Extraction of Cu(II), Fe(III), Zn(II), and Mn(II) from Aqueous Solutions with Ionic Liquid R4NCy
Abstract
:1. Introduction
2. Materials and Methods
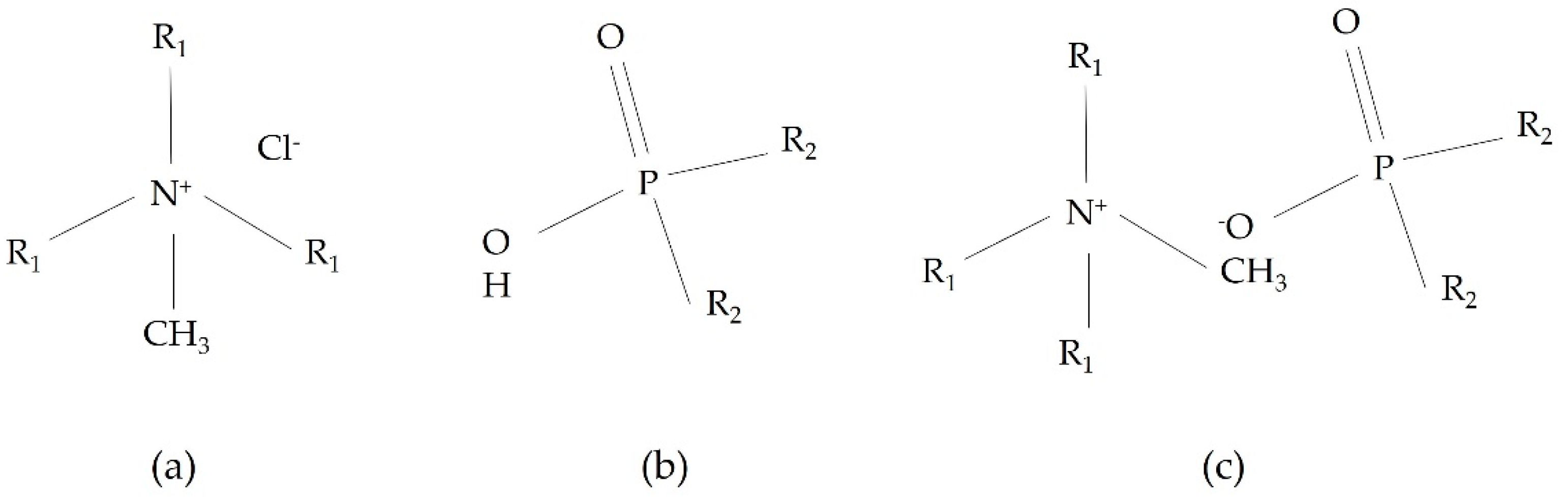
2.1. Reagents and Synthesis of Ionic Liquids
2.2. Liquid/Liquid Extraction
3. Results and Discussion
3.1. Effect of the R4NCy Concentration in Metal Extraction
3.2. Effect of Initial pH in Metal Extraction
3.3. Effect of O/A Ratio in Single Metal Extraction
