Vanadium and Melanoma: A Systematic Review
Abstract
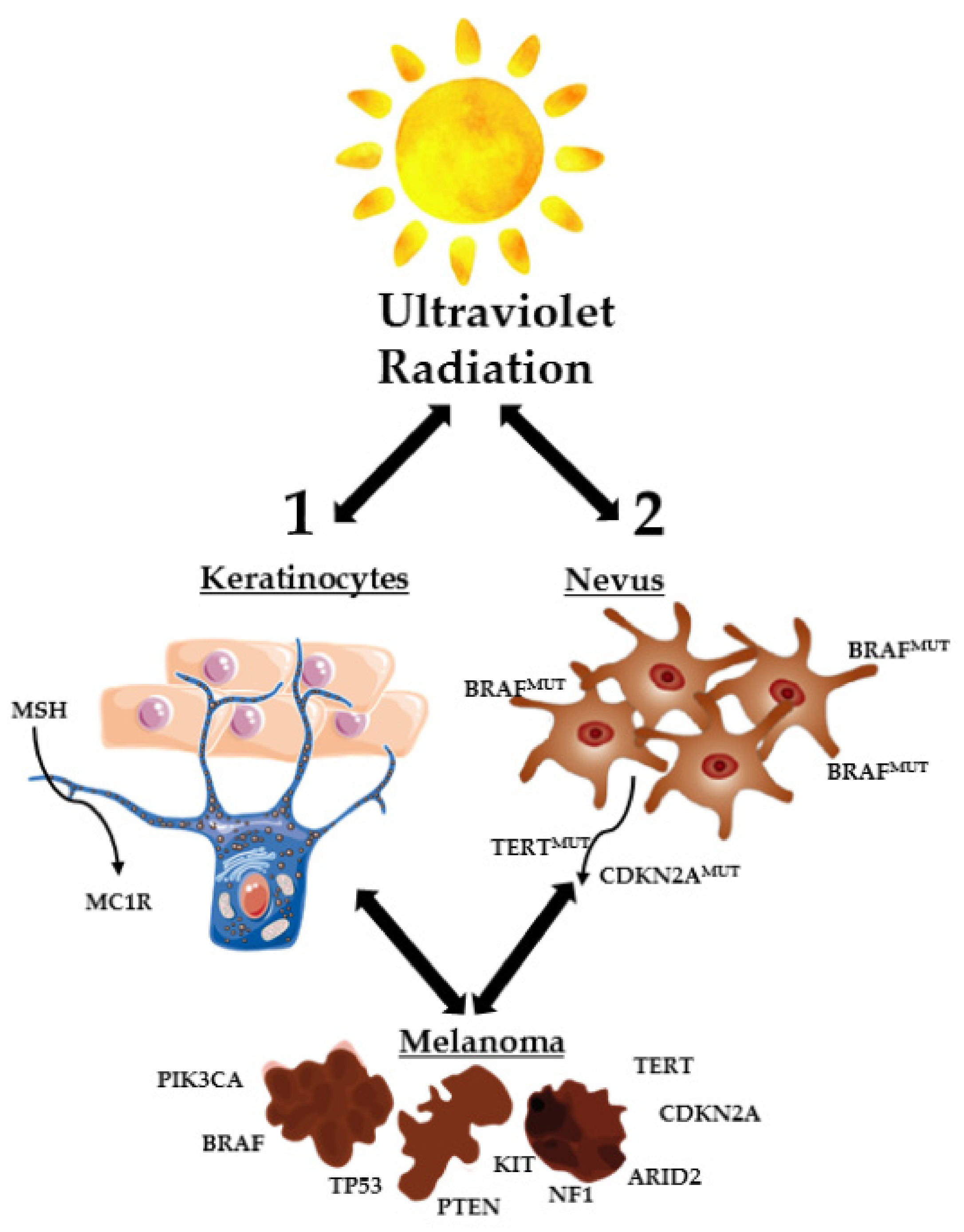
1. Introduction
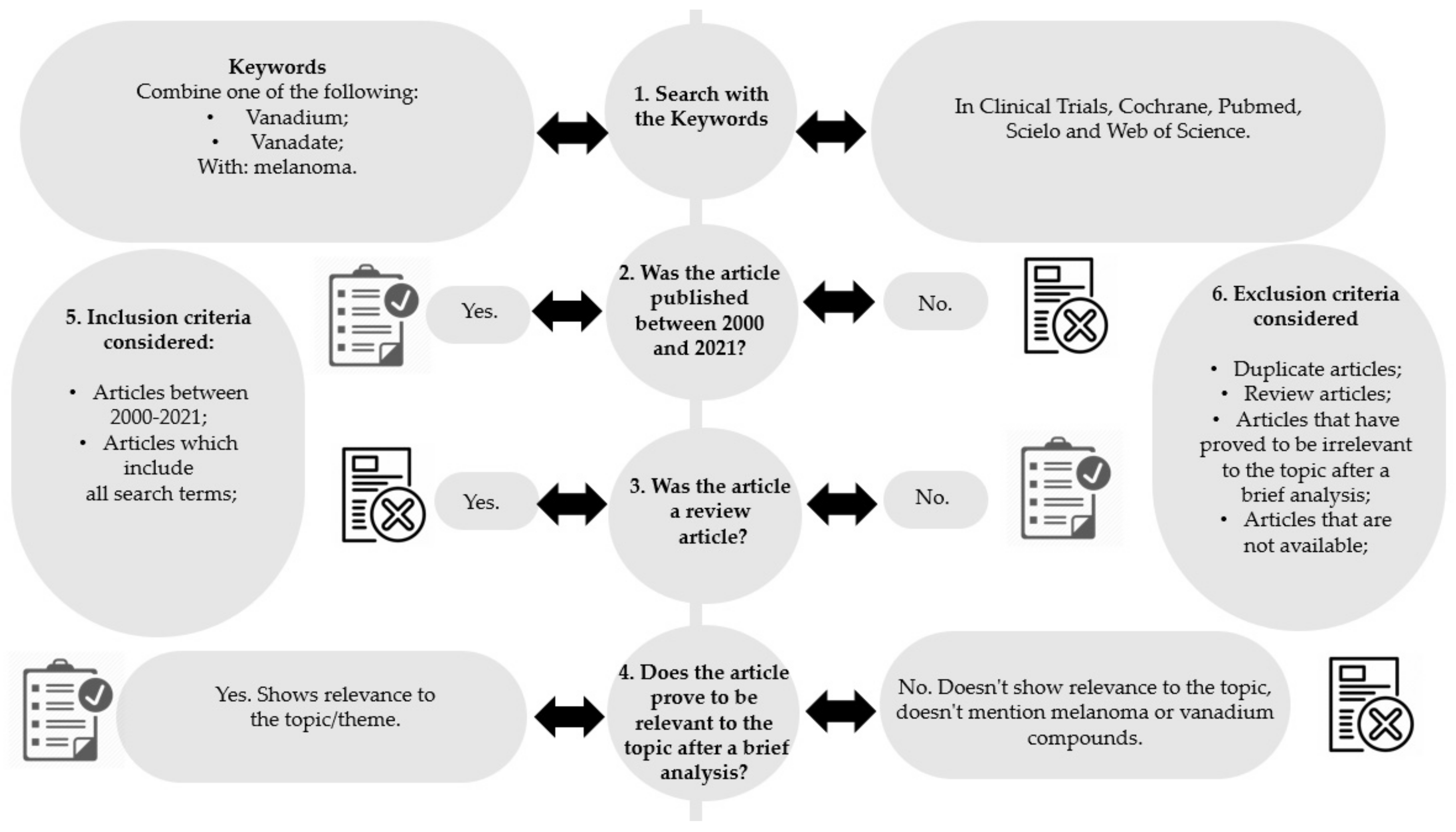
2. Methodology
- Removal of articles not published between 2000 and 2021 (Figure 2, step 2);
- Removal of review articles (Figure 2, step 3);
- Evaluation of titles/abstracts and obtaining all articles potentially relevant;
- Confirmation of the relevance or irrelevance of the articles obtained, checking for factors that would imply inclusion/exclusion, through the reading of the full articles (Figure 2, step 4);
- Organization of selected studies in a reference management program.
