Coupling Flotation Rate Constant and Viscosity Models
Abstract
:1. Introduction
| Researcher | Reported Suspension Viscosity | Flotation Results (Rate Constant or Recovery/Grade) | References |
|---|---|---|---|
| Farrokhpay et al., 2011 | The viscosity of 50 vol.% glycerol–water mixture, used in their study, was 0.0076 Pa s. | The recovery of coarse composite copper-bearing particles (+210 μm) of porphyry copper ore, recovered in the tailings of rougher, at a grind size of d80 = 250 μm, increased from 83 to 90% with the increase in viscosity from 0.001 to 0.0076 Pa s. | [24] |
| Shabalala et al., 2011 | The viscosity of kaolin ore slurry increased exponentially to the maximum values of between 0.03 and 0.08 Pa s with the increase in solid concentration from 15 to 40 wt.% of kaolin. | Bubble size generated within kaolin ore suspension decreased from 1 to 0.65 mm with the increase in solid concentration from 30 to 40 wt.% at an impeller speed of 650 rpm. | [26] |
| Forbes et al., 2014 | The viscosity of pulp containing chalcopyrite and clay minerals (kaolinite and quartz) was found between 0.001 and 0.15 Pa s. | The recovery of chalcopyrite (copper) was 92% at quartz/kaolinite content of 100/0; however, with the change in quartz/kaolinite ratio (i.e., 70/30 and 30/70), the chalcopyrite recovery reduced to 87% and 82%, respectively. | [28] |
| Cruz et al., 2015 | The viscosity of copper–gold ore slurry increased from 0.0035 to 0.014 Pa s with the increase of solid concentration of bentonite from 0 to 15 wt.% at a shear rate of 100 s−1. | The baseline flotation of their ore (i.e., 100 wt.% ore without bentonite/kaolinite) resulted in a copper recovery of 92% at a grade of 10% copper, and 81% gold recovery at a grade of 7 ppm gold. The addition of 15 wt.% bentonite to the ore (100 wt.%) decreased the recovery (i.e., copper recovery from 92 to 83% and gold recovery from 81 to 64%) and slightly decreased in copper and gold grades from 10 to 8% and 7 ppm to 5 ppm, respectively. The addition of 30 wt.% kaolinite to the ore (100 wt.%) did not decrease copper and gold recoveries but did decrease copper and gold grades from 10 to 2% and 7 ppm to 1 ppm, respectively. | [20] |
| Wang et al., 2015 | The apparent viscosity of Telfer clean ore increased from 0.001 to 0.008 Pa s with the increase in solid concentration of bentonite from 5 to 25 wt.% at a shear rate of 100 s−1. | The copper recovery decreased from 76 to 25% with the increase in solid concentration of bentonite from 0 to 20 wt.%; however, it slightly decreased the copper grade from 5.1 to 5%. The copper recovery decreased from 80 to 67% with the increase in solid concentration of kaolin from 0 to 20 wt.%; however, it decreased the copper grade from 5 to 4%. | [16] |
| Zhang & Peng, 2015 | The apparent viscosity of a copper–gold ore increased from 0.0018 to 0.0076 Pa s with the increase in solid concentration of bentonite from 0 to 15 wt.% at a shear rate of 100 s−1. | The copper recovery decreased from 82 to 60% with the increase in solid concentration of bentonite from 0 to 15 wt.%. The gold recovery decreased from 78 to 65% with the increase in solid concentration of bentonite from 0 to 15%. | [14] |