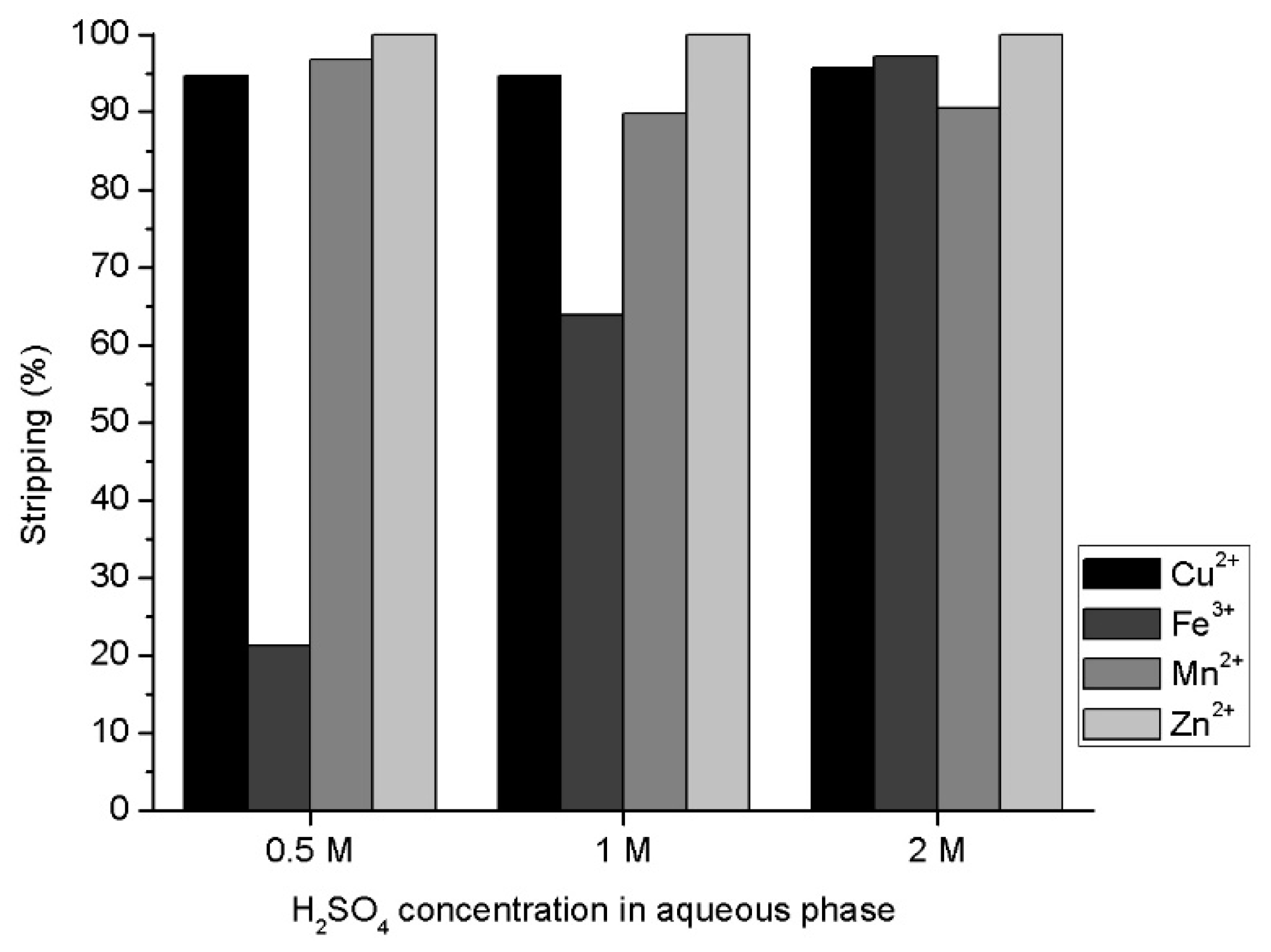
3.4. Effect of Sulfuric Acid in Metal Stripping
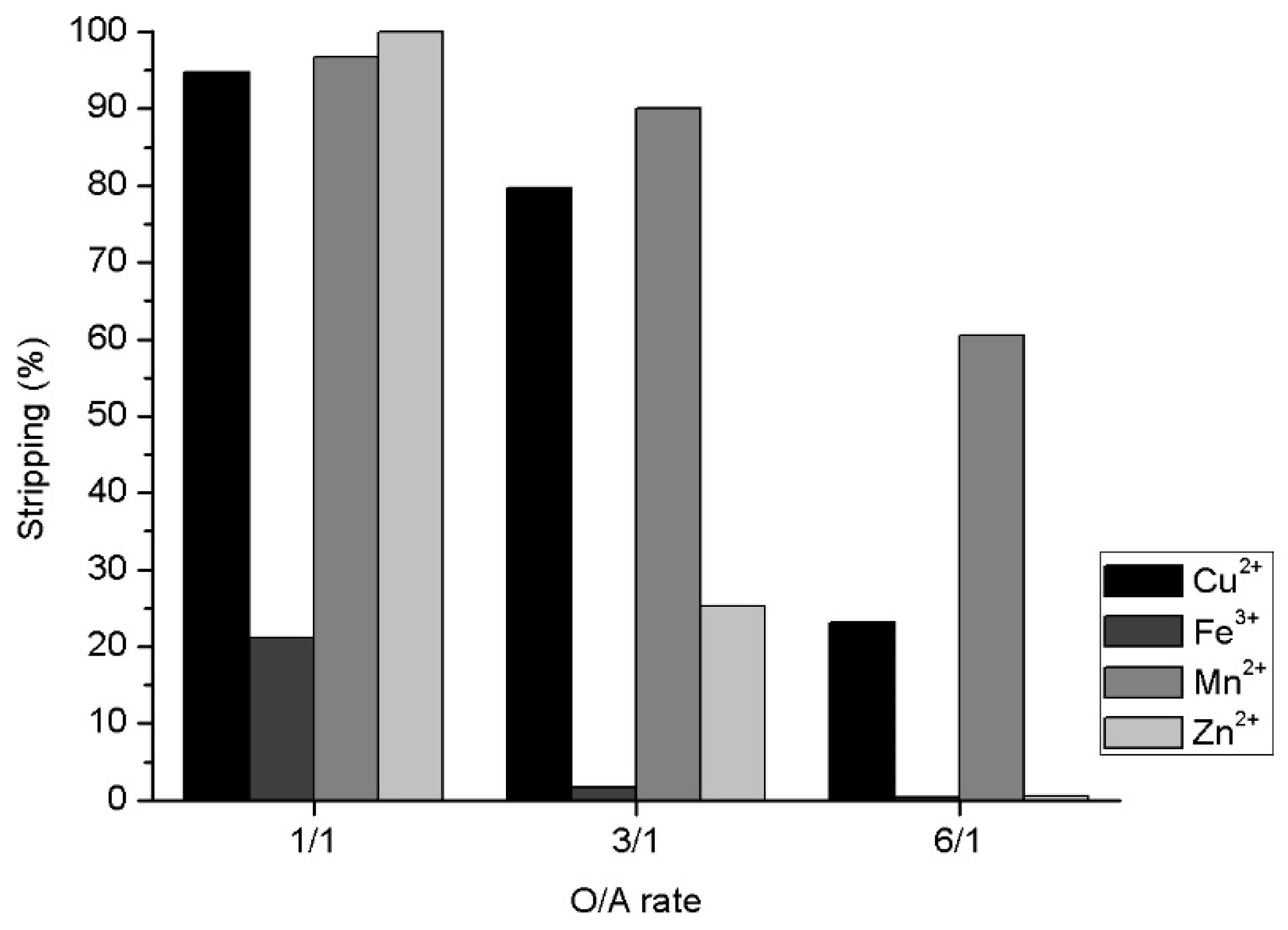
3.5. Effect of O/A in Metal Stripping
4. Conclusions
- (1)
- The extraction tests showed a strongly selective extraction of Fe(III) and Zn(II) over Cu(II) and Mn(II). A practically total extraction of Fe(III) and Zn(II) was obtained for all the conditions under study. In the case of Cu(II) and Mn(II), the extraction efficiency is strongly influenced by the R4NCy concentration, the PLS pH, and the O/A ratio.
- (2)
- The use of 2 M of H2SO4 produces a very efficient stripping of all metals loaded into the ionic liquid. The efficiency does not decline as acid concentration decreases, except for Fe(III). The stripping efficiency of iron loaded into the ionic liquid is close to 20% for 0.5 M of sulfuric acid. This result is very promising because it would allow developing a very selective process concerning Fe(III).