3. Types of Vanadium Based-Compounds Used in Studies against Melanoma
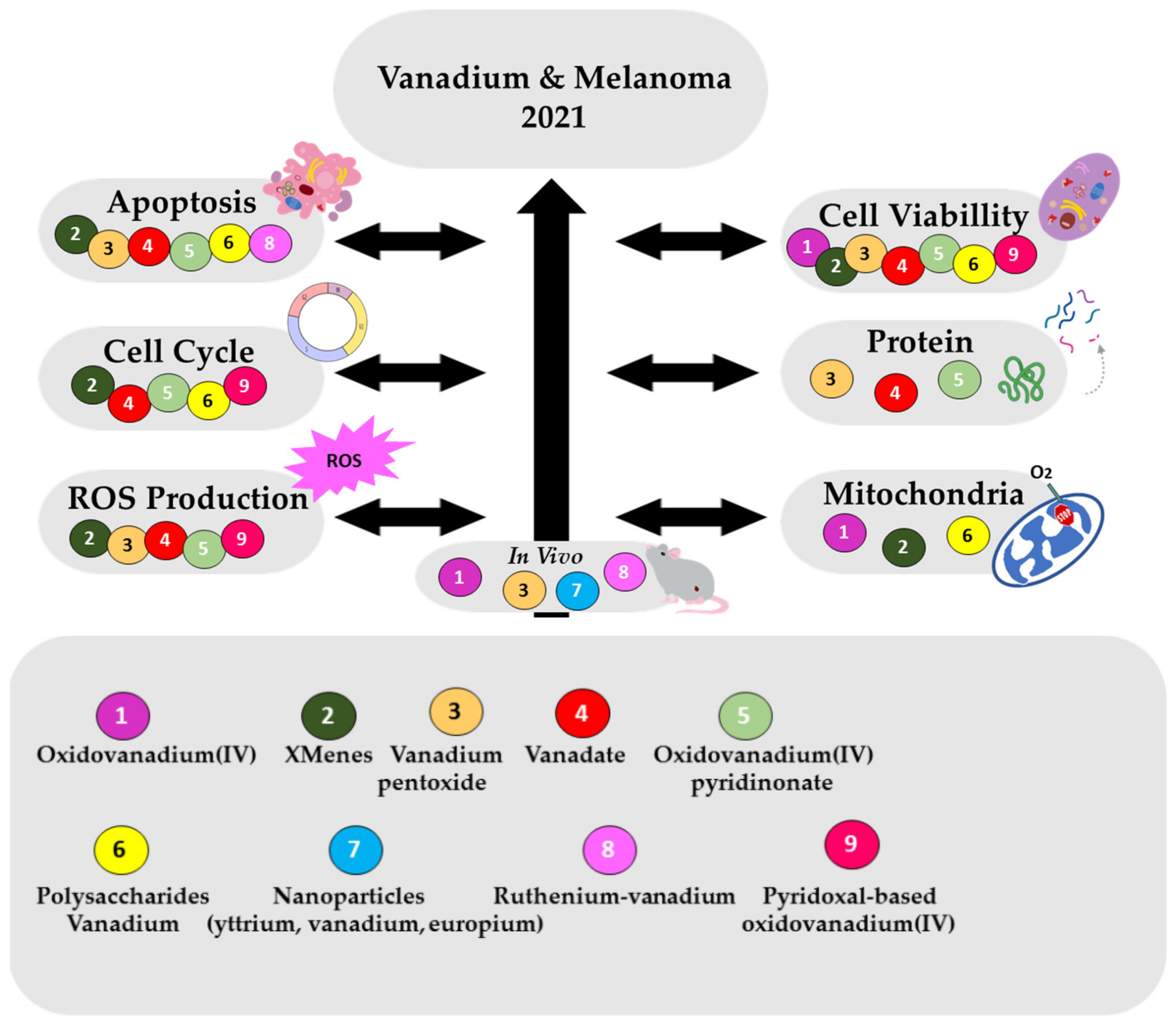
4. Vanadium Species, Complexes and/or Materials Effects in Melanoma Cells
4.1. Cell Viability Effects
4.2. Cell Morphology and Apoptosis Effects
4.3. Cell Cycle Effects
4.4. Effects on ROS Production
4.5. Effects on Mitochondria Function
4.6. Protein Expressions Effects
4.7. Anticancer Effects In Vivo
5. Discussion
5.1. Vanadyl
5.2. XMenes
5.3. Vanadium Pentoxide
5.4. Oxidovanadium(IV) Pyridinonate Compounds
5.5. Polysaccharides Vanadium(IV/V) Complexes
5.6. Mixed-Metal Binuclear Ruthenium(II)–Vanadium(IV) Complexes
5.7. Pyridoxal-Based Oxidovanadium(IV) Complexes
5.8. Nanoparticles of Yttrium Vanadate Doped with Europium
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| A375 | human malignant melanoma |
| A549 | human lung carcinoma |
| ARID2 | AT-rich interaction domain 2 |
| AsPC-1 | human pancreatic cancer |
| B16F10 | mus musculus skin melanoma |
| BRAF | B-Raf proto-oncogene |
| CDDP | cisplatin or cis-diamminedichloroplatinum(II) |
| CDK | cyclin dependent kinase |
| CDKN2A | cyclin-dependent kinase inhibitor 2A |
| CHO | chinese hamster ovary cells |
| CN-mel | human noncutaneous metastatic melanoma |
| CPTES | 3-chloropropyltrimethoxysilane |
| DCF-DA | fluorescent dye |
| DHE | dihydroethidium |
| ΔΨm | mitochondrial membrane potential |
| ERK | extracellular signal-regulated kinase |
| FA | folic acid |
| FITC | fluorescein isothiocyanate |
| FR | folate receptor |
| FSaR | fibrosarcoma cells |
| H69 | human small cell lung cancer |
| HaCaT | immortalized keratinocytes |
| HAM-F10 | F-10 nutient medium |
| HDL | high density lipoprotein |
| HEK-293 | human embryonic cells |
| HepG2 | human hepatocarcinoma |
| HT29 | human colon adenocarcinoma |
| IFN-γ | interferon gamma |
| KIT | KIT proto-oncogene receptor tyrosine kinase |
| LDL | low density lipoprotein |
| L929 | murine fibroblasts |
| MAPK | mitogen-activated protein kinase |
| MC1R | melanocortin 1 receptor |
| MDA-MB 231 | breast adenocarcinoma |
| MCF7 | human breast cancer |
| MOI | multiplicity of infection |
| MSH | melanocyte stimulating hormone |
| MTT | [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide)] |
| Na+/K+-ATPase | Na+/K+ pump ATP dependent |
| NAC | N-acetylcysteine |
| NDV | Newcastle disease virus |
| NEK | normal human epidermal keratinocytes |
| NF1 | neurofibromin 1 |
| NRK-49F | rat kidney cell line |
| NPs | nanoparticles |
| NuLi | normal lung cells |
| O2- | anion superoxide |
| PANC1 | human pancreatic cancer cells |
| PARP | Poly (ADP-ribose) polymerase |
| PBMC | peripheral blood mononuclear cells |
| PBS | phosphate buffer |
| ph-Rb | phosphorylated retinoblastoma protein |
| PI | propidium iodide |
| PIK3CA | phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha |
| POTs | polyoxotungstates |
| PTEN | phosphatase and tensin homolog |
| Pyr2enVO | N,N’-ethylenebis (pyridoxylideneiminat) vanadium(IV) |
| Rb | retinoblastoma protein |
| ROS | reactive oxygen species |
| RuVO | [Ru-(pbt)2(tpphz)VO(sal-L-tryp)]Cl2 |
| SCCVII | squamous carcinoma cells |
| SU-5416 | semaxanib |
| TdLNs | tumor-draining lymph nodes |
| TERT | telomerase reverse transcriptase |
| TME | tumor microenvironment |
| TP53 | tumor protein p53 |
| U266 | human multiple myeloma |
| VN | monomeric vanadate |
| V79 | hamster lung fibroblast cell line |
| VO | oxidovanadium (IV) |
| VOSO4 | vanadyl sulphate |
| VP | Vanadium pentoxide |
| VS2 | [VIVO(dhp)2], 1,2-dimethyl-3-hydroxy-4(1H)-pyridinonate |
| VS3 | [VIVO (mpp)2], mpp: 1-methyl-3-hydroxy-4(1H) pyridinonate |
| VS4 | [VIVO(ppp)2], ppp: 1-phenyl-2-methyl-3-hydroxy-4(1H)-pyridinonate |
| W10 | decatungstate |
| XGC | Xyloglucan |
| XGC:VO | Xyloglucan oxovanadium |
| XMenes | transition metal carbides/nitrides |
References
- Mondal, A.H.; Behera, T.; Swain, P.; Das, R.; Sahoo, S.N.; Mishra, S.S.; Das, J.; Ghosh, K. Nano zinc vis-à-vis inorganic Zinc as feed additives: Effects on growth, activity of hepatic enzymes and non-specific immunity in rohu, Labeo rohita (Hamilton) fingerlings. Aquac. Nutr. 2020, 26, 1211–1222. [Google Scholar] [CrossRef]
- Yan, S.; Wu, F.; Zhou, S.; Yang, J.; Tang, X.; Ye, W. Zinc oxide nanoparticles alleviate the arsenic toxicity and decrease the accumulation of arsenic in rice (Oryza Sativa L.). BMC Plant Biol. 2021, 21, 150. [Google Scholar] [CrossRef]
