Development of a New AuCuZnGe Alloy and Determination of Its Corrosion Properties
Abstract
1. Introduction
- The white Au alloy has a 14 karat (58.5 wt.%) gold content, a widely-used white Au alloy for general jewelry production.
- The Au alloy should be made as white as possible, reducing the need for additional whitening steps for the final jewelry products with galvanic coatings.
- Germanium content should be kept as low as possible, due to its effects on the alloy’s brittleness and hardness.
- The germanium alloy (as a prealloy) should be implemented easily in noble metal alloy production processes (casting, mechanical treatment, rolling, soldering for jewelry).
2. Materials and Methods
3. Results and Discussions
3.1. SEM/EDX Observations
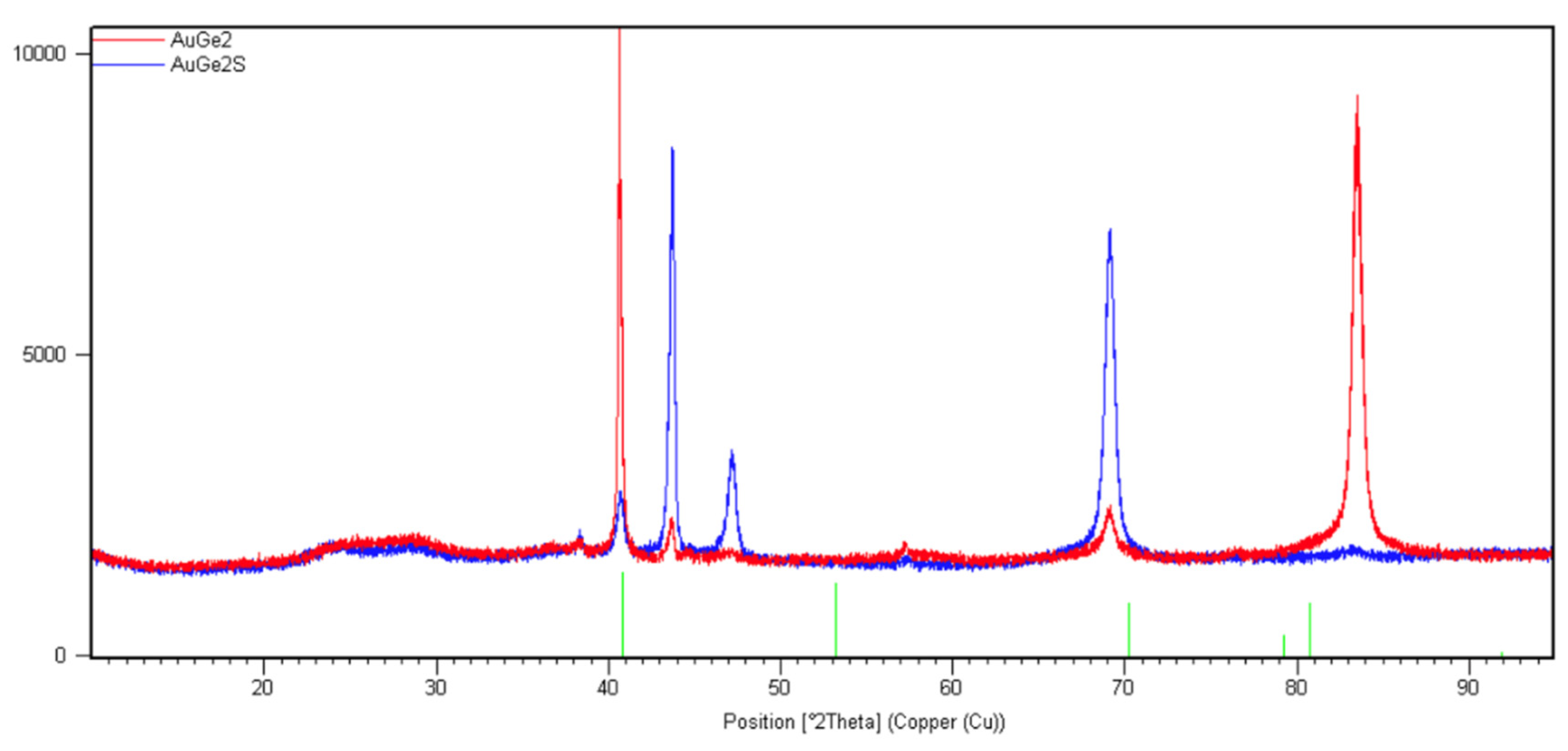
