Thermo-Calc Prediction of Mushy Zone in AlSiFeMn Alloys
Abstract
:1. Introduction
2. Materials and Methods
3. Results
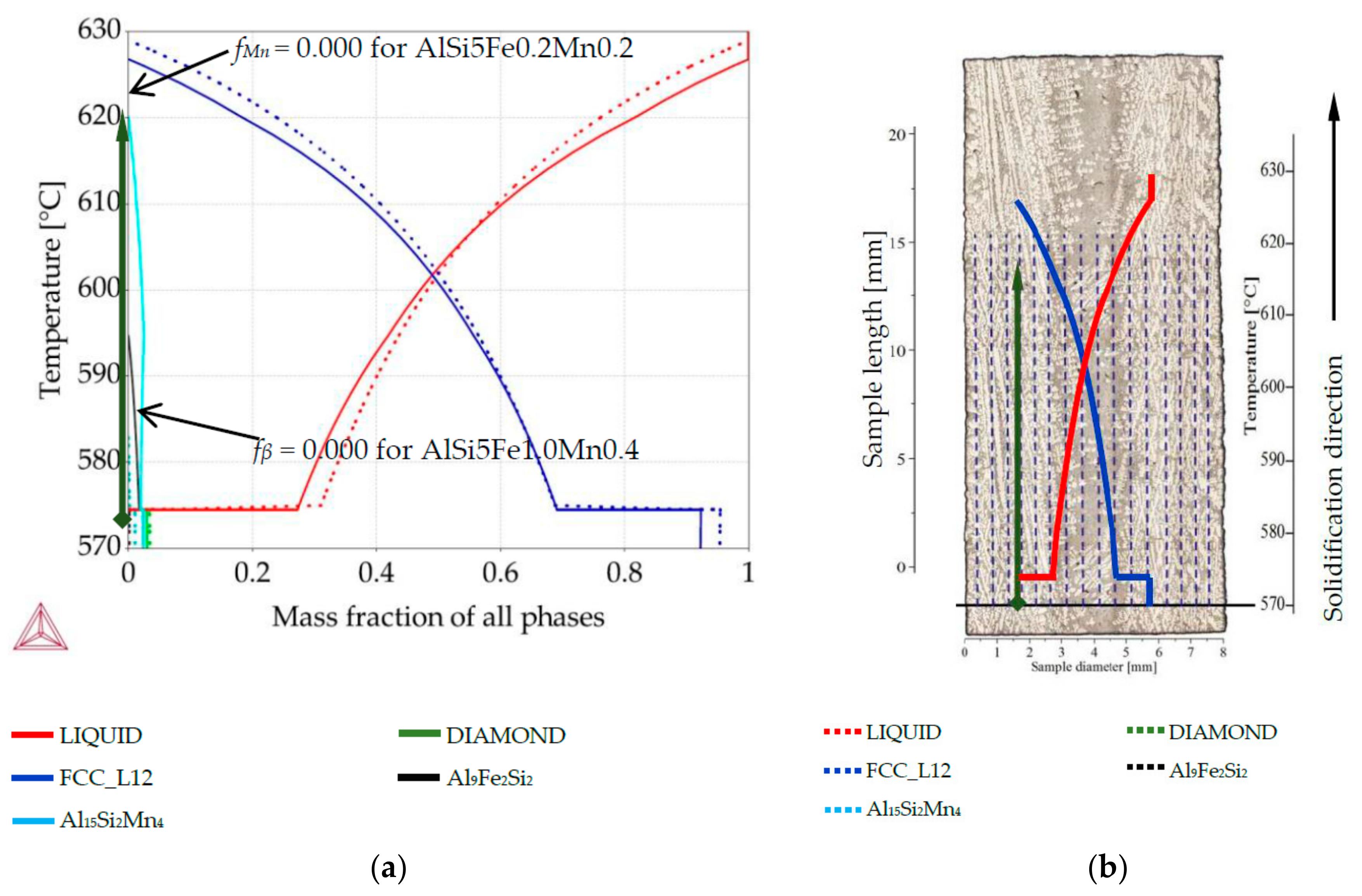
3.1. Precipitation Sequence
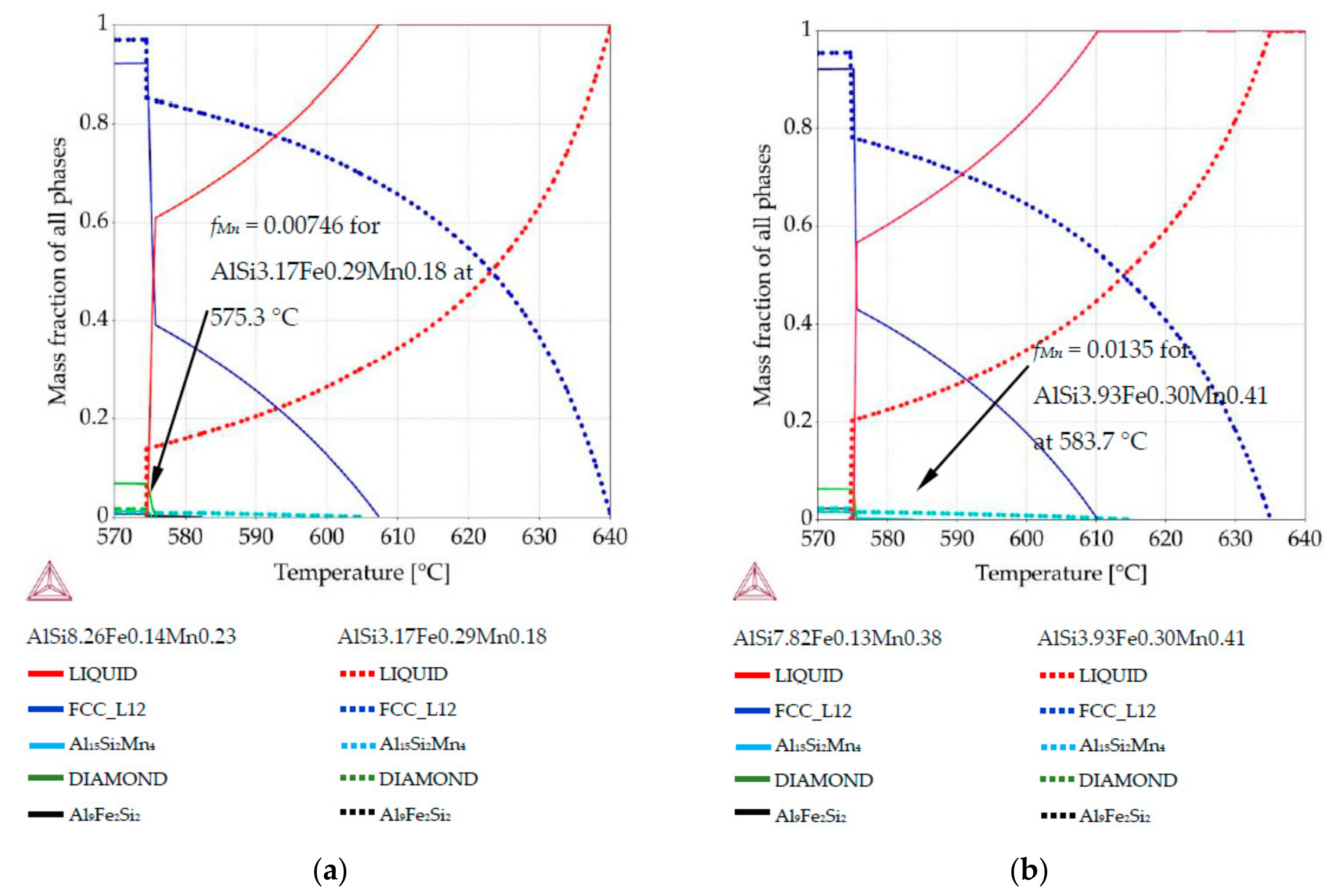
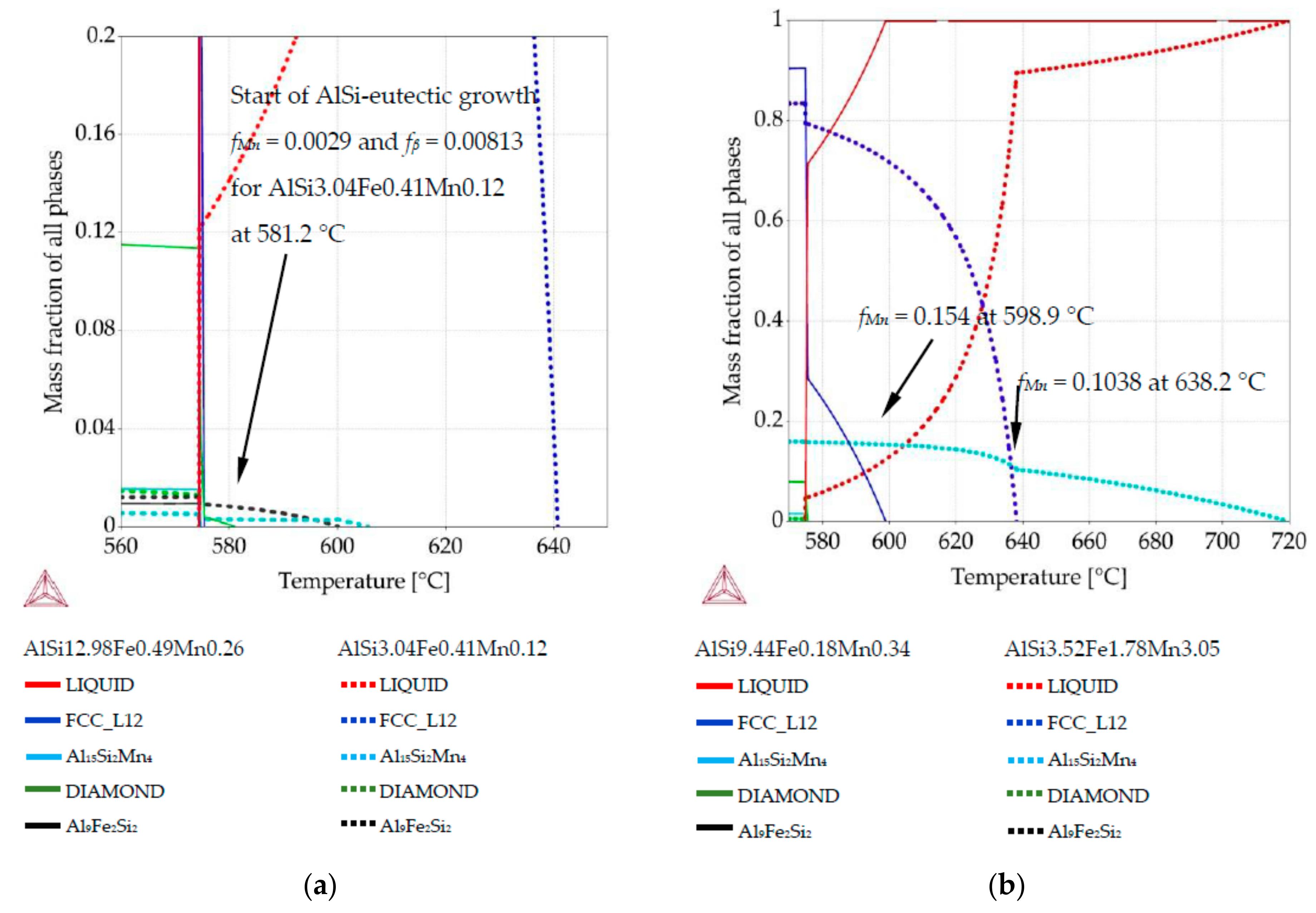
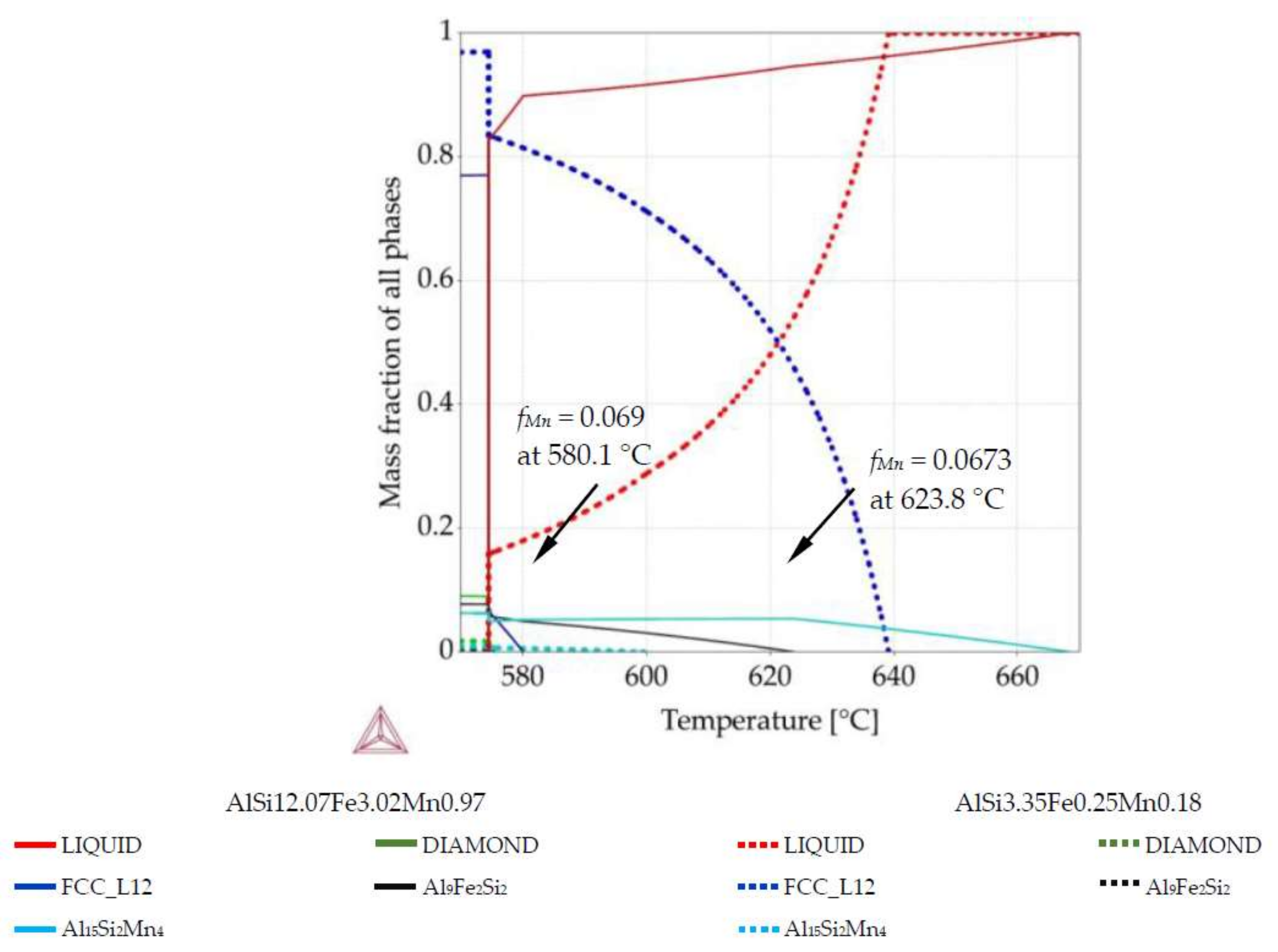
3.2. Phase Mass Fraction Diagrams
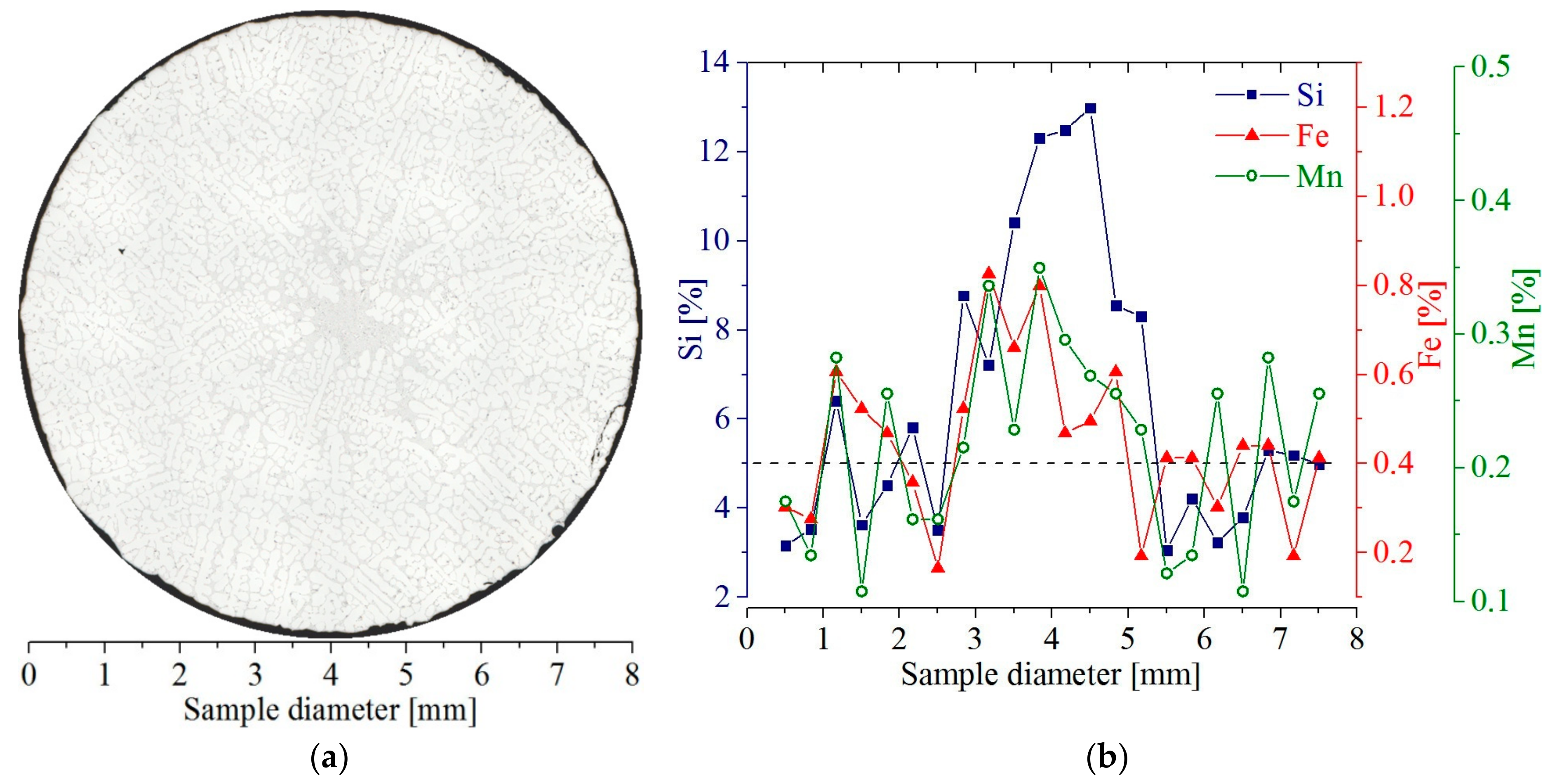
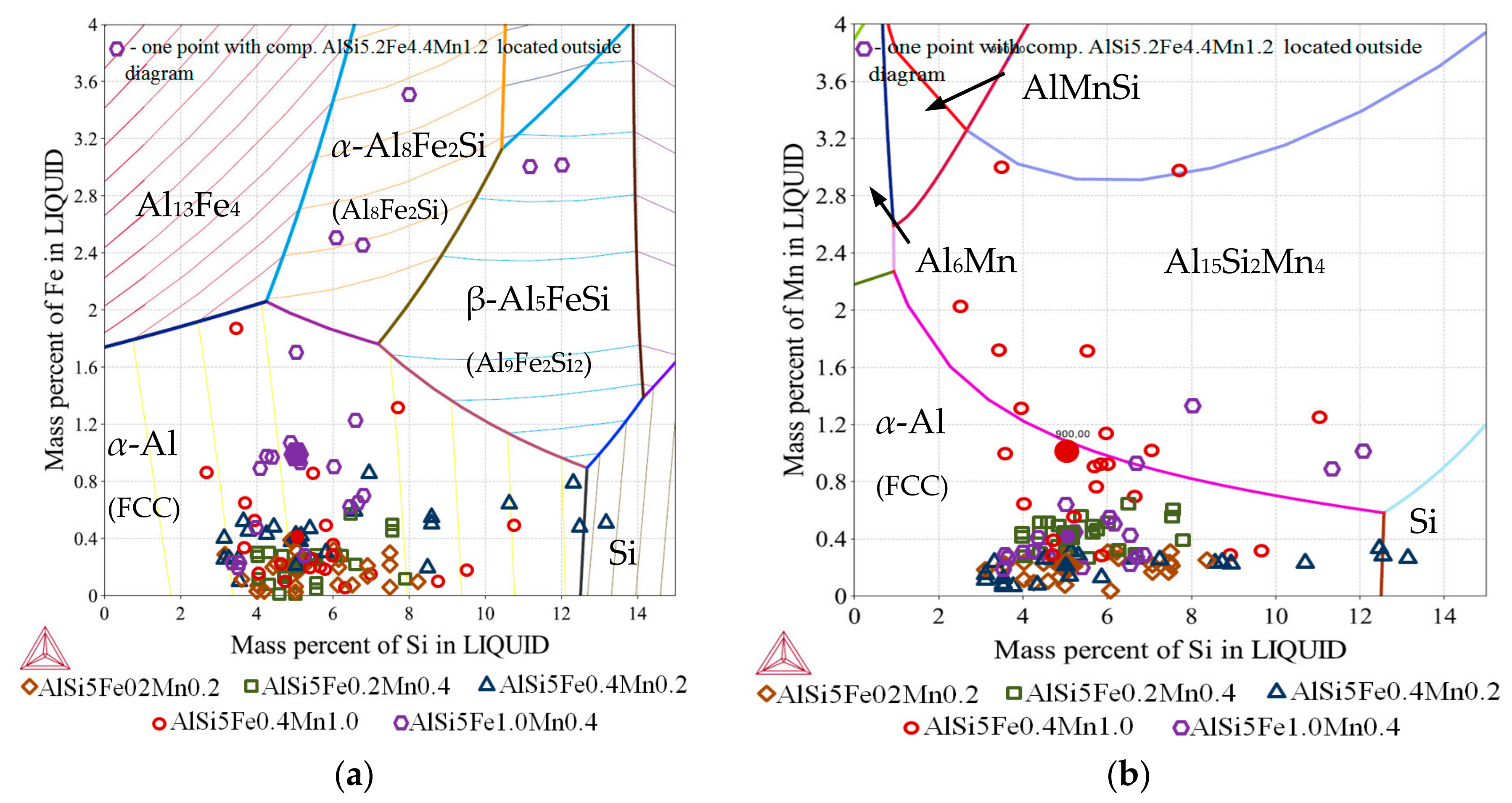
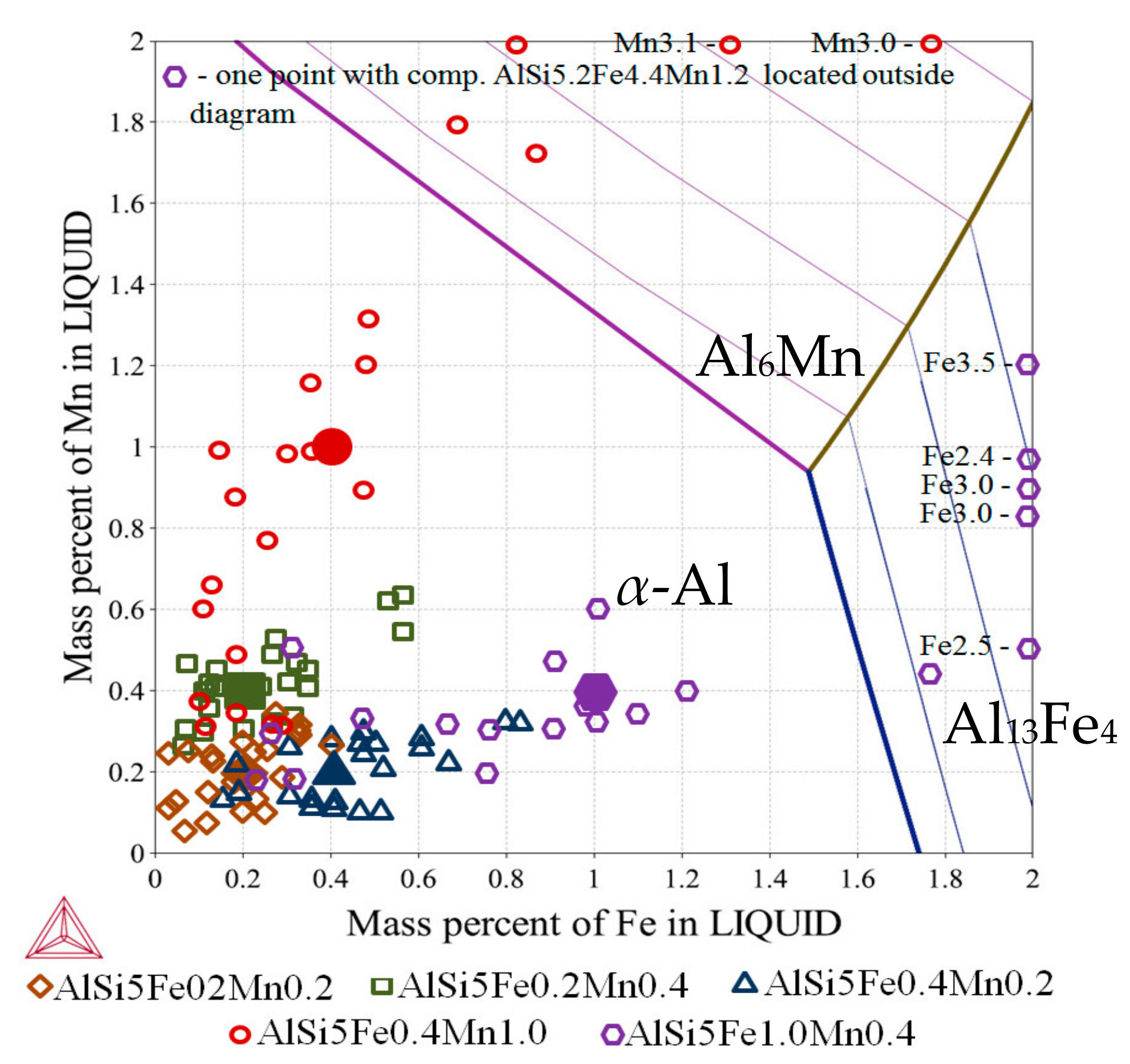
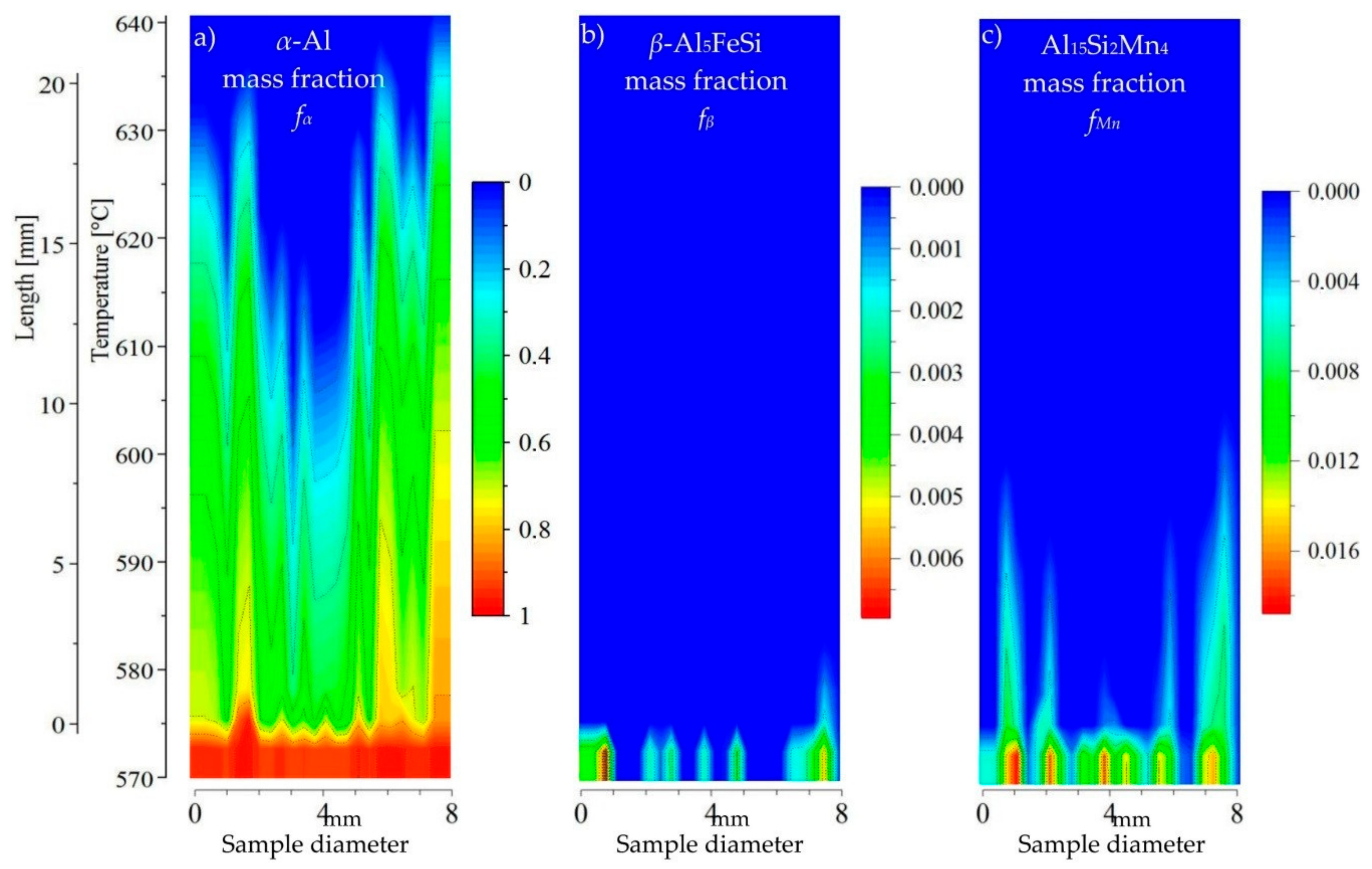
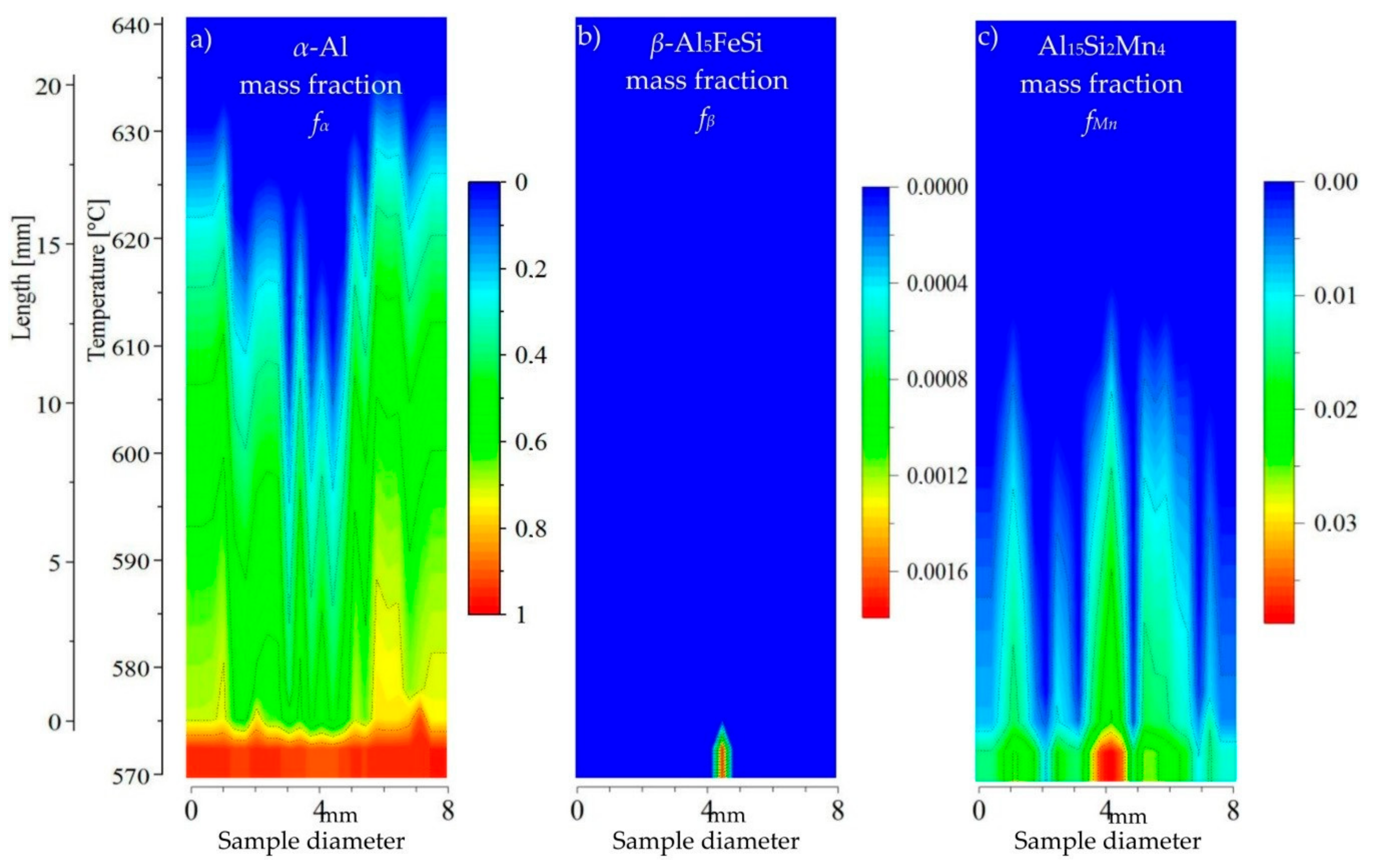
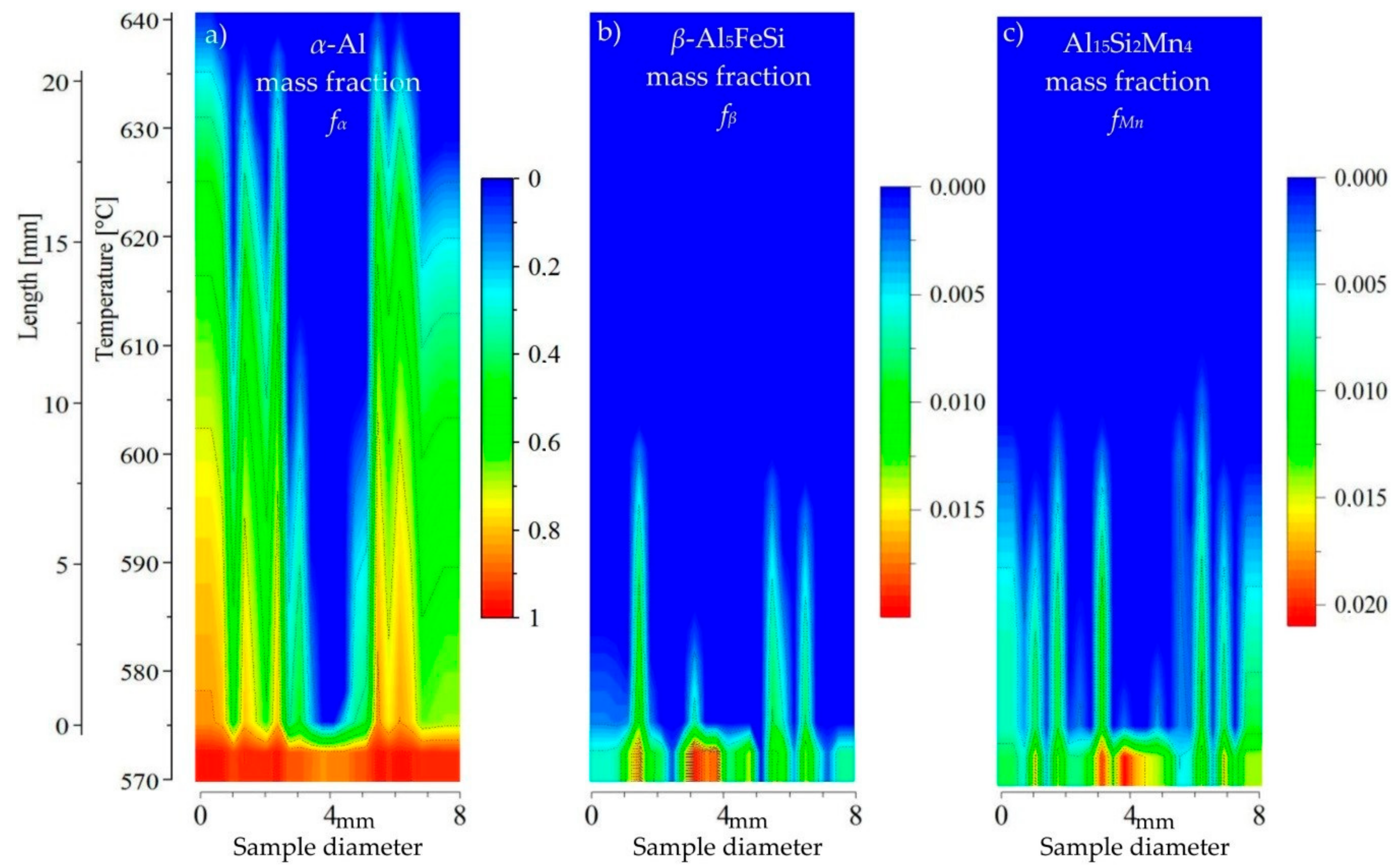