- Carofiglio, M.; Barui, S.; Cauda, V.; Laurenti, M. Doped zinc oxide nanoparticles: Synthesis, characterization and potential use in nanomedicine. Appl. Sci. 2020, 10, 5194. [Google Scholar] [CrossRef]
- Arentz, S.; Hunter, J.; Yang, G.; Goldenberg, J.; Beardsley, J.; Myers, S.P.; Mertz, D.; Leeder, S. Zinc for the prevention and treatment of SARS-CoV-2 and other acute viral respiratory infections: A rapid review. Adv. Integr. Med. 2020, 7, 252–260. [Google Scholar] [CrossRef]
- Cheng, P.; Wang, Y.; Sarakha, M.; Mailhot, G. Enhancement of the photocatalytic activity of decatungstate, W10O324−, for the oxidation of sulfasalazine/sulfapyridine in the presence of hydrogen peroxide. J. Photochem. Photobiol. A Chem. 2021, 404, 112890. [Google Scholar] [CrossRef]
- Pimpão, C.; da Silva, I.V.; Mósca, A.F.; Pinho, J.O.; Gaspar, M.M.; Gumerova, N.I.; Rompel, A.; Aureliano, M.; Soveral, G. The aquaporin-3-inhibiting potential of polyoxotungstates. Int. J. Mol. Sci. 2020, 21, 2467. [Google Scholar] [CrossRef] [PubMed]
- Gumerova, N.; Krivosudsky, L.; Fraqueza, G.; Breibeck, J.; Al-Sayed, E.; Tanuhadi, E.; Bijelic, A.; Fuentes, J.; Aureliano, M.; Rompel, A. The P-type ATPase inhibiting potential of polyoxotungstates. Metallomics 2018, 10, 287–295. [Google Scholar] [CrossRef]
- Fonseca, C.; Fraqueza, G.; Carabineiro, S.A.C.; Aureliano, M. The Ca2+-ATPase inhibition potential of gold (I,III) compounds. Inorganics 2020, 8, 49. [Google Scholar] [CrossRef]
- Li, P.; Chen, P.; Wang, G.; Wang, L.; Wang, X.; Li, Y.; Zhang, W.; Jiang, H.; Chen, H. Uranium elimination and recovery from wastewater with ligand chelation-enhanced electrocoagulation. Chem. Eng. J. 2020, 393, 124819. [Google Scholar] [CrossRef]
- Vijaya, P.; Kaur, H.; Garg, N.; Sharma, S. Protective and therapeutic effects of garlic and tomato on cadmium-induced neuropathology in mice. J. Basic Appl. Zool. 2020, 81, 23. [Google Scholar] [CrossRef]
- Obeng-Gyasi, E. Chronic cadmium exposure and cardiovascular disease in adults. J. Environ. Sci. Health Part A 2020, 55, 726–729. [Google Scholar] [CrossRef] [PubMed]
- Vosahlikova, M.; Roubalova, L.; Cechova, K.; Kaufman, J.; Musil, S.; Miksik, I.; Alda, M.; Svoboda, P. Na+/K+-ATPase and lipid peroxidation in forebrain cortex and hippocampus of sleep-deprived rats treated with therapeutic lithium concentration for different periods of time. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2020, 102, 109953. [Google Scholar] [CrossRef] [PubMed]
- Hosseinzadeh, Z.; Hauser, S.; Singh, Y.; Pelzl, L.; Schuster, S.; Sharma, Y.; Höflinger, P.; Zacharopoulou, N.; Stournaras, C.; Rathbun, D.L.; et al. Decreased Na+/K+ ATPase expression and depolarized cell membrane in neurons differentiated from chorea-acanthocytosis patients. Sci. Rep. 2020, 10, 8391. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Arnaiz, S.; Tate, R.J.; Grant, M.H. Cytotoxicity of cobalt chloride in brain cell lines—A comparison between astrocytoma and neuroblastoma cells. Toxicol. Vitro 2020, 68, 104958. [Google Scholar] [CrossRef]
- Bejarbaneh, M.; Moradi-Shoeili, Z.; Jalali, A.; Salehzadeh, A. Synthesis of cobalt hydroxide nano-flakes functionalized with glutamic acid and conjugated with thiosemicarbazide for anticancer activities against human breast cancer cells. Biol. Trace Elem. Res. 2020, 198, 98–108. [Google Scholar] [CrossRef]
- Treviño, S.; Díaz, A.; Sánchez-Lara, E.; Sanchez-Gaytan, B.L.; Perez-Aguilar, J.M.; González-Vergara, E. Vanadium in biological action: Chemical, pharmacological aspects, and metabolic implications in diabetes mellitus. Biol. Trace Elem. Res. 2019, 188, 68–98. [Google Scholar] [CrossRef]
- Sánchez-Lara, E.; Treviño, S.; Sánchez-Gaytán, B.L.; Sánchez-Mora, E.; Castro, M.E.; Meléndez-Bustamante, F.J.; Méndez-Rojas, M.A.; González-Vergara, E. Decavanadate salts of cytosine and metformin: A combined experimental-theoretical study of potential metallodrugs against diabetes and cancer. Front. Chem. 2018, 6, 402. [Google Scholar] [CrossRef]
- Zhang, Q.K.; Yue, C.P.; Zhang, Y.; Lu, Y.; Hao, Y.P.; Miao, Y.L.; Li, J.P.; Liu, Z.Y. Six metal-organic frameworks assembled from asymmetric triazole carboxylate ligands: Synthesis, crystal structures, photoluminescence properties and antibacterial activities. Inorg. Chim. Acta 2018, 473, 112–120. [Google Scholar] [CrossRef]
- Wang, X.T.; Li, R.Y.; Liu, A.G.; Yue, C.P.; Wang, S.M.; Cheng, J.J.; Li, J.P.; Liu, Z.Y. Syntheses, crystal structures, antibacterial activities of Cu (II) and Ni (II) complexes based on terpyridine polycarboxylic acid ligand. J. Mol. Struct. 2019, 1184, 503–511. [Google Scholar] [CrossRef]