- (3)
- Despite its exploratory nature, this study offers a clear insight into the strong affinity of ionic liquid R4NCy for Fe(III) and Zn(II) over Cu(II) and Mn(II). This fact opens the possibility of using the ionic liquid R4NCy in the pre-treatment of copper-rich solutions, removing the impurities as Fe(III) and Zn(II).
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Cao, X.; Zhang, T.A.; Zhang, W.; Lv, G. Solvent Extraction of Sc(III) by D2EHPA/TBP from the Leaching Solution of Vanadium Slag. Metals 2020, 10, 790. [Google Scholar] [CrossRef]
- Sekisov, A.; Rasskazova, A. Assessment of the Possibility of Hydrometallurgical Processing of Low-Grade Ores in the Oxidation Zone of the Malmyzh Cu-Au Porphyry Deposit. Minerals 2021, 11, 69. [Google Scholar] [CrossRef]
- Mahmoudi, A.; Shakibania, S.; Rezaee, S.; Mokmeli, M. Effect of the Chloride Content of Seawater on the Copper Solvent Extraction Using Acorga M5774 and LIX 984N Extractants. Sep. Purif. Technol. 2020, 251, 117394. [Google Scholar] [CrossRef]
- Rao, M.; Zhang, T.; Li, G.; Zhou, Q.; Luo, J.; Zhang, X.; Zhu, Z.; Peng, Z.; Jiang, T. Solvent Extraction of Ni and Co from the Phosphoric Acid Leaching Solution of Laterite Ore by P204 and P507. Metals 2020, 10, 545. [Google Scholar] [CrossRef] [Green Version]
- Ying, Z.; Ren, X.; Li, J.; Wu, G.; Wei, Q. Recovery of Chromium(VI) in Wastewater Using Solvent Extraction with Amide. Hydrometallurgy 2020, 196, 105440. [Google Scholar] [CrossRef]
- Zhang, W.; Xie, X.; Tong, X.; Du, Y.; Song, Q.; Feng, D. Study on the Effect and Mechanism of Impurity Aluminum on the Solvent Extraction of Rare Earth Elements (Nd, Pr, La) by P204-P350 in Chloride Solution. Minerals 2021, 11, 61. [Google Scholar] [CrossRef]
- Shakibania, S.; Mahmoudi, A.; Mokmeli, M.; Rashchi, F. The Effect of the Chloride Ion on Chemical Degradation of LIX 984N Extractant. Miner. Eng. 2020, 159, 106628. [Google Scholar] [CrossRef]
- Wang, L.Y.; Guo, Q.J.; Lee, M.S. Recent Advances in Metal Extraction Improvement: Mixture Systems Consisting of Ionic Liquid and Molecular Extractant. Sep. Purif. Technol. 2019, 210, 292–303. [Google Scholar] [CrossRef]
- Hong, T.; Liu, M.; Ma, J.; Yang, G.; Li, L.; Mumford, K.A.; Stevens, G.W. Selective Recovery of Rhenium from Industrial Leach Solutions by Synergistic Solvent Extraction. Sep. Purif. Technol. 2020, 236, 116281. [Google Scholar] [CrossRef]
- Hidayah, N.N.; Abidin, S.Z. The Evolution of Mineral Processing in Extraction of Rare Earth Elements Using Liquid-Liquid Extraction: A Review. Miner. Eng. 2018, 121, 146–157. [Google Scholar] [CrossRef]
- Keskin, S.; Kayrak-Talay, D.; Akman, U.; Hortaçsu, Ö. A Review of Ionic Liquids towards Supercritical Fluid Applications. J. Supercrit. Fluids 2007, 43, 150–180. [Google Scholar] [CrossRef]
