Intriguing Prospects of a Novel Magnetic Nanohybrid Material: Ferromagnetic FeRh Nanoparticles Grown on Nanodiamonds
Abstract
1. Introduction
2. Materials and Methods
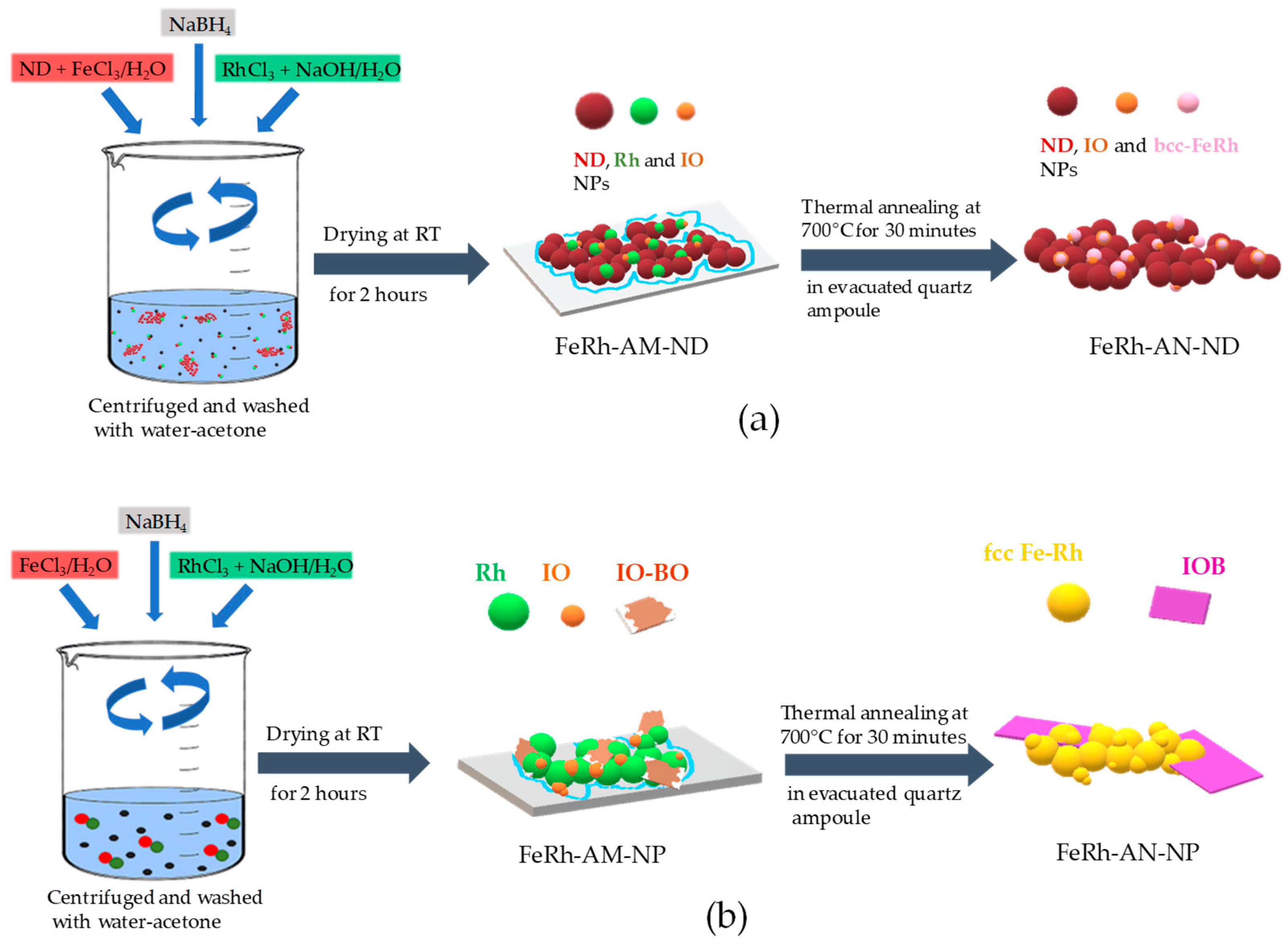
2.1. Materials Synthesis
2.2. Materials Characterization
3. Results
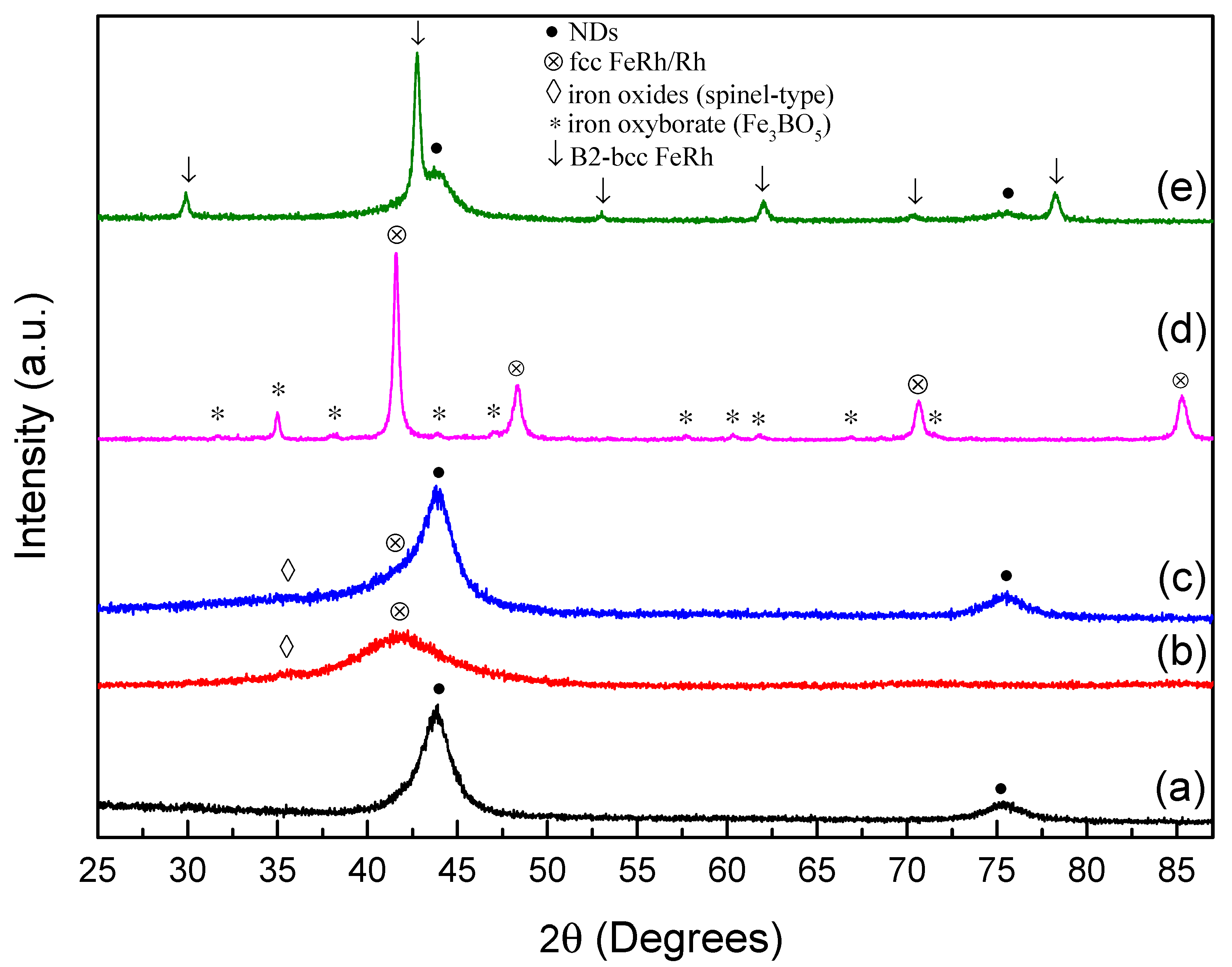
3.1. XRD
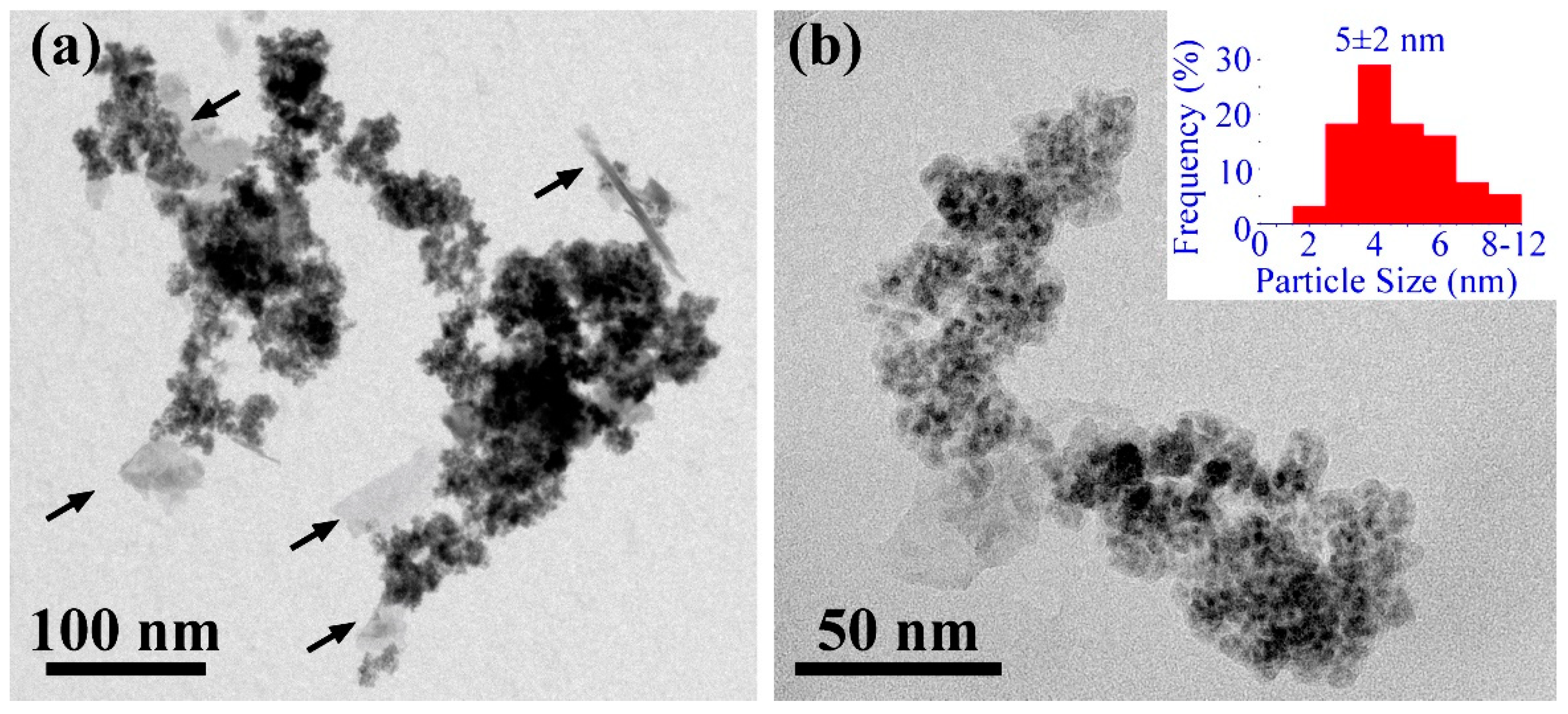
3.2. TEM, STEM and EDS Analysis
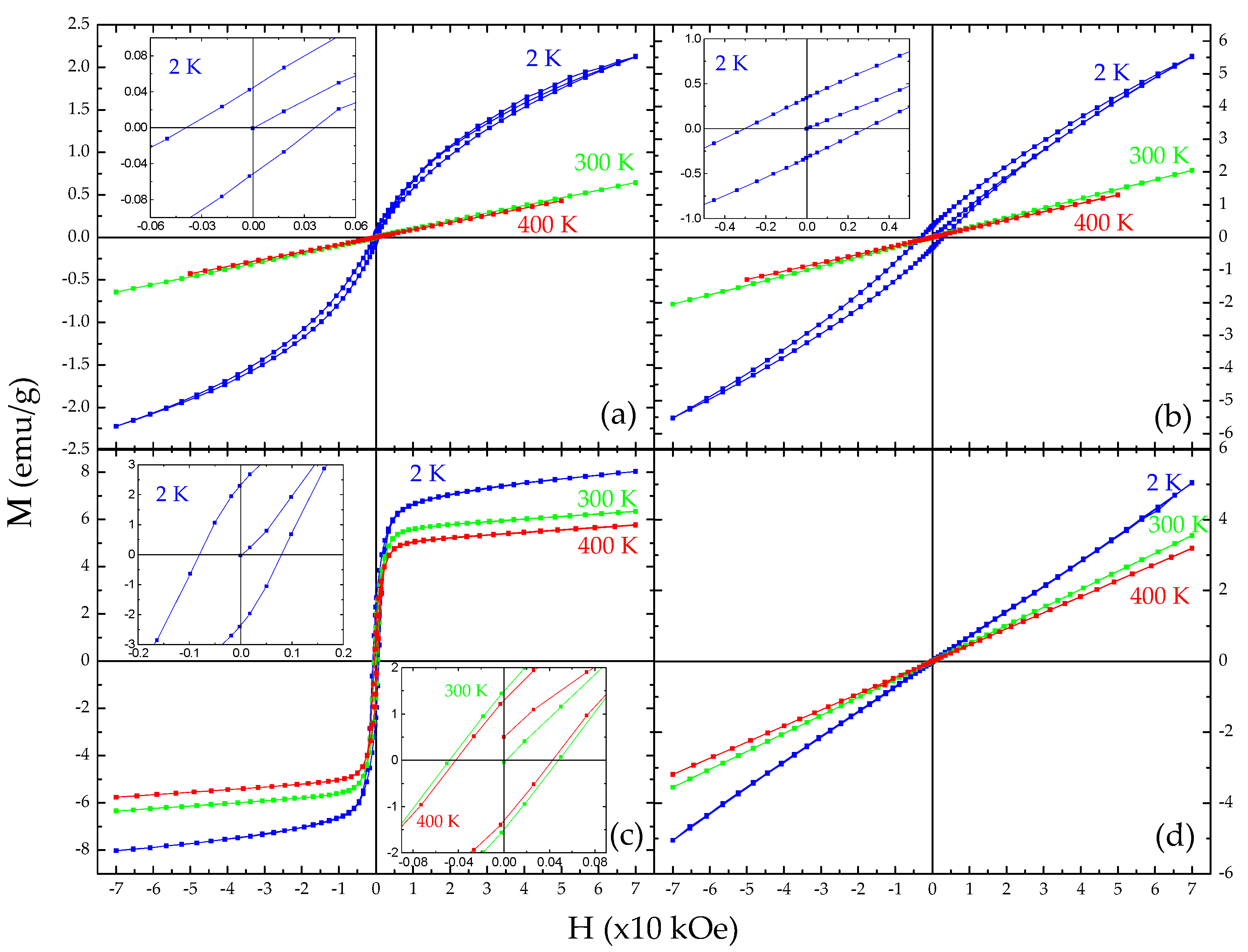
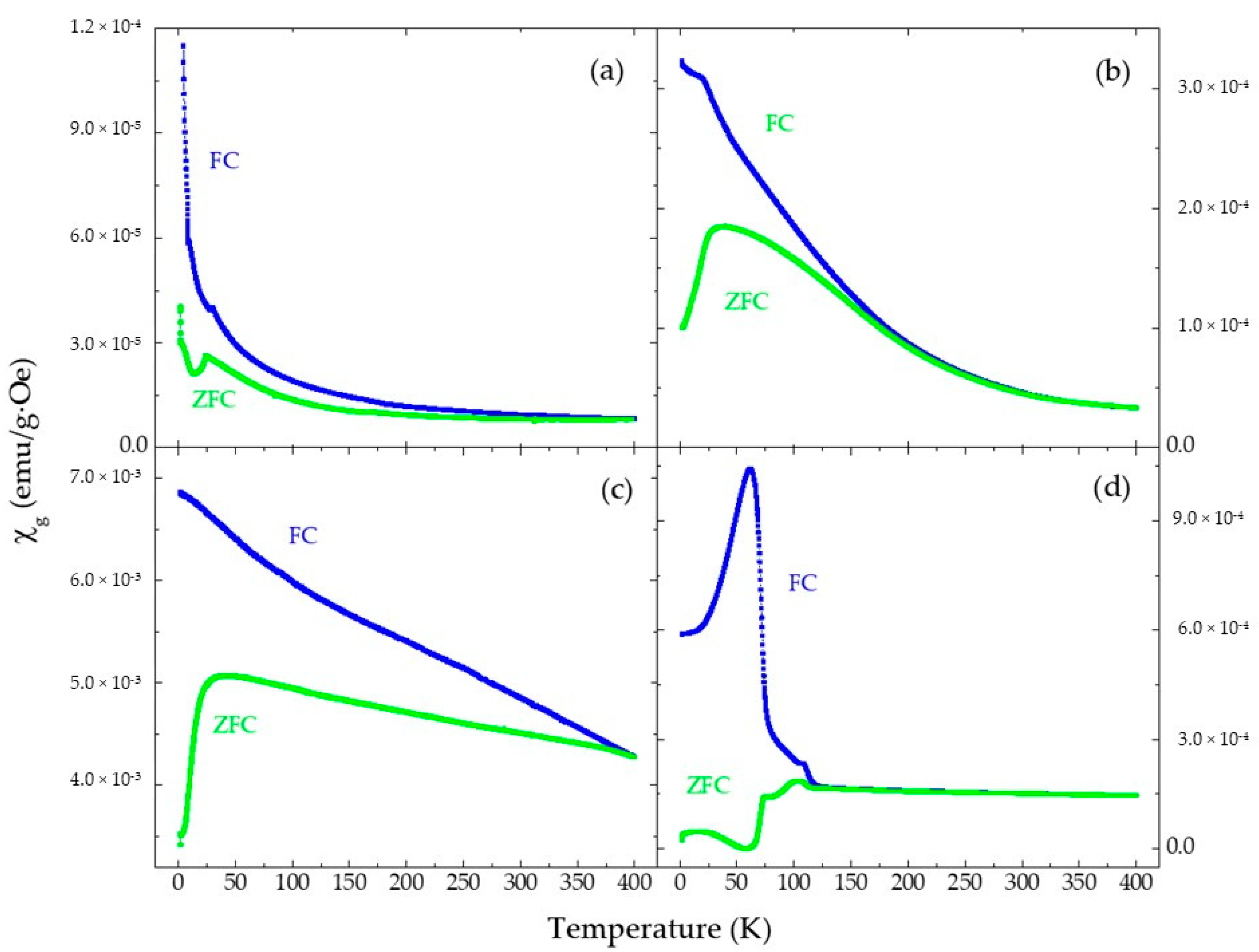
3.3. Magnetization and Magnetic Susceptibility Measurements
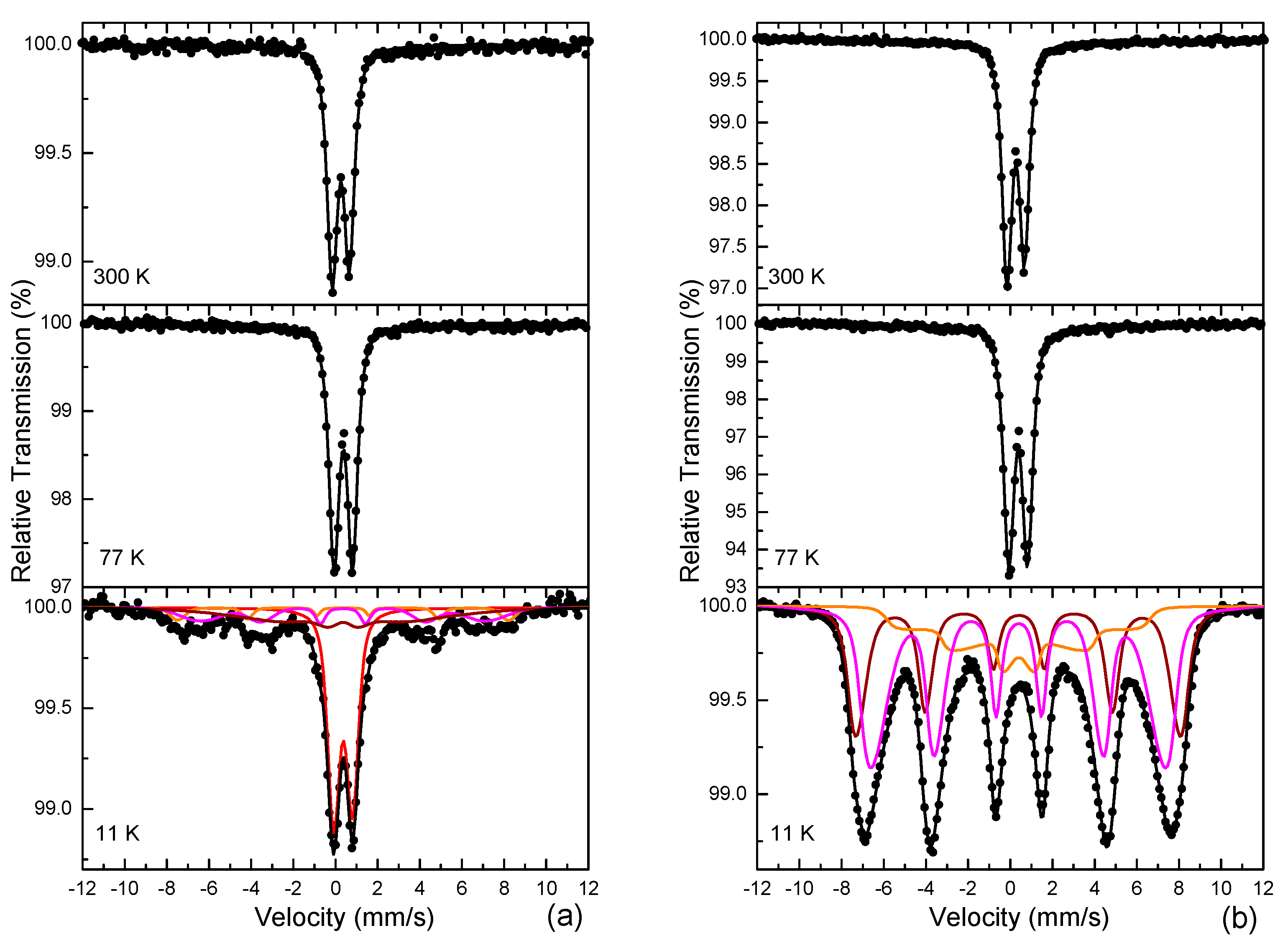
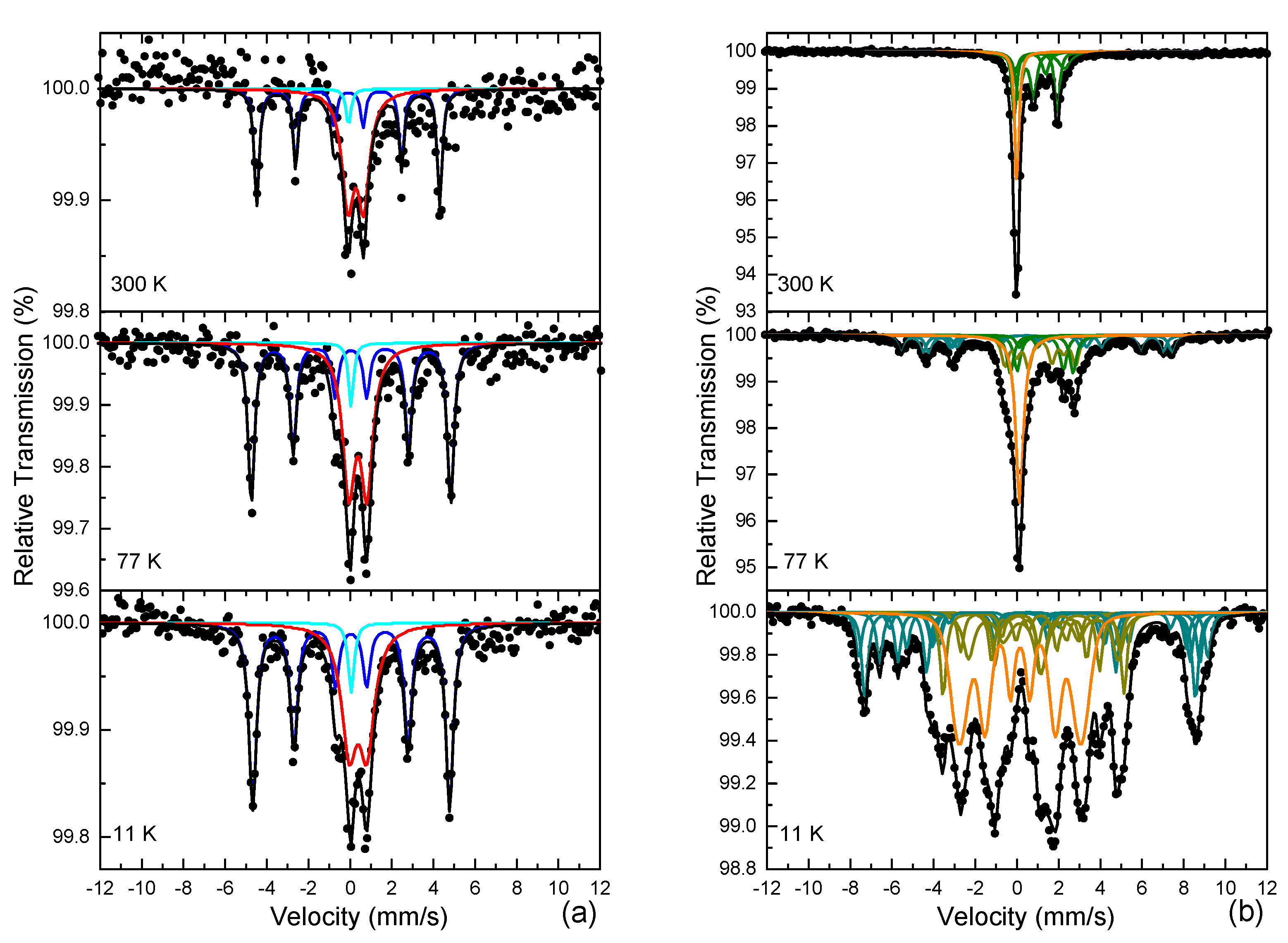
3.4. 57Fe Mössbauer Spectroscopy
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pepperhoff, W.; Acet, M. Constitution and Magnetism of Iron and Its Alloys; Engineering Materials; Springer: Berlin/Heidelberg, Germany, 2001; ISBN 978-3-642-07630-5. [Google Scholar]
- Fallot, M. Les Alliages Du Fer Avec Les Métaux de La Famille Du Platine. Ann. Phys. 1938, 11, 291–332. [Google Scholar] [CrossRef]
- Muldawer, L.; deBergevin, F. Antiferromagnetic-Ferromagnetic Transformation in FeRh. J. Chem. Phys. 1961, 35, 1904–1905. [Google Scholar] [CrossRef]
- Kouvel, J.S.; Hartelius, C.C. Anomalous Magnetic Moments and Transformations in the Ordered Alloy FeRh. In Proceedings of the Seventh Conference on Magnetism and Magnetic Materials; Springer: Boston, MA, USA, 1962; pp. 1343–1344. [Google Scholar]
- Shirane, G.; Chen, C.W.; Flinn, P.A.; Nathans, R. Hyperfine Fields and Magnetic Moments in the Fe–Rh System. J. Appl. Phys. 1963, 34, 1044–1045. [Google Scholar] [CrossRef]
- Lounis, S.; Benakki, M.; Demangeat, C. Ferromagnetic Stabilization of Ordered B2 FeRh Thin Films. Phys. Rev. B 2003, 67, 094432. [Google Scholar] [CrossRef]
- Pressacco, F.; Uhlίř, V.; Gatti, M.; Bendounan, A.; Fullerton, E.E.; Sirotti, F. Stable Room-Temperature Ferromagnetic Phase at the FeRh(100) Surface. Sci. Rep. 2016, 6, 22383. [Google Scholar] [CrossRef] [PubMed]
