Structural Evolution in Wet Mechanically Alloyed Co-Fe-(Ta,W)-B Alloys
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. X-ray Scattering
2.3. Transmission and Scanning Electron Microscopy
2.4. X-ray Absorption Spectroscopy
2.5. Magnetic Measurements
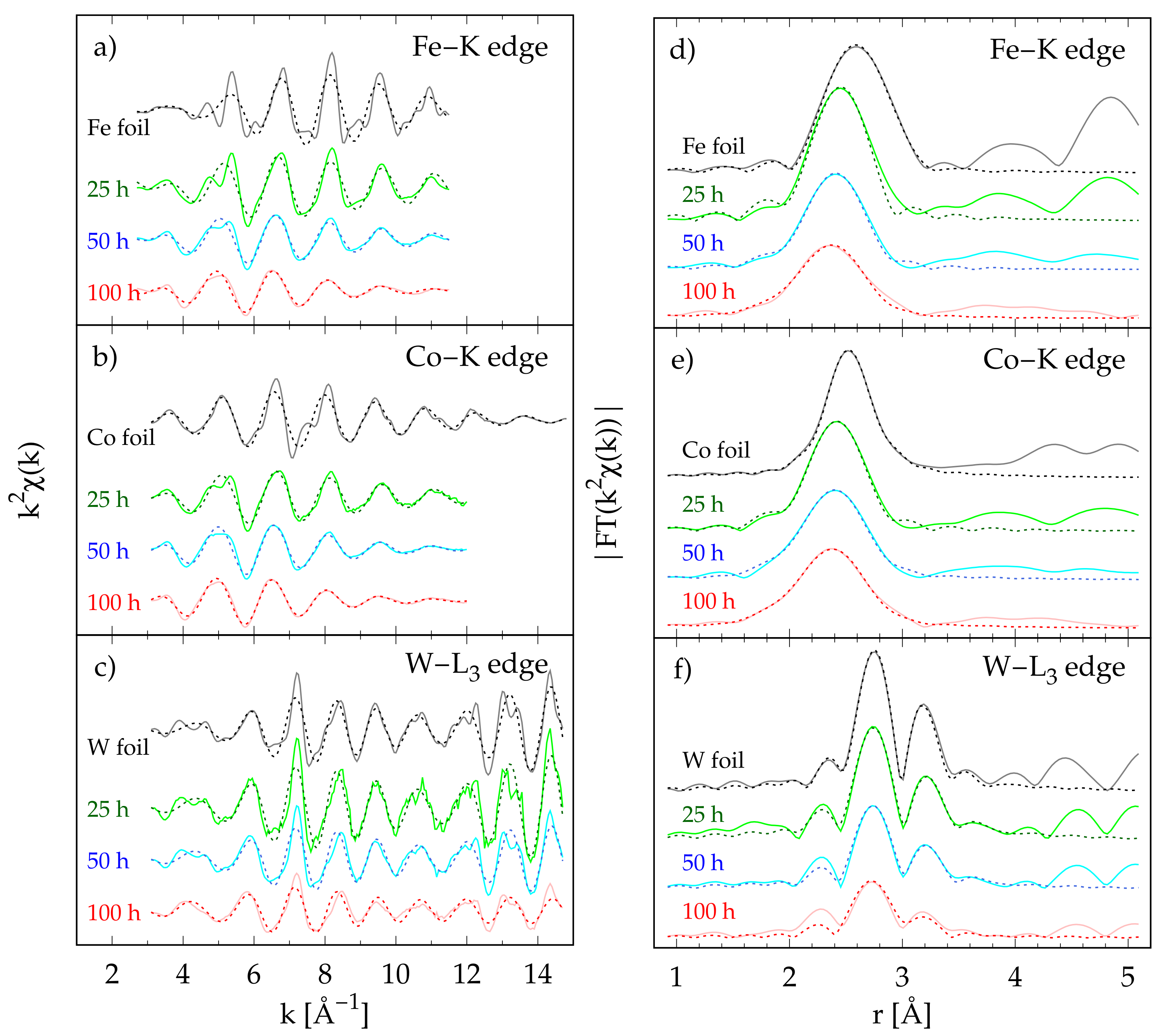
3. Results and Discussion
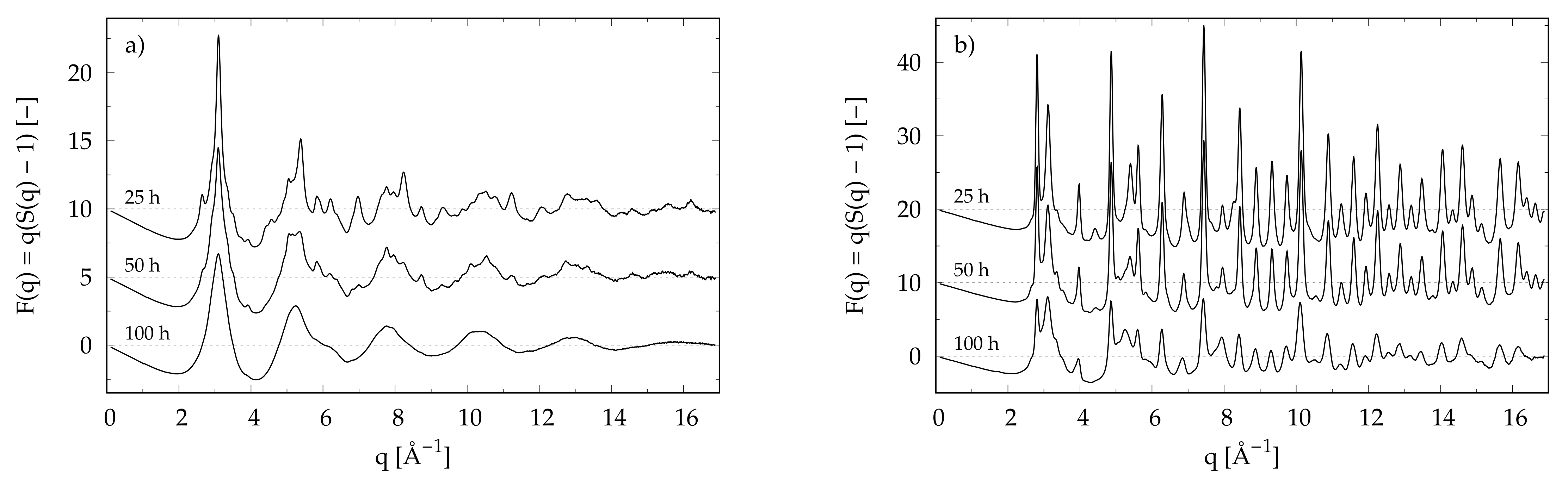
3.1. X-ray Diffraction
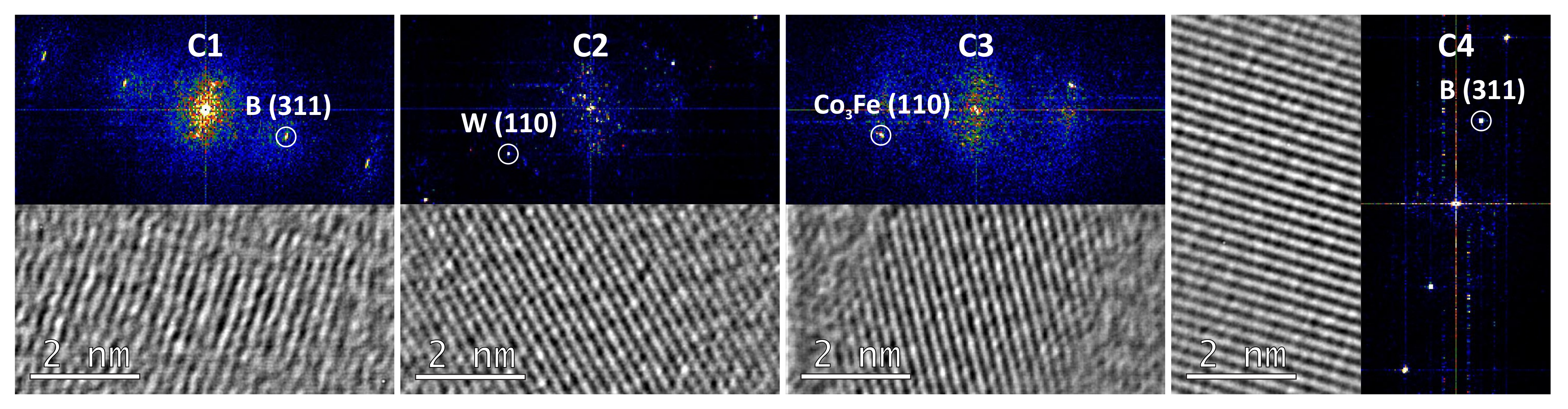
3.2. Transmission Electron Microscopy
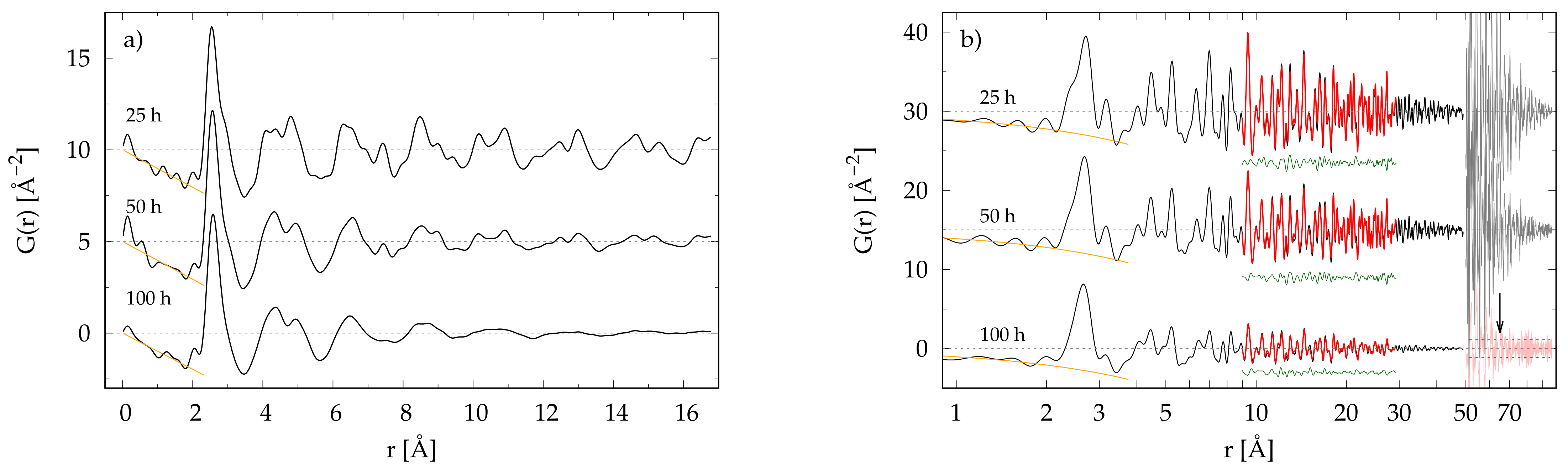
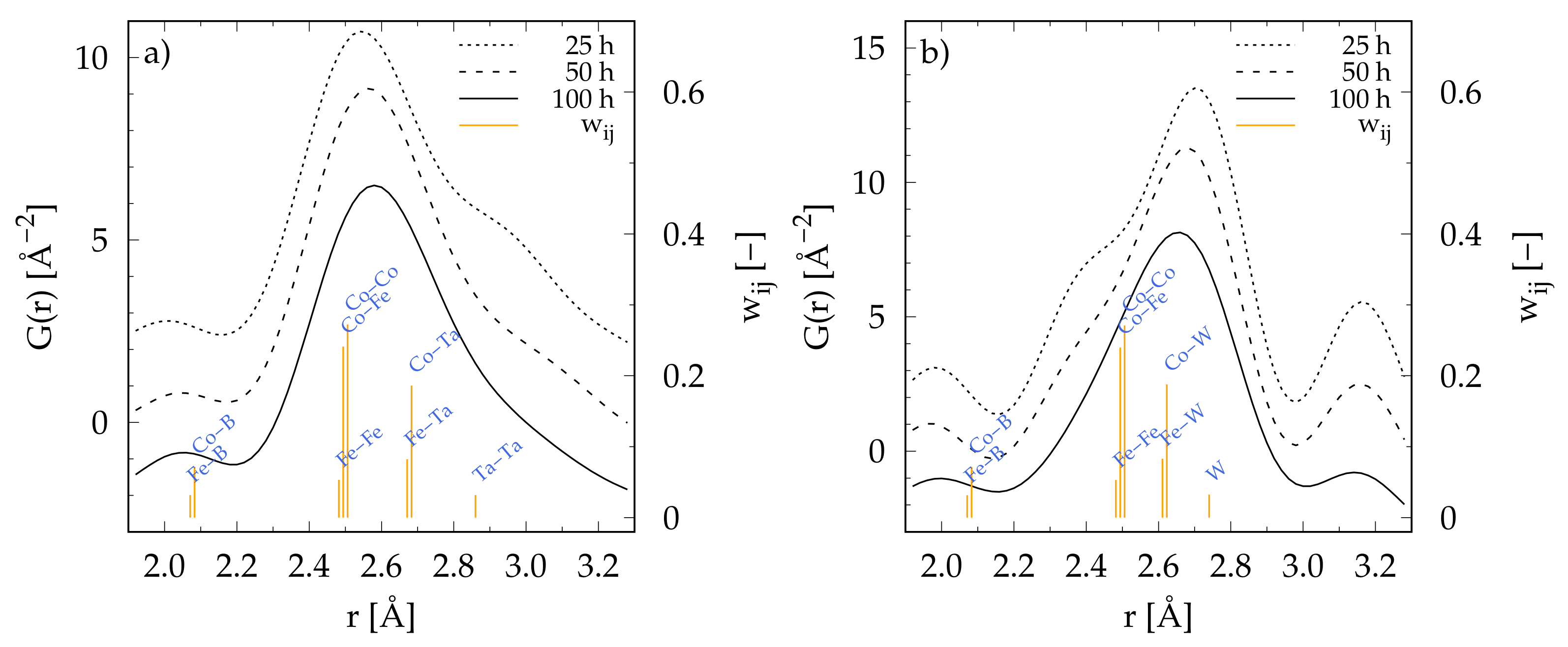
3.3. Local Atomic Structure from Pair Distribution Function
3.4. X-ray Absorption Spectroscopy
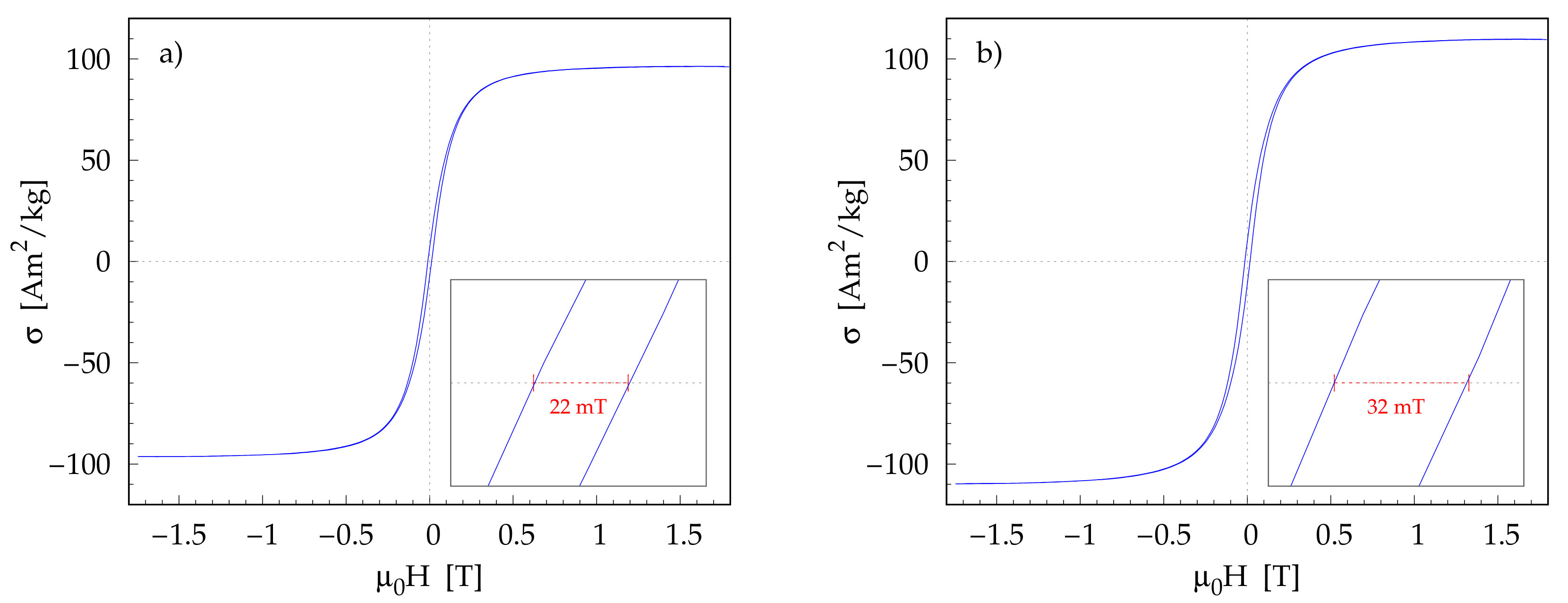
3.5. Magnetic Properties
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 2D | two-dimensional |
| CN | Coordination Number |
| COD | Crystallographic Open Database |
| EDX | Energy Dispersive X-ray |
| EXAFS | Extended X-ray Absorption Spectroscopy |
| FT | Fourier Transform |
| FWHM | Full Width at Half Maximum |
| GFA | Glass Forming Ability |
| HRTEM | High-Resolution Transmission Electron Microscopy |
| HV | Vickers Hardness |
| IRF | Instrumental Resolution Function |
| MA | Mechanical Alloying |
| Pair Distribution Function | |
| SAD | Selected Area Diffraction |
| SEM | Scanning Electron Microscopy |
| TEM | Transmission Electron Microscopy |
| VSM | Vibrating Sample Magnetometer |
| XRD | X-Ray Diffraction |
References
- Wu, S.; Liu, Z.; Qu, R.; Zhang, Z. Designing metallic glasses with optimal combinations of glass-forming ability and mechanical properties. J. Mater. Sci. Technol. 2021, 67, 254–264. [Google Scholar] [CrossRef]
