Corrosion of N10276 in a H2S, HCl, and CO2 Containing Atmosphere at 480 °C and 680 °C
Abstract
:1. Introduction
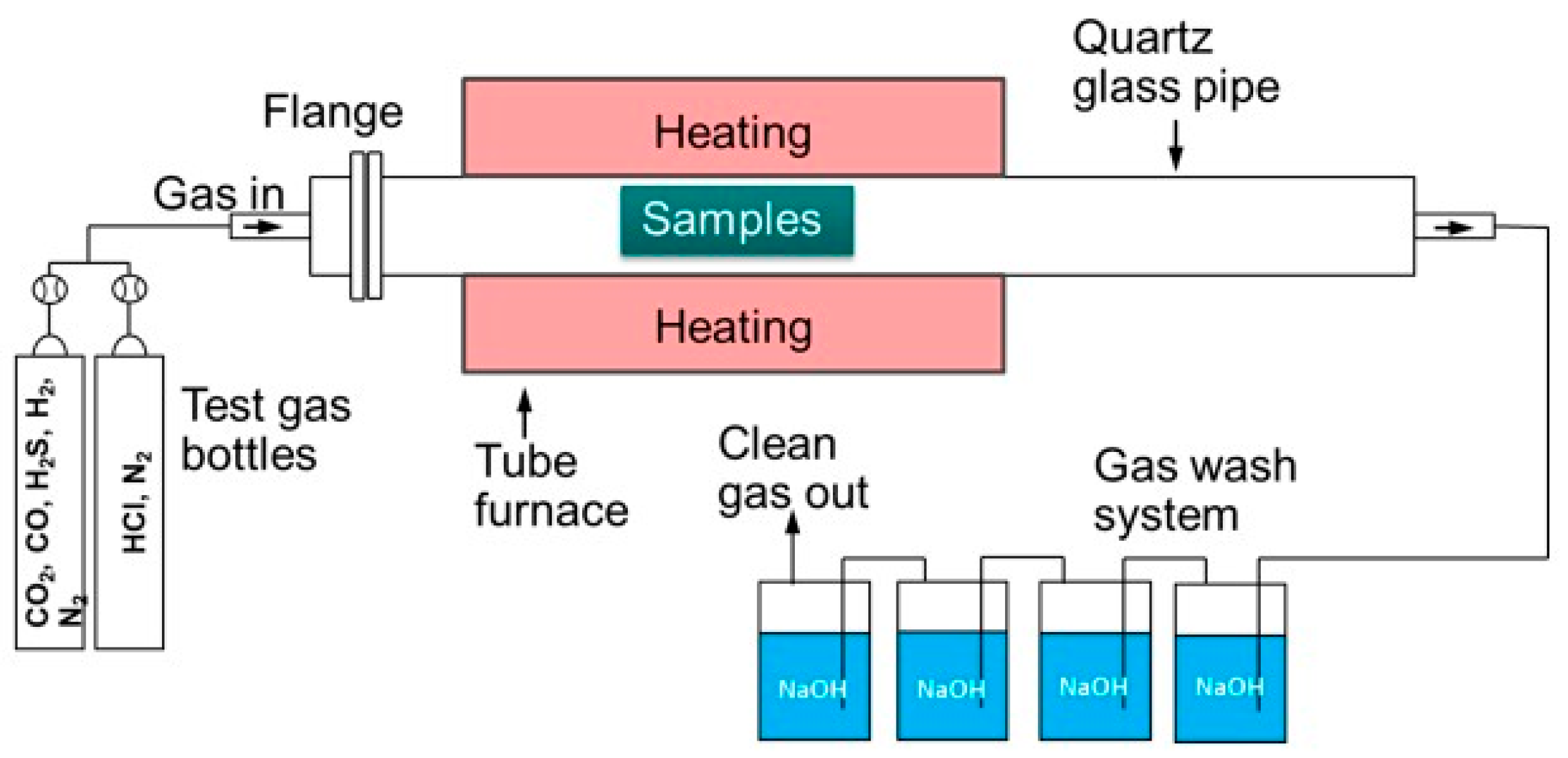
2. Materials and Methods
3. Results
3.1. Evaluation of Corrosion Rate
3.2. Phases Identified in the Corrosion Layer
4. Discussion
4.1. Derivation of the Corrosion Mechanism
Corrosion Reactions
4.2. Suggested Corrosion Mechanisms
4.2.1. Corrosion Mechanism of N10276 at 480 °C
- HCl penetrates the initial oxide layer of the metal and metal chlorides are formed. The formation of metal chlorides is primarily based on the enthalpy of formation. The concentration of the elements is also important, because if a base metal has been removed by corrosion, a more noble metal can subsequently be attacked. In the present case, the last metal to react would be Mo.
