. Mater. Trans. 2006, 47, 433–439. [Google Scholar] [CrossRef]
- Kim, J.M.; Lee, S.J. Microstructure and Castability of Mg–Al–La Alloys for High Conductivity Applications. Int. J. Met. 2015, 9, 15–21. [Google Scholar] [CrossRef]
- Zhang, J.H.; Zhang, M.L.; Meng, J.; Wu, R.Z.; Tang, D.X. Microstructures and mechanical properties of heat-resistant high-pressure die-cast Mg–4Al–xLa–0.3Mn (x = 1, 2, 4, 6) alloys. Mater. Sci. Eng. A 2010, 527, 2527–2537. [Google Scholar] [CrossRef]
- Zhang, J.H.; Liu, S.J.; Zhe, L.; Zhang, M.L.; Meng, J.; Wu, R.Z. Structure stability and mechanical properties of high-pressure die-cast Mg–Al–La–Y–based alloy. Mater. Sci. Eng. A 2012, 531, 70–75. [Google Scholar] [CrossRef]
- Nguyen, Q.; Gupta, M. Increasing significantly the failure strain and work of fracture of solidification processed AZ31B using nano–Al2O3 particulates. J. Alloy. Compd. 2008, 459, 244–250. [Google Scholar] [CrossRef]
- Yang, Q.; Bu, F.; Zheng, F.; Liu, X.; Zhang, D.; Qiu, X.; Meng, J. Influence of trace Sr additions on the microstructures and the mechanical properties of Mg–Al–La–based alloy. Mater. Sci. Eng. A 2014, 619, 256–264. [Google Scholar] [CrossRef]
