Dispersive Solid–Liquid Microextraction Based on the Poly(HDDA)/Graphene Sorbent Followed by ICP-MS for the Determination of Rare Earth Elements in Coal Fly Ash Leachate
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Leaching of CFA Samples
2.3. Synthesis of Poly(HDDA)/Graphene
2.4. Factorial Design of DSPME
2.5. ICP-MS Measurements
3. Results and Discussion
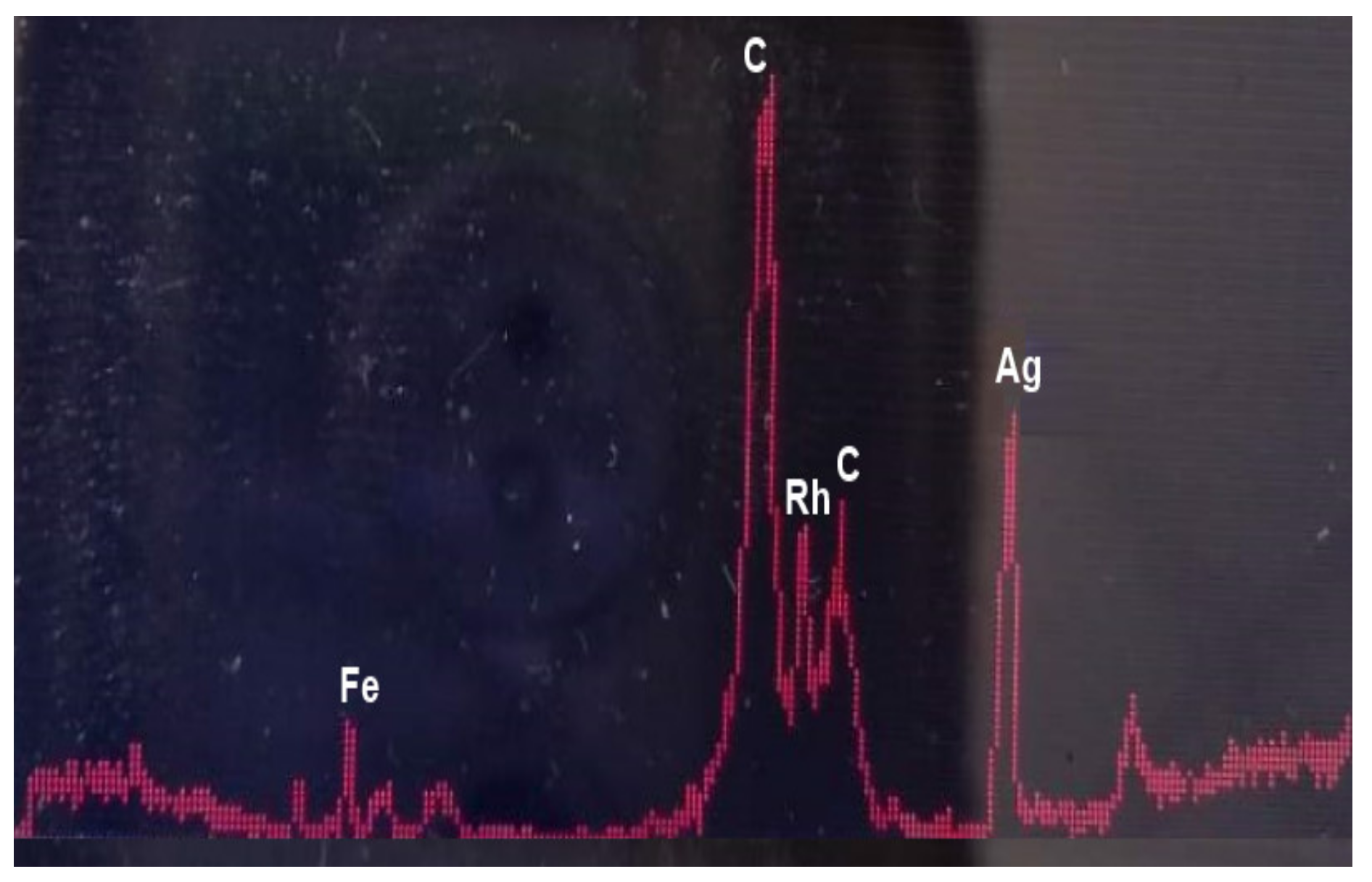
3.1. Characterization of Poly(HDDA)/Graphene Particles
3.2. Factorial Optimization of DSPME
3.2.1. Plackett–Burman Screening
3.2.2. Central Composite Design Optimization
3.3. Analytical Characteristics
3.4. Analytical Applications
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Stopic, S.; Friedrich, B. Advances in Understanding of the Application of Unit Operations in Metallurgy of Rare Earth Elements. Metals 2021, 11, 978. [Google Scholar] [CrossRef]
- Balaram, V. Rare Earth Elements: A Review of Applications, Occurrence, Exploration, Analysis, Recycling, and Environmental Impact. Geosci. Front. 2019, 10, 1285–1303. [Google Scholar] [CrossRef]
- Jyothi, R.K.; Thenepalli, T.; Ahn, J.W.; Parhi, P.K.; Chung, K.W.; Lee, J.-Y. Review of Rare Earth Elements Recovery from Secondary Resources for Clean Energy Technologies: Grand Opportunities to Create Wealth from Waste. J. Clean. Prod. 2020, 267, 122048. [Google Scholar] [CrossRef]
- Palaparthi, J.; Chakrabarti, R.; Banerjee, S.; Guin, R.; Ghosal, S.; Agrahari, S.; Sengupta, D. Economically Viable Rare Earth Element Deposits along Beach Placers of Andhra Pradesh, Eastern Coast of India. Arab. J. Geosci. 2017, 10, 201. [Google Scholar] [CrossRef]
- Vilakazi, A.Q.; Ndlovu, S.; Chipise, L.; Shemi, A. The Recycling of Coal Fly Ash: A Review on Sustainable Developments and Economic Considerations. Sustainability 2022, 14, 1958. [Google Scholar] [CrossRef]
- Fu, B.; Hower, J.C.; Zhang, W.; Luo, G.; Hu, H.; Yao, H. A Review of Rare Earth Elements and Yttrium in Coal Ash: Content, Modes of Occurrences, Combustion Behavior, and Extraction Methods. Prog. Energy Combust. Sci. 2022, 88, 100954. [Google Scholar] [CrossRef]
- Zhang, W.; Noble, A.; Yang, X.; Honaker, R. A Comprehensive Review of Rare Earth Elements Recovery from Coal-Related Materials. Minerals 2020, 10, 451. [Google Scholar] [CrossRef]
- Keller, V.; Stopić, S.; Xakalashe, B.; Ma, Y.; Ndlovu, S.; Mwewa, B.; Simate, G.S.; Friedrich, B. Effectiveness of Fly Ash and Red Mud as Strategies for Sustainable Acid Mine Drainage Management. Minerals 2020, 10, 707. [Google Scholar] [CrossRef]
- Ma, Y.; Stopic, S.; Xakalashe, B.; Ndlovu, S.; Forsberg, K.; Friedrich, B. A Cleaner Approach for Recovering Al and Ti from Coal Fly Ash via Microwave-Assisted Baking, Leaching, and Precipitation. Hydrometallurgy 2021, 206, 105754. [Google Scholar] [CrossRef]