3.2. XRD Analysis
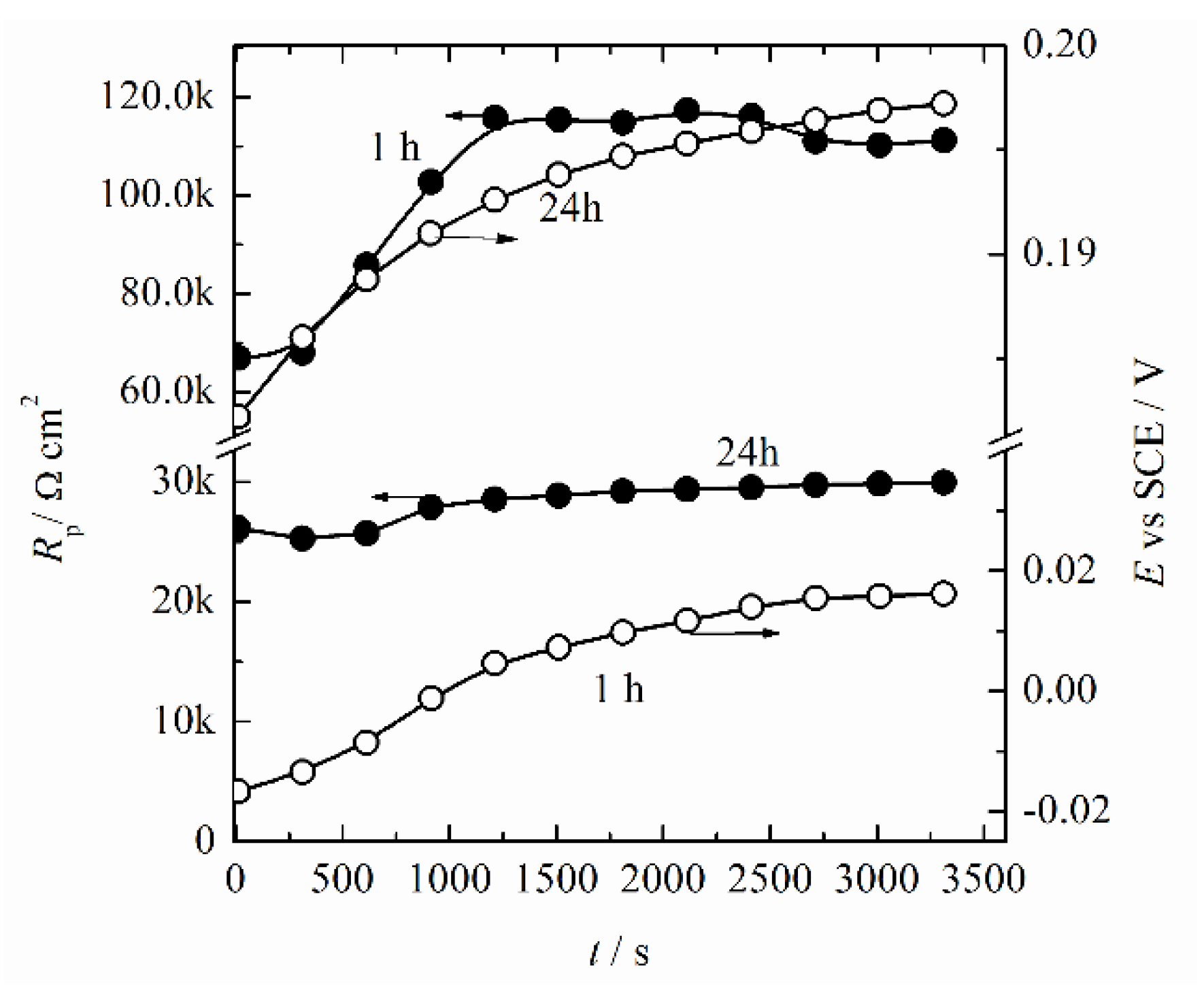
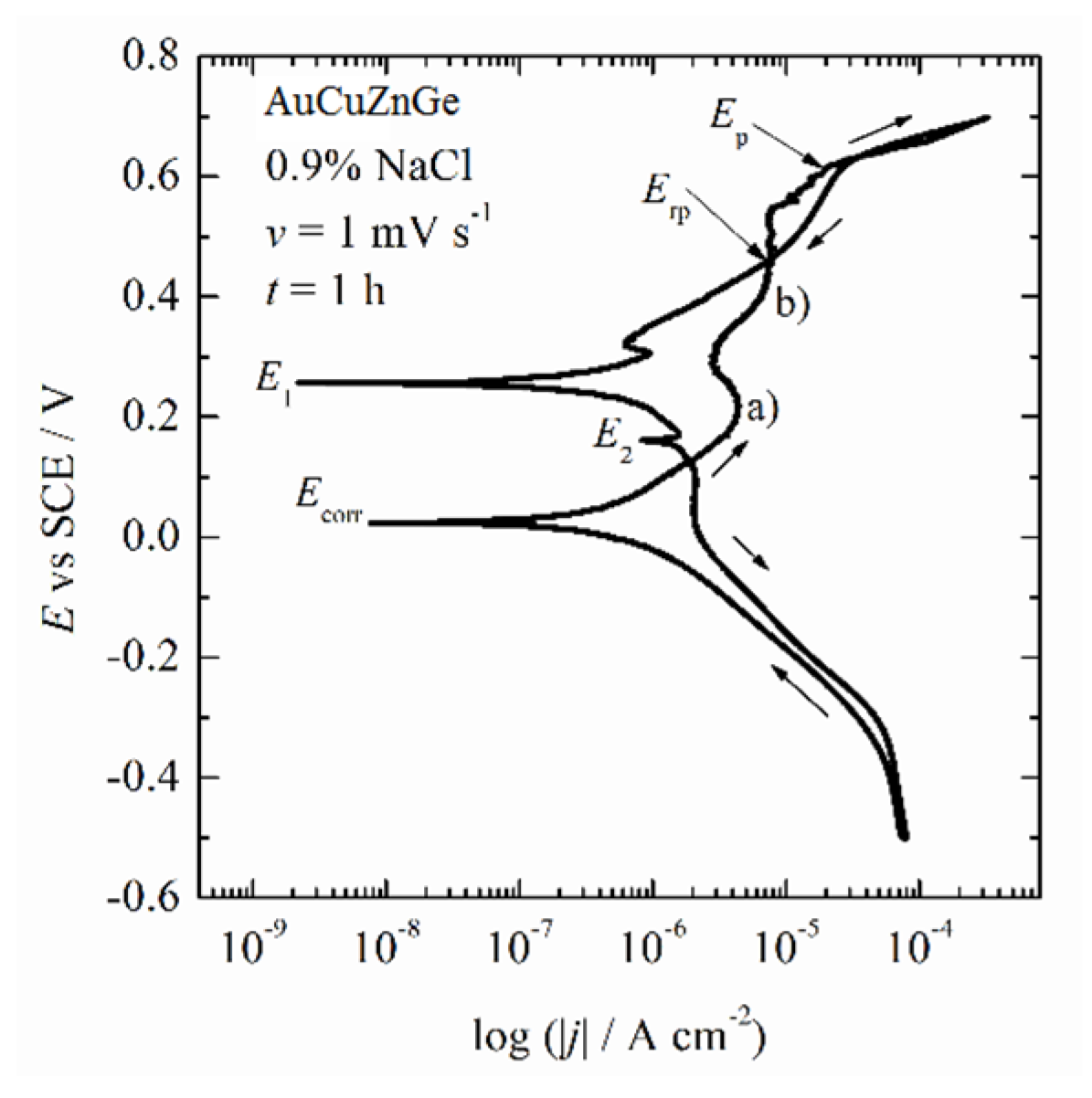
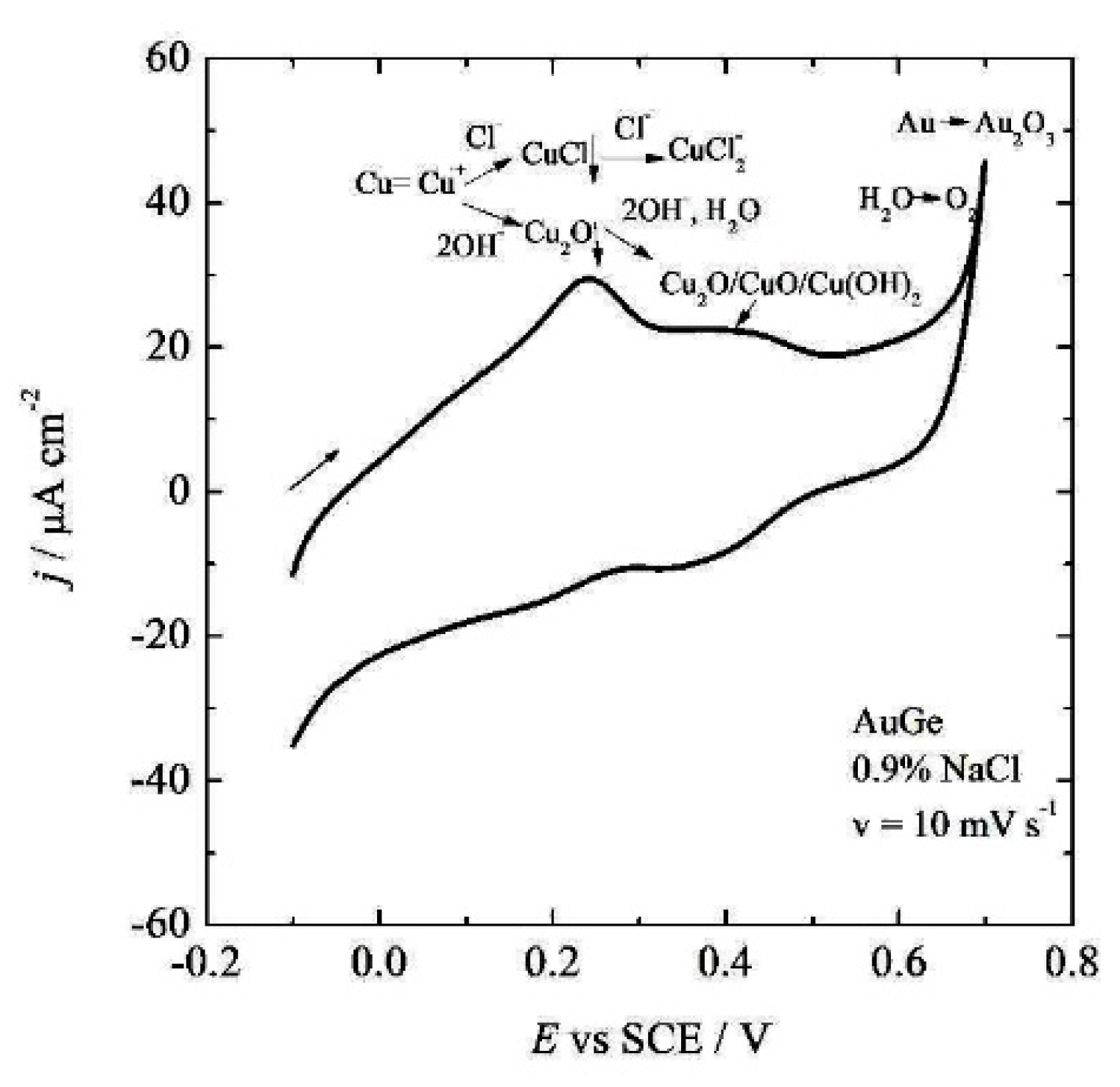
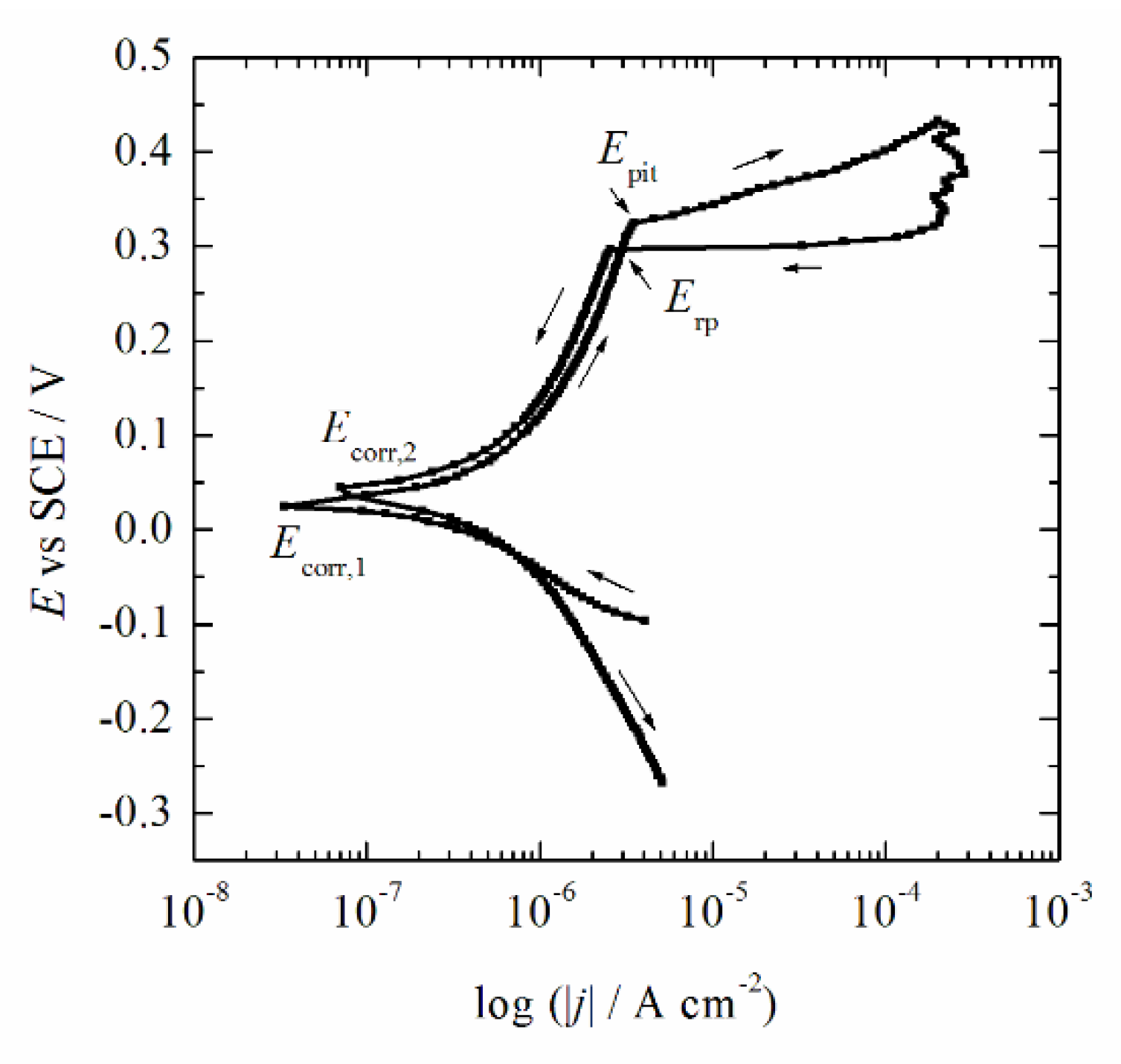
3.3. Corrosion Behavior
4. Conclusions
- It was concluded that during the initial immersion period of 1 h, the alloy established corrosion potential of ~20 mV (SCE), with an estimated corrosion current density lower than 2 × 10−7 A cm−2.
- During the prolonged immersion, complex copper reactions on the alloy surfaces occurred, which led to a decrease in the initial copper contents on the surface. The alloy’s behavior was stabilized after 24 h.
- Using the EDS before and after alloy oxidation, it was concluded that only copper dissolved slightly from the alloy’s surface.
- According to the ADA classification system and our previous works, it was concluded that the investigated new AuCuZnGe alloy belongs to the first group of stability and represents a high noble alloy.
- Even the investigated AuCuZnGe alloy shows acceptable corrosion stability, for the commercialization a lot of work has to be done, mainly in understanding the role of the Ge in the alloy.
5. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cretu, C.; Van Der Lingen, E. Coloured Gold Alloys. Gold Bull. 1999, 32, 115–126. [Google Scholar] [CrossRef]
- Normandeau, G. White Golds: A Review of Commercial Material Characteristics & Alloy Design Alternatives. Gold Bull. 1992, 25, 94–103. [Google Scholar] [CrossRef][Green Version]
- Moon, J.; Reeder, M.; Atwater, A.R. Contact Allergy to Nickel: Still #1 after All These Years. Cutis 2021, 107, 12–15. [Google Scholar] [CrossRef]
- Ahlström, M.G.; Thyssen, J.P.; Wennervaldt, M.; Menné, T.; Johansen, J.D. Nickel Allergy and Allergic Contact Dermatitis: A Clinical Review of Immunology, Epidemiology, Exposure, and Treatment. Contact Dermat. 2019, 81, 227–241. [Google Scholar] [CrossRef] [PubMed]
- Garner, L.A. Contact Dermatitis to Metals. Dermatol. Ther. 2004, 17, 321–327. [Google Scholar] [CrossRef]
- Thyssen, J.P.; Uter, W.; McFadden, J.; Menné, T.; Spiewak, R.; Vigan, M.; Gimenez-Arnau, A.; Lidén, C. The EU Nickel Directive Revisited—Future Steps towards Better Protection against Nickel Allergy. Contact Dermat. 2011, 64, 121–125. [Google Scholar] [CrossRef]
- Ahlström, M.G.; Thyssen, J.P.; Menné, T.; Johansen, J.D. Prevalence of Nickel Allergy in Europe Following the EU Nickel Directive—A Review. Contact Dermat. 2017, 77, 193–200. [Google Scholar] [CrossRef]
- Wataha, J.C.; Shor, K. Palladium Alloys for Biomedical Devices. Expert Rev. Med. Devices 2014, 7, 489–501. [Google Scholar] [CrossRef]
- Erfani, B.; Lidén, C.; Midander, K. Short and Frequent Skin Contact with Nickel. Contact Dermat. 2015, 73, 222–230. [Google Scholar] [CrossRef]
- Peretti, D.; Di Siro, M.; Di Siro, S. Nickel- and Palladium-Free Master Alloys for All Karats of White Gold. In Proceedings of the Santa Fe Symposium on Jewellery Manufacturing Technology, Albuquerque, NM, USA, 21–24 May 2017; pp. 343–362. [Google Scholar]
- Rosenberg, E. Germanium: Environmental Occurrence, Importance and Speciation. Rev. Environ. Sci. Biotechnol. 2009, 8, 29–57. [Google Scholar] [CrossRef]
- Zheng, J.; Yang, L.; Deng, Y.; Zhang, C.; Zhang, Y.; Xiong, S.; Ding, C.; Zhao, J.; Liao, C.; Gong, D. A Review of Public and Environmental Consequences of Organic Germanium. Crit. Rev. Environ. Sci. Technol. 2019, 50, 1384–1409. [Google Scholar] [CrossRef]
- CrystalMaker Software V 10.7; CrystalMaker Software Ltd.: Kidlington, UK, 2021.
- Schluckebier, G.; Predel, B. Investigation on the Structure of Metastable Phases in the Gold-Germanium System. Z. Metallkd. 1980, 71, 535–541. [Google Scholar]
- ASTM Designation: G 59–97 (Reapproved 2003); Standard Test Method for Conducting Potentiodynamic Polarization Resistance Measurements. ASTM International: West Conshohocken, PA, USA, 2003.