- Park, J.; Jung, Y.; Kusumah, P.; Lee, J.; Kwon, K.; Lee, C.K. Application of Ionic Liquids in Hydrometallurgy. Int. J. Mol. Sci. 2014, 15, 15320–15343. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Weng, J.; Wang, C.; Li, H.; Wang, Y. Novel Quaternary Ammonium Ionic Liquids and Their Use as Dual Solvent-Catalysts in the Hydrolytic Reaction. Green Chem. 2006, 8, 96. [Google Scholar] [CrossRef]
- Quijada-maldonado, E.; Olea, F.; Sepúlveda, R.; Cabezas, R.; Merlet, G.; Romero, J. Possibilities and Challenges for Ionic Liquids in Hydrometallurgy. Sep. Purif. Technol. 2020, 251, 117289. [Google Scholar] [CrossRef]
- Lee, J.; Yeo, C.-D.; Hu, Z.; Thalangama-Arachchige, V.D.; Kaur, J.; Quitevis, E.L.; Kumar, G.; Koh, Y.P.; Simon, S. Friction and Wear of Pd-Rich Amorphous Alloy (Pd43Cu27Ni10P20) with Ionic Liquid (IL) as Lubricant at High Temperatures. Metals 2019, 9, 1180. [Google Scholar] [CrossRef] [Green Version]
- Alguacil, F.J.; López, F.A. Permeation of AuCl4− Across a Liquid Membrane Impregnated with A324H+Cl− Ionic Liquid. Metals 2020, 10, 363. [Google Scholar] [CrossRef] [Green Version]
- Diabate, P.D.; Dupont, L.; Boudesocque, S.; Mohamadou, A. Novel Task Specific Ionic Liquids to Remove Heavy Metals from Aqueous Effluents. Metals 2018, 8, 412. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez, M.; Ayala, L.; Robles, P.; Sepúlveda, R.; Torres, D.; Carrillo-Pedroza, F.R.; Jeldres, R.I.; Toro, N. Leaching Chalcopyrite with an Imidazolium-Based Ionic Liquid and Bromide. Metals 2020, 10, 183. [Google Scholar] [CrossRef] [Green Version]
- Salar-García, M.J.; Ortiz-Martínez, V.M.; Hernández-Fernández, F.J.; de los Ríos, A.P.; Quesada-Medina, J. Ionic Liquid Technology to Recover Volatile Organic Compounds (VOCs). J. Hazard. Mater. 2017, 321, 484–499. [Google Scholar] [CrossRef]
- Carlesi, C.; Cortes, E.; Dibernardi, G.; Morales, J.; Muñoz, E. Ionic Liquids as Additives for Acid Leaching of Copper from Sulfidic Ores. Hydrometallurgy 2016, 161, 29–33. [Google Scholar] [CrossRef]
- Jin, Y.; Zhang, J.; Song, J.; Zhang, Z.; Fang, S.; Yang, L.; Hirano, S.I. Functionalized Ionic Liquids Based on Quaternary Ammonium Cations with Two Ether Groups as New Electrolytes for Li/LiFePO4 Secondary Battery. J. Power Sources 2014, 254, 137–147. [Google Scholar] [CrossRef]
- Egashira, M.; Okada, S.; Yamaki, J.I.; Yoshimoto, N.; Morita, M. Effect of Small Cation Addition on the Conductivity of Quaternary Ammonium Ionic Liquids. Electrochim. Acta 2005, 50, 3708–3712. [Google Scholar] [CrossRef]
- Matsumoto, M.; Yamaguchi, T.; Tahara, Y. Extraction of Rare Earth Metal Ions with an Undiluted Hydrophobic Pseudoprotic Ionic Liquid. Metals 2020, 10, 502. [Google Scholar] [CrossRef]