- Wen, Z.; Zhou, C.; Pan, J.; Cao, S.; Hu, T.; Ji, W.; Nie, T. Recovery of Rare-Earth Elements from Coal Fly Ash via Enhanced Leaching. Int. J. Coal Prep. Util. 2020, 284, 124725. [Google Scholar] [CrossRef]
- Tsiridis, V.; Samaras, P.; Kungolos, A.; Sakellaropoulos, G.P. Application of Leaching Tests for Toxicity Evaluation of Coal Fly Ash. Environ. Toxicol. 2006, 21, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Hong, W.; Zhang, B.; Yang, C.; Wang, J.; Gao, J.; Mi, F.; Zhang, H.; Zhao, X.; Li, Q. Study on the Determination of Rare Earth Elements in Coal Ash by ICP-MS. Integr. Ferroelectr. 2019, 198, 116–121. [Google Scholar] [CrossRef]
- Wysocka, I. Determination of Rare Earth Elements Concentrations in Natural Waters—A Review of ICP-MS Measurement Approaches. Talanta 2021, 221, 121636. [Google Scholar] [CrossRef] [PubMed]
- Hann, S.; Boeck, K.; Koellensperger, G. Immunoaffinity Assisted LC-ICP-MS—a Versatile Tool in Biomedical Research. J. Anal. Spectrom. 2010, 25, 18–20. [Google Scholar] [CrossRef]
- Fisher, A.; Kara, D. Determination of Rare Earth Elements in Natural Water Samples—A Review of Sample Separation, Preconcentration and Direct Methodologies. Anal. Chim. Acta 2016, 935, 1–29. [Google Scholar] [CrossRef] [Green Version]
- Manousi, N.; Gomez-Gomez, B.; Madrid, Y.; Deliyanni, E.A.; Zachariadis, G.A. Determination of Rare Earth Elements by Inductively Coupled Plasma-Mass Spectrometry after Dispersive Solid Phase Extraction with Novel Oxidized Graphene Oxide and Optimization with Response Surface Methodology and Central Composite Design. Microchem. J. 2020, 152, 104428. [Google Scholar] [CrossRef]
- Ebihara, M.; Hayano, K.; Shirai, N. Determination of Trace Rare Earth Elements in Rock Samples Including Meteorites by ICP-MS Coupled with Isotope Dilution and Comparison Methods. Anal. Chim. Acta 2020, 1101, 81–89. [Google Scholar] [CrossRef]
- Milicic, L.; Terzic, A.; Pezo, L.; Mijatovic, N.; Brceski, I.; Vukelic, N. Assessment of Efficiency of Rare Earth Elements Recovery from Lignite Coal Combustion Ash via Five-Stage Extraction. Sci. Sinter. 2021, 53, 169–185. [Google Scholar] [CrossRef]
- Rubinos, D.A.; Barral, M.T. Sorptive Removal of Hg II by Red Mud (Bauxite Residue) in Contaminated Landfill Leachate. J. Environ. Sci. Health Part A 2017, 52, 84–98. [Google Scholar] [CrossRef]
- Balaram, V.; Subramanyam, K.S.V. Sample Preparation for Geochemical Analysis: Strategies and Significance. Adv. Sample Prep. 2022, 1, 100010. [Google Scholar] [CrossRef]