- Hofer, E.M.; Cucka, P. Magnetic Properties of Rh-Rich FeRh Alloy. J. Phys. Chem. Solids 1966, 27, 1552–1555. [Google Scholar] [CrossRef]
- Lommel, J.M.; Kouvel, J.S. Effects of Mechanical and Thermal Treatment on the Structure and Magnetic Transitions in FeRh. J. Appl. Phys. 1967, 38, 1263–1264. [Google Scholar] [CrossRef]
- Kim, J.; Ramesh, R.; Kioussis, N. Revealing the Hidden Structural Phases of FeRh. Phys. Rev. B 2016, 94, 180407. [Google Scholar] [CrossRef]
- Zakharov, A.I.; Kadomtseva, A.M.; Levitin, R.Z.; Ponyatovskil, E.G. Magnetic and Magnetoelastic Properties of a Metamagnetic Iron-Rhodium Alloy. J. Exptl. Theoret. Phys. 1964, 19, 2003–2010. [Google Scholar]
- Miyajima, H.; Yuasa, S. Structural Phase Transition and Magnetic Properties of FeRh1−xCox Alloys. J. Magn. Magn. Mater. 1992, 104–107, 2025–2026. [Google Scholar] [CrossRef]
- Ohtani, Y.; Hatakeyama, I. Antiferro-ferromagnetic Transition and Microstructural Properties in a Sputter Deposited FeRh Thin Film System. J. Appl. Phys. 1993, 74, 3328–3332. [Google Scholar] [CrossRef]
- Ko, H.Y.Y.; Suzuki, T.; Phuoc, N.N.; Cao, J. Fabrication and Characterization of FeRh Nanoparticles. J. Appl. Phys. 2008, 103, 07D508. [Google Scholar] [CrossRef]
- Chao, C.C.; Duwez, P.; Tsuei, C.C. Metastable Fcc Fe–Rh Alloys and the Fe–Rh Phase Diagram. J. Appl. Phys. 1971, 42, 4282–4284. [Google Scholar] [CrossRef]
- Rosenberg, M.; Kuncser, V.; Crisan, O.; Hernando, A.; Navarro, E.; Filoti, G. A Mössbauer Spectroscopy and Magnetic Study of FeRh. J. Magn. Magn. Mater. 1998, 177–181, 135–136. [Google Scholar] [CrossRef]
- Peng, L.S.-J.; Collins, G.S. Point Defects and The B2 to Fcc Transformation in Milled FeRh. MRS Proc. 1997, 481, 631. [Google Scholar] [CrossRef]