| Farrokhpay et al., 2016 | The apparent viscosity of their copper ore slurry in the presence of 15 wt.% montmorillonite, 30 wt.% of kaolinite, and 30 wt.% illite at a shear rate of 100 s−1 was 0.17, 0.03, and 0.02 Pa s, respectively. | The copper recovery decreased (90 to 80%) in the presence of 15 wt.% swelling clay (montmorillonite); however, in the presence of 15 wt.% non-swelling clays (illite and kaolinite), the copper recovery decreased slightly to 87% and 88%, respectively. The copper grade decreased from 18% to about 1% in the presence of both 30 wt.% of kaolinite and 15 wt.% of montmorillonite, respectively; however, it decreased to about 5% in the presence of 30% illite. The copper ore flotation rate constants were 0.51 s−1 and 0.49 s−1 in the presence of 15 wt.% of kaolinite and 15 wt.% of illite, respectively, and 0.33 s−1 in the presence 15 wt.% of montmorillonite, as compared with the copper ore flotation rate constant of 0.70 s−1 in the absence of clay minerals. | [25] |
| Basnayaka et al., 2017 | The viscosity of gold ore increased from 0.0018 to 0.0035 Pa s by the addition of 10 wt.% kaolin at pH 7, a shear rate of 100 s−1, and polyacrylate depressant concentration of 0 and 200 g/t, respectively; however, by the addition of 5 wt.% bentonite, the viscosity increased to 0.0060 Pa s, under the same conditions. | The flotation rate constant of their gold-bearing pyrite ore was decreased from 13.71 to 3.37 s−1 (822.6 to 202.2 min−1) without and with the presence of 10 wt.% kaolin at pH 7, air rate of 5 L/min, and polyacrylate depressant concentration of 0 and 200 g/t, respectively. The presence of bentonite under same conditions reduced the flotation rate constant to 4.14 s−1 (248.4 min−1). | [33] |
| Chen et al., 2017 | The apparent viscosity of amorphous silica and quartz suspension increased from 0.109 to 0.147 Pa s with the increase in the concentration of amorphous silica (in amorphous silica and quartz suspension) from 30 to 50 vol.%, at a shear rate of 100 s−1. | The copper recovery dropped sharply from 95 to 63.6% after the percentage of amorphous silica increased from 30 to 50 vol.%. The copper grade decreased slightly from 3.9 to 3.8% with the increase in the concentration of amorphous silica (in amorphous silica and quartz suspension) from 30 to 50 vol.%. | [18] |
| Farrokhpay et al., 2018 | The viscosity of their copper ore slurry increased from 0.010 to 0.038 Pa s with the increase in solid concentration of muscovite from 0 to 30 wt.% at a shear rate of 100 s−1; however, with the increase in solid concentration of talc from 0 to 7.5 wt.%, there was a slight increase in the slurry viscosity from 0.010 to 0.012 Pa s, at a shear rate of 100 s−1. | The flotation grade decreased (19 to 2%) with the increase in solid concentration of muscovite from 0 to 30 wt.%; however, the change in the recovery was reported negligible. The flotation recovery (90 to 83%) and grade (19 to 2%) decreased with the increase in solid concentration of talc from 0 to 7.5 wt.%. | [17] |
2. Theory
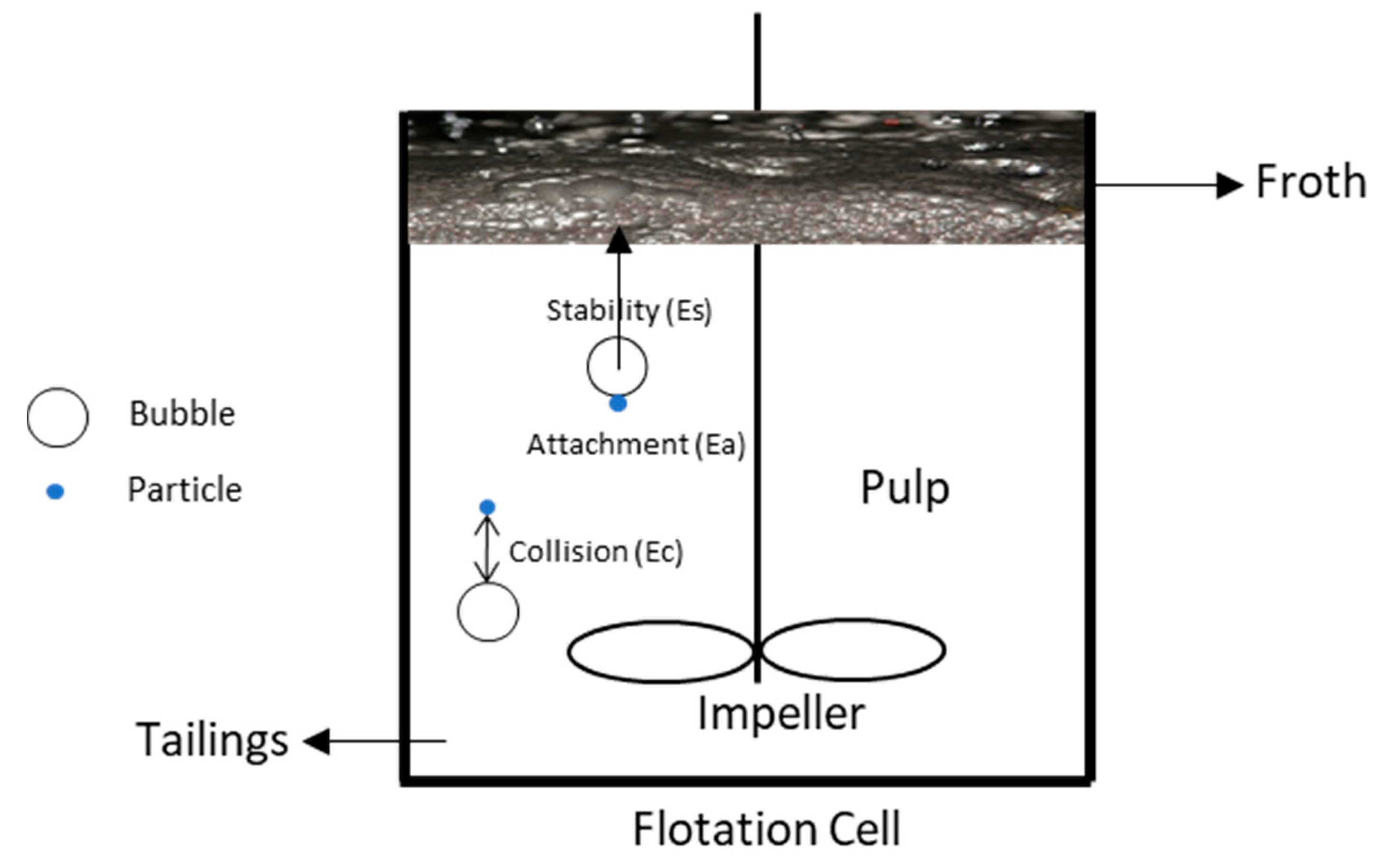
2.1. Collection Efficiency ()
2.1.1. Collision Efficiency ()
2.1.2. Attachment Efficiency
2.1.3. Stability Efficiency
2.2. Flotation Rate Constant under Turbulent Flow Condition
2.3. Viscosity Modeling and Factors Affecting Viscosity
2.3.1. Hard Sphere Suspensions