- Ma, Y.; Stopic, S.; Gronen, L.; Milivojevic, M.; Obradovic, S.; Friedrich, B. Neural Network Modeling for the Extraction of Rare Earth Elements from Eudialyte Concentrate by Dry Digestion and Leaching. Metals 2018, 8, 267. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Peng, G.; He, Q.; Zhu, H.; Al-Hamadani, S.M.Z.F. Dispersive Liquid–Liquid Microextraction Based on the Solidification of Floating Organic Drop Followed by ICP-MS for the Simultaneous Determination of Heavy Metals in Wastewaters. Spectrochim. Acta. A Mol. Biomol. Spectrosc. 2015, 140, 156–161. [Google Scholar] [CrossRef] [PubMed]
- Smith, R.C.; Taggart, R.K.; Hower, J.C.; Wiesner, M.R.; Hsu-Kim, H. Selective Recovery of Rare Earth Elements from Coal Fly Ash Leachates Using Liquid Membrane Processes. Environ. Sci. Technol. 2019, 53, 4490–4499. [Google Scholar] [CrossRef] [PubMed]
- Marjanovic, V.; Peric-Grujic, A.; Ristic, M.; Marinkovic, A.; Markovic, R.; Onjia, A.; Sljivic-Ivanovic, M. Selenate Adsorption from Water Using the Hydrous Iron Oxide-Impregnated Hybrid Polymer. Metals 2020, 10, 1630. [Google Scholar] [CrossRef]
- Suručić, L.; Tadić, T.; Janjić, G.; Marković, B.; Nastasović, A.; Onjia, A. Recovery of Vanadium (V) Oxyanions by a Magnetic Macroporous Copolymer Nanocomposite Sorbent. Metals 2021, 11, 1777. [Google Scholar] [CrossRef]
- Peiravi, M.; Ackah, L.; Guru, R.; Mohanty, M.; Liu, J.; Xu, B.; Zhu, X.; Chen, L. Chemical Extraction of Rare Earth Elements from Coal Ash. Miner. Metall. Process. 2017, 34, 170–177. [Google Scholar] [CrossRef]
- Aguirre, M.Á.; Baile, P.; Vidal, L.; Canals, A. Metal Applications of Liquid-Phase Microextraction. TrAC Trends Anal. Chem. 2019, 112, 241–247. [Google Scholar] [CrossRef]
- Rajakovic, L.; Todorovic, Z.; Rajakovic-Ognjanovic, V.; Onjia, A. Analytical Methods for Arsenic Speciation Analysis. J. Serb. Chem. Soc. 2013, 78, 1461–1479. [Google Scholar] [CrossRef] [Green Version]
- Sajid, M.; Asif, M.; Ihsanullah, I. Dispersive Liquid–Liquid Microextraction of Multi-Elements in Seawater Followed by Inductively Coupled Plasma-Mass Spectrometric Analysis and Evaluation of Its Greenness. Microchem. J. 2021, 169, 106565. [Google Scholar] [CrossRef]
- Labutin, T.A.; Lednev, V.N.; Ilyin, A.A.; Popov, A.M. Femtosecond Laser-Induced Breakdown Spectroscopy. J. Anal. At. Spectrom. 2016, 31, 90–118. [Google Scholar] [CrossRef]
- Krishnan Chandrasekaran, S.; Dheram Karunasagar, K.; Jayaraman Arunachalam, G. Dispersive Liquid-Liquid Micro-Extraction for Simultaneous Preconcentration of 14 Lanthanides at Parts per Trillion Levels from Groundwater and Determination Using a Micro-Flow Nebulizer in Inductively Coupled Plasma-Quadrupole Mass Spectrometry. J. Anal. Spectrom. 2010, 25, 18–20. [Google Scholar] [CrossRef]