- Jing, X.; Wu, Z.; Zhao, D.; Li, S.; Kong, F.; Chu, Y. Environmentally Friendly Extraction and Recovery of Cobalt from Simulated Solution of Spent Ternary Lithium Batteries Using the Novel Ionic Liquids of [C8H17NH2][Cyanex 272]. ACS Sustain. Chem. Eng. 2021, 9, 2475–2485. [Google Scholar] [CrossRef]
- Łukomska, A.; Wiśniewska, A.; Dąbrowski, Z.; Domańska, U. Liquid-Liquid Extraction of Cobalt(II) and Zinc(II) from Aqueous Solutions Using Novel Ionic Liquids as an Extractants. J. Mol. Liq. 2020, 307, 112955. [Google Scholar] [CrossRef]
- Tran, T.T.; Liu, Y.; Lee, M.S. Recovery of Pure Molybdenum and Vanadium Compounds from Spent Petroleum Catalysts by Treatment with Ionic Liquid Solution in the Presence of Oxidizing Agent. Sep. Purif. Technol. 2021, 255, 117734. [Google Scholar] [CrossRef]
- Binnemans, K. Lanthanides and Actinides in Ionic Liquids. Compr. Inorg. Chem. II Second. Ed. Elem. Appl. 2013, 2, 641–673. [Google Scholar] [CrossRef]
- Maria, L.; Cruz, A.; Carretas, J.M.; Monteiro, B.; Galinha, C.; Gomes, S.S.; Araújo, M.F.; Paiva, I.; Marçalo, J.; Leal, J.P. Improving the Selective Extraction of Lanthanides by Using Functionalised Ionic Liquids. Sep. Purif. Technol. 2020, 237, 116354. [Google Scholar] [CrossRef]
- Devi, N.; Sukla, L.B. Studies on Liquid-Liquid Extraction of Yttrium and Separation from Other Rare Earth Elements Using Bifunctional Ionic Liquids. Miner. Process. Extr. Metall. Rev. 2019, 40, 46–55. [Google Scholar] [CrossRef]
- Rzelewska-Piekut, M.; Regel-Rosocka, M. Separation of Pt(IV), Pd(II), Ru(III) and Rh(III) from Model Chloride Solutions by Liquid-Liquid Extraction with Phosphonium Ionic Liquids. Sep. Purif. Technol. 2019, 212, 791–801. [Google Scholar] [CrossRef]
- Sepúlveda, R.; Romero, J.; Sánchez, J. Copper Removal from Aqueous Solutions by Means of Ionic Liquids Containing a B-Diketone and the Recovery of Metal Complexes by Supercritical Fluid Extraction. J. Chem. Technol. Biotechnol. 2014, 89, 899–908. [Google Scholar] [CrossRef]
- Kim, B.K.; Lee, E.J.; Kang, Y.; Lee, J.J. Application of Ionic Liquids for Metal Dissolution and Extraction. J. Ind. Eng. Chem. 2018, 61, 388–397. [Google Scholar] [CrossRef]
- Valdés Vergara, M.A.; Lijanova, I.V.; Likhanova, N.V.; Olivares Xometl, O.; Jaramillo Vigueras, D.; Morales Ramirez, A.J. Recycling and Recovery of Ammonium-Based Ionic Liquids after Extraction of Metal Cations from Aqueous Solutions. Sep. Purif. Technol. 2014, 155, 110–117. [Google Scholar] [CrossRef]
- Nguyen, V.N.H.; Le, M.N.; Lee, M.S. Comparison of Extraction Ability between a Mixture of Alamine 336/Aliquat 336 and D2EHPA and Ionic Liquid ALi-D2 from Weak Hydrochloric Acid Solution. Metals 2020, 10, 1678. [Google Scholar] [CrossRef]
- Castillo, J.; Coll, M.T.; Fortuny, A.; Navarro Donoso, P.; Sepúlveda, R.; Sastre, A.M. Cu(II) Extraction Using Quaternary Ammonium and Quaternary Phosphonium Based Ionic Liquid. Hydrometallurgy 2014, 141, 89–96. [Google Scholar] [CrossRef]
