Influence of Yttrium on the Phase Composition of the Ti-Al System Obtained by the ‘Hydride Technology’
Abstract
1. Introduction
2. Materials and Methods
2.1. Obtaining Alloy
2.2. Research Methods
3. Results
4. Discussion
5. Conclusions
- Layered compositions, based on intermetallic phases and the alloying additive of 49Ti–49Al-2Y (at.%) yttrium, have been obtained using the “hydride technology”. This technology allows, under hydrogenation-dehydrogenation conditions, to alloy the Ti-Al system with up to 2 at.% yttrium;
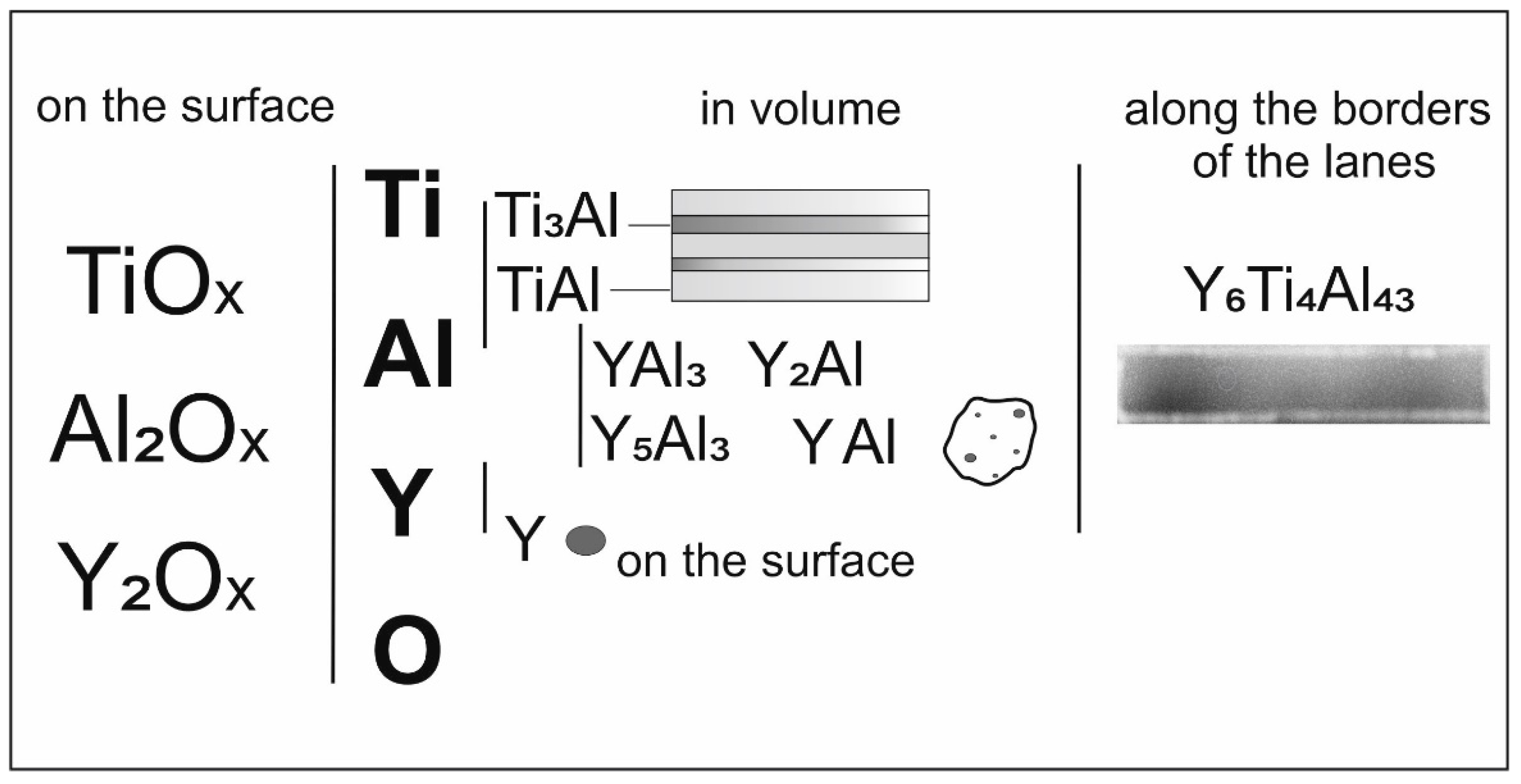
- The following phase compositions have been formed in the sample: Ti3Al, TiAl, Y2O3, Al, α-Ti, Ti3Al5, Y2Al, Y5Al3, YAl3, YAl, Y6Ti4Al43. A lamellar structure has been formed (Ti3Al and TiAl);
- Yttrium is distributed in the matrix of intermetallic phases with a simultaneous increase (two times) in the thickness of the layers and/or separate phases are formed: Y2Al, Y5Al3, YAl3, and YAl. As the alloy particle moves away from the surface into the volume, there is a gradient of changes in the concentration of yttrium and oxygen and, accordingly, a change in the qualitative and quantitative phase composition of yttrium-containing phases;
- The Y6Ti4Al43 (P63/mcm) phase forms at the grain boundaries and/or in the grain volume;
- An oxide layer (Y2O3, TiO2, and Al2O3) is formed at the surface boundary which prevents the sample oxidation.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, Z.; Jiang, H.; Wang, Y.; Zhang, D.; Yan, D.; Rong, L. Effect of minor Sc addition on microstructure and stress corrosion cracking behavior of medium strength Al–Zn–Mg alloy. J. Mater. Sci. Technol. 2018, 34, 1172–1179. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, H.; Yi, D.; Wang, B.; Wang, H. Comparative study of Sc and Er addition on microstructure, mechanical properties, and electrical conductivity of Al-0.2 Zr-based alloy cables. Mater. Charact. 2018, 145, 126–134. [Google Scholar] [CrossRef]
- Wu, S.H.; Zhang, P.; Shao, D.; Cheng, P.M.; Kuang, J.; Wu, K.; Zhang, J.Y.; Liu, G.; Sun, J. Grain size-dependent Sc microalloying effect on the yield strength-pitting corrosion correlation in Al-Cu alloys. Mat. Sci. Eng. A Struct. 2018, 721, 200–214. [Google Scholar] [CrossRef]