- Chen, S.; Yan, J.; Li, J.; Lu, D. Magnetic ZnFe2O4 Nanotubes for Dispersive Micro Solid-Phase Extraction of Trace Rare Earth Elements Prior to Their Determination by ICP-MS. Microchim. Acta 2019, 186, 228. [Google Scholar] [CrossRef] [PubMed]
- Sereshti, H.; Khojeh, V.; Samadi, S. Optimization of Dispersive Liquid–Liquid Microextraction Coupled with Inductively Coupled Plasma-Optical Emission Spectrometry with the Aid of Experimental Design for Simultaneous Determination of Heavy Metals in Natural Waters. Talanta 2011, 83, 885–890. [Google Scholar] [CrossRef]
- Pinheiro, F.C.; Aguirre, M.Á.; Nóbrega, J.A.; González-Gallardo, N.; Ramón, D.J.; Canals, A. Dispersive Liquid-Liquid Microextraction Based on Deep Eutectic Solvent for Elemental Impurities Determination in Oral and Parenteral Drugs by Inductively Coupled Plasma Optical Emission Spectrometry. Anal. Chim. Acta 2021, 1185, 339052. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Moshtaghibana, S.; Zhu, T.; Fayemiwo, K.A.; Price, A.; Vladisavljević, G. Microfluidic Fabrication of Novel Polymeric Core-shell Microcapsules for Storage of CO2 Solvents and Organic Chelating Agents. J. Polym. Sci. 2022, pol.20210959. [Google Scholar] [CrossRef]
- Derringer, G.; Suich, R. Simultaneous Optimization of Several Response Variables. J. Qual. Technol. 1980, 12, 214–219. [Google Scholar] [CrossRef]
- Park, S.; Kim, M.; Lim, Y.; Yu, J.; Chen, S.; Woo, S.W.; Yoon, S.; Bae, S.; Kim, H.S. Characterization of Rare Earth Elements Present in Coal Ash by Sequential Extraction. J. Hazard. Mater. 2021, 402, 123760. [Google Scholar] [CrossRef]
- Wang, Z.; Dai, S.; Zou, J.; French, D.; Graham, I.T. Rare Earth Elements and Yttrium in Coal Ash from the Luzhou Power Plant in Sichuan, Southwest China: Concentration, Characterization and Optimized Extraction. Int. J. Coal Geol. 2019, 203, 1–14. [Google Scholar] [CrossRef]
- Tang, M.; Zhou, C.; Pan, J.; Zhang, N.; Liu, C.; Cao, S.; Hu, T.; Ji, W. Study on Extraction of Rare Earth Elements from Coal Fly Ash through Alkali Fusion—Acid Leaching. Miner. Eng. 2019, 136, 36–42. [Google Scholar] [CrossRef]
- King, J.F.; Taggart, R.K.; Smith, R.C.; Hower, J.C.; Hsu-Kim, H. Aqueous Acid and Alkaline Extraction of Rare Earth Elements from Coal Combustion Ash. Int. J. Coal Geol. 2018, 195, 75–83. [Google Scholar] [CrossRef]
- Rybak, A.; Rybak, A. Characteristics of Some Selected Methods of Rare Earth Elements Recovery from Coal Fly Ashes. Metals 2021, 11, 142. [Google Scholar] [CrossRef]



| No. | Variable | Symbol | Level | |
|---|---|---|---|---|
| −1 | +1 | |||
| 1. | poly(HDDA)/graphene adsorbent amount (mg) | ma | 10 | 50 |
| 2. | pH value | pH | 3 | 11 |
| 3. | D2EHPA chelating (10%) agent content (μL) | Che | 20 | 200 |