2.3.2. Effect of Shear Rate
2.3.3. Colloidal Suspensions
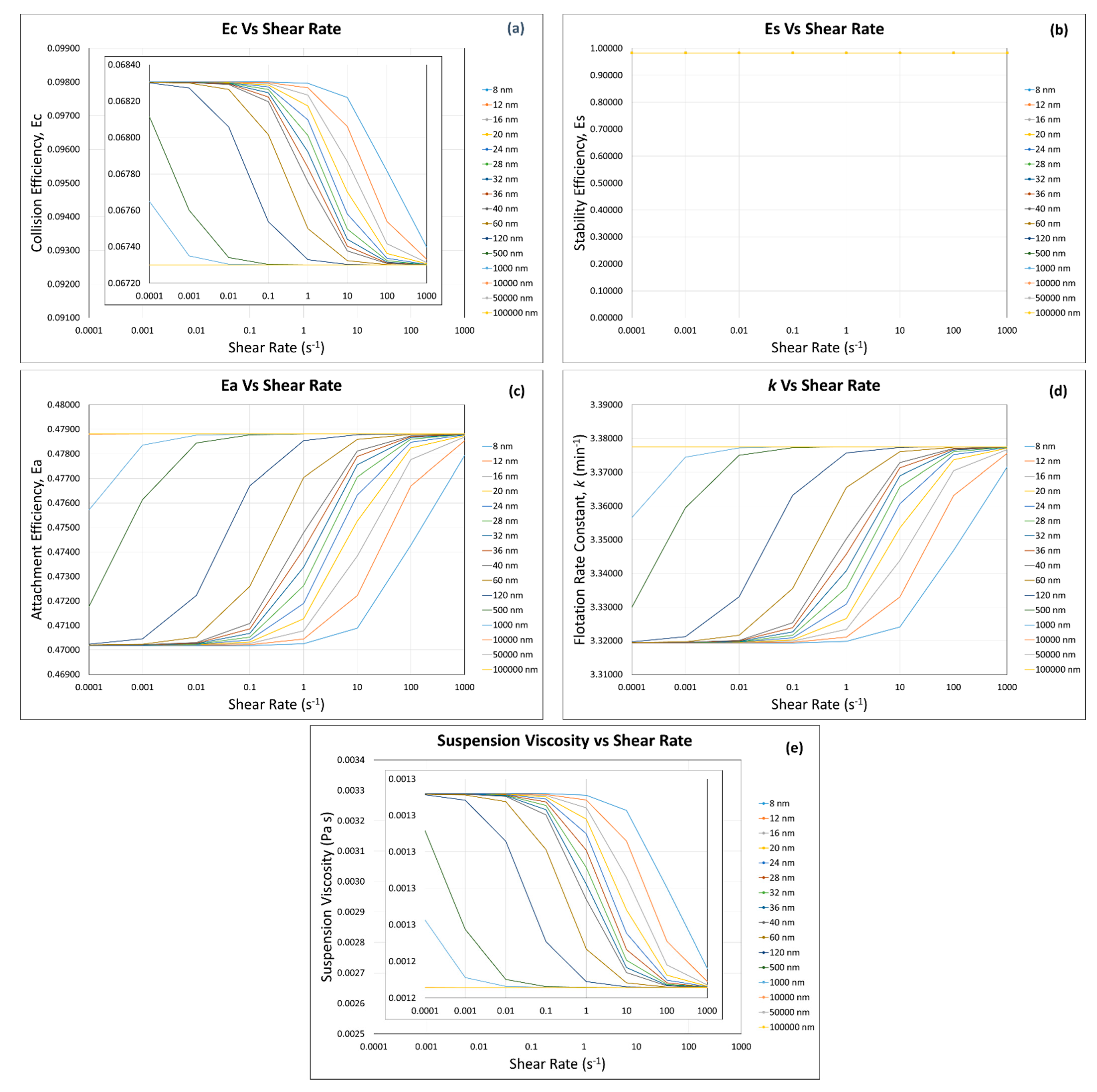
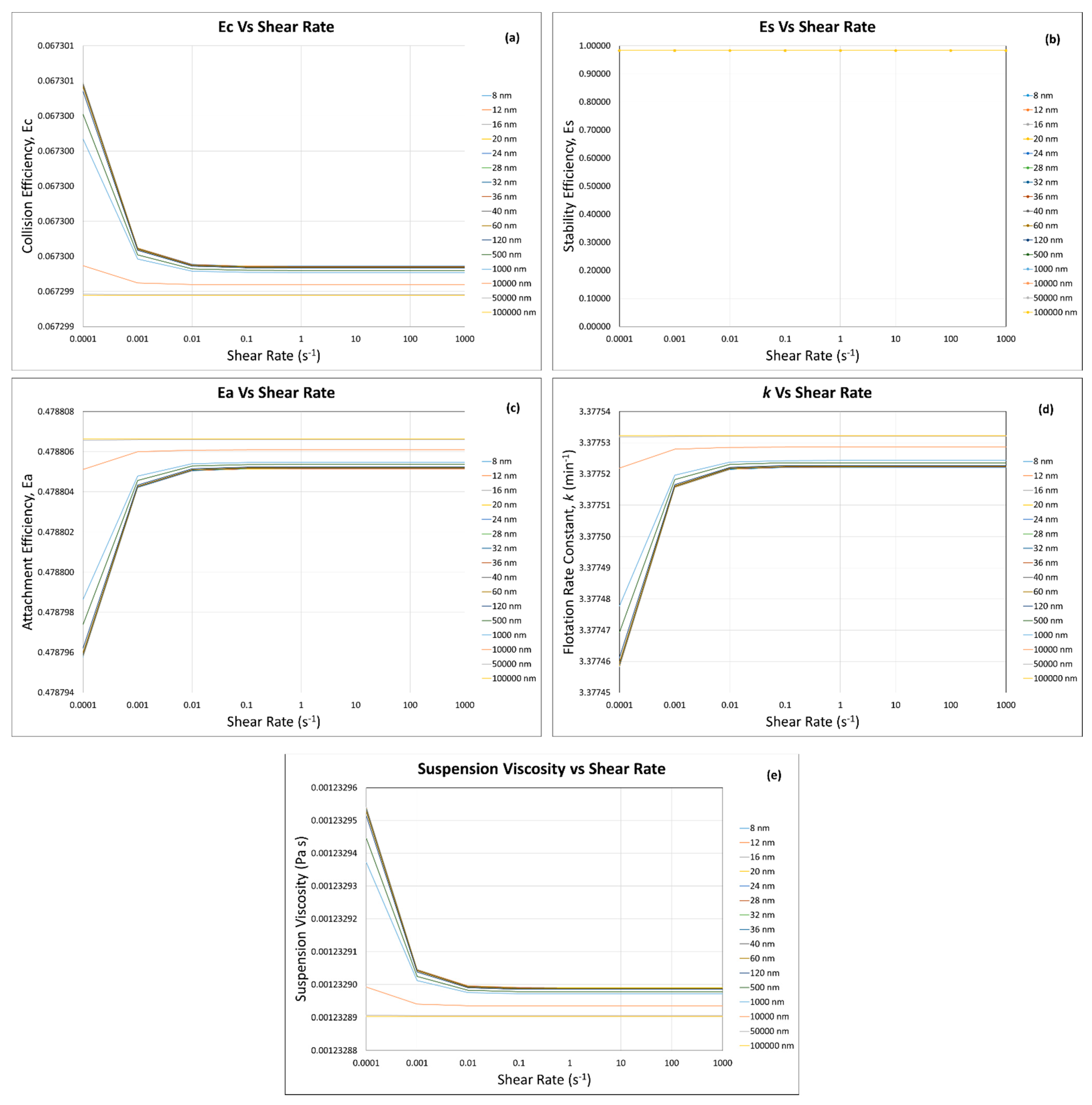
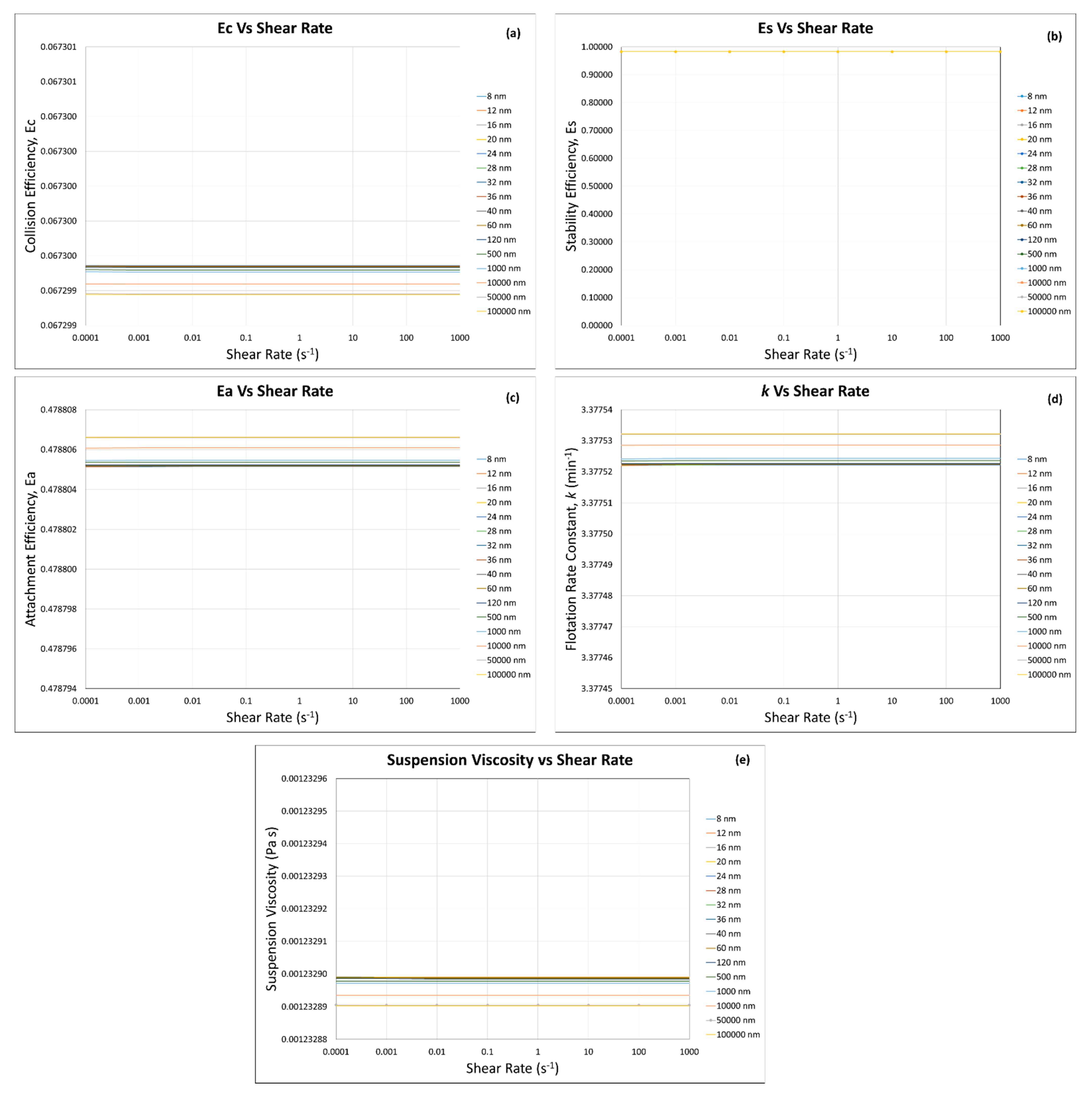
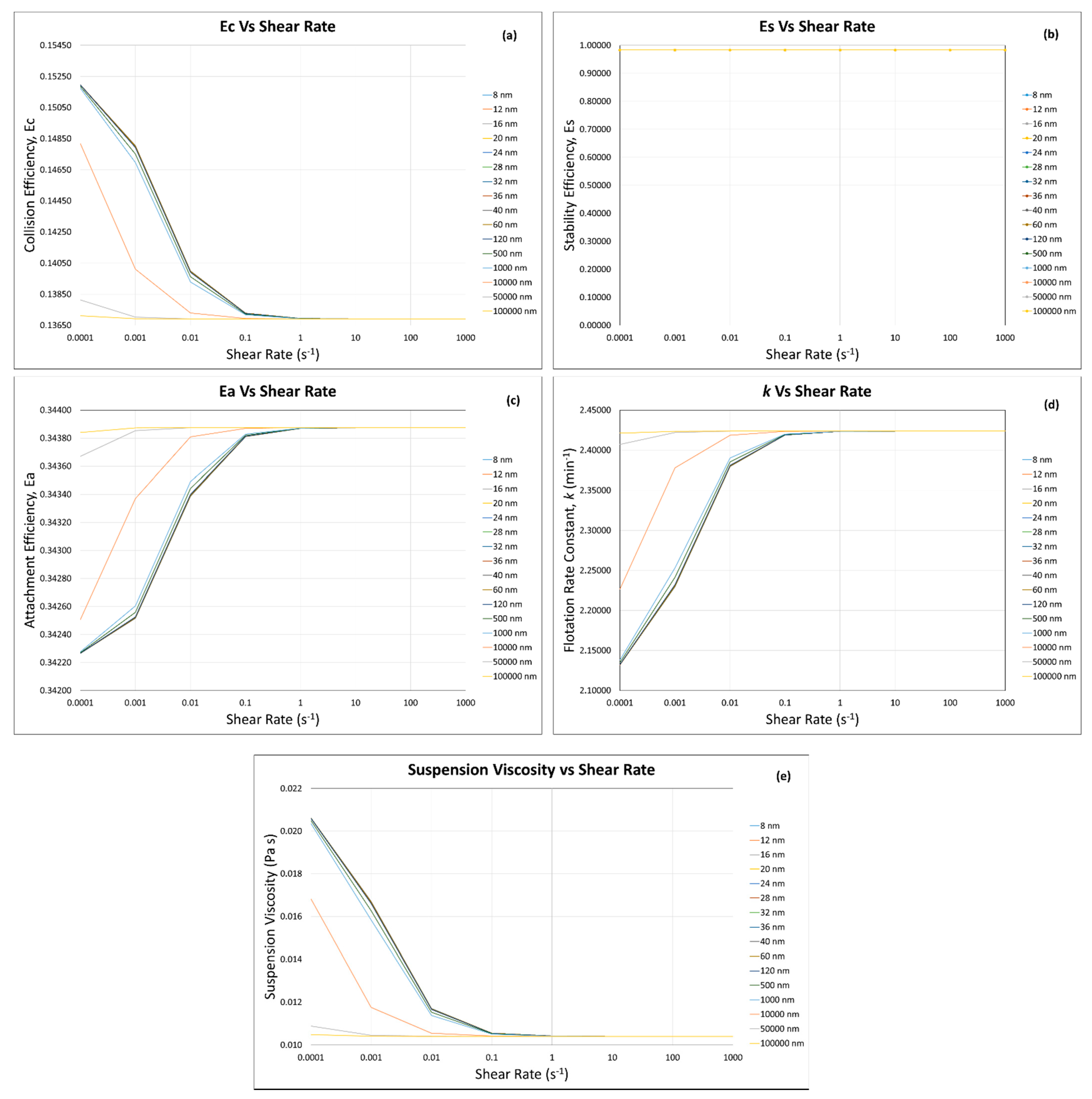
3. Results and Discussion
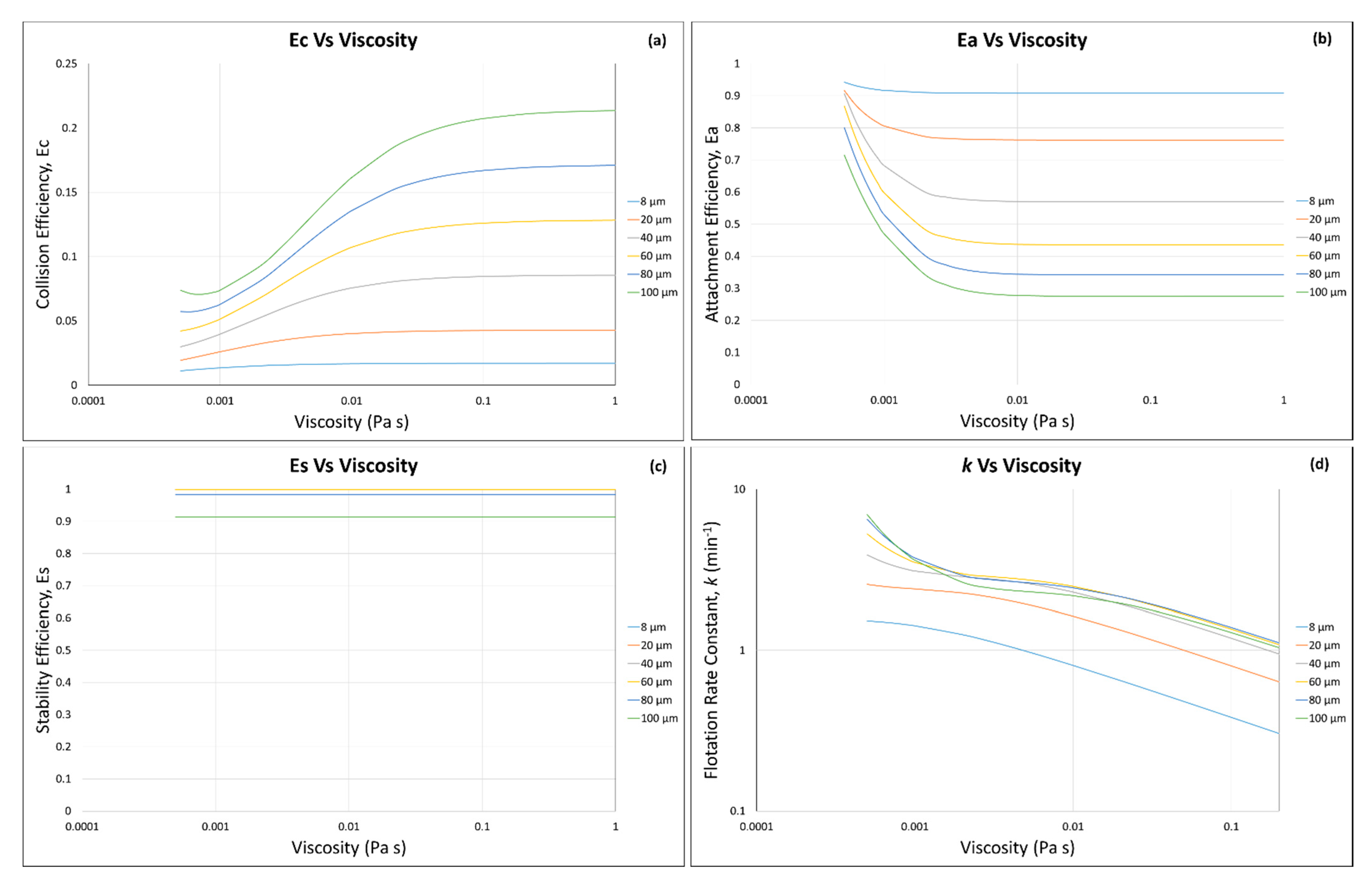
3.1. Calculated Flotation Collection Efficiencies , , , and Rate Constant
3.2. Effect of Suspension Viscosity on , , , and Rate Constant
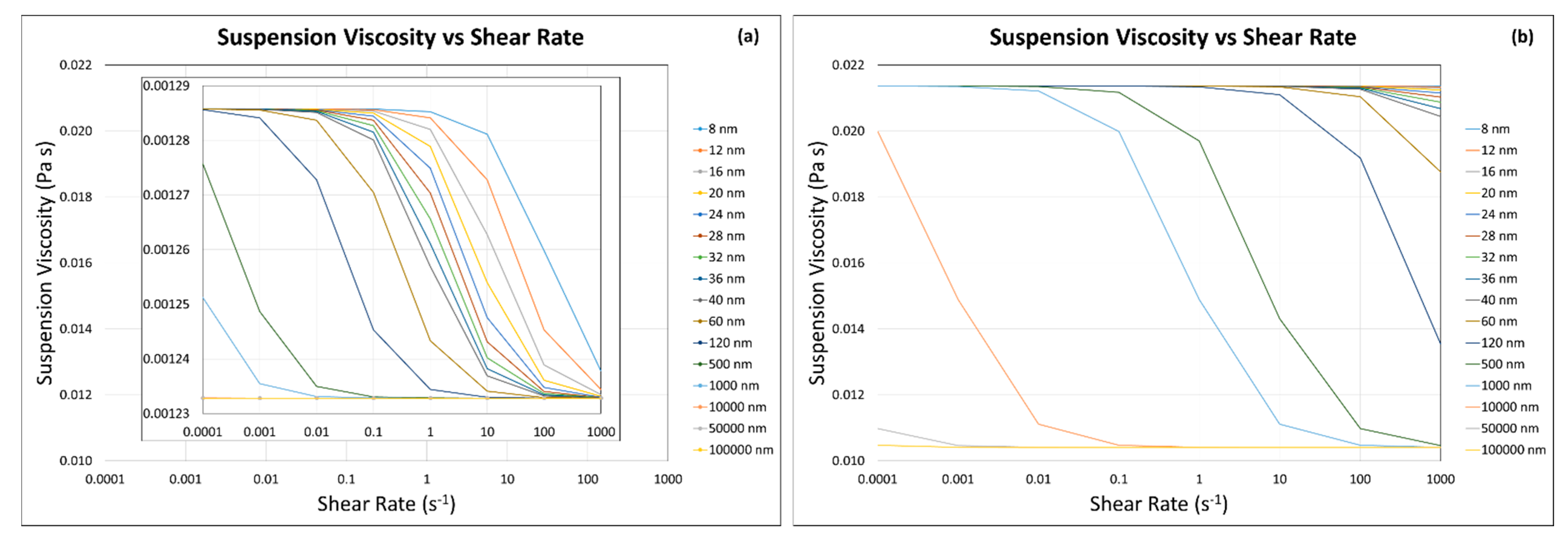
3.3. Suspension Viscosity Calculation
3.3.1. Modified Krieger and Dougherty Model—Hard Sphere Suspensions
3.3.2. Our Predictive Model—Hard Sphere and Interacting Colloidal Particle Suspensions
Homogeneous Case (i.e., Same Particle with Same Sizes)
Homogeneous Case (i.e., Same Particles with Different Particle Sizes)
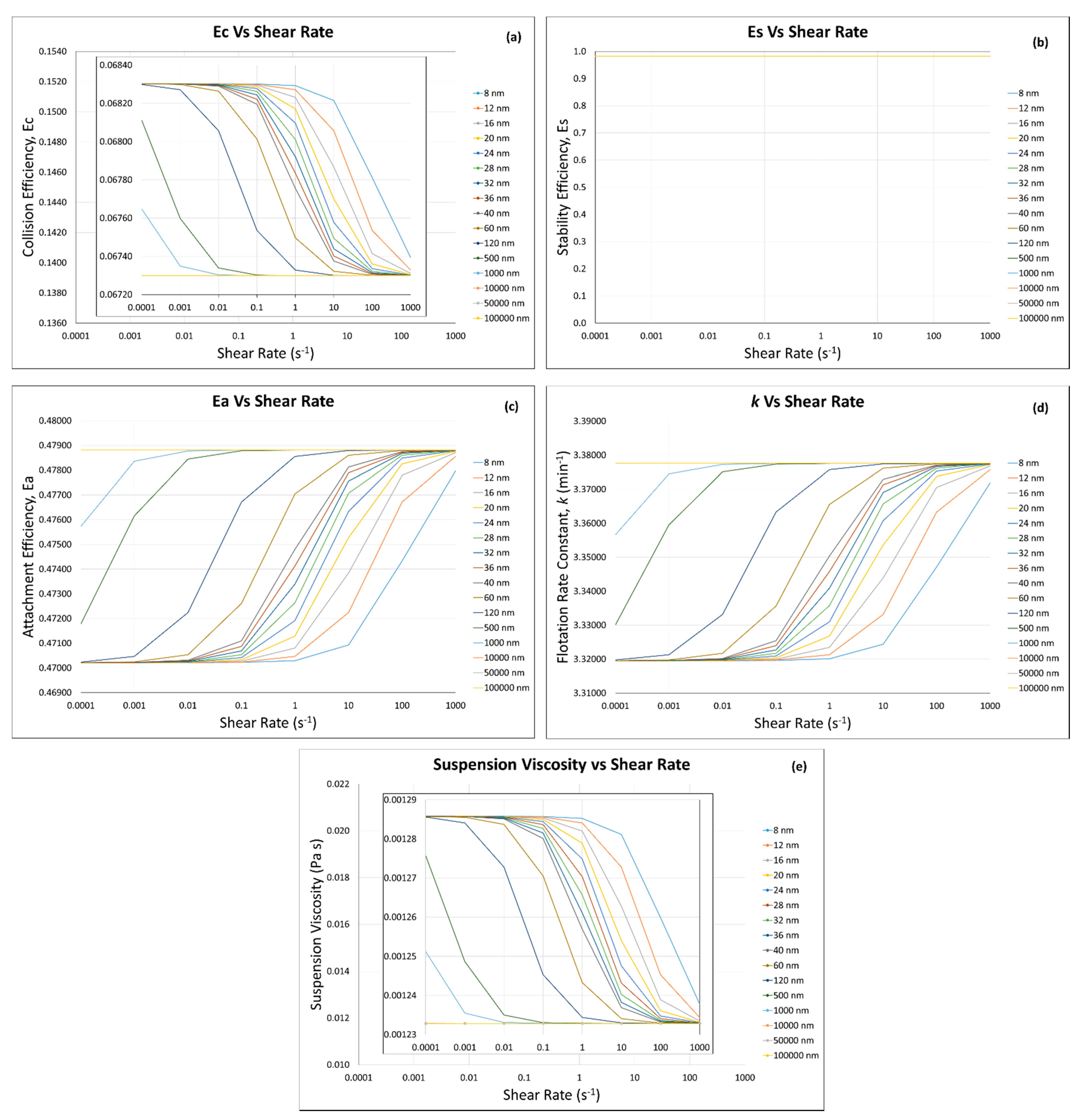
3.4. Flotation Efficiencies and Rate Constant Calculations
3.4.1. Incorporation of the Modified Krieger and Dougherty Model—Hard Sphere Suspensions
3.4.2. Incorporation of Our Predictive Model—Hard Sphere and Interacting Colloidal Particle Suspensions
Homogeneous Case—Same Particles with the Same Sizes
Homogeneous Case—Same Particle with Different Particle Sizes
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| Hamaker constant | |
| Radius of hard spheres | |
| Bond number | |
| Concentration of ions | |
| Bubble diameter | |
| Particle diameter | |
| Elementary charge | |
| Collection efficiency | |
| Collision efficiency | |
| Attachment efficiency | |
| Stability efficiency | |
| Attractive forces | |
| Buoyancy force | |
| Capillary force | |
| Machine acceleration force | |
| Detachment forces | |
| Gravitational force | |
| Hydrostatic force | |
| Capillary pressure force | |
| Gas flow rate | |
| Interparticle separation distance | |
| Flotation rate constant | |
| Boltzman’s constant | |
| Avogadro’s number | |
| Number concentration of ions | |
| Peclet number | |
| Characteristic Peclet number | |
| Bubble radius | |
| Reynolds number | |
| Particle radius | |
| Absolute temperature | |
| Induction time | |
| Volume of flotation cell | |
| Kinematic viscosity | |
| Bubble velocity | |
| Particle velocity | |
| Ionic valence | |
| Collision angle | |
| Maximum collision angle | |
| Contact angle | |
| Particle density | |
| Fluid density | |
| Dynamic viscosity | |
| Suspension viscosity | |
| Liquid’s viscosity | |
| Relative viscosity | |
| Intrinsic viscosity of the particles | |
| Solid volume fraction | |
| Maximum packing fraction | |
| Shear rate | |
| Reduced surface potential | |
| Debye–Huckel reciprocal length | |
| Dielectric constant of the medium | |
| Permittivity of free space | |
| Zeta potential |
Appendix A

References
- Venditti, R.A. A Simple Flotation De-Inking Experiment for the Recycling of Paper. J. Chem. Educ. 2004, 81, 693. [Google Scholar] [CrossRef]
- Wang, C.; Wang, H.; Fu, J.; Liu, Y. Flotation separation of waste plastics for recycling-A review. Waste Manag. 2015, 41, 28–38. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Zhao, C. A study of the kinetics on induced-air flotation for oil-water separation. Sep. Sci. Technol. 1993, 28, 1233–1240. [Google Scholar] [CrossRef]
- Rubio, J.; Souza, M.; Smith, R.W. Overview of flotation as a wastewater treatment technique. Miner. Eng. 2002, 15, 139–155. [Google Scholar] [CrossRef]