- Belov, N.; Naumova, E.; Akopyan, T. Effect of 0.3% Sc on microstructure, phase composition and hardening of Al–Ca–Si eutectic alloys. Trans. Nonferrous Met. Soc. China 2017, 27, 741–746. [Google Scholar] [CrossRef]
- Erdeniz, D.; Nasim, W.; Malik, J.; Yost, A.R.; Park, S.; De Luca, A.; Vo, N.Q.; Karaman, I.; Mansoor, B.; Seidman, D.N.; et al. Effect of vanadium micro-alloying on the microstructural evolution and creep behavior of Al-Er-Sc-Zr-Si alloys. Acta Mater. 2017, 124, 501–512. [Google Scholar] [CrossRef]
- Li, J.H.; Wiessner, M.; Albu, M.; Wurster, S.; Sartory, B.; Hofer, F.; Schumacher, P. Correlative characterization of primary Al3(Sc, Zr) phase in an Al–Zn–Mg based alloy. Mater. Charact. 2015, 102, 62–70. [Google Scholar] [CrossRef]
- Saumitra, S.; Todorova, T.Z.; Zwanziger, J.W. Temperature dependent lattice misfit and coherency of Al3X (X = Sc, Zr, Ti and Nb) particles in an Al matrix. Acta Mater. 2015, 89, 109–115. [Google Scholar]
- Dorin, T.; Ramajayam, M.; Lamb, J.; Langan, T. Effect of Sc and Zr additions on the microstructure/strength of Al–Cu binary alloys. Mater. Sci. Eng. 2017, 707, 58–64. [Google Scholar] [CrossRef]
- Hallem, H.; Lefebvre, W.; Forbord, B.; Danoix, F.; Marthinsen, K. The formation of Al3(ScxZryHf1−x−y)-dispersoids in aluminium alloys. Mater. Sci. Eng. 2006, 421, 154–160. [Google Scholar] [CrossRef]
- Hallem, H.; Forbord, B.; Marthinsen, K. An investigation of dilute Al–Hf and Al–Hf–Si alloys. Mater. Sci. Eng. 2004, 387–389, 940–943. [Google Scholar] [CrossRef]
- Seijirau, S.; Fukai, Y. Metal-Hydrogen Systems: Fundamentals and Applications. In Proceedings of the International Symposium on Research Involving Palladium and Other Platinum Group Metals, Conference Centre, Tokyo, Japan, 1 November 1994. [Google Scholar]
- Daou, J.N.; Vajda, P. Hydrogen ordering and metal-semiconductor transitions in the system YH2+x. Phys. Rev. B Condens. Matter. 1992, 45, 10907–10913. [Google Scholar] [CrossRef] [PubMed]
- Van Gogh, A.T.M.; van der Molen, S.J.; Kerssemakers, J.W.J.; Koeman, N.J.; Griessen, R. Performance enhancement of metal-hydride switchable mirrors using Pd/AlOx composite cap layers. Appl. Phys. Lett. 2000, 6, 815–817. [Google Scholar] [CrossRef]