- Esmailzadeh, S.; Aliofkhazraei, M.; Sarlak, H. Interpretation of Cyclic Potentiodynamic Polarization Test Results for Study of Corrosion Behavior of Metals: A Review. Prot. Met. Phys. Chem. Surf. 2018, 54, 976–989. [Google Scholar] [CrossRef]
- Wang, J.; Qin, H.; Chen, J.; Yang, D.; Zhang, G. First-Principles Study on the Elastic Mechanical Properties and Anisotropies of Gold–Copper Intermetallic Compounds. Metals 2022, 12, 959. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, B.; Pan, B.; Wang, C. Germanium electrochemical study and its CMP application. App. Surf. Sci. 2017, 422, 247–256. [Google Scholar] [CrossRef]
- Lgaz, H.; Saha, S.K.; Lee, H.-S.; Kang, N.; Thari, F.Z.; Karrouchi, K.; Salghi, R.; Bougrin, K.; Ali, I.H. Corrosion Inhibition Properties of Thiazolidinedione Derivatives for Copper in 3.5 wt.% NaCl Medium. Metals 2021, 11, 1861. [Google Scholar] [CrossRef]
- Cai, Y.; Xu, Y.; Zhao, Y.; Zhang, W.; Yao, J.; Wei, M.; Zhou, K.; Ma, X. Quantitative Understanding of the Environmental Effect on B10 Copper Alloy Corrosion in Seawater. Metals 2021, 11, 1080. [Google Scholar] [CrossRef]
- Antonijevic, M.M.; Alagic, S.C.; Petrovic, M.B.; Radovanovic, M.B.; Stamenkovic, A.T. The Influence of pH on Electrochemical Behavior of Copper in Presence of Chloride Ions. Int. J. Electrochem. Sci. 2009, 4, 516–524. [Google Scholar]
- Azzaroni, O.; Cipollone, M.; Vela, M.E.; Salvarezza, R.C. Protective Properties of Dodecanethiol Layers on Copper Surfaces: The Effect of Chloride Anions in Aqueous Environments. Langmuir 2001, 17, 1483–1487. [Google Scholar] [CrossRef]
- Milazzo, G.; Caroli, S. Tables of Standard Electrode Potentials; John Wiley & Sons: Chichester, NY, USA, 1978. [Google Scholar]
- Tasić, Ž.Z.; Mihajlović, M.B.P.; Radovanović, M.B.; Simonović, A.T.; Antonijević, M.M. Cephradine as corrosion inhibitor for copper in 0.9% NaCl solution. J. Mol. Struct. 2018, 1159, 46–54. [Google Scholar] [CrossRef]
- Wang, D.; Xiang, B.; Liang, Y.; Song, S.; Liu, C. Corrosion control of copper in 3.5wt.% NaCl Solution by Domperidone: Experimental and Theoretical Study. Corr. Sci. 2014, 85, 77–86. [Google Scholar] [CrossRef]