- Pyke, B.; Fornasiero, D.; Ralston, J. Bubble particle heterocoagulation under turbulent conditions. J. Colloid Interface Sci. 2003, 265, 141–151. [Google Scholar] [CrossRef]
- Sajjad, M.; Otsuki, A. Correlation between Flotation and Rheology of Fine Particle Suspensions. Metals 2022, 12, 270. [Google Scholar] [CrossRef]
- Dai, Z.; Dukhin, S.; Fornasiero, D.; Ralston, J. The Inertial Hydrodynamic Interaction of Particles and Rising Bubbles with Mobile Surfaces. J. Colloid Interface Sci. 1998, 197, 275–292. [Google Scholar] [CrossRef]
- Szatkowski, M. A model of flotation selectivity. Int. J. Miner. Process. 1988, 23, 25–31. [Google Scholar] [CrossRef]
- Schulze, H.J. Physico–Chemical Elementary Processes in Flotation: An Analysis from the Point of View of Colloid Science Including Process Engineering Considerations; Elsevier: Amsterdam, The Netherlands, 1984. [Google Scholar]
- Dukhin, S.S.; Kretzschmar, G.; Miller, R. Dynamics of Adsorption at Liquid Interfaces, 1st ed.; Elsevier Science: Amsterdam, The Netherlands, 1995. [Google Scholar]
- Duan, J.; Fornasiero, D.; Ralston, J. Calculation of the flotation rate constant of chalcopyrite particles in an ore. Int. J. Miner. Process. 2003, 72, 227–237. [Google Scholar] [CrossRef]
- Jameson, G.J.; Nam, S.; Young, M.M. Physical Factors Affecting Recovery Rates in Flotation. Miner. Sci. Eng. 1997, 9, 103–118. [Google Scholar]
- Radoev, B.P.; Alexandrova, L.; Tchaljovska, S. On the kinetics of froth flotation. Int. J. Miner. Process. 1990, 28, 127–138. [Google Scholar] [CrossRef]
- Zhang, M.; Peng, Y. Effect of clay minerals on pulp rheology and the flotation of copper and gold minerals. Miner. Eng. 2015, 70, 8–13. [Google Scholar] [CrossRef]
- Xu, D.; Ametov, I.; Grano, S.R. Quantifying rheological and fine particle attachment contributions to coarse particle recovery in flotation. Miner. Eng. 2012, 39, 89–98. [Google Scholar] [CrossRef]
- Wang, Y.; Peng, Y.; Nicholson, T.M.; Lauten, R.A. The different effects of bentonite and kaolin on copper flotation. Appl. Clay Sci. 2015, 114, 48–52. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Ndlovu, B.N.; Bradshaw, D.J. Behavior of talc and mica in copper ore flotation. Appl. Clay Sci. 2018, 160, 270–275. [Google Scholar] [CrossRef]
- Chen, X.; Hadde, E.; Liu, S.; Peng, Y. The effect of amorphous silica on pulp rheology and copper flotation. Miner. Eng. 2017, 113, 41–46. [Google Scholar] [CrossRef]
- Zhang, M.; Peng, Y.; Xu, N. The effect of sea water on copper and gold flotation in the presence of bentonite. Miner. Eng. 2015, 77, 93–98. [Google Scholar] [CrossRef]
- Cruz, N.; Peng, Y.; Wightman, E.M.; Xu, N. The interaction of clay minerals with gypsum and its effects on copper–gold flotation. Miner. Eng. 2015, 77, 121–130. [Google Scholar] [CrossRef]
- Genovese, D.B. Shear rheology of hard-sphere, dispersed, and aggregated suspensions, and filler-matrix composites. Adv. Colloid Interface Sci. 2012, 171, 1–16. [Google Scholar] [CrossRef]
- Ogawa, A.; Yamada, H.; Matsuda, S.; Okajima, K. Viscosity equation for concentrated suspensions of charged colloidal particles. J. Rheol. 1997, 41, 769–785. [Google Scholar] [CrossRef]