- Leonardi, G.C.; Falzone, L.; Salemi, R.; Zanghì, A.; Spandidos, D.A.; Mccubrey, J.A.; Candido, S.; Libra, M. Cutaneous melanoma: From pathogenesis to therapy. Int. J. Oncol. 2018, 52, 1071–1080. [Google Scholar] [CrossRef]
- Rastrelli, M.; Tropea, S.; Rossi, C.R.; Alaibac, M. Melanoma: Epidemiology, risk factors, pathogenesis, diagnosis and classification. Vivo 2014, 28, 1005–1011, PMID: 25398793. [Google Scholar] [PubMed]
- Luke, J.J.; Flaherty, K.T.; Ribas, A.; Long, G.V. Targeted agents and immunotherapies: Optimizing outcomes in melanoma. Nat. Rev. Clin. Oncol. 2017, 14, 463–482. [Google Scholar] [CrossRef] [PubMed]
- Paluncic, J.; Kovacevic, Z.; Jansson, P.J.; Kalinowski, D.; Merlot, A.M.; Huang, M.L.-H.; Sahni, S.; Lane, D.J.R.; Richardson, D.R. Roads to melanoma: Key pathways and emerging players in melanoma progression and oncogenic signaling. Biochim. Biophys. Acta 2016, 1863, 770–784. [Google Scholar] [CrossRef] [PubMed]
- Grayschopfer, V.; Wellbrock, C.; Marais, R. Melanoma biology and new targeted therapy. Nature 2007, 445, 851–857. [Google Scholar] [CrossRef] [PubMed]
- Ali, Z.; Yousaf, N.; Larkin, J. Melanoma epidemiology, biology and prognosis. Eur. J. Cancer Suppl. 2013, 11, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, A.; Toh, Y. Human pluripotent stem cell-derived neural crest cells for tissue regeneration and disease modeling. Front. Mol. Neurosci. 2019, 12, 39. [Google Scholar] [CrossRef]
- Mcausland, T.M.; Vloten, J.P.V.; Santry, L.A.; Guilleman, M.M.; Rghei, A.D.; Ferreira, E.M.; Ingrao, J.C.; Arulanandam, R.; Major, P.P.; Susta, L.; et al. Combining vanadyl sulfate with Newcastle disease virus potentiates rapid innate immune-mediated regression with curative potential in murine cancer models. Mol. Ther. Oncolytics 2021, 20, 306–324. [Google Scholar] [CrossRef]
- Jastrzębska, A.M.; Scheibe, B.; Szuplewska, A.; Rozmysłowska-Wojciechowska, A.; Chudy, M.; Aparicio, C.; Scheibe, M.; Janica, I.; Ciesielski, A.; Otyepka, M.; et al. On the rapid in situ oxidation of two-dimensional V 2 CT z MXene in culture cell media and their cytotoxicity. Mater Sci. Eng. C 2021, 119, 111431. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Roy, A.; Barui, A.K.; Alabbasi, M.M.A.; Kuncha, M.; Sistla, R.; Sreedhar, B.; Patra, C.R. Anti-angiogenic vanadium pentoxide nanoparticles for the treatment of melanoma and their in vivo toxicity study. Nanoscale 2020, 12, 7604–7621. [Google Scholar] [CrossRef]
- Pisano, M.; Arru, C.; Serra, M.; Galleri, G.; Sanna, D.; Garribba, E.; Palmieri, G.; Rozzo, C. Antiproliferative activity of vanadium compounds: Effects on the major malignant melanoma molecular pathways. Metallomics 2019, 11, 1687–1699. [Google Scholar] [CrossRef]
- Rozzo, C.; Sanna, D.; Garribba, E.; Serra, M.; Cantara, A.; Palmieri, G.; Pisano, M. Antitumoral effect of vanadium compounds in malignant melanoma cell lines. J. Inorg. Biochem. 2017, 174, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Farias, C.L.A.; Martinez, G.R.; Cadena, S.M.S.C.; Mercê, A.L.R.; Petkowicz, C.L.; Noleto, G.R. Cytotoxicity of xyloglucan from Copaifera langsdorffii and its complex with oxovanadium (IV/V) on B16F10 cells. Int. J. Biol. Macromol. 2018, 121, 1019–1028. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, N.H.; Furtado, R.A.; Ribeiro, A.B.; Oliveira, P.F.; Ozelin, S.D.; Souza, L.D.R.; Neto, F.R.; Miura, B.A.; Magalhães, G.M.; Nassar, E.J.; et al. Europium (III)—Doped yttrium vanadate nanoparticles reduce the toxicity of cisplatin. J. Inorg. Biochem. 2018, 182, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Holder, A.A.; Taylor, P.; Magnusen, A.R.; Moffett, E.T.; Meyer, K.; Hong, Y.; Ramsdale, S.E.; Gordon, M.; Stubbs, J.; Seymour, L.A.; et al. Preliminary anti-cancer photodynamic therapeutic in vitro studies with mixed-metal binuclear ruthenium (II)–vanadium (IV) complexes. Dalton Trans. 2013, 42, 11881–11899. [Google Scholar] [CrossRef] [PubMed]
- Strianese, M.; Basile, A.; Mazzone, A.; Morello, S.; Turco, M.C.; Pellecchia, C. Therapeutic potential of a pyridoxal-based vanadium (IV) complex showing selective cytotoxicity for cancer vs. healthy cells. J. Cell. Physiol. 2013, 228, 2202–2209. [Google Scholar] [CrossRef]
- Burman, B.; Pesci, G.; Zamarin, D. Newcastle disease virus at the forefront of cancer immunotherapy. Cancers 2020, 12, 3552. [Google Scholar] [CrossRef]