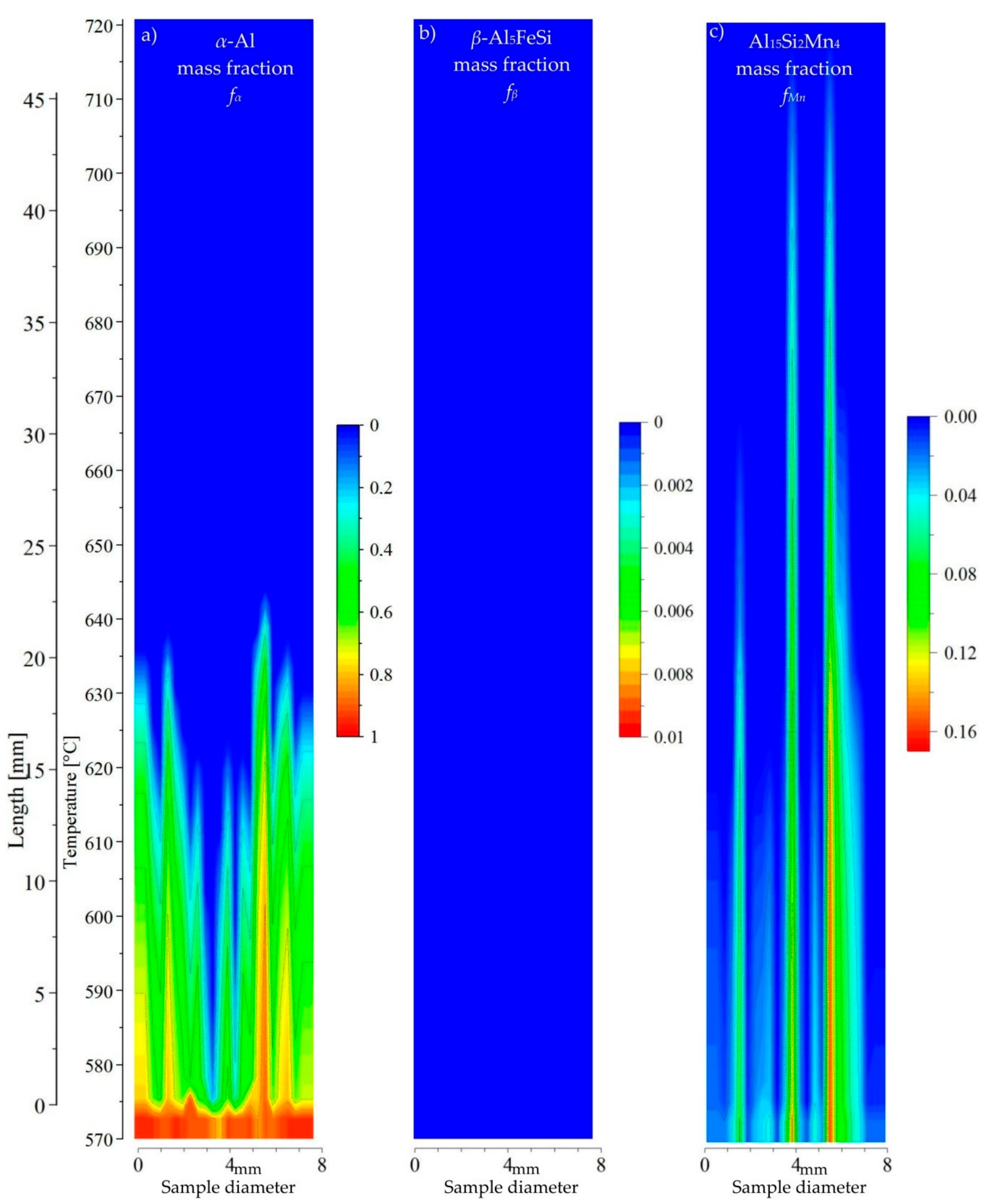
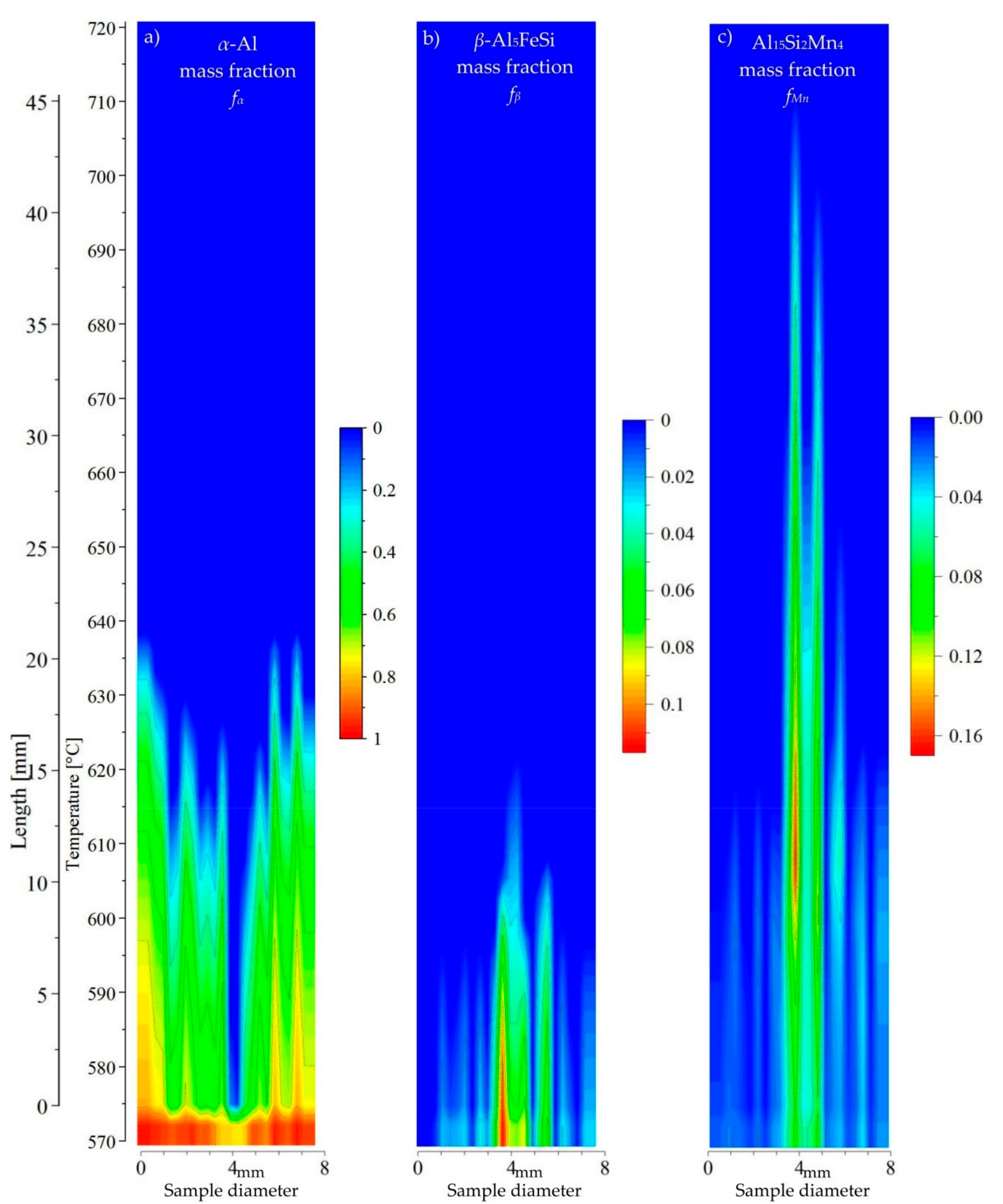
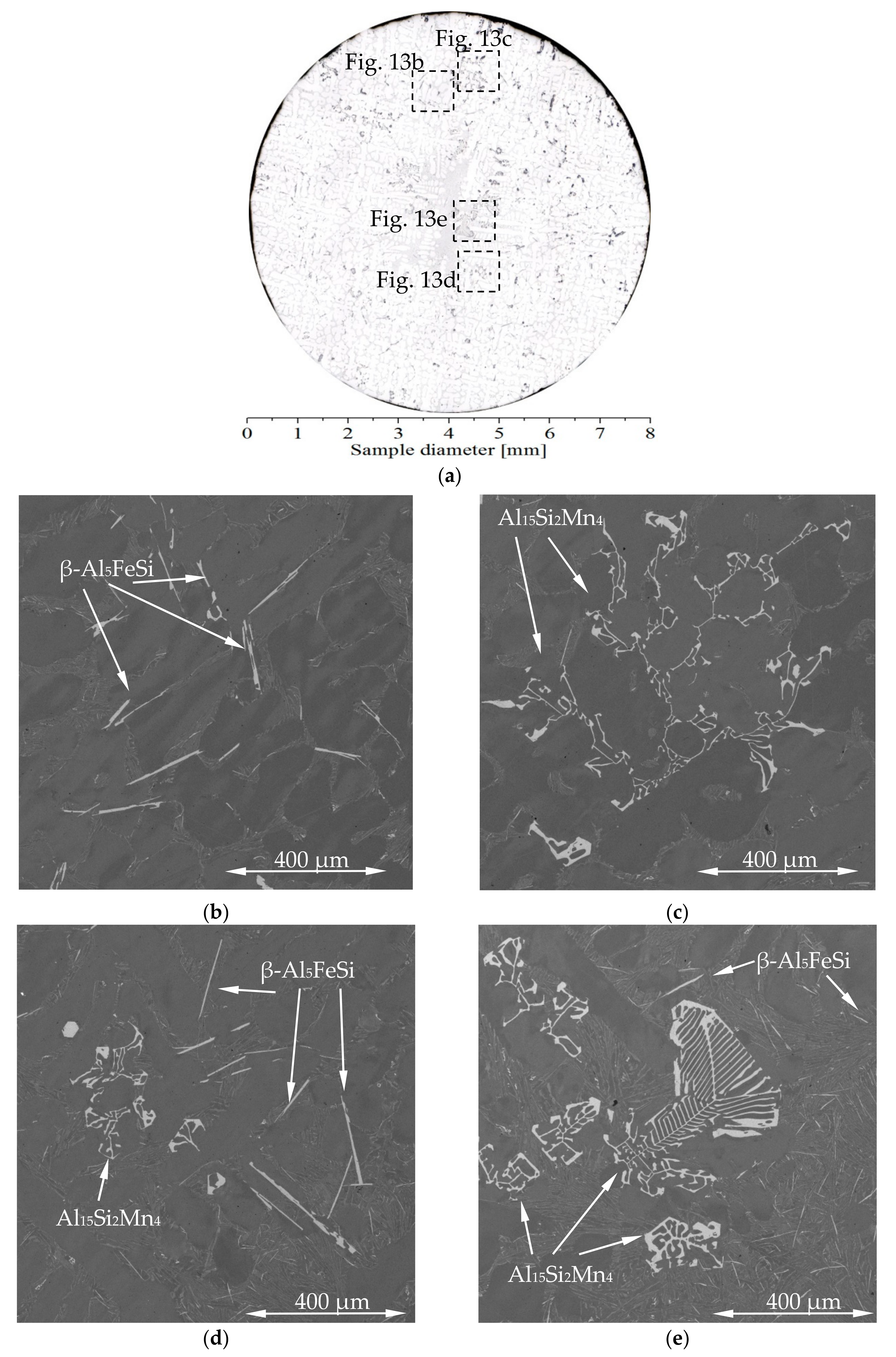
3.3. 2D Maps of Mushy Zone
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Massalski, T.B.; Okamoto, H. Binary Alloy Phase Diagrams; ASM International: Geauga County, OH, USA, 1990; ISBN 978-0871704030. [Google Scholar]
- Kaufman, L.; Bernstein, H. Computer Calculation of Phase Diagrams with Special Reference to Refractory Metals; Academic Press: New York, NY, USA, 1970; ISBN 9780124020504. [Google Scholar]