- Qiang, J.; Estevez, D.; Chang, C.; Man, Q.; Li, R.W.; Wang, X.; Inoue, A. High strength CoFe-based glassy alloy with high thermal stability. Mater. Lett. 2014, 114, 126–128. [Google Scholar] [CrossRef]
- Narushima, T. New-generation metallic biomaterials. In Metals for Biomedical Devices; Elsevier: Amsterdam, The Netherlands, 2010; pp. 355–378. [Google Scholar] [CrossRef]
- Coimbrao, D.D.; Zepon, G.; Koga, G.Y.; Pérez, D.A.G.; de Almeida, F.H.P.; Roche, V.; Lepretre, J.C.; Jorge, A.M.; Kiminami, C.S.; Bolfarini, C.; et al. Corrosion properties of amorphous, partially, and fully crystallized Fe68Cr8Mo4Nb4B16 alloy. J. Alloy. Compd. 2020, 826, 154123. [Google Scholar] [CrossRef]
- Taghvaei, A.H.; Stoica, M.; Khoshkhoo, M.S.; Kaban, I.; Bednarčik, J.; Jóvári, P.; Janghorban, K.; Eckert, J. DSC, XRD and TEM characterization of glassy Co40Fe22Ta8B30 alloy with very high thermal stability. Mater. Lett. 2013, 93, 322–325. [Google Scholar] [CrossRef]
- Shen, B.; Inoue, A. Enhancement of the fracture strength and glass-forming ability of CoFeTaB bulk glassy alloy. J. Phys. Condens. Matter 2005, 17, 5647–5653. [Google Scholar] [CrossRef]
- Shen, B.; Pang, S.; Zhang, T.; Kimura, H.; Inoue, A. Corrosion properties of Co43Fe20Ta5.5B31.5 bulk glassy alloy. J. Alloy. Compd. 2008, 460, L11–L13. [Google Scholar] [CrossRef]
- Nicula, R.; Stir, M.; Ishizaki, K.; Catalá-Civera, J.M.; Vaucher, S. Nanocrystallization of amorphous alloys using microwaves:In situtime-resolved synchrotron radiation studies. J. Phys. Conf. Ser. 2009, 144, 012109. [Google Scholar] [CrossRef]
- Inoue, A.; Shen, B.; Koshiba, H.; Kato, H.; Yavari, A.R. Cobalt-based bulk glassy alloy with ultrahigh strength and soft magnetic properties. Nat. Mater. 2003, 2, 661–663. [Google Scholar] [CrossRef]