- Navarro, E.; Fiorani, D.; Yavari, R.; Rosenberg, M.; Multigner, M.; Hernando, A.; Caciuffo, R.; Rinaldi, D.; Gialanella, S. Low Temperature Magnetic Properties of FCC FeRh Obtained by Ball Milling. Mater. Sci. Forum 1998, 269–272, 133–138. [Google Scholar] [CrossRef]
- Hillion, A.; Cavallin, A.; Vlaic, S.; Tamion, A.; Tournus, F.; Khadra, G.; Dreiser, J.; Piamonteze, C.; Nolting, F.; Rusponi, S.; et al. Low Temperature Ferromagnetism in Chemically Ordered FeRh Nanocrystals. Phys. Rev. Lett. 2013, 110, 087207. [Google Scholar] [CrossRef]
- Herr, U.; Jing, J.; Birringer, R.; Gonser, U.; Gleiter, H. Investigation of Nanocrystalline Iron Materials by Mössbauer Spectroscopy. Appl. Phys. Lett. 1987, 50, 472–474. [Google Scholar] [CrossRef]
- Thiele, J.-U.; Maat, S.; Fullerton, E.E. FeRh/FePt Exchange Spring Films for Thermally Assisted Magnetic Recording Media. Appl. Phys. Lett. 2003, 82, 2859–2861. [Google Scholar] [CrossRef]
- Bordel, C.; Juraszek, J.; Cooke, D.W.; Baldasseroni, C.; Mankovsky, S.; Minár, J.; Ebert, H.; Moyerman, S.; Fullerton, E.E.; Hellman, F. Fe Spin Reorientation across the Metamagnetic Transition in Strained FeRh Thin Films. Phys. Rev. Lett. 2012, 109, 117201. [Google Scholar] [CrossRef]
- Thiele, J.-U.; Maat, S.; Robertson, J.L.; Fullerton, E.E. Magnetic and Structural Properties of FePt–FeRh Exchange Spring Films for Thermally Assisted Magnetic Recording Media. IEEE Trans. Magn. 2004, 40, 2537–2542. [Google Scholar] [CrossRef]
- Wolloch, M.; Gruner, M.E.; Keune, W.; Mohn, P.; Redinger, J.; Hofer, F.; Suess, D.; Podloucky, R.; Landers, J.; Salamon, S.; et al. Impact of Lattice Dynamics on the Phase Stability of Metamagnetic FeRh: Bulk and Thin Films. Phys. Rev. B 2016, 94, 174435. [Google Scholar] [CrossRef]
- Yu, B.; Liu, M.; Egolf, P.W.; Kitanovski, A. A Review of Magnetic Refrigerator and Heat Pump Prototypes Built before the Year 2010. Int. J. Refrig. 2010, 33, 1029–1060. [Google Scholar] [CrossRef]