- Lukas, H.; Fries, S.G.; Sundman, B. Computational Thermodynamics—The Calphad. Method; Cambridge University Press: Cambridge, UK, 2007; ISBN 978-0-521-86811. [Google Scholar]
- Hillert, M. Phase Equilibria, Phase Diagrams and Phase Transformations—Their Thermodynamic Basis; Cambridge University Press: Cambridge, UK, 1998; ISBN 978-0-521-85351-4. [Google Scholar]
- Shi, P.; Sundman, B. Thermo-Calc. Software System—Thermodynamic Framework and Data; Thermo-Calc Software AB; Foundation of Computational Thermodynamics: Stockholm, Sweden, 2013. [Google Scholar]
- Firouzdor, V.; Rajabi, M.; Nejati, E.; Khomamizadeh, F. Effect of microstructural constituents on the thermal fatigue life of A319 Al alloy. Mater. Sci. Eng. A 2007, 454–455, 528–535. [Google Scholar] [CrossRef]
- Yi, J.Z.; Gao, Y.X.; Lee, P.D.; Lindley, T.C. Effect of Fe-content on fatigue crack initiation and propagation in a cast aluminium-silicon alloy (A356-T6). Mater. Sci. Eng. A 2004, 386, 396–407. [Google Scholar] [CrossRef]
- Ma, Z.; Samuel, A.M.; Samuel, F.H.; Doty, H.W.; Valtierra, S. A study of tensile properties in Al-Si-Cu and Al-Si-Mg alloys. Effect of beta-iron intermetallics and porosity. Mater. Sci. Eng. A 2008, 490, 36–51. [Google Scholar] [CrossRef]