- Lacal, P.M.; Failla, C.M.; Pagani, E.; Odorisio, T.; Schietroma, C.; Falcinelli, S.; Zambruno, G.; D’Atr, S. Human melanoma cells secrete and respond to placenta growth factor and vascular endothelial growth factor. J. Investig. Dermatol. 2000, 115, 1000–1007. [Google Scholar] [CrossRef]
- Ogawara, K.; Abe, S.; Un, K.; Yoshizawa, Y.; Kimura, T.; Higaki, K. Determinants for in vivo antitumor effect of angiogenesis inhibitor SU5416 formulated in PEGylated emulsion. J. Pharm. Sci. 2014, 103, 2464–2469. [Google Scholar] [CrossRef]
- Aldini, G.; Altomare, A.; Baron, G.; Vistoli, G.; Carini, M.; Borsani, L.; Sergio, F. N-Acetylcysteine as an antioxidant and disulphide breaking agent: The reasons why. Free Radic. Res. 2018, 52, 751–762. [Google Scholar] [CrossRef]
- Scibior, A.; Pietrzyk, L.; Plewa, Z.; Skiba, A. Vanadium: Risks and possible benefits in the light of a comprehensive overview of its pharmacotoxicological mechanisms and multi-applications with a summary of further research trends. J. Trace. Elem. Med. Biol. 2020, 61. [Google Scholar] [CrossRef]
- Evangelou, A.M. Vanadium in cancer treatment. Crit. Rev. Oncol. Hematol. 2002, 42, 249–265. [Google Scholar] [CrossRef]
- Pessoa, J.C.; Etcheverry, S.; Gambino, D. Vanadium compounds in medicine. Coord. Chem. Rev. 2015, 301, 24–48. [Google Scholar] [CrossRef] [PubMed]
- Barrio, D.A.; Etcheverry, S.B. Potential use of vanadium compounds in therapeutics. Curr. Med. Chem. 2010, 17, 3632–3642. [Google Scholar] [CrossRef]
- Bijelic, A.; Aureliano, M.; Rompel, A. Polyoxometalates as potential next-generation metallodrugs in the combat against cancer. Angew. Chem. Int. Ed. Engl. 2019, 58, 2980–2999. [Google Scholar] [CrossRef]
- Bijelic, A.; Aureliano, M.; Rompel, A. The antibacterial activity of polyoxometalates: Structures, antibiotic effects and future perspectives. Chem. Commun. 2018, 54, 1153–1169. [Google Scholar] [CrossRef]
- Imbert, V.; Rupec, R.A.; Livolsi, A.; Pahl, H.L.; Traenckner, E.B.; Mueller-Dieckmann, C.; Farahifar, D.; Rossi, B.; Auberger, P.; Baeuerle, P.A.; et al. Tyrosine phosphorylation of I kappa B-alpha activates NF-kappa B without proteolytic degradation of I kappa B-alpha. Cell 1996, 86, 787–798. [Google Scholar] [CrossRef]
- Yamamoto, F.; Fujioka, H.; Iinuma, M.; Takano, M.; Maeno, K.; Nagai, Y.; Ito, Y. Enhancement of Newcastle disease virus-induced fusion of mouse L Cells by sodium vanadate. Microbiol. Immun. 1984, 28, 75–83. [Google Scholar] [CrossRef] [PubMed]
- Korbecki, J.; Baranowska-Bosiacka, I.; Gutowska, I.; Chlubek, D. Biochemical and medical importance of vanadium compounds. Acta Biochim. Pol. 2012, 59, 195–200. [Google Scholar] [CrossRef]
- Rojas, E.; Valverde, M.; Herrera, L.A.; Altamirano-lozano, M.; Ostrosky-Wegman, P. Genotoxicity of vanadium pentoxide evaluate by the single cell gel electrophoresis assay in human lymphocytes. Mutat. Res. 1996, 359, 77–84. [Google Scholar] [CrossRef]
- Kang, Y.; Liu, J.; Wu, J.; Yin, Q.; Liang, H.; Chen, A.; Shao, L. Graphene oxide and reduced graphene oxide induced neural pheochromocytoma-derived PC12 cell lines apoptosis and cell cycle alterations via the ERK signaling pathways. Int. J. Nanomed. 2017, 12, 5501–5510. [Google Scholar] [CrossRef]
- Nandi, A.; Ghosh, C.; Bajpai, A.; Basu, S. Graphene oxide nanocells for impairing topoisomerase and DNA in cancer cells. J. Mater. Chem. B 2019, 7, 4191–4197. [Google Scholar] [CrossRef]
- Pérez-Torres, I.; Guarner-Lans, V.; Rubio-Ruiz, M.E. Reductive stress in inflammation-associated diseases and the pro-oxidant effect of antioxidant agents. Int. J. Mol. Sci. 2017, 18, 2098. [Google Scholar] [CrossRef] [PubMed]
- Skulachev, V.P. Role of uncoupled and non-coupled oxidations in maintenance of safely low levels of oxygen and its one-electron reductants. Q. Rev. Biophys. 1996, 29, 169–202. [Google Scholar] [CrossRef] [PubMed]
- Starkov, A.A.; Fiskum, G. Regulation of brain mitochondrial H2O2 production by membrane potential and NAD (P) H redox state. J. Neurochem. 2003, 86, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- Suma, P.R.P.; Padmanabhan, R.A.; Telukutla, S.R.; Ravindran, R.; Velikkakath, A.K.G.; Dekiwadia, C.D.; Paul, W.; Shenoy, S.J.; Laloraya, M.; Srinivasula, S.M.; et al. Paradigm of Vanadium pentoxide nanoparticle-induced autophagy and apoptosis in triple-negative breast cancer cells. bioRxiv 2019. [Google Scholar] [CrossRef]
- Ivanković, S.; Musić, S.; Gotić, M.; Ljubesić, N. Cytotoxicity of nanosize V2O5 particles to selected fibroblast and tumor cells. Toxicol. Vitro 2006, 20, 286–294. [Google Scholar] [CrossRef]