- Zverev, V.I.; Pyatakov, A.P.; Shtil, A.A.; Tishin, A.M. Novel Applications of Magnetic Materials and Technologies for Medicine. J. Magn. Magn. Mater. 2018, 459, 182–186. [Google Scholar] [CrossRef]
- Astefanoaei, I.; Gimaev, R.; Zverev, V.; Stancu, A. Modelling of Working Parameters of Gd and FeRh Nanoparticles for Magnetic Hyperthermia. Mater. Res. Express 2019, 6, 125089. [Google Scholar] [CrossRef]
- Barbic, M.; Dodd, S.J.; Morris, H.D.; Dilley, N.; Marcheschi, B.; Huston, A.; Harris, T.D.; Koretsky, A.P. Magnetocaloric Materials as Switchable High Contrast Ratio MRI Labels. Magn. Reson. Med. 2019, 81, 2238–2246. [Google Scholar] [CrossRef]
- Marti, X.; Fina, I.; Frontera, C.; Liu, J.; Wadley, P.; He, Q.; Paull, R.J.; Clarkson, J.D.; Kudrnovský, J.; Turek, I.; et al. Room-Temperature Antiferromagnetic Memory Resistor. Nat. Mater. 2014, 13, 367–374. [Google Scholar] [CrossRef]
- Ibarra, M.R.; Algarabel, P.A. Giant Volume Magnetostriction in the FeRh Alloy. Phys. Rev. B 1994, 50, 4196–4199. [Google Scholar] [CrossRef]
- Algarabel, P.A.; Ibarra, M.R.; Marquina, C.; del Moral, A.; Galibert, J.; Iqbal, M.; Askenazy, S. Giant Room-temperature Magnetoresistance in the FeRh Alloy. Appl. Phys. Lett. 1995, 66, 3061–3063. [Google Scholar] [CrossRef]
- Douvalis, A.P.; Bourlinos, A.B.; Tucek, J.; Čépe, K.; Bakas, T.; Zboril, R. Development of Novel FePt/Nanodiamond Hybrid Nanostructures: L10 Phase Size-Growth Suppression and Magnetic Properties. J. Nanopart. Res. 2016, 18, 115. [Google Scholar] [CrossRef]
- Ziogas, P.; Bourlinos, A.B.; Tucek, J.; Malina, O.; Douvalis, A.P. Novel Magnetic Nanohybrids: From Iron Oxide to Iron Carbide Nanoparticles Grown on Nanodiamonds. Magnetochemistry 2020, 6, 73. [Google Scholar] [CrossRef]
- Ho, D. Nanodiamonds: Applications in Biology and Nanoscale Medicine; Springer US: Boston, MA, USA, 2010; ISBN 978-1-4419-0530-7. [Google Scholar]