- Zhang, J.; Zhang, D.; Tian, Z.; Wang, J.; Liu, K.; Lu, H.; Tang, D.X.; Meng, J. Microstructures, tensile properties and corrosion behavior of die–cast Mg–4Al–based alloys containing La and/or Ce. Mater. Sci. Eng. A 2008, 489, 113–119. [Google Scholar] [CrossRef]
- Wei, L.; Dunlop, G. The solidification behaviour of Mg–Al–rare earth alloys. J. Alloy. Compd. 1996, 232, 264–268. [Google Scholar] [CrossRef]
- Zou, H.; Zeng, X.; Zhai, C.; Ding, W. Effects of Nd on the microstructure of ZA52 alloy. Mater. Sci. Eng. A 2005, 392, 229–234. [Google Scholar] [CrossRef]
- Sankaranarayanan, S.; Nayak, U.P.; Stbat, R.K.; Suwas, S.; Almajid, A.; Gupta, M. Nano–ZnO particle addition to monolithic magnesium for enhanced tensile and compressive response. J. Alloy. Compd. 2014, 615, 211–219. [Google Scholar] [CrossRef]
- Goodfellowc. Metals and Materials for Research and Industry. Alloys, and Ceramics Compounds. Available online: http://www.goodfellow.com/E/M.html (accessed on 14 March 2017).
- Nguyen, Q.B.; Nai, M.L.S.; Nguyen, A.S.; Seetharaman, S.; Jayalakshmi, S.; Leong, E.W.W.; Gupta, M. Microstructure and damping characteristics of Mg and its composites containing metastable Al85Ti15 particle. J. Compos. Mater. 2016, 50, 2565–2573. [Google Scholar] [CrossRef]