- Litz, J.; Warshamana-Greene, G.S.; Sulanke, G.; Lipson, K.E.; Krystal, G.W. The multi-targeted kinase inhibitor SU5416 inhibits small cell lung cancer growth and angiogenesis, in part by blocking Kit-mediated VEGF expression. Lung Cancer 2004, 46, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Kanapathipillai, M. Treating p53 Mutant Aggregation-Associated Cancer. Cancers 2018, 10, 154. [Google Scholar] [CrossRef]
- Cheung, C.H.A.; Chang, Y.; Lin, T.; Cheng, S.M.; Leung, E. Anti-apoptotic proteins in the autophagic world: An update on functions of XIAP, Survivin, and BRUCE. J. Biomed. Sci. 2020, 27, 31. [Google Scholar] [CrossRef]
- Kulkarni, A.; Kumar, G.S.; Kaur, J.; Tikoo, K. A comparative study of the toxicological aspects of vanadium pentoxide and vanadium oxide nanoparticles. Inhal. Toxicol. 2014, 26, 772–788. [Google Scholar] [CrossRef]
- Cohen, M.D.; Sisco, M.; Prophete, C.; Yoshida, K.; Chen, L.; Zelikoff, J.T.; Smee, J.; Holder, A.A.; Stonehuerner, J.; Crans, D.C.; et al. Effects of metal compounds with distinct physicochemical properties on iron homeostasis and antibacterial activity in the lungs: Chromium and vanadium. Inhal. Toxicol. 2010, 22, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Al-Qatati, A.; Fontes, F.L.; Barisas, B.G.; Zhang, D.; Roess, D.A.; Crans, D.C. Raft localization of Type I Fcε receptor and degranulation of RBL-2H3 cells exposed to decavanadate, a structural model for V2O5. Dalton Trans. 2013, 42, 11912–11920. [Google Scholar] [CrossRef]
- Aureliano, M.; Ohlin, C.A. Decavanadate in vitro and in vivo effects: Facts and opinions. J. Inorg. Biochem. 2014, 137, 123–130. [Google Scholar] [CrossRef]
- Wu, J.; Hong, Y.; Yang, X. Bis (acetylacetonato)-oxidovanadium (IV) and sodium metavanadate inhibit cell proliferation via ROS-induced sustained MAPK/ERK activation but with elevated AKT activity in human pancreatic cancer AsPC-1 cells. J. Biol. Inorg. Chem. 2016, 21, 919–929. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Wang, Q.; Yang, X.G.; Yang, X.D.; Wang, K. Vanadyl bisacetylacetonate induced G1/S cell cycle arrest via high-intensity ERK phosphorylation in HepG2 cells. J. Biol. Inorg. Chem. 2008, 13, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
- Rivadeneira, J.; Virgilio, A.L.D.; Barrio, D.A.; Muglia, C.I.; Bruzzone, L.; Etcheverry, S.B. Cytotoxicity of a vanadyl (IV) complex with a multidentate oxygen donor in osteoblast cell lines in culture. Med. Chem. 2010, 6, 9–23. [Google Scholar] [CrossRef]
- Dankner, M.; Rose, A.A.N.; Rajkumar, S.; Siegel, P.M.; Watson, I.R. Classifying BRAF alterations in cancer: New rational therapeutic strategies for actionable mutations. Oncogene 2018, 37, 3183–3199. [Google Scholar] [CrossRef]
- Broude, E.V.; Swift, M.E.; Vivo, C.; Chang, B.; Davis, B.M.; Kalurupalle, S.; Blagosklonny, M.V.; Roninson, I.B. p21(Waf1/Cip1/Sdi1) mediates retinoblastoma protein degradation. Oncogene 2007, 26, 6954–6958. [Google Scholar] [CrossRef]
- Kastenhuber, E.R.; Lowe, S.W. Putting p53 in context. Cell 2017, 170, 1062–1078. [Google Scholar] [CrossRef]
- Padua, M.M.C.; Cadena, S.M.S.C.; Petkowicz, C.L.O.; Martinez, G.R.; Rocha, M.E.M.; Mercê, A.L.R.; Noleto, G.R. Toxicity of native and oxovanadium (IV/V) galactomannan complexes on HepG2 cells is related to impairment of mitochondrial functions. Carbohydr. Polym. 2017, 173, 665–675. [Google Scholar] [CrossRef]
- Cao, Y.; Ikeda, I. Antioxidant activity and antitumor activity (in vitro) of xyloglucan selenious ester and surfated xyloglucan. Int. J. Biol. Macromol. 2009, 45, 231–235. [Google Scholar] [CrossRef]
- Kulkarni, A.D.; Joshi, A.A.; Patil, C.L.; Amale, P.D.; Patel, H.M.; Surana, S.J.; Belgamwar, V.S.; Chaudhari, K.S.; Pardeshi, C.V. Xyloglucan: A functional biomacromolecule for drug delivery applications. Int. J. Biol. Macromol. 2017, 104, 799–812. [Google Scholar] [CrossRef]