- Zhang, W. Evaluation of effect of viscosity changes on bubble size in a mechanical flotation cell. Trans. Nonferrous Met. Soc. China 2014, 24, 2964–2968. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Ametov, I.; Grano, S.R. Improving the recovery of low grade coarse composite particles in porphyry copper ores. Adv. Powder Technol. 2011, 22, 464–470. [Google Scholar] [CrossRef]
- Farrokhpay, S.; Bulelwa, N.; Dee, B. Behaviour of swelling clays versus non-swelling clays in flotation. Miner. Eng. 2016, 96, 59–66. [Google Scholar] [CrossRef]
- Shabalala, N.Z.; Harris, M.C.; Leal-Filho, L.D.S.; Deglon, D.A. Effect of slurry rheology on gas dispersion in a pilot-scale mechanical flotation cell. Miner. Eng. 2011, 24, 1448–1453. [Google Scholar] [CrossRef]
- Becker, M.; Yorath, G.; Ndlovu, B.; Harris, M.C.; Deglon, D.A.; Franzidis, J.P. A rheological investigation of the behaviour of two Southern African platinum ores. Miner. Eng. 2013, 49, 92–97. [Google Scholar] [CrossRef]
- Forbes, E.; Davey, K.J.; Smith, L. Decoupling rheology and slime coatings effect on the natural floatability of chalcopyrite in a clay-rich flotation pulp. Miner. Eng. 2014, 56, 136–144. [Google Scholar] [CrossRef]
- Bakker, C.W.; Meyer, C.J.; Deglon, D.A. Numerical modelling of non-Newtonian slurry in a mechanical flotation cell. Miner. Eng. 2009, 22, 944–950. [Google Scholar] [CrossRef]
- Otsuki, A.; Chen, Y.; Zhao, Y. Characterisation and Beneficiation of Complex Ores for Sustainable Use of Mineral Resources: Refractory Gold Ore Beneficiation as an Example. Int. J. Soc. Mater. Eng. Resour. 2014, 20, 126–135. [Google Scholar] [CrossRef] [Green Version]
- Derjaguin, B.V. Theory of Stability of Colloids and Thin Films; English Translation; Plenum: New York, NY, USA, 1989. (In Russian) [Google Scholar]
- Verwey, E.J.W. Theory of the Stability of Lyophobic Colloids; Elsevier Publishing Company Inc.: Amsterdam, The Netherlands, 1948. [Google Scholar]
- Basnayaka, L.; Subasinghe, N.; Albijanic, B. Influence of clays on the slurry rheology and flotation of a pyritic gold ore. Appl. Clay Sci. 2017, 136, 230–238. [Google Scholar] [CrossRef]
- Levich, V. Physicochemical Hydrodynamics; Prentice-Hall: Englewood Cliffs, NJ, USA, 1962. [Google Scholar]
- van de Ven, T. Colloidal Hydrodynamics; Academic Press: London, UK, 1989. [Google Scholar]
- Churaev, N.V.; Derjaguin, B.V.; Muller, V. Surface Forces; Springer: New York, NY, USA, 1987. [Google Scholar]
- Edwards, D.A.; Brenner, H.; Wasan, D.T. Interfacial Transport Processes and Rheology; Butterworth-Heinemann: London, UK, 1991. [Google Scholar]
- Ivanov, I.; Dimitrov, D. Thin Liquid Films; Dekker: New York, NY, USA, 1988. [Google Scholar]
- Derjaguin, B.V.; Churaev, N.V. Wetting Films; Nauka: Moscow, Russia, 1984. [Google Scholar]
- Overbeek, J.T.G. Recent developments in the understanding of colloid stability. J. Colloid Interface Sci. 1977, 58, 408–422. [Google Scholar] [CrossRef]
- Kihira, H.; Matijevic, E. An assessment of heterocoagulation theories. Adv. Colloid Interface Sci. 1992, 42, 1–31. [Google Scholar] [CrossRef]
- Kouachi, S.; Hassas, B.V.; Hassanzadeh, A.; Çelik, M.S.; Bouhengue, M. Effect of negative inertial forces on bubble-particle collision via implementation of Schulze collision efficiency in general flotation rate constant equation. Colloids Surf. A Physicochem. Eng. Asp. 2017, 517, 72–83. [Google Scholar] [CrossRef]