- Mochalin, V.N.; Shenderova, O.; Ho, D.; Gogotsi, Y. The Properties and Applications of Nanodiamonds. Nat. Nanotechnol. 2012, 7, 11–23. [Google Scholar] [CrossRef]
- Arnault, J.C. Nanodiamonds: Advanced Material Analysis, Properties and Applications; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 978-0-323-43032-6. [Google Scholar]
- Al Zoubi, W.; Kamil, M.P.; Fatimah, S.; Nashrah, N.; Ko, Y.G. Recent Advances in Hybrid Organic-Inorganic Materials with Spatial Architecture for State-of-the-Art Applications. Prog. Mater. Sci. 2020, 112, 100663. [Google Scholar] [CrossRef]
- Bourlinos, A.B.; Panagiotopoulos, I.; Niarchos, D.; Petridis, D. Hydrophilic Co–Pt Alloy Nanoparticles: Synthesis, Characterization, and Perspectives. J. Mater. Res. 2004, 19, 1227–1233. [Google Scholar] [CrossRef]
- Douvalis, A.P.; Polymeros, A.; Bakas, T. IMSG09: A 57Fe-119Sn Mössbauer Spectra Computer Fitting Program with Novel Interactive User Interface. J. Phys. Conf. Ser. 2010, 217, 012014. [Google Scholar] [CrossRef]
- Cullity, B.D.; Stock, S.R. Elements of X-ray Diffraction, 3rd ed.; Pearson Education: London, UK, 2014; ISBN 978-0201610918. [Google Scholar]
- Ayoub, J.P.; Gatel, C.; Roucau, C.; Casanove, M.J. Structure and Chemical Order in FeRh Nanolayers Epitaxially Grown on MgO(001). J. Cryst. Growth 2011, 314, 336–340. [Google Scholar] [CrossRef]
- Douvalis, A.P.; Moukarika, A.; Bakas, T.; Kallias, G.; Papaefthymiou, V. Mössbauer and Magnetization Studies of Fe3BO5. J. Phys. Condens. Matter 2002, 14, 3303. [Google Scholar] [CrossRef]
- He, S.; Zhang, H.; Xing, H.; Li, K.; Cui, H.; Yang, C.; Sun, S.; Zeng, H. Room Temperature Ferromagnetic (Fe1–x Cox)3BO5 Nanorods. Nano Lett. 2014, 14, 3914–3918. [Google Scholar] [CrossRef]
- Mørup, S.; Hansen, M.F. Superparamagnetic Particles. In Handbook of Magnetism and Advanced Magnetic Materials; Kronmüller, H., Parkin, S., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2007; ISBN 978-0-470-02217-7. [Google Scholar]