- Kim, H.Y.; Han, S.W.; Lee, H.M. The influence of Mn and Cr on the tensile properties of A356-0.20Fe alloy. Mater. Lett. 2006, 60, 1880–1883. [Google Scholar] [CrossRef]
- Hwang, J.Y.; Doty, H.W.; Kaufman, M.J. The effects of Mn additions on the microstructure and mechanical properties of Al-Si-Cu casting alloys. Mater. Sci. Eng. A 2008, 488, 496–504. [Google Scholar] [CrossRef]
- Sreeja Kumari, S.S.; Pillai, R.M.; Pai, B.C. A study on the structural, age hardening and mechanical characteristics of Mn and Ca added Al-7Si-0.3Mg-0.6Fe alloy. J. Alloys Compd. 2008, 453, 167–173. [Google Scholar] [CrossRef]
- Khalifa, W.; Samuel, F.H.; Gruzleski, J.E. Iron intermetallic phases in Al corner of the Al-Si-Fe system. Metall. Mater. Trans. A 2003, 34, 807–825. [Google Scholar] [CrossRef]
- Bridgman, P.W. Certain physical properties of single crystals of tungsten, antimony, bismuth, tellurium, cadmium, zinc and tin. Proc. Am. Acad. Arts Sci. 1925, 60, 305–383. [Google Scholar] [CrossRef]
- Stockbarger, D.C. The production of large single crystals of lithium fluoride. Rev. Sci. Instrum. 1936, 7, 133–136. [Google Scholar] [CrossRef]
- Jung, H.; Mangelinck-Noël, N.; Nguyen-Thi, H.; Billia, B. Columnar to equiaxed transition during directional solidification in refined Al-based alloys. J. Alloys Compd. 2009, 484, 739–746. [Google Scholar] [CrossRef]
- Ruvalcaba, D.; Mathiesen, R.H.; Eskin, D.G.; Arnberg, L.; Katgerman, L. In situ observations of dendritic fragmentation due to local solute-enrichment during directional solidification of an aluminum alloy. Acta Mater. 2007, 55, 4287–4292. [Google Scholar] [CrossRef]
- Thermo-Calc 4.1—Software Package from Thermo-Calc Software AB, Stockholm, Sweden. Available online: www.thermocalc.se (accessed on 16 March 2015).