- Hewitt, D.J.; Fornasiero, D.; Ralston, J. Bubble–particle attachment. J. Chem. Soc. Faraday Trans. 1995, 91, 1997–2001. [Google Scholar] [CrossRef]
- Dai, Z.; Fornasiero, D.; Ralston, J. Particle–Bubble Attachment in Mineral Flotation. J. Colloid Interface Sci. 1999, 217, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Dobby, G.S.; Finch, J.A. Particle size dependence in flotation derived from a fundamental model of the capture process. Int. J. Miner. Process. 1987, 21, 241–260. [Google Scholar] [CrossRef]
- Crawford, R.; Ralston, J. The influence of particle size and contact angle in mineral flotation. Int. J. Miner. Process. 1988, 23, 1–24. [Google Scholar] [CrossRef]
- van Nguyen, A.; Ralston, J.; Schulze, H.J. On modelling of bubble-particle attachment probability in flotation. Int. J. Miner. Process. 1998, 53, 225–249. [Google Scholar] [CrossRef]
- Ahmed, N.; Jameson, G.J. The Effect of Bubble Size on the Rate of Flotation of Fine Particles. Int. J. Miner. Process. 1985, 14, 195–215. [Google Scholar] [CrossRef]
- Dai, Z.; Fornasiero, D.; Ralston, J. Particle–bubble collision models—A review. Adv. Colloid Interface Sci. 2000, 85, 231–256. [Google Scholar] [CrossRef]
- Phan, C.M.; van Nguyen, A.; Miller, J.D.; Evans, G.M.; Jameson, G.J. Investigations of bubble–particle interactions. Int. J. Miner. Process. 2003, 72, 239–254. [Google Scholar] [CrossRef]
- Sutherland, K. Physical Chemistry of Flotation. XI. Kinetics of the flotation process. J. Phys. Colloid Chem. 1948, 52, 394–425. [Google Scholar] [CrossRef] [PubMed]
- Dukhin, S.S. Role of inertial forces in flotation of small particles. Colloid J. USSR 1982, 44, 388–397. [Google Scholar]
- Ralston, J.; Fornasiero, D.; Hayes, R. Bubble–particle attachment and detachment in flotation. Int. J. Miner. Process. 1999, 56, 133–164. [Google Scholar] [CrossRef]
- Ralston, J.; Dukhin, S.S.; Mishchuk, N.A. Inertial hydrodynamic particle–bubble interaction in flotation. Int. J. Miner. Process. 1999, 56, 207–256. [Google Scholar] [CrossRef]
- van Nguyen, A.; Schulze, H.J.; Ralston, J. Elementary steps in particle—bubble attachment. Int. J. Miner. Process. 1997, 51, 183–195. [Google Scholar] [CrossRef]
- Hassas, B.V.; Kouachi, S.; Eskanlou, A.; Bouhenguel, M.; Çelik, M.S.; Miller, J.D. The significance of positive and negative inertial forces in Particle-Bubble interaction and their role in the general flotation kinetics model. Miner. Eng. 2021, 170, 107006. [Google Scholar] [CrossRef]
- Schulze, H.J. Hydrodynamics of Bubble-Mineral Particle Collisions. Miner. Process. Extr. Metall. Rev. 1989, 5, 43–76. [Google Scholar] [CrossRef]
- Koh, P.T.; Schwarz, M.P. CFD modelling of bubble–particle attachments in flotation cells. Miner. Eng. 2006, 19, 619–626. [Google Scholar] [CrossRef]
- Karimi, M.; Akdogan, G.A.; Bradshaw, S.M. A computational fluid dynamics model for the flotation rate constant, Part I: Model development. Miner. Eng. 2014, 69, 214–222. [Google Scholar] [CrossRef]
- Brader, J.M. Nonlinear rheology of colloidal dispersions. J. Phys. Condens. Matter 2010, 22, 363101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, Z.; Scales, P.J.; Boger, D.V. Chemical and physical control of the rheology of concentrated metal oxide suspensions. Chem. Eng. Sci. 2001, 56, 2901–2920. [Google Scholar] [CrossRef]