- Weaver, J.H.; Peterson, D.T.; Benbow, R.L. Electronic structure of metal hydrides. Phys. Rev. B 1979, 20, 5301. [Google Scholar] [CrossRef]
- Snitovsky, Y.P. Phase composition and concentration of yttrium in films during the deposition of aluminum and aluminum alloys from the gas phase. Bull. Yugra State Univ. 2021, 4, 16–31. [Google Scholar] [CrossRef]
- Yu, L.; Tang, J.; Qiao, J.; Wang, H.; Wang, Y.; Apreutesei, M.; Chamas, M.; Duan, M. Effect of yttrium addition on corrosion resistance of Zr-based bulk metallic glasses in NaCl solution. Int. J. Electrochem. Sci. 2017, 12, 6506–6519. [Google Scholar] [CrossRef]
- Peterman, D.J.; Harmon, B.N.; Marchiando, J.; Weaver, J.H. Band theory of ScH2 and YH2. Phys. Rev. B 1979, 19, 4867. [Google Scholar] [CrossRef]
- Wei, Q.; Zhang, Z.Y.; Wang, X.L.; Wang, Y.; Zhao, Y.T. Effect erbium, yttrium on properties and microstructure of 6082 alloy. Mater. Sci. 2016, 34, 529. [Google Scholar]
- Emamy, M.; Nodooshan, H.R.J.; Malekan, A. The microstructure, hardness and tensile properties of Al-15%Mg2Si in situ composite with yttrium addition. Mater. Des. 2011, 8–9, 4559–4566. [Google Scholar] [CrossRef]
- Kim, Y.W. Ordered intermetallic alloys, part III: Gamma titanium aluminides. JOM 1994, 46, 30–39. [Google Scholar] [CrossRef]
- Becker, S.; Rahmel, A.; Schorr, M.; Schutze, M. Mechanism of isothermal oxidation of the intermetallic TiAl and of TiAl alloys. Oxid. Met. 1992, 38, 425–464. [Google Scholar] [CrossRef]
- Heo, S.H.; Kim, I.B.; Han, J.W.; Hwang, W.S.; You, B.D.; Kim, M.S. Comparisons of high temperature oxidation behavior between reactive-sintered and melted Ti-45at.%Al-1.6at.%Mn. Met. Mater. Int. 2000, 6, 449–453. [Google Scholar] [CrossRef]
- Tang, Z.; Niewolak, L.; Shemet, V.; Singheiser, L.; Quadakkers, W.J.; Wang, F.; Wu, W.; Gil, A. First-principles investigation of site. Preference and bonding properties of alloying element in TiAl with O impurity. Mater. Sci. Eng. 2002, 328, 297. [Google Scholar] [CrossRef]
- Yoshihara, M.; Miura, K. Effects of Nb addition on oxidation behavior of TiAl. Intermetallics 1995, 3, 357–363. [Google Scholar] [CrossRef]
- Anada, H.; Shida, Y. The effect of various ternary additives on the oxidation behavior of TiAl in high-temperature air. Oxid. Met. 1996, 45, 197–219. [Google Scholar]