- If the vapor pressures of the formed chlorides are high enough, the MeCln will diffuse outward of the corrosion layer. With decreasing vapor pressure, a bigger amount of MeCln will remain inside the layer. Thus, iron and nickel chlorides will diffuse faster since vapor pressures of those species are higher than for molybdenum and chromium chlorides (FeCl2 > NiCl2 > MoCl4 > CrCl2).
- As soon as the MeCln reach the surface of the corrosion layer, they can react with H2S of the gas phase and form the corresponding sulfides.
- CO2 reacts with H2 to form H2O, which further reacts with the remaining MeCln in the corrosion layer to form oxides. In the present case the H2O will mainly react with CrCl2 to form Cr2O3 since it has the lowest vapor pressure and therefore most of the chromium chloride will remain in the corrosion layer.
- With progressive corrosion, the porosity increases due to evaporation of metal chlorides. Consequently, H2S has direct access to the base metal, where it can react with the main alloying elements to the corresponding sulfides.
4.2.2. Corrosion Mechanism of N10276 at 680 °C
- HCl penetrates the initial oxide layer and metal chlorides are formed. The formation of FeCl2 and CrCl2 is favored, but the formation of small amounts of nickel and molybdenum chlorides is also likely.
- Depending on the vapor pressure the formed metal chlorides can diffuse outward (FeCl2 > CrCl2 > NiCl2 > MoCl4).
- On the surface, the metal chlorides react with the H2S, whereby Cr2S3 and nickel sulfides are preferentially formed. As shown in Figure 5, it is noticeable that nickel sulfide and Cr2S3 crystallites are clearly separated from each other. After an initial nucleation, the two phases grow separately. Small amounts of Mo were measured evenly in the two sulfides formed. Due to the very high vapor pressure of FeCl2 and the fast evaporation of this compounds no further reaction with the gas phase takes place. Thus, no iron sulfides were detected.
- CO2 reacts with H2 to H2O, which reacts with the remaining metal chlorides inside the corrosion layer to form oxides.
- With progressive corrosion, the porosity increases due to evaporation of metal chlorides. Consequently, H2S has direct access to the corroded zone of the base metal and can react with metallic Ni and Mo to the corresponding sulfides.
- Main driving force for the mass loss at 680 °C is the faster evaporation of the metal chlorides due to higher vapor pressures compared to 480 °C. This could also be verified by the increasing amount of detected FeCl2 at the colder parts of the test equipment at higher temperatures.
4.3. Comparison of N10276 with Previously Investigated Steels
5. Conclusions
- In general, the corrosion rates of all materials increased with rising temperature.
- ○
- At 480 °C, N10276 showed a better corrosion resistance compared to previous tested materials without molybdenum.
- ○
- At 680 °C, N10276 showed the worst performance compared to the materials without molybdenum.
- During corrosion of N10276 in a H2S, HCl, and CO2 containing environment, HCl attacks besides the main elements Fe, Cr, and Ni also Mo to form the corresponding metal chlorides.
- Corrosion products that formed on N10276, where different to those formed on previously tested Fe-Cr-Ni alloys.
- ○
- At 480 °C, all materials formed a layer of nickel sulfide containing some iron, which formed above a layer enriched in chromium. Contrary to the other materials, N10276 also showed an accumulation of molybdenum in the corrosion zone.