- Inoue, A.; Shen, B.; Koshiba, H.; Kato, H.; Yavari, A.R. Ultra-high strength above 5000 MPa and soft magnetic properties of Co–Fe–Ta–B bulk glassy alloys. Acta Mater. 2004, 52, 1631–1637. [Google Scholar] [CrossRef]
- Hitit, A.; Talasb, S.; Kara, R. Effects of silicon and chromium additions on glass forming ability and microhardness of Co-based bulk metallic glasses. Indian J. Eng. Mater. Sci. 2014, 21, 111–115. [Google Scholar]
- Raanaei, H.; Fakhraee, M. Synthesis and characterization of nanocrystalline Co-Fe-Nb-Ta-B alloy. J. Magn. Magn. Mater. 2017, 438, 144–151. [Google Scholar] [CrossRef]
- Yazici, Z.O.; Hitit, A.; Yalcin, Y.; Ozgul, M. Effects of minor Cu and Si additions on glass forming ability and mechanical properties of Co-Fe-Ta-B Bulk metallic glass. Met. Mater. Int. 2016, 22, 50–57. [Google Scholar] [CrossRef]
- Hitit, A.; Gecgin, M.; Ozturk, P. Effect of Annealing on Microstructure and Microhardness of Co–Fe–Ni–Ta–B–Si Bulk Metallic Glass. J. Mater. Sci. Technol. 2015, 31, 148–152. [Google Scholar] [CrossRef]
- Hitit, A.; Sahin, H. The Effect of Iron Content on Glass Forming Ability and Thermal Stability of Co–Fe–Ni–Ta–Nb–B–Si Bulk Metallic Glass. Metals 2016, 7, 7. [Google Scholar] [CrossRef]
- Suryanarayana, C. Mechanical alloying and milling. Prog. Mater. Sci. 2001, 46, 1–184. [Google Scholar] [CrossRef]
- Tan, J.H.; Wong, W.L.E.; Dalgarno, K.W. An overview of powder granulometry on feedstock and part performance in the selective laser melting process. Addit. Manuf. 2017, 18, 228–255. [Google Scholar] [CrossRef]
- Pauly, S.; Löber, L.; Petters, R.; Stoica, M.; Scudino, S.; Kühn, U.; Eckert, J. Processing metallic glasses by selective laser melting. Mater. Today 2013, 16, 37–41. [Google Scholar] [CrossRef]