| 4. | Vortex time (min) | tv | 1 | 5 |
| 5. | Extraction temperature (°C) | T | 10 | 40 |
| 6. | Centrifuge time (min) | tc | 1 | 5 |
| 7. | Centrifuge speed (rpm) | wR | 2000 | 10000 |
| 8. | Eluent type * | E | EA | EB |
| 9. | Eluent time (min) | te | 1 | 5 |
| ICP-MS Parameter | Value | Isotope (Interference) |
|---|---|---|
| Plasma power | 1550 W | Analytes: 45Sc (COO, COOH) 89Y 139La 140Ce 141Pr 146Nd 147Sm 153Eu (BaO) 157Gd (CeOH, PrO) 159Tb (NdO) 163Dy (SmO) 165Ho (SmO) 166Er (SmO, NdO) 169Tm (SmO, EuO) 172Yb (GdO) 175Lu (GdO, TbO) 115In (I.S.) |
| Cool flow (Ar) | 13.8 L/min | |
| Auxiliary flow (Ar) | 0.82 L/min | |
| Nebulizer flow (Ar) | 0.97 L/min | |
| KED mode gas flow (He) | 5 mL/min | |
| Peristaltic pump speed | 35 rpm | |
| Injector | Quartz, 2.5 mm ID | |
| Interface cones | Nickel | |
| Sweeps/reading | 20 | |
| Replicates | 3 | |
| Points per peak | 3 | |
| Dwell times | 10–40 ms | |
| Scan mode | Peak hopping | |
| Sweps | 30 | |
| Sample flush time | 4 s | |
| Read delay time | 20 s | |
| Wash time | 60 s | |
| Calibration type | Matrix-matched, external |
| No. | REE | CAS No. | LOD (ng/L) | Linear Equation (a + b·x) | R2 | Recovery (%) | RSD (%) |
|---|---|---|---|---|---|---|---|
| 1. | Scandium (Sc) | 7440-45-1 | 83 | 6.55 + 7707·x | 0.9965 | 59 | 17 |
| 2. | Yttrium (Y) | 7429-91-6 | 27 | 730 + 76,388·x | 0.9992 | 67 | 8.7 |
| 3. | Lanthanum (La) | 7440-52-0 | 2.4 | 46.6 + 263,993·x | 0.9997 | 96 | 9.2 |
| 4. | Cerium (Ce) | 7440-53-1 | 1.8 | 230 + 328,932·x | 0.9996 | 85 | 8.1 |
| 5. | Praseodymium (Pr) | 7440-54-2 | 1.5 | 6.66 + 352,101·x | 0.9998 | 90 | 7.9 |
| 6. | Neodymium (Nd) | 7440-60-0 | 3.1 | 10.0 + 72,372·x | 0.9996 | 112 | 9.8 |
| 7. | Samarium (Sm) | 7439-91-0 | 3.3 | 3.33 + 64,019·x | 0.9997 | 92 | 6.6 |
| 8. | Europium (Eu) | 7439-94-3 | 2.7 | 3.38 + 226,836·x | 0.9997 | 93 | 8.7 |
| 9. | Gadolinium (Gd) | 7440-00-8 | 2.7 | 3.37 + 116,690·x | 0.9997 | 94 | 12 |
| 10. | Terbium (Tb) | 7440-10-0 | 1.9 | 3.31 + 603,632·x | 0.9999 | 87 | 13 |
| 11. | Dysprosium (Dy) | 7440-20-2 | 2.1 | 3.37 + 153,391·x | 0.9998 | 79 | 11 |
| 12. | Holmium (Ho) | 7440-19-9 | 2.3 | 6.67 + 641,315·x | 0.9999 | 68 | 13 |
| 13. | Erbium (Er) | 7440-27-9 | 2.4 | 16.7 + 222,524·x | 0.9997 | 67 | 14 |
| 14. | Thulium (Tm) | 7440-29-1 | 3.6 | 1.21 + 724,602·x | 0.9999 | 56 | 16 |
| 15. | Ytterbium (Yb) | 7440-30-4 | 4.5 | 1.03 + 177,804·x | 0.9997 | 44 | 19 |
| 16. | Lutetium (Lu) | 7440-65-5 | 6.7 | 1.12 + 430,574·x | 0.9999 | 43 | 22 |
| No. | REE | AEP | BEP | CEP | DEP | EEP | ATCLP | BTCLP | CTCLP | DTCLP | ETCLP |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1. | Sc | n.d. | n.d. | n.d. | n.d. | n.d. | 211 | 186 | 172 | 198 | 249 |