- ○
- At 680 °C, N10276 formed separated nickel sulfides containing molybdenum and chromium sulfides compared to previous tested Fe-Cr-Ni alloys, which formed a chromium sulfide layer below a chromium oxide layer but no nickel sulfides. The nickel sulfide and chromium sulfide crystals that form on N10276 do not form a tight diffusion barrier and further corrosion can take place. These circumstances lead to the high rate of corrosion of N10276.
- At high operation temperatures alloys with high molybdenum content are not necessarily beneficial for the corrosion resistance of materials in complex gases.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Doublet, S.; Schütz, M.; Latreche, H. Development of corrosion assessment diagrams for high temperature chlorine corrosion—Part I. Oxid. Met. 2009, 72, 1–30. [Google Scholar]
- Schmid, A.; Mori, G.; Haubner, R.; Weil, M.; Hönig, S. Behaviour of S31400 and S32205 steels in HCl- and H2S-containing gas atmospheres under a low oxygen partial pressure between 480 and 680 °C. Mater. Corros. 2018, 69, 1–10. [Google Scholar] [CrossRef]
- Schmid, A.; Mori, G.; Strobl, S.; Haubner, R.; Hönig, S. Corrosion of various Fe and Ni based alloys in HCl, H2S containing environments, with low oxygen partial pressure, at 680 °C. In Proceedings of the EUROCORR 2017, Prague, Czech Republic, 3–7 September 2017. [Google Scholar]
- Bramhoff, D.; Grabke, H.J.; Schmidt, H.P. Influence of HCl and N2 on high temperature corrosion in atmospheres with low oxygen pressures. Werkst. Korros. 1989, 40, 642–650. [Google Scholar] [CrossRef]
- Asteman, H.; Spiegel, M. Investigation of the HCl (g) attack on pre-oxidized pure Fe, Cr, Ni and commercial 304 steel at 400 °C. Corr. Sci. 2007, 49, 3626–3637. [Google Scholar] [CrossRef]
- Bramhoff, D.; Grabke, H.J.; Schmidt, H.P. The Role of Active Elements in the Oxidation Behaviour of High Temperature Metals and Alloys; Elsevier: Brussels, Belgium; Luxembourgh, 1989; pp. 335–349. [Google Scholar]
- Bakker, W.T. The effect of chlorine on mixed oxidant corrosion of stainless steel. Mater. High Temp. 1997, 14, 197–206. [Google Scholar] [CrossRef]
- Przybylski, K.; Grzesik, Z. Sulfidation of metallic materials. In Developments in High-Temperature Corrosion and Protection of Materials; Woodhead Publishing Limited: Camebridge, UK, 2008; pp. 599–638. [Google Scholar]
- Schütze, M. Fundamentals of High Temperature Corrosion. Mater. Sci. Technol. 2013, 67–130. [Google Scholar] [CrossRef]
- Mrowec, S. Similarities and Differences in Defect Dependent Properties of Transition Metal Sulphides and Oxides. Inorg. Chem. 1989, 9, 55–99. [Google Scholar]
- Rau, H. Estimation of the Homogeneity Range of MoS2. J. Phys. Chem. Solids 1980, 41, 765–767. [Google Scholar] [CrossRef]
- Bakker, W.T.; Perkins, R.A. Beyond mixed oxidant corrosion-corrosion phenomena in gasifiers. Presented at Ninth Annual EPRI Conference on Gasification Power Plants 1990, Palo Alto, CA, USA, 16–19 October 1990. [Google Scholar]
- Bakker, W.T.; Perkins, R.A. Materials for Coal Gasification; ASM International: Cincinnati, OH, USA, 1988; pp. 85–96. [Google Scholar]
- Pan, T.J. Corrosion behaviour of three iron-based alloy in reducing atmospheres containing HCl and H2S at 600 °C. Corr. Sci. 2007, 49, 1362–1377. [Google Scholar] [CrossRef]