- Batista, Â.G.; Ferrari, A.S.; Cunha, D.C.; Silva, J.K.; Cazarin, C.B.B.; Correa, L.C.; Prado, M.A.; Carvalho-Silva, L.B.; Esteves, E.A.; Júnior, M.R.M. Polyphenols, antioxidants, and antimutagenic effects of copaifera langsdorffii fruit. Food Chem. 2016, 197, 1153–1159. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.S.; Gutiérrez-Merino, C.; Aureliano, M. Decavanadate induces mitochondrial membrane depolarization and inhibits oxygen consumption. J. Inorg. Biochem. 2007, 101, 789–796. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Prasad, P.; Hussain, A.; Khan, I.; Kondaiah, P.; Chakravarty, A.R. Remarkable photocytotoxicity of curcumin in HeLa cells in visible light and arresting its degradation on oxovanadium (IV) complex formation. Chem. Commun. 2012, 48, 7702–7704. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Hussain, A.; Prasad, P.; Khan, I.; Banik, B.; Kondaiah, P.; Chakravarty, A.R. Photocytotoxic oxidovanadium (IV) complexes of polypyridyl ligands showing DNA-cleavage activity in near-IR light. Eur. J. Inorg. Chem. 2012, 2012, 3899–3908. [Google Scholar] [CrossRef]
- Sasmal, P.K.; Patra, A.K.; Nethaji, M.; Chakravarty, A.R. DNA cleavage by new oxovanadium (IV) complexes of N-salicylidene alpha-amino acids and phenanthroline bases in the photodynamic therapy window. Inorg. Chem. 2007, 46, 11112–11121. [Google Scholar] [CrossRef] [PubMed]
- Kawashima, T.; Ohkubo, K.; Fukuzumi, S. Photoinduced DNA cleavage by formation of ROS from oxygen with a neurotransmitter and aromatic amino acids. Org. Biomol. Chem. 2010, 8, 994–996. [Google Scholar] [CrossRef]
- Siveen, K.S.; Kuttan, G. Inhibition of B16F-10 Melanoma—induced lung metastasis in C57BL/6 Mice by aerva lanata via induction of apoptosis. Integr. Cancer Ther. 2013, 12, 81–92. [Google Scholar] [CrossRef]
- Pattanayak, P.; Pratihar, J.L.; Patra, D.; Mitra, S.; Bhattacharyya, A.; Lee, H.M.; Chattopadhyay, S. Synthesis, structure and reactivity of azosalophen complexes of vanadium (IV): Studies on cytotoxic properties. Dalton Trans. 2009, 31, 6220–6230. [Google Scholar] [CrossRef]
- Sheth, S.; Mukherjea, D.; Rybak, L.P.; Ramkumar, V. Mechanisms of cisplatin-induced ototoxicity and otoprotection. Front. Cell. Neurosci. 2017, 11, 338. [Google Scholar] [CrossRef] [PubMed]
- Bahrami, B.; Mohammadnia-Afrouzi, M.; Bakhshaei, P.; Yazdani, Y.; Ghalamfarsa, G.; Yousefi, M.; Sadreddini, S.; Jadidi-Niaragh, F.; Hojjat-Farsangi, M. Folate-conjugated nanoparticles as a potent therapeutic approach in targeted cancer therapy. Tumour Biol. 2015, 36, 5727–5742. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.; Gao, C.; Liu, Q.; Yu, C.; Zhang, Z.; Cai, L.; Yang, B.; Qian, Y.; Yang, J.; Liao, X. Research progress in modern structure of platinum complexes. Eur. J. Med. Chem. 2017, 140, 349–382. [Google Scholar] [CrossRef] [PubMed]
- Ursic, K.; Kos, S.; Kamensek, U.; Cemazar, M.; Scancar, J.; Bucek, S.; Kranjc, S.; Staresinic, B.; Sersa, G. Comparable effectiveness and immunomodulatory actions of oxaliplatin and cisplatin in electrochemotherapy of murine melanoma. Bioelectrochemistry 2018, 119, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Doherty, A.T. The in vitro micronucleus assay. Methods Mol. Biol. 2012, 817, 121–141. [Google Scholar] [CrossRef] [PubMed]
- Gumerova, N.I.; Rompel, A. Interweaving disciplines to advance chemistry: Applying polyoxometalates in biology. Inorg. Chem. 2021, 60, 6109–6114. [Google Scholar] [CrossRef]
| Vanadium Compound/Material | Structure | Year (Ref) |
|---|---|---|
| VOSO4 | (VO2+)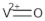 | 2021 [27] |
| Vanadyl sulfate (Abreviated VO) | ||
| 1 | ||
| V2CTz-ox24/ V2CTz-ox48 (V2CTz) |  | 2020 [28] |
| Vanadium carbides (MXenes) | ||
| 2 | ||
| V2O5 | 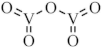 | 2020 [29] |
| Vanadium pentoxide (VP) | ||
| 3 | ||
| H2VO4— | 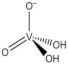 | 2019 [30], 2017 [31] |
| monomeric vanadate (VN) | ||
| 4 | ||
| VIVO(dhp)2 (VS2) | 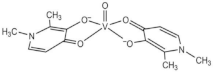 | 2019 [30], 2017 [31] |
| dhp: 1,2-dimethyl-3-hydroxy-4(1H)-pyridinonate | ||
| 5 | ||
| Xyloglucan oxovanadium (IV/V) (XGCVO) | 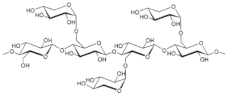 + 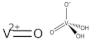 | 2018 [32] |
| 6 | ||
| YIIIVVO4: EuIII (YVEu NPs) | 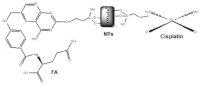 | 2018 [33] |
| Europium(III)-doped yttrium vanadate nanoparticles | ||
| 7 | ||
| [VIVO (mpp)2] (VS3) | 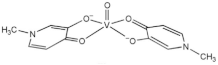 | 2017 [31] |
| mpp: 1-methyl-3-hydroxy-4(1H) pyridinonate | ||
| 8 | ||
| [VIVO(ppp)2] (VS4) | 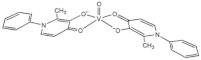 | 2017 [31] |
| ppp: 1-phenyl-2-methyl-3-hydroxy-4(1H)-pyridinonate | ||
| 9 | ||
| [Ru-(pbt)2(tpphz)VO(sal-L-tryp)]Cl2 | 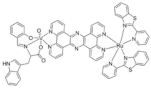 | 2013 [34] |
| (RuVO) | ||
| pbt = 2-(2′-pyridyl)benzothiazole | ||
| tpphz = tetrapyrido [3,2-a:2′,3′-c:3′′,2′′-h:2′′′,3′′′-j]phenazine | ||
| sal-L-tryp = N -salicylidene-L-tryptophanate | ||
| 10 | ||