- Batra, R.C.; Wei, Z.G. Instability strain and shear band spacing in simple tensile/compressive deformations of thermoviscoplastic materials. Int. J. Impact. Eng. 2007, 34, 448–463. [Google Scholar] [CrossRef]
- Wang, T.S.; Hou, R.J.; Lv, B.; Zhang, M.; Zhang, F.C. Microstructure evolution and deformation mechanism change in 0.98C–8.3Mn–0.04N steel during compressive deformation. Mater. Sci. Eng. A 2007, 465, 68–71. [Google Scholar] [CrossRef]

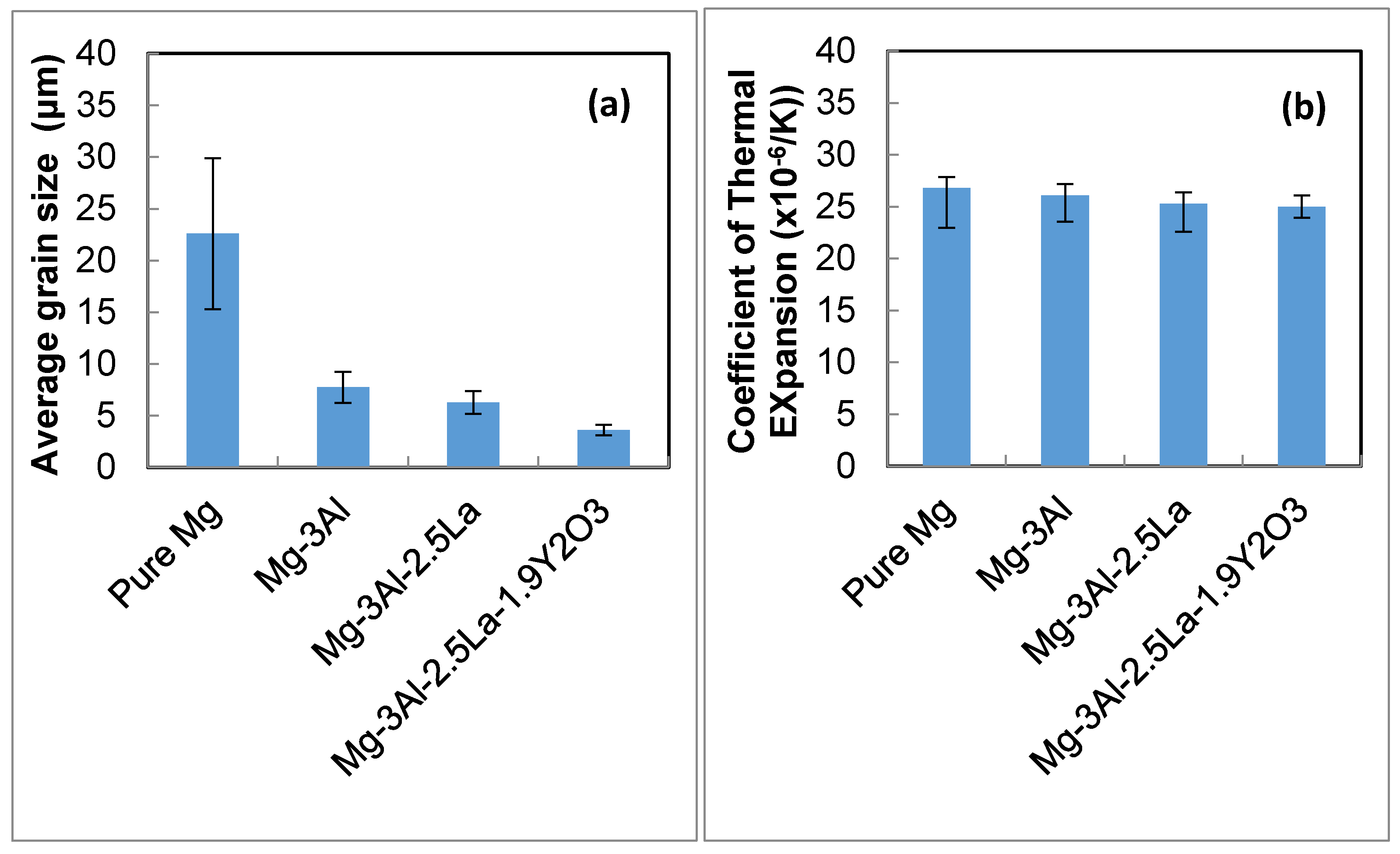

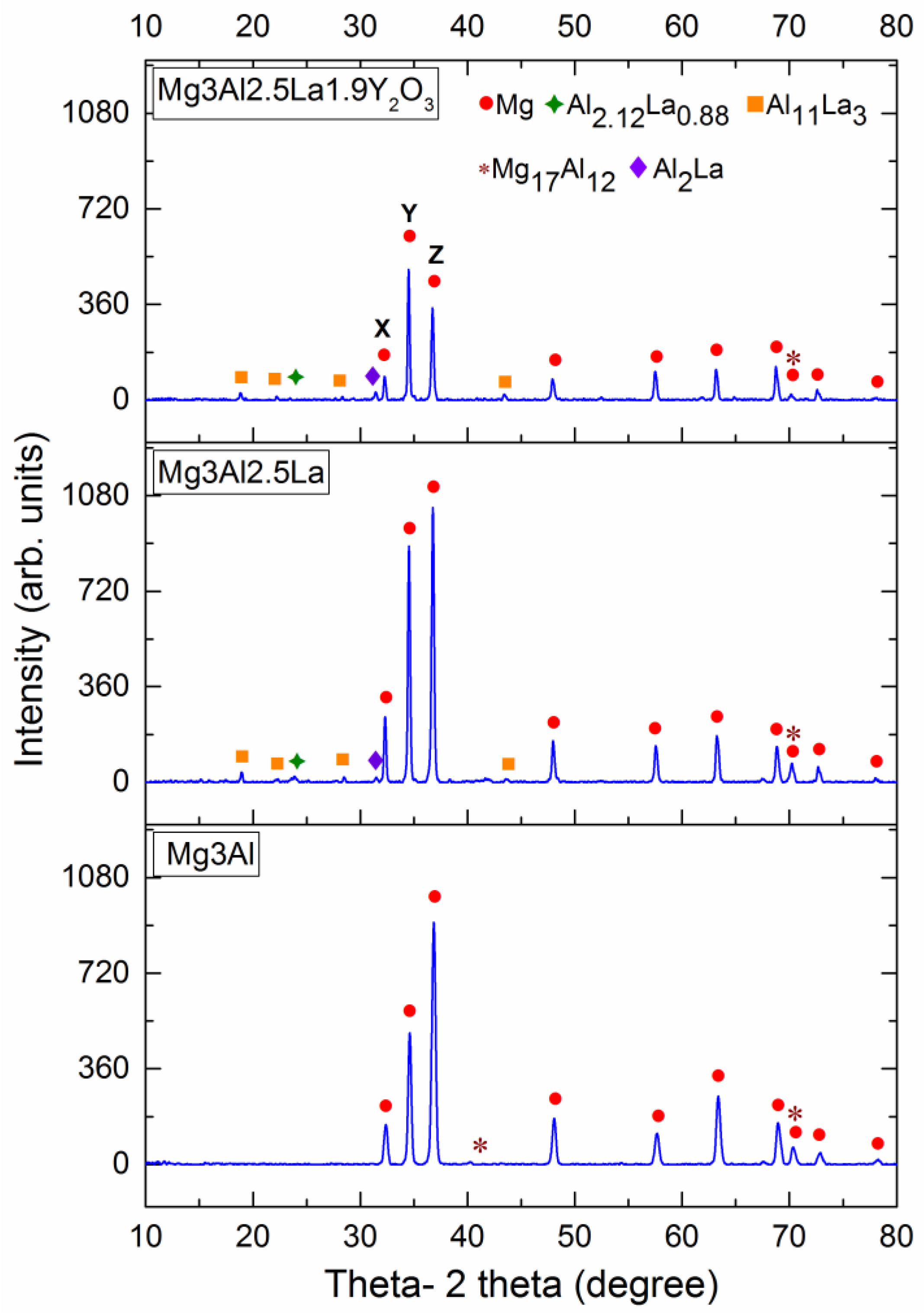
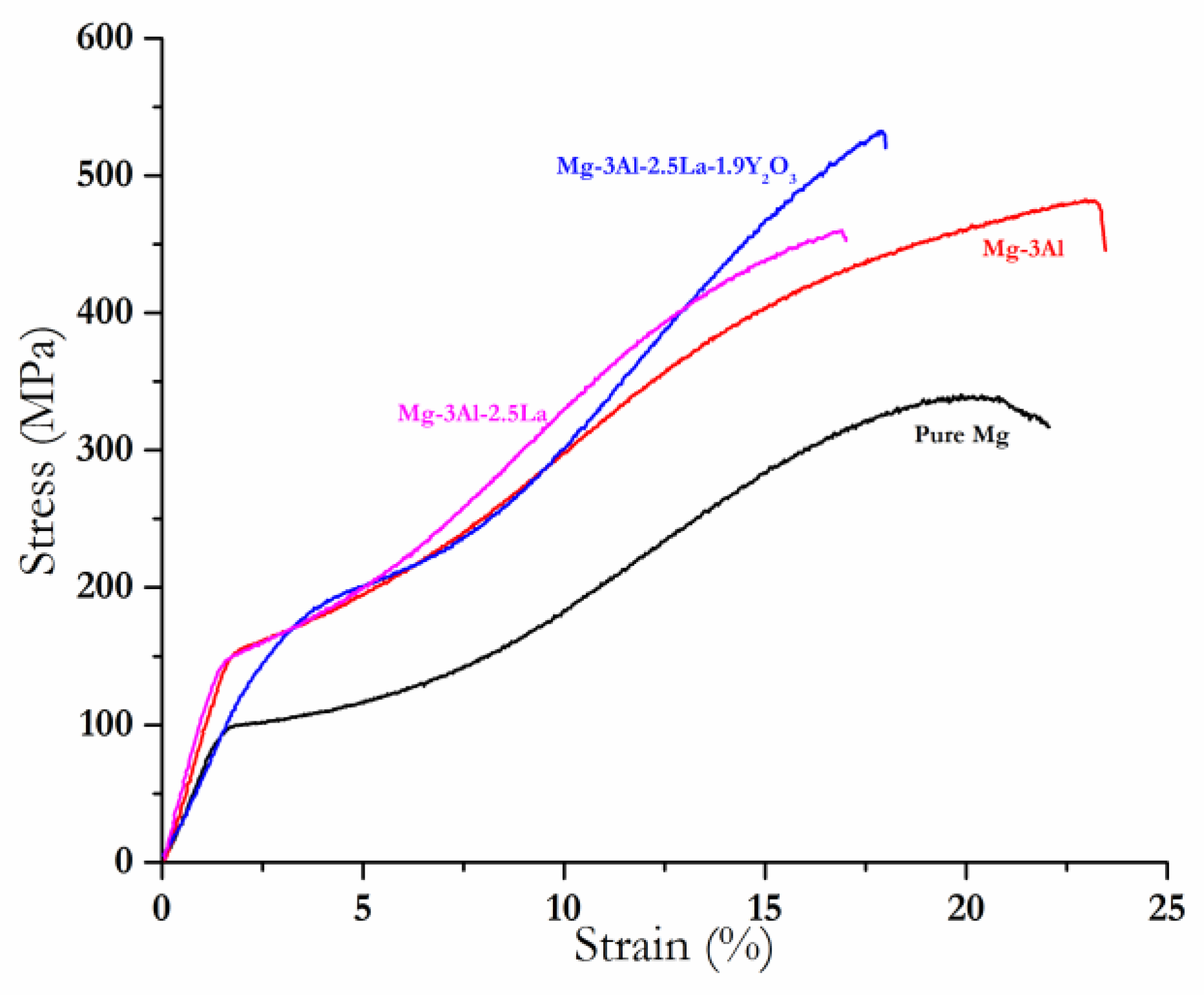
| Material (wt %) | Average Grain Size (μm) | Density and Porosity Measurements | CTE (×10−6/K) | ||
|---|---|---|---|---|---|
| Theoretical Density (g/cc) | Experimental Density (g/cc) | Porosity (%) | |||
| Pure Mg | 22.6 ± 7.3 | 1.738 | 1.737 | 0.15 | 26.8 ± 3.9 |
| Mg–3Al | 7.74 ± 1.5 | 1.758 | 1.753 | 0.29 | 26.1 ± 2.6 |
| Mg–3Al–2.5La | 6.26 ± 1.1 | 1.791 | 1.788 | 0.17 | 25.3 ± 2.7 |
| Mg–3Al–2.5La–1.9Y2O3 | 3.6 ± 0.5 | 1.818 | 1.813 | 0.16 | 25.0 ± 1.1 |
| Material | 0.2% CYS (MPa) | UCS (MPa) | Fracture Strain (%) | Damping Loss Rate | Damping Capacity |
|---|---|---|---|---|---|
| Pure Mg [8] | 90 ± 6 | 333 ± 4 | 23 ± 0.74 | 8.00 ± 1.000 | 0.000456 |
| Mg–3Al [8] | 154 ± 2 | 481 ± 7 | 24 ± 0.5 | 6.16 ± 0.377 | 0.000204 |