- Deng, L.; Gebert, A.; Zhang, L.; Chen, H.Y.; Gu, D.D.; Kühn, U.; Zimmermann, M.; Kosiba, K.; Pauly, S. Mechanical performance and corrosion behaviour of Zr-based bulk metallic glass produced by selective laser melting. Mater. Des. 2020, 189, 108532. [Google Scholar] [CrossRef]
- Hammersley, A.P.; Svensson, S.O.; Hanfland, M.; Fitch, A.N.; Hausermann, D. Two-dimensional detector software: From real detector to idealised image or two-theta scan. High Press. Res. 1996, 14, 235–248. [Google Scholar] [CrossRef]
- Caglioti, G.; Paoletti, A.; Ricci, F.P. Choice of collimators for a crystal spectrometer for neutron diffraction. Nucl. Instrum. 1958, 3, 223–228. [Google Scholar] [CrossRef]
- Egami, T.; Billinge, S. Underneath the Bragg Peaks: Structural Analysis of Complex Materials; Pergamon Press: Oxford, UK; Elsevier: Amsterdam, The Netherlands, 2003; Volume 16. [Google Scholar]
- Waseda, Y. The Structure of Non-Crystalline Materials: Liquids and Amorphous Solids; McGraw-Hill Inc.: New York, NY, USA, 1980. [Google Scholar]
- Faber, T.E.; Ziman, J.M. A theory of the electrical properties of liquid metals. Philos. Mag. 1965, 11, 153–173. [Google Scholar] [CrossRef]
- Waasmaier, D.; Kirfel, A. New analytical scattering-factor functions for free atoms and ions. Acta Crystallogr. Sect. Found. Crystallogr. 1995, 51, 416–431. [Google Scholar] [CrossRef]
- Qiu, X.; Thompson, J.W.; Billinge, S.J.L. PDFgetX2: A GUI-driven program to obtain the pair distribution function from X-ray powder diffraction data. J. Appl. Crystallogr. 2004, 37, 678. [Google Scholar] [CrossRef]