- Fortuny, A.; Coll, M.T.; Sastre, A.M. Use of Methyltrioctyl/Decylammonium Bis 2,4,4-(Trimethylpentyl)Phosphinate Ionic Liquid (ALiCY IL) on the Boron Extraction in Chloride Media. Sep. Purif. Technol. 2012, 97, 137–141. [Google Scholar] [CrossRef]
- Swain, S.S.; Nayak, B.; Devi, N.; Das, S.; Swain, N. Liquid-Liquid Extraction of Cadmium(II) from Sulfate Medium Using Phosphonium and Ammonium Based Ionic Liquids Diluted in Kerosene. Hydrometallurgy 2016, 162, 63–70. [Google Scholar] [CrossRef]
- Deferm, C.; Onghena, B.; Nguyen, V.T.; Banerjee, D.; Fransaer, J.; Binnemans, K. Non-Aqueous Solvent Extraction of Indium from an Ethylene Glycol Feed Solution by the Ionic Liquid Cyphos IL 101: Speciation Study and Continuous Counter-Current Process in Mixer–Settlers. RSC Adv. 2020, 10, 24595–24612. [Google Scholar] [CrossRef]
- Padhan, E.; Sarangi, K. Recovery of Nd and Pr from NdFeB Magnet Leachates with Bi-Functional Ionic Liquids Based on Aliquat 336 and Cyanex 272. Hydrometallurgy 2017, 167, 134–140. [Google Scholar] [CrossRef]
- Rybka, P.; Regel-Rosocka, M. Nickel(II) and Cobalt(II) Extraction from Chloride Solutions with Quaternary Phosphonium Salts. Sep. Sci. Technol. 2012, 47, 1296–1302. [Google Scholar] [CrossRef]
- Quijada-Maldonado, E.; Romero, J.; Osorio, I. Selective Removal of Iron(III) from Synthetic Copper(II) Pregnant Leach Solutions Using [Bmim][Tf2N] as Diluent and TFA as Extracting Agent. Hydrometallurgy 2016, 159, 54–59. [Google Scholar] [CrossRef]
- Devi, N. Solvent Extraction and Separation of Copper from Base Metals Using Bifunctional Ionic Liquid from Sulfate Medium. Trans. Nonferrous Met. Soc. China 2016, 26, 874–881. [Google Scholar] [CrossRef]
- Regel-Rosocka, M.; Nowak, Ł.; Wiśniewski, M. Removal of Zinc(II) and Iron Ions from Chloride Solutions with Phosphonium Ionic Liquids. Sep. Purif. Technol. 2012, 97, 158–163. [Google Scholar] [CrossRef]
- Baczyńska, M.; Regel-Rosocka, M.; Coll, M.T.; Fortuny, A.; Sastre, A.M.; Wiśniewski, M. Transport of Zn(II), Fe(II), Fe(III) across Polymer Inclusion Membranes (PIM) and Flat Sheet Supported Liquid Membranes (SLM) Containing Phosphonium Ionic Liquids as Metal Ion Carriers. Sep. Sci. Technol. 2016, 51, 2639–2648. [Google Scholar] [CrossRef]
- Ola, P.D.; Kurobe, Y.; Matsumoto, M. Solvent Extraction and Stripping of Fe and Mn from Aqueous Solution Using Ionic Liquids as Extractants. Chem. Eng. Trans. 2017, 57, 1135–1140. [Google Scholar] [CrossRef]
- Nguyen, V.N.H.; Lee, M.S. Separation of Co(II), Ni(II), Mn(II) and Li(I) from Synthetic Sulfuric Acid Leaching Solution of Spent Lithium Ion Batteries by Solvent Extraction. J. Chem. Technol. Biotechnol. 2021, 96, 1205–1217. [Google Scholar] [CrossRef]