- Brady, M.P.; Smialek, J.L.; Humphrey, D.L.; Smith, J. The role of Cr in promoting protective alumina scale formation by γ-based TiAlCr. Alloys. Acta. Mater. 1997, 45, 2371–2382. [Google Scholar] [CrossRef]
- Maki, K.; Shioda, M.; Sayashi, M. Effect of silicon and niobium on oxidation resistance of titanium aluminide (TiAl) intermetallics. Mater. Sci. Eng. 1992, 153, 591. [Google Scholar] [CrossRef]
- Wu, Y.; Hwang, S.K. High-temperature oxidation of elemental powder metallurgy processed TiAl-Mn-Mo-C alloys with yttrium addition. Acta. Mater. 2004, 45, 1272–1281. [Google Scholar] [CrossRef][Green Version]
- Park, H.S.; Hwang, S.K.; Lee, C.M.; Yoo, Y.C.; Nam, S.W.; Kim, N.J. Microstructural refinement and mechanical properties improvement of elemental powder metallurgy processed Ti-46.6Al-1.4Mn-2Mo alloy by carbon addition. Metall. Mater. Trans. 2001, 32, 251. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Tsukihashi, F. Thermodynamics of yttrium and oxygen in molten zirconium. Metall. Mater. Trans. B 1998, 30, 352–354. [Google Scholar] [CrossRef]
- Menand, A.; Huguet, A.; Nerac-Partaix, A. Interstitial solubility in γ and α2 phases of TiAl-based alloys. Acta. Mater. 1996, 44, 4729–4737. [Google Scholar] [CrossRef]
- Karakchieva, N.; Lepakova, O.; Abzaev, Y.; Sachkov, V.; Kurzina, I. The Influence of Scandium on the Composition and Structure of the Ti-Al Alloy Obtained by “Hydride Technology”. Nanomaterials 2021, 11, 918. [Google Scholar] [CrossRef]
- Belgibayeva, A.; Abzaev, Y.; Karakchieva, N.; Erkasov, R.; Sachkov, V.; Kurzina, I. The Structural and Phase State of the TiAl System Alloyed with Rare-Earth Metals of the Controlled Composition Synthesized by the “Hydride Technology”. Metals 2020, 10, 859. [Google Scholar] [CrossRef]
- Soroka, O.; Sturm, J.M.; van de Kruijs, R.W.E.; Lee, C.J.; Bijkerk, F. Control of YH3 formation and stability via hydrogen surface adsorption and desorption. Appl. Surf. Sci. 2018, 455, 70–74. [Google Scholar] [CrossRef]
- Gromov, V.E.; Sosnin, K.V.; Ivanov, Y.F.; Zenina, E.V.; Rubannikova, Y.A. Formation of structure and phase composition of Ti-Y surface layer by electro explosion and electron-beam treatment. Russ. Univ. Rep. Math. 2016, 21, 850–852. [Google Scholar] [CrossRef]