- Qin, K.; Zaman, A.A. Viscosity of concentrated colloidal suspensions: Comparison of bidisperse models. J. Colloid Interface Sci. 2003, 266, 461–467. [Google Scholar] [CrossRef]
- Russel, W.B.; Sperry, P.R. Effect of microstructure on the viscosity of hard sphere dispersions and modulus of composites. Prog. Org. Coat. 1994, 23, 305–324. [Google Scholar] [CrossRef]
- Quemada, D.C.; Berli, C.L.A. Energy of interaction in colloids and its implications in rheological modeling. Adv. Colloid Interface Sci. 2002, 98, 51–85. [Google Scholar] [CrossRef]
- Metzner, A. Rheology of Suspensions in Polymeric Liquids. J. Rheol. 1985, 29, 739–775. [Google Scholar] [CrossRef]
- Russel, W.B.; Saville, D.A.; Schowalter, W.R. Rheology. In Colloidal Dispersions; Cambridge University Press: Cambridge, UK, 1889; pp. 456–506. [Google Scholar]
- Krieger, I.M.; Dougherty, T.J. A Mechanism for Non-Newtonian Flow in Suspensions of Rigid Spheres. J. Rheol. 1959, 3, 137–152. [Google Scholar] [CrossRef]
- Potanin, A.A. On the Self-Consistent Calculations of the Viscosity of Colloidal Dispersions. J. Colloid Interface Sci. 1993, 156, 143–152. [Google Scholar] [CrossRef]
- Chen, H.; Ding, Y.; Tan, C. Rheological behaviour of nanofluids. New J. Phys. 2007, 9, 367. [Google Scholar] [CrossRef]
- Johnson, S.B.; Franks, G.V.; Scales, P.J.; Boger, D.V.; Healy, T.W. Surface chemistry–rheology relationships in concentrated mineral suspensions. Int. J. Miner. Process. 2000, 58, 267–304. [Google Scholar] [CrossRef]
- Derjaguin, B.V.; Landau, L.D. Theory of the Stability of Strongly Charged Lyophobic Sols and of the Adhesion of Strongly Charged Particles in Solutions of Electrolytes. Acta Physicochim. 1941, 14, 633–662. [Google Scholar] [CrossRef]
- Diao, M.; Taran, E.; Mahler, S.M.; van Nguyen, A. A concise review of nanoscopic aspects of bioleaching bacteria–mineral interactions. Adv. Colloid Interface Sci. 2014, 212, 45–63. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Otsuki, A.; Lonio, H.N. Zeta potential of inorganic fine particle—Na-bentonite binder mixture systems. Electrophoresis 2020, 41, 1405–1412. [Google Scholar] [CrossRef] [PubMed]
- Elphick, K.; Yamaguchi, A.; Otsuki, A.; Hayagan, N.; Hirohata, A. Non-Destructive Imaging on Synthesised Nanoparticles. Materials 2021, 14, 613. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Han, Z.; He, C.L.; Feng, Q.; Wang, K.; Wang, Y.; Luo, N.; Dodbiba, G.; Wei, Y.; Otsuki, A.; et al. Long-Term Stability of Different Kinds of Gas Nanobubbles in Deionized and Salt Water. Materials 2021, 14, 1808. [Google Scholar] [CrossRef]
- Otsuki, A.; Dodbiba, G.; Fujita, T. Two-Liquid Flotation: Heterocoagulation of Fine Particles in Polar Organic Solvent. Mater. Trans. 2007, 48, 1095–1104. [Google Scholar] [CrossRef] [Green Version]
- Kurosawa, S.; Nagasaki, S.; Tanaka, S. Evaluation of particle dispersion from montmorillonite gel in groundwater based on DLVO theory. Trans. At. Energy Soc. Jpn. 2007, 6, 205–213. [Google Scholar] [CrossRef]
- Water—Density Viscosity Specific Weight. Engineering Edge. Available online: https://www.engineersedge.com/physics/water__density_viscosity_specific_weight_13146.htm (accessed on 27 August 2021).