- Zhu, Z.; Yoko, P.; Cheng, C.Y. Recovery of Cobalt and Manganese from Nickel Laterite Leach Solutions Containing Chloride by Solvent Extraction Using Cyphos IL 101. Hydrometallurgy 2017, 169, 213–218. [Google Scholar] [CrossRef]
- Sobekova Foltova, S.; vander Hoogerstraete, T.; Banerjee, D.; Binnemans, K. Samarium/Cobalt Separation by Solvent Extraction with Undiluted Quaternary Ammonium Ionic Liquids. Sep. Purif. Technol. 2019, 210, 209–218. [Google Scholar] [CrossRef]
- Tran, T.T.; Iqbal, M.; Lee, M.S. Comparison of the Extraction and Stripping Behavior of Iron (III) from Weak Acidic Solution between Ionic Liquids and Commercial Extractants. J. Korean Inst. Met. Mater. 2019, 57, 787–794. [Google Scholar] [CrossRef]

| Metal | Concentration (mg/L) | |
|---|---|---|
| Literature Report [41] | This Study | |
| Cu(II) | 3360 | 3360 |
| Fe(III) | 1570 | 1570 |
| Mn(II) | >1000 | 1000 |
| Zn(II) | 107 | 110 |
| Metal | IL Concentration [M] | E(%) | D |
|---|---|---|---|
| Cu(II) | 0.1 | 44.14 | 0.79 |
| 0.27 | 84.73 | 5.54 | |
| 0.54 | 97.64 | 41.4 | |
| Fe(III) | 0.1 | 99.77 | 435 |
| 0.27 | 99.82 | 559 | |
| 0.54 | 99.73 | 372 | |
| Mn(II) | 0.1 | 30.63 | 0.44 |
| 0.27 | 71.58 | 2.51 | |
| 0.54 | 93.22 | 13.7 | |
| Zn(II) | 0.1 | 95.55 | 21.4 |
| 0.27 | 96.09 | 24.6 | |
| 0.54 | 92.82 | 12.9 |
| Metal | O/A Rate | E(%) | D |
|---|---|---|---|
| Cu(II) | 1:1 | 84.73 | 5.54 |
| 1:3 | 28.60 | 0.40 | |
| 1:6 | 9.08 | 0.09 | |
| Fe(III) | 1:1 | 99.82 | 559 |
| 1:3 | 99.82 | 540 | |
| 1:6 | 91.40 | 10.6 | |
| Mn(II) | 1:1 | 71.58 | 2.51 |
| 1:3 | 22.70 | 0.29 | |
| 1:6 | 11.90 | 0.13 | |
| Zn(II) | 1:1 | 96.09 | 24.58 |
| 1:3 | 96.73 | 29.55 | |
| 1:6 | 82.73 | 4.78 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Castillo, J.; Toro, N.; Hernández, P.; Navarro, P.; Vargas, C.; Gálvez, E.; Sepúlveda, R. Extraction of Cu(II), Fe(III), Zn(II), and Mn(II) from Aqueous Solutions with Ionic Liquid R4NCy. Metals 2021, 11, 1585. https://doi.org/10.3390/met11101585
Castillo J, Toro N, Hernández P, Navarro P, Vargas C, Gálvez E, Sepúlveda R. Extraction of Cu(II), Fe(III), Zn(II), and Mn(II) from Aqueous Solutions with Ionic Liquid R4NCy. Metals. 2021; 11(10):1585. https://doi.org/10.3390/met11101585
Chicago/Turabian StyleCastillo, Jonathan, Norman Toro, Pía Hernández, Patricio Navarro, Cristian Vargas, Edelmira Gálvez, and Rossana Sepúlveda. 2021. "Extraction of Cu(II), Fe(III), Zn(II), and Mn(II) from Aqueous Solutions with Ionic Liquid R4NCy" Metals 11, no. 10: 1585. https://doi.org/10.3390/met11101585
APA StyleCastillo, J., Toro, N., Hernández, P., Navarro, P., Vargas, C., Gálvez, E., & Sepúlveda, R. (2021). Extraction of Cu(II), Fe(III), Zn(II), and Mn(II) from Aqueous Solutions with Ionic Liquid R4NCy. Metals, 11(10), 1585. https://doi.org/10.3390/met11101585