- Farrow, C.L.; Juhas, P.; Liu, J.W.; Bryndin, D.; Božin, E.S.; Bloch, J.; Proffen, T.; Billinge, S.J.L. PDFfit2 and PDFgui: Computer programs for studying nanostructure in crystals. J. Phys. Condens. Matter 2007, 19, 335219. [Google Scholar] [CrossRef] [PubMed]
- Welter, E.; Chernikov, R.; Herrmann, M.; Nemausat, R. A beamline for bulk sample x-ray absorption spectroscopy at the high brilliance storage ring PETRA III. AIP Conf. Proc. 2019, 2054, 040002. [Google Scholar] [CrossRef]
- Ravel, B.; Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: Data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 2005, 12, 537–541. [Google Scholar] [CrossRef]
- Gražulis, S.; Chateigner, D.; Downs, R.T.; Yokochi, A.F.T.; Quirós, M.; Lutterotti, L.; Manakova, E.; Butkus, J.; Moeck, P.; Bail, A.L. Crystallography Open Database—An open-access collection of crystal structures. J. Appl. Crystallogr. 2009, 42, 726–729. [Google Scholar] [CrossRef]
- Taghvaei, A.H.; Stoica, M.; Vaughan, G.; Ghaffari, M.; Maleksaeedi, S.; Janghorban, K. Microstructural characterization and amorphous phase formation in Co40Fe22Ta8B30 powders produced by mechanical alloying. J. Alloy. Compd. 2012, 512, 85–93. [Google Scholar] [CrossRef]
- Msetra, Z.; Khitouni, N.; Suñol, J.J.; Khitouni, M.; Chemingui, M. Characterization and thermal analysis of new amorphous Co60Fe18Ta8B14alloy produced by mechanical alloying. Mater. Lett. 2021, 292, 129532. [Google Scholar] [CrossRef]
- Williamson, G.K.; Hall, W.H. X-ray line broadening from filed aluminium and wolfram. Acta Metall. 1953, 1, 22–31. [Google Scholar] [CrossRef]
- Shan, L.; Wang, X.; Wang, Y. Extension of Solid Solubility and Structural Evolution in Nano-Structured Cu-Cr Solid Solution Induced by High-Energy Milling. Materials 2020, 13, 5532. [Google Scholar] [CrossRef]