- Alkemper, J.; Sous, S.; Stoker, C.; Ratke, L. Directional solidification in an aerogel furnace with high resolution optical temperature measurements. J. Cryst. Growth 1998, 191, 252–260. [Google Scholar] [CrossRef]
- Steinbach, S.; Ratke, L. In situ optical determination of fraction solid. Scr. Mater. 2004, 50, 1135–1138. [Google Scholar] [CrossRef]
- Steinbach, S. The Influence of Fluid Flow on the Microstructure Evolution of Directional Solidified Al-Si and A-Si-Mg Alloys. Ph.D. Thesis, Rheinisch-Westfälische Technische Hochschule (RWTH) Aachen University, Aachen, Germany, 28 November 2005. (In German). [Google Scholar]
- Mikolajczak, P.; Ratke, L. Effect of stirring induced by rotating magnetic field on β-Al5FeSi intermetallic phases during directional solidification in AlSi alloys. Int. J. Cast Met. Res. 2013, 26, 339–353. [Google Scholar] [CrossRef]
- Mikolajczak, P.; Ratke, L. Interplay between melt flow and the 3D distribution and morphology of Fe-rich phases in AlSi alloys. Metall. Mater. Trans. A 2015, 46, 1312–1327. [Google Scholar] [CrossRef]
- Hainke, M. Computation of Convection and Alloys Solidification with the Software Package CrysVUn. Ph.D. Thesis, Technical Faculty of Friedrich-Alexander Universität Erlangen-Nürnberg, Erlangen, Germany, July 2004. [Google Scholar]
- Steinbach, S.; Ratke, L. The influence of fluid flow on the microstructure of directionally solidified AlSi-base alloys. Metall. Mater. Trans. A 2007, 38, 1388–1394. [Google Scholar] [CrossRef]
- Steinbach, S.; Ratke, L. Experimental study on interaction of fluid flow and solidification in Al-Si-Cu alloys. Int. J. Cast Met. Res. 2007, 20, 140–144. [Google Scholar] [CrossRef]
- Steinbach, S.; Euskirchen, N.; Witusewicz, V.; Ratke, L. Fluid flow effects on intermetallic phases in Al-cast alloys. Trans. Indian Inst. Met. 2007, 60, 137–141. [Google Scholar]
- Kovacs, J.; Sveda, M.; Ronafoldi, A.; Roosz, A. Influence of high rotating magnetic field (RMF) on the solidified structure of Al-7 wt. % Si-1 wt. % Fe alloy. In Proceedings of the 6th Decennial Conference on Solidification Processing, Beaumont Estate, Old Windsor, UK, 25–28 July 2017. [Google Scholar]
- Budenkowa, O.; Nagy, C.; Du Terrail Couvat, Y.; Steinbach, S.; Roosz, A.; Fautrelle, Y. Comparison of 2D and 3D simulations of solidification of binary and ternary Al-based alloys under RMF. In Proceedings of the 6th Decennial Conference on Solidification Processing, Beaumont Estate, Old Windsor, UK, 25–28 July 2017. [Google Scholar]
- Kovacs, J.; Ronafoldi, A.; Roosz, A. Unidirectional solidification of Pb-Sn alloys in a rotating magnetic field. Mater. Sci. Forum 2013, 790–791, 408–413. [Google Scholar] [CrossRef]
- Sveda, M.; Sycheva, A.; Kovacs, J.; Ronafoldi, A.; Roosz, A. The effect of rotating magnetic field on the solidified structure of Sn-Cd peritectic alloys. Mater. Sci. Forum 2013, 790–791, 414–419. [Google Scholar] [CrossRef]
- Mikolajczak, P.; Ratke, L. Thermodynamic assessment of mushy zone in directional solidification. Arch. Foundry Eng. AFE 2015, 15, 101–109. [Google Scholar] [CrossRef]
- Steinbach, S.; Ratke, L. In situ optical determination of fraction solid of Al-Si-Mg alloys. Mater. Sci. Forum 2006, 508, 491–496. [Google Scholar] [CrossRef]
- Mikolajczak, P.; Genau, A.; Ratke, L. Mushy Zone Morphology Prediction with Application of CALPHAD Technique. Metals 2017, 7, 363. [Google Scholar] [CrossRef]
- Mikolajczak, P.; Ratke, L. Three dimensional morphology of Mn rich intermetallics in AlSi alloys investigated with X-ray tomography. Mater. Sci. Forum 2014, 790–791, 335–340. [Google Scholar] [CrossRef]
- Mikolajczak, P. Microstructural evolution in AlMgSi Alloys during solidification under electromagnetic stirring. Metals 2017, 7, 89. [Google Scholar] [CrossRef]
- Inoue, A.; Takeuchi, A. Recent progress in bulk glassy alloys. Mater. Trans. 2002, 43, 1892–1906. [Google Scholar] [CrossRef]
- Ghidelli, M.; Volland, A.; Blandin, J.-J.; Pardoen, T.; Raskin, J.-P.; Mompiou, F.; Djemia, P.; Gravier, S. Exploring the mechanical size effects in Zr65Ni35 thin film metallic glasses. J. Alloys Compd. 2014, 615, 90–92. [Google Scholar] [CrossRef]
- Ghidelli, M.; Gravier, S.; Blandin, J.-J.; Pardoen, T.; Raskin, J.-P.; Mompiou, F. Compositional-induced structural change in ZrxNi100−x thin film metallic glasses. J. Alloys Compd. 2014, 615, 348–351. [Google Scholar] [CrossRef]
- Hainke, M.; Friedrich, J.; Müller, G. Numerical study on directional solidification of AlSi alloys with rotating magnetic field under microgravity conditions. J. Mater. Sci. 2004, 39, 2011–2015. [Google Scholar] [CrossRef]
- Roosz, A.; Kovacs, J.; Ronafoldi, A.; Kovacs, A. Effect of high rotating magnetic field on the solidified structure of Al-7 wt. % Si-1 wt. % Fe alloy. Mater. Sci. Forum 2013, 752, 57–65. [Google Scholar] [CrossRef]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mikolajczak, P.; Genau, A.; Janiszewski, J.; Ratke, L. Thermo-Calc Prediction of Mushy Zone in AlSiFeMn Alloys. Metals 2017, 7, 506. https://doi.org/10.3390/met7110506
Mikolajczak P, Genau A, Janiszewski J, Ratke L. Thermo-Calc Prediction of Mushy Zone in AlSiFeMn Alloys. Metals. 2017; 7(11):506. https://doi.org/10.3390/met7110506
Chicago/Turabian StyleMikolajczak, Piotr, Amber Genau, Jerzy Janiszewski, and Lorenz Ratke. 2017. "Thermo-Calc Prediction of Mushy Zone in AlSiFeMn Alloys" Metals 7, no. 11: 506. https://doi.org/10.3390/met7110506
APA StyleMikolajczak, P., Genau, A., Janiszewski, J., & Ratke, L. (2017). Thermo-Calc Prediction of Mushy Zone in AlSiFeMn Alloys. Metals, 7(11), 506. https://doi.org/10.3390/met7110506