- Niemann, S.; Jeitschko, W. Ternary Aluminides A6T4Al43 (A = Y, Nd, Sm, Gd-Lu, and U.; T = Ti, V, Nb, and Ta) with Ho6Mo4Al43 Type Structure. J. Solid State Chem. 1995, 116, 131–135. [Google Scholar] [CrossRef]
- Wolff, M.W.; Niemann, S.; Ebel, T.; Jeitschko, W. Magnetic properties of rare-earth transition metal aluminides R6T4Al43 with Ho6Mo4Al43-type structure. J. Magn. Magn. Mater. 2001, 223, 1–15. [Google Scholar] [CrossRef]
- Radu, I.; Li, D.Y.; Llewellyn, R. Tribological behavior of Stellite 21 modified with yttrium. Wear 2004, 257, 1154–1166. [Google Scholar] [CrossRef]
- Raghavan, V. Al-Ti-Y (Aluminum-Titanium-Yttrium). J. Phase Equilib. Diffus. 2005, 26, 191. [Google Scholar] [CrossRef]

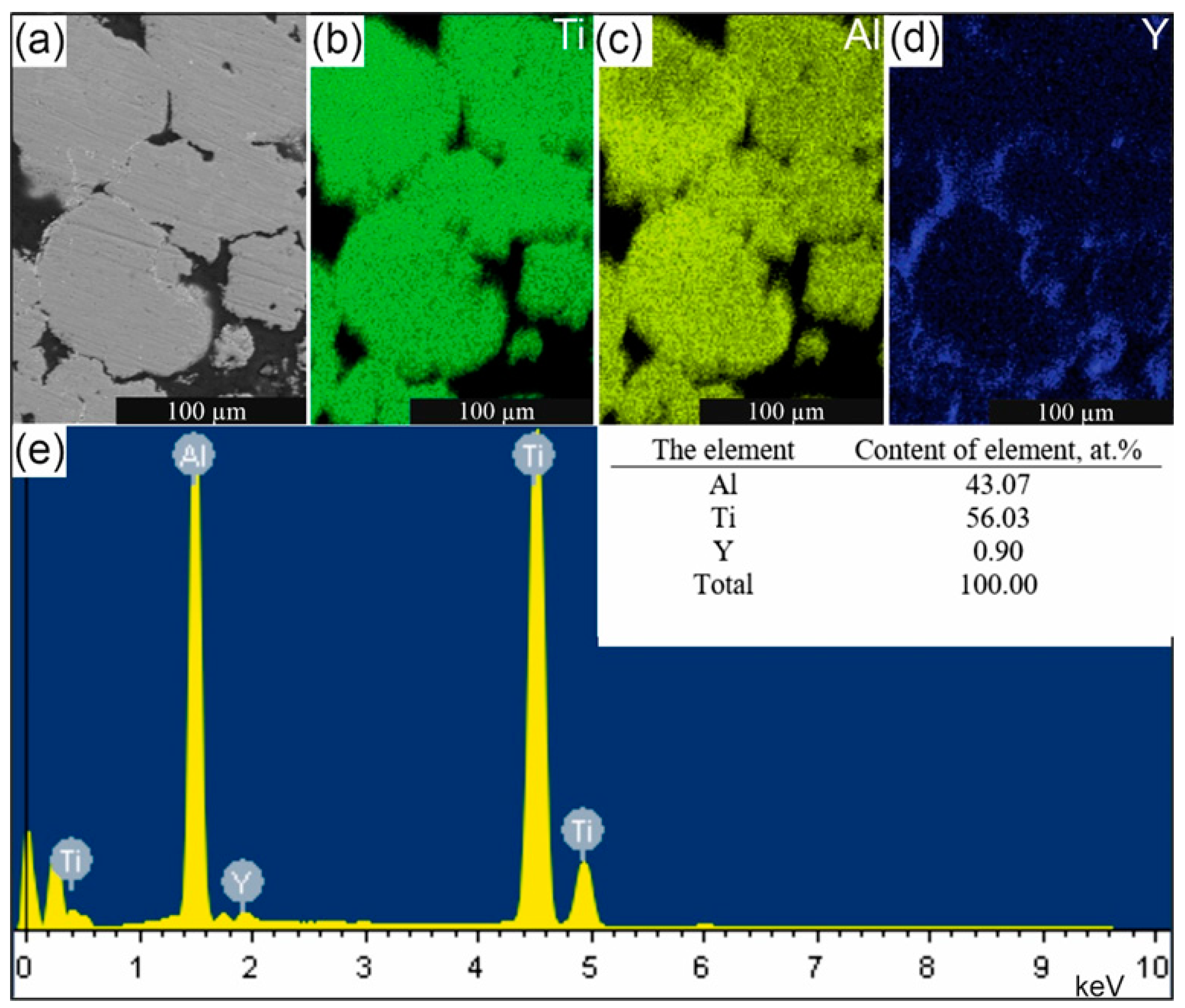





| No. of Spectrum | at.% | Formula | |||
|---|---|---|---|---|---|
| Ti | Al | Y | O | ||
| 1 | 75.12 | 24.09 | - | 0.78 | Ti3Al, TiAl |
| 2 | 61.52 | 38.00 | - | 0.48 | Ti2Al |
| 3 | 4.06 | 1.2 | 67.64 | 27.10 | Y, Y2O3, TiO2, Al2O3 |
| 4 | 35.94 | 19.31 | 37.23 | 7.52 | Ti2Al, Al2O3, Y |
| 5 | 10.12 | 4.61 | 72.11 | 13.17 | Ti2Al, Y, Al2O3, |
| 6 | 54.6 | 29.11 | 0.17 | 16.12 | Ti2Al, Al2O3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karakchieva, N.; Artemenko, A.; Lepakova, O.; Sachkov, V.; Kurzina, I. Influence of Yttrium on the Phase Composition of the Ti-Al System Obtained by the ‘Hydride Technology’. Metals 2022, 12, 1481. https://doi.org/10.3390/met12091481
Karakchieva N, Artemenko A, Lepakova O, Sachkov V, Kurzina I. Influence of Yttrium on the Phase Composition of the Ti-Al System Obtained by the ‘Hydride Technology’. Metals. 2022; 12(9):1481. https://doi.org/10.3390/met12091481
Chicago/Turabian StyleKarakchieva, Natalia, Alina Artemenko, Olga Lepakova, Victor Sachkov, and Irina Kurzina. 2022. "Influence of Yttrium on the Phase Composition of the Ti-Al System Obtained by the ‘Hydride Technology’" Metals 12, no. 9: 1481. https://doi.org/10.3390/met12091481
APA StyleKarakchieva, N., Artemenko, A., Lepakova, O., Sachkov, V., & Kurzina, I. (2022). Influence of Yttrium on the Phase Composition of the Ti-Al System Obtained by the ‘Hydride Technology’. Metals, 12(9), 1481. https://doi.org/10.3390/met12091481