- Toby, B.H.; Egami, T. Accuracy of pair distribution function analysis applied to crystalline and non-crystalline materials. Acta Crystallogr. Sect. Found. Crystallogr. 1992, 48, 336–346. [Google Scholar] [CrossRef]
- Takeuchi, A.; Inoue, A. Classification of Bulk Metallic Glasses by Atomic Size Difference, Heat of Mixing and Period of Constituent Elements and Its Application to Characterization of the Main Alloying Element. Mater. Trans. 2005, 46, 2817–2829. [Google Scholar] [CrossRef]
- Kaban, I.; Jóvári, P.; Stoica, M.; Eckert, J.; Hoyer, W.; Beuneu, B. Topological and chemical ordering in Co43Fe20Ta5.5B31.5metallic glass. Phys. Rev. B 2009, 79, 212201. [Google Scholar] [CrossRef]
- Koningsberger, D.C.; Prins, R. X-ray Absorption; Chemical Analysis: A Series of Monographs on Analytical Chemistry and Its Applications; John Wiley & Sons: Hoboken, NJ, USA, 1988. [Google Scholar]
- Kronmüller, H.; Gröger, B. Domains, domain walls and the coercive field of amorphous ferromagnets. J. Phys. 1981, 42, 1285–1292. [Google Scholar] [CrossRef]
- Leonowicz, M.; Manaf, A.; Davies, H.A. Pinning controlled coercivity in rapidly solidified, low boron Fe-Nd-B-Nb alloys. Mater. Lett. 1992, 14, 277–280. [Google Scholar] [CrossRef]
- Gerling, R. Evidence for Different Ferromagnetic Phases in Amorphous Fe40Ni40B20 Detected by EPR. Z. Naturforschung A 1983, 38, 20–26. [Google Scholar] [CrossRef]





| CoFeTaB | CoFeWB | ||||||
|---|---|---|---|---|---|---|---|
| Atomic Pair | [Å] | [–] | kJ/mol | Atomic Pair | [Å] | [–] | kJ/mol |
| Co–Co | 2.506 | 0.269 | – | Co–Co | 2.506 | 0.268 | – |
| Co–Fe | 2.494 | 0.240 | −1 | Co–Fe | 2.494 | 0.239 | −1 |