- Fiorani, D. Surface Effects in Magnetic Nanoparticles. In Nanostructure Science and Technology; Springer: New York, NY, USA, 2005; ISBN 978-0-387-23279-9. [Google Scholar]
- Mørup, S.; Tronc, E. Superparamagnetic Relaxation of Weakly Interacting Particles. Phys. Rev. Lett. 1994, 72, 3278–3281. [Google Scholar] [CrossRef]
- Mørup, S.; Hansen, M.F.; Frandsen, C. Magnetic Interactions between Nanoparticles. Beilstein J. Nanotechnol. 2010, 1, 182–190. [Google Scholar] [CrossRef]
- Mørup, S.; Frandsen, C.; Hansen, M.F. Uniform Excitations in Magnetic Nanoparticles. Beilstein J. Nanotechnol. 2010, 1, 48–54. [Google Scholar] [CrossRef]
- Swinnea, J.S.; Steinfink, H. Crystal Structure and Mossbauer Spectrum of Vonsenite, 2FeO·FeBO3. Am. Mineral. 1983, 68, 827–832. [Google Scholar]
- Paul Attfield, J.; Clarke, J.F.; Perkins, D.A. Magnetic and Crystal Structures of Iron Borates. Phys. B Condens. Matter 1992, 180–181, 581–584. [Google Scholar] [CrossRef]
- Cornell, R.M.; Schwertmann, U. The Iron Oxides: Structure, Properties, Reactions, Occurences and Uses; Wiley-VCH Verlag: Weinheim, Germany, 2003. [Google Scholar]
- Murad, E.; Jhonson, J.H. Iron Oxides and Oxyhydroxides. In Mossbauer Spectroscopy Applied to Inorganic Chemistry; Long, G.J., Ed.; Plenum Press: New York, NY, USA, 1987; Volume II, ISBN 0-306-42507-6. [Google Scholar]
- Mørup, S. Mössbauer Effect in Small Particles. Hyperfine Interact. 1990, 60, 959–973. [Google Scholar] [CrossRef]
- Mørup, S. Magnetic Hyperfine Splitting in Mössbauer Spectra of Microcrystals. J. Magn. Magn. Mater. 1983, 37, 39–50. [Google Scholar] [CrossRef]
- Vignes, A. Extractive Metallurgy; Wiley: London, UK, 2011; ISBN 978-1-84821-287-9. [Google Scholar]
- Swartzendruber, L.J. The Fe−Rh (Iron-Rhodium) System. Bull. Alloy Phase Diagr. 1984, 5, 456–462. [Google Scholar] [CrossRef]
- Okamoto, H. Fe-Rh (Iron-Rhodium). J. Phase Equilibria Diffus. 2011, 32, 472. [Google Scholar] [CrossRef]