| N,N’-ethylenebis (pyridoxylideneiminat) | 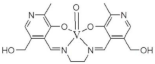 | 2013 [35] |
| vanadium (IV) complex (Pyr2enVO) | ||
| 11 |
| Compound | Cell Viability | Cell Morphology and Apoptosis Effects | Cell Cycle Effects | ROS Production | Mitochondrial Effects | Protein Expressions Studies | In Vivo Anticancer Activity |
|---|---|---|---|---|---|---|---|
| VO 1 | B16F10 cells | Mice Tumor regression upon VOSO4 (40 mg/kg), 96 h | |||||
| 200 µM produces 90% inhibition, after 48 h | |||||||
| V2CTz 2 | A375 cells | A375 cells The population of apoptotic cells was negligible, for 24 h | A375 cells Cellular cycle arrest in the G0/G1 phase, triggering apoptosis | A375 cells V2CTz-ox24 increased ROS to 225%, (100 µg/mL) V2CTz-ox48 increased ROS to 140% (100 µg/mL) | A375 cells s-V2CTz-ox48, slight increase mitochondrial membrane potential (ΔΨm) | ||
| 50% inhibition for 1–5 µg/mL V2CTz-ox48 and for 25–50 µg/mL V2CTz-ox24 | |||||||
| VP 3 | B16F10 cells | B16F10 cells Increase of apoptosis (10 µg/mL), for 18 h DNA damage (10 µg/mL), for 18 h | B16F10 cells Anion superoxide formation (10 µg/mL) | B16F10 cells Upregulation of p53 downregulation of anti-apoptotic survivin (10, 20 µg/mL) | Mice The survival rate increased up to 47 days, (10 mg/kg) No changes: body weight; in feed intake. No toxicity in vital organs | ||
| 50% inhibition for 10 µg/mL | |||||||
| VN 4 | A375 cells IC50 = 4.7 µM, 72 h | A375 cells Pisano, et al. Increased apoptotic population to 48%, (20 µg/mL), 48 h. Rozzo, et al. Increased apoptotic population to 70%, (20 µg/mL), 72 h | A375 cells Cells did not go through the G2/M phase, dying through apoptosis | A375 cells ROS increased to 80% (20 µg/mL), 48 h. | A375 cells ph-ERK, ph-Rb and ph-Cdc25c levels decreased to 20% (20 μM), 24 h, p21Cip1 rised up to 10-14 times (20 μM) | ||
| CN-mel IC50 = 6.5 µM, 72 h | |||||||
| VS2 5 | A375 cells IC50 = 2.6 µM, 72 h | A375 cells Pisano, et al. Increased apoptotic population up to 52%, at the highest concentration used (20 µg/mL) in 48 h assays. Rozzo, et al. Increased apoptotic population up to 67%, at the highest concentration used (20 µg/mL) in 72 h assays | A375 cells Cell cycle arrested in G0/G1 phase, showing not to be able to enter the S phase. | A375 cells ROS levels increasing up to 80% as well, at the highest duration (48 h) and dose (20µg/mL). | Pisano, et al. A375 cells ph-ERK, ph-Rb and ph-Cdc25c levels decreased to 20% (20 μM), p21Cip1 levels incresed up to 18 times (20 μM) Rozzo, et al. Increasing of the cleaved PARP band (85 kD) | ||
| CN-mel IC50 = 12.4 µM, 72 h | |||||||
| XGCVO 6 | B16F10 cells | B16F10 cells 50% increase of apoptosis (200 µg/mL) | B16F10 cells Cell cycle has not changed | B16F10 cells Mitocondrial respiration decreased to 43% (5 μg/mL) 34% pyruvate decrease No effects lactate production | |||
| 50% inhibition at 300 μg/mL | |||||||
| YVEu NPs 7 | Mice Tumor regression upon YIIIVVO4:EuIII: CPTES:FA: CDDP (15 mg/kg), 5 days; CDDP toxicity reduced | ||||||
| VS3 8 | A375 cells IC50 = 2.4 µM, 72 h | ||||||
| CN-mel IC50 = 10.4 µM, 72 h | |||||||
| VS4 9 | A375 cells IC50 = 4.2 µM, 72 h | ||||||
| CN-mel IC50 = 14.0 µM, 72 h | |||||||
| RuVO 10 | Amelanotic melanoma In the absence of light, changes in cell morphology (20 µM). In the presence of light, apoptosis observed, 20 µM. | Mice The survival rate was 100%. Tumor weight reduction. Proliferative activity reduction | |||||
| Pyr2enVO 11 | A375 cells | A375 cells Increase of G0/G1 cells to 60% (100 µM), 72 h | A375 cells ROS increased to 23% (100 µM), 24 h | A375 cells Percentage of cells with loss of ΔΨm increased up to 35% (100 μM), 48 h, and 73%, after 72 h | |||
| IC50 = 61.5 (24 h) IC50 = 13.0 (48 h) IC50 = 6.0 (72 h) | |||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amante, C.; De Sousa-Coelho, A.L.; Aureliano, M. Vanadium and Melanoma: A Systematic Review. Metals 2021, 11, 828. https://doi.org/10.3390/met11050828
Amante C, De Sousa-Coelho AL, Aureliano M. Vanadium and Melanoma: A Systematic Review. Metals. 2021; 11(5):828. https://doi.org/10.3390/met11050828
Chicago/Turabian StyleAmante, Cristina, Ana Luísa De Sousa-Coelho, and Manuel Aureliano. 2021. "Vanadium and Melanoma: A Systematic Review" Metals 11, no. 5: 828. https://doi.org/10.3390/met11050828
APA StyleAmante, C., De Sousa-Coelho, A. L., & Aureliano, M. (2021). Vanadium and Melanoma: A Systematic Review. Metals, 11(5), 828. https://doi.org/10.3390/met11050828