| Co–Ta | 2.683 | 0.186 | −24 | Co–W | 2.623 | 0.187 | −1 |
| Co–B | 2.083 | 0.073 | −24 | Co–B | 2.083 | 0.073 | −24 |
| Fe–Fe | 2.482 | 0.054 | – | Fe–Fe | 2.482 | 0.054 | – |
| Fe–Ta | 2.671 | 0.083 | −15 | Fe–W | 2.611 | 0.084 | 0 |
| Fe–B | 2.071 | 0.033 | −26 | Fe–B | 2.071 | 0.032 | −26 |
| Ta–Ta | 2.860 | 0.032 | – | W–W | 2.740 | 0.033 | – |
| Ta–B | 2.260 | 0.025 | −54 | W–B | 2.200 | 0.025 | −31 |
| B–B | 1.660 | 0.005 | – | B–B | 1.660 | 0.005 | – |
| Sample | [Å] | CN [-] | [g/cm] | Sample | [Å] | CN [-] | [g/cm] |
|---|---|---|---|---|---|---|---|
| Ta-25 | 0.0813 (28) | 15.0(4) | 6.73 (23) | W-25 | 0.0890 (24) | 17.3(3) | 7.39 (20) |
| Ta-50 | 0.0821 (41) | 14.9(6) | 6.80 (34) | W-50 | 0.0884 (59) | 16.7(8) | 7.34 (49) |
| Ta-100 | 0.0786 (19) | 14.2(3) | 6.51 (16) | W-100 | 0.0829 (26) | 15.1(4) | 6.88 (21) |
| Fe K-Edge | Co K-Edge | |||||||
|---|---|---|---|---|---|---|---|---|
| Sample | Pair | [-] | [Å] | [ Å] | Pair | [-] | [Å] | [ Å] |
| Ta-25 | Fe-B | 3.3(8) | 2.06(5) | 116(8) | Co-B | 4.3(9) | 2.11(5) | 129(13) |
| Fe-Co | 11.9(8) | 2.43(1) | Co-Co | 10.9(9) | 2.42(2) | |||
| Ta-50 | Fe-B | 2.4(8) | 2.02(6) | 132(7) | Co-B | 3.4(4) | 2.05(3) | 148(5) |
| Fe-Co | 12.5(8) | 2.44(1) | Co-Co | 11.5(4) | 2.44(1) | |||
| Ta-100 | Fe-B | 4.1(4) | 2.02(2) | 117(5) | Co-B | 3.3(4) | 2.01(2) | 139(4) |
| Fe-Co | 10.1(4) | 2.44(1) | Co-Co | 10.9(4) | 2.44(1) | |||
| W-25 | Fe-B | 5.3(2.8) | 2.06(9) | 76(24) | Co-B | 6.3(2.2) | 2.13(8) | 108(27) |
| Fe-Co | 12.0(2.8) | 2.44(3) | Co-Co | 11.0(2.2) | 2.41(3) | |||
| W-50 | Fe-B | 4.9(1.5) | 2.05(5) | 107(15) | Co-B | 5.1(1.0) | 2.05(4) | 136(11) |
| Fe-Co | 11.8(1.5) | 2.44(2) | Co-Co | 11.5(1.0) | 2.44(2) | |||