- van Driel, J.; Coehoorn, R.; Strijkers, G.J.; Brück, E.; de Boer, F.R. Compositional Dependence of the Giant Magnetoresistance in FexRh1−x Thin Films. J. Appl. Phys. 1999, 85, 1026–1036. [Google Scholar] [CrossRef][Green Version]
- McKinnon, J.B.; Melville, D.; Lee, E.W. The Antiferromagnetic-Ferromagnetic Transition in Iron-Rhodium Alloys. J. Phys. C Solid State Phys. 1970, 3, S46–S58. [Google Scholar] [CrossRef]



| Sample | NDs | γ-FeRh/Rh (fcc) | α′-FeRh (B2-bcc) | Spinel-Type Iron Oxide | Iron Oxyborate (Fe3BO5) |
|---|---|---|---|---|---|
| Pristine NDs | 3 (1) | - | - | - | - |
| FeRh-AM-ND | 3 (1) | non-resolved | - | non-resolved | - |
| FeRh-AM-NP | - | 2 (1) | - | non-resolved | - |
| FeRh-AN-ND | 3 (1) | - | 15 (1) | non-resolved | - |
| FeRh-AN-NP | - | 9 (1) | - | non-resolved | 30 (1) |
| Sample | T (K) | Mmax+ (emu/g) | Mmax− (emu/g) | MR+ (emu/g) | MR− (emu/g) | HC+ (Oe) | HC− (Oe) |
|---|---|---|---|---|---|---|---|
| FeRh-AM-ND | 400 | 0.43 | −0.43 | 0.00 | 0.00 | 0.00 | 0.00 |
| 300 | 0.64 | −0.64 | 0.00 | 0.00 | 0.00 | 0.00 | |
| 2 | 2.23 | −2.22 | 0.04 | −0.05 | 400 | −390 | |
| FeRh-AM-NP | 400 | 1.29 | −1.29 | 0.00 | 0.00 | 0.00 | 0.00 |
| 300 | 2.05 | −2.05 | 0.00 | 0.00 | 0.00 | 0.00 | |
| 2 | 5.53 | −5.53 | 0.34 | −0.33 | 2870 | −3030 | |
| FeRh-AN-ND | 400 | 5.75 | −5.76 | 1.29 | −1.29 | −425 | 425 |
| 300 | 6.35 | −6.35 | 1.43 | −1.56 | 500 | −505 | |
| 2 | 8.03 | −8.03 | 2.29 | −2.40 | 800 | −795 | |
| FeRh-AN-NP | 400 | 3.19 | −3.19 | 0.00 | 0.00 | 0.00 | 0.00 |
| 300 | 3.55 | −3.55 | 0.00 | 0.00 | 0.00 | 0.00 | |
| 2 | 5.03 | −5.06 | 0.00 | 0.00 | 0.00 | 0.00 |
| Sample | IS | Γ/2 | QSC or 2ε | Bhf | ΔBhf or ΔQS | AA | Site–Phase | Color |
|---|---|---|---|---|---|---|---|---|
| mm/s | mm/s | mm/s | kOe | kOe or mm/s | % | |||
| FeRh-AM-ND | 0.35 | 0.22 | 0.81 | 0 | 0.23 | 100 | Fe3+–IO/IHO (SPM) | Black |