- Grgur, B.N.; Lazić, V.; Stojić, D.; Rudolf, R. Electrochemical testing of noble metal dental alloys: The influence of their chemical composition on the corrosion resistance. Corr. Sci. 2021, 184, 109412. [Google Scholar] [CrossRef]
- Brantley, W.A.; Laub, L.W.; Drago, C.J. Framework Design and Metal Selection. In Contemporary Fixed Prosthodontics, 5th ed.; Rosenstiel, S., Land, M., Fujimoto, J., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; Chapter 19; pp. 529–541. [Google Scholar]
- Yi, Y.; Cho, P.; Al Zaabi, A.; Addad, Y.; Jang, C. Potentiodynamic polarization behaviour of AISI type 316 stainless steel in NaCl solution. Corr. Sci. 2013, 74, 92–97. [Google Scholar] [CrossRef]
- Zakeri, M.; Naghizadeh, M.; Nakhaie, D.; Moayed, M.H. Pit transition potential and repassivation potential of stainless steel in thiosulfate solution. J. Electrochem. Soc. 2016, 163, C275–C281. [Google Scholar] [CrossRef]


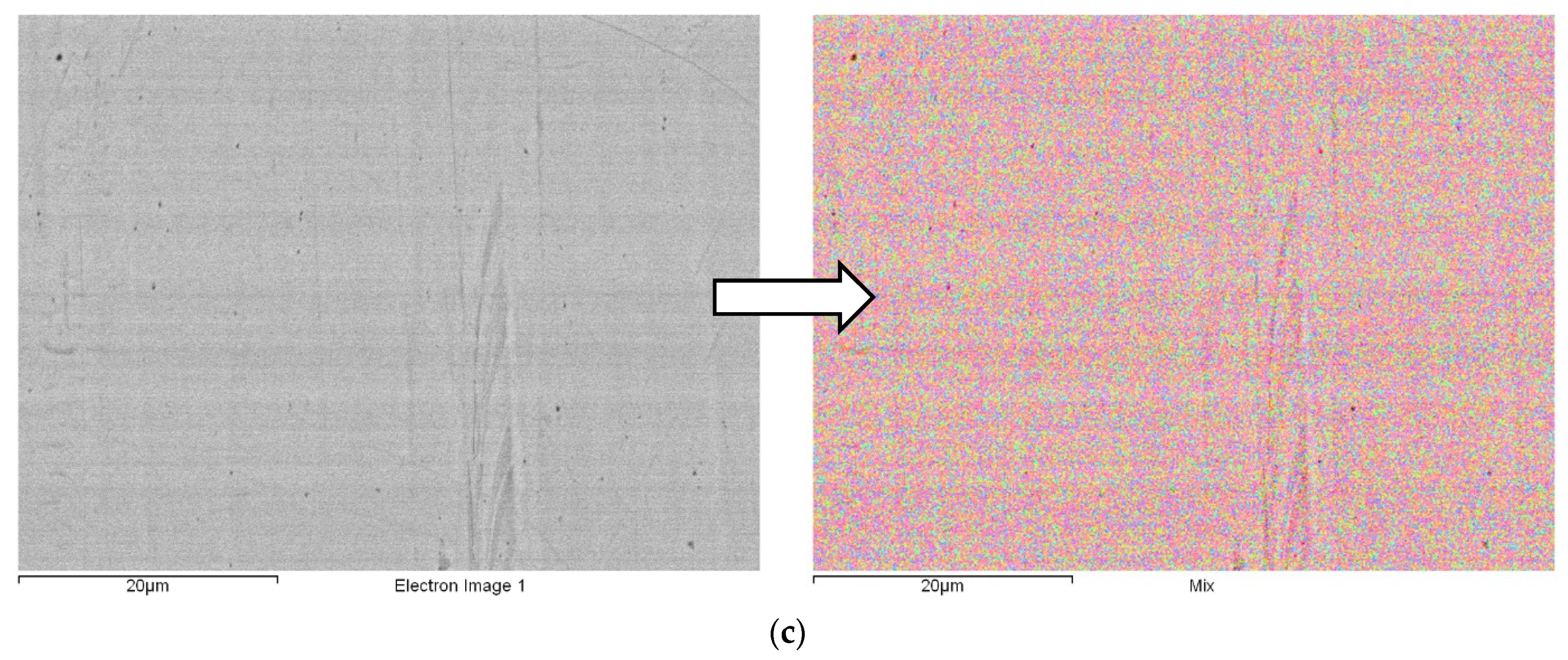

| Spectrum | Cu | Zn | Ge | Au | Total |
|---|---|---|---|---|---|
| Mean | 33.44 | 2.64 | 1.79 | 62.13 | 100.00 |
| Std. Deviation | 0.48 | 0.14 | 0.37 | 0.32 | |
| Max. | 33.85 | 2.88 | 2.35 | 62.59 | |
| Min. | 32.51 | 2.50 | 1.33 | 61.78 |
| Spectrum | Cu | Zn | Ge | Au | Total |
|---|---|---|---|---|---|
| Mean | 28.47 | 4.23 | 4.09 | 63.21 | 100.00 |
| Std. Deviation | 0.40 | 0.24 | 0.54 | 0.73 | |
| Max. | 29.06 | 4.55 | 4.75 | 64.51 | |
| Min. | 27.99 | 3.95 | 3.48 | 62.37 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rudolf, R.; Majerič, P.; Lazić, V.; Grgur, B. Development of a New AuCuZnGe Alloy and Determination of Its Corrosion Properties. Metals 2022, 12, 1284. https://doi.org/10.3390/met12081284
Rudolf R, Majerič P, Lazić V, Grgur B. Development of a New AuCuZnGe Alloy and Determination of Its Corrosion Properties. Metals. 2022; 12(8):1284. https://doi.org/10.3390/met12081284
Chicago/Turabian StyleRudolf, Rebeka, Peter Majerič, Vojkan Lazić, and Branimir Grgur. 2022. "Development of a New AuCuZnGe Alloy and Determination of Its Corrosion Properties" Metals 12, no. 8: 1284. https://doi.org/10.3390/met12081284
APA StyleRudolf, R., Majerič, P., Lazić, V., & Grgur, B. (2022). Development of a New AuCuZnGe Alloy and Determination of Its Corrosion Properties. Metals, 12(8), 1284. https://doi.org/10.3390/met12081284