- Nimmervoll, M.; Mori, G.; Schmid, A.; Haubner, R.; Hönig, S. Surface sulphide formation on high-temperature corrosion resistant alloys in a H2S-HCl-CO2 mixed atmosphere. Corr. Sci. 2021, 181, 109241. [Google Scholar] [CrossRef]
- Schmid, A.; Mori, G.; Bucher, E.; Haubner, R. Model about the course of corrosion reactions of austenitic steels in H2S-, HCl- and CO2-containing atmospheres at 680 °C. Oxid. Met. 2019, 91, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Schmid, A.; Mori, G.; Haubner, R.; Weil, M.; Strobl, S.; Hönig, S. Comparison of the high-temperature chloride-induced corrosion between duplex steel and Ni based alloy in presence of H2S. Corr. Sci. 2018, 139, 76–82. [Google Scholar] [CrossRef]
- Schmid, A.; Mori, G.; Hönig, S.; Strobl, S.; Haubner, R. High Temperature Corrosion of Steel X15CrNiSi25-21 (1.4841) in a Mixed Gas Atmosphere. Prakt. Metallogr. Pract. Metallogr. 2019, 56, 457–468. [Google Scholar] [CrossRef]
- Schmid, A. Chlorine and Sulphur Induced High Temperature Corrosion in a Thermal Cracking Reactor for Anthropogenic Resources. Ph.D. Thesis, University of Leoben, Leoben, Austria, 2018. [Google Scholar]
- Kunze, E. Korrosion und Korrosionsschutz; Wiley–VCH: Berlin, Gremany, 2001. [Google Scholar]
- Young, D.J. High Temperature Oxidation and Corrosion of Metals; Elsevier Ltd.: Amsterdam, The Netherlands, 2016. [Google Scholar]
- Stott, F.H.; Prescott, R.; Elliott, P. Corrosion resistance of high temperature alloys in gas containing hydrogen chloride. Mater. Sci. Technol. 1990, 6, 364. [Google Scholar] [CrossRef]
- Abels, J.M.; Strehblow, H.H. A surface analytical approach to the high temperature chlorination behaviour of inconel 600 at 700 °C. Corr. Sci. 1997, 39, 115–132. [Google Scholar] [CrossRef]
- Jehn, H.; Kurtz, W.; Schneider, D.; Trobisch, U.; Wagner, J. Mo Molybdenum, Gmelin Handbook of Inorganic and Organometallic Chemistry; Springer: Berlin/Heidelberg, Germany, 1989. [Google Scholar]








| Species | Partial Pressure at 480 °C | Partial Pressure at 680 °C |
|---|---|---|
| S2 | 1.6 × 10−09 | 1.3 × 10−09 |
| Cl2 | 4.7 × 10−14 | 7.5 × 10−13 |
| O2 | 3.6 × 10−29 | 9.1 × 10−23 |
| Material | Fe | Ni | Cr | C | Mn | Si | Mo | W/Co |
| N10276 | 5 | 59 | 15 | 0.01 | 1 | - | 16 | 4/2.5 |
| S31400 | 52 | 20 | 25 | 0.07 | 1.5 | 1.5 | - | - |
| N06600 | <10 | 72.5 | 16.5 | <0.05 | 0.8 | 0.4 | 1 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nimmervoll, M.; Mori, G.; Bucher, E.; Schmid, A.; Haubner, R. Corrosion of N10276 in a H2S, HCl, and CO2 Containing Atmosphere at 480 °C and 680 °C. Metals 2021, 11, 1817. https://doi.org/10.3390/met11111817
Nimmervoll M, Mori G, Bucher E, Schmid A, Haubner R. Corrosion of N10276 in a H2S, HCl, and CO2 Containing Atmosphere at 480 °C and 680 °C. Metals. 2021; 11(11):1817. https://doi.org/10.3390/met11111817
Chicago/Turabian StyleNimmervoll, Manuela, Gregor Mori, Edith Bucher, Alexander Schmid, and Roland Haubner. 2021. "Corrosion of N10276 in a H2S, HCl, and CO2 Containing Atmosphere at 480 °C and 680 °C" Metals 11, no. 11: 1817. https://doi.org/10.3390/met11111817
APA StyleNimmervoll, M., Mori, G., Bucher, E., Schmid, A., & Haubner, R. (2021). Corrosion of N10276 in a H2S, HCl, and CO2 Containing Atmosphere at 480 °C and 680 °C. Metals, 11(11), 1817. https://doi.org/10.3390/met11111817