| 2. | Y | 105 | 56 | 430 | 35 | 37 | 10,136 | 18,986 | 22,716 | 8958 | 7343 |
| 3. | La | 92 | 72 | 101 | 48 | 95 | 6199 | 11,264 | 17,285 | 4235 | 2465 |
| 4. | Ce | 194 | 102 | 331 | 22 | 39 | 23,413 | 39,866 | 51,249 | 16,625 | 9660 |
| 5. | Pr | 25 | 28 | 42 | 2.8 | 5.2 | 2977 | 5181 | 6338 | 2168 | 1206 |
| 6. | Nd | 97 | 72 | 179 | 11 | 20 | 12,305 | 21,778 | 25,608 | 9189 | 5402 |
| 7. | Sm | 26 | 26 | 55 | 6.4 | 8.3 | 2838 | 4832 | 5361 | 2096 | 1323 |
| 8. | Eu | 14 | 17 | 15 | 6.1 | n.d. | 642 | 1088 | 1204 | 512 | 324 |
| 9. | Gd | 30 | 25 | 61 | 4.3 | 6.6 | 2823 | 4854 | 5437 | 2090 | 1403 |
| 10. | Tb | 16 | 13 | 13 | n.d. | n.d. | 388 | 664 | 764 | 291 | 219 |
| 11. | Dy | 34 | 22 | 84 | 4.8 | 7.6 | 2129 | 3704 | 4234 | 1632 | 1339 |
| 12. | Ho | 17 | 12 | 17 | 2.9 | n.d. | 407 | 728 | 844 | 327 | 278 |
| 13. | Er | 22 | 14 | 47 | n.d. | 6.2 | 1094 | 2016 | 2374 | 918 | 819 |
| 14. | Tm | 12 | 6.0 | 5.1 | n.d. | n.d. | 135 | 253 | 311 | 113 | 117 |
| 15. | Yb | 11 | 6.3 | 18 | n.d. | n.d. | 598 | 1405 | 1829 | 604 | 704 |
| 16. | Lu | n.d. | n.d. | n.d. | n.d. | n.d. | 168 | 125 | 98 | 42 | 102 |
| 17. | ΣREEs | 0.70 | 0.47 | 1.40 | 0.14 | 0.22 | 66 | 117 | 146 | 50 | 33 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Slavković-Beškoski, L.; Ignjatović, L.; Bolognesi, G.; Maksin, D.; Savić, A.; Vladisavljević, G.; Onjia, A. Dispersive Solid–Liquid Microextraction Based on the Poly(HDDA)/Graphene Sorbent Followed by ICP-MS for the Determination of Rare Earth Elements in Coal Fly Ash Leachate. Metals 2022, 12, 791. https://doi.org/10.3390/met12050791
Slavković-Beškoski L, Ignjatović L, Bolognesi G, Maksin D, Savić A, Vladisavljević G, Onjia A. Dispersive Solid–Liquid Microextraction Based on the Poly(HDDA)/Graphene Sorbent Followed by ICP-MS for the Determination of Rare Earth Elements in Coal Fly Ash Leachate. Metals. 2022; 12(5):791. https://doi.org/10.3390/met12050791
Chicago/Turabian StyleSlavković-Beškoski, Latinka, Ljubiša Ignjatović, Guido Bolognesi, Danijela Maksin, Aleksandra Savić, Goran Vladisavljević, and Antonije Onjia. 2022. "Dispersive Solid–Liquid Microextraction Based on the Poly(HDDA)/Graphene Sorbent Followed by ICP-MS for the Determination of Rare Earth Elements in Coal Fly Ash Leachate" Metals 12, no. 5: 791. https://doi.org/10.3390/met12050791
APA StyleSlavković-Beškoski, L., Ignjatović, L., Bolognesi, G., Maksin, D., Savić, A., Vladisavljević, G., & Onjia, A. (2022). Dispersive Solid–Liquid Microextraction Based on the Poly(HDDA)/Graphene Sorbent Followed by ICP-MS for the Determination of Rare Earth Elements in Coal Fly Ash Leachate. Metals, 12(5), 791. https://doi.org/10.3390/met12050791