| FeRh-AM-NP | 0.36 | 0.22 | 0.83 | 0 | 0.21 | 100 | Fe3+–IO/IHO (SPM) | Black |
| FeRh-AN-ND | 0.03 | 0.14 | −0.02 | 273 | 0.00 | 45 | Fe–bcc B2 α′-FeRh | Blue |
| 0.03 | 0.14 | 0.00 | 0 | 0.00 | 4 | Fe–bcc B2 α′-FeRh (SPM) | Cyan | |
| 0.38 | 0.35 | 0.76 | 0 | 0.00 | 51 | Fe3+–IO/IHO (SPM) | Red | |
| FeRh-AN-NP | 0.11 | 0.10 | 0.00 | 0 | 0.00 | 22 | Fe–fcc γ-FeRh | Orange |
| 0.55 | 0.17 | 0.71 | 0 | 0.00 | 23 | Fe2.ν+–Fe3BO5 | Olive | |
| 0.74 | 0.19 | 1.49 | 0 | 0.00 | 13 | Fe2.5+–Fe3BO5 | Olive | |
| 1.01 | 0.12 | 2.06 | 0 | 0.00 | 25 | Fe2+–Fe3BO5 | Olive | |
| 1.19 | 0.36 | 2.39 | 0 | 0.00 | 17 | Fe2+–Fe3BO5 | Olive |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ziogas, P.; Bourlinos, A.B.; Chatzopoulou, P.; Dimitrakopulos, G.P.; Kehagias, T.; Markou, A.; Douvalis, A.P. Intriguing Prospects of a Novel Magnetic Nanohybrid Material: Ferromagnetic FeRh Nanoparticles Grown on Nanodiamonds. Metals 2022, 12, 1355. https://doi.org/10.3390/met12081355
Ziogas P, Bourlinos AB, Chatzopoulou P, Dimitrakopulos GP, Kehagias T, Markou A, Douvalis AP. Intriguing Prospects of a Novel Magnetic Nanohybrid Material: Ferromagnetic FeRh Nanoparticles Grown on Nanodiamonds. Metals. 2022; 12(8):1355. https://doi.org/10.3390/met12081355
Chicago/Turabian StyleZiogas, Panagiotis, Athanasios B. Bourlinos, Polyxeni Chatzopoulou, George P. Dimitrakopulos, Thomas Kehagias, Anastasios Markou, and Alexios P. Douvalis. 2022. "Intriguing Prospects of a Novel Magnetic Nanohybrid Material: Ferromagnetic FeRh Nanoparticles Grown on Nanodiamonds" Metals 12, no. 8: 1355. https://doi.org/10.3390/met12081355
APA StyleZiogas, P., Bourlinos, A. B., Chatzopoulou, P., Dimitrakopulos, G. P., Kehagias, T., Markou, A., & Douvalis, A. P. (2022). Intriguing Prospects of a Novel Magnetic Nanohybrid Material: Ferromagnetic FeRh Nanoparticles Grown on Nanodiamonds. Metals, 12(8), 1355. https://doi.org/10.3390/met12081355