- Yoon, R.-H.; Yordan, J.L. Induction time measurements for the quartz-amine flotation system. J. Colloid Interface Sci. 1991, 141, 374–383. [Google Scholar] [CrossRef]
- Otsuki, A.; Barry, S.; Fornasiero, D. Rheological studies of nickel oxide and quartz/hematite mixture systems. Adv. Powder Technol. 2011, 22, 471–475. [Google Scholar] [CrossRef]
- Schubert, H. On the optimization of hydrodynamics in fine particle flotation. Miner. Eng. 2008, 21, 930–936. [Google Scholar] [CrossRef]
- Darabi, H.; Koleini, S.M.J.; Deglon, D.A.; Rezai, B.; Abdollahy, M. Investigation of bubble-particle attachment, detachment and collection efficiencies in a mechanical flotation cell. Powder Technol. 2020, 375, 109–123. [Google Scholar] [CrossRef]
- Ndlovu, B.N.; Becker, M.; Forbes, E.; Deglon, D.A.; Franzidis, J.-P. The influence of phyllosilicate mineralogy on the rheology of mineral slurries. Miner. Eng. 2011, 24, 1314–1322. [Google Scholar] [CrossRef]
- Sakairi, N.; Kobayashi, M.; Adachi, Y. Effects of salt concentration on the yield stress of sodium montmorillonite suspension. J. Colloid Interface Sci. 2005, 283, 245–250. [Google Scholar] [CrossRef]






| ϕ/a | 8 (nm) | 12 (nm) | 16 (nm) | 20 (nm) | 24 (nm) | 28 (nm) | 32 (nm) | 36 (nm) | 40 (nm) | 60 (nm) | 120 (nm) | 500 (nm) | 1000 (nm) | 10,000 (nm) | 50,000 (nm) | 100,000 (nm) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.1 | 3.36 | 1.91 | 1.31 | 1.07 | 9.70 × 10−1 | 9.26 × 10−1 | 9.06 × 10−1 | 8.98 × 10−1 | 8.94 × 10−1 | 8.92 × 10−1 | 8.92 × 10−1 | 8.92 × 10−1 | 8.92 × 10−1 | 8.92 × 10−1 | 8.92 × 10−1 | 8.92 × 10−1 |
| 0.176 | 1.51 × 101 | 9.63 | 5.24 | 3.04 | 1.99 | 1.47 | 1.20 | 1.05 | 9.57 × 10−1 | 8.35 × 10−1 | 8.24 × 10−1 | 8.24 × 10−1 | 8.24 × 10−1 | 8.24 × 10−1 | 8.24 × 10−1 | 8.24 × 10−1 |
| 0.2 | 2.40 × 101 | 1.80 × 101 | 1.01 × 101 | 5.53 | 3.30 | 2.19 | 1.62 | 1.30 | 1.11 | 8.33 × 10−1 | 7.99 × 10−1 | 7.99 × 10−1 | 7.99 × 10−1 | 7.99 × 10−1 | 7.99 × 10−1 | 7.99 × 10−1 |
| 0.3 | 1.42 × 102 | 3.19 × 102 | 3.69 × 102 | 2.85 × 102 | 1.76 × 102 | 9.81 × 101 | 5.30 × 101 | 2.91 × 101 | 1.67 × 101 | 2.39 | 6.91 × 10−1 | 6.59 × 10−1 | 6.59 × 10−1 | 6.59 × 10−1 | 6.59 × 10−1 | 6.59 × 10−1 |
| 0.4 | 5.20 × 102 | 5.55 × 103 | 3.16 × 104 | 1.07 × 105 | 2.39 × 105 | 3.78 × 105 | 4.54 × 105 | 4.38 × 105 | 3.57 × 105 | 3.12 × 104 | 1.63 × 101 | 3.99 × 10−1 | 3.99 × 10−1 | 3.99 × 10−1 | 3.99 × 10−1 | 3.99 × 10−1 |
| 0.5 | 1.13 × 101 | 7.41 × 102 | 4.18 × 104 | 2.05 × 106 | 8.72 × 107 | 3.25 × 109 | 1.06 × 1011 | 3.05 × 1012 | 7.73 × 1013 | 1.40 × 1020 | 3.80 × 1032 | 4.60 × 1025 | 5.73 × 103 | 1.69 × 10−3 | 1.69 × 10−3 | 1.69 × 10−3 |
| ϕ/a | 8 (nm) | 12 (nm) | 16 (nm) | 20 (nm) | 24 (nm) | 28 (nm) | 32 (nm) | 36 (nm) | 40 (nm) | 60 (nm) | 120 (nm) | 500 (nm) | 1000 (nm) | 10,000 (nm) | 50,000 (nm) | 100,000 (nm) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.1 | 9.98 × 10−1 | 9.97 × 10−1 | 9.97 × 10−1 | 9.96 × 10−1 | 9.96 × 10−1 | 9.96 × 10−1 | 9.95 × 10−1 | 9.95 × 10−1 | 9.95 × 10−1 | 9.94 × 10−1 | 9.91 × 10−1 | 9.82 × 10−1 | 9.75 × 10−1 | 9.30 × 10−1 | 8.94 × 10−1 | 8.92 × 10−1 |
| 0.176 | 9.96 × 10−1 | 9.95 × 10−1 | 9.95 × 10−1 | 9.94 × 10−1 | 9.93 × 10−1 | 9.93 × 10−1 | 9.92 × 10−1 | 9.92 × 10−1 | 9.91 × 10−1 | 9.89 × 10−1 | 9.85 × 10−1 | 9.70 × 10−1 | 9.58 × 10−1 | 8.86 × 10−1 | 8.29 × 10−1 | 8.25 × 10−1 |
| 0.2 | 9.96 × 10−1 | 9.95 × 10−1 | 9.94 × 10−1 | 9.93 × 10−1 | 9.92 × 10−1 | 9.92 × 10−1 | 9.91 × 10−1 | 9.91 × 10−1 | 9.90 × 10−1 | 9.88 × 10−1 | 9.83 × 10−1 | 9.65 × 10−1 | 9.52 × 10−1 | 8.69 × 10−1 | 8.04 × 10−1 | 8.00 × 10−1 |
| 0.3 | 9.92 × 10−1 | 9.90 × 10−1 | 9.88 × 10−1 | 9.87 × 10−1 | 9.86 × 10−1 | 9.85 × 10−1 | 9.83 × 10−1 | 9.82 × 10−1 | 9.82 × 10−1 | 9.77 × 10−1 | 9.68 × 10−1 | 9.37 × 10−1 | 9.12 × 10−1 | 7.69 × 10−1 | 6.66 × 10−1 | 6.60 × 10−1 |
| 0.4 | 9.82 × 10−1 | 9.78 × 10−1 | 9.74 × 10−1 | 9.71 × 10−1 | 9.69 × 10−1 | 9.66 × 10−1 | 9.64 × 10−1 | 9.62 × 10−1 | 9.60 × 10−1 | 9.51 × 10−1 | 9.31 × 10−1 | 8.66 × 10−1 | 8.16 × 10−1 | 5.61 × 10−1 | 4.09 × 10−1 | 4.01 × 10−1 |
| 0.5 | 9.27 × 10−1 | 8.87 × 10−1 | 8.56 × 10−1 | 8.32 × 10−1 | 8.12 × 10−1 | 7.95 × 10−1 | 7.80 × 10−1 | 7.67 × 10−1 | 7.54 × 10−1 | 7.06 × 10−1 | 6.10 × 10−1 | 3.67 × 10−1 | 2.44 × 10−1 | 1.81 × 10−2 | 2.01 × 10−3 | 1.76 × 10−3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sajjad, M.; Otsuki, A. Coupling Flotation Rate Constant and Viscosity Models. Metals 2022, 12, 409. https://doi.org/10.3390/met12030409
Sajjad M, Otsuki A. Coupling Flotation Rate Constant and Viscosity Models. Metals. 2022; 12(3):409. https://doi.org/10.3390/met12030409
Chicago/Turabian StyleSajjad, Mohsin, and Akira Otsuki. 2022. "Coupling Flotation Rate Constant and Viscosity Models" Metals 12, no. 3: 409. https://doi.org/10.3390/met12030409
APA StyleSajjad, M., & Otsuki, A. (2022). Coupling Flotation Rate Constant and Viscosity Models. Metals, 12(3), 409. https://doi.org/10.3390/met12030409