| W-100 | Fe-B | 3.9(1.0) | 2.02(5) | 128(11) | Co-B | 3.5(4) | 2.00(2) | 149(4) |
| Fe-Co | 11.2(1.0) | 2.43(1) | Co-Co | 11.6(4) | 2.43(1) | |||
| Ta-Alloy, Ta L-Edge | W-Alloy, W L-Edge | |||||||
|---|---|---|---|---|---|---|---|---|
| Time | Pair | [-] | [Å] | [ Å] | Pair | [-] | [Å] | [ Å] |
| 25 h | Ta-B | 7.2(1.6) | 2.15(4) | 128(19) | W-W | 5.9(7) | 2.74(1) | 43(3) |
| Ta-Co | 8.0(1.6) | 2.52(1) | W-W | 8.1(7) | 3.16(1) | |||
| 50 h | Ta-B | 6.9(1.5) | 2.15(4) | 117(17) | W-W | 5.6(1.1) | 2.74(1) | 60(6) |
| Ta-Co | 8.0(1.5) | 2.55(1) | W-W | 8.4(1.1) | 3.16(1) | |||
| 100 h | Ta-B | 5.5(1.4) | 2.19(4) | 112(15) | W-W | 3.4(2.8) | 2.72(2) | 102(16) |
| Ta-Co | 8.7(1.4) | 2.57(1) | W-W | 10.6(2.8) | 3.14(2) | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Girman, V.; Lisnichuk, M.; Yudina, D.; Matvija, M.; Sovák, P.; Bednarčík, J. Structural Evolution in Wet Mechanically Alloyed Co-Fe-(Ta,W)-B Alloys. Metals 2021, 11, 800. https://doi.org/10.3390/met11050800
Girman V, Lisnichuk M, Yudina D, Matvija M, Sovák P, Bednarčík J. Structural Evolution in Wet Mechanically Alloyed Co-Fe-(Ta,W)-B Alloys. Metals. 2021; 11(5):800. https://doi.org/10.3390/met11050800
Chicago/Turabian StyleGirman, Vladimír, Maksym Lisnichuk, Daria Yudina, Miloš Matvija, Pavol Sovák, and Jozef Bednarčík. 2021. "Structural Evolution in Wet Mechanically Alloyed Co-Fe-(Ta,W)-B Alloys" Metals 11, no. 5: 800. https://doi.org/10.3390/met11050800
APA StyleGirman, V., Lisnichuk, M., Yudina, D., Matvija, M., Sovák, P., & Bednarčík, J. (2021). Structural Evolution in Wet Mechanically Alloyed Co-Fe-(Ta,W)-B Alloys. Metals, 11(5), 800. https://doi.org/10.3390/met11050800