| Mg–3Al–2.5La [8] | 141 ± 4 | 456 ± 3 | 18 ± 1 | 8.29 ± 0.827 | 0.000265 |
| Mg–3Al–2.5La–1.9Y2O3 | 156 ± 5 | 520 ± 8 | 18 ± 0.70 | 7.60 ± 0.701 | 0.000272 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license ( http://creativecommons.org/licenses/by/4.0/.
Share and Cite
Kumar, A.; Tun, K.S.; Kohadkar, A.D.; Gupta, M. Improved Compressive, Damping and Coefficient of Thermal Expansion Response of Mg–3Al–2.5La Alloy Using Y2O3 Nano Reinforcement. Metals 2017, 7, 104. https://doi.org/10.3390/met7030104
Kumar A, Tun KS, Kohadkar AD, Gupta M. Improved Compressive, Damping and Coefficient of Thermal Expansion Response of Mg–3Al–2.5La Alloy Using Y2O3 Nano Reinforcement. Metals. 2017; 7(3):104. https://doi.org/10.3390/met7030104
Chicago/Turabian StyleKumar, Amit, Khin Sandar Tun, Amit Devendra Kohadkar, and Manoj Gupta. 2017. "Improved Compressive, Damping and Coefficient of Thermal Expansion Response of Mg–3Al–2.5La Alloy Using Y2O3 Nano Reinforcement" Metals 7, no. 3: 104. https://doi.org/10.3390/met7030104
APA StyleKumar, A., Tun, K. S., Kohadkar, A. D., & Gupta, M. (2017). Improved Compressive, Damping and Coefficient of Thermal Expansion Response of Mg–3Al–2.5La Alloy Using Y2O3 Nano Reinforcement. Metals, 7(3), 104. https://doi.org/10.3390/met7030104








